Trends of Adverse Events Following Immunization (AEFI) Reports of Human Papillomavirus Vaccine in the Valencian Community—Spain (2008–2018)
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Source
2.2. Data Extraction
2.3. Descriptive Analysis
2.4. Trend Analysis
2.5. Ethical Statement
3. Results
3.1. Trends over Time by Setting
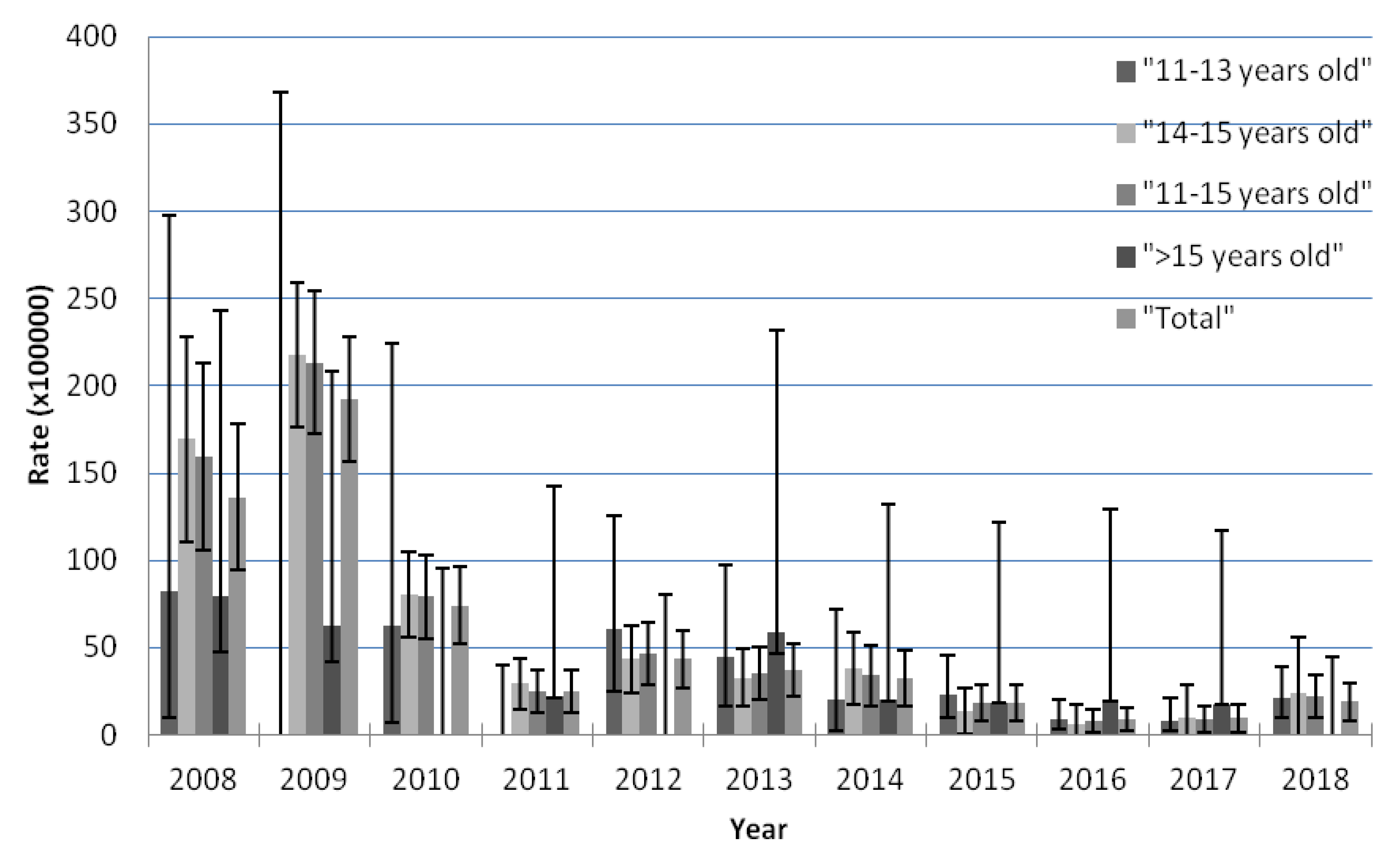
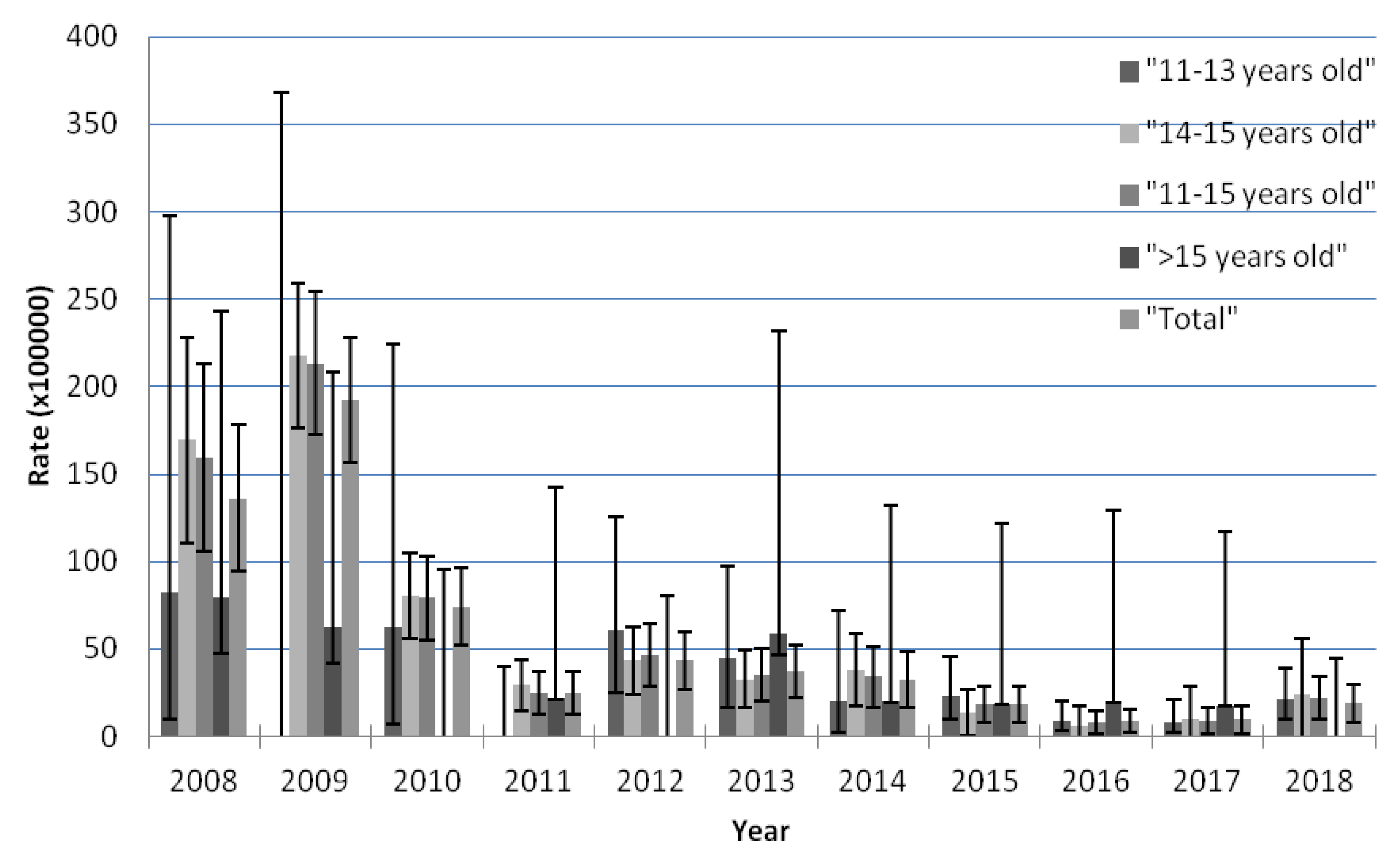
3.2. Trend in AEFI Notifications by Age According to the Joinpoints Identified by the Analysis
3.3. The Trend of AEFIs Reported as ‘Serious’
3.4. Events Following Immunization Reactions
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A
| Years | 11 to 13 Years Old | 14 to 15 Years Old | >15 Years Old * | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ICSR | Doses | AEFI Rate | 95%CI | ICSR | Doses | AEFI Rate | 95%CI | ICSR | Doses | AEFI Rate | 95%CI | |
| 2008 | 2 | 2424 | 82.51 | 9.99–298.05 | 32 | 18,875 | 169.54 | 110.8–228.28 | 7 | 8813 | 79.43 | 31.93–163.65 |
| 2009 | 0 | 1003 | 0 | 0.00–367.78 | 105 | 48,220 | 217.75 | 176.10–259.4 | 5 | 8010 | 62.42 | 20.27–145.67 |
| 2010 | 2 | 3217 | 62.17 | 7.53–224.58 | 41 | 51,121 | 80.2 | 55.65–104.75 | 0 | 3863 | 0 | 0.00–95.49 |
| 2011 | 0 | 9130 | 0 | 0.00–40.40 | 16 | 54,350 | 29.44 | 15.01–43.86 | 1 | 4609 | 21.7 | 0.55–120.89 |
| 2012 | 7 | 11,494 | 60.9 | 24.49–125.48 | 20 | 46,171 | 43.32 | 24.33–62.30 | 0 | 4592 | 0 | 0.00–80.33 |
| 2013 | 6 | 13,418 | 44.72 | 16.41–97.33 | 15 | 45,625 | 32.88 | 16.24–49.51 | 3 | 5083 | 59.02 | 12.17–172.48 |
| 2014 | 2 | 10,083 | 19.84 | 2.40–71.65 | 13 | 33,958 | 38.28 | 17.47–59.09 | 1 | 4978 | 20.09 | 0.51–111.93 |
| 2015 | 8 | 34,885 | 22.93 | 9.90–45.19 | 4 | 29,914 | 13.37 | 0.27–26.48 | 1 | 5381 | 18.58 | 0.47–103.54 |
| 2016 | 5 | 56,405 | 8.86 | 2.88–20.69 | 1 | 17,283 | 5.79 | 0.00–17.13 | 1 | 5073 | 19.71 | 0.50–109.83 |
| 2017 | 4 | 47,565 | 8.41 | 2.29–21.53 | 1 | 10,241 | 9.76 | 0.00–28.90 | 1 | 5596 | 17.87 | 0.45–99.56 |
| 2018 | 10 | 46,685 | 21.42 | 10.27–39.39 | 2 | 8475 | 23.6 | 0.00–56.31 | 0 | 8182 | 0 | 0.00–45.09 |
| Total | 46 | 236,309 | 19.47 | 13.84–25.09 | 250 | 364,233 | 68.64 | 60.13–77.15 | 20 | 64,180 | 31.16 | 17.50–44.82 |
References
- World Health Organization. Human papillomavirus vaccines: WHO position paper, October 2014. Wkly. Epidemiol. Rec. 2014, 89, 465–491. [Google Scholar]
- Kjaer, S.K.; Nygård, M.; Dillner, J.; Brooke Marshall, J.; Radley, D.; Li, M.; Munk, C.; Hansen, B.T.; Sigurdardottir, L.G.; Hortlund, M.; et al. A 12-Year Follow-up on the Long-Term Effectiveness of the Quadrivalent Human Papillomavirus Vaccine in 4 Nordic Countries. Clin. Infect. Dis. 2018, 66, 339–345. [Google Scholar] [CrossRef] [PubMed]
- Drolet, M.; Bénard, É; Pérez, N.; Brisson, M. HPV Vaccination Impact Study Group. Population-level impact and herd effects following the introduction of human papillomavirus vaccination programmes: Updated systematic review and meta-analysis. Lancet 2019, 394, 497–509. [Google Scholar] [CrossRef]
- Arbyn, M.; Xu, L. Efficacy and safety of prophylactic HPV vaccines. A Cochrane review of randomized trials. Expert Rev. Vaccines 2018, 17, 1085–1091. [Google Scholar] [CrossRef] [PubMed]
- Gardasil. Available online: https://www.fda.gov/vaccines-blood-biologics/vaccines/gardasil (accessed on 30 December 2019).
- Cervarix. Available online: https://www.fda.gov/vaccines-blood-biologics/vaccines/cervarix (accessed on 30 December 2019).
- Gardasil 9. Available online: https://www.fda.gov/vaccines-blood-biologics/vaccines/gardasil-9 (accessed on 30 December 2019).
- Cervarix. Available online: https://www.ema.europa.eu/en/medicines/human/EPAR/cervarix (accessed on 30 December 2019).
- Gardasil. Available online: https://www.ema.europa.eu/en/medicines/human/EPAR/gardasil (accessed on 30 December 2019).
- Gardasil 9. Product Information Last Up Date 10/10/2019. Available online: https://www.ema.europa.eu/en/medicines/human/EPAR/gardasil-9 (accessed on 30 December 2019).
- Chen, R.; Rastogi, S.; Mullen, J.; Hayes, S.; Cochi, S.; Donlon, J.; Wassilak, S. The vaccine adverse event reporting system (VAERS). Vaccine 1994, 12, 542–550. [Google Scholar] [CrossRef]
- Gee, J.; Weinbaum, C.; Sukumaran, L.; Markowitz, L.E. Quadrivalent HPV vaccine safety review and safety monitoring plans for nine-valent HPV vaccine in the United States. Hum. Vaccin. Immunother. 2016, 12, 1406–1417. [Google Scholar] [CrossRef]
- Chandler, R.E. Modernising vaccine surveillance systems to improve detection of rare or poorly defined adverse events. BMJ 2019, 365, l2268. [Google Scholar] [CrossRef]
- Limia, A.; Pachón, I. Coverage of human papillomavirus vaccination during the first year of its introduction in Spain. Euro Surveill. 2011, 16, 19873. [Google Scholar]
- Human Papilomavirus. Current Situation, Vaccines and Prospect of Their Use. February 2007. Available online: http://www.mscbs.gob.es/profesionales/saludPublica/prevPromocion/vacunaciones/docs/VPH_2007.pdf (accessed on 12 January 2020).
- History of Childhood Immunization Schedules in Valencia (1992–2015). Available online: http://www.sp.san.gva.es/DgspPortal/docs/HistoricoCalendariosVacunacionInfantil(vf).pdf (accessed on 12 January 2020).
- Acquisition of Vaccines against Human Papillomavirus (HPV). DOCV Num 6656/22.11.2011. Available online: http://www.dogv.gva.es/datos/2011/11/22/pdf/2011_11678.pdf (accessed on 7 February 2020).
- ORDER of 16 June 2008, of the Department of Health, which Modifies the Schedule of Systematic Childhood Vaccination in the Valencian Community. DOGV núm. 5812/23.07.2008. Available online: http://www.dogv.gva.es/es/disposicio?sig=008852/2008&&L=1 (accessed on 7 February 2020).
- Valencian Community. Municipal Register of Inhabitants. 2018. Available online: http://www.pegv.gva.es/es/noticias/-/asset_publisher/CWK0IEKbs79H/content/padron-municipal-continuo-explotacion-estadistica-2018 (accessed on 30 December 2019).
- Vaccination Coverages. Statistical data. Available online: https://www.mscbs.gob.es/profesionales/saludPublica/prevPromocion/vacunaciones/coberturas.htm (accessed on 30 December 2019).
- Agency European Medicines. European Medicines Agency recommends continued vaccination with Gardasil. Press Release. EMEA/CHMP/103339/2009. 19 February 2009. Available online: http://www.emea.europa.eu (accessed on 24 January 2020).
- Tuells, J.; Duro Torrijos, J.L.; Chilet Rosell, E.; Pastor Villalba, E.; Portero Alonso, A.; Navarro Ortiz, C.; Galiana de la Villa, E.M. [News items on human papillomavirus and its vaccine in the Valencian press (2006–2011)]. Gac. Sanit. 2013, 27, 374–377. [Google Scholar] [CrossRef]
- Rodríguez-Galán, M.A.; Pérez-Vilar, S.; Díez-Domingo, J.; Tuells, J.; Gomar-Fayos, J.; Morales-Olivas, F.; Pastor-Villalba, E. Adverse reactions to human papillomavirus vaccine in the Valencian Community (2007–2011). An. Pediatr. 2014, 81, 303–309. [Google Scholar] [CrossRef]
- Royal Decree 577/2013, of July 26, Which Regulates the Pharmacovigilance of Medicines for Human Use. Available online: https://www.boe.es/eli/es/rd/2013/07/26/577 (accessed on 7 February 2020).
- Agencia Española de Medicamentos y Productos Sanitarios (AEMPS) Sistema Español de Farmacovigilancia de Medicamentos de Uso Humano (SEFV-H). [Spanish Agency for Medicines and Health Products (AEMPS). Spanish Pharmacovigilance System for Medicines for Human Use (SEFV-H).]. Available online: https://www.aemps.gob.es/vigilancia/medicamentosUsoHumano/SEFV-H/home.htm (accessed on 7 February 2020).
- Manual of the Vaccine Information System. Valencia: Ministry of Health. Generalitat Valenciana. Available online: http://www.sp.san.gva.es/biblioteca/publicacion_dgsp.jsp?cod_pub_ran=19281&tacc=15 (accessed on 29 December 2019).
- Brown, E.G.; Wood, L.; Wood, S. The Medical Dictionary for Regulatory Activities (MedDRA). Drug Saf. 1999, 20, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Patil, V.V.; Kulkarni, H.V. Comparison of Confidence Intervals for the Poisson Mean: Some New Aspects. Revstat Stat. J. 2012, 10, 211–227. [Google Scholar]
- National Cancer Institute. Joinpoint Trend Analysis Software. Available online: https://surveillance.cancer.gov/joinpoint/ (accessed on 30 December 2019).
- Kim, H.J.; Fay, M.P.; Feuer, E.J.; Midthune, D.N. Permutation tests for joinpoint regression with applications to cancer rates. Stat. Med. 2000, 19, 335–351. [Google Scholar] [CrossRef]
- Mahajan, D.; Dey, A.; Cook, J.; Harvey, B.; Menzies, R.; Macartney, K. Surveillance of adverse events following immunisation in Australia annual report, 2013. Commun Dis Intell 2015, 39, 369–386. [Google Scholar]
- Australian Institute of Health and Welfare 2009 Adult Vaccination Survey: Summary Results. Available online: https://www.aihw.gov.au/getmedia/91c13f90-7a4f-44ff-b09d-bcf344f7ca6d/11936.pdf.aspx?inline=true (accessed on 12 January 2020).
- Mauro, A.B.; Fernandes, E.G.; Miyaji, K.T.; Arantes, B.A.; Valente, M.G.; Sato, H.K.; Sartori, A.M.C. Adverse events following Quadrivalent HPV vaccination reported in Sao Paulo State, Brazil, in the first three years after introducing the vaccine for routine immunization (March 2014 to December 2016). Rev. Inst. Med. Trop. Sao Paulo 2019, 61, e43. [Google Scholar] [CrossRef] [PubMed]
- Wallenstein, E.J.; Fife, D. Temporal patterns of NSAID spontaneous adverse event reports: The weber effect revisited. Drug Saf. 2001, 24, 233–237. [Google Scholar] [CrossRef]
- Loharikar, A.; Suragh, T.A.; MacDonald, N.E.; Balakrishnan, M.R.; Benes, O.; Lamprianou, S.; Hyde, T.B.; McNeil, M.M. Anxiety-related adverse events following immunization (AEFI): A systematic review of published clusters of illness. Vaccine 2018, 36, 299–305. [Google Scholar] [CrossRef]
- Caballero-Pérez, P.; Tuells, J.; Rementería, J.; Nolasco, A.; Navarro-López, V.; Arístegui, J. [Acceptability of the HPV vaccine among Spanish university students in the pre-vaccine era: A cross-sectional study]. Rev. Esp. Quimioter. 2015, 28, 21–28. [Google Scholar]
- Šubelj, M.; Učakar, V.; Kraigher, A.; Klavs, I. Adverse events following school-based vaccination of girls with quadrivalent human papillomavirus vaccine in Slovenia, 2009 to 2013. Euro Surveill. 2016, 21, 14. [Google Scholar] [CrossRef]
- Cervarix HPV Vaccine: Update on UK Safety Experience at End of 4 Years Use in the HPV Routine Immunisation Programme. Available online: https://assets.publishing.service.gov.uk/media/547307f5ed915d1380000039/con213228.pdf (accessed on 29 February 2020).
- Thomsen, P.O.; Saldaña, V.A.; Cerda, L.J.; Abarca, V.K. Vaccine safety: Description of the adverse events reported to the surveillance system in Chile, 2014 to 2016. Rev. Chil. Infectol. 2019, 36, 461–468. [Google Scholar]
- Law, B.; Laflèche, J.; Ahmadipour, N.; Anyoti, H. Canadian Adverse Events Following Immunization Surveillance System (CAEFISS): Annual report for vaccines administered in 2012. Canada Commun. Dis. Rep. 2014, 40, 7–23. [Google Scholar] [CrossRef] [PubMed]
- Suragh, T.A.; Lewis, P.; Arana, J.; Mba-Jonas, A.; Li, R.; Stewart, B.; Shimabukuro, T.T.; Cano, M. Safety of bivalent human papillomavirus vaccine in the US vaccine adverse event reporting system (VAERS), 2009–2017. Br. J. Clin. Pharmacol. 2018, 84, 2928–2932. [Google Scholar] [CrossRef] [PubMed]
- Slade, B.A.; Leidel, L.; Vellozzi, C.; Woo, E.J.; Hua, W.; Sutherland, A.; Izurieta, H.S.; Ball, R.; Miller, N.; Braun, M.M.; et al. Postlicensure safety surveillance for quadrivalent human papillomavirus recombinant vaccine. JAMA 2009, 302, 750–757. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.C.; Bell, C.A.; Simmonds, K.A.; Svenson, L.W.; Russell, M.L. Adverse events following HPV vaccination, Alberta 2006–2014. Vaccine 2016, 34, 1800–1805. [Google Scholar] [CrossRef] [PubMed]
- Evaluation of the National Human Papillomavirus Vaccination Program Final Report. Available online: https://www.health.gov.au/resources/publications/evaluation-of-the-national-hpv-program-final-report (accessed on 29 February 2020).
- Johnson, K.; Anyoti, H.; Coulby, C. Vaccine safety surveillance in Canada: Reports to CAEFISS, 2017. Canada Commun. Dis. Rep. 2018, 44, 333–339. [Google Scholar] [CrossRef] [PubMed]
- Tafuri, S.; Fortunato, F.; Gallone, M.S.; Stefanizzi, P.; Calabrese, G.; Boccalini, S.; Martinelli, D.; Prato, R. Systematic causality assessment of adverse events following HPV vaccines: Analysis of current data from Apulia region (Italy). Vaccine 2018, 36, 1072–1077. [Google Scholar] [CrossRef]
- Harris, T.; Williams, D.M.; Fediurek, J.; Scott, T.; Deeks, S.L. Adverse events following immunization in Ontario’s female school-based HPV program. Vaccine 2014, 32, 1061–1066. [Google Scholar] [CrossRef]
- Buttery, J.P.; Madin, S.; Crawford, N.W.; Elia, S.; La Vincente, S.; Hanieh, S.; Smith, L.; Bolam, B. Mass psychogenic response to human papillomavirus vaccination. Med. J. Aust. 2008, 189, 261–262. [Google Scholar] [CrossRef]
- Castro, C.M. The media influence on the Weber effect: Correlation between the news published about the HPV vaccine and the alerts recorded in pharmacovigilance. Panacea 2015, 16, 195–205. [Google Scholar]
- Ulendorf Jacobsen, S.; Valentiner-Branth, P.; Mølbak, K. Examining determinants for reporting suspected adverse events following HPV vaccination in Denmark. Vaccine 2018, 36, 6158–6162. [Google Scholar] [CrossRef]
- Dey, A.; Wang, H.; Quinn, H.; Cook, J.; Macartney, K. Surveillance of adverse events following immunisation in Australia, 2015. Commun. Dis. Intell. Q. Rep. 2017, 41, E264–E278. [Google Scholar] [PubMed]
- Vaccine Report Post Marketing Surveillance in Italy. 2017. Available online: http://www.agenziafarmaco.gov.it/sites/default/files/Rapp_Vaccini_2017_0.pdf (accessed on 14 January 2020).
- Chandler, R.E.; Juhlin, K.; Fransson, J.; Caster, O.; Edwards, I.R.; Norén, G.N. Current Safety Concerns with Human Papillomavirus Vaccine: A Cluster Analysis of Reports in VigiBase®. Drug Saf. 2017, 40, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Ward, D.; Thorsen, N.M.; Frisch, M.; Valentiner-Branth, P.; Mølbak, K.; Hviid, A. A cluster analysis of serious adverse event reports after human papillomavirus (HPV) vaccination in Danish girls and young women, September 2009 to August 2017. Euro. Surveill. 2019, 24, 19. [Google Scholar] [CrossRef] [PubMed]
- Suragh, T.A.; Lamprianou, S.; MacDonald, N.E.; Loharikar, A.R.; Balakrishnan, M.R.; Benes, O.; Hyde, T.B.; McNeil, M.M. Cluster anxiety-related adverse events following immunization (AEFI): An assessment of reports detected in social media and those identified using an online search engine. Vaccine 2018, 36, 5949–5954. [Google Scholar] [CrossRef]
- Bernard, D.M.; Cooper Robbins, S.C.; McCaffery, K.J.; Scott, C.M.; Skinner, S.R. The domino effect: Adolescent girls’ response to human papillomavirus vaccination. Med. J. Aust. 2011, 194, 297–300. [Google Scholar] [CrossRef]
- Forster, A.S.; McBride, K.A.; Davies, C.; Stoney, T.; Marshall, H.; McGeechan, K.; Cooper, S.C.; Skinner, S.R. Development and validation of measures to evaluate adolescents’ knowledge about human papillomavirus (HPV), involvement in HPV vaccine decision-making, self-efficacy to receive the vaccine and fear and anxiety. Public Health 2017, 147, 77–83. [Google Scholar] [CrossRef]
- O’Neill, J.; Newall, F.; Antolovich, G.; Lima, S.; Danchin, M. The uptake of adolescent vaccinations through the School Immunisation Program in specialist schools in Victoria, Australia. Vaccine 2019, 37, 272–279. [Google Scholar] [CrossRef]
- Guide to Interpreting VAERS Data. Available online: https://vaers.hhs.gov/data/dataguide.html (accessed on 24 January 2020).
- Lei, J.; Balakrishnan, M.R.; Gidudu, J.F.; Zuber, P.L.F. Use of a new global indicator for vaccine safety surveillance and trends in adverse events following immunization reporting 2000–2015. Vaccine 2018, 36, 1577–1582. [Google Scholar] [CrossRef]
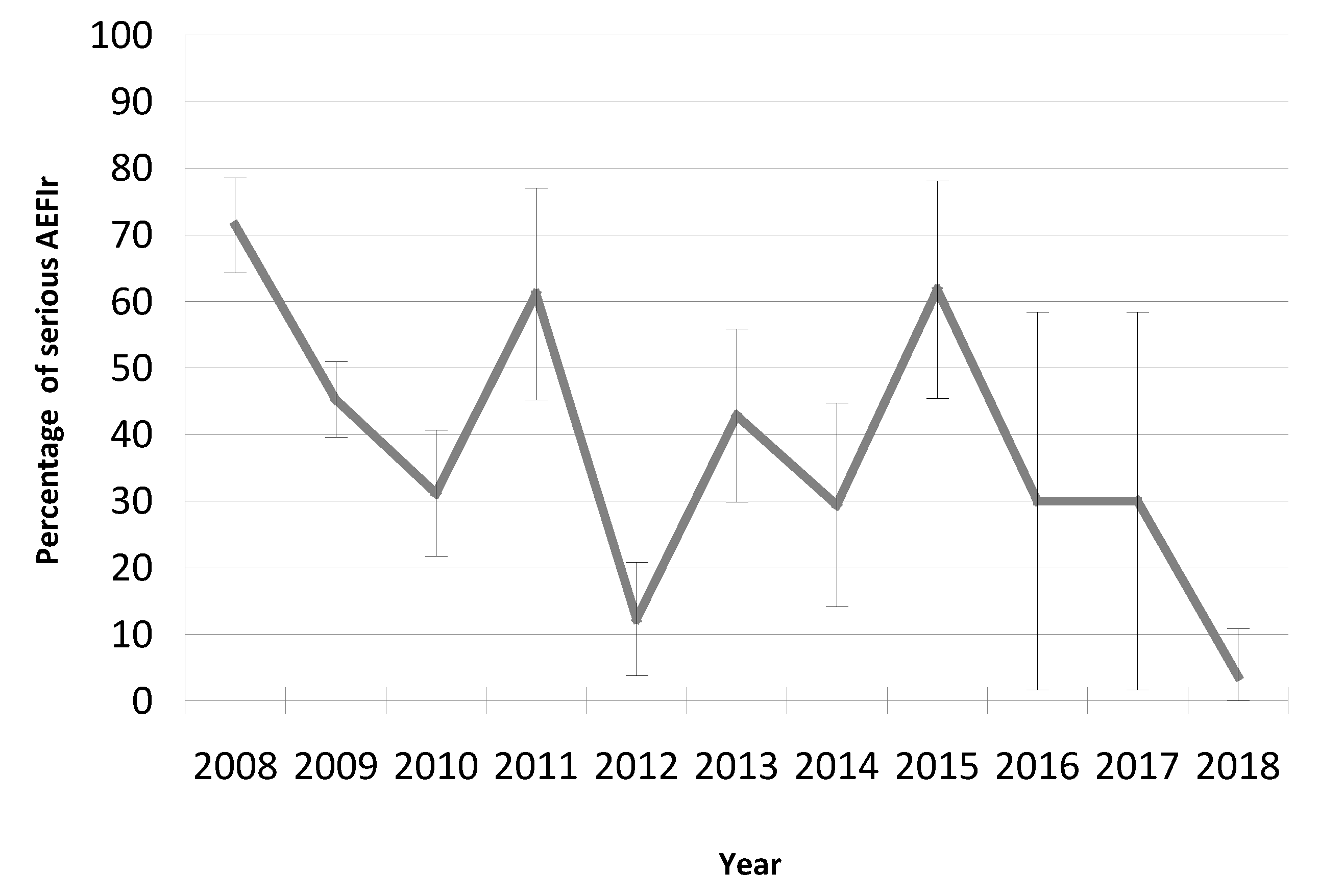
| Years | AEFIr | Doses | AEFIr Rate 1 | 95%CI | Total AE | AEFIr with Serious AE | Total Serious AE | Total AE/AEFIr | Total AE Serious/AEFIr | AEFIr Serious Rate 1 |
|---|---|---|---|---|---|---|---|---|---|---|
| 2008 | 41 | 30,112 | 136.16 | 94.48–177.84 | 154 | 18 | 110 | 3.76 | 2.68 | 59.78 |
| 2009 | 110 | 57,233 | 192.2 | 156.28–228.11 | 296 | 24 | 134 | 2.69 | 1.22 | 41.93 |
| 2010 | 43 | 58,201 | 73.88 | 51.80–95.96 | 93 | 13 | 29 | 2.16 | 0.67 | 22.34 |
| 2011 | 17 | 68,089 | 24.97 | 13.10–36.84 | 36 | 6 | 22 | 2.12 | 1.29 | 8.81 |
| 2012 | 27 | 62,257 | 43.37 | 27.01–59.73 | 57 | 3 | 7 | 2.11 | 0.26 | 4.82 |
| 2013 | 24 | 64,126 | 37.43 | 22.45–52.40 | 56 | 8 | 24 | 2.33 | 1.00 | 12.48 |
| 2014 | 16 | 49,019 | 32.64 | 16.65–48.63 | 34 | 2 | 10 | 2.13 | 0.63 | 4.08 |
| 2015 | 13 | 70,180 | 18.52 | 8.45–28.59 | 34 | 6 | 21 | 2.62 | 1.62 | 8.55 |
| 2016 | 7 | 78,761 | 11.04 | 2.30–15.47 | 15 | 1 | 1 | 1.43 | 0.43 | 1.27 |
| 2017 | 7 | 63,402 | 9.46 | 1.89–17.04 | 10 | 1 | 3 | 1.67 | 0.50 | 1.58 |
| 2018 | 12 | 63,342 | 18.94 | 8.23–29.66 | 27 | 1 | 1 | 2.25 | 0.08 | 1.58 |
| Total | 317 | 66,4722 | 47.54 | 42.30–52.78 | 812 | 83 | 362 | 2.57 | 1.14 | 12.49 |
| 2008 | 2009 | 2010 | 2011 | 2012 | 2013 | 2014 | 2015 | 2016 | 2017 | 2018 | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| AEFI a | (n) | (n) | (n) | (n) | (n) | (n) | (n) | (n) | (n) | (n) | (n) |
| Injection site reaction b | 8 | 24 | 11 | 6 | 9 | 4 | 2 | 1 | 6 | 3 | 8 |
| Headache | 7 | 33 | 6 | 4 | 7 | 11 | 3 | 5 | 2 | 0 | 1 |
| Dizziness | 12 | 32 | 7 | 2 | 0 | 3 | 6 | 4 | 1 | 1 | 4 |
| Fever | 2 | 26 | 12 | 6 | 7 | 9 | 2 | 3 | 1 | 2 | 0 |
| Syncope | 11 | 21 | 12 | 3 | 5 | 2 | 3 | 3 | 1 | 0 | 1 |
| Myalgia and malaise | 1 | 16 | 6 | 2 | 4 | 3 | 1 | 6 | 1 | 2 | 1 |
| Nausea/ vomiting | 4 | 20 | 4 | 1 | 3 | 4 | 3 | 1 | 0 | 0 | 1 |
| Presyncope/Pallor | 6 | 11 | 2 | 1 | 2 | 0 | 3 | 1 | 1 | 1 | 5 |
| Hypersensitivity reaction c | 3 | 11 | 5 | 0 | 6 | 1 | 2 | 1 | 0 | 1 | 1 |
| Seizure | 0 | 9 | 3 | 0 | 1 | 2 | 0 | 0 | 0 | 0 | 0 |
| Somnolence | 3 | 4 | 2 | 1 | 1 | 0 | 0 | 1 | 0 | 0 | 1 |
| Others | 97 | 89 | 23 | 10 | 12 | 17 | 9 | 8 | 2 | 0 | 4 |
| Total | 154 | 296 | 93 | 36 | 57 | 56 | 34 | 34 | 15 | 10 | 27 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Egoavil, C.M.; Tuells, J.; Carreras, J.J.; Montagud, E.; Pastor-Villalba, E.; Caballero, P.; Nolasco, A. Trends of Adverse Events Following Immunization (AEFI) Reports of Human Papillomavirus Vaccine in the Valencian Community—Spain (2008–2018). Vaccines 2020, 8, 117. https://doi.org/10.3390/vaccines8010117
Egoavil CM, Tuells J, Carreras JJ, Montagud E, Pastor-Villalba E, Caballero P, Nolasco A. Trends of Adverse Events Following Immunization (AEFI) Reports of Human Papillomavirus Vaccine in the Valencian Community—Spain (2008–2018). Vaccines. 2020; 8(1):117. https://doi.org/10.3390/vaccines8010117
Chicago/Turabian StyleEgoavil, Cecilia M., José Tuells, Juan José Carreras, Emilia Montagud, Eliseo Pastor-Villalba, Pablo Caballero, and Andreu Nolasco. 2020. "Trends of Adverse Events Following Immunization (AEFI) Reports of Human Papillomavirus Vaccine in the Valencian Community—Spain (2008–2018)" Vaccines 8, no. 1: 117. https://doi.org/10.3390/vaccines8010117
APA StyleEgoavil, C. M., Tuells, J., Carreras, J. J., Montagud, E., Pastor-Villalba, E., Caballero, P., & Nolasco, A. (2020). Trends of Adverse Events Following Immunization (AEFI) Reports of Human Papillomavirus Vaccine in the Valencian Community—Spain (2008–2018). Vaccines, 8(1), 117. https://doi.org/10.3390/vaccines8010117







