N-terminal VP1 Truncations Favor T = 1 Norovirus-Like Particles
Abstract
1. Introduction
2. Materials and Methods
2.1. VLP Production and Preparation
2.2. VP1 Mapping
2.3. Sample Preparation
2.4. Mass Spectrometry
2.5. Electron Microscopy
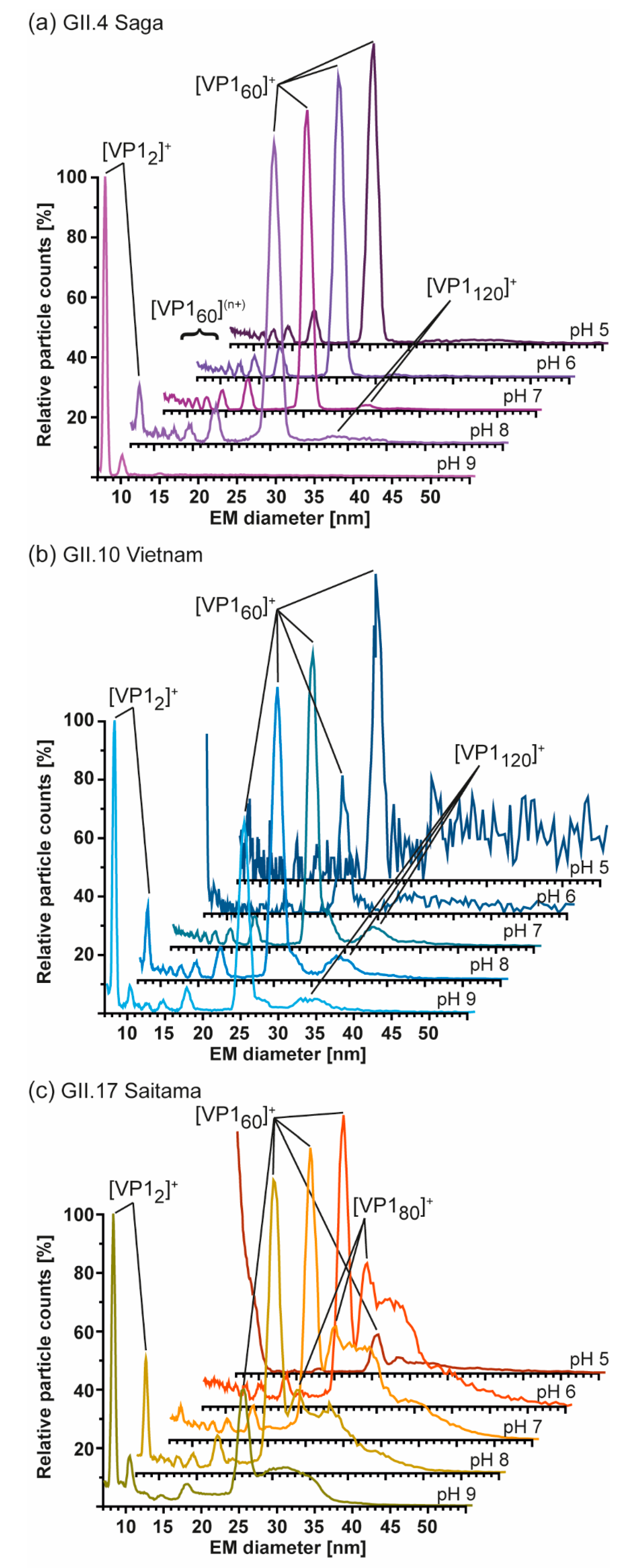
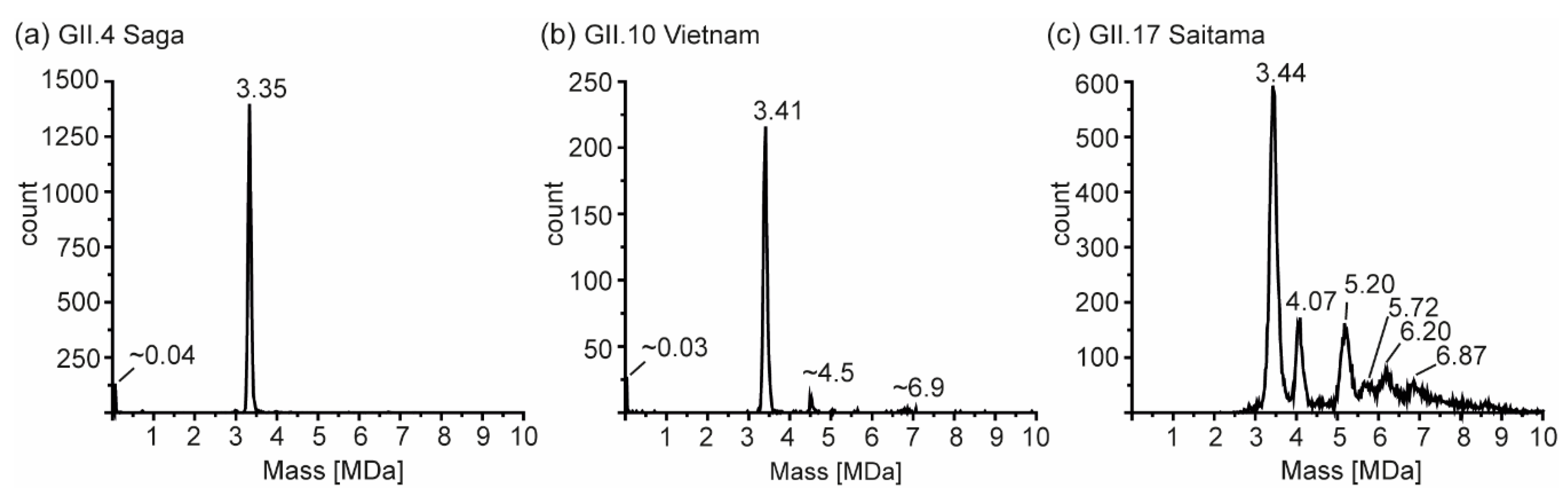
3. Results
3.1. Truncated GII.4 Saga VP1 Forms Homogeneous T = 1 Particles
3.2. T = 1 Capsid Formation Is Genotype-Independent
3.3. Heterologous Expression of GI.1 West Chester Results in Either T = 1 or T = 3 Preparations
3.4. Detailed nES GEMMA and CDMS Profiling
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ahmed, S.M.; Hall, A.J.; Robinson, A.E.; Verhoef, L.; Premkumar, P.; Parashar, U.D.; Koopmans, M.; Lopman, B.A. Global prevalence of norovirus in cases of gastroenteritis: A systematic review and meta-analysis. Lancet Infect. Dis. 2014, 14, 725–730. [Google Scholar] [CrossRef]
- Teunis, P.F.; Moe, C.L.; Liu, P.; Miller, S.E.; Lindesmith, L.; Baric, R.S.; Le Pendu, J.; Calderon, R.L. Norwalk virus: How infectious is it? J. Med. Virol. 2008, 80, 1468–1476. [Google Scholar] [CrossRef]
- Xi, J.N.; Graham, D.Y.; Wang, K.N.; Estes, M.K. Norwalk virus genome cloning and characterization. Science 1990, 250, 1580–1583. [Google Scholar] [CrossRef]
- Jiang, X.; Wang, M.; Wang, K.; Estes, M.K. Sequence and genomic organization of Norwalk virus. Virology 1993, 195, 51–61. [Google Scholar] [CrossRef]
- Chhabra, P.; de Graaf, M.; Parra, G.I.; Chan, M.C.; Green, K.; Martella, V.; Wang, Q.; White, P.A.; Katayama, K.; Vennema, H.; et al. Updated classification of norovirus genogroups and genotypes. J. Gen. Virol. 2019, 100, 1393–1406. [Google Scholar] [CrossRef]
- Kapikian, A.Z.; Wyatt, R.G.; Dolin, R.; Thornhill, T.S.; Kalica, A.R.; Chanock, R.M. Visualization by immune electron microscopy of a 27-nm particle associated with acute infectious nonbacterial gastroenteritis. J. Virol. 1972, 10, 1075–1081. [Google Scholar] [CrossRef]
- Eden, J.S.; Tanaka, M.M.; Boni, M.F.; Rawlinson, W.D.; White, P.A. Recombination within the Pandemic Norovirus GII.4 Lineage. J. Virol. 2013, 87, 6270–6282. [Google Scholar] [CrossRef]
- De Graaf, M.; van Beek, J.; Vennema, H.; Podkolzin, A.T.; Hewitt, J.; Bucardo, F.; Templeton, K.; Mans, J.; Nordgren, J.; Reuter, G.; et al. Emergence of a novel GII.17 norovirus—End of the GII.4 era? Eurosurveillance 2015, 20, 8–15. [Google Scholar] [CrossRef]
- Ettayebi, K.; Crawford, S.E.; Murakami, K.; Broughman, J.R.; Karandikar, U.; Tenge, V.R.; Neill, F.H.; Blutt, S.E.; Zeng, X.L.; Qu, L.; et al. Replication of human noroviruses in stem cell-derived human enteroids. Science 2016. [Google Scholar] [CrossRef]
- Tome-Amat, J.; Fleischer, L.; Parker, S.A.; Bardliving, C.L.; Batt, C.A. Secreted production of assembled Norovirus virus-like particles from Pichia pastoris. Microb. Cell Fact. 2014, 13, 134. [Google Scholar] [CrossRef]
- Taube, S.; Kurth, A.; Schreier, E. Generation of recombinant norovirus-like particles (VLP) in the human endothelial kidney cell line 293T. Arch. Virol. 2005, 150, 1425–1431. [Google Scholar] [CrossRef] [PubMed]
- Diamos, A.G.; Mason, H.S. High-level expression and enrichment of norovirus virus-like particles in plants using modified geminiviral vectors. Protein Expr. Purif. 2018, 151, 86–92. [Google Scholar] [CrossRef] [PubMed]
- Mason, H.S.; Ball, J.M.; Shi, J.J.; Jiang, X.; Estes, M.K.; Arntzen, C.J. Expression of Norwalk virus capsid protein in transgenic tobacco and potato and its oral immunogenicity in mice. Proc. Natl. Acad. Sci. USA 1996, 93, 5335–5340. [Google Scholar] [CrossRef] [PubMed]
- Prasad, B.V.; Rothnagel, R.; Jiang, X.; Estes, M.K. Three-dimensional structure of baculovirus-expressed Norwalk virus capsids. J. Virol. 1994, 68, 5117–5125. [Google Scholar] [CrossRef] [PubMed]
- Prasad, B.V.; Hardy, M.E.; Dokland, T.; Bella, J.; Rossmann, M.G.; Estes, M.K. X-ray crystallographic structure of the Norwalk virus capsid. Science 1999, 286, 287–290. [Google Scholar] [CrossRef]
- Jiang, X.; Wang, M.; Graham, D.Y.; Estes, M.K. Expression, self-assembly, and antigenicity of the Norwalk virus capsid protein. J. Virol. 1992, 66, 6527–6532. [Google Scholar] [CrossRef] [PubMed]
- Tan, M.; Jiang, X. The p domain of norovirus capsid protein forms a subviral particle that binds to histo-blood group antigen receptors. J. Virol. 2005, 79, 14017–14030. [Google Scholar] [CrossRef]
- Bertolotti-Ciarlet, A.; White, L.J.; Chen, R.; Prasad, B.V.; Estes, M.K. Structural requirements for the assembly of Norwalk virus-like particles. J. Virol. 2002, 76, 4044–4055. [Google Scholar] [CrossRef]
- White, L.J.; Hardy, M.E.; Estes, M.K. Biochemical characterization of a smaller form of recombinant Norwalk virus capsids assembled in insect cells. J. Virol. 1997, 71, 8066–8072. [Google Scholar] [CrossRef]
- Devant, J.M.; Hofhaus, G.; Bhella, D.; Hansman, G.S. Heterologous expression of human norovirus GII.4 VP1 leads to assembly of T = 4 virus-like particles. Antivir. Res. 2019, 168, 175–182. [Google Scholar] [CrossRef]
- Jung, J.; Grant, T.; Thomas, D.R.; Diehnelt, C.W.; Grigorieff, N.; Joshua-Tor, L. High-resolution cryo-EM structures of outbreak strain human norovirus shells reveal size variations. Proc. Natl. Acad. Sci. USA 2019, 116, 12828–12832. [Google Scholar] [CrossRef] [PubMed]
- Taniguchi, K.; Urasawa, S.; Urasawa, T. Further studies of 35–40 nm virus-like particles associated with outbreaks of acute gastroenteritis. J. Med. Microbiol. 1981, 14, 107–118. [Google Scholar] [CrossRef] [PubMed]
- Dulfer, J.; Kadek, A.; Kopicki, J.D.; Krichel, B.; Uetrecht, C. Structural mass spectrometry goes viral. Adv. Virus Res. 2019, 105, 189–238. [Google Scholar] [CrossRef] [PubMed]
- Shoemaker, G.K.; van Duijn, E.; Crawford, S.E.; Uetrecht, C.; Baclayon, M.; Roos, W.H.; Wuite, G.J.; Estes, M.K.; Prasad, B.V.; Heck, A.J. Norwalk virus assembly and stability monitored by mass spectrometry. Mol. Cell Proteom. 2010, 9, 1742–1751. [Google Scholar] [CrossRef] [PubMed]
- Pogan, R.; Schneider, C.; Reimer, R.; Hansman, G.; Uetrecht, C. Norovirus-like VP1 particles exhibit isolate dependent stability profiles. J. Phys. Condens. Matter 2018, 30, 064006. [Google Scholar] [CrossRef]
- Kaufman, S.L.; Skogen, J.W.; Dorman, F.D.; Zarrin, F.; Lewis, K.C. Macromolecule analysis based on electrophoretic mobility in air: Globular proteins. Anal. Chem. 1996, 68, 1895–1904. [Google Scholar] [CrossRef]
- Weiss, V.U.; Bereszcazk, J.Z.; Havlik, M.; Kallinger, P.; Gosler, I.; Kumar, M.; Blaas, D.; Marchetti-Deschmann, M.; Heck, A.J.; Szymanski, W.W.; et al. Analysis of a common cold virus and its subviral particles by gas-phase electrophoretic mobility molecular analysis and native mass spectrometry. Anal. Chem. 2015, 87, 8709–8717. [Google Scholar] [CrossRef]
- Weiss, V.U.; Pogan, R.; Zoratto, S.; Bond, K.M.; Boulanger, P.; Jarrold, M.F.; Lyktey, N.; Pahl, D.; Puffler, N.; Schelhaas, M.; et al. Virus-like particle size and molecular weight/mass determination applying gas-phase electrophoresis (native nES GEMMA). Anal. Bioanal. Chem. 2019, 411, 5951–5962. [Google Scholar] [CrossRef]
- Hansman, G.S.; Natori, K.; Oka, T.; Ogawa, S.; Tanaka, K.; Nagata, N.; Ushijima, H.; Takeda, N.; Katayama, K. Cross-reactivity among sapovirus recombinant capsid proteins. Arch. Virol. 2005, 150, 21–36. [Google Scholar] [CrossRef]
- Hansman, G.S.; Saito, H.; Shibata, C.; Ishizuka, S.; Oseto, M.; Oka, T.; Takeda, N. Outbreak of gastroenteritis due to sapovirus. J. Clin. Microbiol. 2007, 45, 1347–1349. [Google Scholar] [CrossRef]
- Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef]
- Shevchenko, A.; Tomas, H.; Havlis, J.; Olsen, J.V.; Mann, M. In-gel digestion for mass spectrometric characterization of proteins and proteomes. Nat. Protoc. 2006, 1, 2856–2860. [Google Scholar] [CrossRef]
- Perez-Riverol, Y.; Csordas, A.; Bai, J.; Bernal-Llinares, M.; Hewapathirana, S.; Kundu, D.J.; Inuganti, A.; Griss, J.; Mayer, G.; Eisenacher, M.; et al. The PRIDE database and related tools and resources in 2019: Improving support for quantification data. Nucleic Acids Res. 2019, 47, D442–D450. [Google Scholar] [CrossRef] [PubMed]
- Van den Heuvel, R.H.; van Duijn, E.; Mazon, H.; Synowsky, S.A.; Lorenzen, K.; Versluis, C.; Brouns, S.J.; Langridge, D.; van der Oost, J.; Hoyes, J.; et al. Improving the performance of a quadrupole time-of-flight instrument for macromolecular mass spectrometry. Anal. Chem. 2006, 78, 7473–7483. [Google Scholar] [CrossRef] [PubMed]
- Lorenzen, K.; Versluis, C.; van Duijn, E.; van den Heuvel, R.H.H.; Heck, A.J.R. Optimizing macromolecular tandem mass spectrometry of large non-covalent complexes using heavy collision gases. Int. J. Mass Spectrom. 2007, 268, 198–206. [Google Scholar] [CrossRef][Green Version]
- Morgner, N.; Robinson, C.V. Massign: An assignment strategy for maximizing information from the mass spectra of heterogeneous protein assemblies. Anal. Chem. 2012, 84, 2939–2948. [Google Scholar] [CrossRef]
- Contino, N.C.; Pierson, E.E.; Keifer, D.Z.; Jarrold, M.F. Charge detection mass spectrometry with resolved charge states. J. Am. Soc. Mass Spectrom. 2013, 24, 101–108. [Google Scholar] [CrossRef]
- Tycova, A.; Prikryl, J.; Foret, F. Reproducible preparation of nanospray tips for capillary electrophoresis coupled to mass spectrometry using 3D printed grinding device. Electrophoresis 2016, 37, 924–930. [Google Scholar] [CrossRef]
- Bacher, G.; Szymanski, W.W.; Kaufman, S.L.; Zollner, P.; Blaas, D.; Allmaier, G. Charge-reduced nano electrospray ionization combined with differential mobility analysis of peptides, proteins, glycoproteins, noncovalent protein complexes and viruses. J. Mass Spectrom. 2001, 36, 1038–1052. [Google Scholar] [CrossRef]
- Caspar, D.L.; Klug, A. Physical principles in the construction of regular viruses. Cold Spring Harb. Symp. Quant. Biol. 1962, 27, 1–24. [Google Scholar] [CrossRef]
- Gotoh, T.; Miyazaki, Y.; Kikuchi, K.; Bentley, W.E. Investigation of sequential behavior of carboxyl protease and cysteine protease activities in virus-infected Sf-9 insect cell culture by inhibition assay. Appl. Microbiol. Biotechnol. 2001, 56, 742–749. [Google Scholar] [CrossRef]
- Gotoh, T.; Ono, H.; Kikuchi, K.; Nirasawa, S.; Takahashi, S. Purification and characterization of aspartic protease derived from Sf9 insect cells. Biosci. Biotechnol. Biochem. 2010, 74, 2154–2157. [Google Scholar] [CrossRef] [PubMed]
- Uetrecht, C.; Barbu, I.M.; Shoemaker, G.K.; van Duijn, E.; Heck, A.J. Interrogating viral capsid assembly with ion mobility-mass spectrometry. Nat. Chem. 2011, 3, 126–132. [Google Scholar] [CrossRef] [PubMed]
- Pogan, R.; Dulfer, J.; Uetrecht, C. Norovirus assembly and stability. Curr. Opin. Virol. 2018, 31, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Grimes, J.M.; Burroughs, J.N.; Gouet, P.; Diprose, J.M.; Malby, R.; Zientara, S.; Mertens, P.P.; Stuart, D.I. The atomic structure of the bluetongue virus core. Nature 1998, 395, 470–478. [Google Scholar] [CrossRef] [PubMed]
- Baker, T.S.; Olson, N.H.; Fuller, S.D. Adding the third dimension to virus life cycles: Three-dimensional reconstruction of icosahedral viruses from cryo-electron micrographs. Microbiol. Mol. Biol. Rev. 1999, 63, 862–922. [Google Scholar] [CrossRef] [PubMed]
- Krol, M.A.; Olson, N.H.; Tate, J.; Johnson, J.E.; Baker, T.S.; Ahlquist, P. RNA-controlled polymorphism in the in vivo assembly of 180-subunit and 120-subunit virions from a single capsid protein. Proc. Natl. Acad. Sci. USA 1999, 96, 13650–13655. [Google Scholar] [CrossRef]
- Lutomski, C.A.; Lyktey, N.A.; Zhao, Z.; Pierson, E.E.; Zlotnick, A.; Jarrold, M.F. Hepatitis B Virus Capsid Completion Occurs through Error Correction. J. Am. Chem. Soc. 2017, 139, 16932–16938. [Google Scholar] [CrossRef]
- Pierson, E.E.; Keifer, D.Z.; Kukreja, A.A.; Wang, J.C.; Zlotnick, A.; Jarrold, M.F. Charge Detection Mass Spectrometry Identifies Preferred Non-Icosahedral Polymorphs in the Self-Assembly of Woodchuck Hepatitis Virus Capsids. J. Mol. Biol. 2016, 428, 292–300. [Google Scholar] [CrossRef]
- Salunke, D.M.; Caspar, D.L.; Garcea, R.L. Polymorphism in the assembly of polyomavirus capsid protein VP1. Biophys. J. 1989, 56, 887–900. [Google Scholar] [CrossRef]
- Huo, Y.Q.; Wan, X.; Wang, Z.J.; Meng, S.L.; Shen, S. Production of Norovirus VLPs to size homogeneity. Virus Res. 2015, 204, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Someya, Y.; Shirato, H.; Hasegawa, K.; Kumasaka, T.; Takeda, N. Assembly of homogeneous norovirus-like particles accomplished by amino acid substitution. J. Gen. Virol. 2011, 92, 2320–2323. [Google Scholar] [CrossRef] [PubMed]
- Hasegawa, K.; Someya, Y.; Shigematsu, H.; Kimura-Someya, T.; Nuemket, N.; Kumasaka, T. Crystallization and X-ray analysis of 23 nm virus-like particles from Norovirus Chiba strain. Acta Crystallogr. F Struct. Biol. Commun. 2017, 73, 568–573. [Google Scholar] [CrossRef] [PubMed]
- Tan, M.; Fang, P.; Chachiyo, T.; Xia, M.; Huang, P.; Fang, Z.; Jiang, W.; Jiang, X. Noroviral P particle: Structure, function and applications in virus-host interaction. Virology 2008, 382, 115–123. [Google Scholar] [CrossRef]
- Tan, M.; Jiang, X. Norovirus P particle: A subviral nanoparticle for vaccine development against norovirus, rotavirus and influenza virus. Nanomedicine 2012, 7, 889–897. [Google Scholar] [CrossRef] [PubMed]
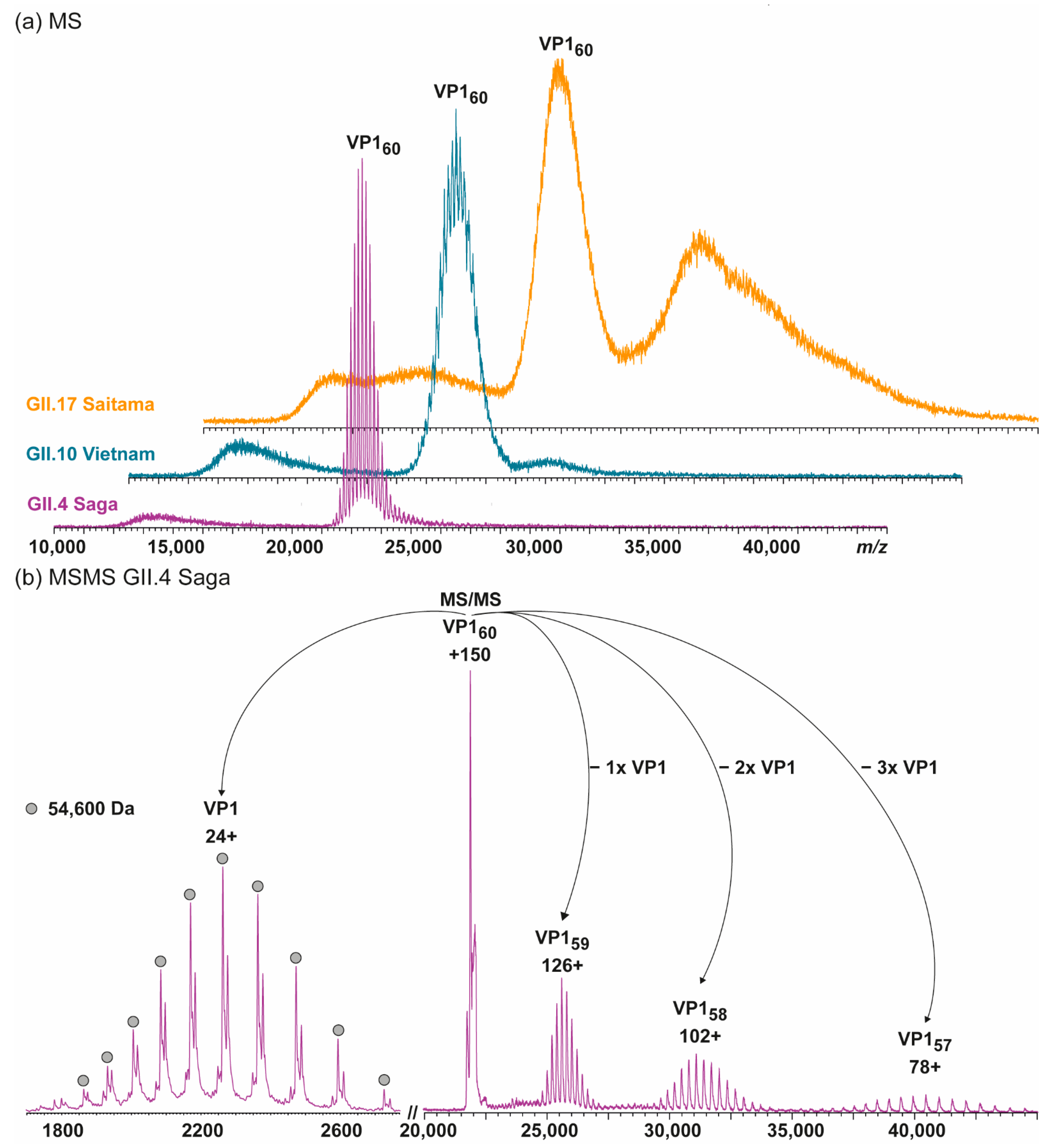
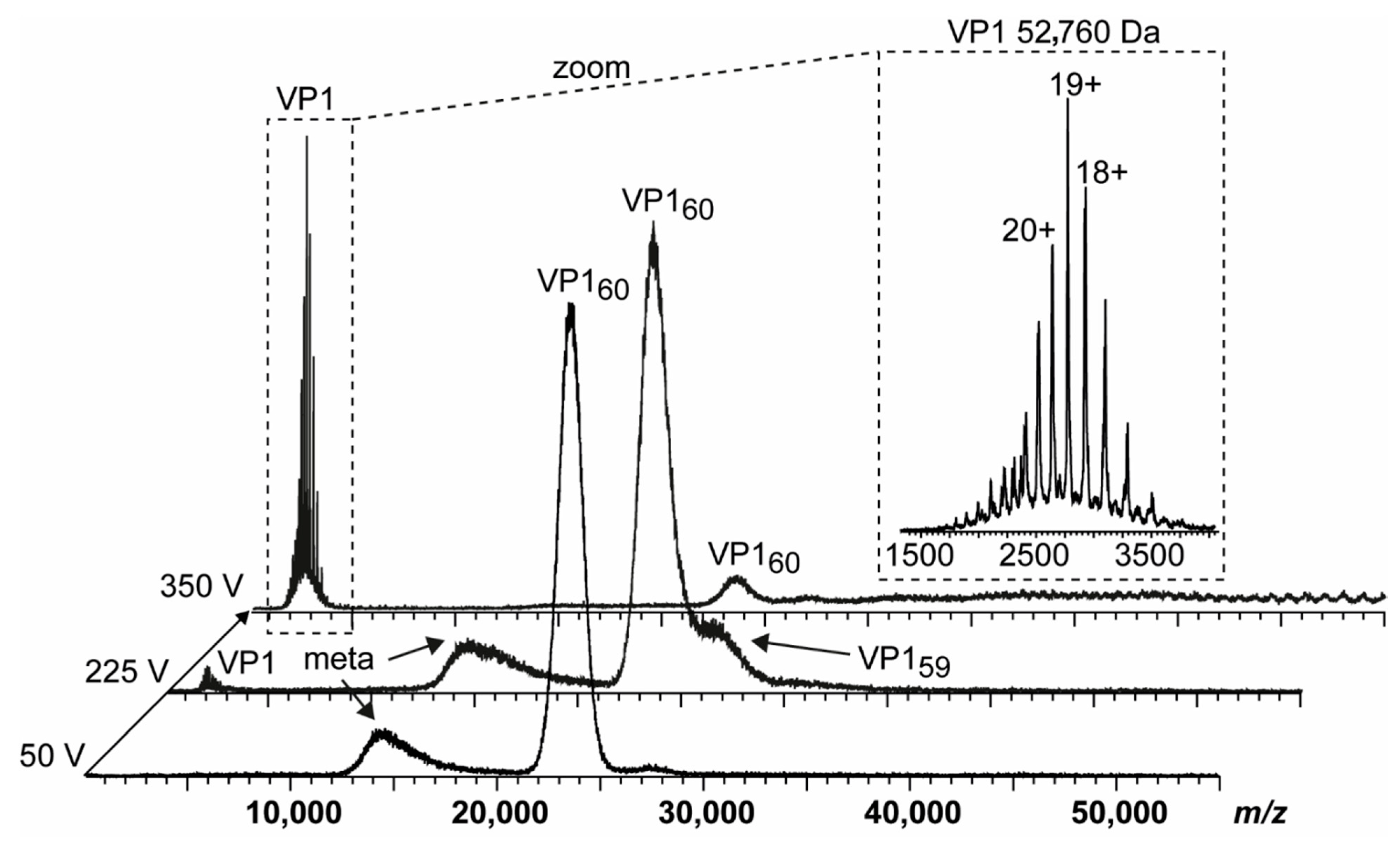
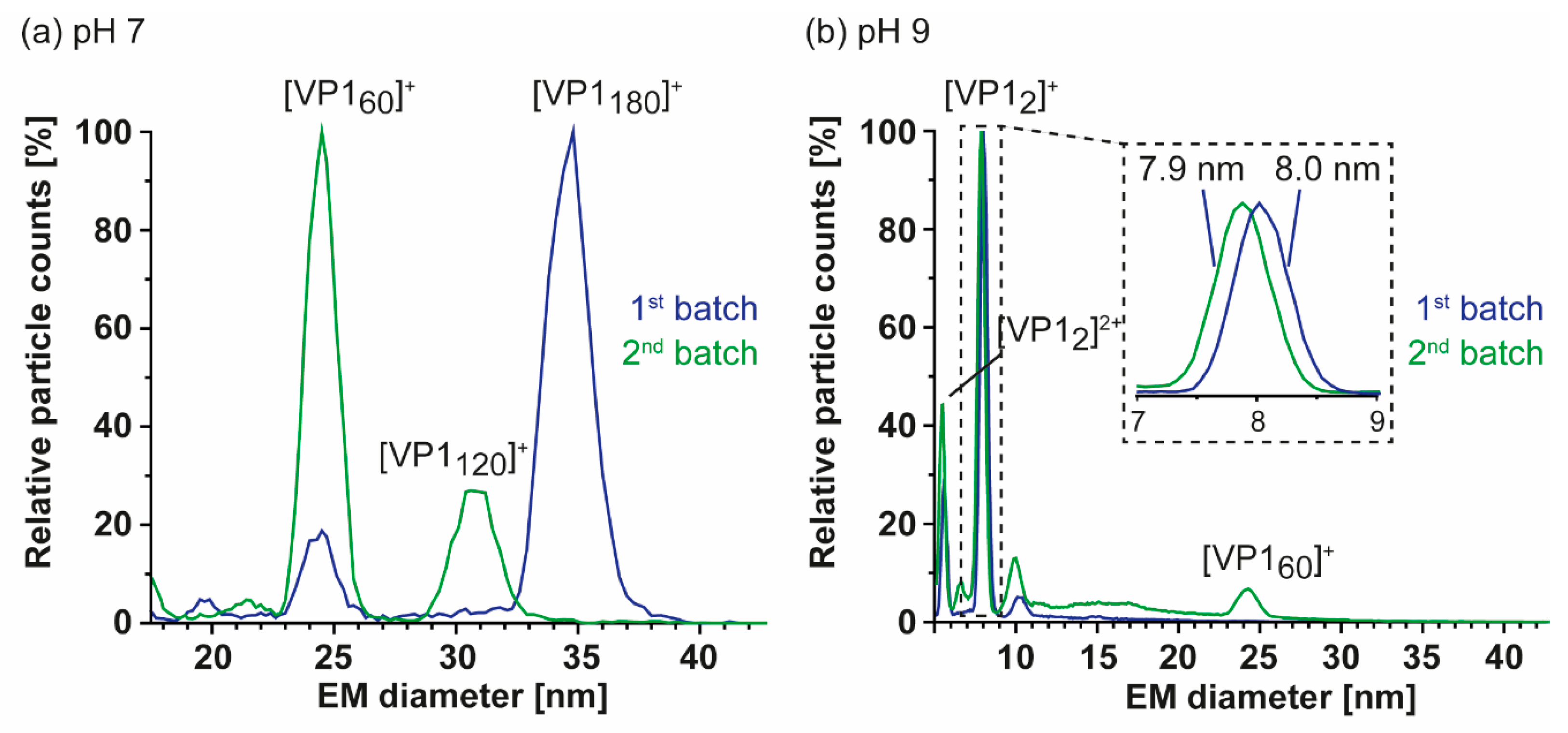
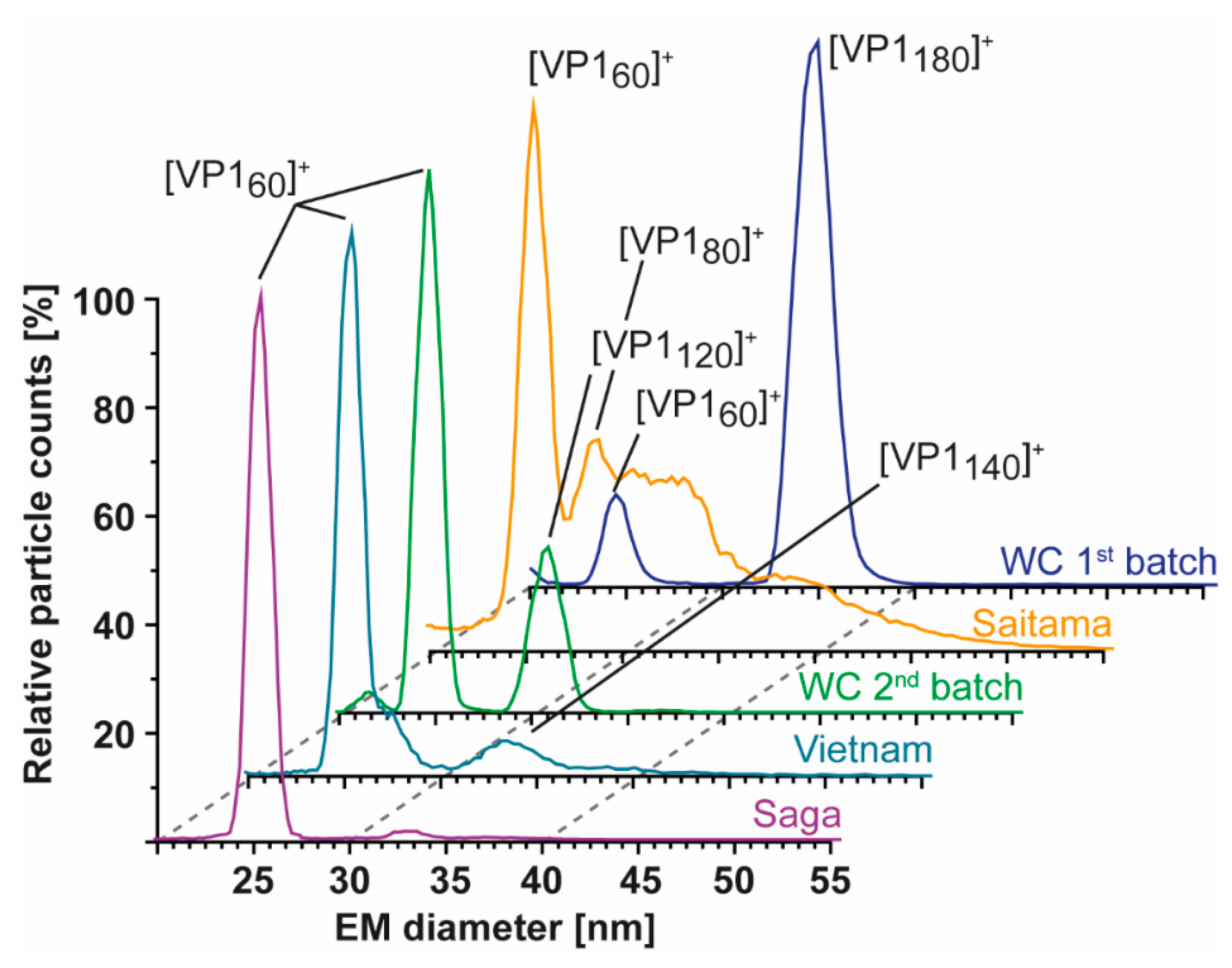
| Variant | VP1 th. | VP1 exp. | Putative Cleavage Site | Trypsin Digestion | Pepsin Digestion |
|---|---|---|---|---|---|
| MW, Total | Main Species MW Truncation | According to exp. VP1 MW | Sequence Coverage %, Minimal N-terminal Truncation | ||
| GI.1 West Chester | 56,609 Da, 530 aa | 52,760 Da, −40 aa | LAMDPVAGSS/TAVATAGQVN | 80%, −6 aa | 98%, −2 aa |
| GII.4 Saga | 59,005 Da 540 aa | 54,600 Da, −45 aa | AIAAPVAGQQ/NVIDPWIRNN | 95%, −25 aa | 90%, −27 aa |
| GII.10 Vietnam | 59,901 Da 548 aa | 55,560 Da, −aa | SLAAPVTGQT/NIIDPWIRMN | 95%, −27 aa | 94%, −27 aa |
| GII.17 Saitama | 58,957 Da 540 aa | 57,300 Da, −17 aa | SNDGATGLVP/EINNETLPLE | 91%, −32 aa | 99%, −3 aa |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pogan, R.; Weiss, V.U.; Bond, K.; Dülfer, J.; Krisp, C.; Lyktey, N.; Müller-Guhl, J.; Zoratto, S.; Allmaier, G.; Jarrold, M.F.; et al. N-terminal VP1 Truncations Favor T = 1 Norovirus-Like Particles. Vaccines 2021, 9, 8. https://doi.org/10.3390/vaccines9010008
Pogan R, Weiss VU, Bond K, Dülfer J, Krisp C, Lyktey N, Müller-Guhl J, Zoratto S, Allmaier G, Jarrold MF, et al. N-terminal VP1 Truncations Favor T = 1 Norovirus-Like Particles. Vaccines. 2021; 9(1):8. https://doi.org/10.3390/vaccines9010008
Chicago/Turabian StylePogan, Ronja, Victor U. Weiss, Kevin Bond, Jasmin Dülfer, Christoph Krisp, Nicholas Lyktey, Jürgen Müller-Guhl, Samuele Zoratto, Günter Allmaier, Martin F. Jarrold, and et al. 2021. "N-terminal VP1 Truncations Favor T = 1 Norovirus-Like Particles" Vaccines 9, no. 1: 8. https://doi.org/10.3390/vaccines9010008
APA StylePogan, R., Weiss, V. U., Bond, K., Dülfer, J., Krisp, C., Lyktey, N., Müller-Guhl, J., Zoratto, S., Allmaier, G., Jarrold, M. F., Muñoz-Fontela, C., Schlüter, H., & Uetrecht, C. (2021). N-terminal VP1 Truncations Favor T = 1 Norovirus-Like Particles. Vaccines, 9(1), 8. https://doi.org/10.3390/vaccines9010008







