Tetravalent Immunogen Assembled from Conserved Regions of HIV-1 and Delivered as mRNA Demonstrates Potent Preclinical T-Cell Immunogenicity and Breadth
Abstract
1. Introduction
2. Materials and Methods
2.1. mRNA Synthesis and Lipid Nanoparticle Formulation
2.2. Mice, Immunizations and Preparation of Splenocytes
2.3. Peptides and Peptide Pools
2.4. INF-γ ELISPOT Assay
2.5. Intracellular Cytokine Staining (ICS) Assay
2.6. Statistical Analysis
3. Results
3.1. Tetravalent mRNA Vaccine HIVconsvM
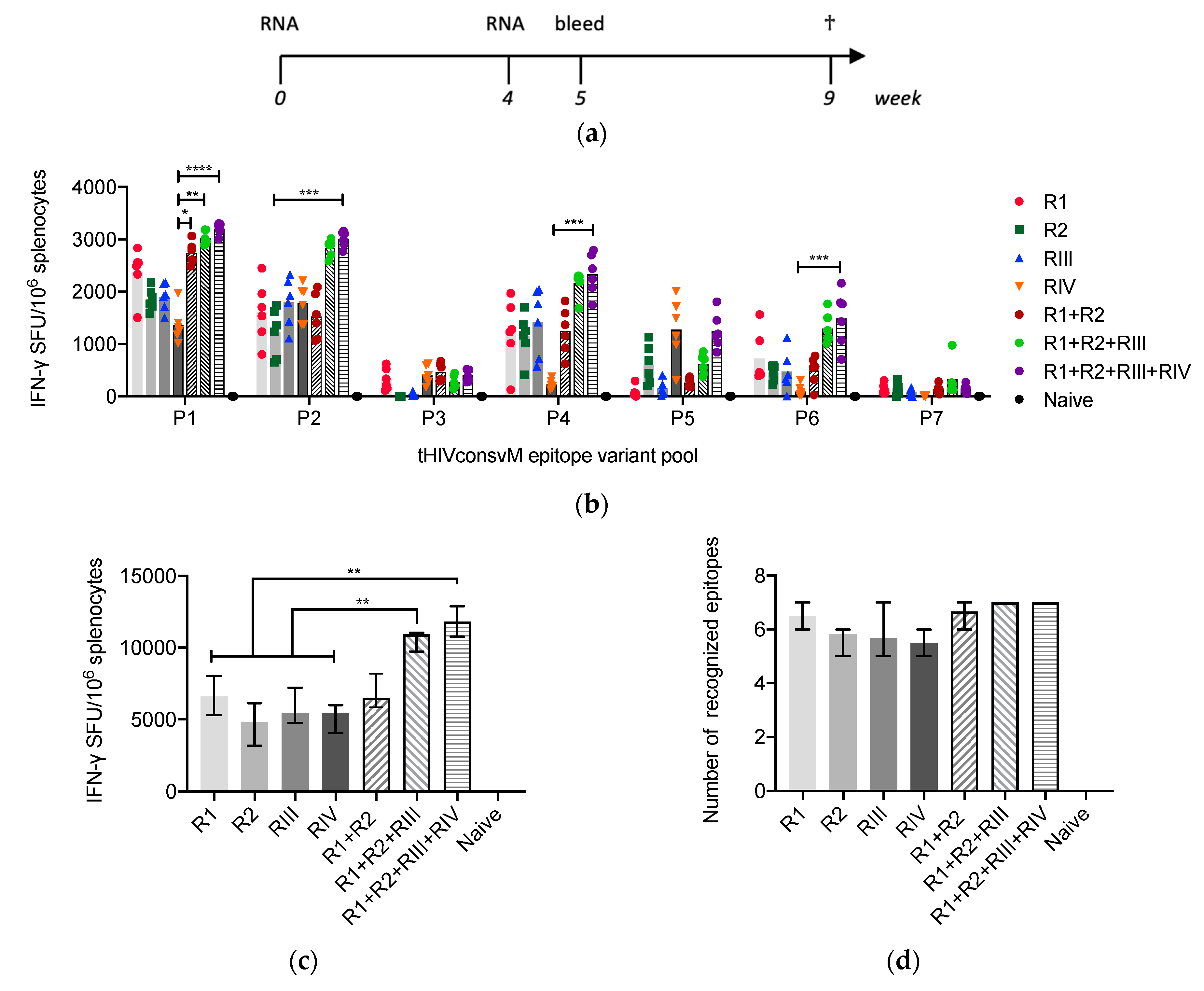
3.2. mRNA Induces Strong and Broad T-Cell Responses
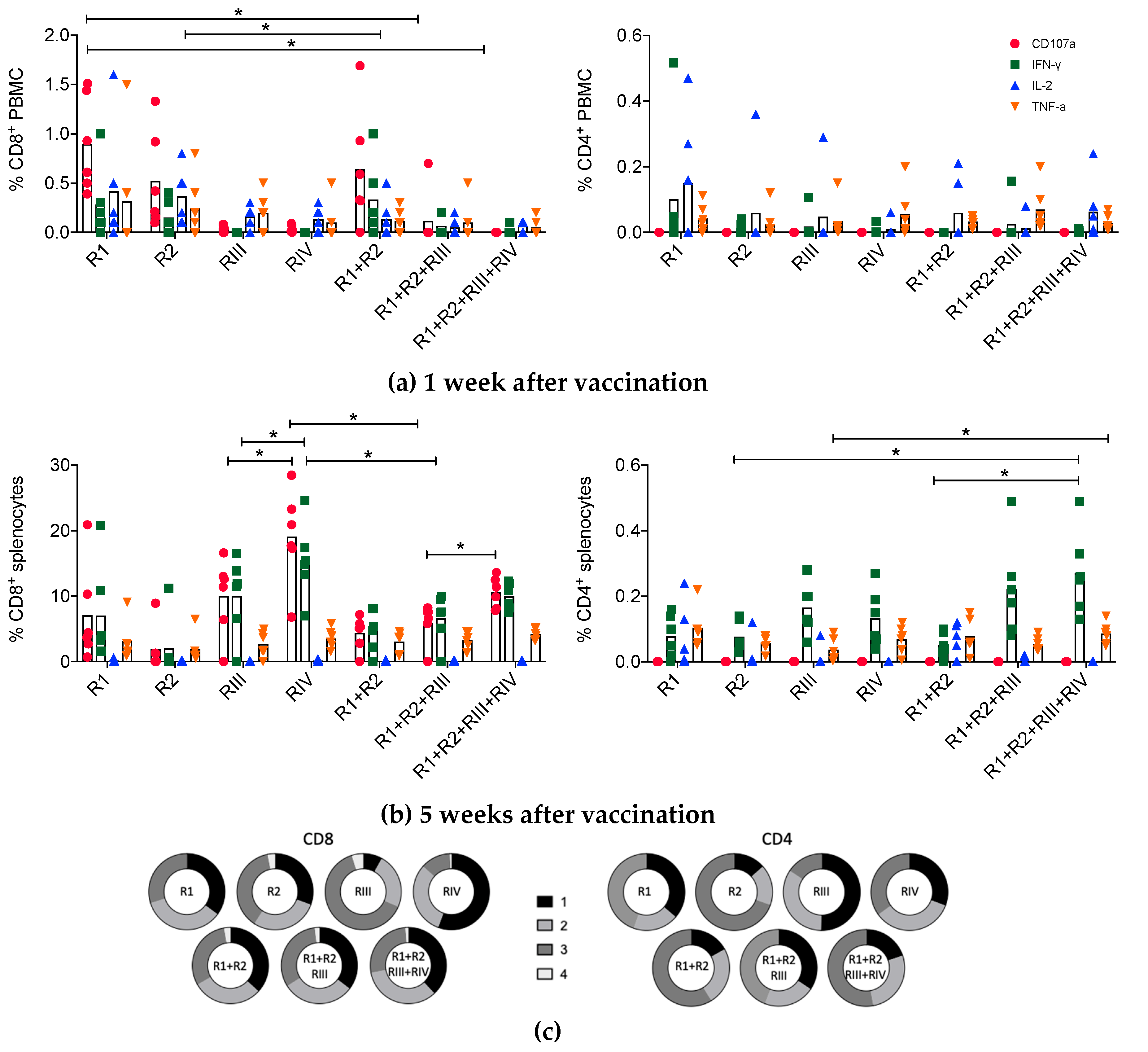
3.3. Kinetics and Quality of the HIVconsvM mRNA-Induced T-cell Responses
4. Discussion and Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Frahm, N.; Kiepiela, P.; Adams, S.; Linde, C.H.; Hewitt, H.S.; Sango, K.; E Feeney, M.; Addo, M.M.; Lichterfeld, M.; Lahaie, M.P.; et al. Control of human immunodeficiency virus replication by cytotoxic T lymphocytes targeting subdominant epitopes. Nat. Immunol. 2006, 7, 173–178. [Google Scholar] [CrossRef] [PubMed]
- Mothe, B.; Llano, A.; Ibarrondo, J.; Daniels, M.; Miranda, C.; Zamarreño, J.; Bach, V.; Zuniga, R.; Pérez-Álvarez, S.; Berger, C.T.; et al. Definition of the viral targets of protective HIV-1-specific T cell responses. J. Transl. Med. 2011, 9, 208. [Google Scholar] [CrossRef]
- Murakoshi, H.; Akahoshi, T.; Koyanagi, M.; Chikata, T.; Naruto, T.; Maruyama, R.; Tamura, Y.; Ishizuka, N.; Gatanaga, H.; Oka, S.; et al. Clinical control of HIV-1 by cytotoxic T cells specific for multiple conserved epitopes. J. Virol. 2015, 89, 5330–5339. [Google Scholar] [CrossRef]
- Murakoshi, H.; Zou, C.; Kuse, N.; Akahoshi, T.; Chikata, T.; Gatanaga, H.; Oka, S.; Hanke, T.; Takiguchi, M. CD8+ T cells specific for conserved, cross-reactive Gag epitopes with strong ability to suppress HIV-1 replication. Retrovirology 2018, 15, 46. [Google Scholar] [CrossRef] [PubMed]
- Ondondo, B.; Murakoshi, H.; Clutton, G.; Abdul-Jawad, S.; Wee, E.G.-T.; Gatanaga, H.; Oka, S.; McMichael, A.J.; Takiguchi, M.; Korber, B.T.; et al. Novel Conserved-region T-cell Mosaic Vaccine With High Global HIV-1 Coverage Is Recognized by Protective Responses in Untreated Infection. Mol. Ther. 2016, 24, 832–842. [Google Scholar] [CrossRef] [PubMed]
- Zou, C.; Murakoshi, H.; Kuse, N.; Akahoshi, T.; Chikata, T.; Gatanaga, H.; Oka, S.; Hanke, T.; Takiguchi, M. Effective suppression of HIV-1 replication by cytotoxic T lymphocytes specific for Pol Epitopes in conserved mosaic vaccine immunogens. J. Virol. 2019, 93, e02142-18. [Google Scholar] [CrossRef] [PubMed]
- Goonetilleke, N.; Liu, M.K.; Salazar-Gonzalez, J.F.; Ferrari, G.; Giorgi, E.; Ganusov, V.; Keele, B.F.; Learn, G.H.; Turnbull, E.L.; Salazar, M.G.; et al. The first T cell response to transmitted/founder virus contributes to the control of acute viremia in HIV-1 infection. J. Exp. Med. 2009, 206, 1253–1272. [Google Scholar] [CrossRef]
- Phillips, R.E.; Jones, S.L.R.; Nixon, D.F.; Gotch, F.M.; Edwards, J.P.; Ogunlesi, A.O.; Elvin, J.G.; Rothbard, J.A.; Bangham, C.R.M.; Rizza, C.R.; et al. Human immunodeficiency virus genetic variation that can escape cytotoxic T cell recognition. Nat. 1991, 354, 453–459. [Google Scholar] [CrossRef]
- Haynes, B.F.; Shaw, G.M.; Korber, B.T.; Kelsoe, G.; Sodroski, J.; Hahn, B.H.; Borrow, P.; McMichael, A.J. HIV-host interactions: Implications for vaccine design. Cell Host Microbe 2016, 19, 292–303. [Google Scholar] [CrossRef]
- Walker, B.; McMichael, A. The T-cell response to HIV. Cold Spring Harb. Perspect. Med. 2012, 2, 1–19. [Google Scholar] [CrossRef]
- Borthwick, N.; Ahmed, T.; Ondondo, B.; Hayes, P.; Rose, A.; Ebrahimsa, U.; Hayton, E.-J.; Black, A.; Bridgeman, A.M.; Rosario, M.; et al. Vaccine-elicited human T cells recognizing conserved protein regions inhibit HIV-1. Mol. Ther. 2014, 22, 464–475. [Google Scholar] [CrossRef]
- Mothe, B.; Manzardo, C.; Sanchez-Bernabeu, A.; Coll, P.; Morón-López, S.; Puertas, M.C.; Rosas-Umbert, M.; Cobarsi, P.; Escrig, R.; Perez-Alvarez, N.; et al. Therapeutic vaccination refocuses T-cell responses towards conserved regions of HIV-1 in early treated individuals (BCN 01 study). EClinicalMedicine 2019, 11, 65–80. [Google Scholar] [CrossRef]
- Mothe, B.; Rosás-Umbert, M.; Coll, P.; Manzardo, C.; Puertas, M.C.; Morón-López, S.; Llano, A.; Miranda, C.; Cedeño, S.; López, M.; et al. HIVconsv vaccines and romidepsin in early-treated HIV-1-infected individuals: Safety, immunogenicity and effect on the viral reservoir (study BCN02). Front. Immunol. 2020, 11, 823. [Google Scholar] [CrossRef] [PubMed]
- Mutua, G.; Farah, B.; Langat, R.; Indangasi, J.; Ogola, S.; Onsembe, B.; Kopycinski, J.T.; Hayes, P.; Ashraf, A.; Borthwick, N.J.; et al. Broad HIV-1 inhibition in vitro by vaccine-elicited CD8+ T cells in African adults. Mol. Ther. Methods Clin. Dev. 2016, 3, 16061. [Google Scholar] [CrossRef] [PubMed]
- Hanke, T. Aiming for protective T-cell responses: A focus on the first generation conserved-region HIVconsv vaccines in preventive and therapeutic clinical trials. Expert Rev. Vaccines 2019, 18, 1029–1041. [Google Scholar] [CrossRef] [PubMed]
- Létourneau, S.; Im, E.-J.; Mashishi, T.; Brereton, C.; Bridgeman, A.M.; Yang, H.; Dorrell, L.; Dong, T.; Korber, B.T.; McMichael, A.J.; et al. Design and Pre-Clinical Evaluation of a Universal HIV-1 Vaccine. PLoS ONE 2007, 2, e984. [Google Scholar] [CrossRef] [PubMed]
- Moyo, N.; Vogel, A.B.; Buus, S.; Erbar, S.; Wee, E.G.; Sahin, U.; Hanke, T. Efficient induction of T cells against conserved HIV-1 regions by mosaic vaccines delivered as self-amplifying mRNA. Mol. Ther. Methods Clin. Dev. 2018, 12, 32–46. [Google Scholar] [CrossRef]
- Wee, E.G.; Ondondo, B.; Berglund, P.; Archer, J.; McMichael, A.J.; Baltimore, D.; Ter Meulen, J.H.; Hanke, T. HIV-1 conserved mosaics delivered by regimens with integration-deficient DC-targeting lentiviral vector induce robust T cells. Mol. Ther. 2017, 25, 494–503. [Google Scholar] [CrossRef]
- Pastor, F.; Berraondo, P.; Etxeberria, I.; Frederick, J.; Sahin, U.; Gilboa, E.; Melero, I. An RNA toolbox for cancer immunotherapy. Nat. Rev. Drug Discov. 2018, 17, 751–767. [Google Scholar] [CrossRef]
- Kaczmarek, J.C.; Kowalski, P.; Anderson, D.G. Advances in the delivery of RNA therapeutics: From concept to clinical reality. Genome Med. 2017, 9, 60. [Google Scholar] [CrossRef]
- Richner, J.M.; Himansu, S.; Dowd, K.A.; Butler, S.L.; Salazar, V.; Fox, J.M.; Julander, J.G.; Tang, W.W.; Shresta, S.; Pierson, T.C.; et al. Modified mRNA vaccines protect against zika virus infection. Cell 2017, 169, 176. [Google Scholar] [CrossRef] [PubMed]
- Theiler, J.; Yoon, H.; Yusim, K.; Picker, L.J.; Früh, K.; Korber, B. Epigraph: A vaccine design tool applied to an HIV therapeutic vaccine and a pan-filovirus vaccine. Sci. Rep. 2016, 6, 33987. [Google Scholar] [CrossRef]
- Ondondo, B.; Abdul-Jawad, S.; Roshorm, Y.; Bridgeman, A.; Hanke, T. Vector delivery-dependant effect of human tissue plasminogen activator signal peptide on vaccine induction of T cells. J. HIV AIDS 2016, 2, 2. [Google Scholar] [CrossRef]
- Sabnis, S.; Kumarasinghe, E.S.; Salerno, T.; Mihai, C.; Ketova, T.; Senn, J.J.; Lynn, A.; Bulychev, A.; McFadyen, I.J.; Chan, J.; et al. A novel amino lipid series for mRNA delivery: Improved endosomal escape and sustained pharmacology and safety in non-human primates. Mol. Ther. 2018, 26, 1509–1519. [Google Scholar] [CrossRef] [PubMed]
- Fischer, W.; Perkins, S.; Theiler, J.; Bhattacharya, T.; Yusim, K.; Funkhouser, R.; Kuiken, C.; Haynes, B.; Letvin, N.L.; Walker, B.D.; et al. Polyvalent vaccines for optimal coverage of potential T-cell epitopes in global HIV-1 variants. Nat. Med. 2007, 13, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Los Alamos National Laboratory HIV Molecular Immunoloy Database. 2019. Available online: www.hiv.lanl.gov (accessed on 9 September 2017).

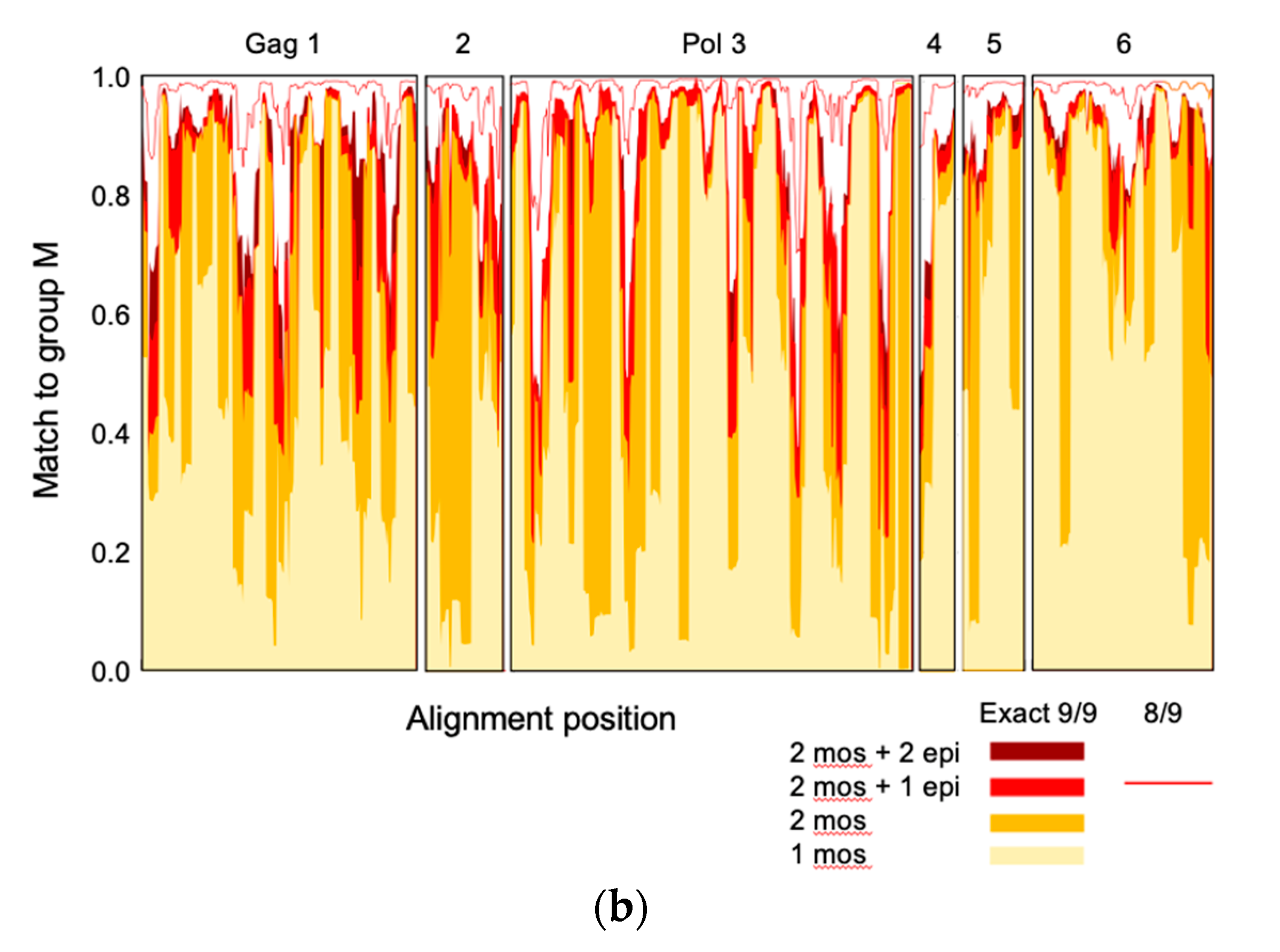
| Peptide Pools | Peptides | Vaccine Component |
|---|---|---|
| P1 | VLVGPTPVNI | Mosaic 1 |
| VLIGPTPVNI | Mosaic 2 | |
| VLVGPTPINI | Epigraph III | |
| VLVGPTPANI | Epigraph IV | |
| P2 | AMQMLKDTI | Mosaic 1 |
| AMQMLKETI | Mosaic 2 | |
| AMQILKDTI | Epigraph III | |
| AMQILKETI | Epigraph IV | |
| P3 | IFQSSMTKI | Mosaic 1 |
| IFQCSMTKI | Mosaic 2 | |
| IFQSSMTRI | Epigraph III | |
| IFQASMTKI | Epigraph IV | |
| P4 | SPAIFQSSM | Mosaic 1 |
| SPAIFQCSM | Mosaic 2 | |
| SPAIFQASM | Epigraph III | |
| SPSIFQSSM | Epigraph IV | |
| P5 | REHLLKWGF | Mosaic 1 |
| RQHLLRWGF | Mosaic 2 | |
| RAHLLSWGF | Epigraph III | |
| RQHLLKWGF | Epigraph IV | |
| P6 | ITKIQNFRVYY | Mosaic 1 |
| IIKIQNFRVYY | Mosaic 2 | |
| IIKVQNFRVYF | Epigraph III | |
| ITKLQNFRVYY | Epigraph IV | |
| P7 | VYYRDSRDPI | Mosaic 1 |
| VYYRDSRDPL | Mosaic 2 | |
| VYYRDNRDPL | Epigraph III | |
| VYFRDSRDPV | Epigraph IV |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moyo, N.; Wee, E.G.; Korber, B.; Bahl, K.; Falcone, S.; Himansu, S.; Wong, A.L.; Dey, A.K.; Feinberg, M.; Hanke, T. Tetravalent Immunogen Assembled from Conserved Regions of HIV-1 and Delivered as mRNA Demonstrates Potent Preclinical T-Cell Immunogenicity and Breadth. Vaccines 2020, 8, 360. https://doi.org/10.3390/vaccines8030360
Moyo N, Wee EG, Korber B, Bahl K, Falcone S, Himansu S, Wong AL, Dey AK, Feinberg M, Hanke T. Tetravalent Immunogen Assembled from Conserved Regions of HIV-1 and Delivered as mRNA Demonstrates Potent Preclinical T-Cell Immunogenicity and Breadth. Vaccines. 2020; 8(3):360. https://doi.org/10.3390/vaccines8030360
Chicago/Turabian StyleMoyo, Nathifa, Edmund G. Wee, Bette Korber, Kapil Bahl, Samantha Falcone, Sunny Himansu, Adrianne L. Wong, Antu K. Dey, Mark Feinberg, and Tomáš Hanke. 2020. "Tetravalent Immunogen Assembled from Conserved Regions of HIV-1 and Delivered as mRNA Demonstrates Potent Preclinical T-Cell Immunogenicity and Breadth" Vaccines 8, no. 3: 360. https://doi.org/10.3390/vaccines8030360
APA StyleMoyo, N., Wee, E. G., Korber, B., Bahl, K., Falcone, S., Himansu, S., Wong, A. L., Dey, A. K., Feinberg, M., & Hanke, T. (2020). Tetravalent Immunogen Assembled from Conserved Regions of HIV-1 and Delivered as mRNA Demonstrates Potent Preclinical T-Cell Immunogenicity and Breadth. Vaccines, 8(3), 360. https://doi.org/10.3390/vaccines8030360





