Dendritic Cells and Myeloid Derived Suppressor Cells Fully Responsive to Stimulation via Toll-Like Receptor 4 Are Rapidly Induced from Bone-Marrow Cells by Granulocyte-Macrophage Colony-Stimulating Factor
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Isolation of BM Cells from Mice
2.3. GM-CSF Derived DC Culture
2.4. DC Activation by Lipopolysaccharide
2.5. Preparation and Incubation of Fluorescent Particles in BM Culture
2.6. Fluorochrome-Conjugated Antibody Cocktail Preparation and Cell Surface Staining
2.7. T Cell Purification
2.8. Mixed Leukocyte Reaction Assay
2.9. T Cell Co-Stimulation Assay
2.10. Cytokine Detection
2.11. Statistical Analyses
3. Results
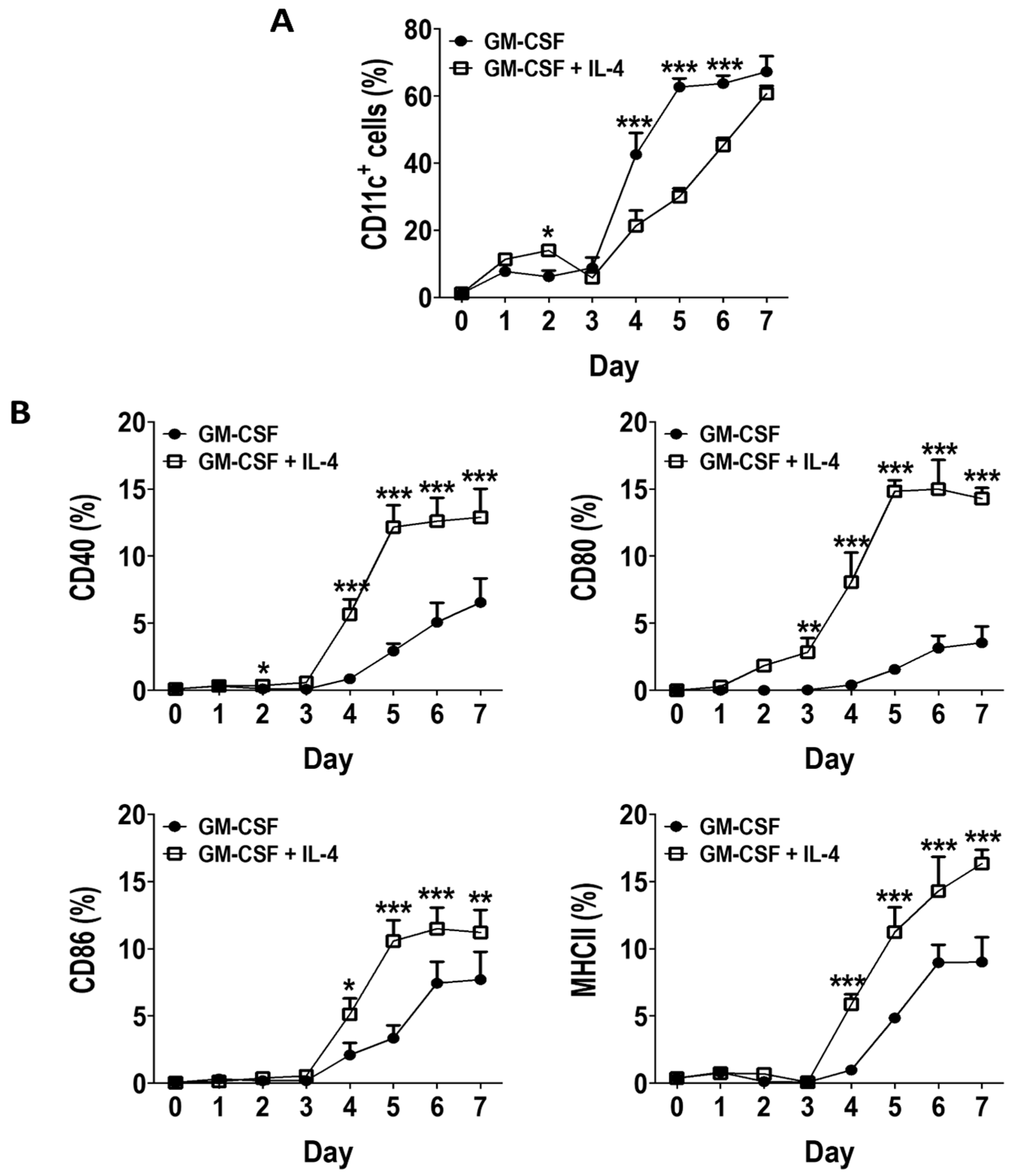
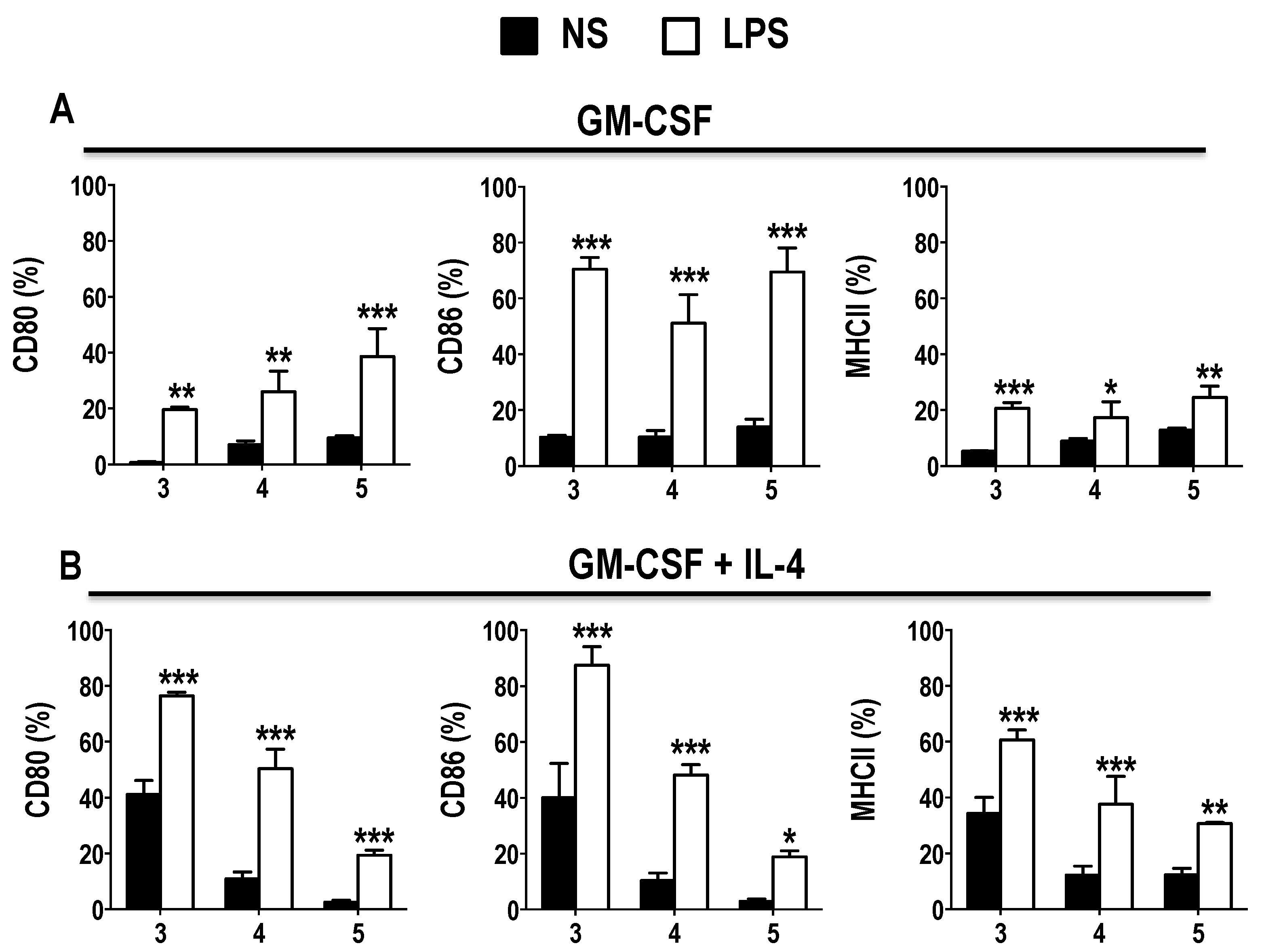
3.1. GM-CSF Induces an Early Surge in DC Generation without Inducing Expression of Activation Markers in BM Cultures
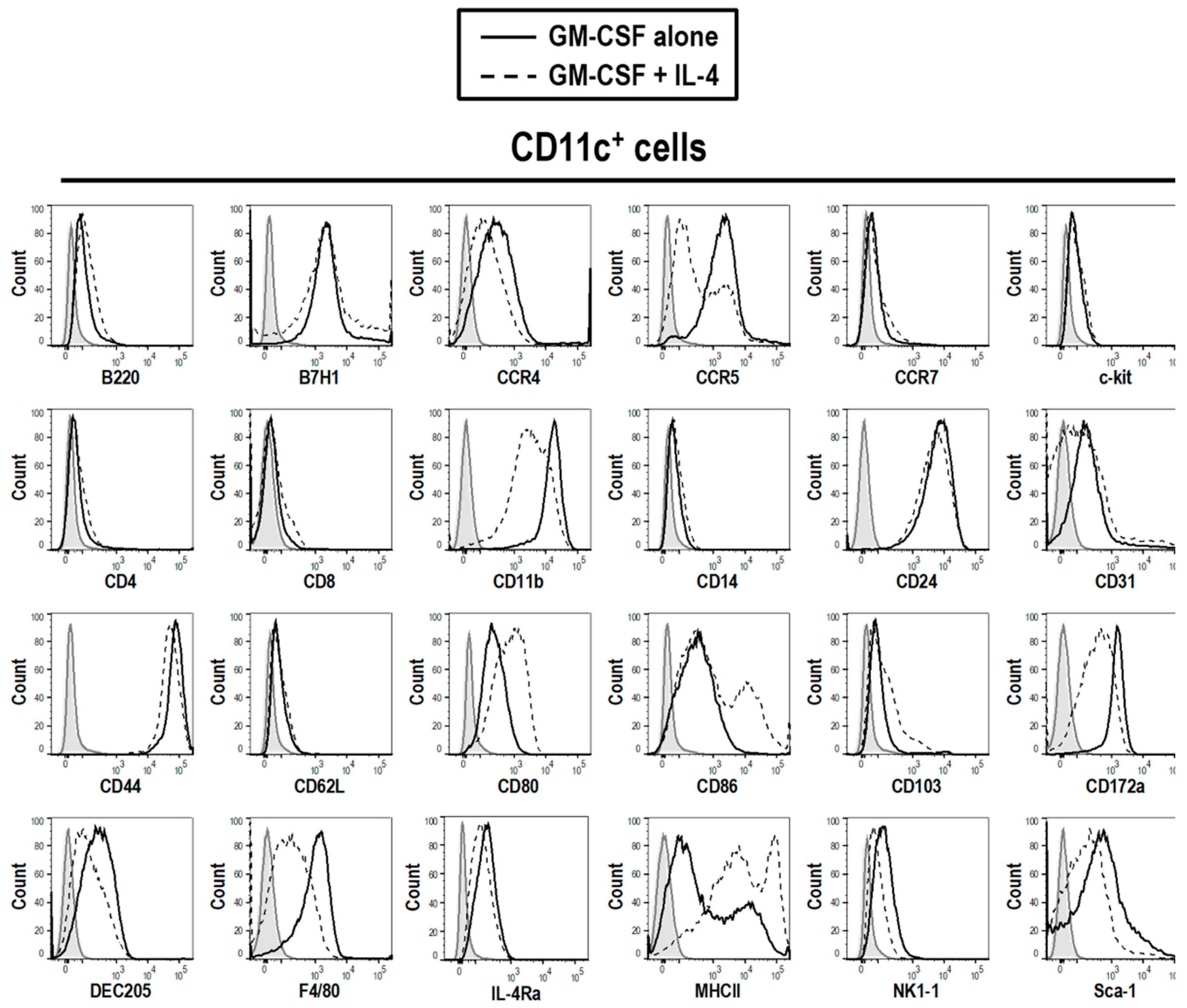
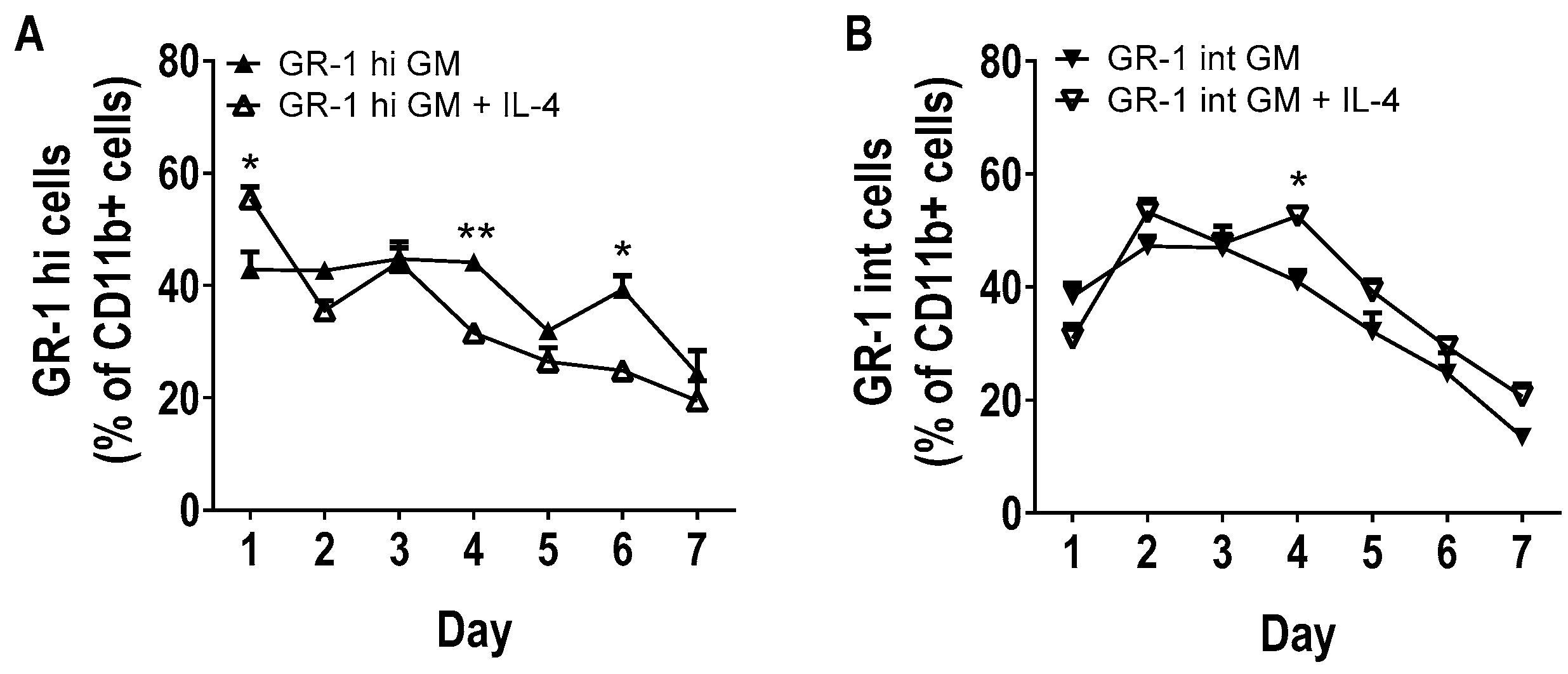
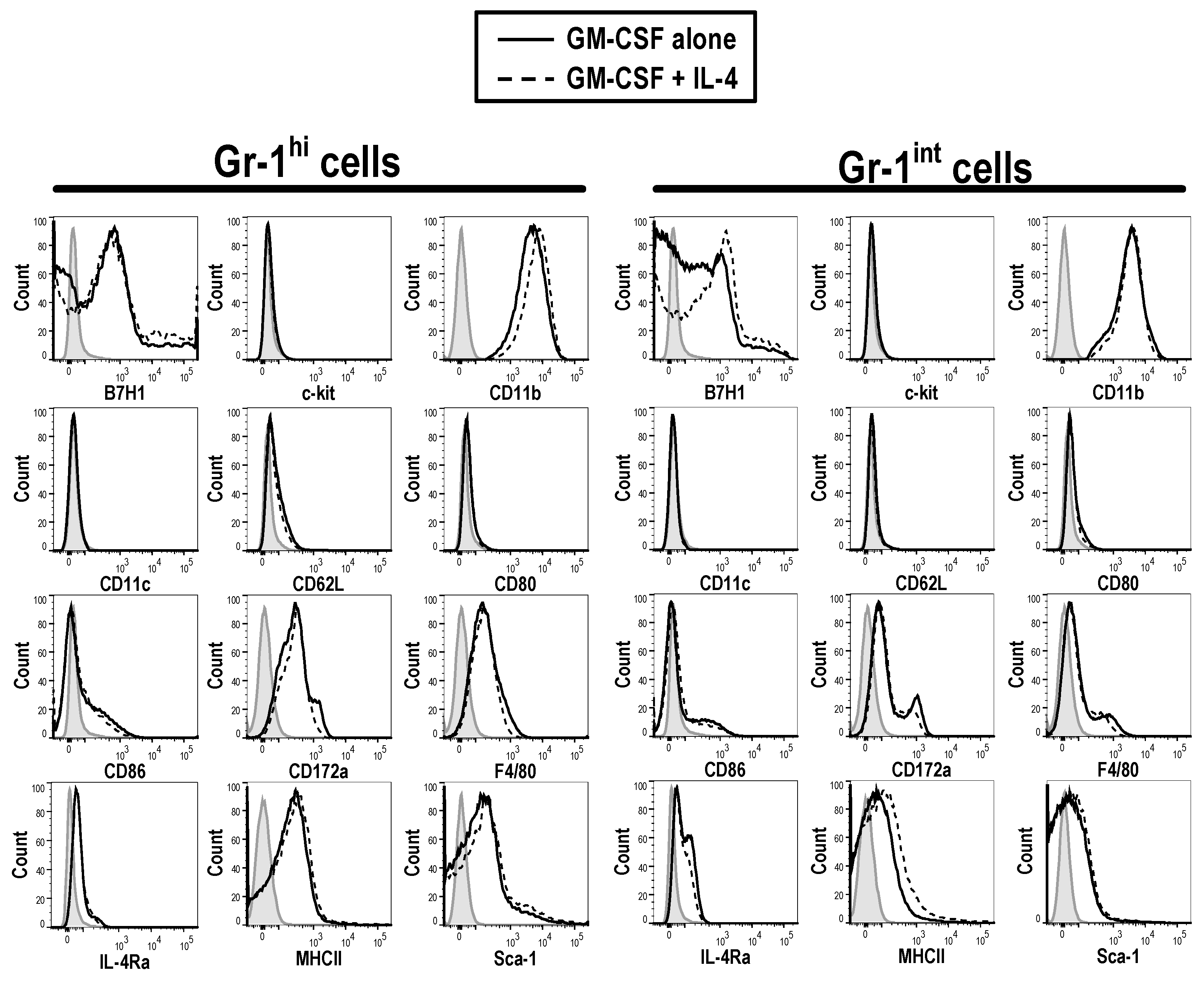
3.2. Phenotype of GM-CSF Cultured Cells Yield Conventional DCs, Granulocytic Gr-1hi and Monocytic Gr-1int MDSCs
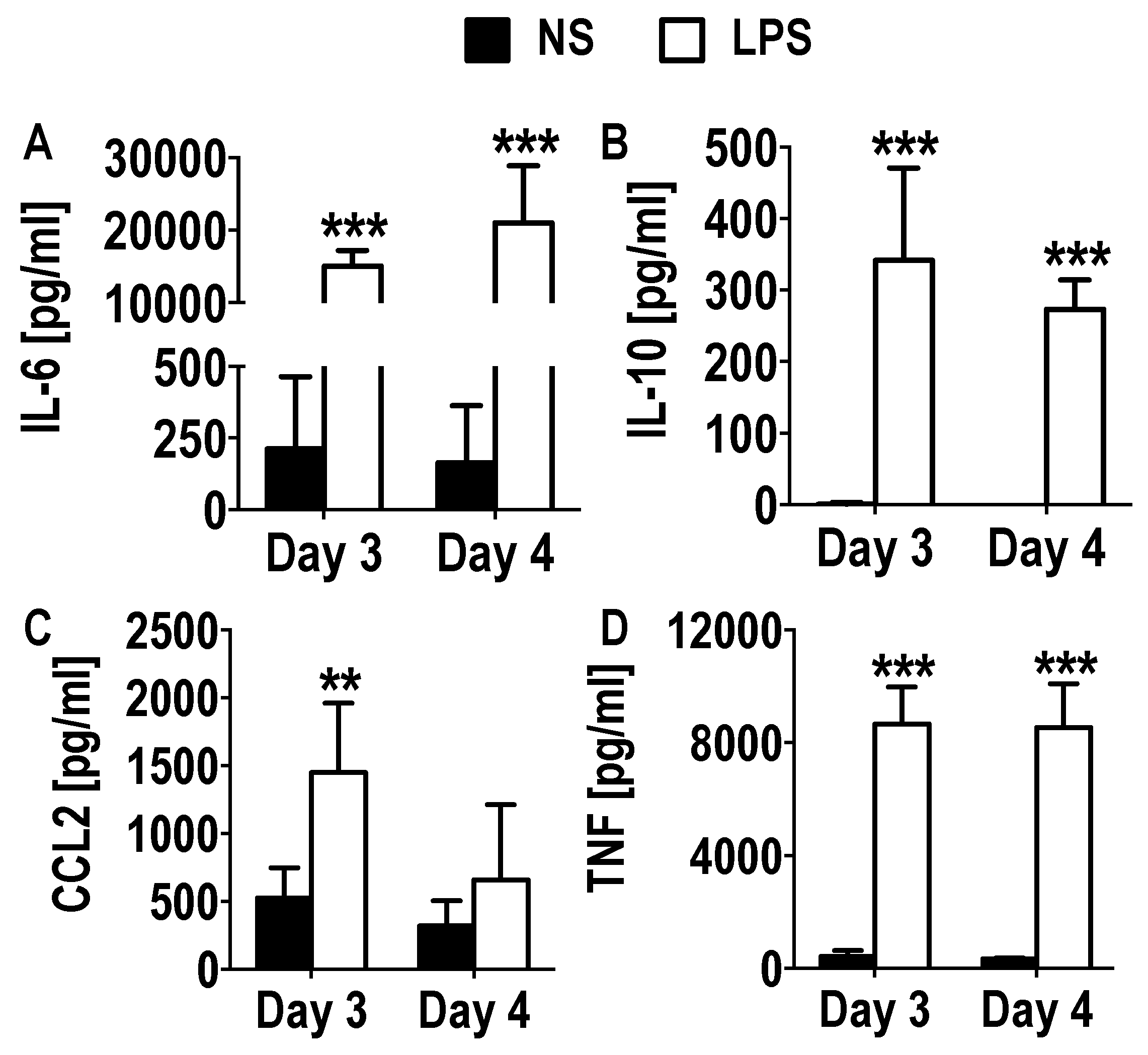
3.3. Functional Activation and Cytokine Secretion in Early GM-CSF Derived BM Cultures upon LPS Stimulation
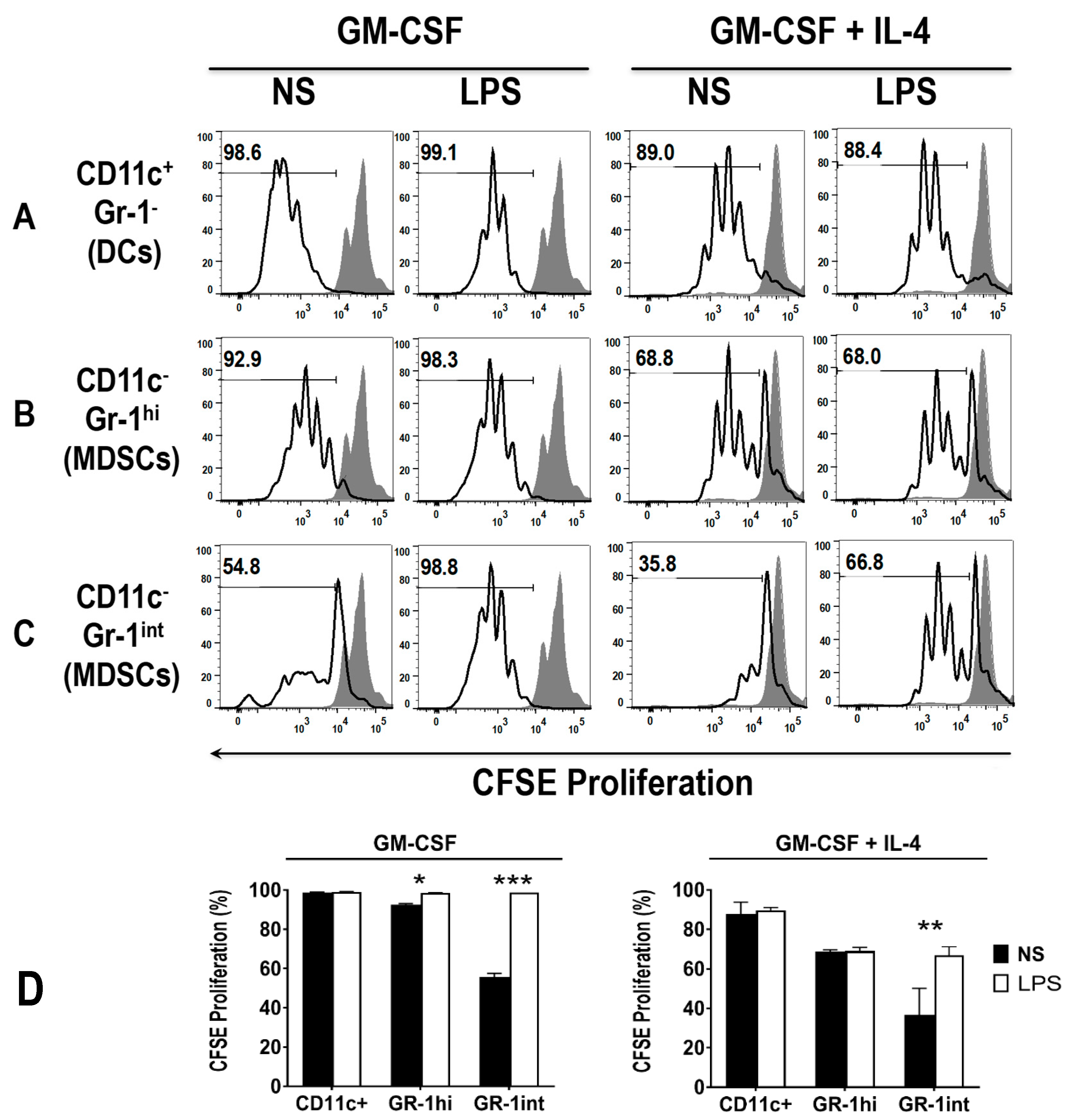
3.4. Early DCs Stimulate T Cells upon TLR4 Stimulation
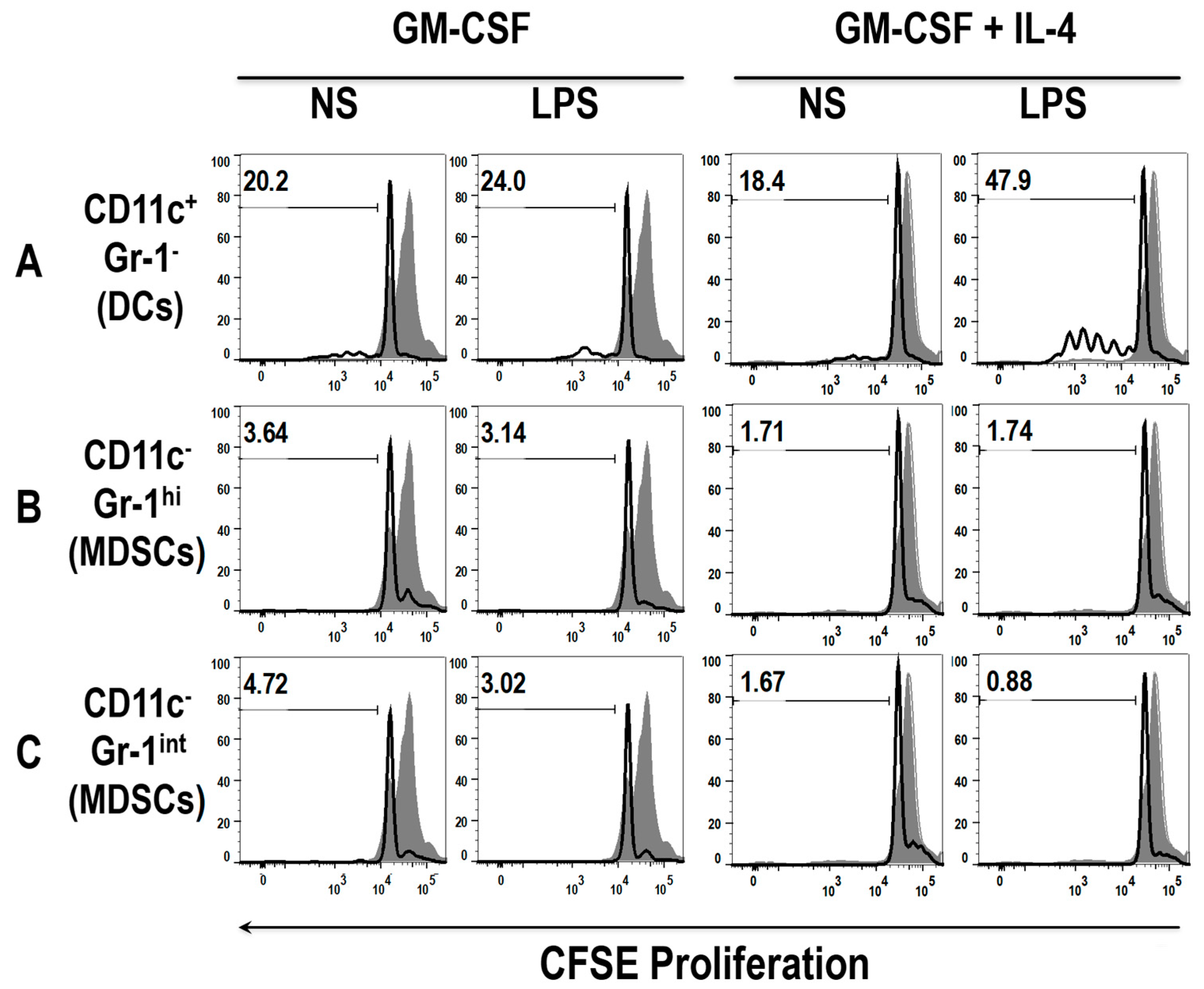
3.5. The Addition of LPS Enhances the Ability of Early MDSCs to Co-Stimualte T Cell Proliferation, in the Presence of Anti-CD3 and Anti-CD28
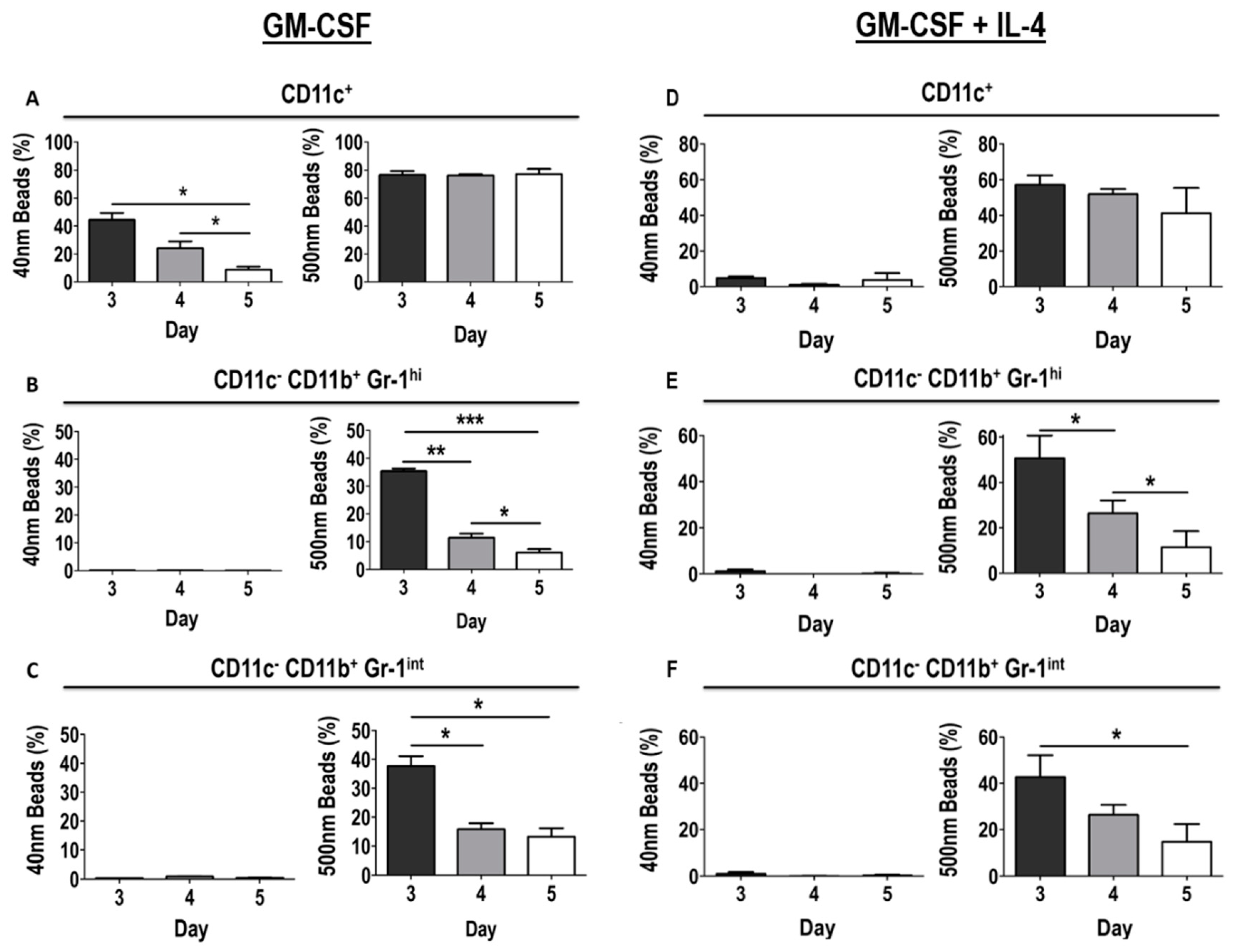
3.6. Differential Uptake Capacity by Early GM-CSF Derived DCs and MDSCs
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Geissmann, F.; Jung, S.; Littman, D.R. Blood monocytes consist of two principal subsets with distinct migratory properties. Immunity 2003, 19, 71–82. [Google Scholar] [CrossRef]
- Rossner, S.; Voigtlander, C.; Wiethe, C.; Hanig, J.; Seifarth, C.; Lutz, M.B. Myeloid dendritic cell precursors generated from bone marrow suppress T cell responses via cell contact and nitric oxide production in vitro. Eur. J. Immunol. 2005, 35, 3533–3544. [Google Scholar] [CrossRef] [PubMed]
- Dolcetti, L.; Peranzoni, E.; Ugel, S.; Marigo, I.; Fernandez Gomez, A.; Mesa, C.; Geilich, M.; Winkels, G.; Traggiai, E.; Casati, A.; et al. Hierarchy of immunosuppressive strength among myeloid-derived suppressor cell subsets is determined by GM-CSF. Eur. J. Immunol. 2010, 40, 22–35. [Google Scholar] [CrossRef] [PubMed]
- Morales, J.K.; Kmieciak, M.; Knutson, K.L.; Bear, H.D.; Manjili, M.H. GM-CSF is one of the main breast tumor-derived soluble factors involved in the differentiation of CD11b-Gr1- bone marrow progenitor cells into myeloid-derived suppressor cells. Breast Cancer Res. Treat. 2010, 123, 39–49. [Google Scholar] [CrossRef]
- Zhang, X.; Munegowda, M.A.; Yuan, J.; Wei, Y.; Xiang, J. Optimal TLR9 signal converts tolerogenic CD4-8- DCs into immunogenic ones capable of stimulating antitumor immunity via activating CD4+ Th1/Th17 and NK cell responses. J. Leukoc. Biol. 2010, 88, 393–403. [Google Scholar] [CrossRef] [PubMed]
- Movahedi, K.; Guilliams, M.; Van den Bossche, J.; Van den Bergh, R.; Gysemans, C.; Beschin, A.; De Baetselier, P.; Van Ginderachter, J.A. Identification of discrete tumor-induced myeloid-derived suppressor cell subpopulations with distinct T cell-suppressive activity. Blood 2008, 111, 4233–4244. [Google Scholar] [CrossRef] [PubMed]
- Huang, B.; Lei, Z.; Zhao, J.; Gong, W.; Liu, J.; Chen, Z.; Liu, Y.; Li, D.; Yuan, Y.; Zhang, G.M.; et al. CCL2/CCR2 pathway mediates recruitment of myeloid suppressor cells to cancers. Cancer Lett. 2007, 252, 86–92. [Google Scholar] [CrossRef]
- Bingisser, R.M.; Tilbrook, P.A.; Holt, P.G.; Kees, U.R. Macrophage-derived nitric oxide regulates T cell activation via reversible disruption of the Jak3/STAT5 signaling pathway. J. Immunol. 1998, 160, 5729–5734. [Google Scholar]
- Bronte, V.; Serafini, P.; Mazzoni, A.; Segal, D.M.; Zanovello, P. L-arginine metabolism in myeloid cells controls T-lymphocyte functions. Trends Immunol. 2003, 24, 302–306. [Google Scholar] [CrossRef]
- Huber, A.; Dammeijer, F.; Aerts, J.; Vroman, H. Current State of Dendritic Cell-Based Immunotherapy: Opportunities for in vitro Antigen Loading of Different DC Subsets? Front. Immunol. 2018, 9, 2804. [Google Scholar] [CrossRef]
- Hamilton, J.A.; Achuthan, A. Colony stimulating factors and myeloid cell biology in health and disease. Trends Immunol. 2013, 34, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Ushach, I.; Zlotnik, A. Biological role of granulocyte macrophage colony-stimulating factor (GM-CSF) and macrophage colony-stimulating factor (M-CSF) on cells of the myeloid lineage. J. Leukoc. Biol. 2016, 100, 481–489. [Google Scholar] [CrossRef]
- Hamilton, J.A. GM-CSF as a target in inflammatory/autoimmune disease: Current evidence and future therapeutic potential. Expert Rev. Clin. Immunol. 2015, 11, 457–465. [Google Scholar] [CrossRef] [PubMed]
- Helft, J.; Bottcher, J.; Chakravarty, P.; Zelenay, S.; Huotari, J.; Schraml, B.U.; Goubau, D.; e Sousa, C.R. GM-CSF Mouse Bone Marrow Cultures Comprise a Heterogeneous Population of CD11c(+)MHCII(+) Macrophages and Dendritic Cells. Immunity 2015, 42, 1197–1211. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Dai, X.; Hsu, C.; Ming, C.; He, Y.; Zhang, J.; Wei, L.; Zhou, P.; Wang, C.Y.; Yang, J.; et al. Discrimination of the heterogeneity of bone marrowderived dendritic cells. Mol. Med. Rep. 2017, 16, 6787–6793. [Google Scholar] [CrossRef]
- Zhang, W.; Ding, Y.; Sun, L.; Hong, Q.; Sun, Y.; Han, L.; Zi, M.; Xu, Y. Bone marrow-derived inflammatory and steady state DCs are different in both functions and survival. Cell. Immunol. 2018, 331, 100–109. [Google Scholar] [CrossRef]
- Chen, S.; Li, X.; Zhang, W.; Zi, M.; Xu, Y. Inflammatory compound lipopolysaccharide promotes the survival of GM-CSF cultured dendritic cell via PI3 kinase-dependent upregulation of Bcl-x. Immunol. Cell Biol. 2018, 96, 912–921. [Google Scholar] [CrossRef]
- Kong, Y.Y.; Fuchsberger, M.; Plebanski, M.; Apostolopoulos, V. Alteration of early dendritic cell activation by cancer cell lines predisposes immunosuppression, which cannot be reversed by TLR4 stimulation. Acta Biochim. Biophys. Sin. 2016, 48, 1101–1111. [Google Scholar] [CrossRef]
- Labeur, M.S.; Roters, B.; Pers, B.; Mehling, A.; Luger, T.A.; Schwarz, T.; Grabbe, S. Generation of tumor immunity by bone marrow-derived dendritic cells correlates with dendritic cell maturation stage. J. Immunol. 1999, 162, 168–175. [Google Scholar]
- Xu, Y.; Zhan, Y.; Lew, A.M.; Naik, S.H.; Kershaw, M.H. Differential development of murine dendritic cells by GM-CSF versus Flt3 ligand has implications for inflammation and trafficking. J. Immunol. 2007, 179, 7577–7584. [Google Scholar] [CrossRef]
- Mayordomo, J.I.; Zorina, T.; Storkus, W.J.; Zitvogel, L.; Garcia-Prats, M.D.; DeLeo, A.B.; Lotze, M.T. Bone marrow-derived dendritic cells serve as potent adjuvants for peptide-based antitumor vaccines. Stem Cells 1997, 15, 94–103. [Google Scholar] [CrossRef] [PubMed]
- Li, O.; Chang, X.; Zhang, H.; Kocak, E.; Ding, C.; Zheng, P.; Liu, Y. Massive and destructive T cell response to homeostatic cue in CD24-deficient lymphopenic hosts. J. Exp. Med. 2006, 203, 1713–1720. [Google Scholar] [CrossRef] [PubMed]
- Hegde, V.L.; Singh, N.P.; Nagarkatti, P.S.; Nagarkatti, M. CD44 mobilization in allogeneic dendritic cell-T cell immunological synapse plays a key role in T cell activation. J. Leukoc. Biol. 2008, 84, 134–142. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Wang, Z.; Wu, L.; Zhang, M.; Li, W.; Ding, J.; Zhu, J.; Wei, H.; Zhao, K. Circulating and tumor-infiltrating myeloid-derived suppressor cells in patients with colorectal carcinoma. PLoS ONE 2013, 8, e57114. [Google Scholar] [CrossRef] [PubMed]
- Sheng, K.C.; Pietersz, G.A.; Wright, M.D.; Apostolopoulos, V. Dendritic cells: Activation and maturation—Applications for cancer immunotherapy. Curr. Med. Chem. 2005, 12, 1783–1800. [Google Scholar] [CrossRef]
- Constantino, J.; Gomes, C.; Falcao, A.; Neves, B.M.; Cruz, M.T. Dendritic cell-based immunotherapy: A basic review and recent advances. Immunol. Res. 2017, 65, 798–810. [Google Scholar] [CrossRef]
- Schuler-Thurner, B.; Schultz, E.S.; Berger, T.G.; Weinlich, G.; Ebner, S.; Woerl, P.; Bender, A.; Feuerstein, B.; Fritsch, P.O.; Romani, N.; et al. Rapid induction of tumor-specific type 1 T helper cells in metastatic melanoma patients by vaccination with mature, cryopreserved, peptide-loaded monocyte-derived dendritic cells. J. Exp. Med. 2002, 195, 1279–1288. [Google Scholar] [CrossRef]
- Mastelic-Gavillet, B.; Balint, K.; Boudousquie, C.; Gannon, P.O.; Kandalaft, L.E. Personalized Dendritic Cell Vaccines-Recent Breakthroughs and Encouraging Clinical Results. Front. Immunol. 2019, 10, 766. [Google Scholar] [CrossRef]
- Bhargava, A.; Mishra, D.; Banerjee, S.; Mishra, P.K. Dendritic cell engineering for tumor immunotherapy: From biology to clinical translation. Immunotherapy 2012, 4, 703–718. [Google Scholar] [CrossRef]
- Inaba, K.; Inaba, M.; Deguchi, M.; Hagi, K.; Yasumizu, R.; Ikehara, S.; Muramatsu, S.; Steinman, R.M. Granulocytes, macrophages, and dendritic cells arise from a common major histocompatibility complex class II-negative progenitor in mouse bone marrow. Proc. Natl. Acad Sci. USA 1993, 90, 3038–3042. [Google Scholar] [CrossRef]
- Zhao, Y.; Wu, T.; Shao, S.; Shi, B.; Zhao, Y. Phenotype, development, and biological function of myeloid-derived suppressor cells. Oncoimmunology 2016, 5, e1004983. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, P.C.; Ernstoff, M.S.; Hernandez, C.; Atkins, M.; Zabaleta, J.; Sierra, R.; Ochoa, A.C. Arginase I-producing myeloid-derived suppressor cells in renal cell carcinoma are a subpopulation of activated granulocytes. Cancer Res. 2009, 69, 1553–1560. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Tan, L.; Zhu, W.; Lei, L.; Kuang, Y.; Liu, P.; Li, J.; Chen, X.; Peng, C. Targeting Myeloid-Derived Suppressor Cells Is a Novel Strategy for Anti-Psoriasis Therapy. Med. Inflamm. 2020, 2020, 8567320. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Zheng, L.; Mo, J.; Yao, X.; Fan, C.; Bao, Y. Protective Effects of MitoTEMPO on Nonalcoholic Fatty Liver Disease via Regulating Myeloid-Derived Suppressor Cells and Inflammation in Mice. Biomed. Res. Int. 2020, 2020, 9329427. [Google Scholar] [CrossRef]
- Weber, R.; Riester, Z.; Huser, L.; Sticht, C.; Siebenmorgen, A.; Groth, C.; Hu, X.; Altevogt, P.; Utikal, J.S.; Umansky, V. IL-6 regulates CCR5 expression and immunosuppressive capacity of MDSC in murine melanoma. J. Immunother. Cancer 2020, 8, e000949. [Google Scholar] [CrossRef]
- Dawod, B.; Liu, J.; Gebremeskel, S.; Yan, C.; Sappong, A.; Johnston, B.; Hoskin, D.W.; Marshall, J.S.; Wang, J. Myeloid-derived suppressor cell depletion therapy targets IL-17A-expressing mammary carcinomas. Sci. Rep. 2020, 10, 13343. [Google Scholar] [CrossRef]
- Loeuillard, E.; Yang, J.; Buckarma, E.; Wang, J.; Liu, Y.; Conboy, C.B.; Pavelko, K.D.; Li, Y.; O’Brien, D.; Wang, C.; et al. Targeting tumor-associated macrophages and granulocytic-myeloid-derived suppressor cells augments pd-1 blockade in cholangiocarcinoma. J. Clin. Investig. 2020, 137110. [Google Scholar] [CrossRef]
- Tavazoie, M.F.; Pollack, I.; Tanqueco, R.; Ostendorf, B.N.; Reis, B.S.; Gonsalves, F.C.; Kurth, I.; Andreu-Agullo, C.; Derbyshire, M.L.; Posada, J.; et al. LXR/ApoE Activation Restricts Innate Immune Suppression in Cancer. Cell 2018, 172, 825–840.e818. [Google Scholar] [CrossRef]
- Kong, Y.Y.; Fuchsberger, M.; Xiang, S.D.; Apostolopoulos, V.; Plebanski, M. Myeloid derived suppressor cells and their role in diseases. Curr. Med. Chem. 2013, 20, 1437–1444. [Google Scholar] [CrossRef]
- Brudecki, L.; Ferguson, D.A.; McCall, C.E.; El Gazzar, M. Myeloid-derived suppressor cells evolve during sepsis and can enhance or attenuate the systemic inflammatory response. Infect. Immun. 2012, 80, 2026–2034. [Google Scholar] [CrossRef]
- Zoglmeier, C.; Bauer, H.; Noerenberg, D.; Wedekind, G.; Bittner, P.; Sandholzer, N.; Rapp, M.; Anz, D.; Endres, S.; Bourquin, C. CpG blocks immunosuppression by myeloid-derived suppressor cells in tumor-bearing mice. Clin. Cancer Res. 2011, 17, 1765–1775. [Google Scholar] [CrossRef] [PubMed]
- Greifenberg, V.; Ribechini, E.; Rossner, S.; Lutz, M.B. Myeloid-derived suppressor cell activation by combined LPS and IFN-gamma treatment impairs DC development. Eur. J. Immunol. 2009, 39, 2865–2876. [Google Scholar] [CrossRef]
- Bunt, S.K.; Clements, V.K.; Hanson, E.M.; Sinha, P.; Ostrand-Rosenberg, S. Inflammation enhances myeloid-derived suppressor cell cross-talk by signaling through Toll-like receptor 4. J. Leukoc. Biol. 2009, 85, 996–1004. [Google Scholar] [CrossRef] [PubMed]
- Michels, T.; Shurin, G.V.; Naiditch, H.; Sevko, A.; Umansky, V.; Shurin, M.R. Paclitaxel promotes differentiation of myeloid-derived suppressor cells into dendritic cells in vitro in a TLR4-independent manner. J. Immunotoxicol. 2012, 9, 292–300. [Google Scholar] [CrossRef] [PubMed]
- Hong, E.H.; Chang, S.Y.; Lee, B.R.; Kim, Y.S.; Lee, J.M.; Kang, C.Y.; Kweon, M.N.; Ko, H.J. Blockade of Myd88 signaling induces antitumor effects by skewing the immunosuppressive function of myeloid-derived suppressor cells. Int. J. Cancer 2013, 132, 2839–2848. [Google Scholar] [CrossRef]
- Wang, S.; Sun, X.; Zhou, H.; Zhu, Z.; Zhao, W.; Zhu, C. Interleukin-4 affects the mature phenotype and function of rat bone marrow-derived dendritic cells. Mol. Med. Rep. 2015, 12, 233–237. [Google Scholar] [CrossRef]
- Karlson Tde, L.; Kong, Y.Y.; Hardy, C.L.; Xiang, S.D.; Plebanski, M. The signalling imprints of nanoparticle uptake by bone marrow derived dendritic cells. Methods 2013, 60, 275–283. [Google Scholar] [CrossRef]
- Platt, C.D.; Ma, J.K.; Chalouni, C.; Ebersold, M.; Bou-Reslan, H.; Carano, R.A.; Mellman, I.; Delamarre, L. Mature dendritic cells use endocytic receptors to capture and present antigens. Proc. Natl. Acad. Sci. USA 2010, 107, 4287–4292. [Google Scholar] [CrossRef]
- Karalkin, P.A.; Lupatov, A.Y.; Yarygin, K.N. Endocytosis of Micro- and Nanosized Particles in vitro by Human Dendritic Cells. Biol. Membr. 2009, 26, 394–400. [Google Scholar] [CrossRef]
- Nayak, J.V.; Hokey, D.A.; Larregina, A.; He, Y.; Salter, R.D.; Watkins, S.C.; Falo, L.D., Jr. Phagocytosis induces lysosome remodeling and regulated presentation of particulate antigens by activated dendritic cells. J. Immunol. 2006, 177, 8493–8503. [Google Scholar] [CrossRef]
| Antibody | Clone | Fluorophore | Company | Catalogue Number |
|---|---|---|---|---|
| B220 | RA3-6B2 | FITC | BD Biosciences | 553088 |
| B7H1 | MIH5 | PE | BD Biosciences | 557495 |
| c-kit | 2B8 | FITC | BD Biosciences | 553354 |
| CCR4 | 2G12 | PECy7 | BioLegend | 131214 |
| CCR5 | HM-CCR5 | APC | BioLegend | 107011 |
| CCR7 | 4B12 | PerCP Cy5.5 | eBioscience | 45-1971-82 |
| CD103 | M290 | PE | BD Biosciences | 557495 |
| CD11b | M1/70 | PE | BD Biosciences | 553311 |
| CD11b | M1/70 | PECy7 | BD Biosciences | 552850 |
| CD11c | HL3 | V450 | BD Biosciences | 560521 |
| CD14 | rmC5-3 | FITC | BD Biosciences | 553739 |
| CD172a | P84 | APC | BD Biosciences | 560106 |
| CD205 | 205yekta | PECy7 | BD Biosciences | 25-2051-41 |
| CD205 | NLDC-145 | Biotin | Cederlane | CL89145B |
| CD24 | M1/69 | PECy7 | BD Biosciences | 560536 |
| CD3 | 17A2 | FITC | BD Biosciences | 555274 |
| CD3 | 500A2 | AlexaFluor 700 | BD Biosciences | 557984 |
| CD31 | MEC13.3 | APC | BD Biosciences | 551262 |
| CD4 | GK1.5 | PE | BD Biosciences | 557308 |
| CD4 | RM4-5 | PerCP | BD Biosciences | 553052 |
| CD40 | 3/23 | FITC | BD Biosciences | 553791 |
| CD44 | IM7 | Biotin | BD Biosciences | 553132 |
| CD62L | MEL-14 | FITC | BD Biosciences | 553150 |
| CD80 | 16-10A1 | Biotin | BD Biosciences | 553767 |
| CD86 | GL1 | PE | BD Biosciences | 553692 |
| CD8a | 53-6.7 | PE | BD Biosciences | 553033 |
| CD8a | 53-6.7 | PerCP | BD Biosciences | 553036 |
| CD8a | 53-6.7 | APC | BD Biosciences | 553035 |
| F4/80 | BM8 | PE | eBioscience | 12-4801-80 |
| Gr-1 | RB6-8C5 | PerCP Cy5.5 | BD Biosciences | 552093 |
| Gr-1 | RB6-8C5 | Biotin | BD Biosciences | 553125 |
| IL-4Ra | mIL4R-M1 | PE | BD Biosciences | 561695 |
| MHCI class II | M5/114.15.2 | APCCy7 | BD Biosciences | 47-5321-80 |
| NK1.1 | PK136 | FITC | eBioscience | 12-5941-83 |
| Sca-1 | D7 | PE-Cy7 | BD Biosciences | 558162 |
| Streptavidin | AlexaFluor 700 | Invitrogen | S21383 | |
| Streptavidin | APC | BD Biosciences | 554067 | |
| Streptavidin | APCCy7 | eBioscience | 47-4317-82 | |
| Streptavidin | FITC | BD Biosciences | 554060 | |
| Streptavidin | PE | BD Biosciences | 554061 |
| Surface Markers | GM-CSF DCs | GM-CSF + IL-4 DCs | p-Value | Significance |
|---|---|---|---|---|
| Mean MFI ± SD | Mean MFI ± SD | |||
| CD11b | 14,969 ± 2668 | 4072 ± 473.1 | 0.0233 | * |
| CD44 | 75,858 ± 5882 | 52,938 ± 3483 | 0.0038 | ** |
| CD80 | 751.3 ± 10.79 | 1368 ± 13.5 | <0.0001 | *** |
| CD86 | 991 ± 26.96 | 3229 ± 427 | 0.0114 | * |
| CD103 | 4933 ± 253.2 | 2444 ± 153.3 | 0.0029 | ** |
| CD172α | 1943 ± 65.59 | 1599 ± 113.4 | 0.0098 | ** |
| F4/80 | 1499 ± 124.2 | 733.3 ± 48.68 | 0.0049 | ** |
| MHCII | 2052 ± 248.6 | 12,634 ± 452.3 | 0.0004 | *** |
| Sca-1 | 3499 ± 339.2 | 2892 ± 278.9 | 0.007 | ** |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kong, Y.Y.; Wilson, K.; Apostolopoulos, V.; Plebanski, M. Dendritic Cells and Myeloid Derived Suppressor Cells Fully Responsive to Stimulation via Toll-Like Receptor 4 Are Rapidly Induced from Bone-Marrow Cells by Granulocyte-Macrophage Colony-Stimulating Factor. Vaccines 2020, 8, 522. https://doi.org/10.3390/vaccines8030522
Kong YY, Wilson K, Apostolopoulos V, Plebanski M. Dendritic Cells and Myeloid Derived Suppressor Cells Fully Responsive to Stimulation via Toll-Like Receptor 4 Are Rapidly Induced from Bone-Marrow Cells by Granulocyte-Macrophage Colony-Stimulating Factor. Vaccines. 2020; 8(3):522. https://doi.org/10.3390/vaccines8030522
Chicago/Turabian StyleKong, Ying Ying, Kirsty Wilson, Vasso Apostolopoulos, and Magdalena Plebanski. 2020. "Dendritic Cells and Myeloid Derived Suppressor Cells Fully Responsive to Stimulation via Toll-Like Receptor 4 Are Rapidly Induced from Bone-Marrow Cells by Granulocyte-Macrophage Colony-Stimulating Factor" Vaccines 8, no. 3: 522. https://doi.org/10.3390/vaccines8030522
APA StyleKong, Y. Y., Wilson, K., Apostolopoulos, V., & Plebanski, M. (2020). Dendritic Cells and Myeloid Derived Suppressor Cells Fully Responsive to Stimulation via Toll-Like Receptor 4 Are Rapidly Induced from Bone-Marrow Cells by Granulocyte-Macrophage Colony-Stimulating Factor. Vaccines, 8(3), 522. https://doi.org/10.3390/vaccines8030522







