Electroporation as the Immunotherapy Strategy for Cancer in Veterinary Medicine: State of the Art in Latin America
Abstract
1. Introduction
2. Irreversible Electroporation
3. Reversible Electroporation
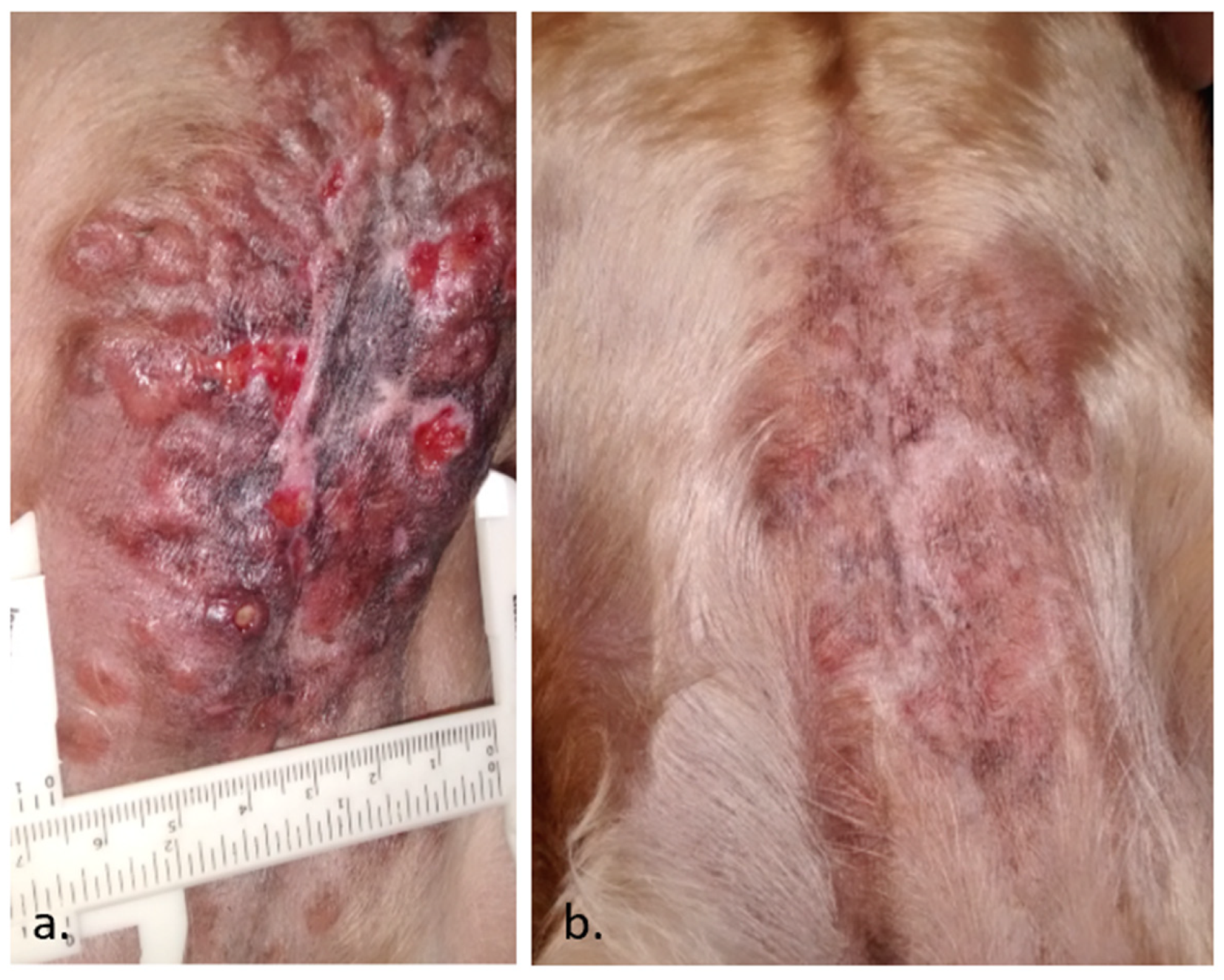
3.1. Electrochemotherapy
3.2. Gene Electrotransfer
4. ECT and GET Immunomodulatory Effects: A Promising Combination Strategy
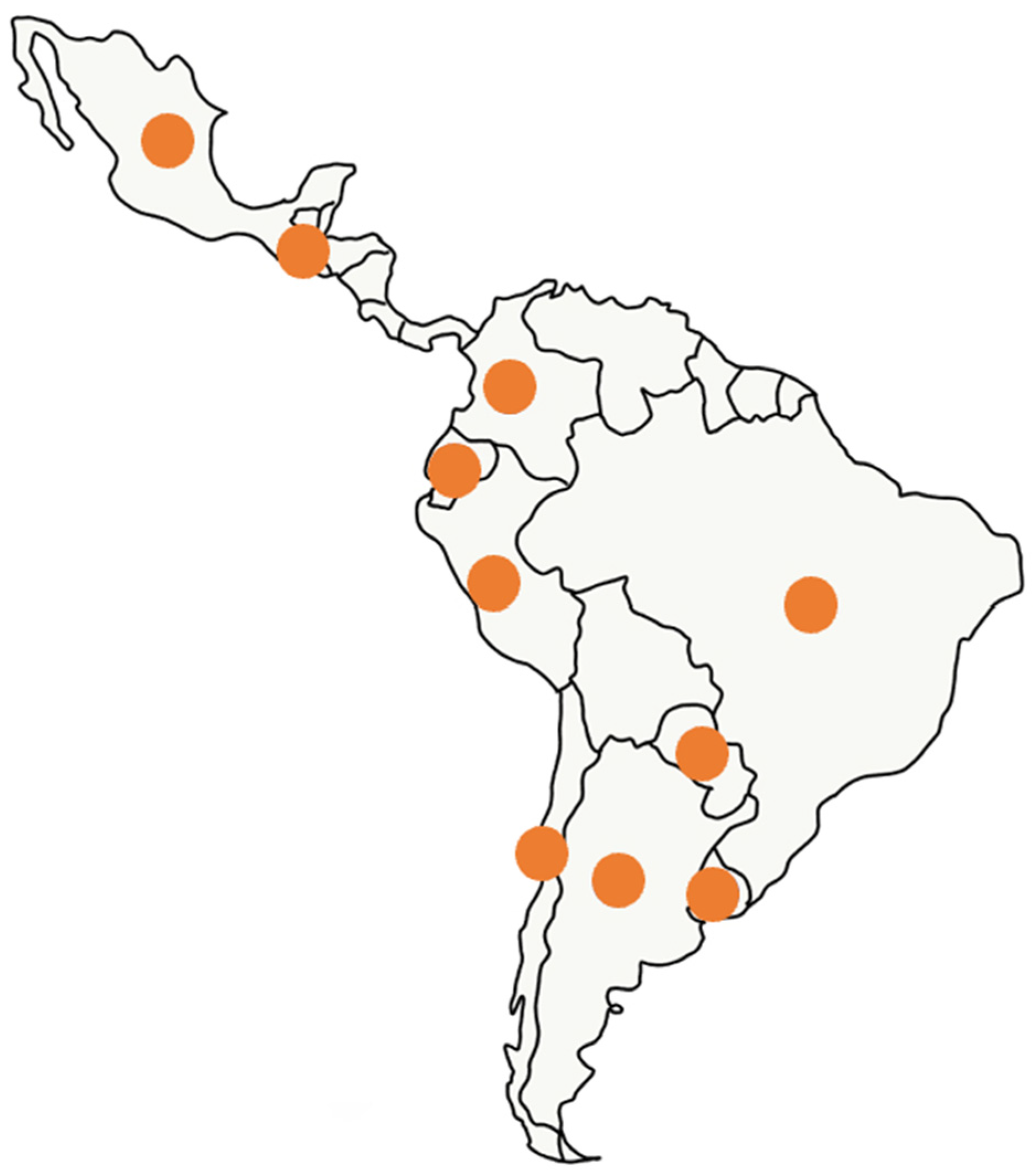
5. State of the Art of Electroporation-Based Treatments in Latin America
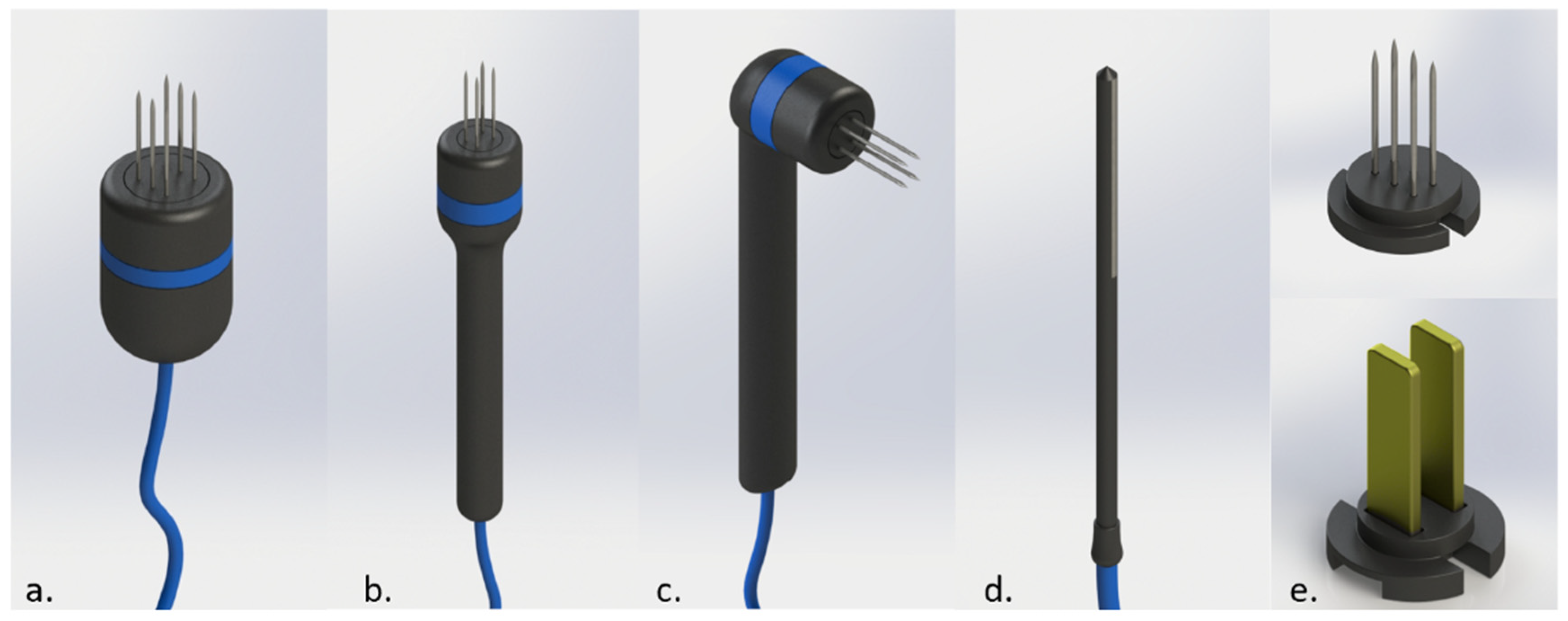
6. The Electroporators Available for Veterinary Medicine in Latin America
7. Final Considerations and Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| BLM | Bleomycin |
| CDDP | Cisplatin |
| ECT | Electrochemotherapy |
| GET | Gene Electrotransfer |
| HV+LV | High-Voltage followed by Low-Voltage pulses |
| IL | Interleukin |
| INF | Interferon |
| IRE | Irreversible Electroporation |
| NK | Natural Killer |
| TGF | Transforming Growth Factor |
| TNF | Tumor Necrosis Factor |
| Tregs | T regulatory cells |
References
- Albert, A.; Bulcroft, K. Pets and Urban Life. Anthrozoös 1987, 1, 9–25. [Google Scholar] [CrossRef]
- Mir, L.M.; Orlowski, S.; Belehradek, J., Jr.; Paoletti, C. Electrochemotherapy potentiation of antitumour effect of bleomycin by local electric pulses. Eur. J. Cancer 1991, 27, 68–72. [Google Scholar] [CrossRef]
- Mir, L.M.; Belehradek, M.; Domenge, C.; Orlowski, S.; Poddevin, B.; Belehradek, J., Jr.; Schwaab, G.; Luboinski, B.; Paoletti, C. Electrochemotherapy, a new antitumor treatment: First clinical trial. C. R. Acad. Sci. III 1991, 313, 613–618. [Google Scholar] [PubMed]
- Neumann, E.; Schaefer-Ridder, M.; Wang, Y.; Hofschneider, P.H. Gene transfer into mouse lyoma cells by electroporation in high electric fields. EMBO J. 1982, 1, 841–845. [Google Scholar] [CrossRef] [PubMed]
- Frandsen, S.K.; Vissing, M.; Gehl, J. A Comprehensive Review of Calcium Electroporation—A Novel Cancer Treatment Modality. Cancers 2020, 12, 290. [Google Scholar] [CrossRef] [PubMed]
- Kotnik, T.; Kramar, P.; Pucihar, G.; Miklavcic, D.; Tarek, M. Cell membrane electroporation—Part 1: The phenomenon. IEEE Electr. Insul. Mag. 2012, 28, 14–23. [Google Scholar] [CrossRef]
- Mir, L.M. Bases and rationale of the electrochemotherapy. In Proceedings of the 11th Mediterranean Conference on Medical and Biomedical Engineering and Computing, Ljubljana, Slovenia, 26–30 June 2007; p. 622. [Google Scholar]
- Rubinsky, B. Irreversible Electroporation; Springer: Berlin/Heidelberg, Germany, 2009; ISBN 9783642054204. [Google Scholar]
- Miklavcic, D.; Corovic, S.; Pucihar, G.; Pavselj, N. Importance of tumour coverage by sufficiently high local electric field for effective electrochemotherapy. Eur. J. Cancer Suppl. 2006, 4, 45–51. [Google Scholar] [CrossRef]
- Maglietti, F.; Tellado, M.; Olaiz, N.; Michinski, S.; Marshall, G. Combined local and systemic bleomycin administration in electrochemotherapy to reduce the number of treatment sessions. Radiol. Oncol. 2016, 50, 58–63. [Google Scholar] [CrossRef]
- Neal, R.E.; Rossmeisl, J.H., Jr.; Robertson, J.L.; Arena, C.B.; Davis, E.M.; Singh, R.N.; Stallings, J.; Davalos, R.V. Improved local and systemic anti-tumor efficacy for irreversible electroporation in immunocompetent versus immunodeficient mice. PLoS ONE 2013, 8, e64559. [Google Scholar] [CrossRef]
- Mir, L.M.; Orlowski, S. Mechanisms of electrochemotherapy. Adv. Drug Deliv. Rev. 1999, 35, 107–118. [Google Scholar] [CrossRef]
- Meijerink, M.R.; Scheffer, H.J.; Narayanan, G. Irreversible Electroporation in Clinical Practice; Springer: Berlin/Heidelberg, Germany, 2017; ISBN 9783319551135. [Google Scholar]
- Scheltema, M.; de la Rosette, J. Irreversible Electroporation of Prostate Tumors. In Irreversible Electroporation in Clinical Practice; Springer: Cham, Switzerland, 2018; pp. 215–222. [Google Scholar]
- Koukounaras, J.; Kavnoudias, H.; Thomson, K.R. Irreversible Electroporation of Kidney Tumours. In Irreversible Electroporation in Clinical Practice; Springer: Cham, Switzerland, 2018; pp. 201–213. [Google Scholar]
- Kodama, H.; Srimathveeravalli, G.; Solomon, S.B. Irreversible Electroporation of Lung Tumors. In Irreversible Electroporation in Clinical Practice; Springer: Cham, Switzerland, 2018; pp. 239–245. [Google Scholar]
- Wiggermann, P.; Brünn, K.; Bäumler, W. Irreversible electroporation (IRE): A minimally invasive therapeutic option in prostate cancer. Radiologe 2017, 57, 637–640. [Google Scholar] [CrossRef] [PubMed]
- Niessen, C.; Thumann, S.; Beyer, L.; Pregler, B.; Kramer, J.; Lang, S.; Teufel, A.; Jung, E.M.; Stroszczynski, C.; Wiggermann, P. Percutaneous Irreversible Electroporation: Long-term survival analysis of 71 patients with inoperable malignant hepatic tumors. Sci. Rep. 2017, 7, 43687. [Google Scholar] [CrossRef] [PubMed]
- Chan, G.; Pua, U. Irreversible Electroporation of the Pancreas. Semin. Intervent. Radiol. 2019, 36, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.-Y.; Shi, J.-J.; Chen, J.-B.; Xu, K.-C.; Niu, L.-Z. Irreversible electroporation for liver metastasis from pancreatic cancer: A case report. World J. Clin. Cases 2020, 8, 390–397. [Google Scholar] [CrossRef]
- Van Es, R.; Konings, M.K.; Du Pré, B.C.; Neven, K.; van Wessel, H.; van Driel, V.J.H.M.; Westra, A.H.; Doevendans, P.A.F.; Wittkampf, F.H.M. High-frequency irreversible electroporation for cardiac ablation using an asymmetrical waveform. Biomed. Eng. Online 2019, 18, 75. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Wen, X.; Tian, L.; Li, T.; Xu, C.; Wen, X.; Melancon, M.P.; Gupta, S.; Shen, B.; Peng, W.; et al. Irreversible electroporation reverses resistance to immune checkpoint blockade in pancreatic cancer. Nat. Commun. 2019, 10, 899. [Google Scholar] [CrossRef] [PubMed]
- Mir, L.M.; Orlowski, S. The Basis of Electrochemotherapy. In Electrochemotherapy, Electrogenetherapy, and Transdermal Drug Delivery; Humana Press: Totowa, NJ, USA, 2000; pp. 99–117. [Google Scholar]
- Cemazar, M.; Tamzali, Y.; Sersa, G.; Tozon, N.; Mir, L.M.; Miklavcic, D.; Lowe, R.; Teissie, J. Electrochemotherapy in veterinary oncology. J. Vet. Intern. Med. 2008, 22, 826–831. [Google Scholar] [CrossRef]
- Gehl, J.; Sersa, G.; Matthiessen, L.W.; Muir, T.; Soden, D.; Occhini, A.; Quaglino, P.; Curatolo, P.; Campana, L.G.; Kunte, C.; et al. Updated standard operating procedures for electrochemotherapy of cutaneous tumours and skin metastases. Acta Oncol. 2018, 57, 874–882. [Google Scholar] [CrossRef]
- Miklavčič, D.; Mali, B.; Kos, B.; Heller, R.; Serša, G. Electrochemotherapy: From the drawing board into medical practice. Biomed. Eng. Online 2014, 13, 29. [Google Scholar] [CrossRef]
- Spugnini, E.P.; Vincenzi, B.; Citro, G.; Dotsinsky, I.; Mudrov, T.; Baldi, A. Evaluation of Cisplatin as an electrochemotherapy agent for the treatment of incompletely excised mast cell tumors in dogs. J. Vet. Intern. Med. 2011, 25, 407–411. [Google Scholar] [CrossRef]
- Tounekti, O.; Pron, G.; Belehradek, J., Jr.; Mir, L.M. Bleomycin, an apoptosis-mimetic drug that induces two types of cell death depending on the number of molecules internalized. Cancer Res. 1993, 53, 5462–5469. [Google Scholar] [PubMed]
- Chen, J.; Ghorai, M.K.; Kenney, G.; Stubbe, J. Mechanistic studies on bleomycin-mediated DNA damage: Multiple binding modes can result in double-stranded DNA cleavage. Nucleic Acids Res. 2008, 36, 3781–3790. [Google Scholar] [CrossRef] [PubMed]
- Cepeda, V.; Fuertes, M.A.; Castilla, J.; Alonso, C.; Quevedo, C.; Pérez, J.M. Biochemical mechanisms of cisplatin cytotoxicity. Anti-Cancer Agents Med. Chem. 2007, 7, 3–18. [Google Scholar] [CrossRef] [PubMed]
- Jarm, T.; Cemazar, M.; Miklavcic, D.; Sersa, G. Antivascular effects of electrochemotherapy: Implications in treatment of bleeding metastases. Expert Rev. Anti-Cancer Ther. 2010, 10, 729–746. [Google Scholar] [CrossRef]
- Sersa, G.; Jarm, T.; Kotnik, T.; Coer, A.; Podkrajsek, M.; Sentjurc, M.; Miklavcic, D.; Kadivec, M.; Kranjc, S.; Secerov, A.; et al. Vascular disrupting action of electroporation and electrochemotherapy with bleomycin in murine sarcoma. Br. J. Cancer 2008, 98, 388–398. [Google Scholar] [CrossRef]
- Calvet, C.Y.; Mir, L.M. The promising alliance of anti-cancer electrochemotherapy with immunotherapy. Cancer Metastasis Rev. 2016, 35, 165–177. [Google Scholar] [CrossRef]
- Calvet, C.Y.; Famin, D.; André, F.M.; Mir, L.M. Electrochemotherapy with bleomycin induces hallmarks of immunogenic cell death in murine colon cancer cells. Oncoimmunology 2014, 3, e28131. [Google Scholar] [CrossRef]
- Sersa, G.; Teissie, J.; Cemazar, M.; Signori, E.; Kamensek, U.; Marshall, G.; Miklavcic, D. Electrochemotherapy of tumors as in situ vaccination boosted by immunogene electrotransfer. Cancer Immunol. Immunother. 2015, 64, 1315–1327. [Google Scholar] [CrossRef]
- Dunn, G.P.; Old, L.J.; Schreiber, R.D. The immunobiology of cancer immunosurveillance and immunoediting. Immunity 2004, 21, 137–148. [Google Scholar] [CrossRef]
- Bugaut, H.; Bruchard, M.; Berger, H.; Derangère, V.; Odoul, L.; Euvrard, R.; Ladoire, S.; Chalmin, F.; Végran, F.; Rébé, C.; et al. Bleomycin exerts ambivalent antitumor immune effect by triggering both immunogenic cell death and proliferation of regulatory T cells. PLoS ONE 2013, 8, e65181. [Google Scholar] [CrossRef]
- National Institute for Health and Care Excellence. Electrochemotherapy for Metastases in the Skin from Tumours of Non-Skin Origin and Melanoma; NICE: London, UK, 2013. [Google Scholar]
- Jamsek, C.; Sersa, G.; Bosnjak, M.; Groselj, A. Long term response of electrochemotherapy with reduced dose of bleomycin in elderly patients with head and neck non-melanoma skin cancer. Radiol. Oncol. 2020, 54, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Spugnini, E.P.; Baldi, A. Electrochemotherapy in Veterinary Oncology: State-of-the-Art and Perspectives. Vet. Clin. N. Am. Small Anim. Pract. 2019, 49, 967–979. [Google Scholar] [CrossRef]
- Spugnini, E.P.; Vincenzi, B.; Amadio, B.; Baldi, A. Adjuvant electrochemotherapy with bleomycin and cisplatin combination for canine soft tissue sarcomas: A study of 30 cases. Open Vet. J. 2019, 9, 88–93. [Google Scholar] [CrossRef] [PubMed]
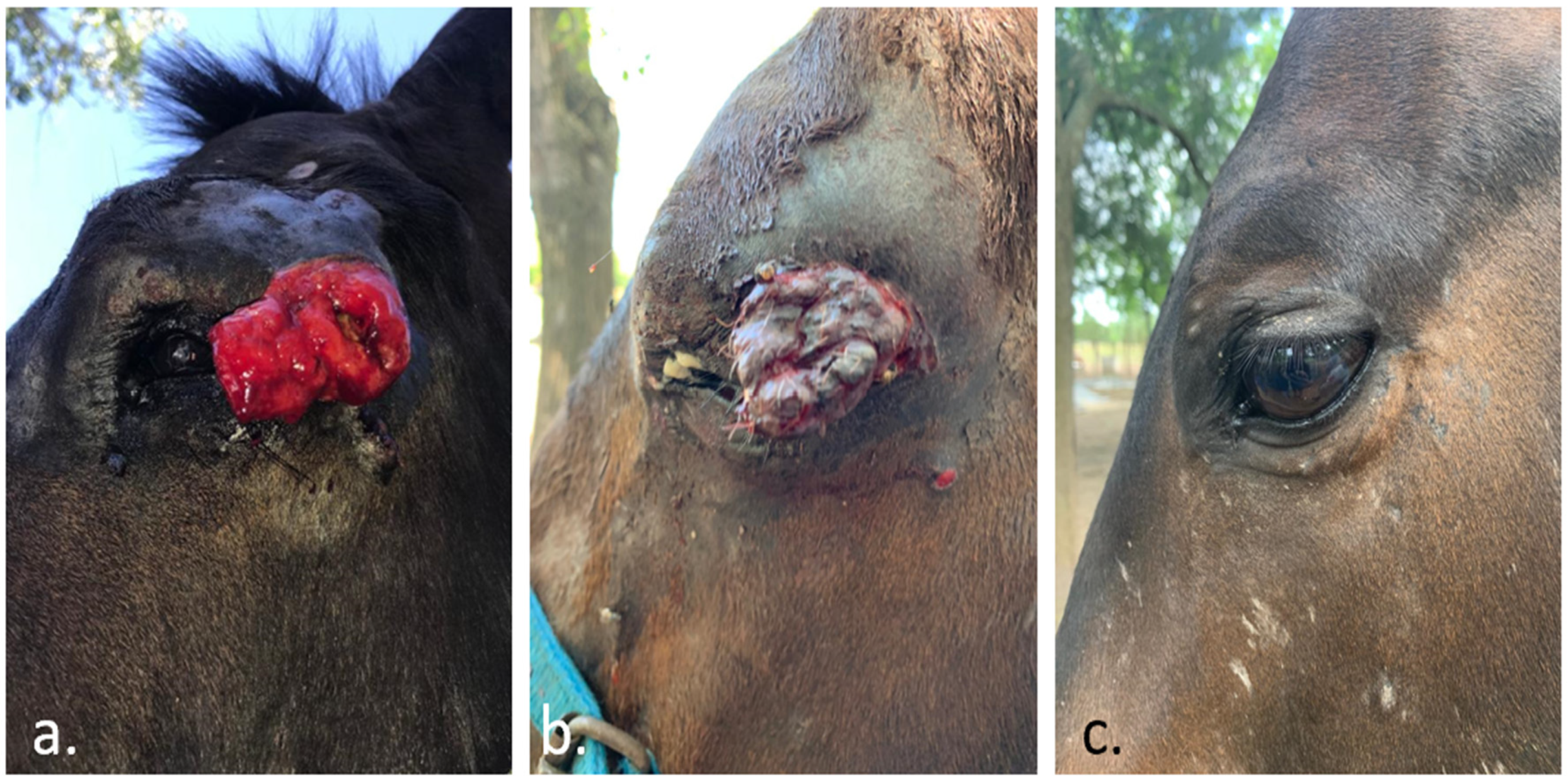
- Spugnini, E.P.; Pizzuto, M.; Filipponi, M.; Romani, L.; Vincenzi, B.; Menicagli, F.; Lanza, A.; De Girolamo, R.; Lomonaco, R.; Fanciulli, M.; et al. Electroporation Enhances Bleomycin Efficacy in Cats with Periocular Carcinoma and Advanced Squamous Cell Carcinoma of the Head. J. Vet. Intern. Med. 2015, 29, 1368–1375. [Google Scholar] [CrossRef] [PubMed]
- Edhemovic, I.; Brecelj, E.; Gasljevic, G.; Marolt Music, M.; Gorjup, V.; Mali, B.; Jarm, T.; Kos, B.; Pavliha, D.; Grcar Kuzmanov, B.; et al. Intraoperative electrochemotherapy of colorectal liver metastases. J. Surg. Oncol. 2014, 110, 320–327. [Google Scholar] [CrossRef] [PubMed]
- Casadei, R.; Ricci, C.; Ingaldi, C.; Alberici, L.; Di Marco, M.; Guido, A.; Minni, F.; Serra, C. Intraoperative electrochemotherapy in locally advanced pancreatic cancer: Indications, techniques and results-a single-center experience. Updates Surg. 2020. [Google Scholar] [CrossRef]
- Impellizeri, J.; Aurisicchio, L.; Forde, P.; Soden, D.M. Electroporation in veterinary oncology. Vet. J. 2016, 217, 18–25. [Google Scholar] [CrossRef]
- Cemazar, M.; Sersa, G. Recent Advances in Electrochemotherapy. Bioelectricity 2019, 1, 204–213. [Google Scholar] [CrossRef]
- REVEAL 2 Trial (Evaluation of VGX-3100 and Electroporation for the Treatment of Cervical HSIL)—Full Text View—ClinicalTrials.gov. Available online: https://clinicaltrials.gov/ct2/show/NCT03721978 (accessed on 4 June 2020).
- André, F.; Mir, L.M. DNA electrotransfer: Its principles and an updated review of its therapeutic applications. Gene Ther. 2004, 11, S33–S42. [Google Scholar] [CrossRef]
- Satkauskas, S.; Bureau, M.F.; Puc, M.; Mahfoudi, A.; Scherman, D.; Miklavcic, D.; Mir, L.M. Mechanisms of in vivo DNA electrotransfer: Respective contributions of cell electropermeabilization and DNA electrophoresis. Mol. Ther. 2002, 5, 133–140. [Google Scholar] [CrossRef]
- André, F.M.; Gehl, J.; Sersa, G.; Préat, V.; Hojman, P.; Eriksen, J.; Golzio, M.; Cemazar, M.; Pavselj, N.; Rols, M.-P.; et al. Efficiency of high- and low-voltage pulse combinations for gene electrotransfer in muscle, liver, tumor, and skin. Hum. Gene Ther. 2008, 19, 1261–1271. [Google Scholar] [CrossRef] [PubMed]
- Gothelf, A.; Gehl, J. Gene electrotransfer to skin; review of existing literature and clinical perspectives. Curr. Gene Ther. 2010, 10, 287–299. [Google Scholar] [CrossRef] [PubMed]
- Gothelf, A.; Gehl, J. Gene Electrotransfer to Skin. In Clinical Aspects of Electroporation; Springer: New York, NY, USA, 2011; pp. 189–201. [Google Scholar]
- Andre, F.M.; Mir, L.M. Nucleic acids electrotransfer in vivo: Mechanisms and practical aspects. Curr. Gene Ther. 2010, 10, 267–280. [Google Scholar] [CrossRef]
- Villemejane, J.; Mir, L.M. Physical methods of nucleic acid transfer: General concepts and applications. Br. J. Pharmacol. 2009, 157, 207–219. [Google Scholar] [CrossRef]
- Riccardo, F.; Barutello, G.; Petito, A.; Tarone, L.; Conti, L.; Arigoni, M.; Musiu, C.; Izzo, S.; Volante, M.; Longo, D.L.; et al. Immunization against ROS1 by DNA Electroporation Impairs K-Ras-Driven Lung Adenocarcinomas. Vaccines 2020, 8, 166. [Google Scholar] [CrossRef]
- Combadiere, B.; Liard, C. Transcutaneous and intradermal vaccination. Hum. Vaccin. 2011, 7, 811–827. [Google Scholar] [CrossRef]
- Kutzler, M.A.; Weiner, D.B. DNA vaccines: Ready for prime time? Nat. Rev. Genet. 2008, 9, 776–788. [Google Scholar] [CrossRef]
- Remic, T.; Sersa, G.; Ursic, K.; Cemazar, M.; Kamensek, U. Development of Tumor Cell-Based Vaccine with IL-12 Gene Electrotransfer as Adjuvant. Vaccines 2020, 8, 111. [Google Scholar] [CrossRef]
- Vesely, M.D.; Kershaw, M.H.; Schreiber, R.D.; Smyth, M.J. Natural Innate and Adaptive Immunity to Cancer. Annual Rev. Immunol. 2011, 29, 235–271. [Google Scholar] [CrossRef]
- Braumüller, H.; Wieder, T.; Brenner, E.; Aßmann, S.; Hahn, M.; Alkhaled, M.; Schilbach, K.; Essmann, F.; Kneilling, M.; Griessinger, C.; et al. T-helper-1-cell cytokines drive cancer into senescence. Nature 2013, 494, 361–365. [Google Scholar] [CrossRef]
- Kranjc Brezar, S.; Mrak, V.; Bosnjak, M.; Savarin, M.; Sersa, G.; Cemazar, M. Intratumoral Gene Electrotransfer of Plasmid DNA Encoding shRNA against Melanoma Cell Adhesion Molecule Radiosensitizes Tumors by Antivascular Effects and Activation of an Immune Response. Vaccines 2020, 8, 135. [Google Scholar] [CrossRef] [PubMed]
- Colombo, M.P.; Trinchieri, G. Interleukin-12 in anti-tumor immunity and immunotherapy. Cytokine Growth Factor Rev. 2002, 13, 155–168. [Google Scholar] [CrossRef]
- Yamashita, Y.I.; Shimada, M.; Hasegawa, H.; Minagawa, R.; Rikimaru, T.; Hamatsu, T.; Tanaka, S.; Shirabe, K.; Miyazaki, J.I.; Sugimachi, K. Electroporation-mediated interleukin-12 gene therapy for hepatocellular carcinoma in the mice model. Cancer Res. 2001, 61, 1005–1012. [Google Scholar] [PubMed]
- Lampreht Tratar, U.; Loiacono, L.; Cemazar, M.; Kamensek, U.; Fazio, V.M.; Sersa, G.; Signori, E. Gene Electrotransfer of Plasmid-Encoding IL-12 Recruits the M1 Macrophages and Antigen-Presenting Cells Inducing the Eradication of Aggressive B16F10 Murine Melanoma. Mediat. Inflamm. 2017, 2017, 5285890. [Google Scholar] [CrossRef]
- Daud, A.I.; DeConti, R.C.; Andrews, S.; Urbas, P.; Riker, A.I.; Sondak, V.K.; Munster, P.N.; Sullivan, D.M.; Ugen, K.E.; Messina, J.L.; et al. Phase I trial of interleukin-12 plasmid electroporation in patients with metastatic melanoma. J. Clin. Oncol. 2008, 26, 5896–5903. [Google Scholar] [CrossRef]
- Algazi, A.; Bhatia, S.; Agarwala, S.; Molina, M.; Lewis, K.; Faries, M.; Fong, L.; Levine, L.P.; Franco, M.; Oglesby, A.; et al. Intratumoral delivery of tavokinogene telseplasmid yields systemic immune responses in metastatic melanoma patients. Ann. Oncol. 2020, 31, 532–540. [Google Scholar] [CrossRef]
- Lopes, A.; Vandermeulen, G.; Préat, V. Cancer DNA vaccines: Current preclinical and clinical developments and future perspectives. J. Exp. Clin. Cancer Res. 2019, 38, 146. [Google Scholar] [CrossRef]
- Chiarella, P.; Massi, E.; De Robertis, M.; Sibilio, A.; Parrella, P.; Fazio, V.M.; Signori, E. Electroporation of skeletal muscle induces danger signal release and antigen-presenting cell recruitment independently of DNA vaccine administration. Expert Opin. Biol. Ther. 2008, 8, 1645–1657. [Google Scholar] [CrossRef]
- Calvet, C.Y.; Thalmensi, J.; Liard, C.; Pliquet, E.; Bestetti, T.; Huet, T.; Langlade-Demoyen, P.; Mir, L.M. Optimization of a gene electrotransfer procedure for efficient intradermal immunization with an hTERT-based DNA vaccine in mice. Mol. Ther. Methods Clin. Dev. 2014, 1, 14045. [Google Scholar] [CrossRef]
- Maglietti, F.; Michinski, S.; Olaiz, N.; Castro, M.; Suárez, C.; Marshall, G. The role of pH fronts in tissue electroporation based treatments. PLoS ONE 2013, 8, e80167. [Google Scholar] [CrossRef] [PubMed]
- Peri, D.; Deville, M.; Poignard, C.; Signori, E.; Natalini, R. Numerical optimization of plasmid DNA delivery combined with hyaluronidase injection for electroporation protocol. Comput. Methods Progr. Biomed. 2020, 186, 105204. [Google Scholar] [CrossRef] [PubMed]
- Sieni, E.; Dettin, M.; De Robertis, M.; Bazzolo, B.; Conconi, M.T.; Zamuner, A.; Marino, R.; Keller, F.; Campana, L.G.; Signori, E. The Efficiency of Gene Electrotransfer in Breast-Cancer Cell Lines Cultured on a Novel Collagen-Free 3D Scaffold. Cancers 2020, 12, 1043. [Google Scholar] [CrossRef] [PubMed]
- De Robertis, M.; Pasquet, L.; Loiacono, L.; Bellard, E.; Messina, L.; Vaccaro, S.; Di Pasquale, R.; Fazio, V.M.; Rols, M.-P.; Teissie, J.; et al. In Vivo Evaluation of a New Recombinant Hyaluronidase to Improve Gene Electro-Transfer Protocols for DNA-Based Drug Delivery against Cancer. Cancers 2018, 10, 405. [Google Scholar] [CrossRef] [PubMed]
- McMahon, J.M.; Signori, E.; Wells, K.E.; Fazio, V.M.; Wells, D.J. Optimisation of electrotransfer of plasmid into skeletal muscle by pretreatment with hyaluronidase—Increased expression with reduced muscle damage. Gene Ther. 2001, 8, 1264–1270. [Google Scholar] [CrossRef] [PubMed]
- Serša, G.; Čemažar, M.; Menart, V.; Gaberc-Porekar, V.; Miklavčič, D. Anti-tumor effectiveness of electrochemotherapy with bleomycin is increased by TNF-α on SA-1 tumors in mice. Cancer Lett. 1997, 116, 85–92. [Google Scholar] [CrossRef]
- Mir, L.M.; Orlowski, S.; Poddevin, B.; Belehradek, J., Jr. Electrochemotherapy tumor treatment is improved by interleukin-2 stimulation of the host’s defenses. Eur. Cytokine Netw. 1992, 3, 331–334. [Google Scholar]
- Mir, L.M.; Roth, C.; Orlowski, S.; Quintin-Colonna, F.; Fradelizi, D.; Belehradek, J., Jr.; Kourilsky, P. Systemic antitumor effects of electrochemotherapy combined with histoincompatible cells secreting interleukin-2. J. Immunother. Emphas. Tumor Immunol. 1995, 17, 30–38. [Google Scholar] [CrossRef]
- Ichim, C.V. Revisiting immunosurveillance and immunostimulation: Implications for cancer immunotherapy. J. Transl. Med. 2005, 3, 8. [Google Scholar] [CrossRef]
- Prehn, R.T.; Prehn, L.M. The flip side of immune surveillance: Immune dependency. Immunol. Rev. 2008, 222, 341–356. [Google Scholar] [CrossRef]
- He, Y.; Zha, J.; Wang, Y.; Liu, W.; Yang, X.; Yu, P. Tissue damage–associated “danger signals” influence T-cell responses that promote the progression of preneoplasia to cancer. Cancer Res. 2013. [Google Scholar] [CrossRef] [PubMed]
- Campana, L.G.; Edhemovic, I.; Soden, D.; Perrone, A.M.; Scarpa, M.; Campanacci, L.; Cemazar, M.; Valpione, S.; Miklavčič, D.; Mocellin, S.; et al. Electrochemotherapy—Emerging applications technical advances, new indications, combined approaches, and multi-institutional collaboration. Eur. J. Surg. Oncol. 2019, 45, 92–102. [Google Scholar] [CrossRef] [PubMed]
- Mozzillo, N.; Simeone, E.; Benedetto, L.; Curvietto, M.; Giannarelli, D.; Gentilcore, G.; Camerlingo, R.; Capone, M.; Madonna, G.; Festino, L.; et al. Assessing a novel immuno-oncology-based combination therapy: Ipilimumab plus electrochemotherapy. Oncoimmunology 2015, 4, e1008842. [Google Scholar] [CrossRef] [PubMed]
- Brizio, M.; Fava, P.; Astrua, C.; Cavaliere, G.; Savoia, P. Complete regression of melanoma skin metastases after electrochemotherapy plus ipilimumab treatment: An unusual clinical presentation. Eur. J. Dermatol. 2015, 25, 271–272. [Google Scholar] [CrossRef] [PubMed]
- Pavlin, D.; Tozon, N.; Sersa, G.; Pogacnik, A.; Cemazar, M. Efficient electrotransfection into canine muscle. Technol. Cancer Res. Treat. 2008, 7, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Cutrera, J.; Torrero, M.; Shiomitsu, K.; Mauldin, N.; Li, S. Intratumoral bleomycin and IL-12 electrochemogenetherapy for treating head and neck tumors in dogs. Methods Mol. Biol. 2008, 423, 319–325. [Google Scholar] [CrossRef]
- Reed, S.D.; Fulmer, A.; Buckholz, J.; Zhang, B.; Cutrera, J.; Shiomitsu, K.; Li, S. Bleomycin/interleukin-12 electrochemogene therapy for treating naturally occurring spontaneous neoplasms in dogs. Cancer Gene Ther. 2010, 17, 457–464. [Google Scholar] [CrossRef]
- Pavlin, D.; Cemazar, M.; Cör, A.; Sersa, G.; Pogacnik, A.; Tozon, N. Electrogene therapy with interleukin-12 in canine mast cell tumors. Radiol. Oncol. 2011, 45, 31–39. [Google Scholar] [CrossRef]
- Cemazar, M.; Sersa, G.; Pavlin, D.; Tozon, N. Intramuscular IL-12 electrogene therapy for treatment of spontaneous canine tumors. Targets Gene Ther. 2011, 299, 320. [Google Scholar] [CrossRef]
- Cutrera, J.; King, G.; Jones, P.; Kicenuik, K.; Gumpel, E.; Xia, X.; Li, S. Safe and effective treatment of spontaneous neoplasms with interleukin 12 electro-chemo-gene therapy. J. Cell Mol. Med. 2015, 19, 664–675. [Google Scholar] [CrossRef]
- Maglietti, F.; Michinski, S.; Emanuela, S.; Tellado, M.; Marshall, G. Electrochemotherapy immune response enhancement by gene electrotransfer using IL-2 and IL-12 genes in canine patients. Eur. J. Cancer 2016, 61, S210. [Google Scholar] [CrossRef]
- Cicchelero, L.; Denies, S.; Haers, H.; Vanderperren, K.; Stock, E.; Van Brantegem, L.; de Rooster, H.; Sanders, N.N. Intratumoural interleukin 12 gene therapy stimulates the immune system and decreases angiogenesis in dogs with spontaneous cancer. Vet. Comp. Oncol. 2017, 15, 1187–1205. [Google Scholar] [CrossRef] [PubMed]
- Cemazar, M.; Ambrozic Avgustin, J.; Pavlin, D.; Sersa, G.; Poli, A.; Krhac Levacic, A.; Tesic, N.; Lampreht Tratar, U.; Rak, M.; Tozon, N. Efficacy and safety of electrochemotherapy combined with peritumoral IL-12 gene electrotransfer of canine mast cell tumours. Vet. Comp. Oncol. 2017, 15, 641–654. [Google Scholar] [CrossRef] [PubMed]
- Cicchelero, L.; Denies, S.; Vanderperren, K.; Stock, E.; Van Brantegem, L.; de Rooster, H.; Sanders, N.N. Immunological, anti-angiogenic and clinical effects of intratumoral interleukin 12 electrogene therapy combined with metronomic cyclophosphamide in dogs with spontaneous cancer: A pilot study. Cancer Lett. 2017, 400, 205–218. [Google Scholar] [CrossRef] [PubMed]
- Salvadori, C.; Svara, T.; Rocchigiani, G.; Millanta, F.; Pavlin, D.; Cemazar, M.; Lampreht Tratar, U.; Sersa, G.; Tozon, N.; Poli, A. Effects of Electrochemotherapy with Cisplatin and Peritumoral IL-12 Gene Electrotransfer on Canine Mast Cell Tumors: A Histopathologic and Immunohistochemical Study. Radiol. Oncol. 2017, 51, 286–294. [Google Scholar] [CrossRef] [PubMed]
- Milevoj, N.; Tratar, U.L.; Nemec, A.; Brožič, A.; Žnidar, K.; Serša, G.; Čemažar, M.; Tozon, N. A combination of electrochemotherapy, gene electrotransfer of plasmid encoding canine IL-12 and cytoreductive surgery in the treatment of canine oral malignant melanoma. Res. Vet. Sci. 2019, 122, 40–49. [Google Scholar] [CrossRef]
- Milevoj, N.; Tozon, N.; Licen, S.; Lampreht Tratar, U.; Sersa, G.; Cemazar, M. Health-related quality of life in dogs treated with electrochemotherapy and/or interleukin-12 gene electrotransfer. Vet. Med. Sci. 2020. [Google Scholar] [CrossRef]
- Maglietti, F.; Tellado, M.; Olaiz, N.; Michinski, S.; Marshall, G. Minimally Invasive Electrochemotherapy Procedure for Treating Nasal Duct Tumors in Dogs using a Single Needle Electrode. Radiol. Oncol. 2017, 51, 422–430. [Google Scholar] [CrossRef]
- Mali, B.; Jarm, T.; Snoj, M.; Sersa, G.; Miklavcic, D. Antitumor effectiveness of electrochemotherapy: A systematic review and meta-analysis. Eur. J. Surg. Oncol. 2013, 39, 4–16. [Google Scholar] [CrossRef]
- Chuang, T.-F.; Lee, S.-C.; Liao, K.-W.; Hsiao, Y.-W.; Lo, C.-H.; Chiang, B.-L.; Lin, X.-Z.; Tao, M.-H.; Chu, R.-M. Electroporation-mediated IL-12 gene therapy in a transplantable canine cancer model. Int. J. Cancer 2009, 125, 698–707. [Google Scholar] [CrossRef]
- Kamensek, U.; Cemazar, M.; Lampreht Tratar, U.; Ursic, K.; Sersa, G. Antitumor in situ vaccination effect of TNFα and IL-12 plasmid DNA electrotransfer in a murine melanoma model. Cancer Immunol. Immunother. 2018, 67, 785–795. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, L.; De Smidt, E.; Geukens, N.; Declerck, P.; Hollevoet, K. DNA-Based Delivery of Checkpoint Inhibitors in Muscle and Tumor Enables Long-Term Responses with Distinct Exposure. Mol. Ther. 2020. [Google Scholar] [CrossRef] [PubMed]
- Turjanski, P.; Olaiz, N.; Maglietti, F.; Michinski, S.; Suárez, C.; Molina, F.V.; Marshall, G. The role of pH fronts in reversible electroporation. PLoS ONE 2011, 6, e17303. [Google Scholar] [CrossRef] [PubMed]
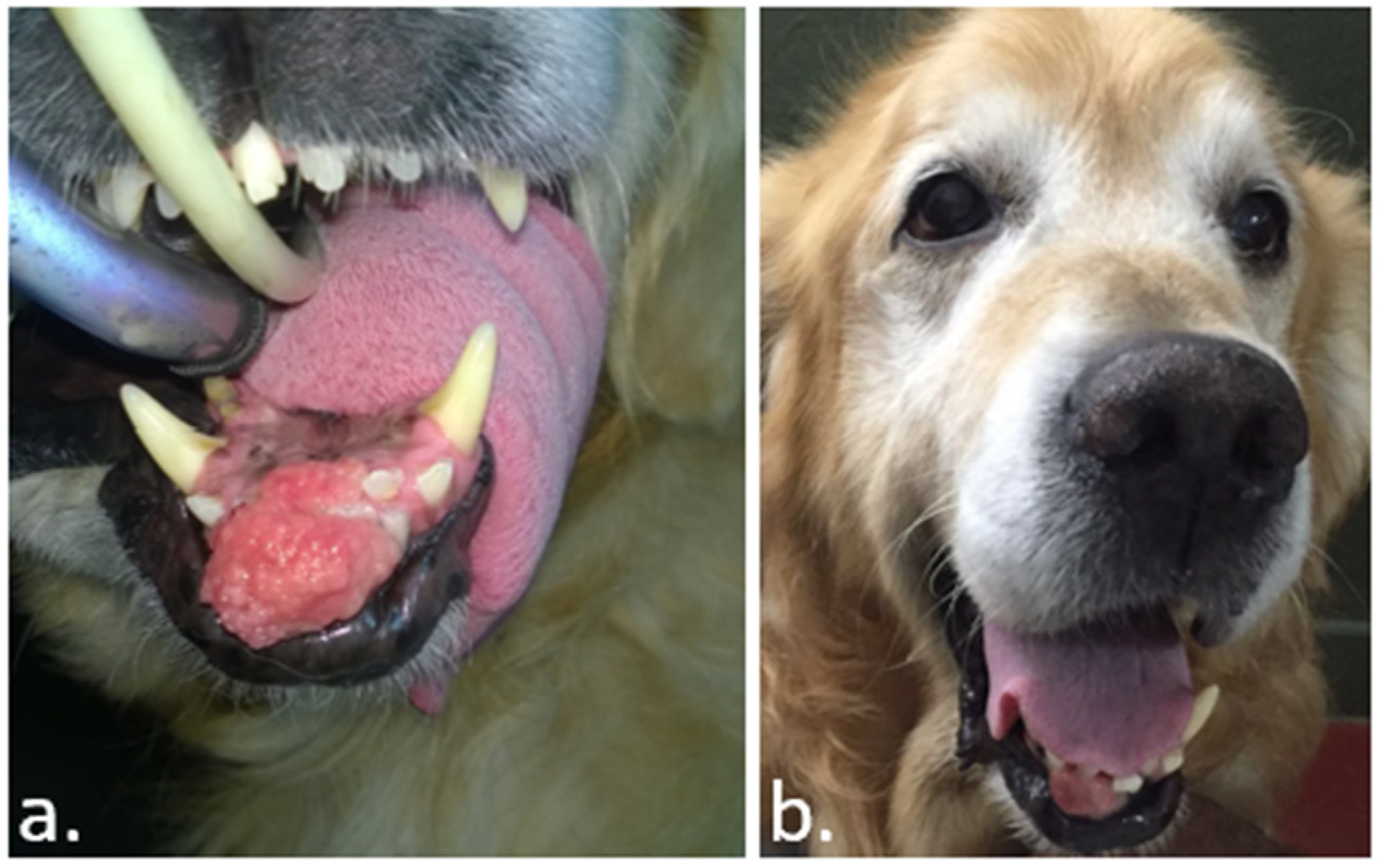
- Tellado, M.N.; Maglietti, F.H.; Michinski, S.D.; Marshall, G.R.; Signori, E. Electrochemotherapy in treatment of canine oral malignant melanoma and factors influencing treatment outcome. Radiol. Oncol. 2020, 54, 68–78. [Google Scholar] [CrossRef]
- Tellado, M.N.; Michinski, S.D.; Olaiz, N.; Maglietti, F.; Marshall, G. Canine Oral Eosinophilic Granuloma Treated with Electrochemotherapy. Case Rep. Vet. Med. 2014, 2014, 519197. [Google Scholar] [CrossRef]
- Suárez, C.; Soba, A.; Maglietti, F.; Olaiz, N.; Marshall, G. The role of additional pulses in electropermeabilization protocols. PLoS ONE 2014, 9, e113413. [Google Scholar] [CrossRef]
- Marino, M.; Olaiz, N.; Signori, E.; Maglietti, F.; Suárez, C.; Michinski, S.; Marshall, G. pH fronts and tissue natural buffer interaction in gene electrotransfer protocols. Electrochim. Acta 2017, 255, 463–471. [Google Scholar] [CrossRef]
- Suárez, C.; Soba, A.; Maglietti, F.; Olaiz, N.; Marshall, G. Effects of Pulse Addition in Electropermeabilization: Theoretical Insights on the Electric Conductivity. In Proceedings of the 1st World Congress on Electroporation and Pulsed Electric Fields in Biology, Medicine and Food & Environmental Technologies, Portoroz, Slovenia, 6–10 September 2016; pp. 215–218. [Google Scholar]
- Maglietti, F.H.; Michinski, S.D.; Ricotti, I.; Maure, P.; Mir, L.M.; Olaiz, N.; Marshall, G. Amelanotic Melanoma of the Root of the Tongue in A Canine Patient Treated by Electrochemotherapy. J. Anal. Oncol. 2014, 3, 217–221. [Google Scholar] [CrossRef]
- Rangel, M.M.M.; Luz, J.C.S.; Oliveira, K.D.; Ojeda, J.; Freytag, J.O.; Suzuki, D.O. Electrochemotherapy in the treatment of neoplasms in dogs and cats. Austral J. Vet. Sci. 2019, 51, 45–51. [Google Scholar] [CrossRef]
- Suzuki, D.O.H. Electrochemotherapy on Bladder—Preliminary Results. Bio. J. Sci. Tech. Res. 2018, 12. [Google Scholar] [CrossRef]
- Suzuki, D.O.H.; Berkenbrock, J.A.; de Oliveira, K.D.; Freytag, J.O.; Rangel, M.M.M. Novel application for electrochemotherapy: Immersion of nasal cavity in dog. Artif. Organs 2017, 41, 767–773. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, D.O.H.; Marques, C.M.G.; Rangel, M.M.M. Conductive Gel Increases the Small Tumor Treatment With Electrochemotherapy Using Needle Electrodes. Artif. Organs 2016, 40, 705–711. [Google Scholar] [CrossRef] [PubMed]
- Rangel, M.M.M.; Chaible, L.M.; Nagamine, M.K.; Mennecier, G.; Cogliati, B.; de Oliveira, K.D.; Fukumasu, H.; Sinhorini, I.L.; Mir, L.M.; Dagli, M.L.Z. Electroporation transiently decreases GJB2 (connexin 26) expression in B16/BL6 melanoma cell line. J. Membr. Biol. 2015, 248, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, D.O.H.; Berkenbrock, J.A.; Frederico, M.J.S.; Fátima, R.M.; Rangel, M.M.M. Oral Mucosa Model for Electrochemotherapy Treatment of Dog Mouth Cancer: Ex Vivo, In Silico, and In Vivo Experiments. Artif. Organs 2018, 42, 297–304. [Google Scholar] [CrossRef] [PubMed]
- Miklavcic, D.; Pucihar, G.; Pavlovec, M.; Ribaric, S.; Mali, M.; Macek-Lebar, A.; Petkovsek, M.; Nastran, J.; Kranjc, S.; Cemazar, M.; et al. The effect of high frequency electric pulses on muscle contractions and antitumor efficiency in vivo for a potential use in clinical electrochemotherapy. Bioelectrochemistry 2005, 65, 121–128. [Google Scholar] [CrossRef]
- Kotnik, T.; Mir, L.M.; Flisar, K.; Puc, M.; Miklavcic, D. Cell membrane electropermeabilization by symmetrical bipolar rectangular pulses. Part I. Increased efficiency of permeabilization. Bioelectrochemistry 2001, 54, 83–90. [Google Scholar] [CrossRef]
- Lanza, A.; Baldi, A.; Rossi, G.; Spugnini, E.P. Electrochemotherapy for the treatment of an incompletely excised subcutaneous low-grade epithelioid hemangioendothelioma in a budgerigar parakeet (Melopsittacus undulatus). Open Vet. J. 2019, 9, 269–272. [Google Scholar] [CrossRef]
- Spugnini, E.P.; Lanza, A.; Sebasti, S.; Baldi, A. Electrochemotherapy palliation of an oral squamous cell carcinoma in an African hedgehog (Atelerix albiventris). Vet. Res. Forum 2018, 9, 379–381. [Google Scholar]
- Racnik, J.; Svara, T.; Zadravec, M.; Gombac, M.; Cemazar, M.; Sersa, G.; Tozon, N. Electrochemotherapy with Bleomycin in the Treatment of Squamous Cell Carcinoma of the Uropygial Gland in a Cockatiel (Nymphicus Hollandicus). J. Exot. Pet Med. 2019, 29, 217–221. [Google Scholar] [CrossRef]
- Racnik, J.; Svara, T.; Zadravec, M.; Gombac, M.; Cemazar, M.; Sersa, G.; Tozon, N. Electrochemotherapy with Bleomycin of Different types of Cutaneous Tumours in a Ferret (Mustela Putorius Furo). Radiol. Oncol. 2018, 52, 98–104. [Google Scholar] [CrossRef]
- Stewart, T.H.M.; Heppner, G.H. Immunological enhancement of breast cancer. Parasitology 1997, 115, 141–153. [Google Scholar] [CrossRef] [PubMed]
- Prehn, R.T. The Immune Reaction as a Stimulator of Tumor Growth. Science 1972, 176, 170–171. [Google Scholar] [CrossRef]
- Parmiani, G.; Maccalli, C. The early antitumor immune response is necessary for tumor growth. OncoImmunology 2012, 1, 930–934. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Jenkins, R.W.; Barbie, D.A.; Flaherty, K.T. Mechanisms of resistance to immune checkpoint inhibitors. Br. J. Cancer 2018, 118, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Parakh, S.; King, D.; Gan, H.K.; Scott, A.M. Current Development of Monoclonal Antibodies in Cancer Therapy. Curr. Immun. Strateg. Cancer 2020, 1–70. [Google Scholar] [CrossRef]
- Andrews, A. Treating with Checkpoint Inhibitors-Figure $1 Million per Patient. Am. Health Drug Benefits 2015, 8, 9. [Google Scholar] [PubMed]
- Sultan, F.; Ganaie, B.A. Comparative oncology: Integrating human and veterinary medicine. Open Vet. J. 2018, 8, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Pinho, S.S.; Carvalho, S.; Cabral, J.; Reis, C.A.; Gärtner, F. Canine tumors: A spontaneous animal model of human carcinogenesis. Transl. Res. 2012, 159, 165–172. [Google Scholar] [CrossRef]
- Rowell, J.L.; McCarthy, D.O.; Alvarez, C.E. Dog models of naturally occurring cancer. Trends Mol. Med. 2011, 17, 380–388. [Google Scholar] [CrossRef]
| ECT | GET | IRE | |
|---|---|---|---|
| Spares non-dividing cells | yes | yes | no |
| Drugs used | bleomycin or cisplatin | plasmids | none |
| Electrical parameters | 8 × 1000 V/cm 100 µs | varies | 80–90 × 1000–30,000 V/cm 100 µs |
| Thermal effect | no | no | depends on the protocol |
| Regular use in human medicine | yes | no | yes |
| Regular use in veterinary medicine | yes | no | no |
| Uses | cancer | cancer, vaccines, gene therapy | cancer, ablation of tissues (cardiac) |
| Year | Technique | Plasmid | Pulse Parameters | Results | Author |
|---|---|---|---|---|---|
| 2008 | GET | IL-12 (human) | 1 × 600 V/cm 100 µs + 4 × 80 V/cm 100 ms | Safety n = 6 | Pavlin [84] |
| 2008 | ECT + GET | IL-12 | 2 × 450 V/cm 25 ms | CR: 100% n = 2 An untreated part of a lesion responded | Cutrera [85] |
| 2010 | ECT + GET | IL-12 (feline) | 2 × 400 V/cm 20 ms | CR: 80% PR: 20% n = 5 | Reed [86] |
| 2011 | GET + standard therapy | IL-12 (human) | 1 × 1200 V/cm 100 µs + 8 × 140 V/cm 50 ms | CR: 36.4% n = 8 (11 tumors) | Pavlin [87] |
| 2011 | GET + standard therapy | IL-12 (human) | 1 × 600 V/cm 100 µs + 4 × 80 V/cm 100 ms | CR: 33% n = 6 | Cemazar [88] |
| 2015 | ECT + GET | IL-12 (canine) | 2 × 350 V/cm 20 ms | CR: 14% PR: 36% n = 13 | Cutrera [89] |
| 2016 | ECT + GET | IL-2 (canine) + IL-12 (canine) | ECT 8 × 1000 V/cm 100 µs GET 1 × 1000 V/cm + 4 × 140 V/cm 100 ms (with hyaluronidase pretreatment) | SD: 100% One systemic response n = 5 | Maglietti [90] |
| 2017 | GET | IL-12 (human) | 2 × 750 V/cm 50 µs + 8 × 183 V/cm 10 ms | Authors describe immunostimulatory effects. n = 9 | Cicchelero [91] |
| 2017 | ECT + GET | IL-12 (human) | ECT: 8 × 1300 V/cm 100 µs GET: 1 × 1200 V/cm 100 µs + 1 × 140 V/cm 400 ms | CR: 72% n = 18 | Cemazar [92] |
| 2017 | GET + ciclophosphamide | IL-12 (human) | 2 × 750 V/cm 50 µs + 8 × 183 V/cm 10 ms | PD Slower tumor growth and improved well-being n = 6 | Cicchelero [93] |
| 2017 | ECT + GET | IL-12 (human) | ECT: 8 × 1300 V/cm 100 µs GET: 1 × 1200 V/cm 100 µs + 1 × 140 V/cm 400 ms | Reduced neoplastic cell proliferation and induced cellular response. n = 11 | Salvadori [94] |
| 2019 | ECT + GET + Surgery | IL-12 (canine) | ECT 8 × 1300 V/cm 100 µs GET 2 × 60 V/cm 150 ms | OR: 67% n = 9 | Milevoj [95] |
| 2020 | GET + various treatments | IL-12 | not reported | OR: 82% n = 44 | Milevoj [96] |
| Device, Company | Pulse-Type (Square) | Max Current [A] | Automatic/Manual | Max Frequency [Hz] | Electrodes Available | Company Location |
|---|---|---|---|---|---|---|
| EPV-100, BIOTEX (www.biotex.com.ar) | monopolar | 55 | Automatic | 5000 | Disposable needles in three versions, plates, nasal, and urethral | Argentina |
| Oncopore, BIOTEX (www.biotex.com.ar) | monopolar | 55 | Automatic | 5000 | Disposable needles in three versions, plates, nasal, and urethral for human use. | Argentina |
| VETCP 125, VETCP (www.vetcancer.com.br) | bipolar | 25 | Manual | 5000 | Reusable needles. | Brazil |
| BK100, BRUNNER | monopolar | 10 | Automatic | 1 | Disposable needles | Brazil |
| ElectroVET EZ, LEROY BIOTECH (www.leroybiotech.com) | monopolar | 25 | Automatic | 5000 | Reusable needles and plates | France |
| OnkoDisruptor, BIOPULSE BIOTECH (www.onkodisruptor.com) | bipolar | 5 | Automatic | Not reported | Reusable needles and plates | Italy |
| ECM 830, BTX (www.btxonline.com) | monopolar | 100 | Manual | 10 | Not provided for vet medicine | United States |
| Clinivet, IGEA (www.igea.it) | monopolar | 25 | Automatic/Manual | 5000 | Reusable needles in three versions and plates | Italy |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maglietti, F.; Tellado, M.; De Robertis, M.; Michinski, S.; Fernández, J.; Signori, E.; Marshall, G. Electroporation as the Immunotherapy Strategy for Cancer in Veterinary Medicine: State of the Art in Latin America. Vaccines 2020, 8, 537. https://doi.org/10.3390/vaccines8030537
Maglietti F, Tellado M, De Robertis M, Michinski S, Fernández J, Signori E, Marshall G. Electroporation as the Immunotherapy Strategy for Cancer in Veterinary Medicine: State of the Art in Latin America. Vaccines. 2020; 8(3):537. https://doi.org/10.3390/vaccines8030537
Chicago/Turabian StyleMaglietti, Felipe, Matías Tellado, Mariangela De Robertis, Sebastián Michinski, Juan Fernández, Emanuela Signori, and Guillermo Marshall. 2020. "Electroporation as the Immunotherapy Strategy for Cancer in Veterinary Medicine: State of the Art in Latin America" Vaccines 8, no. 3: 537. https://doi.org/10.3390/vaccines8030537
APA StyleMaglietti, F., Tellado, M., De Robertis, M., Michinski, S., Fernández, J., Signori, E., & Marshall, G. (2020). Electroporation as the Immunotherapy Strategy for Cancer in Veterinary Medicine: State of the Art in Latin America. Vaccines, 8(3), 537. https://doi.org/10.3390/vaccines8030537