Comparative Immunogenicity of the 2014–2015 Northern Hemisphere Trivalent IIV and LAIV against Influenza A Viruses in Children
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Participants
2.2. Sample Collection
2.3. HAI
2.4. Enzyme-Linked Immunosorbent Assays (ELISAs)
2.5. Statistical Analyses
3. Results
3.1. Participant Characteristics
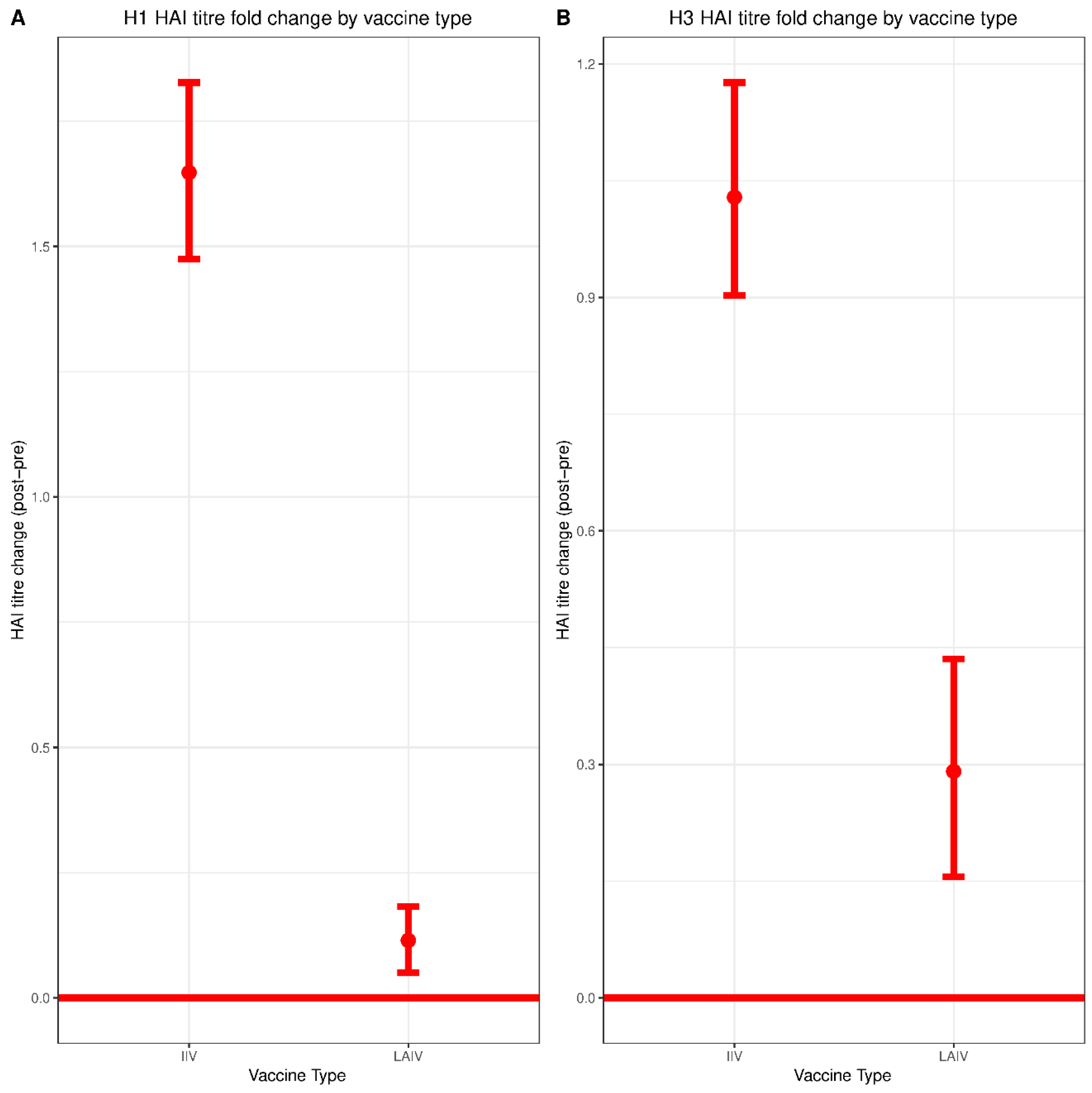
3.2. Serum HAI Responses to IIV and LAIV
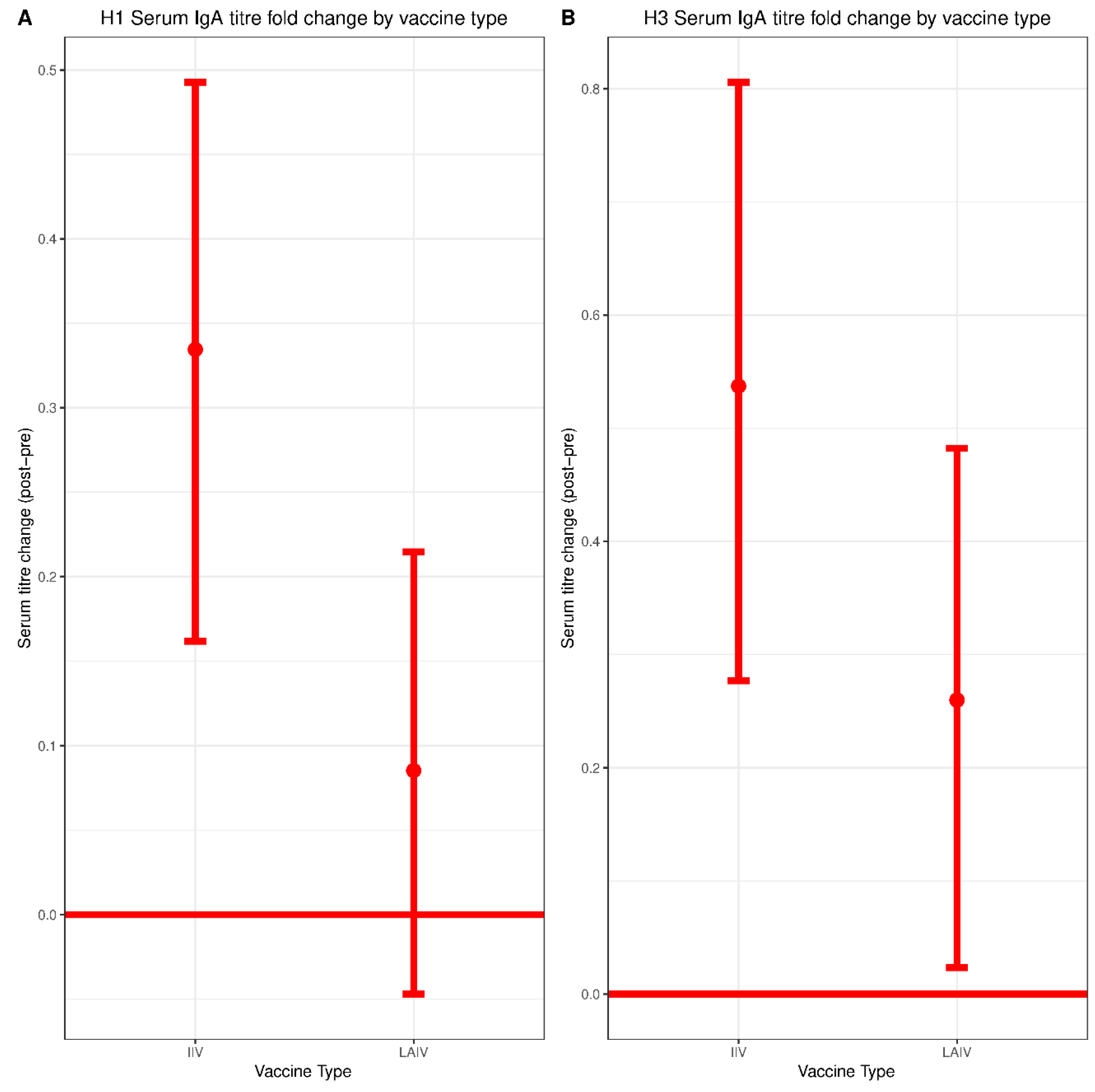
3.3. Serum IgA Responses to IIV and LAIV
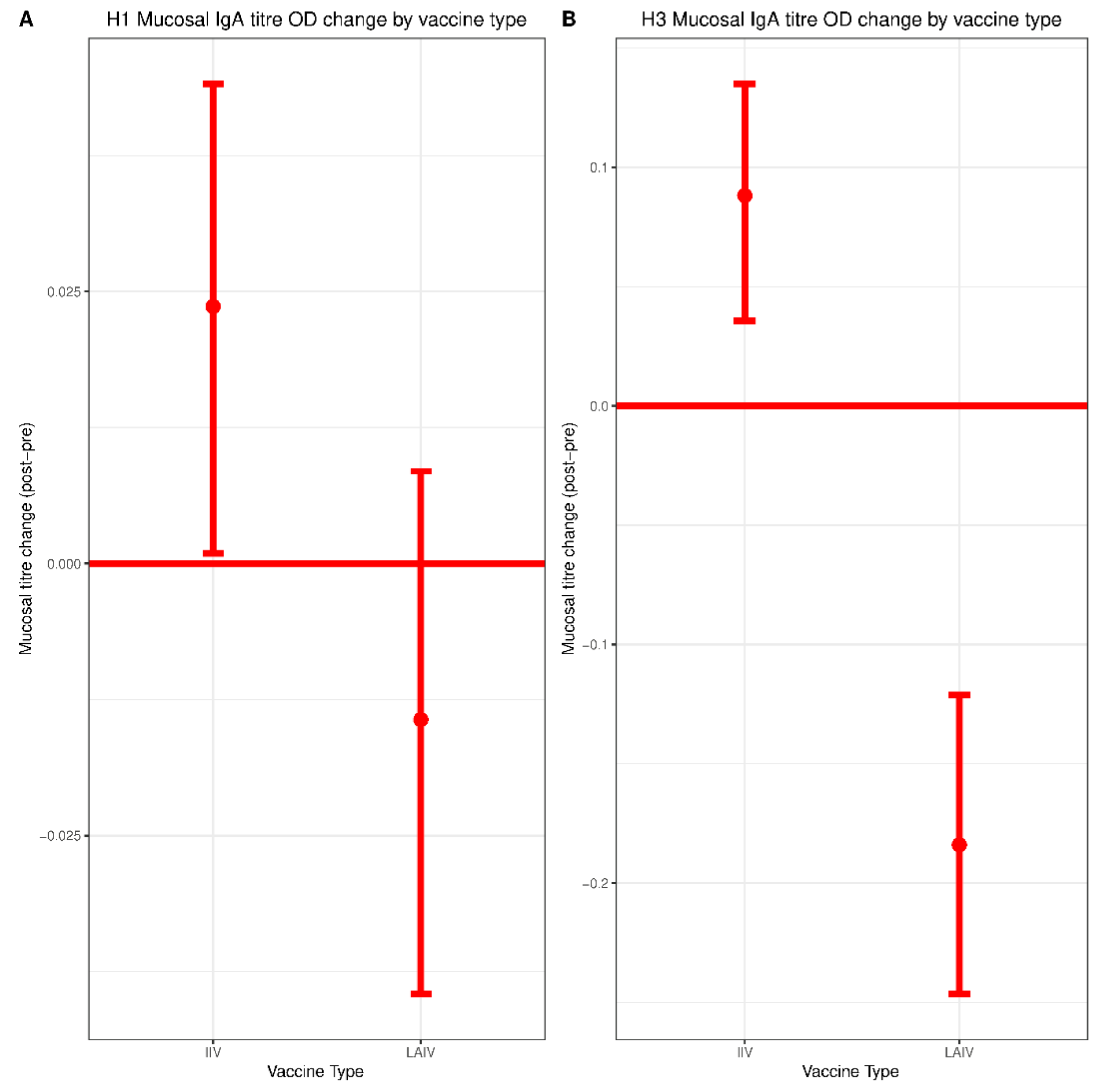
3.4. Mucosal Responses to IIV and LAIV
3.5. Variables Associated with Antibody Response to Vaccination
3.6. Correlates of Protection against H3N2 Infection
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- WHO|Influenza. Available online: http://www.who.int/mediacentre/factsheets/2003/fs211/en/ (accessed on 16 October 2013).
- Thompson, W.W.; Shay, D.K.; Weintraub, E.; Brammer, L.; Cox, N.; Anderson, L.J.; Fukuda, K. Mortality associated with influenza and respiratory syncytial virus in the United States. JAMA 2003, 289, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Lafond, K.E.; Nair, H.; Rasooly, M.H.; Valente, F.; Booy, R.; Rahman, M.; Kitsutani, P.; Yu, H.; Guzman, G.; Coulibaly, D.; et al. Global Role and Burden of Influenza in Pediatric Respiratory Hospitalizations, 1982–2012: A Systematic Analysis. PLoS Med. 2016, 13, e1001977. [Google Scholar] [CrossRef] [PubMed]
- Foy, H.M.; Cooney, M.K.; Allan, I. Longitudinal studies of types A and B influenza among Seattle schoolchildren and families, 1968–1974. J. Infect. Dis. 1976, 134, 362–369. [Google Scholar] [CrossRef] [PubMed]
- Fox, J.P.; Hall, C.E.; Cooney, M.K.; Foy, H.M. Influenzavirus infections in Seattle families, 1975–1979. I. Study design, methods and the occurrence of infections by time and age. Am. J. Epidemiol. 1982, 116, 212–227. [Google Scholar] [CrossRef] [PubMed]
- Monto, A.S.; Koopman, J.S.; Longini, I.M. Tecumseh study of illness. XIII. Influenza infection and disease, 1976–1981. Am. J. Epidemiol. 1985, 121, 811–822. [Google Scholar] [CrossRef] [PubMed]
- Neuzil, K.M.; Hohlbein, C.; Zhu, Y. Illness among schoolchildren during influenza season: Effect on school absenteeism, parental absenteeism from work, and secondary illness in families. Arch. Pediatr. Adolesc. Med. 2002, 156, 986–991. [Google Scholar] [CrossRef]
- Administration, U.S.F. and D. CBER Approval Letter, Influenza Virus Vaccine, Live, Intranasal (Flumist). Available online: https://web.archive.org/web/20070929154324/http://www.fda.gov/cber/approvltr/inflmed061703L.htm (accessed on 14 January 2018).
- Rhorer, J.; Ambrose, C.S.; Dickinson, S.; Hamilton, H.; Oleka, N.A.; Malinoski, F.J.; Wittes, J. Efficacy of live attenuated influenza vaccine in children: A meta-analysis of nine randomized clinical trials. Vaccine 2009, 27, 1101–1110. [Google Scholar] [CrossRef]
- Belshe, R.B.; Edwards, K.M.; Vesikari, T.; Black, S.V.; Walker, R.E.; Hultquist, M.; Kemble, G.; Connor, E.M. Live Attenuated versus Inactivated Influenza Vaccine in Infants and Young Children. N. Engl. J. Med. 2007, 356, 685–696. [Google Scholar] [CrossRef]
- Grohskopf, L.A.; Olsen, S.J.; Sokolow, L.Z.; Bresee, J.S.; Cox, N.J.; Broder, K.R.; Karron, R.A.; Walter, E.B. Prevention and Control of Influenza with Vaccines: Recommendations of the Advisory Committee on Immunization Practices (ACIP)—United States, 2014–2015 Influenza Season. Morb. Mortal. Wkly. Rep. 2014, 63, 691–697. [Google Scholar]
- Grohskopf, L.A.; Sokolow, L.Z.; Olsen, S.J.; Bresee, J.S.; Broder, K.R.; Karron, R.A. Prevention and Control of Influenza with Vaccines: Recommendation of the Advisory Committee on Immunization Practices, United States, 2015–2016 Influenza Season. Morb. Mortal. Wkly. Rep. 2015, 64, 818–825. [Google Scholar] [CrossRef]
- Grohskopf, L.A.; Sokolow, L.Z.; Broder, K.R.; Olsen, S.J.; Karron, R.A.; Jernigan, D.B.; Bresee, J.S. Prevention and Control of Seasonal Influenza with Vaccines. Recommendations of the Advisory Committee on Immunization Practices - United States, 2016–2017 Influenza Season. Morb. Mortal. Wkly. Rep. 2016, 65, 1–54. [Google Scholar]
- Loeb, M.; Russell, M.L.; Manning, V.; Fonseca, K.; Earn, D.J.D.; Horsman, G.; Chokani, K.; Vooght, M.; Babiuk, L.; Schwartz, L.; et al. Live Attenuated Versus Inactivated Influenza Vaccine in Hutterite Children. Ann. Intern. Med. 2016, 165, 617–624. [Google Scholar] [CrossRef] [PubMed]
- Grohskopf, L.A.; Sokolow, L.Z.; Fry, A.M.; Walter, E.B.; Jernigan, D.B. Update: ACIP Recommendations for the Use of Quadrivalent Live Attenuated Influenza Vaccine (LAIV4)—United States, 2018–2019 Influenza Season. MMWR. Morb. Mortal. Wkly. Rep. 2018, 67, 643–645. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Russell, M.L.; Brewer, A.; Newton, J.; Singh, P.; Ward, B.J.; Loeb, M. Single radial haemolysis compared to haemagglutinin inhibition and microneutralization as a correlate of protection against influenza A H3N2 in children and adolescents. Influenza Other Respi. Viruses 2017, 11, 283–288. [Google Scholar] [CrossRef] [PubMed]
- FluWatch Report: December 21, 2014 to January 3, 2015 (Weeks 52–53)—Canada.ca. Available online: https://www.canada.ca/en/public-health/services/publications/diseases-conditions/fluwatch-report-december-21-2014-january-3-2015-weeks-52-53.html (accessed on 11 July 2018).
- Cox, R.J. Correlates of protection to influenza virus, where do we go from here? Hum. Vaccin. Immunother. 2013, 9, 405–408. [Google Scholar] [CrossRef] [PubMed]
- Hoft, D.F.; Lottenbach, K.R.; Blazevic, A.; Turan, A.; Blevins, T.P.; Pacatte, T.P.; Yu, Y.; Mitchell, M.C.; Hoft, S.G.; Belshe, R.B. Comparisons of the Humoral and Cellular Immune Responses Induced by Live Attenuated Influenza Vaccine and Inactivated Influenza Vaccine in Adults. Clin. Vaccine Immunol. 2017, 24, e00414–e00416. [Google Scholar] [CrossRef] [PubMed]
- Belshe, R.B.; Gruber, W.C.; Mendelman, P.M.; Mehta, H.B.; Mahmood, K.; Reisinger, K.; Treanor, J.; Zangwill, K.; Hayden, F.G.; Bernstein, D.I.; et al. Correlates of Immune Protection Induced by Live, Attenuated, Cold-Adapted, Trivalent, Intranasal Influenza Virus Vaccine. J. Infect. Dis. 2000, 181, 1133–1137. [Google Scholar] [CrossRef]
- Barría, M.I.; Garrido, J.L.; Stein, C.; Scher, E.; Ge, Y.; Engel, S.M.; Kraus, T.A.; Banach, D.; Moran, T.M. Localized mucosal response to intranasal live attenuated influenza vaccine in adults. J. Infect. Dis. 2013, 207, 115–124. [Google Scholar] [CrossRef]
- Loeb, M.; Russell, M.L.; Moss, L.; Fonseca, K.; Fox, J.; Earn, D.J.D.; Aoki, F.; Horsman, G.; Van Caeseele, P.; Chokani, K.; et al. Effect of Influenza Vaccination of Children on Infection Rates in Hutterite Communities. JAMA 2010, 303, 943–950. [Google Scholar] [CrossRef]
- Iuliano, A.D.; Roguski, K.M.; Chang, H.H.; Muscatello, D.J.; Palekar, R.; Tempia, S.; Cohen, C.; Gran, J.M.; Schanzer, D.; Cowling, B.J.; et al. Estimates of global seasonal influenza-associated respiratory mortality: A modelling study. Lancet 2018, 391, 1285–1300. [Google Scholar] [CrossRef]
- Edwards, K.M.; Dupont, W.D.; Westrich, M.K.; Plummer, W.D.; Palmer, P.S.; Wright, P.F. A Randomized Controlled Trial of Cold-Adapted and Inactivated Vaccines for the Prevention of Influenza A Disease. J. Infect. Dis. 1994, 169, 68–76. [Google Scholar] [CrossRef] [PubMed]
- Black, S.; Nicolay, U.; Vesikari, T.; Knuf, M.; Del Giudice, G.; Della Cioppa, G.; Tsai, T.; Clemens, R.; Rappuoli, R. Hemagglutination Inhibition Antibody Titers as a Correlate of Protection for Inactivated Influenza Vaccines in Children. Pediatr. Infect. Dis. J. 2011, 30, 1081–1085. [Google Scholar] [CrossRef] [PubMed]
- Rose, M.A.; Zielen, S.; Baumann, U. Mucosal immunity and nasal influenza vaccination. Expert Rev. Vaccines 2012, 11, 595–607. [Google Scholar] [CrossRef] [PubMed]
- Gorse, G.J.; Otto, E.E.; Powers, D.C.; Chambers, G.W.; Eickhoff, C.S.; Newman, F.K. Induction of Mucosal Antibodies by Live Attenuated and Inactivated Influenza Virus Vaccines in the Chronically III Elderly. J. Infect. Dis. 1996, 173, 285–290. [Google Scholar] [CrossRef] [PubMed]
- Mohn, K.G.-I.; Smith, I.; Sjursen, H.; Cox, R.J. Immune responses after live attenuated influenza vaccination. Hum. Vaccin. Immunother. 2018, 14, 571–578. [Google Scholar] [CrossRef] [PubMed]
- Miller, M.S.; Palese, P. Peering into the crystal ball: Influenza pandemics and vaccine efficacy. Cell 2014, 157, 294–299. [Google Scholar] [CrossRef]
- Brickley, E.B.; Wright, P.F.; Khalenkov, A.; Neuzil, K.M.; Ortiz, J.R.; Rudenko, L.; Levine, M.Z.; Katz, J.M.; Brooks, W.A. The effect of pre-existing immunity on virus detection and immune responses in a phase II randomized trial of a Russian-backbone live attenuated influenza vaccine in Bangladeshi children. Clin. Infect. Dis. 2018. [Google Scholar] [CrossRef]
- Stacey, H.; Barjesteh, N.; Mapletoft, J.; Miller, M. “Gnothi Seauton”: Leveraging the Host Response to Improve Influenza Virus Vaccine Efficacy. Vaccines 2018, 6, 23. [Google Scholar] [CrossRef]
- Dougan, S.K.; Ashour, J.; Karssemeijer, R.A.; Popp, M.W.; Avalos, A.M.; Barisa, M.; Altenburg, A.F.; Ingram, J.R.; Cragnolini, J.J.; Guo, C.; et al. Antigen-specific B-cell receptor sensitizes B cells to infection by influenza virus. Nature 2013, 503, 406–409. [Google Scholar] [CrossRef]
- Mohn, K.G.I.; Zhou, F.; Brokstad, K.A.; Sridhar, S.; Cox, R.J. Boosting of Cross-Reactive and Protection-Associated T Cells in Children After Live Attenuated Influenza Vaccination. J. Infect. Dis. 2017, 215, 1527–1535. [Google Scholar] [CrossRef]
- Eichelberger, M.C.; Rivers, K.H.; Ream, R.; Gao, J.; Hassantoufighi, A.S.; Bulte, M.R.; Straight, T.M. Qualitative Differences in T cell responses to Live, Attenuated and Inactivated Influenza Vaccines. J. Clin. Cell. Immunol. 2012, 1, 1–8. [Google Scholar] [CrossRef]
- McElhaney, J.E.; Ewen, C.; Zhou, X.; Kane, K.P.; Xie, D.; Hager, W.D.; Barry, M.B.; Kleppinger, A.; Wang, Y.; Bleackley, R.C. Granzyme B: Correlates with protection and enhanced CTL response to influenza vaccination in older adults. Vaccine 2009, 27, 2418–2425. [Google Scholar] [CrossRef] [PubMed]
| Variable | Vaccination Type | ||
|---|---|---|---|
| IIV | LAIV | ||
| Number of Participants | 278 | 340 | |
| Previously Study-Vaccinated | 241 (87%) | 294 (86%) | |
| Number of Infections (H3N2) | 11 (3.9%) | 8 (2.4%) | |
| Age, mean(SD) | 9.3 (3.3) | 9.4 (3.2) | |
| 3–9 years, No. (%) | 143 (51.4%) | 170 (50%) | |
| 10–15 years, No. (%) | 135 (48.6%) | 170 (50%) | |
| Female Sex | 151 (54.3%) | 177 (52.1%) | |
| Pre and Post vaccination antibody titer for H1N1 | |||
| HAI (log2 titer) | |||
| Pre, mean (SD) | 4.67 (2.07) | 4.17 (1.91) | |
| Post, mean (SD) | 6.32 (1.59) | 4.29 (1.87) | |
| Serum IgA (log2 titer) | |||
| Pre, mean (SD) | 2.90 (2.51) | 2.88 (2.27) | |
| Post, mean (SD) | 3.23 (2.74) | 2.96 (2.36) | |
| Mucosal IgA (OD) | |||
| Pre, mean(SD) | 0.08 (0.16) | 0.11 (0.25) | |
| Post, mean(SD) | 0.10 (0.14) | 0.10 (0.14) | |
| Pre and Post vaccination antibody titer for H3N2 | |||
| HAI (log2 titer) | |||
| Pre, mean (SD) | 5.29 (1.69) | 4.23 (1.83) | |
| Post, mean (SD) | 6.32 (1.14) | 4.52 (1.76) | |
| Serum IgA (log2 titer) | |||
| Pre, mean (SD) | 4.25 (2.76) | 4.12 (2.50) | |
| Post, mean (SD) | 4.79 (2.79) | 4.38 (2.59) | |
| Mucosal IgA (OD) | |||
| Pre, mean (SD) | 0.21 (0.33) | 0.38 (0.50) | |
| Post, mean (SD) | 0.29 (0.30) | 0.21 (0.25) | |
| Antibody Measures | |||||||
|---|---|---|---|---|---|---|---|
| Variable | HAI Titer Change (log) | Serum IgA Titer Change (log) | Mucosal IgA Change (OD, ^0.3) | ||||
| β (95% CI) | p-Value | β (95% CI) | p-Value | β (95% CI) | p-Value | ||
| Vaccine Type | LAIV vs. IIV | −1.66 (−1.81 to −1.50) | <0.01 | −0.25 (−0.46 to −0.04) | 0.02 | ||
| Pre vaccination titer | Per 1-unit | −0.25 (−0.29 to −0.21) | <0.01 | −0.09 (−0.14 to −0.05) | <0.01 | −0.71 (−0.78 to −0.64) | <0.01 |
| Age | Per 1 year | −0.03 (−0.06 to −0.01) | <0.01 | ||||
| Antibody Measures | |||||||
|---|---|---|---|---|---|---|---|
| Variable | HAI Titer Change (Log) | Serum IgA Titer Change (Log) | Mucosal IgA Change (OD, ^0.3) | ||||
| β (95% CI) | p-Value | β (95% CI) | p-Value | β (95% CI) | p-Value | ||
| Vaccine Type | LAIV vs. IIV | −0.29 (−0.34 to −0.24) | <0.01 | −0.33 (−0.66 to 0.00) | 0.05 | −0.10 (−0.14 to −0.06) | <0.01 |
| Pre vaccination titer | Per 1-unit | −0.30 (−0.34 to −0.25) | <0.01 | −0.35 (−0.41 to −0.28) | <0.01 | −0.94 (−0.10 to −0.87) | <0.01 |
| Age | Per 1 year | −0.05 (−0.07 to −0.02) | <0.01 | ||||
| Cut-Off | Titer Value | IIV | LAIV | ||||
|---|---|---|---|---|---|---|---|
| Protectiveness (95% CI) | p Value | Infected above the Cut-Off † | Protectiveness (95% CI) | p Value | Infected above the Cut-Off ‡ | ||
| 3 | 20 | 11 | 60 (−180 to 94) | 0.36 | 6 | ||
| 4 | 40 | 81 (−41 to 98) | 0.10 | 10 | 49 (−392 to 95) | 0.56 | 5 |
| 5 | 80 | 77 (44 to 90) | <0.01 | 9 | 21 (−373 to 87) | 0.80 | 4 |
| 6 | 160 | 76 (7 to 94) | 0.04 | 5 | −52 (−654 to 69) | 0.61 | 3 |
| 7 | 320 | 39 (−34 to 72) | 0.22 | 4 | −187 (−1529 to 49) | 0.23 | 2 |
| 8 | 640 | 40 (−164 to 87) | 0.50 | 1 | −443 (−3794 to 24) | 0.09 | 1 |
| Cut-Off | Titer Value | IIV | LAIV | ||||
|---|---|---|---|---|---|---|---|
| Protectiveness (95% CI) | p Value | Infected above the Cut-Off † | Protectiveness (95% CI) | p Value | Infected above the Cut-Off ‡ | ||
| 2 | 50 | 11 (−245 to 77) | 0.86 | 10 | 13 (−482 to 87) | 0.89 | 7 |
| 3 | 100 | 25 (−91 to 70) | 0.55 | 7 | −41 (−562 to 70) | 0.66 | 6 |
| 4 | 200 | 41 (−100 to 83) | 0.40 | 5 | 15 (−218 to 77) | 0.82 | 4 |
| 5 | 400 | 63 (−53 to 91) | 0.17 | 3 | 25 (−166 to 79) | 0.66 | 3 |
| 6 | 800 | 40 (−140 to 85) | 0.47 | 3 | −24 (−342 to 65) | 0.74 | 3 |
| 7 | 1600 | 41 (−111 to 83) | 0.42 | 2 | −22 (−292 to 62) | 0.74 | 2 |
| 8 | 3200 | 11 (−226 to 76) | 0.86 | 2 | −115 (−567 to 31) | 0.18 | 2 |
| 9 | 6400 | −20 (−366 to 69) | 0.80 | 2 | −9 (−377 to 75) | 0.91 | 1 |
| 10 | 12,800 | −202 (−1183 to 29) | 0.14 | 2 | |||
| Cut-Off -Titer Value | IIV | LAIV | ||||
|---|---|---|---|---|---|---|
| Protectiveness (95% CI) | p Value | Infected above the Cut-Off † | Protectiveness (95% CI) | p Value | Infected above the Cut-Off ‡ | |
| 0.0001 | N.D. | N.D. | 10 | 88 (54 to 97) | 0.00 | 4 |
| 0.001 | N.D. | N.D. | 10 | 87 (48 to 97) | 0.00 | 4 |
| 0.01 | N.D. | N.D. | 10 | 88 (18 to 98) | 0.03 | 3 |
| 0.1 | N.D. | N.D. | 10 | 100 (100 to 100) | 0.00 | 0 |
| 1 | 100 (100 to 100) | 0.00 | 0 | 100 (100 to 100) | 0.00 | 0 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ang, J.C.; Wang, B.; Wang, J.J.F.; Zeng, P.Y.F.; Krammer, F.; Ward, B.J.; Russell, M.L.; Loeb, M.; Miller, M.S. Comparative Immunogenicity of the 2014–2015 Northern Hemisphere Trivalent IIV and LAIV against Influenza A Viruses in Children. Vaccines 2019, 7, 87. https://doi.org/10.3390/vaccines7030087
Ang JC, Wang B, Wang JJF, Zeng PYF, Krammer F, Ward BJ, Russell ML, Loeb M, Miller MS. Comparative Immunogenicity of the 2014–2015 Northern Hemisphere Trivalent IIV and LAIV against Influenza A Viruses in Children. Vaccines. 2019; 7(3):87. https://doi.org/10.3390/vaccines7030087
Chicago/Turabian StyleAng, Jann Catherine, Biao Wang, Joanne J.F. Wang, Peter Yu Fan Zeng, Florian Krammer, Brian J. Ward, Margaret L. Russell, Mark Loeb, and Matthew S. Miller. 2019. "Comparative Immunogenicity of the 2014–2015 Northern Hemisphere Trivalent IIV and LAIV against Influenza A Viruses in Children" Vaccines 7, no. 3: 87. https://doi.org/10.3390/vaccines7030087
APA StyleAng, J. C., Wang, B., Wang, J. J. F., Zeng, P. Y. F., Krammer, F., Ward, B. J., Russell, M. L., Loeb, M., & Miller, M. S. (2019). Comparative Immunogenicity of the 2014–2015 Northern Hemisphere Trivalent IIV and LAIV against Influenza A Viruses in Children. Vaccines, 7(3), 87. https://doi.org/10.3390/vaccines7030087






