Strain-Dependent Impact of G and SH Deletions Provide New Insights for Live-Attenuated HMPV Vaccine Development
Abstract
1. Introduction
2. Materials and Methods
2.1. Cells
2.2. Rescue of Recombinant Viruses
2.3. In Vitro Experiments
2.4. Infection of Reconstituted Human Airway Epithelium
2.5. Real-Time (RT)-PCR
2.6. Animal Studies
2.7. Statistical Analysis
3. Results
3.1. In Vitro Characteristics of ΔG- and ΔSH-HMPV Recombinant Viruses Differ Depending on the Viral Strain Background
3.2. ΔG- and ΔSH-Viruses Harbor Different Replicative Properties in Reconstituted Human Airway Epithelium
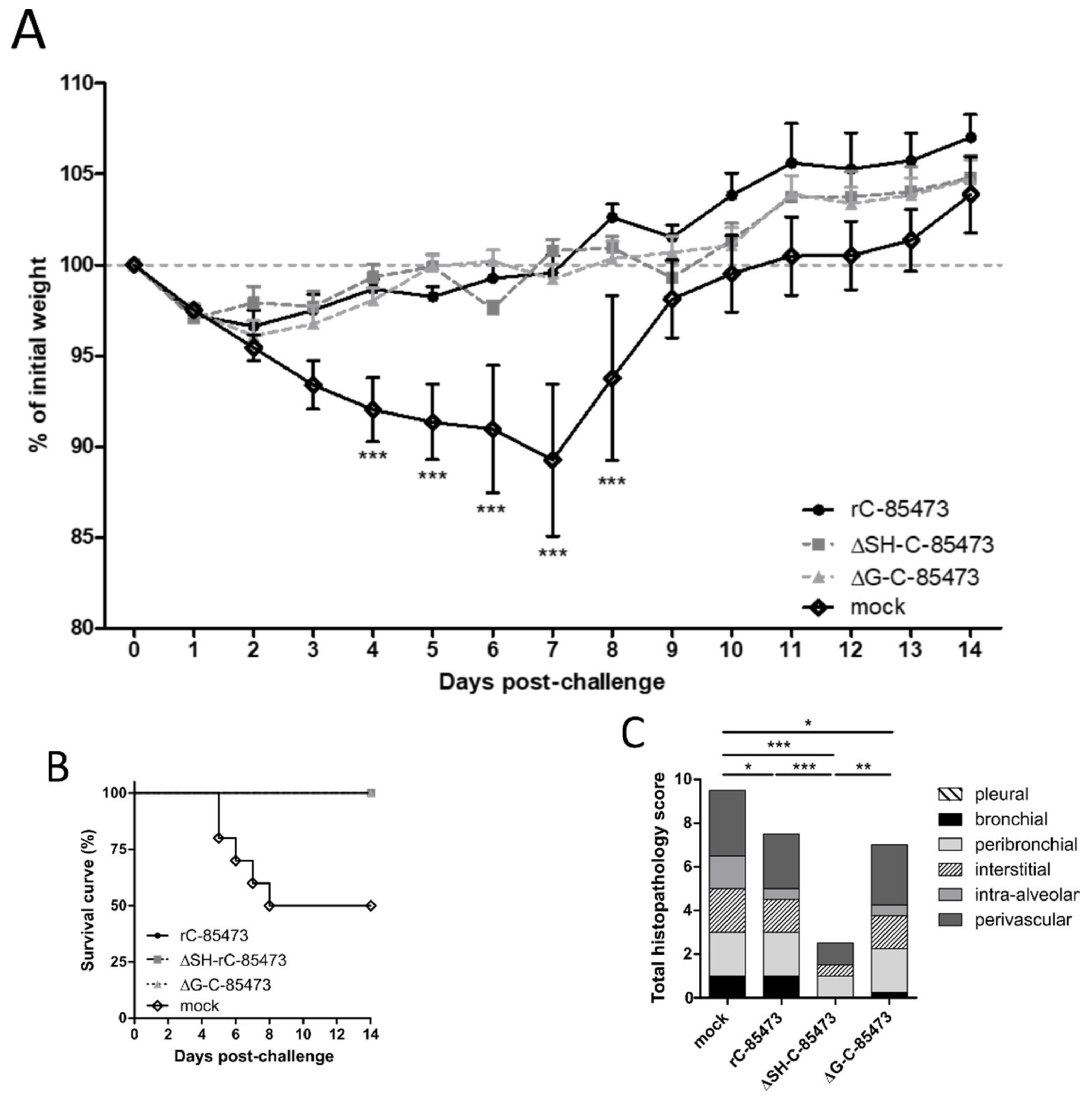
3.3. Immunization with Deleted-C-85473 Viruses Reduces HMPV Disease Severity in Challenged BALB/c Mice
4. Discussion
5. Conclusions
6. Patents
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Van den Hoogen, B.G.; Herfst, S.; Sprong, L.; Cane, P.A.; Forleo-Neto, E.; de Swart, R.L.; Osterhaus, A.D.; Fouchier, R.A. Antigenic and genetic variability of human metapneumoviruses. Emerg. Infect. Dis. 2004, 10, 658–666. [Google Scholar] [CrossRef]
- Feuillet, F.; Lina, B.; Rosa-Calatrava, M.; Boivin, G. Ten years of human metapneumovirus research. J. Clin. Virol. 2012, 53, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Papenburg, J.; Boivin, G. The distinguishing features of human metapneumovirus and respiratory syncytial virus. Rev. Med. Virol. 2010, 20, 245–260. [Google Scholar] [CrossRef] [PubMed]
- Rima, B.; Collins, P.; Easton, A.; Fouchier, R.; Kurath, G.; Lamb, R.A.; Lee, B.; Maisner, A.; Rota, P.; Wang, L.; et al. ICTV Virus Taxonomy Profile: Pneumoviridae. J. Gen. Virol. 2017, 98, 2912–2913. [Google Scholar] [CrossRef] [PubMed]
- Mazur, N.I.; Higgins, D.; Nunes, M.C.; Melero, J.A.; Langedijk, A.C.; Horsley, N.; Buchholz, U.J.; Openshaw, P.J.; McLellan, J.S.; Englund, J.A.; et al. The respiratory syncytial virus vaccine landscape: lessons from the graveyard and promising candidates. Lancet Infect. Dis. 2018, 18, e295–e311. [Google Scholar] [CrossRef]
- Marquez-Escobar, V.A. Current developments and prospects on human metapneumovirus vaccines. Expert Rev. Vaccines 2017, 16, 419–431. [Google Scholar] [CrossRef]
- Hamelin, M.E.; Couture, C.; Sackett, M.K.; Boivin, G. Enhanced lung disease and Th2 response following human metapneumovirus infection in mice immunized with the inactivated virus. J. Gen. Virol. 2007, 88, 3391–3400. [Google Scholar] [CrossRef]
- Karron, R.A.; Buchholz, U.J.; Collins, P.L. Live-attenuated respiratory syncytial virus vaccines. Curr Top Microbiol. Immunol. 2013, 372, 259–284. [Google Scholar]
- Papenburg, J.; Carbonneau, J.; Isabel, S.; Bergeron, M.G.; Williams, J.V.; De Serres, G.; Hamelin, M.E.; Boivin, G. Genetic diversity and molecular evolution of the major human metapneumovirus surface glycoproteins over a decade. J. Clin. Virol. 2013, 58, 541–547. [Google Scholar] [CrossRef]
- Huck, B.; Scharf, G.; Neumann-Haefelin, D.; Puppe, W.; Weigl, J.; Falcone, V. Novel human metapneumovirus sublineage. Emerg. Infect. Dis. 2006, 12, 147–150. [Google Scholar] [CrossRef]
- Peret, T.C.; Boivin, G.; Li, Y.; Couillard, M.; Humphrey, C.; Osterhaus, A.D.; Erdman, D.D.; Anderson, L.J. Characterization of human metapneumoviruses isolated from patients in North America. J. Infect. Dis. 2002, 185, 1660–1663. [Google Scholar] [CrossRef] [PubMed]
- Hamelin, M.E.; Abed, Y.; Boivin, G. Human metapneumovirus: a new player among respiratory viruses. Clin. Infect. Dis. 2004, 38, 983–990. [Google Scholar] [CrossRef] [PubMed]
- Nidaira, M.; Taira, K.; Hamabata, H.; Kawaki, T.; Gushi, K.; Mahoe, Y.; Maeshiro, N.; Azama, Y.; Okano, S.; Kyan, H.; et al. Molecular epidemiology of human metapneumovirus from 2009 to 2011 in Okinawa, Japan. Jpn. J. Infect. Dis. 2012, 65, 337–340. [Google Scholar] [CrossRef] [PubMed]
- Biacchesi, S.; Skiadopoulos, M.H.; Boivin, G.; Hanson, C.T.; Murphy, B.R.; Collins, P.L.; Buchholz, U.J. Genetic diversity between human metapneumovirus subgroups. Virology 2003, 315, 1–9. [Google Scholar] [CrossRef]
- Van den Hoogen, B.G.; Bestebroer, T.M.; Osterhaus, A.D.; Fouchier, R.A. Analysis of the genomic sequence of a human metapneumovirus. Virology 2002, 295, 119–132. [Google Scholar] [CrossRef] [PubMed]
- Skiadopoulos, M.H.; Biacchesi, S.; Buchholz, U.J.; Amaro-Carambot, E.; Surman, S.R.; Collins, P.L.; Murphy, B.R. Individual contributions of the human metapneumovirus F, G, and SH surface glycoproteins to the induction of neutralizing antibodies and protective immunity. Virology 2006, 345, 492–501. [Google Scholar] [CrossRef] [PubMed]
- Cox, R.G.; Williams, J.V. Breaking in: Human metapneumovirus fusion and entry. Viruses 2013, 5, 192–210. [Google Scholar] [CrossRef]
- Adamson, P.; Thammawat, S.; Muchondo, G.; Sadlon, T.; Gordon, D. Diversity in glycosaminoglycan binding amongst hMPV G protein lineages. Viruses 2012, 4, 3785–3803. [Google Scholar] [CrossRef]
- Cox, R.G.; Livesay, S.B.; Johnson, M.; Ohi, M.D.; Williams, J.V. The human metapneumovirus fusion protein mediates entry via an interaction with RGD-binding integrins. J. Virol. 2012, 86, 12148–12160. [Google Scholar] [CrossRef]
- Chang, A.; Masante, C.; Buchholz, U.J.; Dutch, R.E. Human metapneumovirus (HMPV) binding and infection are mediated by interactions between the HMPV fusion protein and heparan sulfate. J. Virol. 2012, 86, 3230–3243. [Google Scholar] [CrossRef]
- Cseke, G.; Maginnis, M.S.; Cox, R.G.; Tollefson, S.J.; Podsiad, A.B.; Wright, D.W.; Dermody, T.S.; Williams, J.V. Integrin αvβ1 promotes infection by human metapneumovirus. PNAS 2009, 106, 1566–1571. [Google Scholar] [CrossRef] [PubMed]
- Bao, X.; Liu, T.; Shan, Y.; Li, K.; Garofalo, R.P.; Casola, A. Human metapneumovirus glycoprotein G inhibits innate immune responses. PLoS Pathog. 2008, 4, e1000077. [Google Scholar] [CrossRef] [PubMed]
- Kolli, D.; Bao, X.; Liu, T.; Hong, C.; Wang, T.; Garofalo, R.P.; Casola, A. Human metapneumovirus glycoprotein G inhibits TLR4-dependent signaling in monocyte-derived dendritic cells. J. Immunol. 2011, 187, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Bao, X.; Kolli, D.; Ren, J.; Liu, T.; Garofalo, R.P.; Casola, A. Human Metapneumovirus Glycoprotein G Disrupts Mitochondrial Signaling in Airway Epithelial Cells. PLoS ONE 2013, 8, e62568. [Google Scholar] [CrossRef] [PubMed]
- Bao, X.; Kolli, D.; Liu, T.; Shan, Y.; Garofalo, R.P.; Casola, A. Human metapneumovirus small hydrophobic protein inhibits NF-κB transcriptional activity. J. Virol. 2008, 82, 8224–8229. [Google Scholar] [CrossRef] [PubMed]
- Masante, C.; El Najjar, F.; Chang, A.; Jones, A.; Moncman, C.L.; Dutch, R.E. The human metapneumovirus small hydrophobic protein has properties consistent with those of a viroporin and can modulate viral fusogenic activity. J. Virol. 2014, 88, 6423–6433. [Google Scholar] [CrossRef]
- Biacchesi, S.; Skiadopoulos, M.H.; Yang, L.; Lamirande, E.W.; Tran, K.C.; Murphy, B.R.; Collins, P.L.; Buchholz, U.J. Recombinant human Metapneumovirus lacking the small hydrophobic SH and/or attachment G glycoprotein: Deletion of G yields a promising vaccine candidate. J. Virol. 2004, 78, 12877–12887. [Google Scholar] [CrossRef]
- Biacchesi, S.; Skiadopoulos, M.H.; Tran, K.C.; Murphy, B.R.; Collins, P.L.; Buchholz, U.J. Recovery of human metapneumovirus from cDNA: Optimization of growth in vitro and expression of additional genes. Virology 2004, 321, 247–259. [Google Scholar] [CrossRef]
- Aerts, L.; Cavanagh, M.H.; Dubois, J.; Carbonneau, J.; Rheaume, C.; Lavigne, S.; Couture, C.; Hamelin, M.E.; Boivin, G. Effect of in vitro syncytium formation on the severity of human metapneumovirus disease in a murine model. PLoS ONE 2015, 10, e0120283. [Google Scholar] [CrossRef]
- Dubois, J.; Cavanagh, M.H.; Terrier, O.; Hamelin, M.E.; Lina, B.; Shi, R.; Rosa-Calatrava, M.; Boivin, G. Mutations in the fusion protein heptad repeat domains of human metapneumovirus impact on the formation of syncytia. J. Gen. Virol. 2017, 98, 1174–1180. [Google Scholar] [CrossRef][Green Version]
- Nao, N.; Sato, K.; Yamagishi, J.; Tahara, M.; Nakatsu, Y.; Seki, F.; Katoh, H.; Ohnuma, A.; Shirogane, Y.; Hayashi, M.; et al. Consensus and variations in cell line specificity among human metapneumovirus strains. PLoS ONE 2019, 14, e0215822. [Google Scholar] [CrossRef] [PubMed]
- Mas, V.; Herfst, S.; Osterhaus, A.D.; Fouchier, R.A.; Melero, J.A. Residues of the human metapneumovirus fusion (F) protein critical for its strain-related fusion phenotype: Implications for the virus replication cycle. J. Virol. 2011, 85, 12650–12661. [Google Scholar] [CrossRef]
- Herfst, S.; Mas, V.; Ver, L.S.; Wierda, R.J.; Osterhaus, A.D.; Fouchier, R.A.; Melero, J.A. Low-pH-induced membrane fusion mediated by human metapneumovirus F protein is a rare, strain-dependent phenomenon. J. Virol. 2008, 82, 8891–8895. [Google Scholar] [CrossRef]
- Le, V.B.; Dubois, J.; Couture, C.; Cavanagh, M.H.; Uyar, O.; Pizzorno, A.; Rosa-Calatrava, M.; Hamelin, M.E.; Boivin, G. Human metapneumovirus activates NOD-like receptor protein 3 inflammasome via its small hydrophobic protein which plays a detrimental role during infection in mice. PLoS Pathog. 2019, 15, e1007689. [Google Scholar] [CrossRef] [PubMed]
- Hamelin, M.E.; Yim, K.; Kuhn, K.H.; Cragin, R.P.; Boukhvalova, M.; Blanco, J.C.; Prince, G.A.; Boivin, G. Pathogenesis of human metapneumovirus lung infection in BALB/c mice and cotton rats. J. Virol. 2005, 79, 8894–8903. [Google Scholar] [CrossRef] [PubMed]
- Meng, J.; Hotard, A.L.; Currier, M.G.; Lee, S.; Stobart, C.C.; Moore, M.L. Respiratory Syncytial Virus Attachment Glycoprotein Contribution to Infection Depends on the Specific Fusion Protein. J. Virol. 2015, 90, 245–253. [Google Scholar] [CrossRef]
- Nicolas de Lamballerie, C.; Pizzorno, A.; Dubois, J.; Julien, T.; Padey, B.; Bouveret, M.; Traversier, A.; Legras-Lachuer, C.; Lina, B.; Boivin, G.; et al. Characterization of cellular transcriptomic signatures induced by different respiratory viruses in human reconstituted airway epithelia. Sci. Rep. 2019, 9, 11493. [Google Scholar] [CrossRef]
- Mayor, S. Acute respiratory infections are world’s third leading cause of death. BMJ 2010, 341, c6360. [Google Scholar] [CrossRef]
- Vekemans, J.; Moorthy, V.; Giersing, B.; Friede, M.; Hombach, J.; Arora, N.; Modjarrad, K.; Smith, P.G.; Karron, R.; Graham, B.; et al. Respiratory syncytial virus vaccine research and development: World Health Organization technological roadmap and preferred product characteristics. Vaccine 2018. [Google Scholar] [CrossRef]
- Karron, R.A.; San Mateo, J.; Wanionek, K.; Collins, P.L.; Buchholz, U.J. Evaluation of a Live Attenuated Human Metapneumovirus Vaccine in Adults and Children. J. Pediatr. Infect. Dis. Soc. 2017, 7, 86–89. [Google Scholar] [CrossRef]
- Biacchesi, S.; Pham, Q.N.; Skiadopoulos, M.H.; Murphy, B.R.; Collins, P.L.; Buchholz, U.J. Infection of nonhuman primates with recombinant human metapneumovirus lacking the SH, G, or M2-2 protein categorizes each as a nonessential accessory protein and identifies vaccine candidates. J. Virol. 2005, 79, 12608–12613. [Google Scholar] [CrossRef] [PubMed]
- Bastien, N.; Liu, L.; Ward, D.; Taylor, T.; Li, Y. Genetic variability of the G glycoprotein gene of human metapneumovirus. J. Clin. Microbiol. 2004, 42, 3532–3537. [Google Scholar] [CrossRef] [PubMed]
- Piyaratna, R.; Tollefson, S.J.; Williams, J.V. Genomic analysis of four human metapneumovirus prototypes. Virus Res. 2011, 160, 200–205. [Google Scholar] [CrossRef] [PubMed]
- Schildgen, O.; Simon, A.; Williams, J. Animal models for human metapneumovirus (HMPV) infections. Veterinary research 2007, 38, 117–126. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Cornell, T.T.; LeVine, A.; Berlin, A.A.; Hinkovska-Galcheva, V.; Fleszar, A.J.; Lukacs, N.W.; Shanley, T.P. Dual role of interleukin-10 in the regulation of respiratory syncitial virus (RSV)-induced lung inflammation. Clin. Exp. Immunol. 2013, 172, 263–279. [Google Scholar] [CrossRef] [PubMed]
- Malashchenko, V.V.; Meniailo, M.E.; Shmarov, V.A.; Gazatova, N.D.; Melashchenko, O.B.; Goncharov, A.G.; Seledtsova, G.V.; Seledtsov, V.I. Direct anti-inflammatory effects of granulocyte colony-stimulating factor (G-CSF) on activation and functional properties of human T cell subpopulations in vitro. Cell. Immunol. 2018, 325, 23–32. [Google Scholar] [CrossRef]
- Kim, H.W.; Canchola, J.G.; Brandt, C.D.; Pyles, G.; Chanock, R.M.; Jensen, K.; Parrott, R.H. Respiratory syncytial virus disease in infants despite prior administration of antigenic inactivated vaccine. Am. J. Epidemiol. 1969, 89, 422–434. [Google Scholar] [CrossRef]
| Target | Virus Background | Forward Primer | Reverse Primer |
|---|---|---|---|
| SH deletion | A1/C-85473 | 5′-GGGACAAGTAGTTATGGA-3′ | 5′-ACTCTGATGTGTTTTTACTAAC-3′ |
| B2/CAN98-75 | 5′-GGGACAAGTGGCCATG-3′ | 5′-ATTCTATTTATTTTTTACT AACTTAAGTAAGCTT-3′ | |
| G deletion | A1/C-85473 | 5′-GAGACAAATAACAATGGATC-3′ | 5′-ATATTATTGTTTTTACTTCTTAAATT-3′ |
| B2/CAN98-75 | 5′-GGGACAAATAACAATGGATCCG TTTGTGAATCC-3′ | 5′-ATTTTCTTGTTTTGTA TTACTCAGTGATTG-3′ | |
| N gene amplification | A1/C-85473 B2/CAN98-75 | 5′-AGAGTCTCAATACACAATAAA AAGAGATGTAGG-3′ | 5′-CCTATCTCTGCAGCA TATTTGTAATCAG-3′ |
| Imm. virus | Imm. Inoculum (log10 TCID50) | Reciprocal Neutralization Titer 21 Days Post-Challenge against Different HMPV Strains (n = 4) 1 | Challenge Virus Replication in Lungs Five Days Post-Challenge (n = 4) | |||
|---|---|---|---|---|---|---|
| rC-85473 | WT C-85473 | WT CAN98-75 | Mice with Detectable Virus (%) | Mean Virus Titer (log10 N copies/µg RNA) | ||
| mock | 10 | 7.5 | 12.5 | 100 | 4.8 ± 4.3 | |
| rC-85473 | 5.7 | >160 | 80 | 60 | 0 | <1 |
| ΔSH-C-85473 | 5.7 | >160 | >160 | >160 | 0 | <1 |
| ΔG-C-85473 | 5.7 | >160 | 80 | 70 | 0 | <1 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dubois, J.; Pizzorno, A.; Cavanagh, M.-H.; Padey, B.; Nicolas de Lamballerie, C.; Uyar, O.; Venable, M.-C.; Carbonneau, J.; Traversier, A.; Julien, T.; et al. Strain-Dependent Impact of G and SH Deletions Provide New Insights for Live-Attenuated HMPV Vaccine Development. Vaccines 2019, 7, 164. https://doi.org/10.3390/vaccines7040164
Dubois J, Pizzorno A, Cavanagh M-H, Padey B, Nicolas de Lamballerie C, Uyar O, Venable M-C, Carbonneau J, Traversier A, Julien T, et al. Strain-Dependent Impact of G and SH Deletions Provide New Insights for Live-Attenuated HMPV Vaccine Development. Vaccines. 2019; 7(4):164. https://doi.org/10.3390/vaccines7040164
Chicago/Turabian StyleDubois, Julia, Andrés Pizzorno, Marie-Hélène Cavanagh, Blandine Padey, Claire Nicolas de Lamballerie, Olus Uyar, Marie-Christine Venable, Julie Carbonneau, Aurélien Traversier, Thomas Julien, and et al. 2019. "Strain-Dependent Impact of G and SH Deletions Provide New Insights for Live-Attenuated HMPV Vaccine Development" Vaccines 7, no. 4: 164. https://doi.org/10.3390/vaccines7040164
APA StyleDubois, J., Pizzorno, A., Cavanagh, M.-H., Padey, B., Nicolas de Lamballerie, C., Uyar, O., Venable, M.-C., Carbonneau, J., Traversier, A., Julien, T., Lavigne, S., Couture, C., Lina, B., Hamelin, M.-È., Terrier, O., Rosa-Calatrava, M., & Boivin, G. (2019). Strain-Dependent Impact of G and SH Deletions Provide New Insights for Live-Attenuated HMPV Vaccine Development. Vaccines, 7(4), 164. https://doi.org/10.3390/vaccines7040164





