Immunotherapy with Checkpoint Inhibitors for Hepatocellular Carcinoma: Where Are We Now?
Abstract
1. Introduction
2. Materials and Methods
3. Results
3.1. Concluded and Ongoing Trials
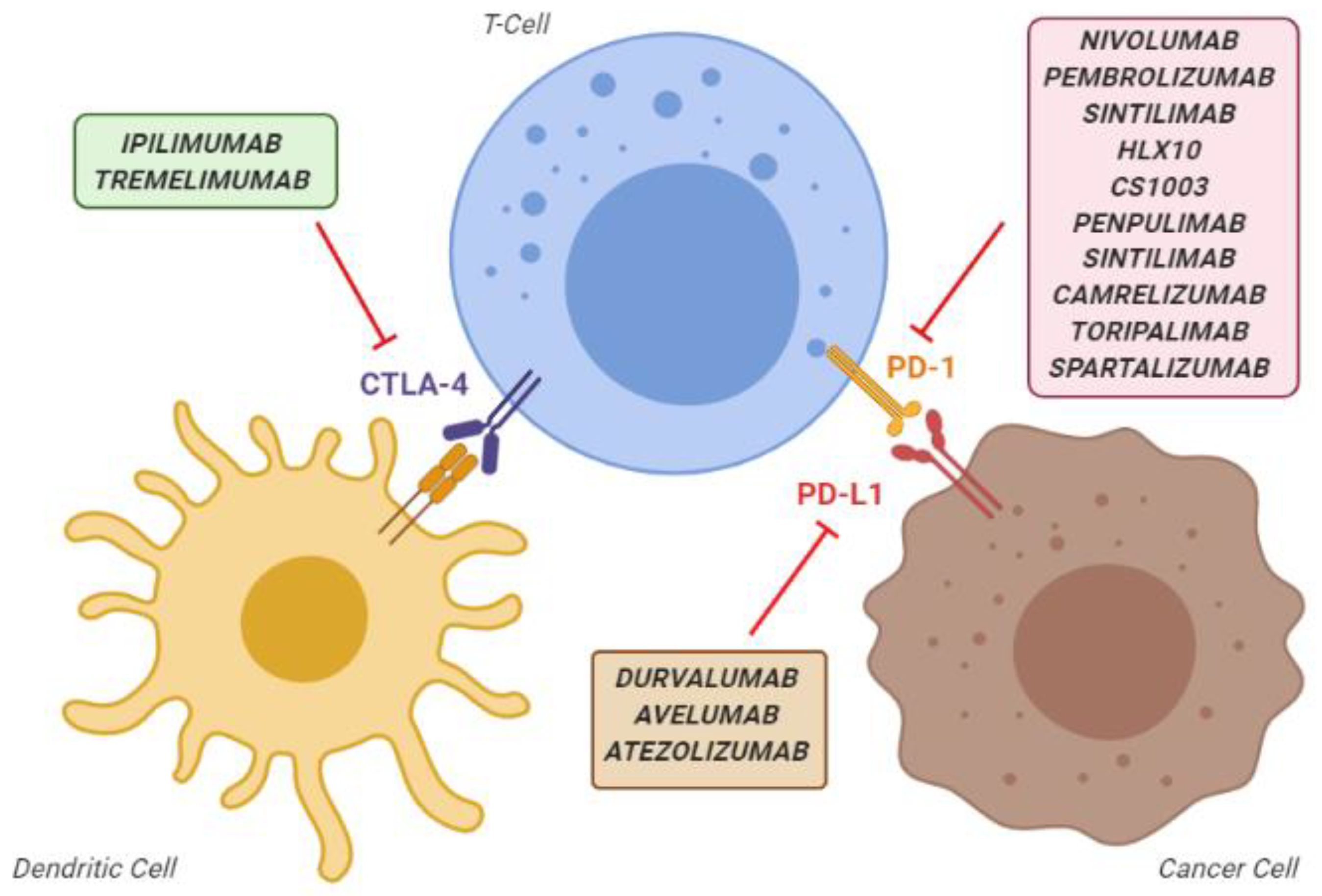
3.1.1. Anti-PD1 Inhibitors as Single Agents
3.1.2. Combination of PD1 Inhibitors with Intravenous Anti-VEGF Agents
3.1.3. Combination of PD1 Inhibitors with Tyrosine Kinase Inhibitors
3.1.4. Combination of PD-1 and CTLA-4 Inhibitors (Dual Checkpoint Blockade)
3.1.5. Other Therapeutic Combination of Immune Checkpoint Inhibitors
3.1.6. Adjuvant and Neoadjuvant Setting
3.1.7. Combination with Local Treatments
3.2. Open Problems
3.2.1. Eligibility in the Real-World Clinical Practice
3.2.2. Safety
3.2.3. Unpredictable Efficacy, the Need for Biomarkers
3.2.4. Resistance to Immune Checkpoint Inhibitors
3.2.5. The Radiological Evaluation of Response
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Villanueva, A. Hepatocellular Carcinoma. N. Engl. J. Med. 2019, 380, 1450–1462. [Google Scholar] [CrossRef] [PubMed]
- Llovet, J.M.; Ricci, S.; Mazzaferro, V.; Hilgard, P.; Gane, E.; Blanc, J.-F.; de Oliveira, A.C.; Santoro, A.; Raoul, J.-L.; Forner, A.; et al. Sorafenib in advanced hepatocellular carcinoma. N. Engl. J. Med. 2008, 359, 378–390. [Google Scholar] [CrossRef] [PubMed]
- Tovoli, F.; Negrini, G.; Benevento, F.; Faggiano, C.; Goio, E.; Granito, A. Systemic treatments for hepatocellular carcinoma: Challenges and future perspectives. Hepatic Oncol. 2018, 5, HEP01. [Google Scholar] [CrossRef] [PubMed]
- Nishida, N.; Arizumi, T.; Hagiwara, S.; Ida, H.; Sakurai, T.; Kudo, M. MicroRNAs for the Prediction of Early Response to Sorafenib Treatment in Human Hepatocellular Carcinoma. Liver Cancer 2017, 6, 113–125. [Google Scholar] [CrossRef]
- Prieto, J.; Melero, I.; Sangro, B. Immunological landscape and immunotherapy of hepatocellular carcinoma. Nat. Rev. Gastroenterol. Hepatol. 2015, 12, 681–700. [Google Scholar] [CrossRef]
- Shi, F.; Shi, M.; Zeng, Z.; Qi, R.-Z.; Liu, Z.-W.; Zhang, J.-Y.; Yang, Y.-P.; Tien, P.; Wang, F.-S. PD-1 and PD-L1 upregulation promotes CD8(+) T-cell apoptosis and postoperative recurrence in hepatocellular carcinoma patients. Int. J. Cancer 2011, 128, 887–896. [Google Scholar] [CrossRef]
- Topalian, S.L.; Drake, C.G.; Pardoll, D.M. Immune checkpoint blockade: A common denominator approach to cancer therapy. Cancer Cell 2015, 27, 450–461. [Google Scholar] [CrossRef]
- Whiteside, T.L.; Demaria, S.; Rodriguez-Ruiz, M.E.; Zarour, H.M.; Melero, I. Emerging Opportunities and Challenges in Cancer Immunotherapy. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2016, 22, 1845–1855. [Google Scholar] [CrossRef]
- Postow, M.A.; Callahan, M.K.; Wolchok, J.D. Immune Checkpoint Blockade in Cancer Therapy. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2015, 33, 1974–1982. [Google Scholar] [CrossRef]
- Ringelhan, M.; Pfister, D.; O’Connor, T.; Pikarsky, E.; Heikenwalder, M. The immunology of hepatocellular carcinoma. Nat. Immunol. 2018, 19, 222–232. [Google Scholar] [CrossRef]
- Yau, T.; Park, J.W.; Finn, R.S.; Cheng, A.-L.; Mathurin, P.; Edeline, J.; Kudo, M.; Han, K.-H.; Harding, J.J.; Merle, P.; et al. CheckMate 459: A randomized, multi-center phase III study of nivolumab (NIVO) vs sorafenib (SOR) as first-line (1L) treatment in patients (pts) with advanced hepatocellular carcinoma (aHCC). Ann. Oncol. 2019, 30, v874–v875. [Google Scholar] [CrossRef]
- Finn, R.S.; Qin, S.; Ikeda, M.; Galle, P.R.; Ducreux, M.; Kim, T.-Y.; Kudo, M.; Breder, V.; Merle, P.; Kaseb, A.O.; et al. Atezolizumab plus Bevacizumab in Unresectable Hepatocellular Carcinoma. N. Engl. J. Med. 2020, 382, 1894–1905. [Google Scholar] [CrossRef] [PubMed]
- Kudo, M.; Finn, R.S.; Qin, S.; Han, K.-H.; Ikeda, K.; Piscaglia, F.; Baron, A.; Park, J.-W.; Han, G.; Jassem, J.; et al. Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellular carcinoma: A randomised phase 3 non-inferiority trial. Lancet Lond. Engl. 2018, 391, 1163–1173. [Google Scholar] [CrossRef]
- Sangro, B.; Gomez-Martin, C.; de la Mata, M.; Iñarrairaegui, M.; Garralda, E.; Barrera, P.; Riezu-Boj, J.I.; Larrea, E.; Alfaro, C.; Sarobe, P.; et al. A clinical trial of CTLA-4 blockade with tremelimumab in patients with hepatocellular carcinoma and chronic hepatitis C. J. Hepatol. 2013, 59, 81–88. [Google Scholar] [CrossRef]
- El-Khoueiry, A.B.; Sangro, B.; Yau, T.; Crocenzi, T.S.; Kudo, M.; Hsu, C.; Kim, T.-Y.; Choo, S.-P.; Trojan, J.; Welling, T.H.; et al. Nivolumab in patients with advanced hepatocellular carcinoma (CheckMate 040): An open-label, non-comparative, phase 1/2 dose escalation and expansion trial. Lancet Lond. Engl. 2017, 389, 2492–2502. [Google Scholar] [CrossRef]
- Zhu, A.X.; Finn, R.S.; Edeline, J.; Cattan, S.; Ogasawara, S.; Palmer, D.; Verslype, C.; Zagonel, V.; Fartoux, L.; Vogel, A.; et al. Pembrolizumab in patients with advanced hepatocellular carcinoma previously treated with sorafenib (KEYNOTE-224): A non-randomised, open-label phase 2 trial. Lancet Oncol. 2018, 19, 940–952. [Google Scholar] [CrossRef]
- Qin, S.; Ren, Z.; Meng, Z.; Chen, Z.; Chai, X.; Xiong, J.; Bai, Y.; Yang, L.; Zhu, H.; Fang, W.; et al. Camrelizumab in patients with previously treated advanced hepatocellular carcinoma: A multicentre, open-label, parallel-group, randomised, phase 2 trial. Lancet Oncol. 2020, 21, 571–580. [Google Scholar] [CrossRef]
- Safety and Clinical Activity of Durvalumab Monotherapy in Patients with Hepatocellular Carcinoma (HCC). | Journal of Clinical Oncology. Available online: https://ascopubs.org/doi/abs/10.1200/JCO.2017.35.15_suppl.4071 (accessed on 21 July 2020).
- Tovoli, F.; Ielasi, L.; Casadei-Gardini, A.; Granito, A.; Foschi, F.G.; Rovesti, G.; Negrini, G.; Orsi, G.; Renzulli, M.; Piscaglia, F. Management of adverse events with tailored sorafenib dosing prolongs survival of hepatocellular carcinoma patients. J. Hepatol. 2019, 71, 1175–1183. [Google Scholar] [CrossRef]
- Finn, R.S.; Ryoo, B.-Y.; Merle, P.; Kudo, M.; Bouattour, M.; Lim, H.Y.; Breder, V.; Edeline, J.; Chao, Y.; Ogasawara, S.; et al. Pembrolizumab As Second-Line Therapy in Patients With Advanced Hepatocellular Carcinoma in KEYNOTE-240: A Randomized, Double-Blind, Phase III Trial. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2020, 38, 193–202. [Google Scholar] [CrossRef]
- Tovoli, F.; Lorenzo, S.D.; Barbera, M.A.; Garajova, I.; Frega, G.; Palloni, A.; Pantaleo, M.A.; Biasco, G.; Brandi, G. Postsorafenib systemic treatments for hepatocellular carcinoma: Questions and opportunities after the regorafenib trial. Future Oncol. Lond. Engl. 2017, 13, 1893–1905. [Google Scholar] [CrossRef]
- Qin, S.; Finn, R.S.; Kudo, M.; Meyer, T.; Vogel, A.; Ducreux, M.; Macarulla, T.M.; Tomasello, G.; Boisserie, F.; Hou, J.; et al. RATIONALE 301 study: Tislelizumab versus sorafenib as first-line treatment for unresectable hepatocellular carcinoma. Future Oncol. Lond. Engl. 2019, 15, 1811–1822. [Google Scholar] [CrossRef]
- Wallin, J.J.; Bendell, J.C.; Funke, R.; Sznol, M.; Korski, K.; Jones, S.; Hernandez, G.; Mier, J.; He, X.; Hodi, F.S.; et al. Atezolizumab in combination with bevacizumab enhances antigen-specific T-cell migration in metastatic renal cell carcinoma. Nat. Commun. 2016, 7, 12624. [Google Scholar] [CrossRef]
- Hegde, P.S.; Wallin, J.J.; Mancao, C. Predictive markers of anti-VEGF and emerging role of angiogenesis inhibitors as immunotherapeutics. Semin. Cancer Biol. 2018, 52, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Kudo, M. Combination Cancer Immunotherapy in Hepatocellular Carcinoma. Liver Cancer 2018, 7, 20–27. [Google Scholar] [CrossRef] [PubMed]
- Kudo, M. Combination Cancer Immunotherapy with Molecular Targeted Agents/Anti-CTLA-4 Antibody for Hepatocellular Carcinoma. Liver Cancer 2019, 8, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Finn, R.S.; Ikeda, M.; Zhu, A.X.; Sung, M.W.; Baron, A.D.; Kudo, M.; Okusaka, T.; Kobayashi, M.; Kumada, H.; Kaneko, S.; et al. Phase Ib Study of Lenvatinib Plus Pembrolizumab in Patients With Unresectable Hepatocellular Carcinoma. J. Clin. Oncol. 2020, JCO.20.00808. [Google Scholar] [CrossRef]
- Nivolumab (NIVO) + ipilimumab (IPI) + cabozantinib (CABO) combination therapy in patients (pts) with advanced hepatocellular carcinoma (aHCC): Results from CheckMate 040. J. Clin. Oncol. Available online: https://ascopubs.org/doi/abs/10.1200/JCO.2020.38.4_suppl.478 (accessed on 21 July 2020).
- Sangro, B.; Chan, S.L.; Meyer, T.; Reig, M.; El-Khoueiry, A.; Galle, P.R. Diagnosis and management of toxicities of immune checkpoint inhibitors in hepatocellular carcinoma. J. Hepatol. 2020, 72, 320–341. [Google Scholar] [CrossRef]
- Nivolumab (NIVO) + ipilimumab (IPI) combination therapy in patients (pts) with advanced hepatocellular carcinoma (aHCC): Results from CheckMate 040. J. Clin. Oncol. Available online: https://ascopubs.org/doi/abs/10.1200/JCO.2019.37.15_suppl.4012 (accessed on 21 July 2020).
- Bruix, J.; Takayama, T.; Mazzaferro, V.; Chau, G.-Y.; Yang, J.; Kudo, M.; Cai, J.; Poon, R.T.; Han, K.-H.; Tak, W.Y.; et al. Adjuvant sorafenib for hepatocellular carcinoma after resection or ablation (STORM): A phase 3, randomised, double-blind, placebo-controlled trial. Lancet Oncol. 2015, 16, 1344–1354. [Google Scholar] [CrossRef]
- Goio, E.; Ielasi, L.; Benevento, F.; Renzulli, M.; Tovoli, F. Long-lasting remission in a metastatic hepatocellular carcinoma patient after combined regorafenib therapy and surgery. Hepatic Oncol. 2020, 7, HEP24. [Google Scholar] [CrossRef]
- Duffy, A.G.; Ulahannan, S.V.; Makorova-Rusher, O.; Rahma, O.; Wedemeyer, H.; Pratt, D.; Davis, J.L.; Hughes, M.S.; Heller, T.; ElGindi, M.; et al. Tremelimumab in combination with ablation in patients with advanced hepatocellular carcinoma. J. Hepatol. 2017, 66, 545–551. [Google Scholar] [CrossRef]
- Tovoli, F.; Casadei-Gardini, A.; Benevento, F.; Piscaglia, F. Immunotherapy for hepatocellular carcinoma: A review of potential new drugs based on ongoing clinical studies as of 2019. Dig. Liver Dis. Off. J. Ital. Soc. Gastroenterol. Ital. Assoc. Study Liver 2019, 51, 1067–1073. [Google Scholar] [CrossRef] [PubMed]
- Mole, R.H. Whole body irradiation; radiobiology or medicine? Br. J. Radiol. 1953, 26, 234–241. [Google Scholar] [CrossRef]
- Giannini, E.G.; Aglitti, A.; Borzio, M.; Gambato, M.; Guarino, M.; Iavarone, M.; Lai, Q.; Levi Sandri, G.B.; Melandro, F.; Morisco, F.; et al. Overview of Immune Checkpoint Inhibitors Therapy for Hepatocellular Carcinoma, and The ITA.LI.CA Cohort Derived Estimate of Amenability Rate to Immune Checkpoint Inhibitors in Clinical Practice. Cancers 2019, 11. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.-F.; Chen, Y.; Song, S.-Y.; Wang, T.-J.; Ji, W.-J.; Li, S.-W.; Liu, N.; Yan, C.-X. Immune-Related Adverse Events Associated with Anti-PD-1/PD-L1 Treatment for Malignancies: A Meta-Analysis. Front. Pharm. 2017, 8, 730. [Google Scholar] [CrossRef] [PubMed]
- Bertrand, A.; Kostine, M.; Barnetche, T.; Truchetet, M.-E.; Schaeverbeke, T. Immune related adverse events associated with anti-CTLA-4 antibodies: Systematic review and meta-analysis. BMC Med. 2015, 13, 211. [Google Scholar] [CrossRef]
- De Velasco, G.; Je, Y.; Bossé, D.; Awad, M.M.; Ott, P.A.; Moreira, R.B.; Schutz, F.; Bellmunt, J.; Sonpavde, G.P.; Hodi, F.S.; et al. Comprehensive Meta-analysis of Key Immune-Related Adverse Events from CTLA-4 and PD-1/PD-L1 Inhibitors in Cancer Patients. Cancer Immunol. Res. 2017, 5, 312–318. [Google Scholar] [CrossRef]
- Abdel-Rahman, O.; Fouad, M. Risk of pneumonitis in cancer patients treated with immune checkpoint inhibitors: A meta-analysis. Adv. Respir. Dis. 2016, 10, 183–193. [Google Scholar] [CrossRef]
- Johnson, D.B.; Balko, J.M.; Compton, M.L.; Chalkias, S.; Gorham, J.; Xu, Y.; Hicks, M.; Puzanov, I.; Alexander, M.R.; Bloomer, T.L.; et al. Fulminant Myocarditis with Combination Immune Checkpoint Blockade. N. Engl. J. Med. 2016, 375, 1749–1755. [Google Scholar] [CrossRef]
- Albillos, A.; Lario, M.; Álvarez-Mon, M. Cirrhosis-associated immune dysfunction: Distinctive features and clinical relevance. J. Hepatol. 2014, 61, 1385–1396. [Google Scholar] [CrossRef]
- Chan, S.L.; Yip, T.C.-F.; Wong, V.W.-S.; Tse, Y.-K.; Yuen, B.W.-Y.; Luk, H.W.-S.; Lui, R.N.-S.; Chan, H.L.-Y.; Mok, T.S.-K.; Wong, G.L.-H. Pattern and impact of hepatic adverse events encountered during immune checkpoint inhibitors—A territory-wide cohort study. Cancer Med. 2020, in press. [Google Scholar] [CrossRef]
- Kudo, M.; Matilla, A.; Santoro, A.; Melero, I.; Gracian, A.C.; Acosta-Rivera, M.; Choo, S.P.; El-Khoueiry, A.B.; Kuromatsu, R.; El-Rayes, B.F.; et al. Checkmate-040: Nivolumab (NIVO) in patients (pts) with advanced hepatocellular carcinoma (aHCC) and Child-Pugh B (CPB) status. J. Clin. Oncol. 2019, 37, 327. [Google Scholar] [CrossRef]
- Prieux-Klotz, C.; Dior, M.; Damotte, D.; Dreanic, J.; Brieau, B.; Brezault, C.; Abitbol, V.; Chaussade, S.; Coriat, R. Immune Checkpoint Inhibitor-Induced Colitis: Diagnosis and Management. Target. Oncol. 2017, 12, 301–308. [Google Scholar] [CrossRef] [PubMed]
- Davis, A.A.; Patel, V.G. The role of PD-L1 expression as a predictive biomarker: An analysis of all US Food and Drug Administration (FDA) approvals of immune checkpoint inhibitors. J. Immunother. Cancer 2019, 7, 278. [Google Scholar] [CrossRef] [PubMed]
- Hirsch, F.R.; McElhinny, A.; Stanforth, D.; Ranger-Moore, J.; Jansson, M.; Kulangara, K.; Richardson, W.; Towne, P.; Hanks, D.; Vennapusa, B.; et al. PD-L1 Immunohistochemistry Assays for Lung Cancer: Results from Phase 1 of the Blueprint PD-L1 IHC Assay Comparison Project. J. Thorac. Oncol. Off. Publ. Int. Assoc. Study Lung Cancer 2017, 12, 208–222. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, J.H.; Lelkaitis, G.; Håkansson, K.; Vogelius, I.R.; Johannesen, H.H.; Fischer, B.M.; Bentzen, S.M.; Specht, L.; Kristensen, C.A.; von Buchwald, C.; et al. Intratumor heterogeneity of PD-L1 expression in head and neck squamous cell carcinoma. Br. J. Cancer 2019, 120, 1003–1006. [Google Scholar] [CrossRef]
- Bensch, F.; van der Veen, E.L.; Lub-de Hooge, M.N.; Jorritsma-Smit, A.; Boellaard, R.; Kok, I.C.; Oosting, S.F.; Schröder, C.P.; Hiltermann, T.J.N.; van der Wekken, A.J.; et al. 89Zr-atezolizumab imaging as a non-invasive approach to assess clinical response to PD-L1 blockade in cancer. Nat. Med. 2018, 24, 1852–1858. [Google Scholar] [CrossRef]
- Gandara, D.R.; Paul, S.M.; Kowanetz, M.; Schleifman, E.; Zou, W.; Li, Y.; Rittmeyer, A.; Fehrenbacher, L.; Otto, G.; Malboeuf, C.; et al. Blood-based tumor mutational burden as a predictor of clinical benefit in non-small-cell lung cancer patients treated with atezolizumab. Nat. Med. 2018, 24, 1441–1448. [Google Scholar] [CrossRef]
- Immuno-Oncology | OncologyPRO. Available online: https://oncologypro.esmo.org/education-library/handbooks/immuno-oncology (accessed on 21 July 2020).
- Le, D.T.; Durham, J.N.; Smith, K.N.; Wang, H.; Bartlett, B.R.; Aulakh, L.K.; Lu, S.; Kemberling, H.; Wilt, C.; Luber, B.S.; et al. Mismatch repair deficiency predicts response of solid tumors to PD-1 blockade. Science 2017, 357, 409–413. [Google Scholar] [CrossRef]
- Sinicrope, F.A.; Sargent, D.J. Molecular pathways: Microsatellite instability in colorectal cancer: Prognostic, predictive, and therapeutic implications. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2012, 18, 1506–1512. [Google Scholar] [CrossRef]
- Sarshekeh, A.M.; Overman, M.J.; Kopetz, S. Nivolumab in the treatment of microsatellite instability high metastatic colorectal cancer. Future Oncol. 2018, 14, 1869–1874. [Google Scholar] [CrossRef]
- Sia, D.; Jiao, Y.; Martinez-Quetglas, I.; Kuchuk, O.; Villacorta-Martin, C.; Castro de Moura, M.; Putra, J.; Camprecios, G.; Bassaganyas, L.; Akers, N.; et al. Identification of an Immune-specific Class of Hepatocellular Carcinoma, Based on Molecular Features. Gastroenterology 2017, 153, 812–826. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, Y.; Yamagiwa, Y.; Fukushima, K.; Ueno, Y.; Shimosegawa, T. Expression of galectin-3 involved in prognosis of patients with hepatocellular carcinoma. Hepatol. Res. Off. J. Jpn. Soc. Hepatol. 2008, 38, 1098–1111. [Google Scholar] [CrossRef] [PubMed]
- Yan, W.; Liu, X.; Ma, H.; Zhang, H.; Song, X.; Gao, L.; Liang, X.; Ma, C. Tim-3 fosters HCC development by enhancing TGF-β-mediated alternative activation of macrophages. Gut 2015, 64, 1593–1604. [Google Scholar] [CrossRef]
- Yarchoan, M.; Xing, D.; Luan, L.; Xu, H.; Sharma, R.B.; Popovic, A.; Pawlik, T.M.; Kim, A.K.; Zhu, Q.; Jaffee, E.M.; et al. Characterization of the Immune Microenvironment in Hepatocellular Carcinoma. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2017, 23, 7333–7339. [Google Scholar] [CrossRef] [PubMed]
- Joyce, J.A.; Fearon, D.T. T cell exclusion, immune privilege, and the tumor microenvironment. Science 2015, 348, 74–80. [Google Scholar] [CrossRef] [PubMed]
- Audrito, V.; Managò, A.; Gaudino, F.; Sorci, L.; Messana, V.G.; Raffaelli, N.; Deaglio, S. NAD-Biosynthetic and Consuming Enzymes as Central Players of Metabolic Regulation of Innate and Adaptive Immune Responses in Cancer. Front. Immunol. 2019, 10, 1720. [Google Scholar] [CrossRef]
- Kalluri, R.; Weinberg, R.A. The basics of epithelial-mesenchymal transition. J. Clin. Investig. 2009, 119, 1420–1428. [Google Scholar] [CrossRef]
- Immune Checkpoint Blockade Therapies for HCC: Current Status and Future Implications. Available online: https://hrjournal.net/article/view/3207 (accessed on 15 August 2020).
- Soundararajan, R.; Fradette, J.J.; Konen, J.M.; Moulder, S.; Zhang, X.; Gibbons, D.L.; Varadarajan, N.; Wistuba, I.I.; Tripathy, D.; Bernatchez, C.; et al. Targeting the Interplay between Epithelial-to-Mesenchymal-Transition and the Immune System for Effective Immunotherapy. Cancers 2019, 11. [Google Scholar] [CrossRef]
- Terry, S.; Savagner, P.; Ortiz-Cuaran, S.; Mahjoubi, L.; Saintigny, P.; Thiery, J.-P.; Chouaib, S. New insights into the role of EMT in tumor immune escape. Mol. Oncol. 2017, 11, 824–846. [Google Scholar] [CrossRef]
- Ye, L.-Y.; Chen, W.; Bai, X.-L.; Xu, X.-Y.; Zhang, Q.; Xia, X.-F.; Sun, X.; Li, G.-G.; Hu, Q.-D.; Fu, Q.-H.; et al. Hypoxia-Induced Epithelial-to-Mesenchymal Transition in Hepatocellular Carcinoma Induces an Immunosuppressive Tumor Microenvironment to Promote Metastasis. Cancer Res. 2016, 76, 818–830. [Google Scholar] [CrossRef]
- Massagué, J. TGFβ signalling in context. Nat. Rev. Mol. Cell Biol. 2012, 13, 616–630. [Google Scholar] [CrossRef] [PubMed]
- Nakao, A.; Afrakhte, M.; Morn, A.; Nakayama, T.; Christian, J.L.; Heuchel, R.; Itoh, S.; Kawabata, M.; Heldin, N.-E.; Heldin, C.-H.; et al. Identification of Smad7, a TGFβ-inducible antagonist of TGF-β signalling. Nature 1997, 389, 631–635. [Google Scholar] [CrossRef] [PubMed]
- The Hallmarks of Cancer: Cell. Available online: https://www.cell.com/cell/fulltext/S0092-8674(00)81683-9?_returnURL=https%3A%2F%2Flinkinghub.elsevier.com%2Fretrieve%2Fpii%2FS0092867400816839%3Fshowall%3Dtrue (accessed on 21 September 2020).
- Blobe, G.C.; Schiemann, W.P.; Lodish, H.F. Role of Transforming Growth Factor β in Human Disease. Available online: https://www.nejm.org/doi/10.1056/NEJM200005043421807 (accessed on 21 September 2020).
- Eisenstein, E.M.; Williams, C.B. The T reg /Th17 Cell Balance: A New Paradigm for Autoimmunity. Pediatr. Res. 2009, 65, 26–31. [Google Scholar] [CrossRef] [PubMed]
- Roes, J.; Choi, B.K.; Cazac, B.B. Redirection of B cell responsiveness by transforming growth factor β receptor. Proc. Natl. Acad. Sci. USA 2003, 100, 7241–7246. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.M.; Chen, D.S. Immune escape to PD-L1/PD-1 blockade: Seven steps to success (or failure). Ann. Oncol. Off. J. Eur. Soc. Med. Oncol. 2016, 27, 1492–1504. [Google Scholar] [CrossRef]
- Kelley, R.K. Atezolizumab plus Bevacizumab—A Landmark in Liver Cancer. N. Engl. J. Med. 2020, 382, 1953–1955. [Google Scholar] [CrossRef]
- Sharma, P.; Hu-Lieskovan, S.; Wargo, J.A.; Ribas, A. Primary, Adaptive and Acquired Resistance to Cancer Immunotherapy. Cell 2017, 168, 707–723. [Google Scholar] [CrossRef]
- Eisenhauer, E.A.; Therasse, P.; Bogaerts, J.; Schwartz, L.H.; Sargent, D.; Ford, R.; Dancey, J.; Arbuck, S.; Gwyther, S.; Mooney, M.; et al. New response evaluation criteria in solid tumours: Revised RECIST guideline (version 1.1). Eur. J. Cancer Oxf. Engl. 1990 2009, 45, 228–247. [Google Scholar] [CrossRef]
- Lencioni, R.; Llovet, J.M. Modified RECIST (mRECIST) assessment for hepatocellular carcinoma. Semin. Liver Dis. 2010, 30, 52–60. [Google Scholar] [CrossRef]
- Lencioni, R.; Montal, R.; Torres, F.; Park, J.-W.; Decaens, T.; Raoul, J.-L.; Kudo, M.; Chang, C.; Ríos, J.; Boige, V.; et al. Objective response by mRECIST as a predictor and potential surrogate end-point of overall survival in advanced HCC. J. Hepatol. 2017, 66, 1166–1172. [Google Scholar] [CrossRef]
- Bruix, J.; Reig, M.; Sangro, B. Assessment of treatment efficacy in hepatocellular carcinoma: Response rate, delay in progression or none of them. J. Hepatol. 2017, 66, 1114–1117. [Google Scholar] [CrossRef] [PubMed]
- Wolchok, J.D.; Rollin, L.; Larkin, J. Nivolumab and Ipilimumab in Advanced Melanoma. N. Engl. J. Med. 2017, 377, 2503–2504. [Google Scholar] [CrossRef] [PubMed]
- Seymour, L.; Bogaerts, J.; Perrone, A.; Ford, R.; Schwartz, L.H.; Mandrekar, S.; Lin, N.U.; Litière, S.; Dancey, J.; Chen, A.; et al. iRECIST: Guidelines for response criteria for use in trials testing immunotherapeutics. Lancet Oncol. 2017, 18, e143–e152. [Google Scholar] [CrossRef]
| NCT | Phase | Study Drugs | Line | Primary Endpoint | Estimated End of Trial |
|---|---|---|---|---|---|
| NCT03794440 | 2–3 | SINTILIMAB + BEVACIZUMAB BIOSIMILAR | 1 | OS, ORR | December 2022 |
| NCT03970616 | 1B/2 | DURVALUMAB + TIVOZANIB | 1 | SAFETY | August 2022 |
| NCT03973112 | 2 | HLX-10+BEVACIZUMAB BIOSIMILAR | 1 | ORR | June 2022 |
| NCT04393220 | 2 | NIVOLUMAB + BEVACIZUMAB | 1 | OS, PFS | October 2021 |
| NCT04072679 | 1 | SINTILIMAB + BEVACIZUMAB BIOSIMILAR | 1 | SAFETY | November 2021 |
| NCT | Phase | Study Drugs | Line | Primary Endpoint | Estimated End of Trial |
|---|---|---|---|---|---|
| NCT04194775 | 3 | CS1003 + LENVATINIB vs. LENVATINIB | 1 | OS, PFS | June 2023 |
| NCT04344158 | 3 | PENPULIMAB + ANLOTINIB vs. SORAFENIB | 1 | OS | December 2024 |
| NCT03755791 | 3 | ATEZOLIZUMAB + CABOZANTINIB vs. SORAFENIB | 1 | OS, PFS | December 2021 |
| NCT03713593 | 3 | PEMBROLIZUMAB + LENVATINIB vs. LENVATINIB | 1 | OS, PFS | May 2022 |
| NCT04411706 | 2 | SINTILIMAB + APATINIB + CAPECITABINE | 1 | ORR | June 2022 |
| NCT04042805 | 2 | SINTILIMAB + LENVATINIB | 1 | ORR | August 2024 |
| NCT04444167 | 2 | BISPECIFIC AK104 + LENVATINIB | 1 | ORR | March 2022 |
| NCT04172571 | 2 | PENPULIMAB + ANLOTINIB | 1 | ORR | June 2021 |
| NCT04183088 | 2 | TISLELIZUMAB + REGORAFENIB | 1 | ORR, PFS, SAFETY | March 2025 |
| NCT04052152 | 2 | SINTILIMAB + ANLOTINIB | 1 | ORR, SAFETY | December 2021 |
| NCT03841201 | 2 | NIVOLUMAB + LENVATINIB | 1 | ORR, SAFETY | October 2021 |
| NCT04310709 | 2 | NIVOLUMAB + REGORAFENIB | 1 | ORR | May 2023 |
| NCT04442581 | 2 | PEMBROLIZUMAB + CABOZANTINIB | 1 | ORR | September 2024 |
| NCT04069949 | 2 | TORIPALIMAB + SORAFENIB | 1 | 6M-PFS, SAFETY | October 2021 |
| NCT03439891 | 2 | NIVOLUMAB + SORAFENIB | 1 | MTD, ORR | May 2022 |
| NCT04170556 | 2 | NIVOLUMAB + REGORAFENIB | 2 | SAFETY | December 2022 |
| NCT04014101 | 2 | CAMRELIZUMAB + APATINIB | 2 | ORR | October 2021 |
| NCT04170179 | 2 | TORIPALIMAB + LENVATINIB + CHEMOTHERAPY | 1 | 6M-PFS | December 2020 |
| NCT04401800 | 1b/2 | TISLELIZUMAB + LENVATINIB | 1 | ORR | December 2022 |
| NCT04443309 | 1b/2 | CAMRELIZUMAB + LENVATINIB | 1 | ORR | August 2024 |
| NCT03347292 | 1 | PEMBROLIZUMAB + REGORAFENIB | 1 | DLT, SAFETY | October 2022 |
| NCT | Phase | Study Drugs | Line | Primary Endpoint | Estimated End of Trial |
|---|---|---|---|---|---|
| NCT03605706 | 3 | CAMRELIZUMAB + FOLFOX4 vs. SORAFENIB OR FOLFOX4 | 1 | OS | December 2021 |
| NCT03519997 | 2 | PEMBROLIZUMAB + BAVITUXIMAB (phosphatidylserine) | 1 | ORR | April 2022 |
| NCT04050462 | 2 | NIVOLUMAB vs. NIVOLUMAB/BMS-986253 (anti-IL8) vs. NIVOLUMAB/Cabiralizumab (anti-CSF1R) | 1 | ORR | August 2020 |
| NCT03695250 | 1–2 | NIVOLUMAB + BMS-986205 (IDO1 inhibitor) | 1–2 | SAFETY, ORR | June 2022 |
| NCT03893695 | 1–2 | NIVOLUMAB + Ascrinvacumab (activin receptor-like kinase 1) | 2 | DLT | September 2020 |
| NCT03419481 | 1–2 | NIVOLUMAB + ABX196 (invariant Natural Killer T cell agonist) | 2 | SAFETY | June 2021 |
| NCT03655613 | 1–2 | NIVOLUMAB + APL-101 (cMET inhibitor) | 2 | DLT | December 2020 |
| NCT02423343 | 1–2 | Nivolumab + galunisertib | 2 | MTD, SAFETY | July 2020 |
| NCT03241173 | 1–2 | Nivolumab + ipilimumab + INCAGN01949 | 2 | SAFETY, ORR | November 2021 |
| NCT02178722 | 1–2 | Pembrolizumab + epacadostat | 2–3 | DLT, ORR | August 2020 |
| NCT02795429 | 1–2 | Spartalizumab (+capmatinib) | 1 | DLT, ORR | October 2020 |
| NCT03095781 | 1 | Pembrolizumab + XL888 | 2 | RP2D | June 2023 |
| NCT | Setting | Study Drug(s) | Phase | Primary Endpoint | Estimated End of Trial |
|---|---|---|---|---|---|
| NCT03383458 | ADJ | NIVOLUMAB vs. PLACEBO | 3 | RFS | 1 June 2025 |
| NCT03867084 | ADJ | PEMBROLIZUMAB vs. PLACEBO | 3 | RFS, OS | 1 June 2025 |
| NCT03847428 | ADJ | DURVALUMAB + BEVACIZUMAB vs. PLACEBO | 3 | RFS | 1 September 2023 |
| NCT04102098 | ADJ | ATEZOLIZUMAB + BEVACIZUMAB vs. PLACEBO | 3 | RFS | 1 July 2027 |
| NCT03859128 | ADJ | TORIPALIMAB vs. PLACEBO | 2–3 | RFS | 1 April 2024 |
| NCT03337841 | ADJ | PEMBROLIZUMAB | 2 | 1Y-RFS | 1 October 2020 |
| NCT03839550 | ADJ | CAMRELIZUMAB + APATINIB | 2 | RFS | 1 February 2023 |
| NCT04418401 | ADJ | ANTI-PD1 + DONAFINIB | 2 | 1Y-RFS | 1 June 2023 |
| NCT03630640 | ADJ, NADJ | NIVOLUMAB * | 2 | RFS | 1 September 2020 |
| NCT03510871 | NADJ | NIVOLUMAB + IPILIMUMAB | 2 | ORR, DOWNSTAGING RATE | 1 December 2022 |
| NCT04297202 | NADJ | CAMRELIZUMAB + APATINIB | 2 | ORR (10%) | 1 December 2021 |
| NCT04297202 | NADJ | CAMRELIZUMAB + APATINIB | 2 | ORR (10%) | 1 December 2021 |
| NCT04123379 | NADJ | NIVOLUMAB + CCR2/5-inhibitor vs. NIVOLUMAB + ANTI-IL8 | 2 | SAFETY | 1 October 2024 |
| NCT03222076 | NADJ | NIVOLUMAB | 2 | SAFETY | 1 September 2022 |
| NCT03682276 | NADJ | NIVOLUMAB + IPILIMUMAB | 1–2 | DELAY TO SURGERY, SAFETY | 1 September 2022 |
| NCT04035876 | NADJ | CAMRELIZUMAB + APATINIB ** | 1–2 | RFS | 1 December 2021 |
| NCT03722875 | ADJ | CAMRELIZUMAB | 1 | RFS | 1 March 2020 |
| NCT03383458 | ADJ | NIVOLUMAB vs. PLACEBO | 1 | RFS | 1 June 2025 |
| NCT03914352 | ADJ | CAMRELIZUMAB *** | n/a | OS, RFS | 1 January 2020 |
| NCT04425226 | NADJ | PEMBROLIZUMAB + LENVATINIB vs. BSC | n/a | RFS, ORR | 1 December 2025 |
| NCT | Phase | Study Drug(s) | Primary Endpoint | Estimated End of Trial |
|---|---|---|---|---|
| NCT03778957 | 3 | TACE + DURVALUMAB + BEVACIZUMAB | PFS | March 2024 |
| NCT04246177 | 3 | TACE + PEMBROLIZUMAB + LENVATINIB | OS, PFS | December 2029 |
| NCT04229355 | 3 | TACE + (SORAFENIB vs. LENVATINIB vs SINTILIMAB) | PFS | December 2022 |
| NCT04268888 | 2–3 | TACE + NIVOLUMAB | OS, TTP | June 2026 |
| NCT03753659 | 2 | RFA/MWA/brachytherapy + PEMBROLIZUMAB | ORR | September 2023 |
| NCT04297280 | 2 | SINTILIMAB + TACE | ORR | May 2023 |
| NCT03857815 | 2 | EBRT + SINTILIMAB | PFS | February 2022 |
| NCT03851939 | 2 | HAIC + TORIPALIMAB | PFS, ORR | March 2021 |
| NCT03033446 | 2 | SIRT + NIVOLUMAB | ORR | December 2019 |
| NCT03482102 | 2 | EBRT + DURVALUMAB + TREMELIMUMAB | ORR | October 2025 |
| NCT03869034 | 2 | HAIC vs. HAIC + PD-1 | PFS | March 2022 |
| NCT03572582 | 2 | TACE + NIVOLUMAB | ORR | September 2022 |
| NCT03937830 | 2 | TACE + DURVALUMAB + BEVACIZUMAB | 6-M PFS | December 2022 |
| NCT04224636 | 2 | TACE + ATEZOLIZUMAB + BEVACIZUMAB | 2Y-OS | March 2025 |
| NCT02821754 | 2 | ABLATIVE PROCEDURE + DURVALUMAB + TREMELIMUMAB | PFS | December 2021 |
| NCT04204577 | 2 | TACE + CAMRELIZUMAB + APATINIB | PFS | November 2023 |
| NCT04220944 | 2 | RFA/TACE + SINTILIMAB | PFS | July 2021 |
| NCT04430452 | 2 | EBRT + DURVALUMAB + TREMELIMUMAB | ORR | August 2024 |
| NCT04191889 | 2 | TAI + CAMRELIZUMAB + APATINIB | ORR, SAFETY | December 2025 |
| NCT04044313 | 2 | HAIC + TORIPALIMAB | PFS | August 2020 |
| NCT04135690 | 2 | HAIC + TORIPALIMAB vs. HAIC + SORAFENIB | PFS | January 2020 |
| NCT04273100 | 2 | TACE + LENVATINIB- + PD-1 | ORR | June 2021 |
| NCT04150744 | 2 | RFA + CARRIZUMAB + APATINIB vs CARRIZUMAB + APATINIB | PFS | December 2026 |
| NCT04167293 | 2 | EBRT + SINTILIMAB | 2-Y PFS | October 2025 |
| NCT03316872 | 2 | EBRT + PEMBROLIZUMAB | ORR | February 2022 |
| NCT04124991 | 1–2 | SIRT + DURVALUMAB | TTP | December 2021 |
| NCT03397654 | 1B | TACE + PEMBROLIZUMAB | SAFETY | December 2020 |
| NCT04104074 | 1 | EBRT + SINTILIMAB | SAFETY | December 2020 |
| NCT02837029 | 1 | SIRT + NIVOLUMAB | MTD | July 2023 |
| Parameter/Endpoint | Nivolumab | Pembrolizumab | Atezolizumab + Bevacizumab | Pembrolizumab + Lenvatinib | Nivolumab + Ipilimumab |
|---|---|---|---|---|---|
| Median OS (months) | 16.4 | 13.9 | >17.0 | 22.0 | 12.2–22.5 |
| Median PFS (months) | 3.7 | 3.0 | 6.8 | 8.6 | not reported |
| ORR | 15% | 18.3% | 27.3% | 36.6% | 31–32% |
| G ≥3 AEs | 22% | 46.3% | 56.5% | 67% * | 37% |
| Discontinuation rate for AEs | 4% | 17.2% | 15.5% | not reported | 2–18% |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tovoli, F.; De Lorenzo, S.; Trevisani, F. Immunotherapy with Checkpoint Inhibitors for Hepatocellular Carcinoma: Where Are We Now? Vaccines 2020, 8, 578. https://doi.org/10.3390/vaccines8040578
Tovoli F, De Lorenzo S, Trevisani F. Immunotherapy with Checkpoint Inhibitors for Hepatocellular Carcinoma: Where Are We Now? Vaccines. 2020; 8(4):578. https://doi.org/10.3390/vaccines8040578
Chicago/Turabian StyleTovoli, Francesco, Stefania De Lorenzo, and Franco Trevisani. 2020. "Immunotherapy with Checkpoint Inhibitors for Hepatocellular Carcinoma: Where Are We Now?" Vaccines 8, no. 4: 578. https://doi.org/10.3390/vaccines8040578
APA StyleTovoli, F., De Lorenzo, S., & Trevisani, F. (2020). Immunotherapy with Checkpoint Inhibitors for Hepatocellular Carcinoma: Where Are We Now? Vaccines, 8(4), 578. https://doi.org/10.3390/vaccines8040578






