Reduction in Oviposition of Poultry Red Mite (Dermanyssus gallinae) in Hens Vaccinated with Recombinant Akirin
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Mites
2.3. Cloning of the Gene Coding for Deg-AKR
2.4. Production of Recombinant Deg-AKR and Vaccine Formulation
2.5. Hen Vaccination and PRM Infestation
2.6. Assessment of Vaccine Efficacy
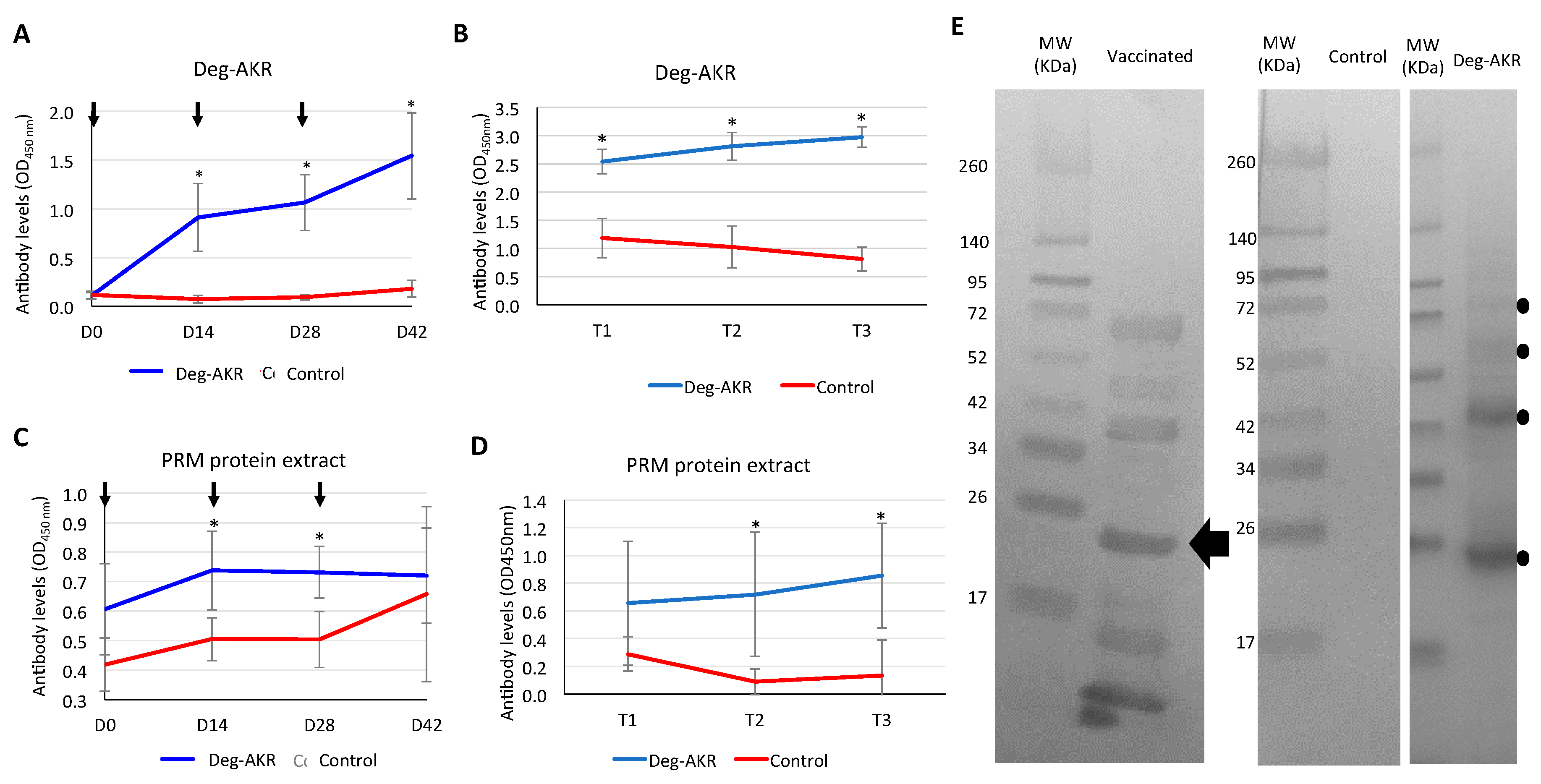
2.7. Analysis of Immune Response to Deg-AKR in Hens by ELISA
2.8. Analysis of Hen Immune Response to Deg-AKR by Western Blot
2.9. Statistical Analysis
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Van Emous, R.A. Verwachtte Schade Bloedluis 21 Miljoen Euro. Pluimveeweb 2017. Available online: https://www.pluimveeweb.nl/artikelen/2017/01/schade-bloedluis-21-miljoen-euro (accessed on 5 January 2018).
- Sparagano, O.A.E.; George, D.R.; Harrington, D.W.J.; Giangaspero, A. Significance and Control of the Poultry Red Mite, Dermanyssus gallinae. Annu. Rev. Entomol. 2014, 59, 447–466. [Google Scholar] [CrossRef] [PubMed]
- Maurer, V.; Bieri, M.; Foelsch, D.W. Das Suchverhalten von Dermanyssus gallinae in Huhnerstallen. Host-finding of Dermanyssus gallinae in poultry-houses. Eur. Poult. Sci. 1988, 52, 209–315. [Google Scholar]
- Koenraadt, C.J.M.; Dicke, M. The role of volatiles in aggregation and host-seeking of the haematophagous poultry red mite Dermanyssus gallinae (Acari: Dermanyssidae). Exp. Appl. Acarol. 2010, 50, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Maurer, V.; Baumgärtner, J. Temperature influence on life table statistics of the chicken mite Dermanyssus gallinae (Acari: Dermanyssidae). Exp. Appl. Acarol. 1992, 15, 27–40. [Google Scholar] [CrossRef] [PubMed]
- Cosoroaba, I. Massive Dermanyssus gallinae invasion in battery-husbandry raised fowls. Rev. Med. Vet. 2001, 152, 89–96. [Google Scholar]
- Kilpinen, O.; Roepstorff, A.; Permin, A.; Nørgaard-Nielsen, G.; Lawson, L.G.; Simonsen, H.B. Influence of Dermanyssus gallinae and Ascaridia galli infections on behaviour and health of laying hens (Gallus gallus domesticus). Br. Poult. Sci. 2005, 46, 26–34. [Google Scholar] [CrossRef]
- Sigognault Flochlay, A.; Thomas, E.; Sparagano, O.A.E. Poultry red mite (Dermanyssus gallinae) infestation: A broad impact parasitological disease that still remains a significant challenge for the egg-laying industry in Europe. Parasit Vectors 2017, 10, 357. [Google Scholar] [CrossRef] [PubMed]
- Sommer, D.; Heffels-Redmann, U.; Köhler, K.; Lierz, M.; Kaleta, E.F. Role of the poultry red mite (Dermanyssus gallinae) in the transmission of avian influenza A virus. Tierärztliche Prax Großtiere 2016, 1, 47–54. [Google Scholar]
- De Luna, C.J.; Moro, C.V.; Guy, J.H.; Zenner, L.; Sparagano, O.A.E. Endosymbiotic bacteria living inside the poultry red mite (Dermanyssus gallinae). Exp. Appl. Acarol. 2009, 48, 105–113. [Google Scholar] [CrossRef]
- Marangi, M.; Cafiero, M.A.; Capelli, G.; Camarda, A.; Sparagano, O.A.E.; Giangaspero, A. Evaluation of the poultry red mite, Dermanyssus gallinae (Acari: Dermanyssidae) susceptibility to some acaricides in field populations from Italy. Exp. Appl. Acarol. 2009, 48, 11–18. [Google Scholar] [CrossRef]
- Marangi, M.; Morelli, V.; Pati, S.; Camarda, A.; Cafiero, M.A.; Giangaspero, A. Acaricide residues in laying hens naturally infested by red mite Dermanyssus gallinae. PLoS ONE 2012, 7, e31795. [Google Scholar] [CrossRef] [PubMed]
- Bartley, K.; Huntley, J.F.; Wright, H.W.; Nath, M.; Nisbet, A.J. Assessment of cathepsin D and L-like proteinases of poultry red mite, Dermanyssus gallinae (De Geer), as potential vaccine antigens. Parasitology 2012, 139, 755–765. [Google Scholar] [CrossRef] [PubMed]
- Bartley, K.; Nisbet, A.J.; Offer, J.E.; Sparks, N.H.C.; Wright, H.W.; Huntley, J.F. Histamine Release Factor from Dermanyssus gallinae (De Geer): Characterization and in vitro assessment as a protective antigen. Int. J. Parasitol. 2009, 39, 447–456. [Google Scholar] [CrossRef] [PubMed]
- Harrington, D.W.J.; Canales, M.; de la Fuente, J.; de Luna, C.; Robinson, K.; Guy, J.; Sparagano, O.A.E. Immunisation with recombinant proteins subolesin and Bm86 for the control of Dermanyssus gallinae in poultry. Vaccine 2009, 27, 4056–4063. [Google Scholar] [CrossRef] [PubMed]
- Wright, H.W.; Bartley, K.; Huntley, J.F.; Nisbet, A.J. Characterisation of tropomyosin and paramyosin as vaccine candidate molecules for the poultry red mite, Dermanyssus gallinae. Parasit Vectors 2016, 9, 544. [Google Scholar] [CrossRef] [PubMed]
- Artigas-Jerónimo, S.; Villar, M.; Cabezas-Cruz, A.; Valdés, J.J.; Estrada-Peña, A.; Alberdi, P.; de la Fuente, J. Functional evolution of Subolesin/Akirin. Front. Physiol. 2018, 9, 1612. [Google Scholar] [CrossRef]
- Merino, O.; Alberdi, P.; Pérez de la Lastra, J.M.; de la Fuente, J. Tick vaccines and the control of tick-borne pathogens. Front. Cell Infect. Microbiol. 2013, 3, 30. [Google Scholar] [CrossRef]
- De la Fuente, J.; Moreno-Cid, J.A.; Galindo, R.C.; Almazan, C.; Kocan, K.M.; Merino, O.; Perez de la Lastra, J.M.; Estrada-Peña, A.; Blouin, E.F. Subolesin/Akirin Vaccines for the Control of Arthropod Vectors and Vectorborne Pathogens. Transbound Emerg. Dis. 2013, 60, 172–178. [Google Scholar] [CrossRef]
- Burgess, S.T.G.; Bartley, K.; Nunn, F.; Wright, H.W.; Hughes, M.; Gemmell, M.; Haldenby, S.; Paterson, S.; Rombauts, S.; Tomley, F.M.; et al. Draft Genome Assembly of the Poultry Red Mite, Dermanyssus gallinae. Microbiol. Resour. Announc. 2018, 7, e01221-18. [Google Scholar] [CrossRef]
- Nunn, F.; Bartley, K.; Palarea-Albaladejo, J.; Innocent, G.T.; Turnbull, F.; Wright, H.W.; Nisbet, A.J. A novel, high-welfare methodology for evaluating poultry red mite interventions in vivo. Vet. Parasitol. 2019, 267, 42–46. [Google Scholar] [CrossRef]
- Bartley, K.; Wright, H.W.; Huntley, J.F.; Manson, E.D.T.; Inglis, N.F.; McLean, K.; Nath, M.; Bartley, Y.; Nisbet, A.J. Identification and evaluation of vaccine candidate antigens from the poultry red mite (Dermanyssus gallinae). Int. J. Parasitol. 2015, 45, 819–830. [Google Scholar] [CrossRef] [PubMed]
- Madeira, F.; Park, Y.; Lee, J.; Buso, N.; Gur, T.; Madhusoodanan, N.; Basutkar, P.; Tivey, A.R.N.; Potter, S.C.; Finn, R.D.; et al. The EMBL-EBI search and sequence analysis tools APIs in 2019. Nucleic Acids Res. 2019, 47, W636–W641. [Google Scholar] [CrossRef] [PubMed]
- Waterhouse, A.M.; Procter, J.B.; Martin, D.M.A.; Clamp, M.; Barton, G.J. Jalview Version 2-a multiple sequence alignment editor and analysis workbench. Bioinformatics 2009, 25, 1189–1191. [Google Scholar] [CrossRef] [PubMed]
- Price, D.R.; Küster, T.; Øines, Ø.; Margaret Oliver, E.; Bartley, K.; Nunn, F.; Lima-Barbero, J.F.; Pritchard, J.; Karp-Tatham, E.; Hauge, H.; et al. Evaluation of vaccine delivery systems for inducing long-lived antibody responses to Dermanyssus gallinae in laying hens. Avian Pathol. 2019, 48 (sup1), S60–S74. [Google Scholar] [CrossRef] [PubMed]
- Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, T.; Rueden, C.; Saalfeld, S.; Schmid, B.; et al. Fiji: An open-source platform for biological-image analysis. Nat. Methods 2012, 9, 676–682. [Google Scholar] [CrossRef] [PubMed]
- Choi, K.S.; Lee, E.K.; Jeon, W.J.; Park, M.J.; Yoo, Y.N.; Kwon, J.H. Diagnostic utility of egg yolk for the detection of avian Metapneumovirus antibodies in laying hens. Avian Dis. 2010, 54, 1230–1236. [Google Scholar] [CrossRef] [PubMed]
- Lima-Barbero, J.F.; Sánchez-Sánchez, M.; Cabezas-Cruz, A.; Mateos-Hernández, L.; Contreras, M.; Fernández de Mera, I.G.; Villar, M.; de la Fuente, J. Clinical gamasoidosis and antibody response in two patients infested with Ornithonyssus bursa (Acari: Gamasida: Macronyssidae). Exp. Appl. Acarol. 2019, 78, 555. [Google Scholar] [CrossRef] [PubMed]
- Contreras, M.; de la Fuente, J. Control of infestations by Ixodes ricinus tick larvae in rabbits vaccinated with aquaporin recombinant antigens. Vaccine 2017, 35, 1323–1328. [Google Scholar] [CrossRef]
- De la Fuente, J.; Estrada-Peña, A. Why new vaccines for the control of ectoparasite vectors have not been registered and commercialized? Vaccines 2019, 7, 75. [Google Scholar] [CrossRef]
- De la Fuente, J.; Contreras, M. Tick vaccines: Current status and future directions. Expert Rev. Vaccines 2015, 14, 1367–1376. [Google Scholar] [CrossRef]
- Nordenfors, H.; Höglung, J.; Uggla, A. Effects of Temperature and Humidity on Oviposition, Molting, and Longevity of Dermanyssus gallinae (Acari: Dermanyssidae). J. Med. Entomol. 1999, 1, 68–72. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Ma, Y.; Huang, Y.; Xu, J.; Cai, J.; Pan, B. An efficient rearing system rapidly producing large quantities of poultry red mites, Dermanyssus gallinae (Acari: Dermanyssidae) under laboratory conditions. Vet. Parasitol. 2018, 258, 38–45. [Google Scholar] [CrossRef] [PubMed]
- De la Fuente, J.; Almazán, C.; Blas-Machado, U.; Naranjo, V.; Mangold, A.J.; Blouin, E.F.; Gortázar, C.; Kocan, K.M. The tick protective antigen, 4D8, is a conserved protein involved in modulation of tick blood ingestion and reproduction. Vaccine 2006, 24, 4082–4095. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Cid, J.A.; Pérez de la Lastra, J.M.; Villar, M.; Jiménez, M.; Pinal, R.; Estrada-Peña, A.; Molina, R.; Lucientes, J.; Gortázar, C.; de la Fuente, J.; et al. Control of multiple arthropod vector infestations with subolesin/akirin vaccines. Vaccine 2013, 31, 1187–1196. [Google Scholar] [CrossRef] [PubMed]
- Merino, M.; Antunes, S.; Mosqueda, J.; Moreno-Cid, J.A.; Pérez de la Lastra, J.M.; Rosario-Cruz, R.; Rodríguez, S.; Domingos, A.; de la Fuente, J. Vaccination with proteins involved in tick-pathogen interactions reduces vector infestations and pathogen infection. Vaccine 2013, 31, 5889–5896. [Google Scholar] [CrossRef] [PubMed]
- Contreras, M.; de la Fuente, J. Control of Ixodes ricinus and Dermacentor reticulatus tick infestations in rabbits vaccinated with the Q38 Subolesin/Akirin chimera. Vaccine 2016, 34, 3010–3013. [Google Scholar] [CrossRef] [PubMed]
- McDevitt, R.; Nisbet, A.J.; Huntley, J.F. Ability of a proteinase inhibitor mixture to kill poultry red mitte, Dermanyssus gallinae in an in vitro feeding system. Vet. Parasitol. 2006, 141, 380–385. [Google Scholar] [CrossRef]

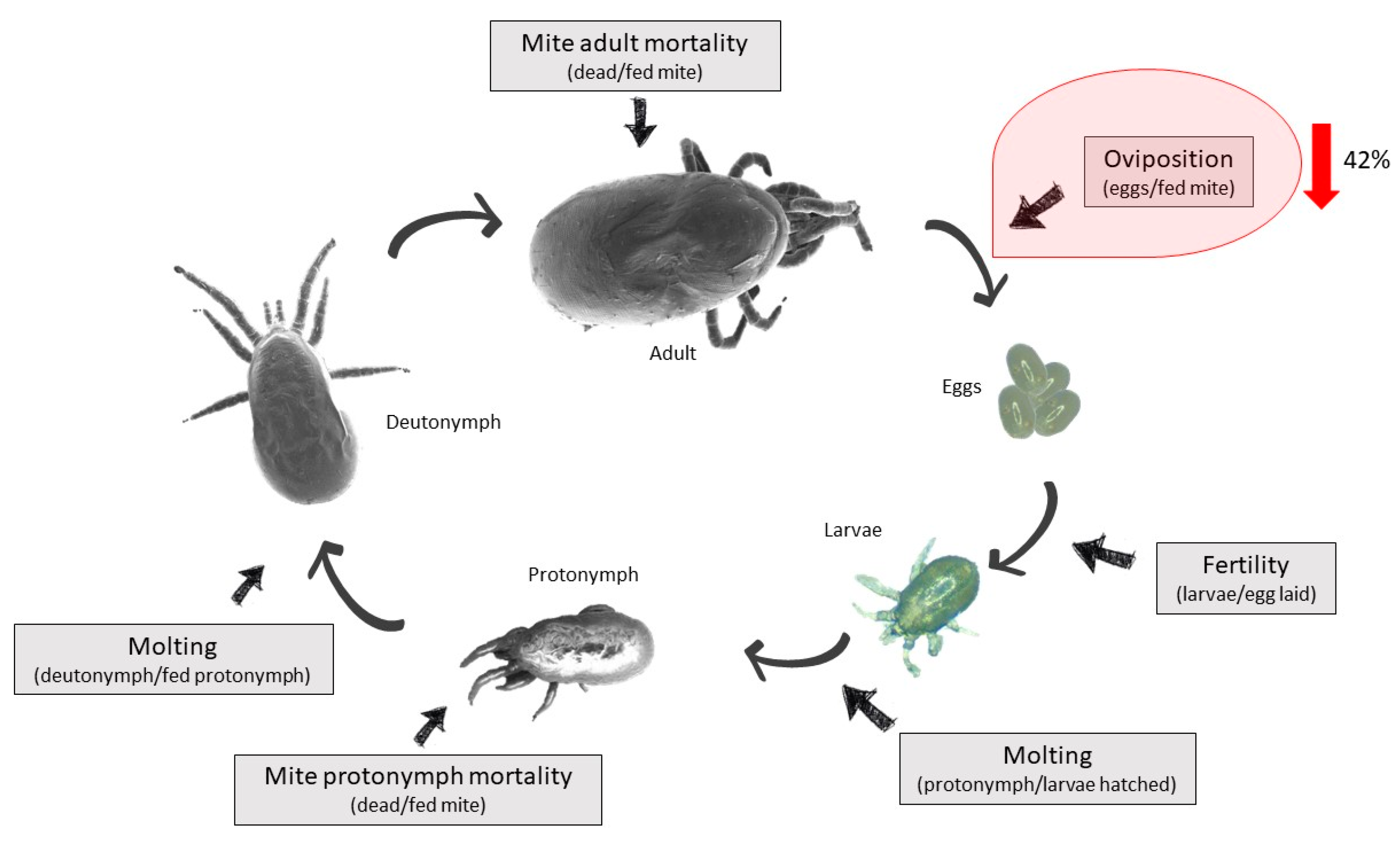

| Group | Hen | Fed Mites | Partially Fed/Unfed Mites | Total | % Fed Mites | Average Feeding ± SD | Reduction | Eggs | Oviposition | Average Oviposition ± SD | Reduction | Dead Mites | Mortality | Average Mortality | Mortality Effect |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Deg-AKR | 1 | 56 | 38/276 = 314 | 370 | 15 | 21 ± 5 | 0% | 34 | 0.6 | 0.4 ± 0.2 | 42% * | 4 | 0.1 | 0.2 ± 0.1 | 6% |
| 2 | 77 | 5/343 = 348 | 425 | 18 | 23 | 0.3 | 19 | 0.2 | |||||||
| 3 | 130 | 23/325 = 348 | 478 | 27 | 34 | 0.3 | 16 | 0.1 | |||||||
| 4 | 120 | 10/391 = 401 | 521 | 23 | 34 | 0.3 | 24 | 0.2 | |||||||
| 5 | 81 | 30/246 = 276 | 357 | 23 | 42 | 0.5 | 19 | 0.2 | |||||||
| Control | 6 | 100 | 23/230 = 253 | 353 | 28 | 18 ± 8 | 65 | 0.6 | 0.7 ± 0.2 | 15 | 0.2 | 0.2 ± 0.0 | |||
| 7 | 32 | 32/304 = 336 | 368 | 9 | 23 | 0.7 | 4 | 0.1 | |||||||
| 8 | 59 | 23/394 = 417 | 476 | 12 | 38 | 0.6 | 10 | 0.2 | |||||||
| 9 | 85 | 23/284 = 307 | 392 | 22 | 86 | 1.0 | 13 | 0.2 | |||||||
| 10 | 44 | 34/190 = 224 | 268 | 16 | 15 | 0.3 | 10 | 0.2 |
| Group | Hen | Fed/PFPN | UnfedPN | Total PN | % Fed | Average Feeding ± SD | Feeding Effect | Molt Fed PN | Molt/Fed | Average Molting ± SD | Molting Effect | Dead PN | Fed | Dead/Fed | Average Mortality | Mortality Effect |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Deg-AKR | 1 | 13/5 = 18 | 133 | 151 | 11.9 | 13.1 ± 4.0 | 0% | 5 | 0.38 | 0.2 ± 0.1 | 0% | 1 | 13 | 0.1 | 0.2± 0.1 | 50% |
| 2 | 11/4 = 15 | 101 | 116 | 12.9 | 2 | 0.18 | 2 | 11 | 0.2 | |||||||
| 3 | 10/5 = 15 | 146 | 161 | 9.3 | 1 | 0.10 | 3 | 10 | 0.3 | |||||||
| 4 | 33/5 = 38 | 153 | 191 | 19.9 | 7 | 0.24 | 6 | 33 | 0.2 | |||||||
| 5 | 19/1 = 20 | 157 | 177 | 11.3 | 6 | 0.32 | 2 | 19 | 0.1 | |||||||
| Control | 6 | 4/9 = 13 | 126 | 139 | 9.4 | 10.6 ± 1.6 | 0 | 0.00 | 0.3 ± 0.3 | 1 | 4 | 0.3 | 0.1 ± 0.2 | |||
| 7 | 12/1 = 13 | 87 | 100 | 13.0 | 7 | 0.58 | 0 | 12 | 0.0 | |||||||
| 8 | 17/3 = 20 | 174 | 194 | 10.3 | 3 | 0.18 | 1 | 17 | 0.1 | |||||||
| 9 | 15/0 = 15 | 119 | 134 | 11.2 | 5 | 0.33 | 5 | 15 | 0.3 | |||||||
| 10 | 5/7 = 12 | 120 | 132 | 9.1 | 3 | 0.60 | 0 | 5 | 0.0 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lima-Barbero, J.F.; Contreras, M.; Bartley, K.; Price, D.R.G.; Nunn, F.; Sanchez-Sanchez, M.; Prado, E.; Höfle, U.; Villar, M.; Nisbet, A.J.; et al. Reduction in Oviposition of Poultry Red Mite (Dermanyssus gallinae) in Hens Vaccinated with Recombinant Akirin. Vaccines 2019, 7, 121. https://doi.org/10.3390/vaccines7030121
Lima-Barbero JF, Contreras M, Bartley K, Price DRG, Nunn F, Sanchez-Sanchez M, Prado E, Höfle U, Villar M, Nisbet AJ, et al. Reduction in Oviposition of Poultry Red Mite (Dermanyssus gallinae) in Hens Vaccinated with Recombinant Akirin. Vaccines. 2019; 7(3):121. https://doi.org/10.3390/vaccines7030121
Chicago/Turabian StyleLima-Barbero, Jose Francisco, Marinela Contreras, Kathryn Bartley, Daniel R. G. Price, Francesca Nunn, Marta Sanchez-Sanchez, Eduardo Prado, Ursula Höfle, Margarita Villar, Alasdair J. Nisbet, and et al. 2019. "Reduction in Oviposition of Poultry Red Mite (Dermanyssus gallinae) in Hens Vaccinated with Recombinant Akirin" Vaccines 7, no. 3: 121. https://doi.org/10.3390/vaccines7030121
APA StyleLima-Barbero, J. F., Contreras, M., Bartley, K., Price, D. R. G., Nunn, F., Sanchez-Sanchez, M., Prado, E., Höfle, U., Villar, M., Nisbet, A. J., & de la Fuente, J. (2019). Reduction in Oviposition of Poultry Red Mite (Dermanyssus gallinae) in Hens Vaccinated with Recombinant Akirin. Vaccines, 7(3), 121. https://doi.org/10.3390/vaccines7030121







