Responsiveness to Influenza Vaccination Correlates with NKG2C-Expression on NK Cells
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Humoral Immune Responses
2.3. Cellular Immune Responses
2.4. Stochastic Neighbor Embedding (SNE) Analysis
2.5. Statistical Analysis
3. Results
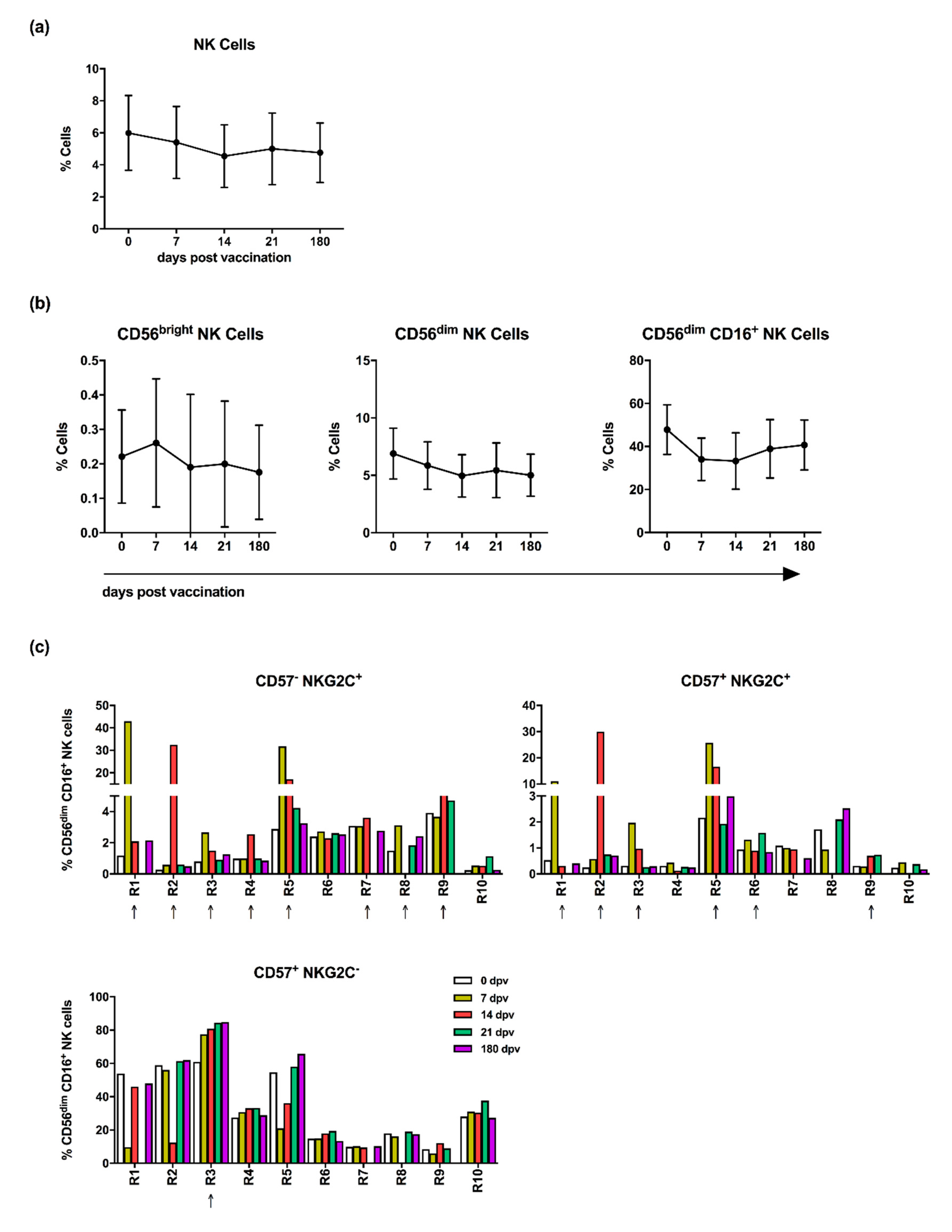
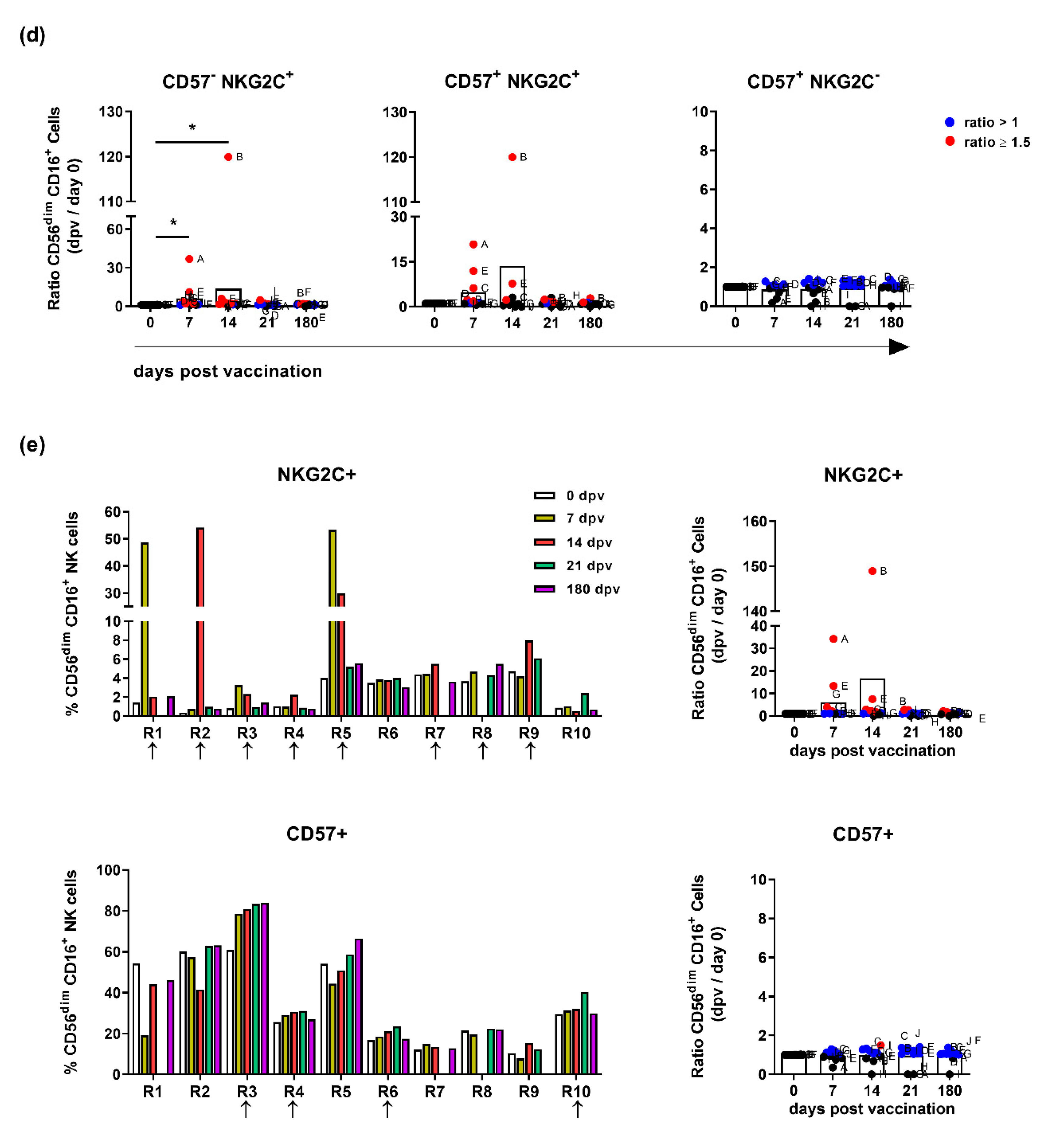
3.1. Influenza Vaccination Leads to Enhanced Frequencies of NKG2C-Expressing NK Cells
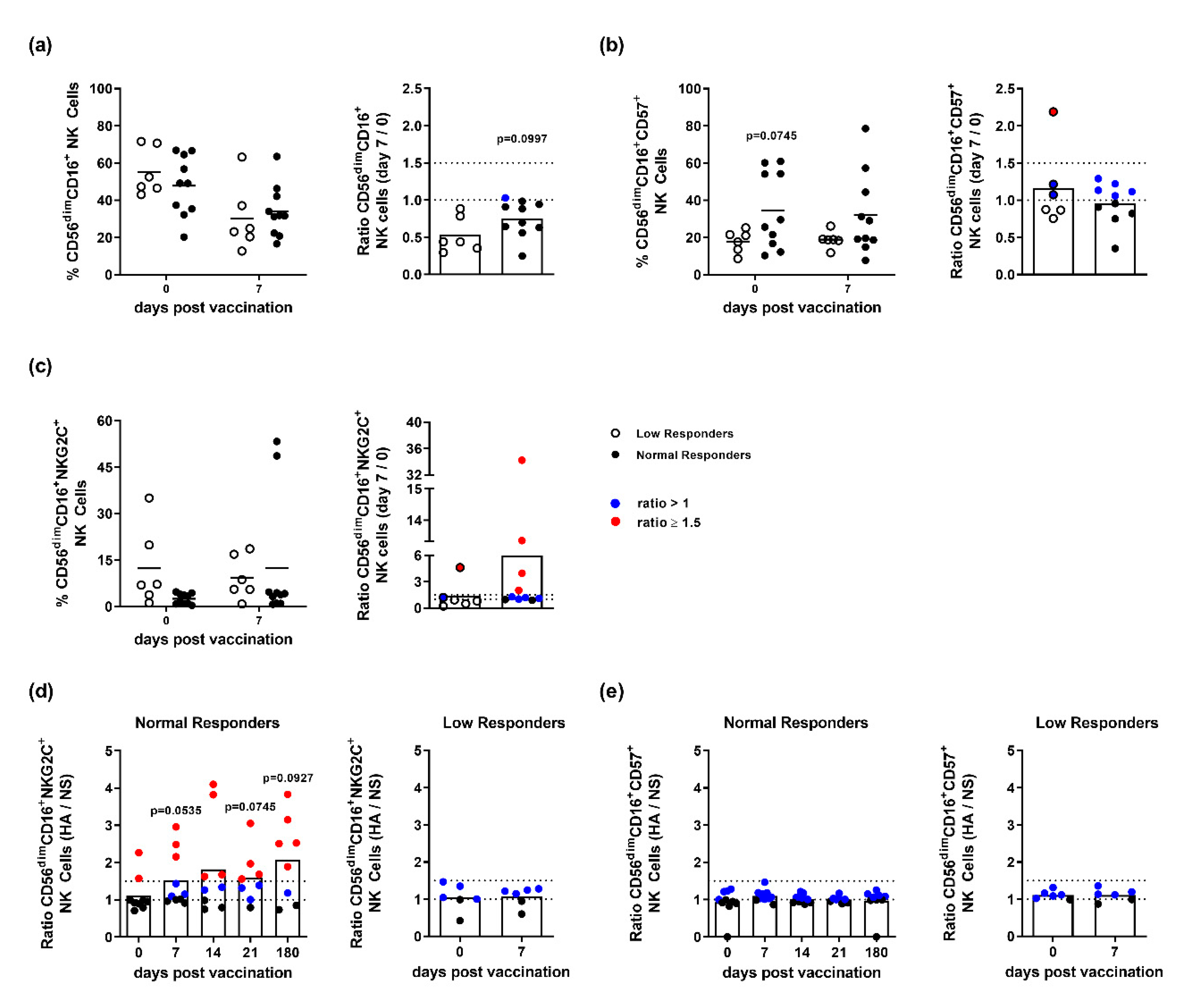
3.2. NK Cells of Influenza Vaccination Responders and Low Responders Display Differences in NKG2C and CD57 Expression
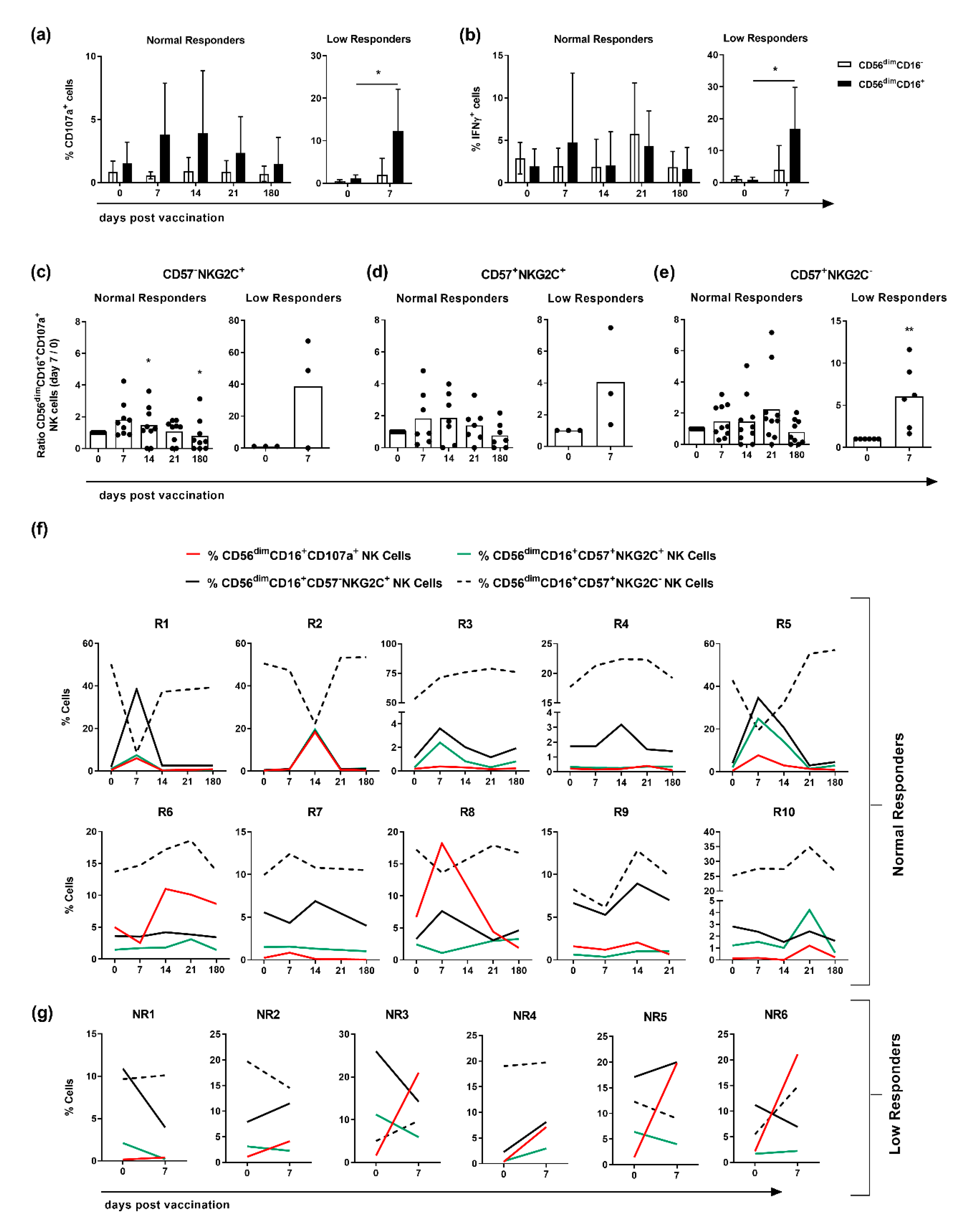
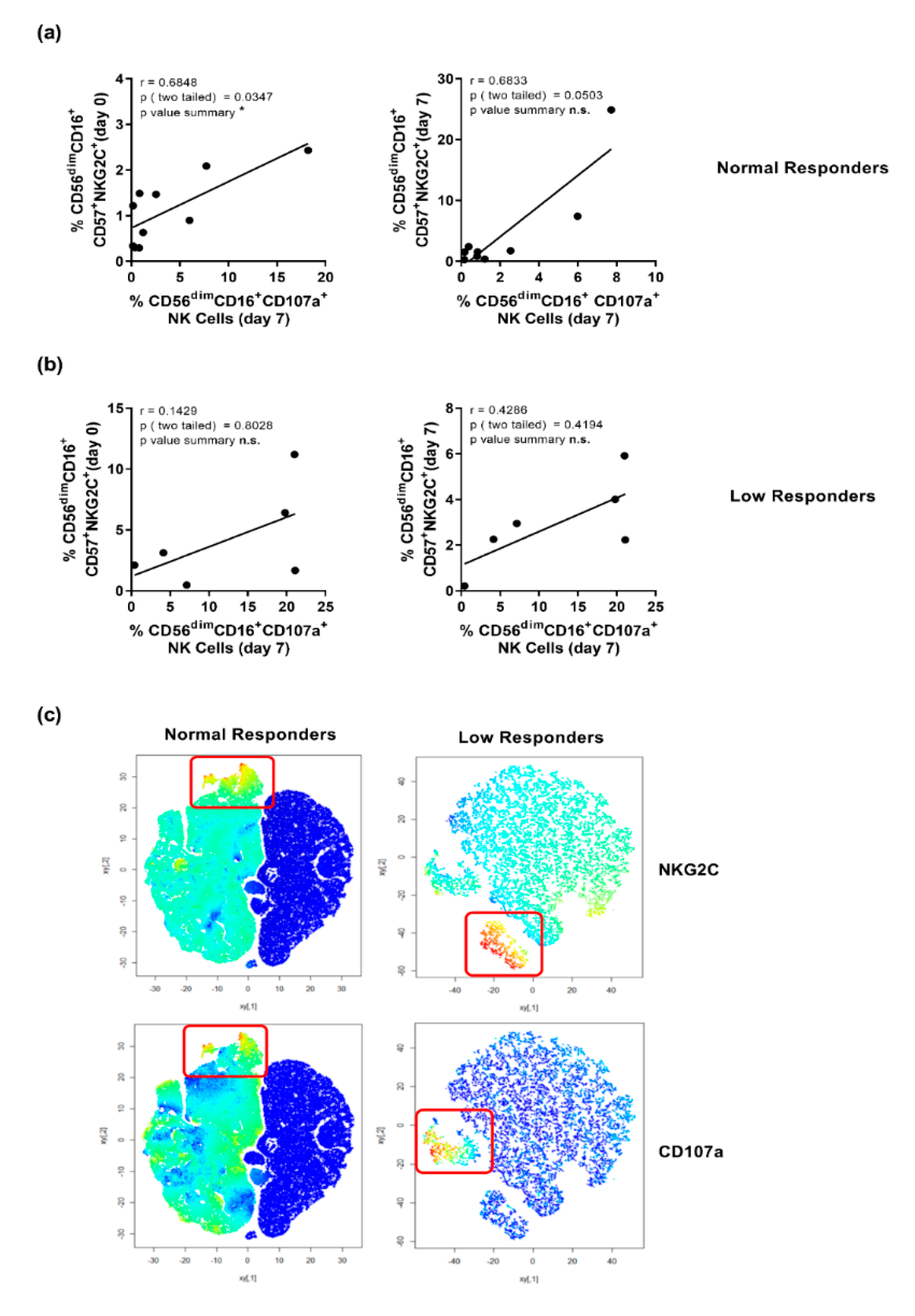
3.3. CD107a Expression Is Confined to CD56dimCD16+NKG2C-Expressing NK Cells in Responders but Not in Low Responders
3.4. Elevated NKG2C-Expression Correlates with a Higher Expression of CD107a
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- WHO. Fact Sheet: Influenza (Seasonal). Available online: https://www.who.int/news-room/fact-sheets/detail/influenza-(seasonal) (accessed on 17 January 2020).
- EMA. Vaccines for Pandemic Influenza. Available online: https://www.ema.europa.eu/en/human-regulatory/overview/public-health-threats/pandemic-influenza/vaccines-pandemic-influenza (accessed on 17 January 2020).
- De Jong, J.C.; Palache, A.M.; Beyer, W.E.P.; Rimmelzwaan, G.F.; Boon, A.C.M.; Osterhaus, A.D.M.E. Haemagglutination-inhibiting antibody to influenza virus. Dev. Biol. 2003, 115, 63–73. [Google Scholar]
- Beyer, W.E.; Palache, A.M.; Lüchters, G.; Nauta, J.; Osterhaus, A.D.M.E. Seroprotection rate, mean fold increase, seroconversion rate: Which parameter adequately expresses seroresponse to influenza vaccination? Virus Res. 2004, 103, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Schultz-Cherry, S. Role of NK cells in influenza infection. Curr. Top. Microbiol. Immunol. 2015, 386, 109–120. [Google Scholar] [PubMed]
- Freud, A.G.; Yokohama, A.; Becknell, B.; Lee, M.T.; Mao, H.C.; Ferketich, A.K.; Caligiuri, M.A. Evidence for discrete stages of human natural killer cell differentiation in vivo. J. Exp. Med. 2006, 203, 1033–1043. [Google Scholar] [CrossRef] [PubMed]
- Björkström, N.K.; Riese, P.; Heuts, F.; Andersson, S.; Fauriat, C.; Ivarsson, M.A.; Björklund, A.T.; Flodström-Tullberg, M.; Michaelsson, J.; Rottenberg, M.E.; et al. Expression patterns of NKG2A, KIR, and CD57 define a process of CD56dim NK-cell differentiation uncoupled from NK-cell education. Blood 2010, 116, 3853–3864. [Google Scholar] [CrossRef]
- Luetke-Eversloh, M.; Killig, M.; Romagnani, C. Signatures of human NK cell development and terminal differentiation. Front. Immunol. 2013, 4, 499. [Google Scholar] [CrossRef]
- Fauriat, C.; Ivarsson, M.A.; Ljunggren, H.G.; Malmberg, K.J.; Michaelsson, J. Education of human natural killer cells by activating killer cell immunoglobulin-like receptors. Blood 2010, 115, 1166–1174. [Google Scholar] [CrossRef]
- Wagner, J.A.; Fehniger, T.A. Human Adaptive Natural Killer Cells: Beyond NKG2C. Trends Immunol. 2016, 37, 351–353. [Google Scholar] [CrossRef]
- Gabrielli, S.; Ortolani, C.; Del Zotto, G.; Luchetti, F.; Canonico, B.; Buccella, F.; Artico, M.; Papa, S.; Zamai, L. The Memories of NK Cells: Innate-Adaptive Immune Intrinsic Crosstalk. J. Immunol. Res. 2016, 2016, 1376595. [Google Scholar] [CrossRef]
- Paust, S.; Gill, H.S.; Wang, B.Z.; Flynn, M.P.; Moseman, E.A.; Senman, B.; Szczepanik, M.; Telenti, A.; Askenase, P.W.; Compans, R.W.; et al. Critical role for the chemokine receptor CXCR6 in NK cell-mediated antigen-specific memory of haptens and viruses. Nat. Immunol. 2010, 11, 1127–1135. [Google Scholar] [CrossRef]
- Marcus, A.; Raulet, D.H. Evidence for natural killer cell memory. Curr. Biol. 2013, 23, R817–R820. [Google Scholar] [CrossRef] [PubMed]
- Min-Oo, G.; Lanier, L.L. Cytomegalovirus generates long-lived antigen-specific NK cells with diminished bystander activation to heterologous infection. J. Exp. Med. 2014, 211, 2669–2680. [Google Scholar] [CrossRef] [PubMed]
- Cooper, M.A.; Elliott, J.M.; Keyel, P.A.; Yang, L.; Carrero, J.A.; Yokoyama, W.M. Cytokine-induced memory-like natural killer cells. Proc. Natl. Acad. Sci. USA 2009, 106, 1915–1919. [Google Scholar] [CrossRef] [PubMed]
- Gumá, M.; Angulo, A.; Vilches, C.; Gómez-Lozano, N.; Malats, N.; López-Botet, M. Imprint of human cytomegalovirus infection on the NK cell receptor repertoire. Blood 2004, 104, 3664–3671. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Verges, S.; Milush, J.M.; Schwarzt, B.S.; Pando, M.J.; Jarjoura, J.; York, V.A.; Houchins, J.P.; Miller, S.; Kang, S.M.; Norris, P.J.; et al. Expansion of a unique CD57(+)NKG2Chi natural killer cell subset during acute human cytomegalovirus infection. Proc. Natl. Acad. Sci. USA 2011, 108, 14725–14732. [Google Scholar] [CrossRef] [PubMed]
- Foley, B.; Cooley, S.; Verneris, M.R.; Curtsinger, J.; Luo, X.; Waller, E.K.; Anasetti, C.; Weisdorf, D.; Miller, J.S. Human cytomegalovirus (CMV)-induced memory-like NKG2C(+) NK cells are transplantable and expand in vivo in response to recipient CMV antigen. J. Immunol. 2012, 189, 5082–5088. [Google Scholar] [CrossRef]
- Romee, R.; Schneider, S.E.; Leong, J.W.; Chase, J.M.; Keppel, C.R.; Sullivan, R.P.; Cooper, M.A.; Fehniger, T.A. Cytokine activation induces human memory-like NK cells. Blood 2012, 120, 4751–4760. [Google Scholar] [CrossRef]
- Berrien-Elliott, M.M.; Wagner, J.A.; Fehniger, T.A. Human Cytokine-Induced Memory-Like Natural Killer Cells. J. Innate Immun. 2015, 7, 563–571. [Google Scholar] [CrossRef]
- Lanier, L.L.; Corliss, B.; Wu, J.; Phillips, J.H. Association of DAP12 with activating CD94/NKG2C NK cell receptors. Immunity 1998, 8, 693–701. [Google Scholar] [CrossRef]
- Lopez-Vergès, S.; Milush, J.M.; Pandey, S.; York, V.A.; Arakawa-Hoyt, J.; Pircher, H.; Norris, P.J.; Nixon, D.F.; Lanier, L.L. CD57 defines a functionally distinct population of mature NK cells in the human CD56dimCD16+ NK-cell subset. Blood 2010, 116, 3865–3874. [Google Scholar] [CrossRef]
- Dou, Y.; Fu, B.; Sun, R.; Li, W.; Hu, W.; Tian, Z.; Wei, H. Influenza vaccine induces intracellular immune memory of human NK cells. PLoS ONE 2015, 10, e0121258. [Google Scholar] [CrossRef] [PubMed]
- Madhun, A.S.; Akselsen, P.E.; Sjursen, H.; Pedersen, G.; Svindland, S.; Nostbakken, J.K.; Nilsen, M.; Mohn, K.; Jul-Larsen, A.; Smith, I.; et al. An adjuvanted pandemic influenza H1N1 vaccine provides early and long term protection in health care workers. Vaccine 2010, 29, 266–273. [Google Scholar] [CrossRef] [PubMed]
- Hengst, J.; Theorell, J.; Deterding, K.; Potthoff, A.; Dettmer, A.; Lunggren, H.G.; Wedemeyer, H.; Björkström, N.K. High-resolution determination of human immune cell signatures from fine-needle liver aspirates. Eur. J. Immunol. 2015, 45, 2154–2157. [Google Scholar] [CrossRef] [PubMed]
- Amir el, A.D.; Davis, K.L.; Tadmor, M.D.; Simonds, E.F.; Levine, J.H.; Bendall, S.C.; Shenfeld, D.K.; Krishnaswamy, S.; Nolan, G.P.; Pe’er, D. viSNE enables visualization of high dimensional single-cell data and reveals phenotypic heterogeneity of leukemia. Nat. Biotechnol. 2013, 31, 545–552. [Google Scholar] [CrossRef] [PubMed]
- Poli, A.; Michel, T.; Thérésine, M.; Andrès, E.; Hentges, F.; Zimmer, J. CD56bright natural killer (NK) cells: An important NK cell subset. Immunology 2009, 126, 458–465. [Google Scholar] [CrossRef]
- Den Elzen, W.P.; Vossen, A.C.M.T.; Cools, H.J.M.; Westendorp, R.G.J.; Kroes, A.C.M.; Gussekloo, J. Cytomegalovirus infection and responsiveness to influenza vaccination in elderly residents of long-term care facilities. Vaccine 2011, 29, 4869–4874. [Google Scholar] [CrossRef]
- Horowitz, A.; Hafalla, J.C.R.; King, E.; Lusingu, J.; Dekker, D.; Leach, A.; Moris, P.; Cohen, J.; Vekemans, J.; Villafana, T.; et al. Antigen-specific IL-2 secretion correlates with NK cell responses after immunization of Tanzanian children with the RTS, S/AS01 malaria vaccine. J. Immunol. 2012, 188, 5054–5062. [Google Scholar] [CrossRef]
- Horowitz, A.; Behrens, R.H.; Okell, L.; Fooks, A.R.; Riley, E.M. NK cells as effectors of acquired immune responses: Effector CD4+ T cell-dependent activation of NK cells following vaccination. J. Immunol. 2010, 185, 2808–2818. [Google Scholar] [CrossRef]
- Marquardt, N.; Ivarsson, M.A.; Blom, K.; Gonzales, V.D.; Braun, M.; Falconer, K.; Gustafsson, R.; Fogdell-Hahn, A.; Sandberg, J.K.; Michaelsson, J. The Human NK Cell Response to Yellow Fever Virus 17D Is Primarily Governed by NK Cell Differentiation Independently of NK Cell Education. J. Immunol. 2015, 195, 3262–3272. [Google Scholar] [CrossRef]
- Long, B.R.; Michaelsson, J.; Loo, C.P.; Ballan, W.M.; Vu, B.A.N.; Hecht, F.M.; Lanier, L.L.; Chapman, J.M.; Nixon, D.F. Elevated frequency of gamma interferon-producing NK cells in healthy adults vaccinated against influenza virus. Clin. Vaccine Immunol. 2008, 15, 120–130. [Google Scholar] [CrossRef]
- Goodier, M.R.; Lusa, C.; Sherratt, S.; Rodriguez-Galan, A.; Behrens, R.; Riley, E.M. Sustained Immune Complex-Mediated Reduction in CD16 Expression after Vaccination Regulates NK Cell Function. Front. Immunol. 2016, 7, 384. [Google Scholar] [CrossRef] [PubMed]
- Petitdemange, C.; Becquart, P.; Wauquier, N.; Béziat, V.; Debré, P.; Leroy, E.M.; Vieillard, V. Unconventional repertoire profile is imprinted during acute chikungunya infection for natural killer cells polarization toward cytotoxicity. PLoS Pathog. 2011, 7, e1002268. [Google Scholar] [CrossRef] [PubMed]
- Brunetta, E.; Fogli, M.; Varchetta, S.; Bozzo, L.; Hudspeth, K.L.; Marcenaro, E.; Moretta, A.; Mavilio, D. Chronic HIV-1 viremia reverses NKG2A/NKG2C ratio on natural killer cells in patients with human cytomegalovirus co-infection. AIDS 2010, 24, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Beziat, V.; Dalgard, O.; Asselah, T.; Halfon, P.; Bedossa, P.; Boudifa, A.; Hervier, B.; Theodoroi, I.; Martinot, M.; Debré, P.; et al. CMV drives clonal expansion of NKG2C+ NK cells expressing self-specific KIRs in chronic hepatitis patients. Eur. J. Immunol. 2012, 42, 447–457. [Google Scholar] [CrossRef]
- Pena, J.; Frías, M.; Castro-Orgaz, L.; Gonzáles, R.; García, F.; Gallart, T.; Gatell, J.M.; Plana, M. Dc2-Manon07 Vaccine Research Group Effects on innate immunity of a therapeutic dendritic cell-based vaccine for HIV-1 infection. Viral Immunol. 2012, 25, 37–44. [Google Scholar] [CrossRef]
- Hodara, V.L.; Parodi, L.M.; Keckler, M.S.; Giavedoni, L.D. Increases in NKG2C Expression on T Cells and Higher Levels of Circulating CD8+ B Cells Are Associated with Sterilizing Immunity Provided by a Live Attenuated SIV Vaccine. AIDS Res. Hum. Retroviruses 2016, 32, 1125–1134. [Google Scholar] [CrossRef] [PubMed]
- Reeves, R.K.; Li, H.; Jost, S.; Blass, E.; Li, H.; Schafer, J.L.; Varner, V.; Manickam, C.; Eslamizar, L.; Altfeld, M.; et al. Antigen-specific NK cell memory in rhesus macaques. Nat. Immunol. 2015, 16, 927–932. [Google Scholar] [CrossRef] [PubMed]
- Della Chiesa, M.; Sivori, S.; Carlomagno, S.; Moretta, L.; Moretta, A. Activating KIRs and NKG2C in Viral Infections: Toward NK Cell Memory? Front. Immunol. 2015, 6, 573. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, C.M.; White, M.J.; Goodier, M.R.; Riley, E.M. Functional Significance of CD57 Expression on Human NK Cells and Relevance to Disease. Front. Immunol. 2013, 4, 422. [Google Scholar] [CrossRef]
- Cook, K.D.; Waggoner, S.N.; Whitmire, J.K. NK cells and their ability to modulate T cells during virus infections. Crit. Rev. Immunol. 2014, 34, 359–388. [Google Scholar] [CrossRef]
- Crouse, J.; Xu, H.C.; Lang, P.A.; Oxenius, A. NK cells regulating T cell responses: Mechanisms and outcome. Trends Immunol. 2015, 36, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Welsh, R.M.; Waggoner, S.N. NK cells controlling virus-specific T cells: Rheostats for acute vs. persistent infections. Virology 2013, 435, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Sinzger, C.; Frascaroli, G.; Reichel, J.; Bayer, C.; Wang, L.; Schirmbeck, R.; Mertens, T. Human cytomegalovirus-induced NKG2C(hi) CD57(hi) natural killer cells are effectors dependent on humoral antiviral immunity. J. Virol. 2013, 87, 7717–7725. [Google Scholar] [CrossRef] [PubMed]
- Krebs, P.; Barnes, M.J.; Lampe, K.; Whitley, K.; Bahjat, K.S.; Beutler, B.; Janssen, E.; Hoebe, K. NK-cell-mediated killing of target cells triggers robust antigen-specific T-cell-mediated and humoral responses. Blood 2009, 113, 6593–6602. [Google Scholar] [CrossRef]
- Goodier, M.R.; Rodrigues-Galan, A.; Lusa, C.; Nielsen, C.M.; Darboe, A.; Moldoveanu, A.L.; White, M.J.; Behrens, R.; Riley, E.M. Influenza Vaccination Generates Cytokine-Induced Memory-like NK Cells: Impact of Human Cytomegalovirus Infection. J. Immunol. 2016, 197, 313–325. [Google Scholar] [CrossRef]
- Ahlenstiel, G.; Titerence, R.H.; Koh, C.; Edlich, B.; Feld, J.J.; Rotman, Y.; Ghany, M.G.; Hoofnagle, J.H.; Liang, T.J.; Heller, T.; et al. Natural killer cells are polarized toward cytotoxicity in chronic hepatitis C in an interferon-alfa-dependent manner. Gastroenterology 2010, 138, 325–335.e2. [Google Scholar] [CrossRef]
- Rolle, A.; Pollmann, J.; Cerwenka, A. Memory of infections: An emerging role for natural killer cells. PLoS Pathog. 2013, 9, e1003548. [Google Scholar] [CrossRef][Green Version]
- Cerwenka, A.; Lanier, L.L. Natural killer cell memory in infection, inflammation and cancer. Nat. Rev. Immunol. 2016, 16, 112–123. [Google Scholar] [CrossRef]
- Mandelboim, O.; Lieberman, N.; Lev, M.; Paul, L.; Arnon, T.I.; Bushkin, Y.; Davis, D.M.; Strominger, J.L.; Yewdell, J.W.; Porgador, A. Recognition of haemagglutinins on virus-infected cells by NKp46 activates lysis by human NK cells. Nature 2001, 409, 1055–1060. [Google Scholar] [CrossRef]
- Fu, X.; Yu, S.; Yang, B.; Lao, S.; Li, B.; Wu, C. Memory-Like Antigen-Specific Human NK Cells from TB Pleural Fluids Produced IL-22 in Response to IL-15 or Mycobacterium tuberculosis Antigens. PLoS ONE 2016, 11, e0151721. [Google Scholar] [CrossRef]
- Malmberg, K.J.; Beziat, V.; Ljunggren, H.G. Spotlight on NKG2C and the human NK-cell response to CMV infection. Eur. J. Immunol. 2012, 42, 3141–3145. [Google Scholar] [CrossRef] [PubMed]
- De Santo, C.; Salio, M.; Masri, S.H.; Lee, L.Y.H.; Dong, T.; Speak, A.O.; Porubsky, S.; Booth, S.; Veerapen, N.; Besra, G.S.; et al. Invariant NKT cells reduce the immunosuppressive activity of influenza A virus-induced myeloid-derived suppressor cells in mice and humans. J. Clin. Investig. 2008, 118, 4036–4048. [Google Scholar] [CrossRef] [PubMed]
- Ho, L.P.; Denney, L.; Luhn, K.; Teoh, D.; Clelland, C.; McMichael, A.J. Activation of invariant NKT cells enhances the innate immune response and improves the disease course in influenza A virus infection. Eur. J. Immunol. 2008, 38, 1913–1922. [Google Scholar] [CrossRef]
- Feng, H.; Nakajima, N.; Wu, L.; Yamashita, M.; Lopes, T.J.S.; Tsuji, M.; Hasegawa, H.; Watanabe, T.; Kawaoka, Y. A Glycolipid Adjuvant, 7DW8-5, Enhances the Protective Immune Response to the Current Split Influenza Vaccine in Mice. Front. Microbiol. 2019, 10, 2157. [Google Scholar] [CrossRef] [PubMed]
- Artiaga, B.L.; Yang, G.; Hackmann, T.J.; Liu, Q.; Richt, J.A.; Salek-Ardakani, S.; Castleman, W.L.; Lednicky, J.A.; Driver, J.P. alpha-Galactosylceramide protects swine against influenza infection when administered as a vaccine adjuvant. Sci. Rep. 2016, 6, 23593. [Google Scholar] [CrossRef]
- Garcon, N.; Vaughn, D.W.; Didierlaurent, A.M. Development and evaluation of AS03, an Adjuvant System containing alpha-tocopherol and squalene in an oil-in-water emulsion. Expert Rev. Vaccines 2012, 11, 349–366. [Google Scholar] [CrossRef]
- Morel, S.; Didierlaurent, A.; Bourguignon, P.; Delhaye, S.; Baras, B.; Jacob, V.; Planty, C.; Elouahabi, A.; Harvengt, P.; Carlsen, H.; et al. Adjuvant System AS03 containing alpha-tocopherol modulates innate immune response and leads to improved adaptive immunity. Vaccine 2011, 29, 2461–2473. [Google Scholar] [CrossRef]
- Draghi, M.; Pashine, A.; Sanjanwala, B.; Gendzekhadze, K.; Cantoni, C.; Cosman, D.; Moretta, A.; Valiante, N.M.; Parham, P. NKp46 and NKG2D recognition of infected dendritic cells is necessary for NK cell activation in the human response to influenza infection. J. Immunol. 2007, 178, 2688–2698. [Google Scholar] [CrossRef]
- Duev-Cohen, A.; Bar-On, Y.; Glasner, A.; Berhani, O.; Ophir, Y.; Levi-Schaffer, F.; Mandelboim, M.; Mandelboim, O. The human 2B4 and NTB-A receptors bind the influenza viral hemagglutinin and co-stimulate NK cell cytotoxicity. Oncotarget 2016, 7, 13093–13105. [Google Scholar] [CrossRef]
- Jegaskanda, S.; Reading, P.C.; Kent, S.J. Influenza-specific antibody-dependent cellular cytotoxicity: Toward a universal influenza vaccine. J. Immunol. 2014, 193, 469–475. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Riese, P.; Trittel, S.; Pathirana, R.D.; Klawonn, F.; Cox, R.J.; Guzmán, C.A. Responsiveness to Influenza Vaccination Correlates with NKG2C-Expression on NK Cells. Vaccines 2020, 8, 281. https://doi.org/10.3390/vaccines8020281
Riese P, Trittel S, Pathirana RD, Klawonn F, Cox RJ, Guzmán CA. Responsiveness to Influenza Vaccination Correlates with NKG2C-Expression on NK Cells. Vaccines. 2020; 8(2):281. https://doi.org/10.3390/vaccines8020281
Chicago/Turabian StyleRiese, Peggy, Stephanie Trittel, Rishi D. Pathirana, Frank Klawonn, Rebecca J. Cox, and Carlos A. Guzmán. 2020. "Responsiveness to Influenza Vaccination Correlates with NKG2C-Expression on NK Cells" Vaccines 8, no. 2: 281. https://doi.org/10.3390/vaccines8020281
APA StyleRiese, P., Trittel, S., Pathirana, R. D., Klawonn, F., Cox, R. J., & Guzmán, C. A. (2020). Responsiveness to Influenza Vaccination Correlates with NKG2C-Expression on NK Cells. Vaccines, 8(2), 281. https://doi.org/10.3390/vaccines8020281





