A COVID-19 Vaccine: Big Strides Come with Big Challenges
Abstract
1. Introduction
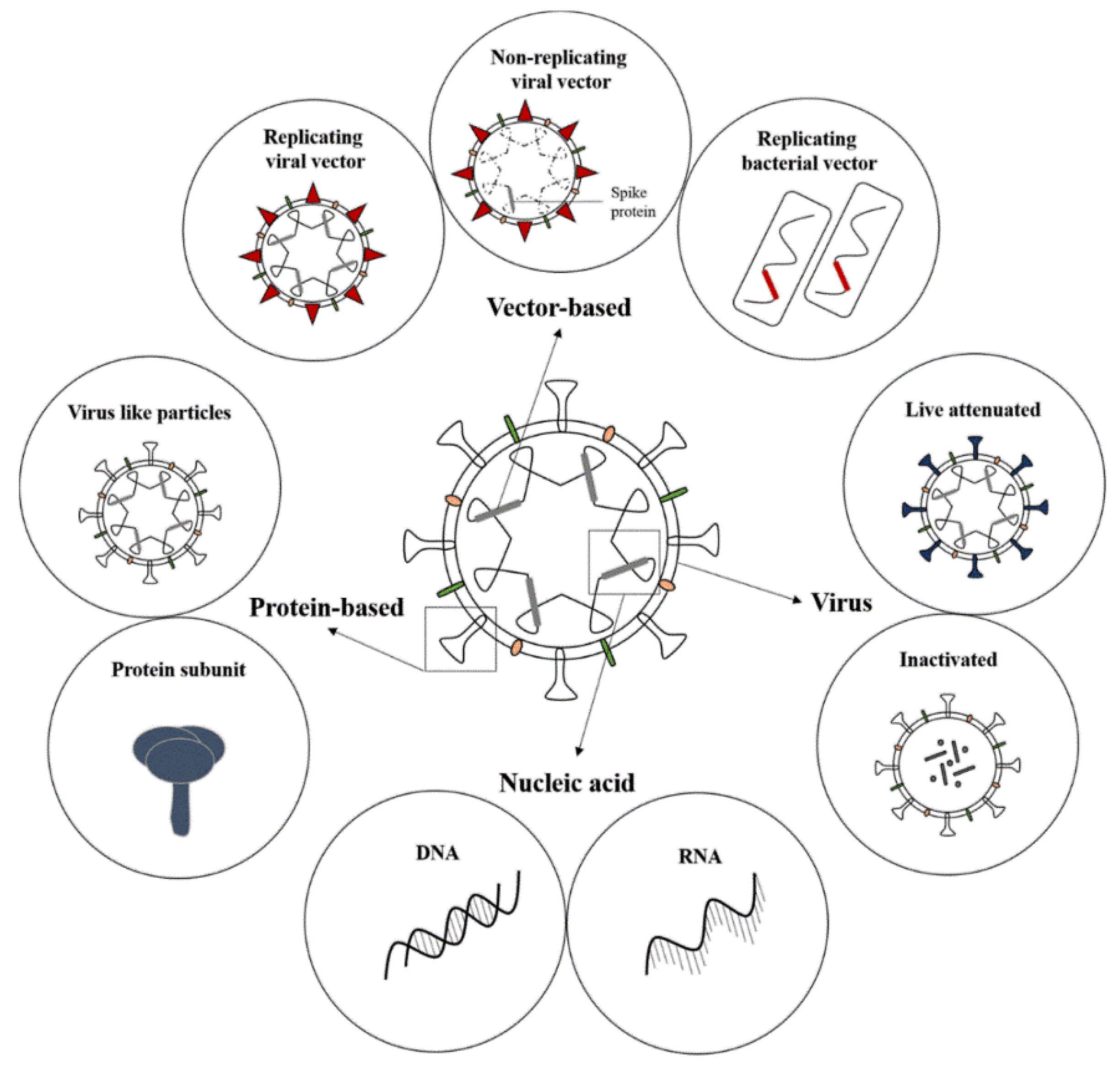
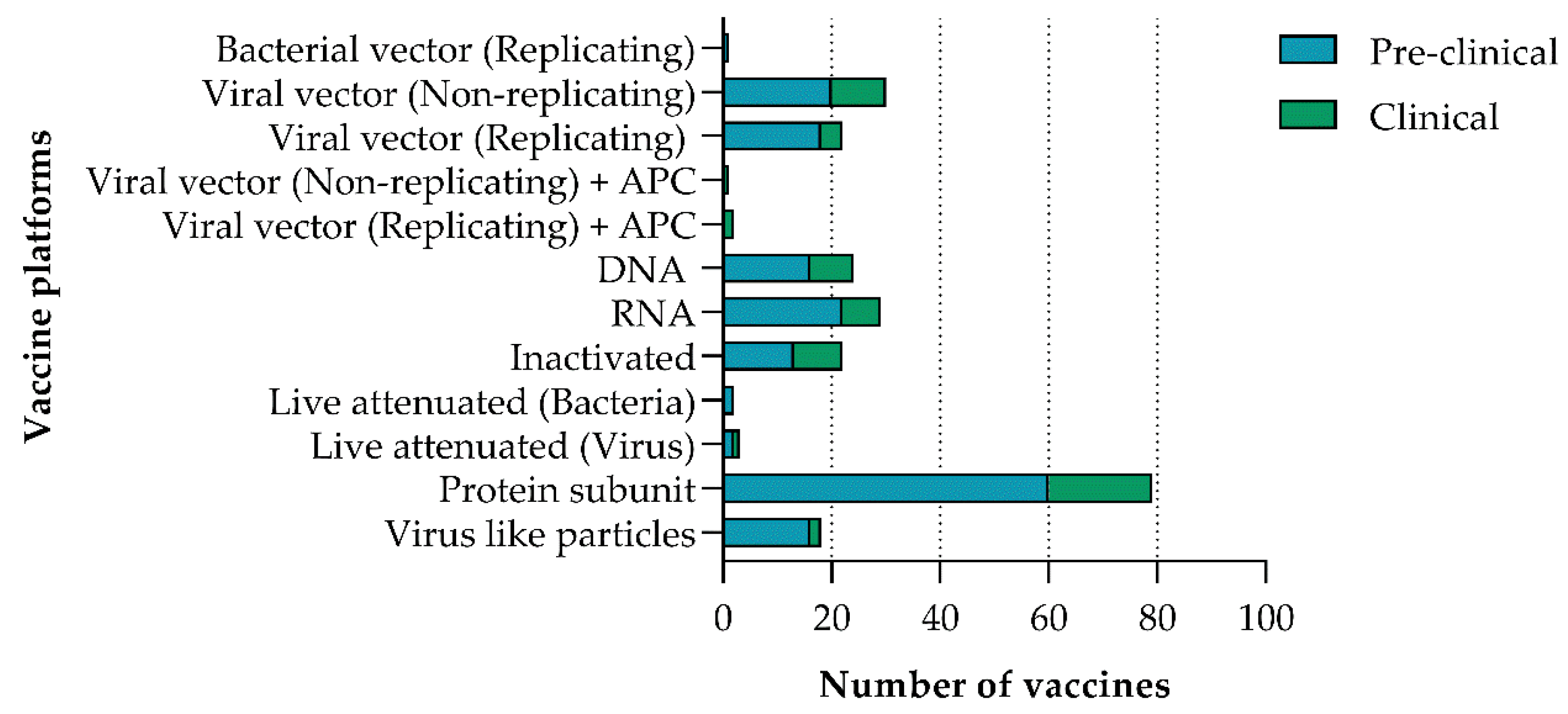
2. Vaccine Platforms
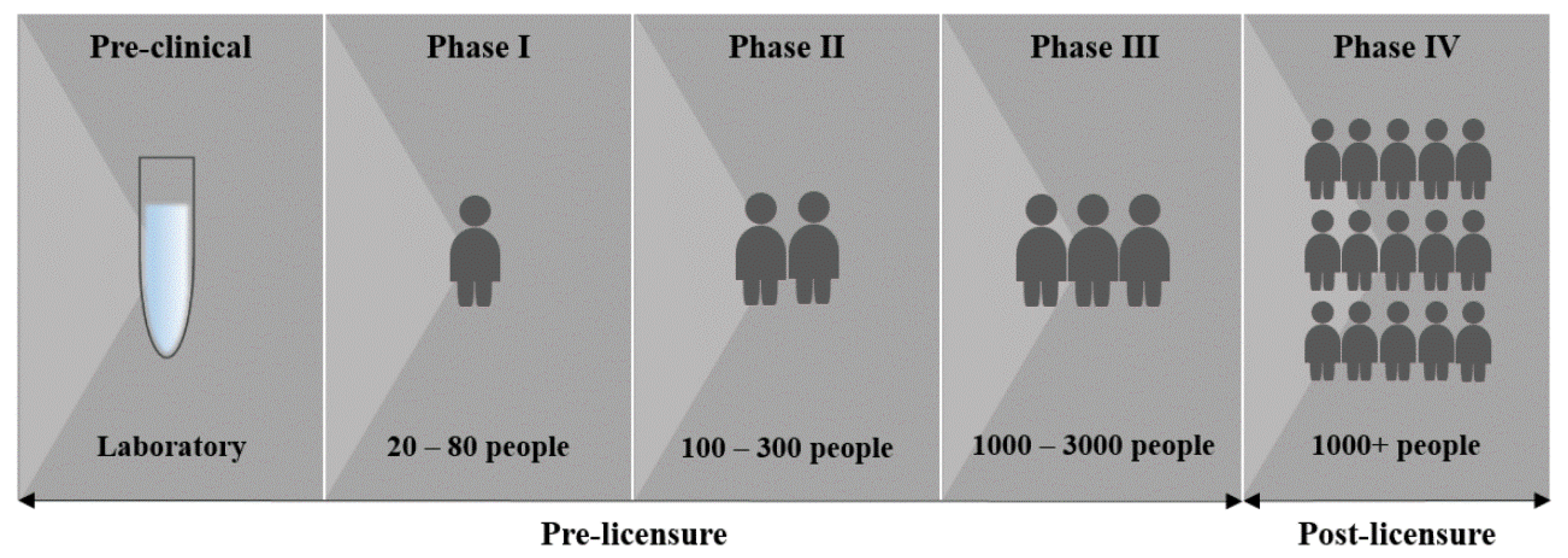
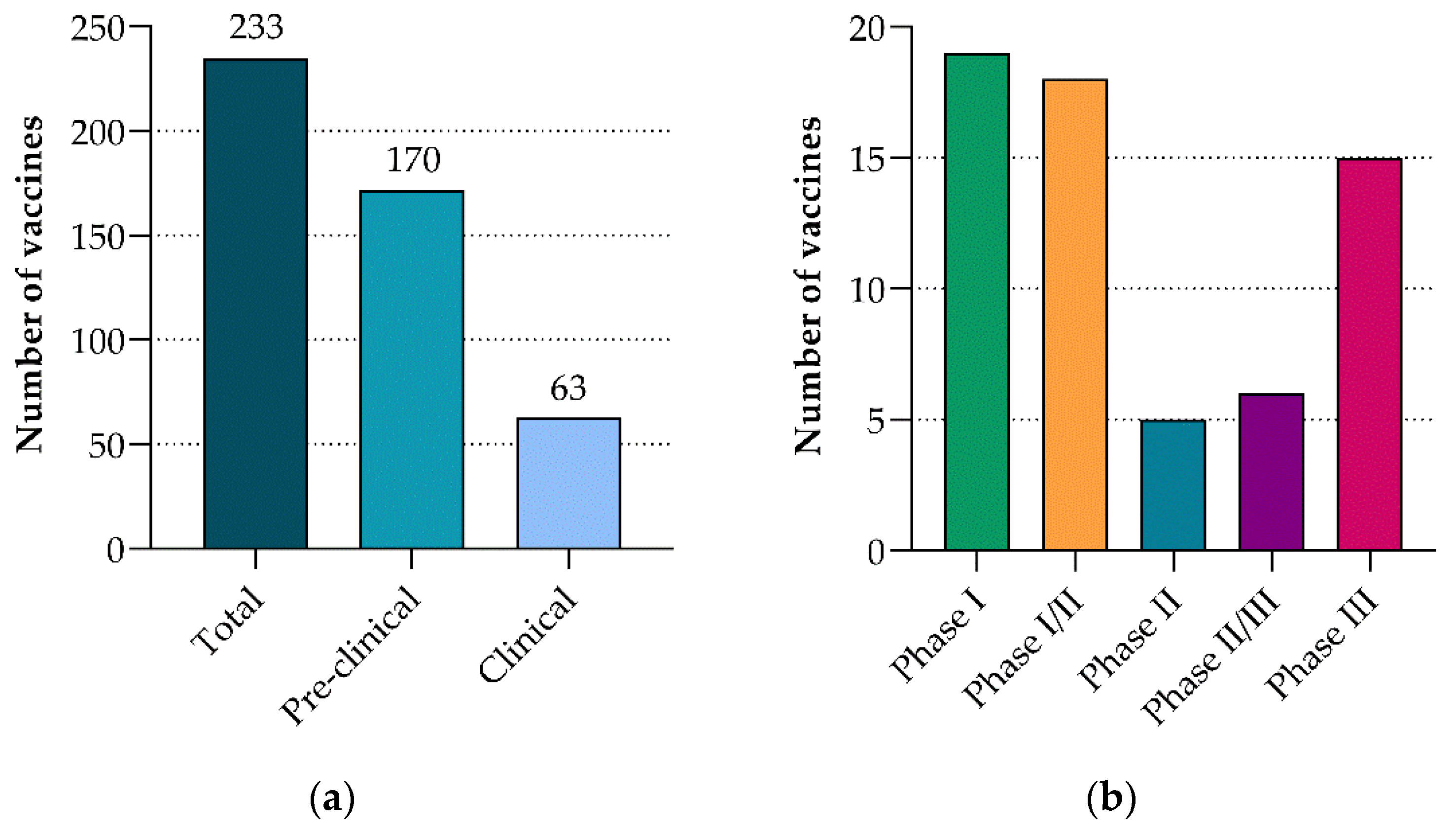
3. Vaccine Development
3.1. Nucleic Acid Vaccines
3.2. Inactivated Vaccines
3.3. Non-Replicating Viral Vector Vaccines
3.4. Protein-Based Vaccines
4. COVID-19 Vaccine Challenges
4.1. Long-Term Immunity
4.2. Antibody-Dependent Enhancement (ADE) of Disease
4.3. Global Distribution
4.4. Vaccine Hesitancy
5. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Novel Coronavirus 2019. Available online: https://www.who.int/emergencies/diseases/novel-coronavirus-2019 (accessed on 8 January 2021).
- World Health Organization. Global Vaccine Action Plan. Available online: https://www.who.int/immunization/global_vaccine_action_plan/GVAP_Introduction_and_Immunization_Landscape_Today.pdf (accessed on 18 September 2020).
- Greenwood, B. The Contribution of Vaccination to Global Health: Past, Present and Future. Philos. Trans. R. Soc. B Biol. Sci. 2014, 369, 20130433. [Google Scholar] [CrossRef] [PubMed]
- Siegrist, C.A. Vaccine Immunology. In Plotkin’s Vaccines; Plotkin, S.A., Orenstein, W.A., Offit, P.A., Edwards, K.M., Eds.; Elsevier Inc.: Philadelphia, PA, USA, 2018; pp. 16–34.e7. ISBN 978-0-323-35761-6. [Google Scholar]
- Braciale, T.J.; Hahn, Y.S. Immunity to Viruses. Immunol. Rev. 2013, 255, 5–12. [Google Scholar] [CrossRef] [PubMed]
- Rodda, L.B.; Netland, J.; Shehata, L.; Pruner, K.B.; Morawski, P.A.; Thouvenel, C.D.; Takehara, K.K.; Eggenberger, J.; Hemann, E.A.; Waterman, H.R.; et al. Functional SARS-CoV-2-Specific Immune Memory Persists after Mild COVID-19. Cell 2020. [Google Scholar] [CrossRef]
- Benn, C.S.; Netea, M.G.; Selin, L.K.; Aaby, P. A Small Jab—A Big Effect: Nonspecific Immunomodulation by Vaccines. Trends Immunol. 2013, 34, 431–439. [Google Scholar] [CrossRef] [PubMed]
- De Gregorio, E.; Rappuoli, R. From Empiricism to Rational Design: A Personal Perspective of the Evolution of Vaccine Development. Nat. Rev. Immunol. 2014, 14, 505–514. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Draft Landscape of COVID-19 Candidate Vaccines. 5 January 2021. Available online: https://www.who.int/who-documents-detail/draft-landscape-of-covid-19-candidate-vaccines (accessed on 7 January 2021).
- Vaccine Types. Available online: https://www.vaccines.gov/basics/types (accessed on 23 July 2020).
- The Washington Post. Elegant but Unproven, RNA Experiments Leap to the Front in Coronavirus Vaccine Race. Will They Work? Available online: https://www.washingtonpost.com/world/europe/coronavirus-vaccine-race-messenger-rna-imperial-college/2020/07/05/6565b2d0-ba2e-11ea-97c1-6cf116ffe26c_story.html (accessed on 20 July 2020).
- Jain, S.; Batra, H.; Yadav, P.; Chand, S. COVID-19 Vaccines Currently under Preclinical and Clinical Studies, and Associated Antiviral Immune Response. Vaccines 2020, 8, 649. [Google Scholar] [CrossRef] [PubMed]
- Folegatti, P.M.; Ewer, K.J.; Aley, P.K.; Angus, B.; Becker, S.; Belij-Rammerstorfer, S.; Bellamy, D.; Bibi, S.; Bittaye, M.; Clutterbuck, E.A.; et al. Safety and Immunogenicity of the ChAdOx1 nCoV-19 Vaccine against SARS-CoV-2: A Preliminary Report of a Phase 1/2, Single-Blind, Randomised Controlled Trial. Lancet 2020, 396, 467–478. [Google Scholar] [CrossRef]
- World Health Organization. COVID-19: Vulnerable and High Risk Groups. Available online: https://www.who.int/westernpacific/emergencies/covid-19/information/high-risk-groups (accessed on 21 July 2020).
- Xiang, Z.; Li, Y.; Cun, A.; Yang, W.; Ellenberg, S.; Switzer, W.M.; Kalish, M.L.; Ertl, H.C.J. Chimpanzee Adenovirus Antibodies in Humans, Sub-Saharan Africa. Emerg. Infect. Dis. 2006, 12, 1596–1599. [Google Scholar] [CrossRef]
- Reuters. Scientists See Downsides to Top COVID-19 Vaccines from Russia. Available online: https://in.reuters.com/article/health-coronavirus-vaccines-adenovirus-f/scientists-see-downsides-to-top-covid-19-vaccines-from-russia-china-idINKBN25R1FW (accessed on 5 September 2020).
- McCoy, K.; Tatsis, N.; Korioth-Schmitz, B.; Lasaro, M.O.; Hensley, S.E.; Lin, S.W.; Li, Y.; Giles-Davis, W.; Cun, A.; Zhou, D.; et al. Effect of Preexisting Immunity to Adenovirus Human Serotype 5 Antigens on the Immune Responses of Nonhuman Primates to Vaccine Regimens Based on Human- or Chimpanzee-Derived Adenovirus Vectors. J. Virol. 2007, 81, 6594–6604. [Google Scholar] [CrossRef]
- Sumida, S.M.; Truitt, D.M.; Kishko, M.G.; Arthur, J.C.; Jackson, S.S.; Gorgone, D.A.; Lifton, M.A.; Koudstaal, W.; Pau, M.G.; Kostense, S.; et al. Neutralizing Antibodies and CD8+ T Lymphocytes both Contribute to Immunity to Adenovirus Serotype 5 Vaccine Vectors. J. Virol. 2004, 78, 2666–2673. [Google Scholar] [CrossRef]
- Calcedo, R.; Vandenberghe, L.H.; Roy, S.; Somanathan, S.; Wang, L.; Wilson, J.M. Host Immune Responses to Chronic Adenovirus Infections in Human and Nonhuman Primates. J. Virol. 2009, 83, 2623–2631. [Google Scholar] [CrossRef] [PubMed]
- Chirmule, N.; Propert, K.J.; Magosin, S.A.; Qian, Y.; Qian, R.; Wilson, J.M. Immune Responses to Adenovirus and Adeno-Associated Virus in Humans. Gene Ther. 1999, 6, 1574–1583. [Google Scholar] [CrossRef] [PubMed]
- Sadoff, J.; Le Gars, M.; Shukarev, G.; Heerwegh, D.; Truyers, C.; de Groot, A.M.; Stoop, J.; Tete, S.; Van Damme, W.; Leroux-Roels, I.; et al. Safety and Immunogenicity of the Ad26.COV2.S COVID-19 Vaccine Candidate: Interim Results of a Phase 1/2a, Double-Blind, Randomized, Placebo-Controlled Trial. medRxiv 2020. [Google Scholar] [CrossRef]
- Sputnik V: Clinical Trials. Available online: https://sputnikvaccine.com/about-vaccine/clinical-trials/ (accessed on 1 October 2020).
- Barouch, D.H.; Kik, S.V.; Weverling, G.J.; Dilan, R.; King, S.L.; Maxfield, L.F.; Clark, S.; Ng’ang’a, D.; Brandariz, K.L.; Abbink, P.; et al. International Seroepidemiology of Adenovirus Serotypes 5, 26, 35, and 48 in Pediatric and Adult Populations. Vaccine 2011, 29, 5203–5209. [Google Scholar] [CrossRef]
- Pronker, E.S.; Weenen, T.C.; Commandeur, H.; Claassen, E.H.J.H.M.; Osterhaus, A.D.M.E. Risk in Vaccine Research and Development Quantified. PLoS ONE 2013, 8, e57755. [Google Scholar] [CrossRef] [PubMed]
- Gao, Q.; Bao, L.; Mao, H.; Wang, L.; Xu, K.; Yang, M.; Li, Y.; Zhu, L.; Wang, N.; Lv, Z.; et al. Development of an Inactivated Vaccine Candidate for SARS-CoV-2. Science 2020, 369, 77–81. [Google Scholar] [CrossRef]
- Yu, J.; Tostanoski, L.H.; Peter, L.; Mercado, N.B.; McMahan, K.; Mahrokhian, S.H.; Nkolola, J.P.; Liu, J.; Li, Z.; Chandrashekar, A.; et al. DNA Vaccine Protection against SARS-CoV-2 in Rhesus Macaques. Science 2020, 369, 806–811. [Google Scholar] [CrossRef]
- Singh, K.; Mehta, S. The Clinical Development Process for a Novel Preventive Vaccine: An Overview. J. Postgrad. Med. 2016, 62, 4. [Google Scholar] [CrossRef]
- Dai, L.; Gao, G.F. Viral targets for vaccines against COVID-19. Nat. Rev. Immunol. 2020. [Google Scholar] [CrossRef]
- Anderson, E.J.; Rouphael, N.G.; Widge, A.T.; Jackson, L.A.; Roberts, P.C.; Makhene, M.; Chappell, J.D.; Denison, M.R.; Stevens, L.J.; Pruijssers, A.J.; et al. Safety and Immunogenicity of SARS-CoV-2 mRNA-1273 Vaccine in Older Adults. N. Engl. J. Med. 2020, 383, 2427–2438. [Google Scholar] [CrossRef]
- Yancy, C.W. COVID-19 and African Americans. JAMA 2020, 323, 1891. [Google Scholar] [CrossRef] [PubMed]
- Centre for Disease Control and Prevention. COVID-19 Hospitalization and Death by Race/Ethnicity. Available online: https://www.cdc.gov/coronavirus/2019-ncov/covid-data/investigations-discovery/hospitalization-death-by-race-ethnicity.html (accessed on 21 December 2020).
- Bassett, M.T.; Chen, J.T.; Krieger, N. Variation in racial/ethnic disparities in COVID-19 mortality by age in the United States: A cross-sectional study. PLOS Med. 2020, 17, e1003402. [Google Scholar] [CrossRef] [PubMed]
- US Food and Drug Association. Drug Trials Snapshot: Summary Report. 2019. Available online: https://www.fda.gov/media/135337/download (accessed on 5 November 2020).
- Jackson, L.A.; Anderson, E.J.; Rouphael, N.G.; Roberts, P.C.; Makhene, M.; Coler, R.N.; McCullough, M.P.; Chappell, J.D.; Denison, M.R.; Stevens, L.J.; et al. An mRNA Vaccine against SARS-CoV-2—Preliminary Report. N. Engl. J. Med. 2020, 383, 1920–1931. [Google Scholar] [CrossRef] [PubMed]
- Moderna. Moderna Announces Primary Efficacy Analysis in Phase 3 COVE Study for Its COVID-19 Vaccine Candidate and Filing Today with U.S. FDA for Emergency Use Authorization. Available online: https://investors.modernatx.com/news-releases/news-release-details/moderna-announces-primary-efficacy-analysis-phase-3-cove-study (accessed on 21 December 2020).
- Widge, A.T.; Rouphael, N.G.; Jackson, L.A.; Anderson, E.J.; Roberts, P.C.; Makhene, M.; Chappell, J.D.; Denison, M.R.; Stevens, L.J.; Pruijssers, A.J.; et al. Durability of Responses after SARS-CoV-2 mRNA-1273 Vaccination. N. Engl. J. Med. 2020. [Google Scholar] [CrossRef]
- FDA News. FDA Takes Additional Action in Fight against COVID-19 by Issuing Emergency Use Authorization for Second COVID-19 Vaccine. Available online: https://www.fda.gov/news-events/press-announcements/fda-takes-additional-action-fight-against-covid-19-issuing-emergency-use-authorization-second-covid (accessed on 21 December 2020).
- The New York Times. Boston Doctor Reports Serious Allergic Reaction after Getting Moderna’s Covid Vaccine. Available online: https://www.nytimes.com/2020/12/25/health/Covid-moderna-vaccine-allergies.html (accessed on 1 January 2021).
- The New York Times. Moderna Plans to Begin Testing Its Coronavirus Vaccine in Children. Available online: https://www.nytimes.com/2020/12/02/health/Covid-Moderna-vaccine-children.html (accessed on 21 December 2020).
- Polack, F.P.; Thomas, S.J.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Pérez Marc, G.; Moreira, E.D.; Zerbini, C.; et al. Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine. N. Engl. J. Med. 2020, 383, 2603–2615. [Google Scholar] [CrossRef] [PubMed]
- FDA News. FDA Takes Key Action in Fight against COVID-19 by Issuing Emergency Use Authorization for First COVID-19 Vaccine. Available online: https://www.fda.gov/news-events/press-announcements/fda-takes-key-action-fight-against-covid-19-issuing-emergency-use-authorization-first-covid-19 (accessed on 21 December 2020).
- The Guardian. Covid Vaccine: UK Woman Becomes First in World to Receive Pfizer Jab. Available online: https://www.theguardian.com/world/2020/dec/08/coventry-woman-90-first-patient-to-receive-covid-vaccine-in-nhs-campaign (accessed on 21 December 2020).
- CNN. Third Alaskan Health Care Worker Has Allergic Reaction to Covid-19 Vaccine. Available online: https://edition.cnn.com/2020/12/18/health/alaska-third-allergic-reaction-vaccine/index.html (accessed on 21 December 2020).
- BBC News. Covid-19 Vaccine: Allergy Warning over New Jab. Available online: https://www.bbc.com/news/health-55244122 (accessed on 21 December 2020).
- Science News. Suspicions Grow That Nanoparticles in Pfizer’s COVID-19 Vaccine Trigger Rare Allergic Reactions. Available online: https://www.sciencemag.org/news/2020/12/suspicions-grow-nanoparticles-pfizer-s-covid-19-vaccine-trigger-rare-allergic-reactions (accessed on 1 January 2021).
- Castells, M.C.; Phillips, E.J. Maintaining Safety with SARS-CoV-2 Vaccines. N. Engl. J. Med. 2020. [Google Scholar] [CrossRef]
- Zhu, F.C.; Guan, X.H.; Li, Y.H.; Huang, J.Y.; Jiang, T.; Hou, L.H.; Li, J.X.; Yang, B.F.; Wang, L.; Wang, W.J.; et al. Immunogenicity and Safety of a Recombinant Adenovirus Type-5-Vectored COVID-19 Vaccine in Healthy Adults Aged 18 Years or Older: A Randomised, Double-Blind, Placebo-Controlled, Phase 2 Trial. Lancet 2020, 396, 479–488. [Google Scholar] [CrossRef]
- Zhu, F.C.; Li, Y.H.; Guan, X.H.; Hou, L.H.; Wang, W.J.; Li, J.X.; Wu, S.P.; Wang, B.S.; Wang, Z.; Wang, L.; et al. Safety, Tolerability, and Immunogenicity of a Recombinant Adenovirus Type-5 Vectored COVID-19 Vaccine: A Dose-Escalation, Open-Label, Non-randomised, First-in-Human Trial. Lancet 2020, 395, 1845–1854. [Google Scholar] [CrossRef]
- Burki, T.K. The Russian Vaccine for COVID-19. Lancet Respir. Med. 2020, 8, e85–e86. [Google Scholar] [CrossRef]
- Science News. Russia’s Approval of a COVID-19 Vaccine Is Less Than Meets the Press Release. Available online: https://www.sciencemag.org/news/2020/08/russia-s-approval-covid-19-vaccine-less-meets-press-release51 (accessed on 20 December 2020).
- Logunov, D.Y.; Dolzhikova, I.V.; Zubkova, O.V.; Tukhvatullin, A.I.; Shcheblyakov, D.V.; Dzharullaeva, A.S.; Grousova, D.M.; Erokhova, A.S.; Kovyrshina, A.V.; Botikov, A.G.; et al. Safety and Immunogenicity of an rAd26 and rAd5 Vector-Based Heterologous Prime-Boost COVID-19 Vaccine in Two Formulations: Two Open, Non-randomised Phase 1/2 Studies from Russia. Lancet 2020, 396, 887–897. [Google Scholar] [CrossRef]
- FDA News. AstraZeneca Partners with Russia’s Gamaleya Research Institute for Combination COVID-19 Vaccine. Available online: https://www.fdanews.com/articles/200473-astrazeneca-partners-with-russias-gamaleya-research-institute-for-combination-covid-19-vaccine2020 (accessed on 20 December 2020).
- Mahase, E. Covid-19: Johnson and Johnson Vaccine Trial Is Paused Because of Unexplained Illness in Participant. BMJ 2020, m3967. [Google Scholar] [CrossRef] [PubMed]
- Pharmaceutical Technology. Janssen Begins Second Phase III Covid-19 Vaccine Trial. Available online: https://www.pharmaceutical-technology.com/news/janssen-ensemble-2-trial/ (accessed on 21 December 2020).
- The Guardian. Oxford Covid Vaccine Works in All Ages, Trial Suggest. Available online: https://www.theguardian.com/world/2020/oct/27/covid-vaccine-uk-oxford-university-astrazeneca-works-in-all-ages-trials-suggest (accessed on 21 December 2020).
- University of the Witwatersrand. Covid19 News. The First Covid-19 Vaccine Trial in South Africa Begins. Available online: https://www.wits.ac.za/covid19/covid19-news/latest/the-first-covid-19-vaccine-trial-in-south-africa-begins.html (accessed on 5 July 2020).
- University of Oxford. Trial of Oxford COVID-19 Vaccine Starts in Brazil. Available online: https://www.research.ox.ac.uk/Article/2020-06-28-trial-of-oxford-covid-19-vaccine-starts-in-brazil (accessed on 21 July 2020).
- The New York Times. AstraZeneca pauses Covid-19 Vaccine Trial for Safety Review. Available online: https://www.deccanherald.com/science-and-environment/astrazeneca-pauses-covid-19-vaccine-trial-for-safety-review-884647.html (accessed on 5 November 2020).
- Voysey, M.; Clemens, S.A.C.; Madhi, S.A.; Weckx, L.Y.; Folegatti, P.M.; Aley, P.K.; Angus, B.; Baillie, V.L.; Barnabas, S.L.; Bhorat, Q.E.; et al. Safety and Efficacy of the ChAdOx1 nCoV-19 Vaccine (AZD1222) against SARS-CoV-2: An Interim Analysis of Four Randomised Controlled Trials in Brazil, South Africa, and the UK. Lancet 2020. [Google Scholar] [CrossRef]
- BBC News. Oxford-AstraZeneca Vaccine: Bogus Reports, Accidental Finds—The Story of the Jab. Available online: https://www.bbc.com/news/health-55308216 (accessed on 20 December 2020).
- Keech, C.; Albert, G.; Cho, I.; Robertson, A.; Reed, P.; Neal, S.; Plested, J.S.; Zhu, M.; Cloney-Clark, S.; Zhou, H.; et al. Phase 1–2 Trial of a SARS-CoV-2 Recombinant Spike Protein Nanoparticle Vaccine. N. Engl. J. Med. 2020, 383, 2320–2332. [Google Scholar] [CrossRef] [PubMed]
- Dai, L.; Zheng, T.; Xu, K.; Han, Y.; Xu, L.; Huang, E.; An, Y.; Cheng, Y.; Li, S.; Liu, M.; et al. A Universal Design of Betacoronavirus Vaccines against COVID-19, MERS, and SARS. Cell 2020, 182, 722–733. [Google Scholar] [CrossRef] [PubMed]
- Liu, A.; Wang, W.; Zhao, X.; Zhou, X.; Yang, D.; Lu, M.; Lv, Y. Disappearance of Antibodies to SARS-CoV-2 in a -COVID-19 Patient after Recovery. Clin. Microbiol. Infect. 2020. [Google Scholar] [CrossRef] [PubMed]
- Cao, W.C.; Liu, W.; Zhang, P.H.; Zhang, F.; Richardus, J.H. Disappearance of Antibodies to SARS-Associated Coronavirus after Recovery. N. Engl. J. Med. 2007, 357, 1162–1163. [Google Scholar] [CrossRef] [PubMed]
- Graham, B.S. Advances in Antiviral Vaccine Development. Immunol. Rev. 2013, 255, 230–242. [Google Scholar] [CrossRef]
- Davis, C.W.; Jackson, K.J.L.; McCausland, M.M.; Darce, J.; Chang, C.; Linderman, S.L.; Chennareddy, C.; Gerkin, R.; Brown, S.J.; Wrammert, J.; et al. Influenza Vaccine-Induced Human Bone Marrow Plasma Cells Decline within a Year after Vaccination. Science 2020, 370, 237–241. [Google Scholar] [CrossRef]
- Stat News. The World Needs Covid-19 Vaccines. It May Also be Overestimating Their Power. Available online: https://www.statnews.com/2020/05/22/the-world-needs-covid-19-vaccines-it-may-also-be-overestimating-their-power/ (accessed on 5 July 2020).
- Yang, L.; Liu, S.; Liu, J.; Zhang, Z.; Wan, X.; Huang, B.; Chen, Y.; Zhang, Y. COVID-19: Immunopathogenesis and Immunotherapeutics. Signal Transduct. Target. Ther. 2020, 5, 128. [Google Scholar] [CrossRef]
- Arvin, A.M.; Fink, K.; Schmid, M.A.; Cathcart, A.; Spreafico, R.; Havenar-Daughton, C.; Lanzavecchia, A.; Corti, D.; Virgin, H.W. A Perspective on Potential Antibody-Dependent Enhancement of SARS-CoV-2. Nature 2020, 584, 353–363. [Google Scholar] [CrossRef]
- Joyner, M.J.; Bruno, K.A.; Klassen, S.A.; Kunze, K.L.; Johnson, P.W.; Lesser, E.R.; Wiggins, C.C.; Senefeld, J.W.; Klompas, A.M.; Hodge, D.O.; et al. Safety Update. Mayo Clin. Proc. 2020, 95, 1888–1897. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, S.; Soni, K.D.; Khanna, P. Convalescent Plasma Is a Clutch at Straws in COVID-19 Management! A Systematic Review and Meta-Analysis. J. Med. Virol. 2020, 93, 1111–1118. [Google Scholar] [CrossRef] [PubMed]
- Joyner, M.J.; Senefeld, J.W.; Klassen, S.A.; Mills, J.R.; Johnson, P.W.; Theel, E.S.; Wiggins, C.C.; Bruno, K.A.; Klompas, A.M.; Lesser, E.R.; et al. Effect of Convalescent Plasma on Mortality among Hospitalized Patients with COVID-19: Initial Three-Month Experience. medRxiv 2020. [Google Scholar] [CrossRef]
- Hansen, J.; Baum, A.; Pascal, K.E.; Russo, V.; Giordano, S.; Wloga, E.; Fulton, B.O.; Yan, Y.; Koon, K.; Patel, K.; et al. Studies in Humanized Mice and Convalescent Humans Yield a SARS-CoV-2 Antibody Cocktail. Science 2020, 369, 1010–1014. [Google Scholar] [CrossRef] [PubMed]
- The New York Times. President Trump Received Experimental Antibody Treatment. Available online: https://www.nytimes.com/2020/10/02/health/trump-antibody-treatment.html (accessed on 20 October 2020).
- Regeneron. Regeneron’s COVID-19 Outpatient Trial Prospectively Demonstrated That REGN-COV2 Antibody Cocktail Significantly Reduced Virus Levels and Need for Further Medical Attention. Available online: https://investor.regeneron.com/news-releases/news-release-details/regenerons-covid-19-outpatient-trial-prospectively-demonstrates (accessed on 11 November 2020).
- The New York Times. Eli Lilly’s Antibody Trial Is Paused over Potential Safety Concern. Available online: https://www.nytimes.com/2020/10/13/health/eli-lilly-antibody.html (accessed on 11 November 2020).
- The New York Times. The, U.S. Commits to Buying Millions of Vaccine Doses—Why That’s Unusual. Available online: https://www.nytimes.com/2020/07/22/upshot/vaccine-coronavirus-government-purchase.html (accessed on 20 October 2020).
- The New York Times. A New Front for Nationalism: The Global Battle against a Virus. Available online: https://www.nytimes.com/2020/04/10/business/coronavirus-vaccine-nationalism.html (accessed on 5 July 2020).
- The Conversation. Pfizer’s Ultra-Cold Vaccine Could Be Difficult to Distribute. Available online: https://theconversation.com/pfizers-ultra-cold-vaccine-could-be-difficult-to-distribute-149848 (accessed on 11 November 2020).
- Eye Witness News. Read: President Ramaphosa’s Full Speech on Easing of Lockdown to Alert Level 1. Available online: https://ewn.co.za/2020/09/16/read-president-ramaphosa-s-full-speech-on-easing-of-lockdown-to-alert-level-1 (accessed on 20 October 2020).
- News 24. Covax: Who Will Be the First in SA to Get the Vaccine. Available online: https://www.news24.com/news24/southafrica/news/covax-who-will-be-the-first-in-sa-to-get-the-vaccine-20201224 (accessed on 30 December 2020).
- Wakefield, A.; Murch, S.; Anthony, A.; Linnell, J.; Casson, D.; Malik, M.; Berelowitz, M.; Dhillon, A.; Thomson, M.; Harvey, P.; et al. Retracted: Ileal-Lymphoid-Nodular Hyperplasia, Non-specific Colitis, and Pervasive Developmental Disorder in Children. Lancet 1998, 351, 637–641. [Google Scholar] [CrossRef]
- Dror, A.A.; Eisenbach, N.; Taiber, S.; Morozov, N.G.; Mizrachi, M.; Zigron, A.; Srouji, S.; Sela, E. Vaccine Hesitancy: The Next Challenge in the Fight against COVID-19. Eur. J. Epidemiol. 2020, 35, 775–779. [Google Scholar] [CrossRef]
| Candidate | Platform | Vaccine Characteristics | Vaccine Targets | Developers | Clinical Stage |
|---|---|---|---|---|---|
| PicoVacc/CoronaVac | Inactivated | Inactivated SARS-CoV-2 alum adjuvant | Whole virus | Sinovac Research and Development Co., Ltd. | Phase I/II NCT04551547 Phase III NCT04456595 |
| Inactivated SARS-CoV-2 vaccine | Inactivated | - | Whole virus | Beijing Institute of Biological Products/Sinopharm | Phase I/II ChiCTR2000032459 Phase III NCT04560881 |
| Inactivated SARS-CoV-2 vaccine | Inactivated | - | Whole virus | Wuhan Institute of Biological Products/Sinopharm | Phase I/II ChiCTR2000031809 Phase III ChiCTR2000034780 |
| BBV152 | Inactivated | Inactivated whole virion | Whole virus | Bharat Biotech International Limited | Phase I/II CTRI/2020/07/026300 CTRI/2020/09/027674 NCT04471519 Phase III CTRI/2020/11/028976 NCT04641481 |
| ChAdOx1 nCoV-19/AZD1222/Covishield | Non-replicating viral vector | Chimpanzee adenovirus containing the genetic sequence of the SARS-CoV-2 surface spike protein | Full-length S protein | University of Oxford/AstraZeneca | Phase I PACTR202005681895696 Phase I/II PACTR202006922165132 Phase II/III NCT04400838 Phase III ISRCTN89951424 |
| Ad5-nCoV | Non-replicating viral vector | Ad5 vector | Full-length S protein | CanSino Biological Inc./Beijing Institute of Biotechnology | Phase I ChiCTR2000030906 Phase II ChiCTR2000031781 Phase III NCT04526990 |
| Sputnik V | Non-replicating viral vector | Recombinant adenovirus type 26 (rAd26) and type 5 (rAd5) vectors carrying the gene for SARS-CoV-2 spike glycoprotein (rAd26-S and rAd5-S) | Full-length S protein | Gamaleya Research Institute/Health Ministry of the Russian Federation | Phase I/II NCT04436471 Phase III NCT04530396 |
| Ad26.COV2.S | Non-replicating viral vector | Ad26 vector | Full-length S protein with 2 proline substitutions (K986P and V987P) and 2 mutations at furin cleavage site (R682S and R685G) | Janssen Pharmaceutical Companies | Phase I NCT04509947 Phase I/II NCT04436276 Phase II EUCTR2020-002584-63-DE Phase III NCT04505722 |
| NVX-CoV2373 | Protein subunit | Full length recombinant SARS-CoV-2 glycoprotein nanoparticle vaccine adjuvanted with Matrix M | Full-length S protein | Novavax | Phase I/II NCT04368988 Phase II NCT04533399 Phase III 2020-004123-16 NCT04611802 |
| - | Protein subunit | Recombinant SARS-CoV-2 vaccine (CHO Cell) | RBD-dimer (residues 319–537 as tandem repeat) | Anhui Zhifei Longcom Biopharmaceutical/Institute of Microbiology, Chinese Academy of Sciences | Phase I NCT04445194 Phase I/II NCT04550351 Phase II NCT04466085 Phase III ChiCTR2000040153 |
| mRNA-1273 | RNA | LNP-encapsulated mRNA encoding the surface spike protein | Full-length S protein with 2 proline substitutions (K986P and V987P) | Moderna/NIAID | Phase I NCT04283461 Phase II NCT04405076 Phase II/III NCT04649151 Phase III NCT04470427 |
| BNT162b2 | RNA | LNP nucleoside-modified mRNA encoding an optimized SARS-CoV-2 RBD antigen | Full-length S protein with 2 proline substitutions (K986P and V987P) | BioNTech/Fosun Pharma/Jiangsu Provincial Center for Disease Prevention and Control/Pfizer | Phase I/II 2020-001038-36 ChiCTR2000034825 NCT04537949 Phase III NCT04368728 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mellet, J.; Pepper, M.S. A COVID-19 Vaccine: Big Strides Come with Big Challenges. Vaccines 2021, 9, 39. https://doi.org/10.3390/vaccines9010039
Mellet J, Pepper MS. A COVID-19 Vaccine: Big Strides Come with Big Challenges. Vaccines. 2021; 9(1):39. https://doi.org/10.3390/vaccines9010039
Chicago/Turabian StyleMellet, Juanita, and Michael S. Pepper. 2021. "A COVID-19 Vaccine: Big Strides Come with Big Challenges" Vaccines 9, no. 1: 39. https://doi.org/10.3390/vaccines9010039
APA StyleMellet, J., & Pepper, M. S. (2021). A COVID-19 Vaccine: Big Strides Come with Big Challenges. Vaccines, 9(1), 39. https://doi.org/10.3390/vaccines9010039






