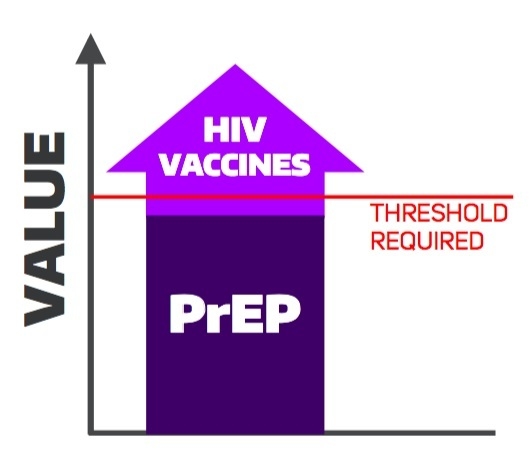
The Potential Cost-Effectiveness of Pre-Exposure Prophylaxis Combined with HIV Vaccines in the United States
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population
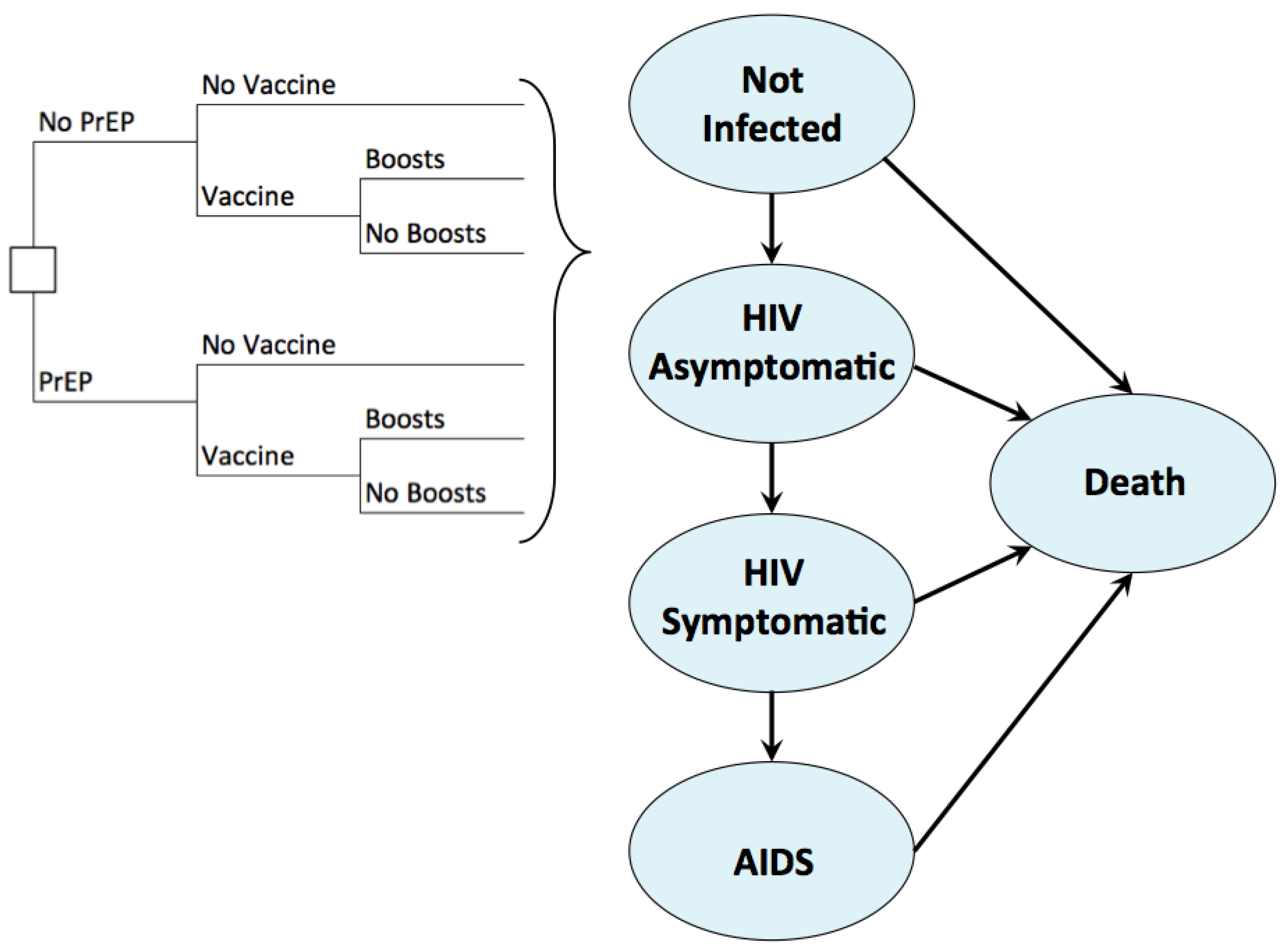
2.2. Model Overview
2.3. Model Inputs
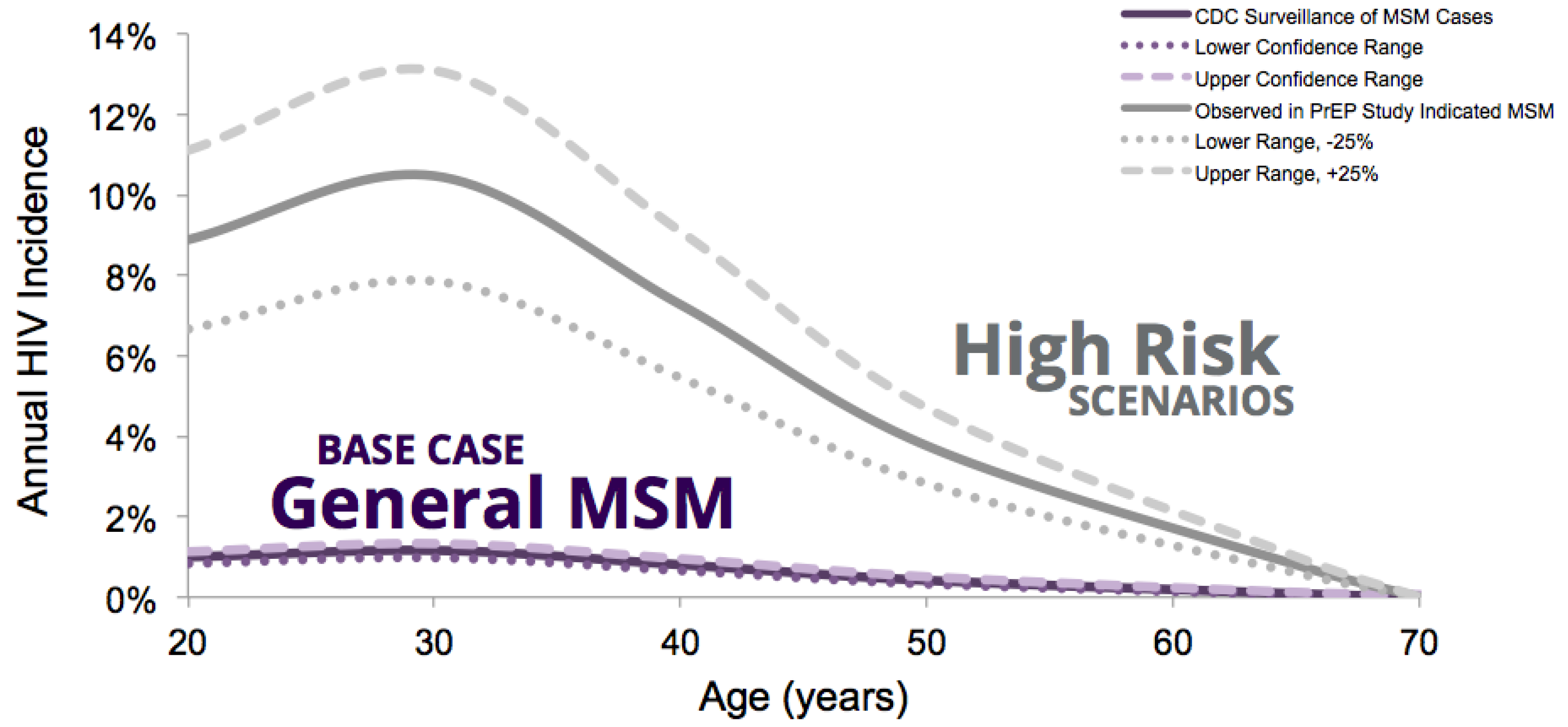
2.3.1. HIV Incidence
2.3.2. Clinical Inputs
2.3.3. Health State Utility
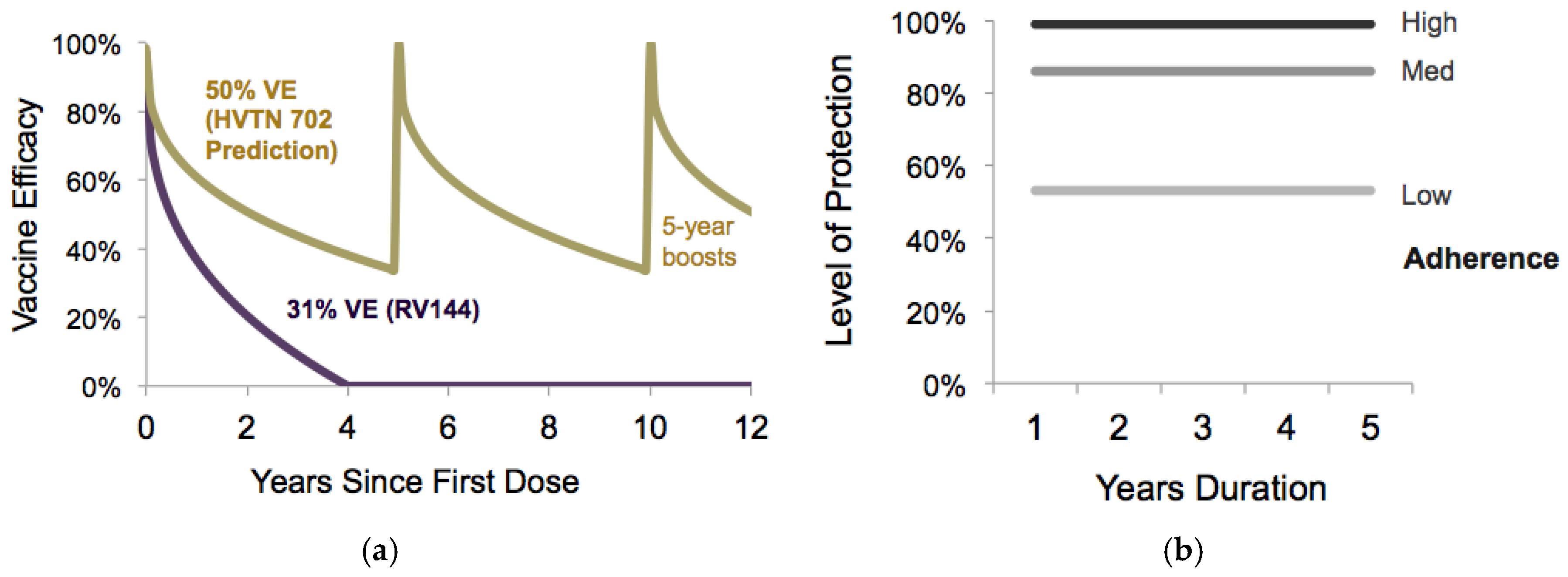
2.3.4. Intervention Effectiveness
2.3.5. Costs
2.4. Model Outputs
2.5. Cost-Effectiveness Analysis
2.6. Sensitivity Analysis
3. Results
3.1. Base Case
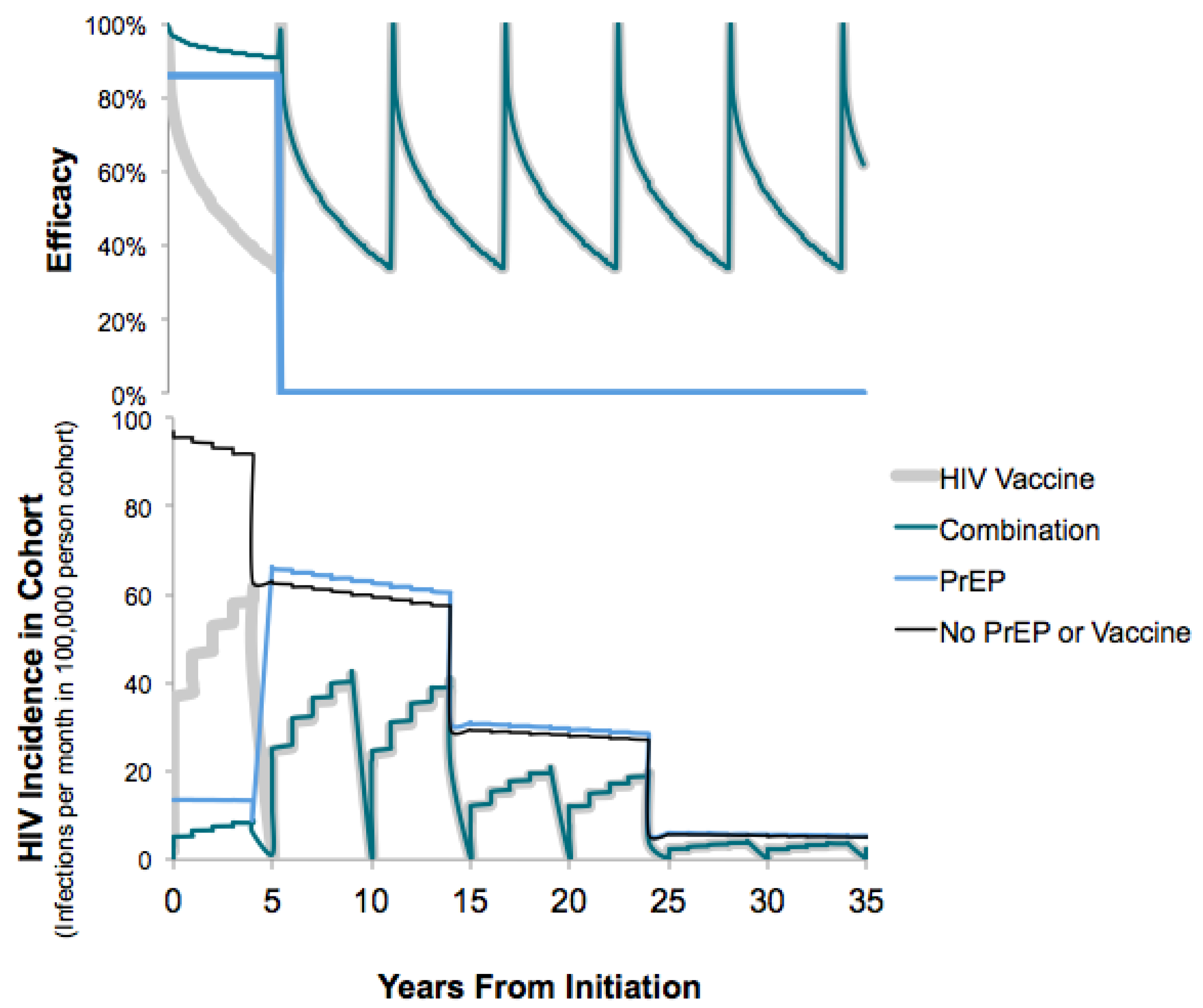
3.1.1. Clinical Outcomes
3.1.2. Costs
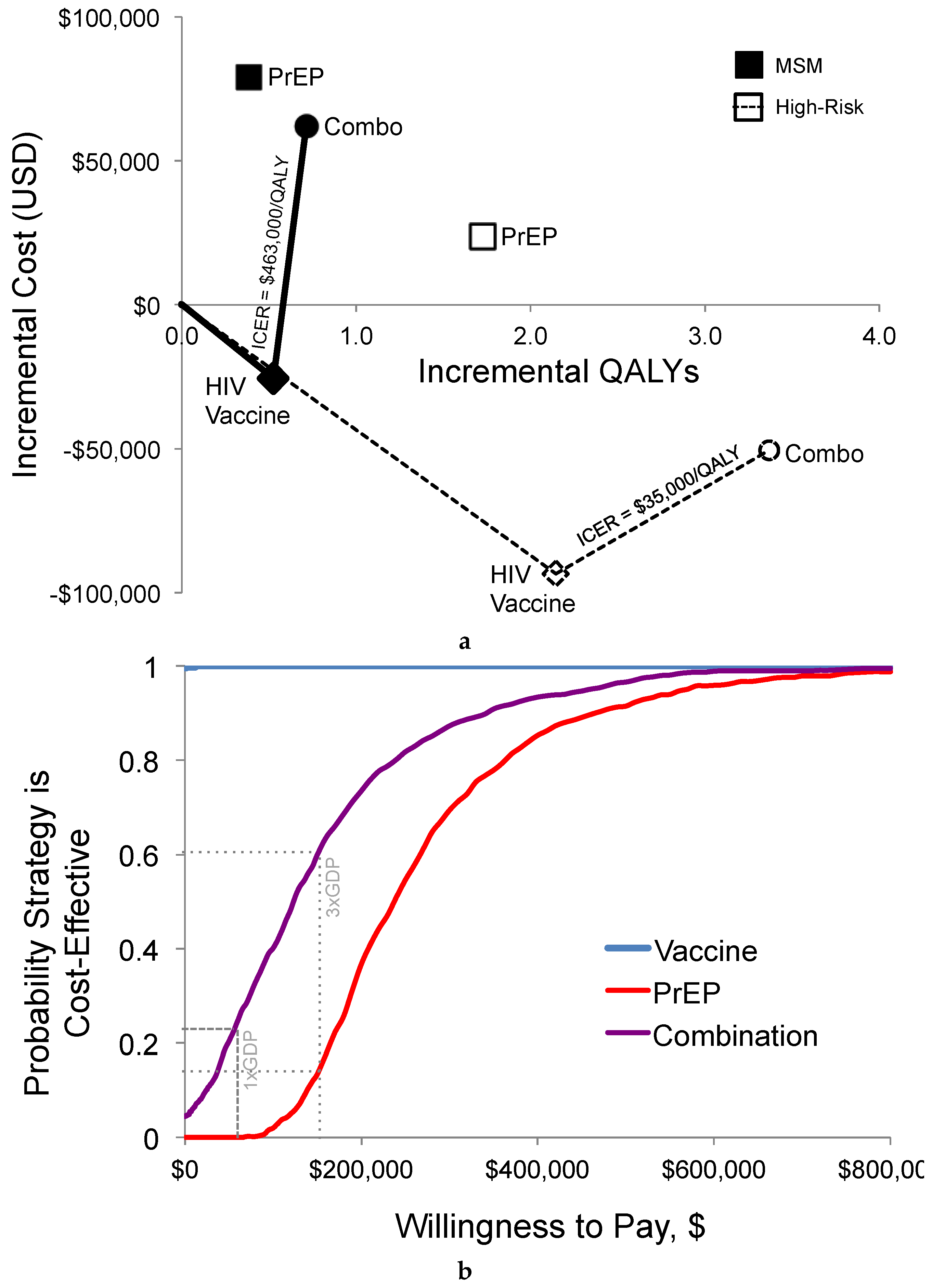
3.1.3. Cost-Effectiveness
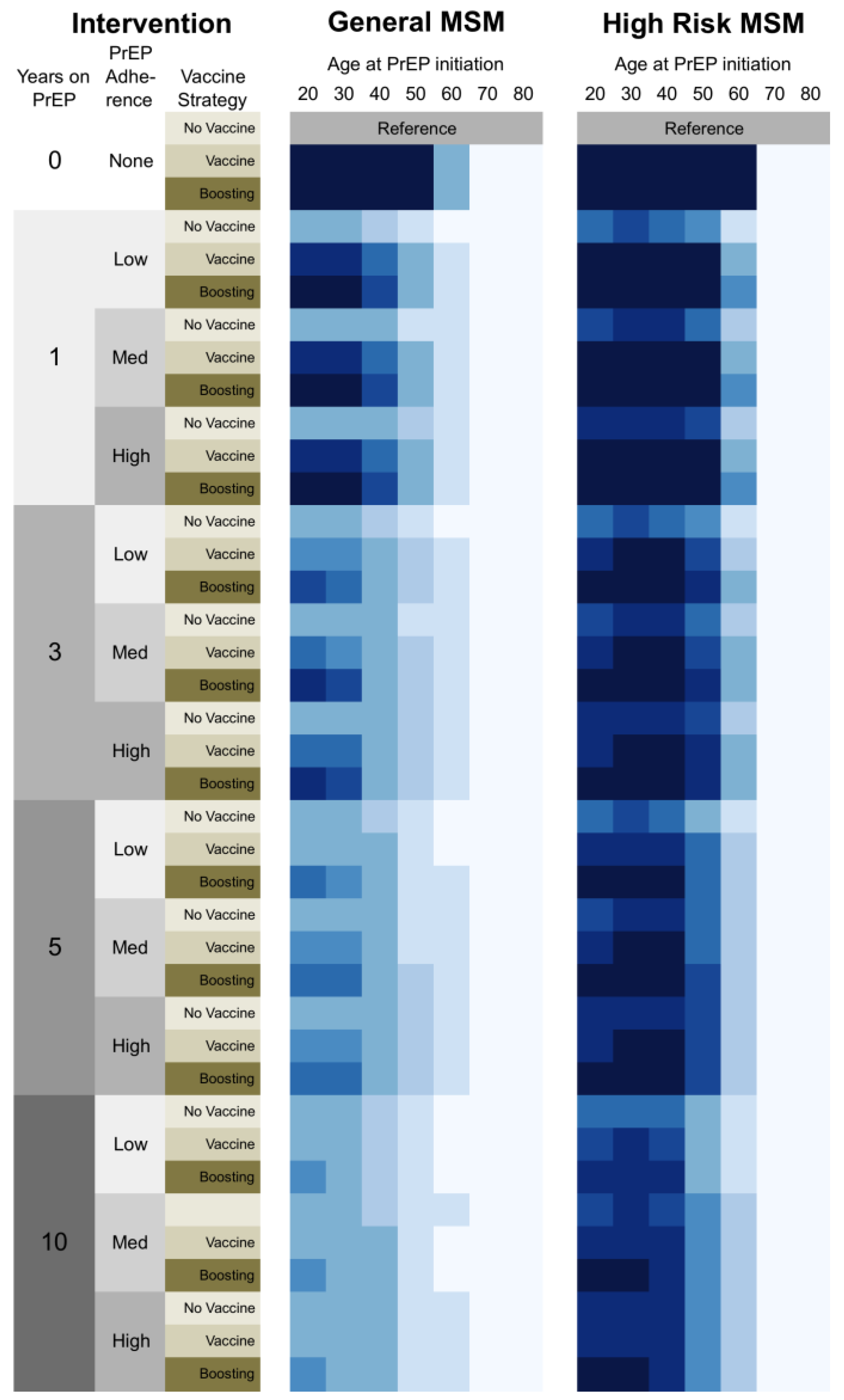
3.2. Sensitivity Analysis
3.2.1. Scenarios
3.2.2. Probabilistic Sensitivity Analysis
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| ART | Antiretroviral Therapy |
| AWP | Average Wholesale Price, $ |
| CEA | Cost-Effectiveness Analysis |
| CPI | Consumer Price Index |
| CR | Credible Range |
| FTC | emtricitabine |
| ICER | Incremental Cost Effectiveness Ratio, $/QALY |
| LY | Life Years |
| MSM | Men Who Have Sex With Men |
| NNT | Number Needed To Treat |
| PrEP | Pre-exposure Prophylaxis |
| PSA | Probabilistic Sensitivity Analysis |
| QALY | Quality-Adjusted Life Year |
| TAF | tenofovir alafenamide fumarate |
| TDF | tenofovir disoproxil fumarate |
| USA | United States of America |
| VE | Vaccine Efficacy, % |
| WTP | Willingness to Pay, $ |
Appendix A
| Vaccine Component | RV144 Thai Trial | HVTN 702 South Africa Trial | ||||
|---|---|---|---|---|---|---|
| DNA Prime | Protein Boost | Adjuvant | DNA Prime | Protein Boost | Adjuvant | |
| Description | ALVAC-HIV recombinant canarypox vaccine, subtype B and E | AIDSVAX® B/E bivalent HIV gp120 envelope glycoprotein vaccine, subtypes B and E | 600 μg of alum adjuvant | Canarypox-based vaccine ALVAC-HIV subtype C | bivalent gp120 protein subunit vaccine, subtype C | MF59 |
| Manufacturer | Developed by Virogenetics Corporation (Troy, NY) and manufactured by Sanofi Pasteur (Marcy-l'Étoille, France) | Originally manufactured by Genentech, Inc., further developed by VaxGen, Inc., later acquired by Global Solutions for Infectious Diseases (San Francisco, CA, USA) | VaxGen, Inc, no IP | Sanofi Pasteur | GlaxoSmithKline (GSK) | GSK |
| Trial Funding | Supported in part by an Interagency Agreement (Y1-AI-2642-12) between the U.S. Army Medical Research and Materiel Command and the National Institute of Allergy and Infectious Diseases and by a cooperative agreement (W81XWH-07-2-0067) between the Henry M. Jackson Foundation for the Advancement of Military Medicine and the U.S. Department of Defense. Sanofi Pasteur provided the ALVAC-HIV vaccine, and Global Solutions for Infectious Diseases (VaxGen) provided the reagents for the immunogenicity assays. | P5 members are National Institute of Allergy and Infectious Diseases (NIAID), the Bill & Melinda Gates Foundation (BMGF), the South African Medical Research Council (SAMRC), HIV Vaccine Trials Network (HVTN), Sanofi Pasteur, GSK, and the U.S. Military HIV Research Program. NIAID, BMGF, and SAMRC fund the P5. The National Institute of Allergy and Infectious Diseases (NIAID) is sponsoring and funding HVTN 702. Sanofi Pasteur and GSK are providing the investigational vaccines for the trial. | ||||
| Sector | Type of Impact (Unit of Measure if Relevant) | Included in this Analysis from the Perspective of | ||
|---|---|---|---|---|
| Payer ☑ | Health Care Sector | Societal | ||
| Formal Health-care sector | Health Outcomes (Effects) | |||
| Longevity effects (Life Years) | ☑ | |||
| Health-related quality of life (QALYs) | ☑ | |||
| Adverse events (QALYS) | ☑ | |||
| Secondary transmissions of infections | ||||
| Medical Costs | ||||
| Paid for by third-party payers ($) | ☑ | |||
| Paid for by patients out-of-pocket ($) | ||||
| Future related medical costs to payers ($) | ☑ | |||
| Future related medical costs to patients ($) | NA | |||
| Future unrelated medical costs to payers ($) | ||||
| Future unrelated medical costs to patients ($) | NA | |||
| Informal Health-care sector | Patient time costs | NA | ||
| Unpaid caregiver time costs ($) | NA | |||
| Transportation costs ($) | NA | |||
| Productivity | Labor market earnings lost ($) | NA | NA | |
| Cost of unpaid lost productivity due to illness ($) | NA | NA | ||
| Cost of uncompensated household production ($) | NA | NA | ||
| Consumption | Future consumption unrelated to health ($) | NA | NA | |
| Social Services | Cost of social services as part of intervention ($) | NA | NA | |
| Education | Impact of intervention on educational achievement of population | NA | NA | |
Model Validation
References
- Holtgrave, D.R.; Hall, H.I.; Wehrmeyer, L.; Maulsby, C. Costs, consequences and feasibility of strategies for achieving the goals of the National HIV/AIDS strategy in the United States: A closing window for success? AIDS Behav. 2012, 16, 1365–1372. [Google Scholar] [CrossRef] [PubMed]
- Farnham, P.G.; Holtgrave, D.R.; Gopalappa, C.; Hutchinson, A.B.; Sansom, S.L. Lifetime costs and quality-adjusted life years saved from HIV prevention in the test and treat era. J. Acquir. Immune Defic. Syndr. 2013, 64, 15–18. [Google Scholar] [CrossRef] [PubMed]
- Schackman, B.R.; Gebo, K.A.; Walensky, R.P.; Losina, E.; Muccio, T.; Sax, P.E.; Weinstein, M.C.; Seage, G.R.; Moore, R.D.; Freedberg, K.A. The lifetime cost of current human immunodeficiency virus care in the United States. Med. Care 2006, 44, 990–997. [Google Scholar] [CrossRef] [PubMed]
- The Henry J. Kaiser Family Foundation. U.S. Federal Funding for HIV/AIDS: Trends Over Time; The Henry J. Kaiser Family Foundation: Menlo Park, CA, USA, 2016. [Google Scholar]
- Jiang, J.; Yang, X.; Ye, L.; Zhou, B.; Ning, C.; Huang, J.; Liang, B.; Zhong, X.; Huang, A.; Tao, R.; et al. Pre-exposure prophylaxis for the prevention of HIV infection in high risk populations: A meta-analysis of randomized controlled trials. PLoS ONE 2014, 9, e87674. [Google Scholar] [CrossRef] [PubMed]
- Liu, A.; Glidden, D.V.; Anderson, P.L.; Amico, K.R.; McMahan, V.; Mehrotra, M.; Lama, J.R.; MacRae, J.; Hinojosa, J.C.; Montoya, O.; et al. Patterns and correlates of PrEP drug detection among MSM and transgender women in the global iPrEx study. J. Acquir. Immune Defic. Syndr. 2014. [Google Scholar] [CrossRef] [PubMed]
- Molina, J.M.; Capitant, C.; Spire, B.; Pialoux, G.; Cotte, L.; Charreau, I.; Tremblay, C.; Gall, J.M.L.; Cua, E.; Pasquet, A.; et al. On-demand preexposure prophylaxis in men at high risk for HIV-1 infection. N. Engl. J. Med. 2015, 373, 2237–2246. [Google Scholar] [CrossRef] [PubMed]
- McCormack, S.; Dunn, D. Pragmatic open-label randomised trial of preexposure prophylaxis: The PROUD study. In Proceedings of the Conference Retroviruses Opportunistic Infect, Seattle, WA, USA, 23–26 February 2015. [Google Scholar]
- Volk, J.E.; Marcus, J.L.; Phengrasamy, T.; Blechinger, D.; Nguyen, D.P.; Follansbee, S.; Hare, C.B. No new HIV infections with increasing use of HIV preexposure prophylaxis in a clinical practice setting. Clin. Infect. Dis. 2015, 61, civ778. [Google Scholar] [CrossRef] [PubMed]
- Express Scripts 2015 Drug Trend Report. Available online: https://www.google.ch/url?sa=t&rct=j&q=&esrc=s&source=web&cd=2&sqi=2&ved=0ahUKEwiKoZG-4IXUAhVLFMAKHemUAPQQFgguMAE&url=https%3A%2F%2Flab.express-scripts.com%2Flab%2F~%2Fmedia%2Fe2c9d19240e94fcf893b706e13068750.ashx&usg=AFQjCNF8GLkNH0aNfbFjCynKBkO3DH_l9Q&cad=rja (accessed on 13 November 2016).
- Smith, D.K.; van Handel, M.; Wolitski, R.J.; Stryker, J.E.; Hall, H.I.; Prejean, J.; Koenig, L.J.; Valleroy, L.A. Vital signs: Estimated percentages and numbers of adults with indications for preexposure prophylaxis to prevent hiv acquisition-United States, 2015. MMWR 2015, 64, 1291–1295. [Google Scholar] [CrossRef] [PubMed]
- Grey, J.A.; Bernstein, K.T.; Sullivan, P.S.; Purcell, D.W.; Chesson, H.W.; Gift, T.L.; Rosenberg, E.S. Estimating the population sizes of men who have sex with men in US States and counties using data from the american community survey. JMIR Public Health Surveill 2016, 2, e14. [Google Scholar] [CrossRef] [PubMed]
- Basow, D.S. Tenofovir Disoproxil Fumarate and Emtricitabine: Drug Information; UpToDate: Waltham, MA, USA, 2015. [Google Scholar]
- Rerks-Ngarm, S.; Pitisuttithum, P.; Nitayaphan, S.; Kaewkungwal, J.; Chiu, J.; Paris, R.; Premsri, N.; Namwat, C.; de Souza, M.; Adams, E.; et al. Vaccination with ALVAC and AIDSVAX to prevent HIV-1 infection in Thailand. N. Engl. J. Med. 2009, 361, 2209–2220. [Google Scholar] [CrossRef] [PubMed]
- Akapirat, S.; Chitraporn, K.; O’Connell, R.J.; Pitisuthithum, P.; Rerks-Ngarm, S.; Michael, N.L.; De Souza, M.A.; Kim, J.H.; Karasavvas, N. Antibody responses in anogenital secretions of rv305 a late boost vaccination of RV144 volunteers. In Proceedings of the Retroviruses Opportunistic Infect, Boston, MA, USA, 3–6 March 2014; pp. 3–6. [Google Scholar]
- National Institute of Allergy and Infectious Diseases. Large-Scale HIV Vaccine Trial to Launch in South Africa: NIH-Funded Study Will Test Safety, Efficacy of Vaccine Regimen. NIH News 2016. Available online: https://www.niaid.nih.gov/news-events/large-scale-hiv-vaccine-trial-launch-south-africa (accessed on 13 November 2016).
- Abbasi, J. Large HIV vaccine trial launches in South Africa. JAMA 2017, 317, 350. [Google Scholar] [CrossRef] [PubMed]
- Paltiel, A.D.; Freedberg, K.A.; Scott, C.A.; Schackman, B.R.; Losina, E.; Wang, B.; Seage, G.R., 3rd; Sloan, C.E.; Sax, P.E.; Walensky, R.P. HIV preexposure prophylaxis in the United States: Impact on lifetime infection risk, clinical outcomes, and cost-effectiveness. Clin. Infect. Dis. 2009, 48, 806–815. [Google Scholar] [CrossRef] [PubMed]
- Juusola, J.L.; Brandeau, M.L.; Owens, D.K.; Bendavid, E. The cost-effectiveness of preexposure prophylaxis for HIV prevention in the United States in men who have sex with men. Ann. Intern. Med. 2012, 156, 541–550. [Google Scholar] [CrossRef] [PubMed]
- Bernard, C.L.; Brandeau, M.L.; Humphreys, K.; Bendavid, E.; Holodniy, M.; Weyant, C.; Owens, D.K.; Goldhaber-Fiebert, J.D. Cost-effectiveness of HIV preexposure prophylaxis for people who inject drugs in the United States. Ann. Intern. Med. 2016, 165, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Gomez, G.B.; Borquez, A.; Case, K.K.; Wheelock, A.; Vassall, A.; Hankins, C. The cost and impact of scaling up pre-exposure prophylaxis for HIV prevention: A systematic review of cost-effectiveness modelling studies. PLoS Med. 2013, 10, e1001401. [Google Scholar] [CrossRef] [PubMed]
- Long, E.F.; Brandeau, M.L.; Owens, D.K. Potential population health outcomes and expenditures of HIV vaccination strategies in the United States. Vaccine 2009, 27, 5402–5410. [Google Scholar] [CrossRef] [PubMed]
- Long, E.F.; Owens, D.K. The cost-effectiveness of a modestly effective HIV vaccine in the United States. Vaccine 2011, 29, 6113–6124. [Google Scholar] [CrossRef] [PubMed]
- Adamson, B.; Dimitrov, D.; Devine, B.; Barnabas, R. The potential cost-effectiveness of HIV vaccines: A systematic review. Pharmacoeconomics 2017, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Schackman, B.R.; Eggman, A.A. Cost-effectiveness of pre-exposure prophylaxis for HIV: A review. Curr. Opin. HIV AIDS 2012, 7, 587–592. [Google Scholar] [CrossRef] [PubMed]
- Hankins, C.A. Untangling the cost-effectiveness knot: Who is oral antiretroviral HIV pre-exposure prophylaxis really for? Expert Rev. Pharm. Outcomes Res. 2014, 14, 167–170. [Google Scholar] [CrossRef] [PubMed]
- US Public Health Service. Preexposure Prophylaxis For The Prevention Of HIV Infection In The United States-2014: A Clinical Practice Guideline; US Public Health Service: Atlanta, GA, USA, 2014. [Google Scholar]
- Neumann, P.J.; Sanders, G.; Russell, L.; Siegel, J.; Ganiats, T. Cost-Effectiveness in Health and Medicine, 2nd ed.; Oxford University Press: New York, NY, USA, 2017. [Google Scholar]
- Husereau, D.; Drummond, M.; Petrou, S.; Carswell, C.; Moher, D.; Greenberg, D.; Augustovski, F.; Briggs, A.H.; Mauskopf, J.; Loder, E. Consolidated health economic evaluation reporting standards (CHEERS) statement. Int. J. Technol. Assess. Health Care 2013, 29, 117–122. [Google Scholar] [CrossRef] [PubMed]
- Sanders, G.D.; Bayoumi, A.M.; Sundaram, V.; Bilir, S.P.; Neukermans, C.P.; Rydzak, C.E.; Douglass, L.R.; Lazzeroni, L.C.; Holodniy, M.; Owens, D.K. Cost-effectiveness of screening for HIV in the era of highly active antiretroviral therapy. N. Engl. J. Med. 2005, 352, 570–585. [Google Scholar] [CrossRef] [PubMed]
- Bayoumi, A.M.; Barnett, P.G.; Joyce, V.R.; Griffin, S.C.; Sun, H.; Bansback, N.J.; Holodniy, M.; Sanders, G.; Brown, S.T.; Kyriakides, T.C.; et al. Cost-effectiveness of newer antiretroviral drugs in treatment-experienced patients with multidrug-resistant HIV disease. J. Acquir. Immune Defic. Syndr. 2013, 64, 382–391. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. HIV/AIDS data through December 2012 provided for the Ryan White HIV/AIDS Program, for fiscal year 2014. HIV Surveill Suppl. Rep. 2015, 20, 1–16. [Google Scholar]
- U.S. Census Bureau Population Division. National Population Projection, 2014; U.S. Census Bureau Population Division: Washington, DC, USA, 2015.
- Arias, E. Division of vs. United States life tables, 2010. Natl. Vital Stat. Rep. 2014, 63, 1–62. [Google Scholar] [PubMed]
- Rodger, A.J.; Lodwick, R.; Schechter, M.; Deeks, S.; Amin, J.; Gilson, R.; Paredes, R.; Bakowska, E.; Engsig, F.N.; Phillips, A.; et al. Mortality in well controlled HIV in the continuous antiretroviral therapy arms of the SMART and ESPRIT trials compared with the general population. AIDS 2013, 27, 973–979. [Google Scholar] [CrossRef] [PubMed]
- Weber, R.; Ruppik, M.; Rickenbach, M.; Spoerri, A.; Furrer, H.; Battegay, M.; Cavassini, M.; Calmy, A.; Bernasconi, E.; Schmid, P.; et al. Decreasing mortality and changing patterns of causes of death in the Swiss HIV Cohort Study. HIV Med. 2013, 14, 195–207. [Google Scholar] [CrossRef] [PubMed]
- Hanmer, J.; Lawrence, W.F.; Anderson, J.P.; Kaplan, R.M.; Fryback, D.G. Report of nationally representative values for the noninstitutionalized US adult population for 7 health-related quality-of-life scores. Med. Decis. Mak. 2006, 26, 391–400. [Google Scholar] [CrossRef] [PubMed]
- Tengs, T.O.; Lin, T.H. A meta-analysis of utility estimates for HIV/AIDS. Med. Decis. Mak. 2002, 22, 475–481. [Google Scholar] [CrossRef] [PubMed]
- Kauf, T.L.; Roskell, N.; Shearer, A.; Gazzard, B.; Mauskopf, J.; Davis, E.A.; Nimsch, C. A predictive model of health state utilities for HIV patients in the modern era of highly active antiretroviral therapy. Value Health 2008, 11, 1144–1153. [Google Scholar] [CrossRef] [PubMed]
- Joyce, V.R.; Barnett, P.G.; Bayoumi, A.M.; Griffin, S.C.; Kyriakides, T.C.; Yu, W.; Sundaram, V.; Holodniy, M.; Brown, S.T.; Cameron, W.; et al. Health-related quality of life in a randomized trial of antiretroviral therapy for advanced HIV disease. J. Acquir. Immune Defic. Syndr. 2009, 50, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Molina, J.M.; Capitant, C.; Spire, B.; Pialoux, G.; Cotte, L.; Charreau, I.; Tremblay, C.; Le Gall, J.M.; Cua, E.; Pasquet, A.; et al. On-Demand Preexposure Prophylaxis in Men at High Risk for HIV-1 Infection. N. Engl. J. Med. 2015, 373, 2237–2246. [Google Scholar] [CrossRef] [PubMed]
- Baeten, J.M.; Donnell, D.; Ndase, P.; Mugo, N.R.; Campbell, J.D.; Wangisi, J.; Tappero, J.W.; Bukusi, E.A.; Cohen, C.R.; Katabira, E.; et al. Antiretroviral prophylaxis for HIV prevention in heterosexual men and women. N. Engl. J. Med. 2012, 367, 399–410. [Google Scholar] [CrossRef] [PubMed]
- Amico, K.R.; Marcus, J.L.; Mcmahan, V.; Liu, A.; Koester, K.A.; Goicochea, P.; Anderson, P.L.; Glidden, D.; Guanira, J.; Grant, R. Study product adherence measurement in the iPrEx placebo-controlled trial: Concordance with drug detection. J. Acquir. Immune Defic. Syndr. 2014, 66, 530–537. [Google Scholar] [CrossRef] [PubMed]
- Hankins, C.A.; Glasser, J.W.; Chen, R.T. Modeling the impact of RV144-like vaccines on HIV transmission. Vaccine 2011, 29, 6069–6071. [Google Scholar] [CrossRef] [PubMed]
- Moody, M.A.; Easterhoff, D.; Gurley, T.C.; Whitesides, J.F.; Marshall, D.J.; Foulger, A.; Lloyd, K.E.; Parks, R.; Pollara, J.; Duffy, R.; et al. Induction of antibodies with long variable heavy third complementarity determining regions by repetitive boosting with aidsvax® B/E in RV144 vaccinees. AIDS Res. Hum. Retroviruses 2014, 30, A36. [Google Scholar]
- United States Department of Labor Bureau of Labor Statistics. Measuring Price Change for Medical Care in the CPI. Consum Price Index 2017. Available online: https://www.bls.gov/cpi/cpifact4.htm (accessed on 22 April 2017).
- Bureau of Labor Statistics. Consumer Price Index Detailed Report: Data for October 2016; Bureau of Labor Statistics: Washington, DC, USA, 2016.
- Gebo, K.A.; Fleishman, J.A.; Conviser, R.; Hellinger, J.; Hellinger, F.J.; Josephs, J.S.; Keiser, P.; Gaist, P.; Moore, R.D. Contemporary costs of HIV healthcare in the HAART era. AIDS 2010, 24, 2705–2715. [Google Scholar] [CrossRef] [PubMed]
- U.S. Department of Veterans Affairs. Department of Veterans Affairs. National Acquisition Center (CCST) n.d. Available online: http://www1.va.gov/nac/ (accessed on 10 October 2016).
- Seage, G.R. Are US populations appropriate for trials of human immunodeficiency virus vaccine?: The HIVNET vaccine preparedness study. Am. J. Epidemiol. 2001, 153, 619–627. [Google Scholar] [CrossRef] [PubMed]
- National Institute of Allergy and Infectious Diseases. NIH-Sponsored HIV Vaccine Trial Launches in South Africa: Early-Stage Trial Aims to Build on RV144 Results. NIH News 2015. Available online: http://www.niaid.nih.gov/news/newsreleases/2015/Pages/HVTN100.aspx (accessed on 31 July 2015).
- Molina, J.-M.; Capitant, C.; Spire, B.; Pialoux, G.; Chidiac, C.; Charreau, I.; Delfraissy, J.F. On Demand PrEP With Oral TDF-FTC in MSM: Results of the ANRS Ipergay Trial. In Proceedings of the Conference on Retroviruses Opportunistic Infection, Seattle, WA, USA, 23–26 Febuary 2017. [Google Scholar]
- Sirivichayakul, S.; Phanuphak, P.; Hanvanich, M.; Ruxrungtham, K.; Panmoung, W.; Thanyanon, W. Clinical correlation of the immunological markers of HIV infection in individuals from Thailand. AIDS 1992, 6, 393–397. [Google Scholar] [CrossRef] [PubMed]
- Gold, M.; Siegel, J.; Russell, L.; Weinstien, M. Cost-Effectiveness in Health and Medicine; Oxford University Press: New York, NY, USA, 1996. [Google Scholar]
- Briggs, A.; Claxton, K.; Sculpher, M. Decision Modelling for Health Economic Evaluation, 1st ed.; Oxford University Press: London, UK, 2006. [Google Scholar]
- Neumann, P.J.; Cohen, J.T. Measuring the value of prescription drugs. N. Engl. J. Med. 2015. [Google Scholar] [CrossRef] [PubMed]
- The Congress of the United States Congressional Budget Office. How Increased Competition from Generic Drugs Has Affected Prices and Returns in the Pharmaceutical Industry; The Congress of the United States Congressional Budget Office: Washington, DC, USA, 1998.
- Bach, P.B. Indication-specific pricing for cancer drugs. JAMA 2014, 312, 1629–1630. [Google Scholar] [CrossRef] [PubMed]
- Bush, S.; Ng, L.; Magnuson, D.; Piontkowsky, D.; Mera Giler, R. Significant uptake of truvada for pre-exposure prophylaxis (PrEP) utilization in the US in Late 2014–1Q2015. In Proceedings of the IAPAC Treatment and Prevention Adherence Conference, Miami, FL, USA, 28–30 June 2015. [Google Scholar]
- Schneider, K.; Gray, R.T.; Wilson, D.P. A cost-effectiveness analysis of HIV preexposure prophylaxis for men who have sex with men in Australia. Clin. Infect. Dis. 2014, 58, 1027–1034. [Google Scholar] [CrossRef] [PubMed]
- Ouellet, E.; Durand, M.; Guertin, J.R.; LeLorier, J.; Tremblay, C.L. Cost effectiveness of “on demand” HIV pre-exposure prophylaxis for non-injection drug-using men who have sex with men in Canada. Can. J. Infect. Dis. Med. Microbiol. 2015, 26, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Hall, H.I.; An, Q.; Tang, T.; Song, R.; Chen, M.; Green, T.; Kang, J. Prevalence of diagnosed and undiagnosed HIV infection-United States, 2008–2012. MMWR 2015, 64, 657–662. [Google Scholar] [PubMed]
- Centers for Disease Control and Prevention. HIV Surveillance Report, Diagnoses of HIV Infection in the United States and Dependent Areas, 2014; Centers for Disease Control and Prevention: Atlanta, GA, USA, 2014.
- Walensky, R.P.; Jacobsen, M.M.; Bekker, L.G.; Parker, R.A.; Wood, R.; Resch, S.C.; Horstman, N.K.; Freedberg, K.A.; Paltiel, A.D. Potential clinical and economic value of long-acting preexposure prophylaxis for South African women at high-risk for HIV infection. J. Infect. Dis. 2015. [Google Scholar] [CrossRef] [PubMed]
- Schackman, B.R.; Fleishman, J.A.; Su, A.E.; Berkowitz, B.K.; Moore, R.D.; Walensky, R.P.; Becker, J.E.; Voss, C.; Paltiel, A.D.; Weinstein, M.C.; et al. The lifetime medical cost savings from preventing HIV in the United States. Med. Care 2015, 53, 293–301. [Google Scholar] [CrossRef] [PubMed]
- Farnham, P.G.; Gopalappa, C.; Sansom, S.L.; Hutchinson, A.B.; Brooks, J.T.; Weidle, P.J.; Marconi, V.C.; Rimland, D. Updates of lifetime costs of care and quality-of-life estimates for HIV-infected persons in the United States. J. Acquir. Immune Defic. Syndr. 2013, 64, 183–189. [Google Scholar] [CrossRef] [PubMed]

| Parameter | Value | Sensitivity Ranges | Reference | |
|---|---|---|---|---|
| Lower | Upper | |||
| HIV Incidence (Per 100 Person-Years) | ||||
| 25–34 year old MSM in United States | 0.66% | 0.56% | 0.76% | [32,33] |
| 35–44 year old MSM in United States | 0.46% | 0.38% | 0.55% | [32,33] |
| 45–54 year old MSM in United States | 0.24% | 0.19% | 0.29% | [32,33] |
| High-risk scenario | 2.0% | 1.0% | 4.0% | [50] |
| Intervention Efficacy | ||||
| Vaccine efficacy, 2 year average with 4 doses | 31.2% | 1.1% | 52.1% | [14] |
| Decay parameter, λ30 | −2.400 | −2.037 | −2.762 | [44] |
| Vaccine Efficacy, 2 year average with 5 doses | 50.0% | 30.0% | 70.0% | Assumed [51] |
| Decay parameter, λ50 | −2.880 | −2.400 | −3.380 | Calculated [44] |
| Vaccine boosting potential, ρ | 100% | 80% | 100% | Assumed |
| PrEP Efficacy | 86% | 39.4% | 98.5% | [52] |
| Disease Progression | ||||
| Probability of HIV symptoms, monthly | 0.008 | 0.000 | 0.015 | [30,53] |
| Probability of AIDS, monthly | 0.081 | 0.009 | 0.700 | [31] |
| Additional hazard of dying with HIV | 1.770 | 1.170 | 2.550 | [35] |
| AIDS mortality rate | 0.43% | 0.37% | 0.51% | [36] |
| Utilities | ||||
| Healthy utility, age 30–39 | 0.918 | 0.912 | 0.925 | [37] |
| Vaccine AE utility decrement | 0.003 | 0.000 | 0.005 | Assumed |
| PrEP AE utility decrement | 0.008 | 0.000 | 0.020 | Assumed |
| HIV Utility, CD4 >500 | 0.798 | 0.696 | 0.900 | [30,38,40] |
| HIV Utility, CD4 200–500 | 0.780 | 0.767 | 0.793 | [30,38,40] |
| AIDS Utility, CD4 <200 | 0.702 | 0.567 | 0.837 | [30,38,40] |
| Costs 1 | ||||
| Vaccine Price, per dose | $500 | $100 | $1000 | Assumed |
| PrEP drug cost, 30-day supply | $1646 | $893 | $2000 | [13,49] |
| PrEP visit cost, including lab tests | $208 | $156 | $260 | [19] |
| HIV Care if CD4 >500, monthly | $1634 | $1579 | $1689 | [48] |
| ART drug cost | $1211 | $1172 | $1251 | [48] |
| Outpatient costs | $45 | $43 | $47 | [48] |
| Other costs | $378 | $364 | $392 | [48] |
| HIV Care, CD4 200–500, monthly | $1924 | $1817 | $2032 | [48] |
| ART drug cost | $1158 | $1103 | $1212 | [48] |
| Outpatient costs | $54 | $51 | $57 | [48] |
| Other costs | $713 | $663 | $763 | [48] |
| HIV Care, CD4 <200, monthly | $2558 | $2334 | $2783 | [48] |
| ART drug cost | $1162 | $1094 | $1229 | [48] |
| Outpatient costs | $62 | $58 | $67 | [48] |
| Other costs | $1334 | $1182 | $1486 | [48] |
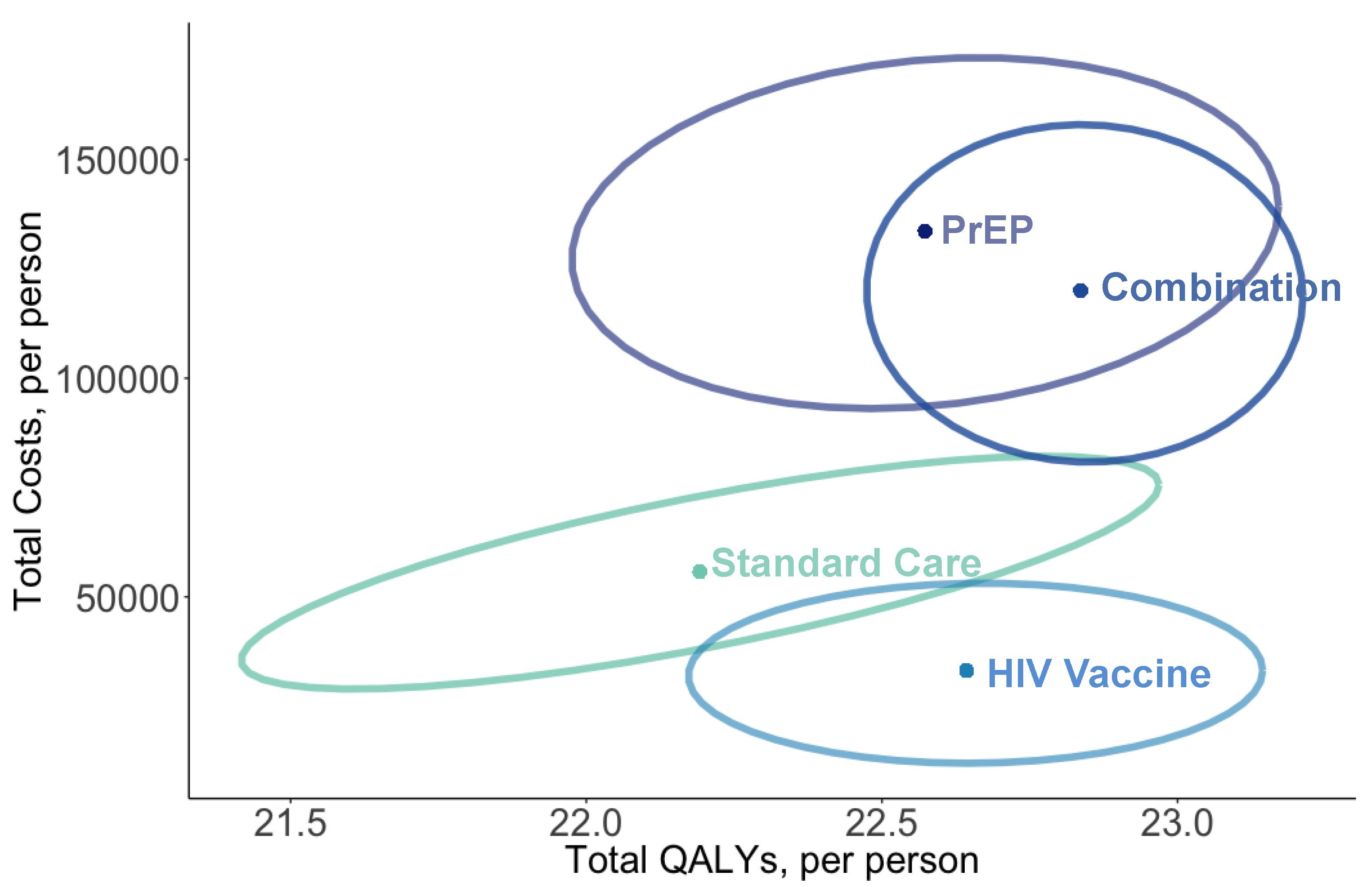
| HIV Prevention Strategy | Total Costs 1 | Total QALYs | HIV Infections | ICER 2 ($/QALY) |
|---|---|---|---|---|
| Standard Care | $51,926 | 22.057 | 170.7 | Dominated |
| PrEP | $130,811 | 22.439 | 128.7 | Dominated |
| HIV Vaccination | $30,870 | 22.580 | 88.3 | Dominant |
| Combination: PrEP and Vaccine | $118,484 | 22.769 | 65.8 | $463,448 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Adamson, B.J.S.; Carlson, J.J.; Kublin, J.G.; Garrison, L.P., Jr. The Potential Cost-Effectiveness of Pre-Exposure Prophylaxis Combined with HIV Vaccines in the United States. Vaccines 2017, 5, 13. https://doi.org/10.3390/vaccines5020013
Adamson BJS, Carlson JJ, Kublin JG, Garrison LP Jr. The Potential Cost-Effectiveness of Pre-Exposure Prophylaxis Combined with HIV Vaccines in the United States. Vaccines. 2017; 5(2):13. https://doi.org/10.3390/vaccines5020013
Chicago/Turabian StyleAdamson, Blythe J. S., Josh J. Carlson, James G. Kublin, and Louis P. Garrison, Jr. 2017. "The Potential Cost-Effectiveness of Pre-Exposure Prophylaxis Combined with HIV Vaccines in the United States" Vaccines 5, no. 2: 13. https://doi.org/10.3390/vaccines5020013
APA StyleAdamson, B. J. S., Carlson, J. J., Kublin, J. G., & Garrison, L. P., Jr. (2017). The Potential Cost-Effectiveness of Pre-Exposure Prophylaxis Combined with HIV Vaccines in the United States. Vaccines, 5(2), 13. https://doi.org/10.3390/vaccines5020013