Seroprevalence of Neutralizing Antibodies against Japanese Encephalitis Virus among Adolescents and Adults in Korea: A Prospective Multicenter Study
Abstract
1. Introduction
2. Materials and Methods
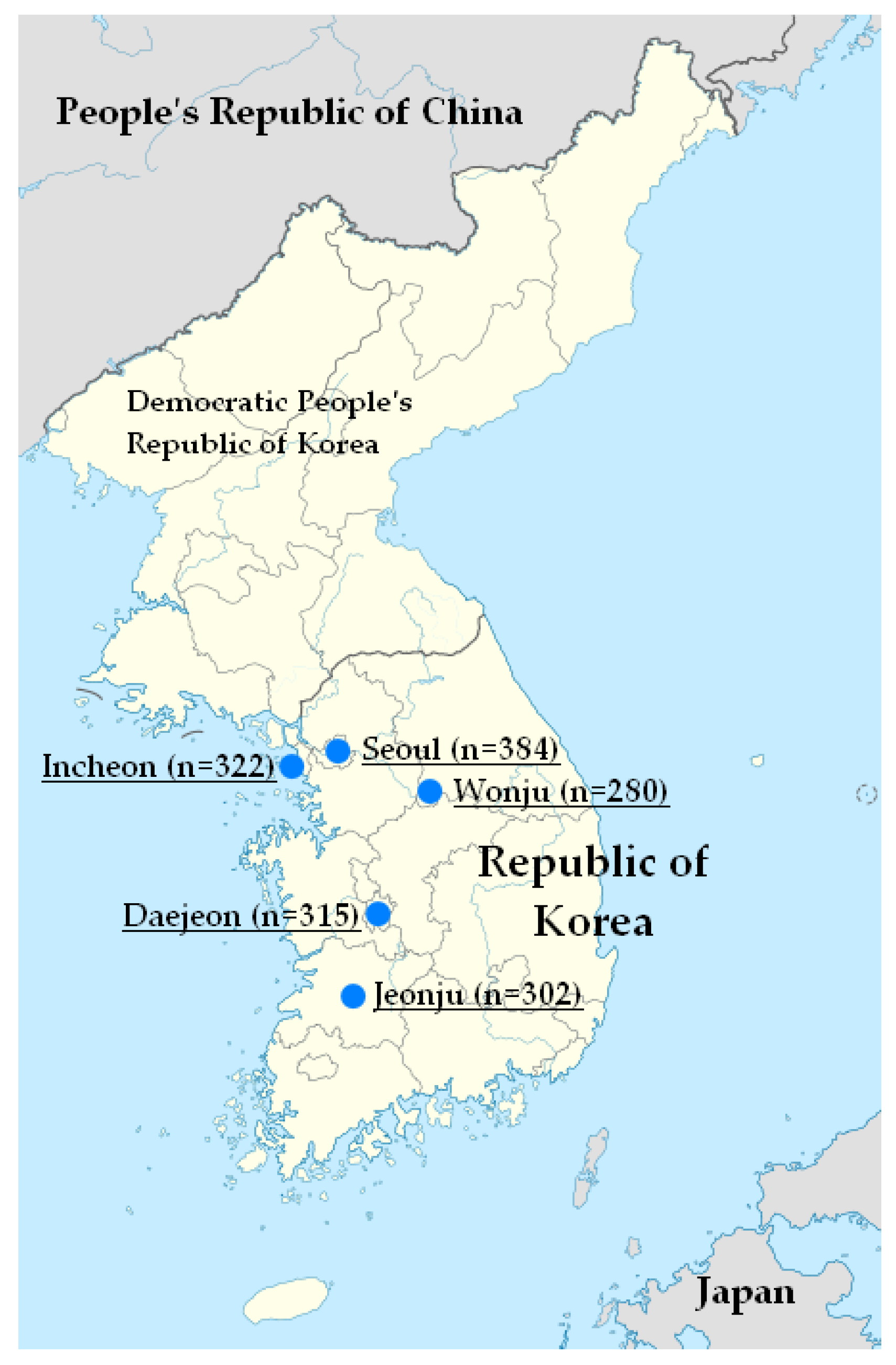
2.1. Study Design and Participants
2.2. Neutralizing Antibody Titers Using Pseudotyped Virus Assay
2.3. Statistical Analyses
3. Results
3.1. Study Population
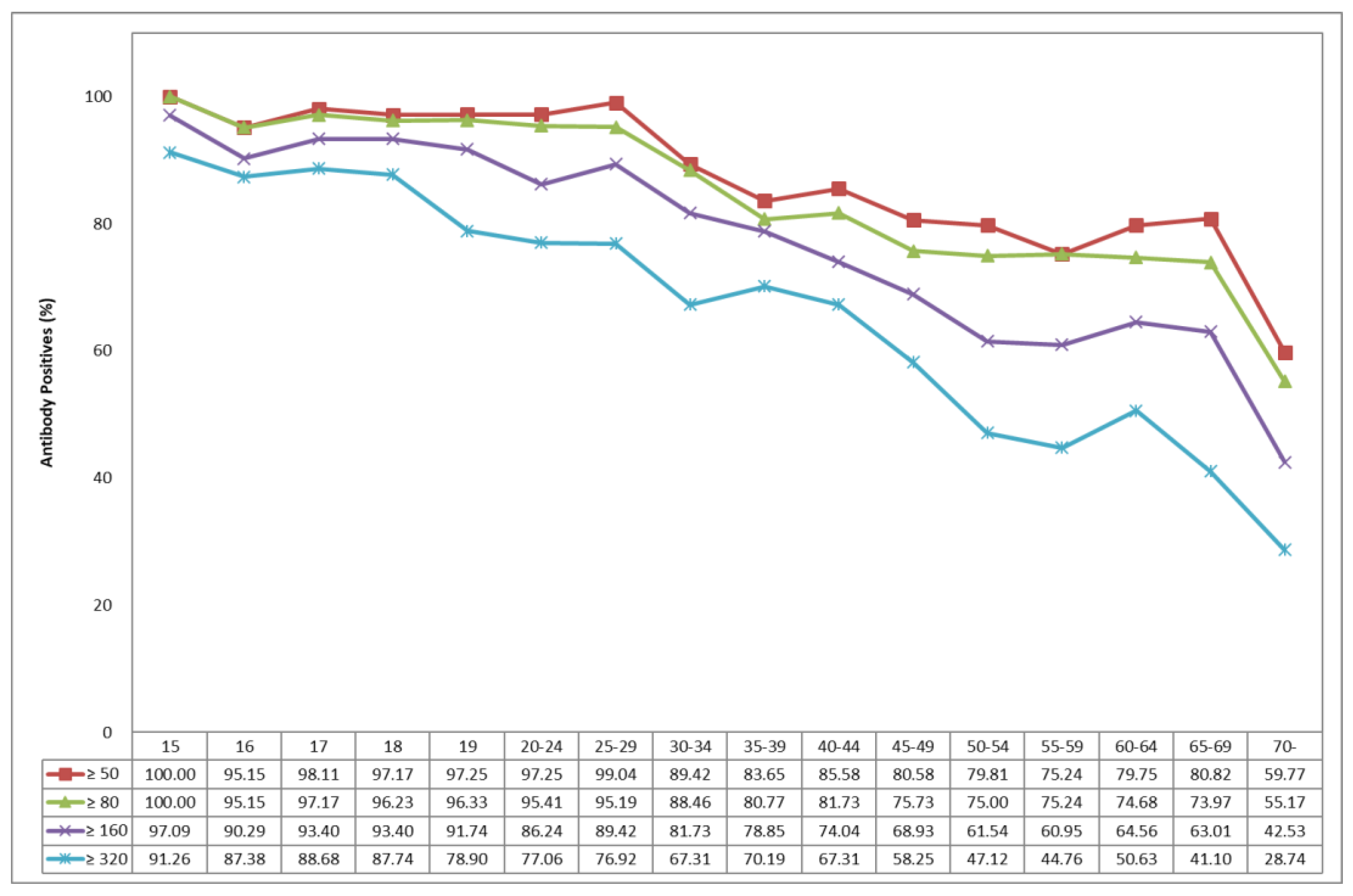
3.2. Seropositive Rates of JE-NTAb Using PV Assay
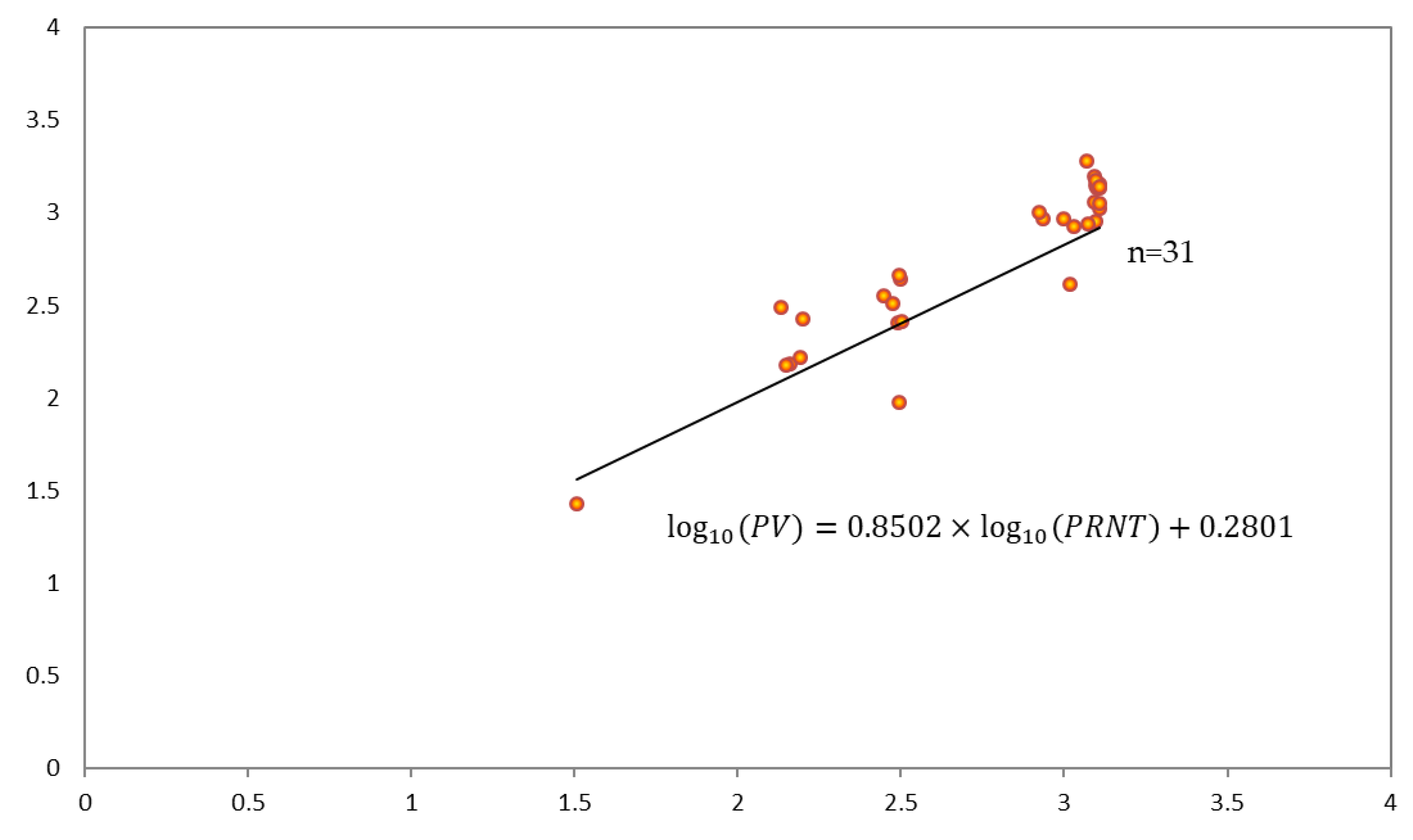
3.3. Model for Validation between PV Assay and PRNT
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Halstead, S.B.; Hills, S.L.; Dubischar, K. Japanese encephalitis vaccines. In Plotkin’s Vaccines, 7th ed.; Plotkin, S.A., Orentein, W.A., Offit, P.A., Edwards, K.M., Eds.; Elsevier: Philadelphia, PA, USA, 2018; pp. 511–548. [Google Scholar]
- Solomon, T. Control of Japanese encephalitis-within our grasp? N. Engl. J. Med. 2006, 355, 869–871. [Google Scholar] [CrossRef] [PubMed]
- Department of Health. Japanese encephalitis. In Immunisation Against Infectious Disease, 3rd ed.; Salisbury, D., Ramsay, M., Moakes, K., Eds.; Crown: London, UK, 2006; pp. 201–208. [Google Scholar]
- Grascenkov, N.I. Japanese encephalitis in the USSR. Bull. World Health Org. 1964, 30, 161–172. [Google Scholar] [PubMed]
- The Korean Pediatric Society. Japanese encephalitis vaccines. In Immunization Guideline, 9th ed.; Kim, J.H., Ed.; The Korean Pediatric Society: Seoul, Korea, 2018; pp. 186–202. [Google Scholar]
- Lee, E.J.; Cha, G.W.; Ju, Y.R.; Han, M.G.; Lee, W.J.; Jeong, Y.E. Prevalence of neutralizing antibodies to Japanese encephalitis virus among high-risk age groups in South Korea, 2010. PLoS ONE 2016, 11, e0147841. [Google Scholar]
- Sohn, Y.M. Japanese encephalitis immunization in South Korea: Past, present, and future. Emerg. Infect. Dis. 2000, 6, 17–24. [Google Scholar] [PubMed]
- Sunwoo, J.S.; Jung, K.H.; Lee, S.T.; Lee, S.K.; Chu, K. Reemergence of Japanese encephalitis in South Korea, 2010–2015. Emerg. Infect. Dis. 2016, 22, 1841–1843. [Google Scholar] [CrossRef] [PubMed]
- Arai, S.; Matsunaga, Y.; Takasaki, T.; Tanaka-Taya, K.; Taniguchi, K.; Okabe, N.; Kurane, I. Vaccine Preventable Diseases Surveillance Program of Japan. Japanese encephalitis: Surveillance and elimination effort in Japan from 1982 to 2004. Jpn. J. Infect. Dis. 2008, 61, 333–338. [Google Scholar] [PubMed]
- Hsu, L.C.; Chen, Y.J.; Hsu, F.K.; Huang, J.H.; Chang, C.M.; Chou, P.; Lin, I.F.; Chang, F.Y. The incidence of Japanese encephalitis in Taiwan—A population-based study. PLoS Negl. Trop. Dis. 2014, 8, e3030. [Google Scholar] [CrossRef] [PubMed]
- Tseng, H.F.; Tan, H.F.; Chang, C.K.; Huang, W.L.; Ho, W.C. Seroepidemiology study of Japanese encephalitis neutralizing antibodies in southern Taiwan: A comparative study between urban city and country townships. Am. J. Infect. Control. 2003, 31, 435–440. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.H.; Hong, Y.J.; Lee, H.J.; Choi, B.Y.; Kim, C.H.; Park, J.O.; Kang, J.H.; Choi, B.J.; Kim, J.H.; Ahn, Y.M.; et al. Immunogenicity and protective effectiveness of Japanese encephalitis vaccine: A prospective multicenter cohort study. Korean. J. Pediatr. Infect. Dis. 2013, 20, 131–138. [Google Scholar] [CrossRef]
- Yun, K.W.; Lee, H.J.; Kang, J.H.; Eun, B.W.; Kim, Y.J.; Kim, K.H.; Kim, N.H.; Hong, Y.J.; Kim, D.H.; Kim, H.M.; et al. Safety and immunogenicity of a freeze-dried, Vero cell culture-derived, inactivated Japanese encephalitis vaccine (KD-287, ENCEVAC®) versus a mouse brain-derived inactivated Japanese encephalitis vaccine in children: A phase III, multicenter, double-blinded, randomized trial. BMC Infect. Dis. 2015, 8, 7. [Google Scholar]
- Choe, Y.J.; Taurel, A.F.; Nealon, J.; Seo, H.S.; Kim, H.S. Systematic review of seroepidemiological studies on Japanese encephalitis in the Republic of Korea. Int. J. Infect. Dis. 2018, 67, 14–19. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Min, H.I.; Park, K.H.; Choi, H.J.; Kim, M.K.; Ahn, C.Y.; Hong, Y.J.; Kim, Y.B. Comparison of JEV neutralization assay using pseudotyped JEV with the conventional plaque-reduction neutralization test. J. Microbiol. 2014, 52, 435–440. [Google Scholar] [CrossRef] [PubMed]
- Deuel, Jr. R.E.; Barwell, M.B.; Matumoto, M.; Sabain, A.B. Status and significance of inapparent infection with virus of Japanese B encephalitis in Korean and Okinawa in 1946. Am. J. Hyg. 1950, 51, 13–20. [Google Scholar] [PubMed]
- Kim, K.H. Recent epidemiological features of Japanese encephalitis in the Republic of Korea. Korean J. Infect. Dis. 1974, 6, 83–88. [Google Scholar]
- Kim, S.O.; Lee, Y.J.; Jung, B.K.; Lee, H.W. The distribution of hemagglutination inhibition antibody for Japanese encephalitis in residents of Seoul areas. Choeshin Ui-hak 1976, 19, 21–26. [Google Scholar]
- Lee, Y.T.; Lee, C.H. Serological study on Japanese encephalitis in Koreans and fowls, 1975. Korean J. Infect. Dis. 1976, 8, 75–82. [Google Scholar]
- Lee, Y.T.; Lee, C.H. The distribution of hemagglutination inhibition antibodies for Japanese encephalitis virus against the Koreas 1976. J. Korean Soc. Microbiol. 1977, 12, 51–56. [Google Scholar]
- Lee, C.H.; Lee, Y.T.; Ko, K.K. The distribution of hemagglutination inhibition antibodies for Japanese encephalitis virus in Korea (1977, 1978). Choeshin Ui-hak 1979, 113–119. [Google Scholar]
- Lee, C.H.; Lee, Y.T.; Ko, K.j.; Moon, K.S.; Kim, O.J. The distribution of hemagglutination inhibition antibodies for Japanese encephalitis virus in Koreans 1979. Korean J. Virol. 1980, 10, 65–69. [Google Scholar]
- Lee, Y.T.; Youm, B.J. A hemagglutination inhibition antibody test for Japanese encephalitis virus among the Koreans, 1984–1985. Korean J. Virol. 1985, 15, 1–9. [Google Scholar]
| C | Provinces | |||||
|---|---|---|---|---|---|---|
| Seoul | Wonju | Incheon | Jeonju | Daejeon | Total | |
| N = 384 | N = 280 | N = 322 | N = 302 | N = 315 | N = 1603 | |
| 15 years | 24 | 20 | 20 | 21 | 18 | 103 |
| F | 12 | 10 | 4 | 11 | 9 | 46 |
| M | 12 | 10 | 16 | 10 | 9 | 57 |
| 16 years | 24 | 20 | 20 | 22 | 17 | 103 |
| F | 12 | 10 | 10 | 10 | 8 | 50 |
| M | 12 | 10 | 10 | 12 | 9 | 53 |
| 17 years | 24 | 20 | 20 | 23 | 19 | 106 |
| F | 12 | 10 | 9 | 10 | 9 | 50 |
| M | 12 | 10 | 11 | 13 | 10 | 56 |
| 18 years | 24 | 20 | 20 | 23 | 19 | 106 |
| F | 12 | 10 | 5 | 11 | 10 | 48 |
| M | 12 | 10 | 15 | 12 | 9 | 58 |
| 19 years | 24 | 20 | 20 | 22 | 23 | 109 |
| F | 12 | 10 | 9 | 12 | 11 | 54 |
| M | 12 | 10 | 11 | 10 | 12 | 55 |
| 20–24 years | 24 | 20 | 20 | 25 | 20 | 109 |
| F | 12 | 10 | 4 | 10 | 10 | 46 |
| M | 12 | 10 | 16 | 15 | 10 | 63 |
| 25–29 years | 24 | 20 | 20 | 20 | 20 | 104 |
| F | 12 | 10 | 5 | 10 | 10 | 47 |
| M | 12 | 10 | 15 | 10 | 10 | 57 |
| 30–34 years | 24 | 20 | 20 | 20 | 20 | 104 |
| F | 12 | 10 | 11 | 10 | 10 | 53 |
| M | 12 | 10 | 9 | 10 | 10 | 51 |
| 35–39 years | 24 | 20 | 20 | 20 | 20 | 104 |
| F | 12 | 10 | 10 | 10 | 10 | 52 |
| M | 12 | 10 | 10 | 10 | 10 | 52 |
| 40–44 years | 24 | 20 | 20 | 20 | 20 | 104 |
| F | 12 | 10 | 9 | 10 | 10 | 51 |
| M | 12 | 10 | 11 | 10 | 10 | 53 |
| 45–49 years | 24 | 20 | 20 | 20 | 19 | 103 |
| F | 12 | 10 | 10 | 10 | 9 | 51 |
| M | 12 | 10 | 10 | 10 | 10 | 52 |
| 50–54 years | 24 | 20 | 20 | 20 | 20 | 104 |
| F | 12 | 10 | 10 | 10 | 10 | 52 |
| M | 12 | 10 | 10 | 10 | 10 | 52 |
| 55–59 years | 25 | 20 | 20 | 20 | 20 | 105 |
| F | 12 | 10 | 13 | 10 | 10 | 55 |
| M | 13 | 10 | 7 | 10 | 10 | 50 |
| 60–64 years | 24 | 8 | 20 | 6 | 21 | 79 |
| F | 12 | 3 | 8 | 3 | 10 | 36 |
| M | 12 | 5 | 12 | 3 | 11 | 43 |
| 65–69 years | 23 | 2 | 20 | 9 | 19 | 73 |
| F | 12 | 2 | 9 | 4 | 10 | 37 |
| M | 11 | 0 | 11 | 5 | 9 | 36 |
| ≥70 years | 24 | 10 | 22 | 11 | 20 | 87 |
| F | 12 | 5 | 10 | 5 | 10 | 42 |
| M | 12 | 5 | 12 | 6 | 10 | 45 |
| Total | 384 | 280 | 322 | 302 | 315 | 1603 |
| F | 192 | 140 | 136 | 146 | 156 | 770 |
| M | 192 | 140 | 186 | 156 | 159 | 833 |
| Age (Years) | Seoul | Wonju | Incheon | Jeonju | Daejeon |
|---|---|---|---|---|---|
| 15–19 | |||||
| Number of participants | 120 | 100 | 100 | 111 | 96 |
| Mean NTAb titer | 1424.51 | 923.92 | 822.68 | 820.33 | 947.22 |
| (95% CI) | (1250.43, 1622.82) | (758.26, 1125.77) | (690.26, 980.52) | (664.26, 1013.07) | (756.54, 1185.95) |
| 20–44 | |||||
| Number of participants | 120 | 100 | 100 | 105 | 100 |
| Mean NTAb titer | 871.94 | 1009.5 | 229.8 | 537.13 | 465.45 |
| (95% CI) | (711.68, 1068.29) | (885.24, 1151.77) | (173.53, 304.30) | (413.71, 697.37) | (365.29, 592.45) |
| ≥45 | |||||
| Number of participants | 144 | 80 | 122 | 86 | 119 |
| Mean NTAb titer | 429.26 | 402.81 | 193.12 | 111.99 | 130.24 |
| (95% CI) | (340.38, 541.36) | (318.22, 509.89) | (151.30, 246.49) | (86.64, 144.77) | (105.43, 160.89) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kwak, B.O.; Kwon, Y.S.; Hong, Y.J.; Nahm, C.H.; Jang, W.; Uh, Y.; Cho, Y.G.; Kim, J.; Kim, M.; Kim, D.H. Seroprevalence of Neutralizing Antibodies against Japanese Encephalitis Virus among Adolescents and Adults in Korea: A Prospective Multicenter Study. Vaccines 2020, 8, 328. https://doi.org/10.3390/vaccines8020328
Kwak BO, Kwon YS, Hong YJ, Nahm CH, Jang W, Uh Y, Cho YG, Kim J, Kim M, Kim DH. Seroprevalence of Neutralizing Antibodies against Japanese Encephalitis Virus among Adolescents and Adults in Korea: A Prospective Multicenter Study. Vaccines. 2020; 8(2):328. https://doi.org/10.3390/vaccines8020328
Chicago/Turabian StyleKwak, Byung Ok, Young Se Kwon, Young Jin Hong, Chung Hyun Nahm, Woori Jang, Young Uh, Yong Gon Cho, Jimyung Kim, Myungshin Kim, and Dong Hyun Kim. 2020. "Seroprevalence of Neutralizing Antibodies against Japanese Encephalitis Virus among Adolescents and Adults in Korea: A Prospective Multicenter Study" Vaccines 8, no. 2: 328. https://doi.org/10.3390/vaccines8020328
APA StyleKwak, B. O., Kwon, Y. S., Hong, Y. J., Nahm, C. H., Jang, W., Uh, Y., Cho, Y. G., Kim, J., Kim, M., & Kim, D. H. (2020). Seroprevalence of Neutralizing Antibodies against Japanese Encephalitis Virus among Adolescents and Adults in Korea: A Prospective Multicenter Study. Vaccines, 8(2), 328. https://doi.org/10.3390/vaccines8020328





