Hepatitis Vaccines
Abstract
:1. Introduction
2. Hepatitis A Virus (HAV)
2.1. History of Vaccine Development
2.2. Recommendations for Administration of HAV Vaccine
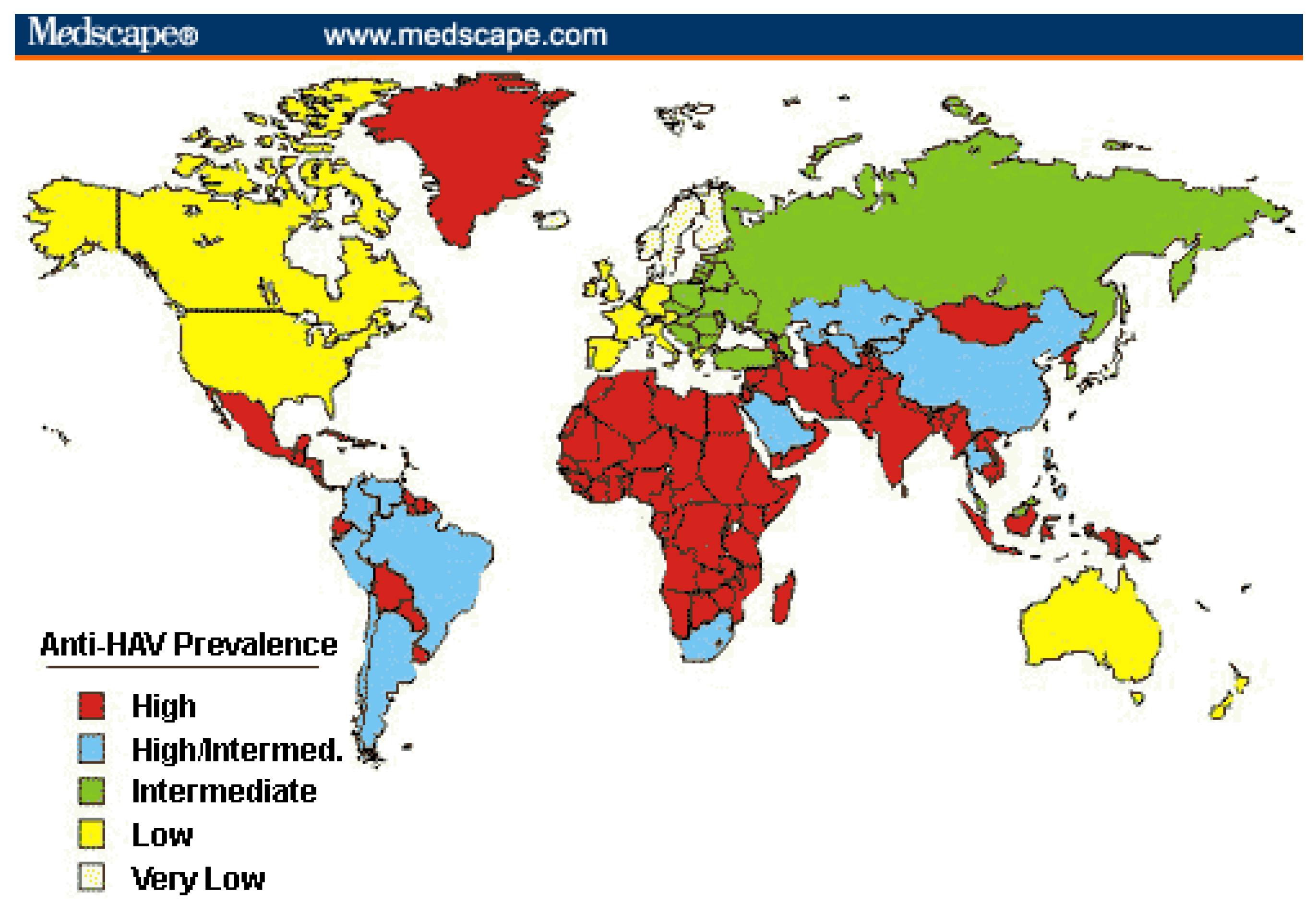
2.3. Impact of Vaccine on Disease Burden
2.4. Remaining Challenges
3. Hepatitis B Virus (HBV)
3.1. History of Vaccine Development
3.2. Recommendations for Administration of HBV Vaccine
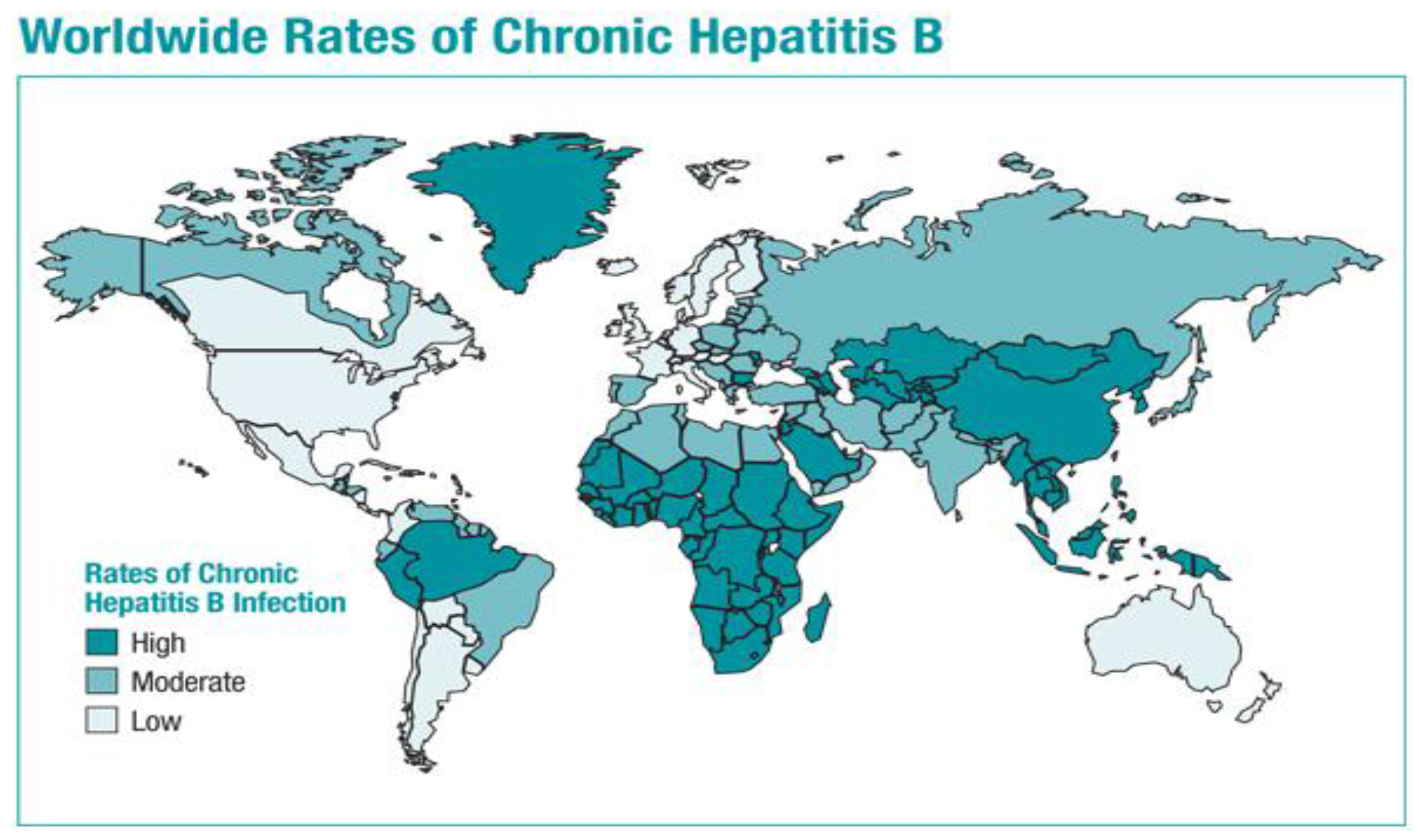
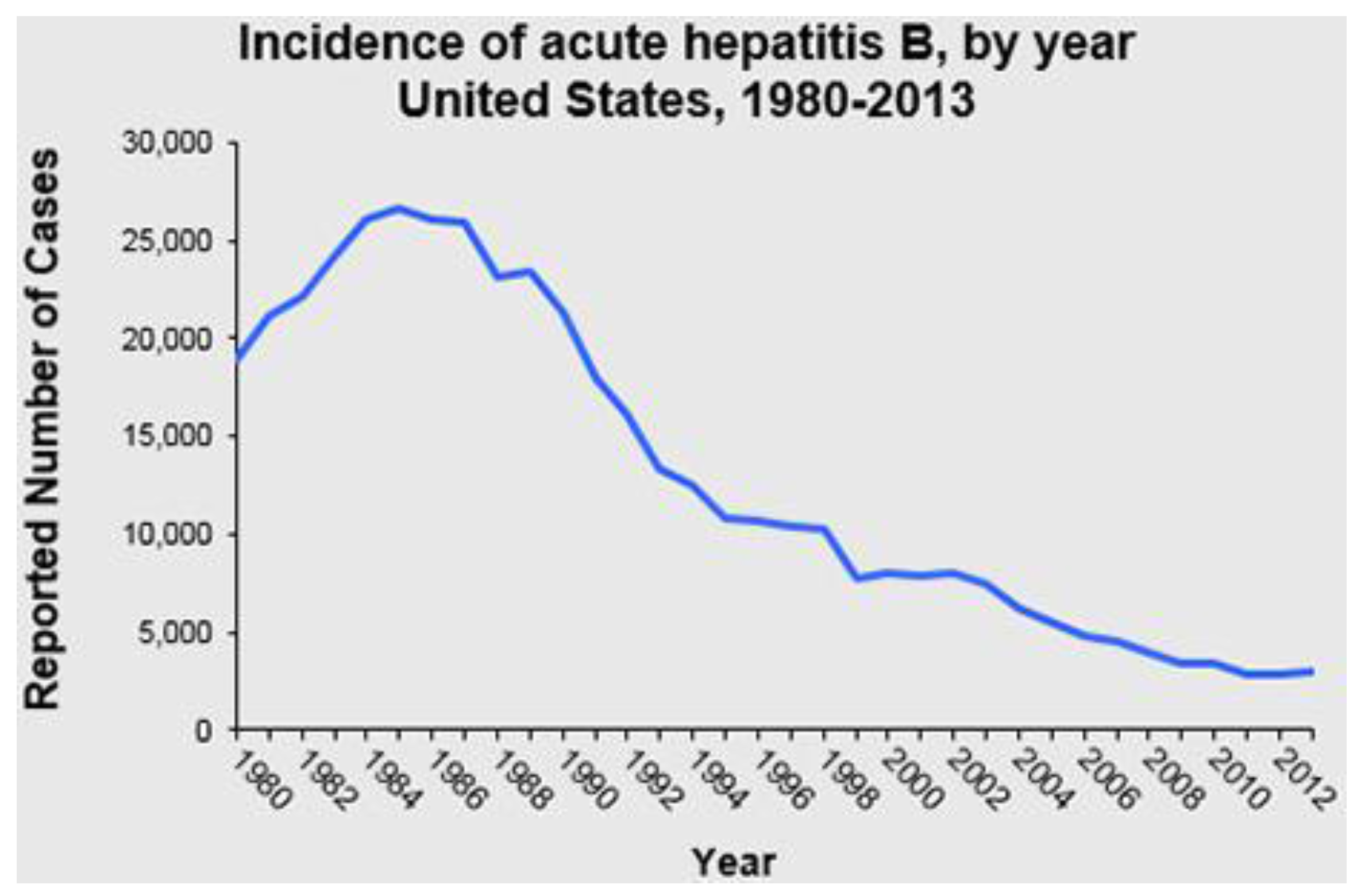
3.3. Impact of the Vaccine on Disease Burden
3.4. Remaining Challenges
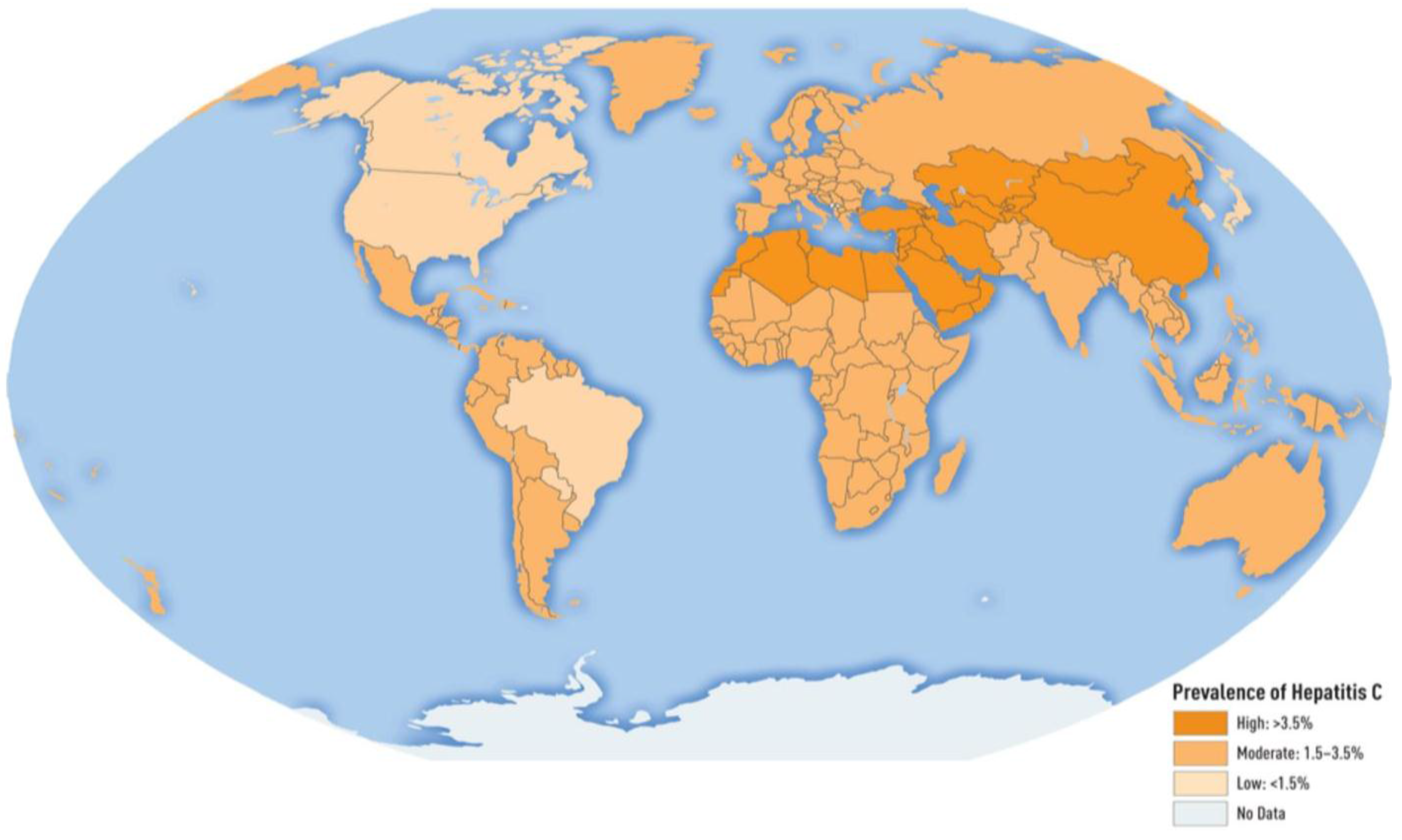
4. Hepatitis C Virus
4.1. History of Vaccine Development
4.2. Remaining Questions
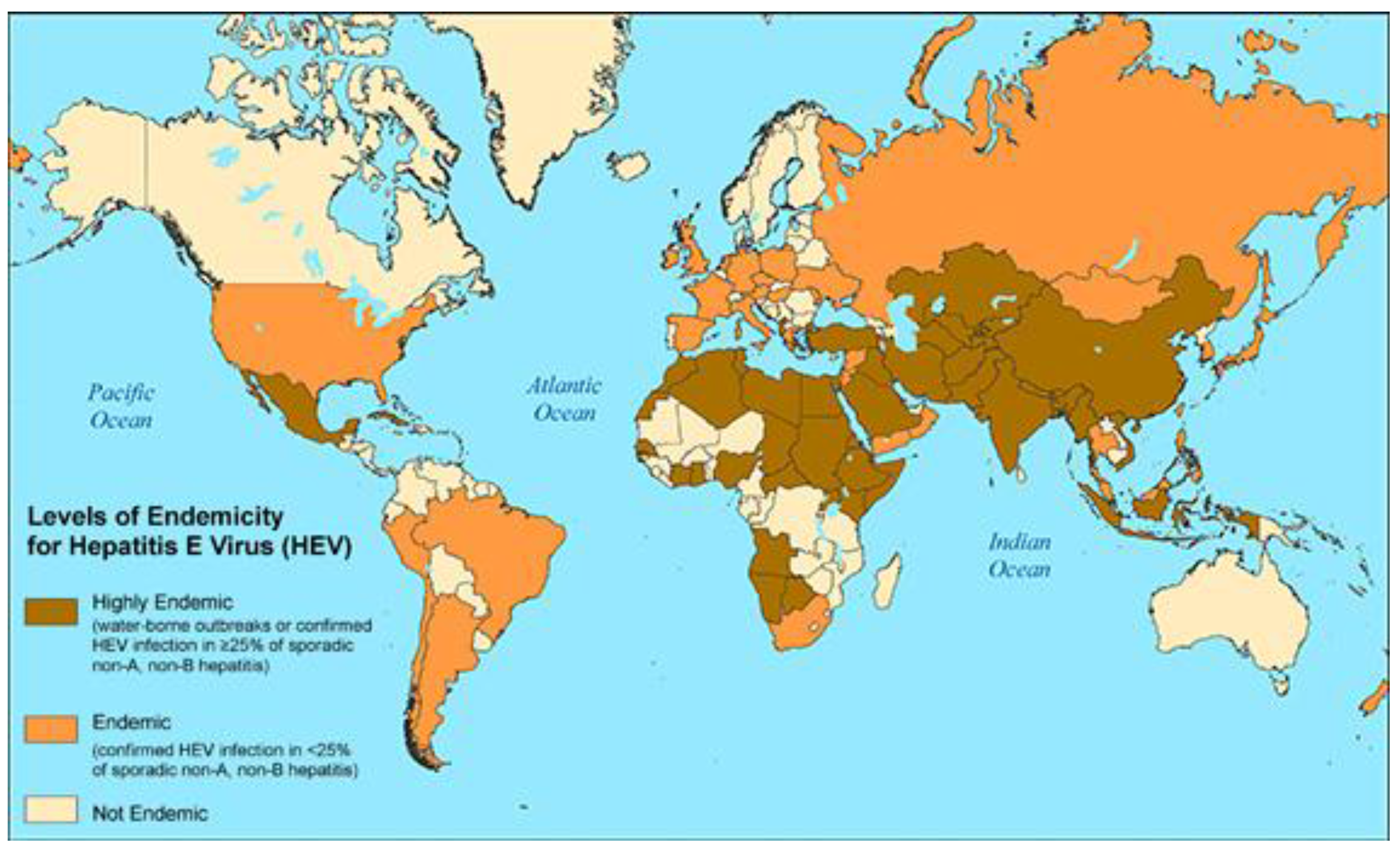
5. Hepatitis E Virus
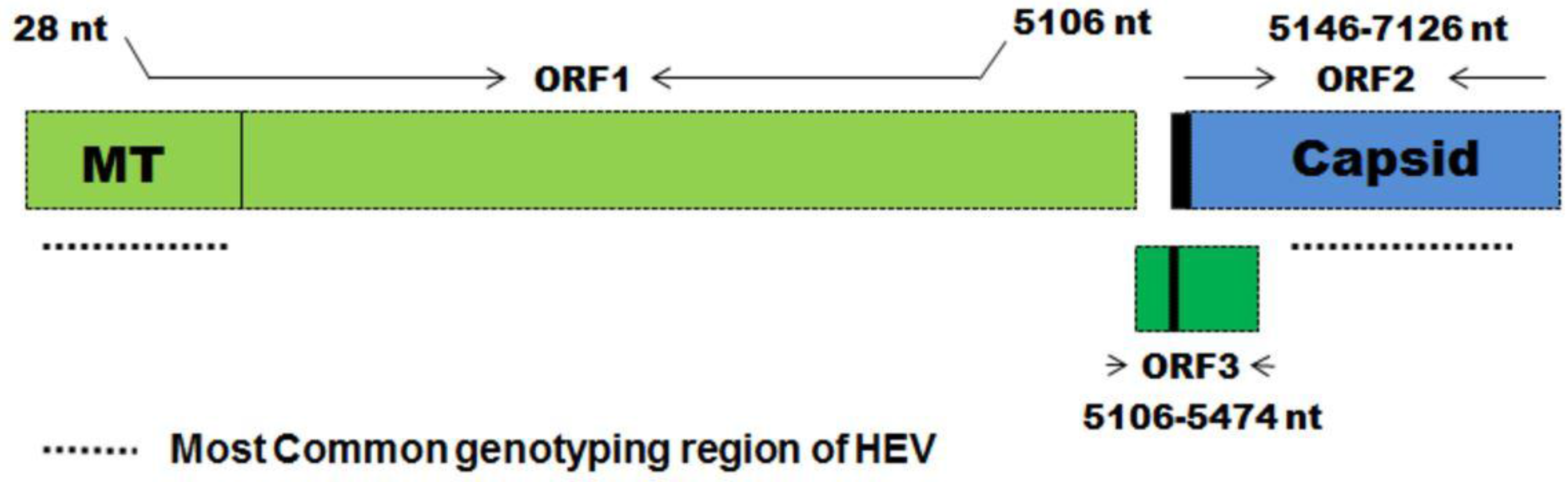
5.1. History of Vaccine Development
5.2. Remaining Questions
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hill, H.A.; Elam-Evans, L.D.; Yankey, D.; Singleton, J.A.; Kolasa, M. National, state, and selected local area vaccination coverage among children aged 19–35 months—United States, 2014. Morb. Mortal. Wkly. Rep. 2015, 64, 889–896. [Google Scholar] [CrossRef]
- Lozano, R.; Naghavi, M.; Foreman, K.; Lim, S.; Shibuya, K.; Aboyans, V.; Abraham, J.; Adair, T.; Aggarwal, R.; Ahn, S.Y.; et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012, 380, 2095–2128. [Google Scholar] [CrossRef]
- Thomas, H.C.; Lok, A.S.F.; Locarnini, S.A.; Zuckerman, J.A. Viral Hepatitis, Fourth Edition; John Wiley & Sons, Ltd.: Oxford, UK, 2013. [Google Scholar]
- Jacobsen, K.H.; Wiersma, S.T. Hepatitis A virus seroprevalence by age and world region, 1990 and 2005. Vaccine 2010, 28, 6653–6665. [Google Scholar] [CrossRef] [PubMed]
- Ciocca, M. Clinical course and consequences of hepatitis A infection. Vaccine 2000, 18, S71–S74. [Google Scholar] [CrossRef]
- WHO position paper on hepatitis A vaccines: June 2012—Recommendations. Vaccine 2013, 31, 285–286.
- Fiore, A.E.; Wasley, A.; Bell, B.P. Prevention of hepatitis A through active or passive immunization: Recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm. Rep. 2006, 55, 1–23. [Google Scholar] [PubMed]
- André, F.E.; D’Hondt, E.; Delem, A.; Safary, A. Clinical assessment of the safety and efficacy of an inactivated hepatitis A vaccine: Rationale and summary of findings. Vaccine 1992, 10, S160–S168. [Google Scholar] [CrossRef]
- Braconier, J.H.; Wennerholm, S.; Norrby, S.R. Comparative immunogenicity and tolerance of Vaqta and Havrix. Vaccine 1999, 17, 2181–2184. [Google Scholar] [CrossRef]
- Bovier, P.A.; Bock, J.; Ebengo, T.F.; Frösner, G.; Glaus, J.; Herzog, C.; Loutan, L. Predicted 30-year protection after vaccination with an aluminum-free virosomal hepatitis A vaccine. J. Med. Virol. 2010, 82, 1629–1634. [Google Scholar] [CrossRef] [PubMed]
- Innis, B.L.; Snitbhan, R.; Kunasol, P.; Laorakpongse, T.; Poopatanakool, W.; Kozik, C.A.; Suntayakorn, S.; Suknuntapong, T.; Safary, A.; Tang, D.B.; et al. Protection against hepatitis A by an inactivated vaccine. JAMA 1994, 271, 1328–1334. [Google Scholar] [CrossRef] [PubMed]
- Victor, J.C.; Monto, A.S.; Surdina, T.Y.; Suleimenova, S.Z.; Vaughan, G.; Nainan, O.V.; Favorov, M.O.; Margolis, H.S.; Bell, B.P. Hepatitis: A vaccine versus immune globulin for post exposure prophylaxis. N. Engl. J. Med. 2007, 357, 1685–1694. [Google Scholar] [CrossRef] [PubMed]
- Wasley, A.; Fiore, A.; Bell, B.P. Hepatitis A in the era of vaccination. Epidemiol. Rev. 2006, 28, 101–111. [Google Scholar] [PubMed]
- Dorell, C.G.; Yankey, D.; Byrd, K.K.; Murphy, T.V. Hepatitis A vaccination coverage among adolescents in the United States. Available online: http://pediatrics.aappublications.org/content/129/2/213 (accessed on 6 September 2015).
- Lopalco, P.L.; Salleras, L.; Barbuti, S.; Germinario, C.; Bruguera, M.; Buti, M.; Dominguez, A. Hepatitis A and B in children and adolescents—What can we learn from Puglia (Italy) and Catalonia (Spain)? Vaccine 2000, 19, 470–474. [Google Scholar] [CrossRef]
- Hanna, J.N.; Hills, S.L.; Humphreys, J.L. Impact of hepatitis A vaccination of Indigenous children on notifications of hepatitis A in north Queensland. Med. J. Aust. 2004, 181, 482–485. [Google Scholar] [PubMed]
- Dagan, R.; Leventhal, A.; Anis, E.; Slater, P.; Ashur, Y.; Shouval, D. Incidence of hepatitis A in Israel following universal immunization of toddlers. JAMA 2005, 294, 202–210. [Google Scholar] [CrossRef] [PubMed]
- Anonychuk, A.M.; Tricco, A.C.; Bauch, C.T.; Pham, B.; Gilca, V.; Duval, B.; John-Baptiste, A.; Woo, G.; Krahn, M. Costeffectiveness analyses of hepatitis A vaccine: A systematic review to explore the effect of methodologicalquality onthe economic attractiveness of vaccination strategies. Pharmacoeconomics 2008, 26, 17–32. [Google Scholar] [CrossRef] [PubMed]
- Michel, M.-L.; Tiollais, P. Hepatitis B vaccines: Protective efficacy and therapeutic potential Vaccins anti-hepatite B: Efficacite protectrice et outil therapeutique potential. Pathol. Biol. 2010, 58, 288–295. [Google Scholar] [CrossRef] [PubMed]
- Maupas, P.; Goudeau, A.; Coursaget, P.; Drucker, J.; Bagros, P. Immunisation against hepatitis B in man. Lancet 1976, 1, 1367–1370. [Google Scholar] [CrossRef]
- Krugman, S.; Giles, J.P.; Hammond, J. Viral hepatitis, type B (MS-2 strain). Studies on active immunization. JAMA 1971, 217, 41–45. [Google Scholar] [CrossRef] [PubMed]
- Dubois, M.-F.; Pourcel, C.; Rousset, S.; Chany, C.; Tiollais, P. Excretion of hepatitis B surface antigen particles from mouse cells transformed with cloned viral DNA. Proc. Natl. Acad. Sci. USA 1980, 77, 4549–4553. [Google Scholar] [CrossRef] [PubMed]
- Valenzuela, P.; Medina, A.; Rutter, W.J.; Ammerer, G.; Hall, B.D. Synthesis and assembly of hepatitis B virus surface antigen particles in yeast. Nature 1982, 298, 347–350. [Google Scholar] [CrossRef] [PubMed]
- Mast, E.E.; Margolis, H.S.; Fiore, A.; Brink, E.W.; Goldstein, S.T.; Wang, S.A.; Moyer, L.A.; Bell, B.P.; Alter, M.J. A comprehensive immunization strategy to eliminate transmission of Hepatitis B Virus infection in the United States. Morb. Mortal. Wkly. Rep. 2005, 54, 1–23. [Google Scholar]
- Kucukoglu, S.; Celebioglu, A.; Caner, I.; Ok, G.; Maden, R. The effects of instrumental touching on infant pain perception and the effects of eutectic mixture of local anesthetics (EMLA) on the reduction of pain. Iran. J. Pediatr. 2015, 25, e532. [Google Scholar] [CrossRef]
- Keck, J.W.; Bulkow, L.R.; Raczniak, G.A.; Negus, S.E.; Zanis, C.L.; Bruce, M.G.; Spradling, P.R.; Teshale, E.H.; McMahon, B.J. Hepatitis B virus antibody levels 7 to 9 years after booster vaccination in Alaska native persons. Clin. Vaccin. Immunol. 2014, 21, 1339–1342. [Google Scholar] [CrossRef] [PubMed]
- Ott, J.J.; Stevens, G.A.; Groeger, J.; Wiersma, S.T. Global epidemiology of hepatitis B virus infection: New estimates of age-specific HBsAg seroprevalence and endemicity. Vaccine 2012, 30, 2212–2219. [Google Scholar] [CrossRef] [PubMed]
- Locarnini, S.; Hatzakis, A.; Chen, D.S.; Lok, A. Strategies to control hepatitis B: Public policy, epidemiology, vaccine and drugs. J. Hepatol. 2015, 62, S76–S86. [Google Scholar] [CrossRef] [PubMed]
- Williams, A. Reduction in the hepatitis B related burden of disease—Measuring the success of universal immunisation programs. Commun. Dis. Intell. Q. Rep. 2002, 26, 458–460. [Google Scholar] [PubMed]
- Thio, C.L.; Guo, N.; Xie, C.; Nelson, K.E.; Ehrhardt, S. Global elimination of mother-to-child transmission of hepatitis B: Revisiting the current strategy. Lancet Infect. Dis. 2015, 15, 981–985. [Google Scholar] [CrossRef]
- Celen, M.K.; Mert, D.; Ay, M.; Dal, T.; Kaya, S.; Yildirim, N.; Gulsun, S.; Barcin, T.; Kalkanli, S.; Dal, M.S. Efficacy and safety of tenofovir disoproxil fumarate in pregnancy for the prevention of vertical transmission of HBV infection. World J. Gastroenterol. 2013, 19, 9377–9382. [Google Scholar] [CrossRef] [PubMed]
- Cao, L.H.; Li, Y.R.; Wang, S.Y.; Liu, Z.M.; Sun, S.C.; Xu, D.B.; Zhang, J.D. Effect of hepatitis B vaccination in hepatitis B surface antibody-negative pregnant mothers on the vertical transmission of hepatitis B virus from father to infant. Exp. Ther. Med. 2015, 10, 279–284. [Google Scholar] [CrossRef] [PubMed]
- Hsu, H.Y.; Chang, M.H.; Ni, Y.H.; Chiang, C.L.; Wu, J.F.; Chen, H.L. Universal infant immunization and occult hepatitis B virus infection in children and adolescents: A population-based study. Hepatology 2015, 61, 1183–11891. [Google Scholar] [CrossRef] [PubMed]
- Travers, C.P.; Connell, J.; Thornton, L.; Keane, E.; Knowles, S.; Murphy, J.F. Follow up of infants born to women with hepatitis B in the National Maternity Hospital. Ir. Med. J. 2015, 108, 147–149. [Google Scholar] [PubMed]
- Geier, D.A.; Kern, J.K.; Hooker, B.S.; King, P.G.; Sykes, L.K.; Geier, M.R. A longitudinal cohort study of the relationship between Thimerosal-containing hepatitis B vaccination and specific delays in development in the United States: Assessment of attributable risk and lifetime care costs. Epidemiol. Glob. Health 2015. [Google Scholar] [CrossRef] [PubMed]
- Alcusky, M.J.; Pawasauskas, J. Adherence to guidelines for Hepatitis B, pneumococcal, and influenza vaccination in patients with diabetes. Clin. Diabetes 2015, 33, 116–122. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Nyamathi, A.; Salem, B.E.; Zhang, S.; Farabee, D.; Hall, B.; Khalilifard, F.; Leake, B. Nursing case management, peer coaching, and hepatitis a and B vaccine completion among homeless men recently released on parole: Randomized clinical trial. Nurs Res. 2015, 64, 177–189. [Google Scholar] [CrossRef] [PubMed]
- Ghomraoui, F.A.; Alfaqeeh, F.A.; Algadheeb, A.S.; Al-Alsheikh, A.S.; Al-Hamoudi, W.K.; Alswat, K.A. Medical students’ awareness of and compliance with the hepatitis B vaccine in a tertiary care academic hospital: An epidemiological study. J. Infect. Public Health. 2015, 9, 60–65. [Google Scholar] [CrossRef] [PubMed]
- Jin, H.; Tan, Z.; Zhang, X.; Wang, B.; Zhao, Y.; Liu, P. Comparison of accelerated and standard Hepatitis B vaccination schedules in high-risk healthy adults: A meta-analysis of randomized controlled trials. PLoS ONE 2015, 10, e0133464. [Google Scholar] [CrossRef] [PubMed]
- LShah, D.P.; Grimes, C.Z.; Nguyen, A.T.; Lai, D.; Hwang, L.Y. Long-term effectiveness of accelerated hepatitis B vaccination schedule in drug users. Am. J. Public Health 2015, 105, e36–e43. [Google Scholar]
- Rey, D.; Piroth, L.; Wendling, M.J.; Miailhes, P.; Michel, M.L.; Dufour, C.; Haour, G.; Sogni, P.; Rohel, A.; Ajana, F.; et al. Safety and immunogenicity of double-dose versus standard-dose hepatitis B revaccination in non-responding adults with HIV-1 (ANRS HB04 B-BOOST): A multicentre, open-label, randomised controlled trial. Lancet Infect. Dis. 2015, 15, 1283–1291. [Google Scholar] [CrossRef]
- Cekic, C.; Aslan, F.; Krc, A.; Gümüs, Z.Z.; Arabul, M.; Yüksel, E.S.; Vatansever, S.; Yurtsever, S.G.; Alper, E.; Ünsal, B. Evaluation of factors associated with response to hepatitis B vaccination in patients with inflammatory bowel disease. Medicine (Baltimore) 2015, 94, e940. [Google Scholar] [CrossRef] [PubMed]
- Mathew, R.O.; Mason, D.L.; Song, R.; Tryniszewski, T.; Kennedy, J.S. Role of T-regulatory cells in the response to hepatitis B vaccine in hemodialysis patients. Hemodial. Int. 2015. [Google Scholar] [CrossRef] [PubMed]
- Yousaf, F.; Gandham, S.; Galler, M.; Spinowitz, B.; Charytan, C. Systematic review of the efficacy and safety of intradermal versus intramuscular hepatitis B vaccination in end-stage renal disease population unresponsive to primary vaccination series. Ren. Fail. 2015, 37, 1080–1088. [Google Scholar] [PubMed]
- Friedrich, P.; Sattler, A.; Müller, K.; Nienen, M.; Reinke, P.; Babel, N. Comparing humoral and cellular immune response against HBV vaccine in Kidney transplant patients. Am. J. Transplant. 2015, 15, 3157–3165. [Google Scholar] [CrossRef] [PubMed]
- Duan, B.W.; Lu, S.C.; Lai, W.; Liu, X.E.; Liu, Y. The detection of (total and ccc) HBV DNA in liver transplant recipients with hepatitis B vaccine against HBV reinfection. Hum. Vaccin. Immunother. 2015, 11, 2490–2494. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.H.; Lee, S.S.; Yu, M.L.; Chang, T.T.; Su, C.W.; Hu, B.S.; Chen, Y.S.; Huang, C.K.; Lai, C.H.; Lin, J.N.; et al. Changing hepatitis D virus epidemiology in a hepatitis B virus endemic area with a national vaccination program. Hepatology 2015, 61, 1870–1879. [Google Scholar] [CrossRef] [PubMed]
- Moon, S.H.; Shin, E.C.; Noh, Y.W.; Lim, Y.T. Evaluation of hyaluronic acid-based combination adjuvant containing monophosphoryl lipid A and aluminum salt for hepatitis B vaccine. Vaccine 2015, 33, 4762–4769. [Google Scholar] [CrossRef] [PubMed]
- Janssen, J.M.; Jackson, S.; Heyward, W.L.; Janssen, R.S. Immunogenicity of an investigational hepatitis B vaccine with a toll-like receptor 9 agonist adjuvant (HBsAg-1018) compared with a licensed hepatitis B vaccine in subpopulations of healthy adults 18–70 years of age. Vaccine 2015, 33, 3614–3618. [Google Scholar] [CrossRef] [PubMed]
- Almeida, M.S.; Borges, O. Nasal vaccines against Hepatitis B: An update. Curr. Pharm. Biotechnol. 2015, 16, 882–890. [Google Scholar] [CrossRef] [PubMed]
- Leroux-Roels, G. Old and new adjuvants for hepatitis B vaccines. Med. Microbiol. Immunol. 2015, 204, 69–78. [Google Scholar] [CrossRef] [PubMed]
- Kosinska, A.D.; Liu, J.; Lu, M.; Roggendorf, M. Therapeutic vaccination and immunomodulation in the woodchuck. Med. Microbiol. Immunol. 2015, 204, 103–114. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Zhang, E.; Ma, Z.; Wu, W.; Kosinska, A.; Zhang, X.; Mὅller, I.; Seiz, P.; Glebe, D.; Wang, B.; et al. Enhancing virus-specific immunity in vivo by combining therapeutic vaccination and PD-L1 blockade in chronic hepadnaviral infection. PLoS Pathog. 2014, 10, e1003856. [Google Scholar] [CrossRef] [PubMed]
- Romanò, L.; Paladini, S.; Galli, C.; Raimondo, G.; Pollicino, T.; Zanetti, A.R. Hepatitis B vaccination. Hum. Vaccin. Immunother. 2015, 11, 53–57. [Google Scholar] [CrossRef] [PubMed]
- Cova, L. Advances and challenges in the development of therapeutic DNA vaccines against hepatitis B virus infection. Curr. Gene Ther. 2014, 14, 149–160. [Google Scholar] [CrossRef] [PubMed]
- Perz, J.F.; Armstrong, G.L.; Farrington, L.A.; Hutin, Y.J.; Bell, B.P. The contributions of hepatitis B virus and hepatitis C virus infections to cirrhosis and primary liver cancer worldwide. J. Hepatol. 2006, 45, 529–538. [Google Scholar] [CrossRef] [PubMed]
- M Mohd Hanafiah, K.; Groeger, J.; Flaxman, A.D.; Wiersma, S.T. Global epidemiology of hepatitis C virus infection: New estimates of age-specific antibody to hepatitis C virus seroprevalence. Hepatology 2013, 57, 1333–1342. [Google Scholar] [CrossRef] [PubMed]
- Simmonds, P. Genetic diversity and evolution of hepatitis C virus—15 years on. J. Gen. Virol. 2004, 85, 3173–3188. [Google Scholar] [CrossRef] [PubMed]
- Sulkoski, M.S.; Gardiner, D.F.; Rodriguez-Torres, M.; Reddy, K.R.; Hassanein, T.; Jacobson, I.M.; Lawitz, E.; Lok, A.; Thuluvath, P.J.; Schwartz, H.; et al. Daclatasvir plus Sofosbuvir for Previously Treated or Untreated Chronic HCV. N. Engl. J. Med. 2014, 370, 211–221. [Google Scholar] [CrossRef] [PubMed]
- Veldt, B.J.; Heathcote, E.J.; Wedemeyer, H.; Rerichen, J.; Hofmann, W.P.; Zeuzem, S.; Manns, M.P.; Hansen, B.E.; Schlam, S.W.; Janssen, H.L. Sustained virologic response and clinical outcomes in patients with chronic hepatitis C and advanced fibrosis. Ann. Intern. Med. 2007, 147, 677–684. [Google Scholar] [CrossRef] [PubMed]
- Osburn, W.O.; Fisher, B.E.; Dowd, K.A.; Urban, G.; Liu, L.; Ray, S.C.; Thomas, D.L.; Cox, A.L. Spontaneous control of primary hepatitis C virus infection and immunity against persistent reinfection. Gastroenterology 2010, 138, 315–324. [Google Scholar] [CrossRef] [PubMed]
- Mehta, S.H.; Cox, A.; Hoover, D.R.; Wang, X.H.; Mao, Q.; Ray, S.; Strathdee, S.A.; Vlahov, D.; Thomas, D.L. Protection against persistence of hepatitis C. Lancet 2002, 359, 1478–1483. [Google Scholar] [CrossRef]
- Fournillier, A.; Frelin, L.; Jacquier, E.; Ahlén, G.; Brass, A.; Gerossier, E.; Holmström, F.; Broderick, K.E.; Sardesai, N.Y.; Bonnefoy, J.Y.; et al. A heterologous prime/boost vaccination strategy enhances the immunogenicity of therapeutic vaccines for hepatitis C virus. J. Infect. Dis. 2013, 208, 1008–1019. [Google Scholar] [CrossRef] [PubMed]
- Swadling, L.; Capone, S.; Antrobus, R.D.; Brown, A.; Richardson, R.; Newell, E.W.; Halliday, J.; Kelly, C.; Bowen, D.; Fergusson, J.; et al. A human vaccine strategy based on chimpanzee adenoviral and MVA vectors that primes, boosts, and sustains functional HCV-specific T cell memory. Sci. Transl. Med. 2014. [Google Scholar] [CrossRef] [PubMed]
- Holtzman, D. Hepatitis C. Available online: http://wwwnc.cdc.gov/travel/yellowbook /2016/infectious-diseases-related-to-travel/hepatitis-c (accessed on 4 March 2016).
- Balayan, M.S.; Andjaparidze, A.G.; Savinskaya, S.S.; Ketiladze, E.S.; Braginsky, D.M.; Savinov, A.P.; Poleschuk, V.F. Evidence for a virus in non-A, non-B hepatitis transmitted via the fecal-oral route. Intervirology 1983, 20, 23–31. [Google Scholar] [PubMed]
- Kamar, N.; Mansuy, J.M.; Cointault, O.; Selves, J.; Abravanel, F.; Danjoux, M.; Otal, P.; Esposito, L.; Durand, D.; Izopet, J.; et al. Hepatitis E virus-related cirrhosis in kidney- and kidney-pancreas-transplant recipients. Am. J. Transplant. 2008, 8, 1744–1748. [Google Scholar] [CrossRef] [PubMed]
- Dalton, H.R.; Stableforth, W.; Thurairajah, P.; Hazeldine, S.; Remnarace, R.; Usama, W.; Farrington, L.; Hamad, N.; Sieberhagen, C.; Ellis, V.; et al. Autochthonous hepatitis E in Southwest England: Natural history, complications and seasonal variation, and hepatitis E virus IgG seroprevalence in blood donors, the elderly and patients with chronic liver disease. Eur. J. Gastroenterol. Hepatol. 2008, 20, 784–790. [Google Scholar] [CrossRef] [PubMed]
- Haffar, S.; Bazerbachi, F.; Lake, J.R. Making the case for the development of a vaccination against hepatitis E. Virus Liver Int. 2015, 35, 311–316. [Google Scholar] [CrossRef] [PubMed]
- Hepatitis E FAQs for Health Professionals. Available online: http://www.cdc.gov/hepatitis/hev/hevfaq.htm (accessed on 31 May 2015).
- Wang, H.; Ji, F.; Liang, H.; Gu, H.; Ning, Z.; Liu, R.; Zhang, G. A Proline-Rich Domain in the Genotype 4 Hepatitis E Virus ORF3 C-Terminus Is Crucial for Downstream V105DLP108 Immunoactivity. PLoS ONE 2015, 10, e0133282. [Google Scholar] [CrossRef] [PubMed]
- Gao, S.; Li, D.; Liu, Y.; Zha, E.; Zhou, T.; Yue, X. Oral immunization with recombinant hepatitis E virus antigen displayed on the Lactococcus lactis surface enhances ORF2-specific mucosal and systemic immune responses in mice. Int. Immunopharmacol. 2015, 24, 140–145. [Google Scholar] [CrossRef] [PubMed]
- Behloul, N.; Wen, J.; Dai, X.; Dong, C.; Meng, J. Antigenic composition and immunoreactivity differences between HEV recombinant capsid proteins generated from different genotypes. Infect. Genet. Evol. 2015, 34, 211–220. [Google Scholar] [CrossRef] [PubMed]
- Shrestha, M.P.; Scott, R.M.; Joshi, D.M.; Mammen, M.P., Jr.; Thapa, G.B.; Thapa, N.; Myint, K.S.; Fourneau, M.; Kuschner, R.A.; Shrestha, S.K.; et al. Safety and efficacy of a recombinant hepatitis E vaccine. N. Engl. J. Med. 2007, 356, 895–903. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhang, X.F.; Huang, S.J.; Wu, T.; Hu, Y.M.; Wang, Z.Z.; Wang, H.; Jiang, H.M.; Wang, Y.J.; Yan, Q.; et al. Long-term efficacy of a hepatitis E vaccine. N. Engl. J. Med. 2015, 372, 914–922. [Google Scholar] [CrossRef] [PubMed]
- Hussain, Z. Genomic Heterogeneity of Hepatitis Viruses (A-E): Role in Clinical Implications and Treatment, Practical Management of Chronic Viral Hepatitis. Available online: http://www.intechopen.com/books/practical-management-of-chronic-viral-hepatitis/genomic-heterogeneity-of-hepatitis-viruses-a-e-role-in-clinical-implications-and-treatment (accessed on 4 March 2016).
- Zhu, F.C.; Zhang, J.; Zhang, X.F.; Zhou, C.; Wang, Z.Z.; Huang, S.J.; Wang, H.; Yang, C.L.; Jiang, H.M.; Cai, J.P.; et al. Efficacy and safety of a recombinant Hepatitis E vaccine in adults: A large scaled, randomized, double blinded placebo controlled, phase 3 trial. Lancet 2010, 376, 895–902. [Google Scholar] [CrossRef]
- Harmanci, H.; Duclos, P.; Hernandez, C.A.R.; Meek, A.; Balakrishnan, M.R.; Arora, N.K.; Stefan, Z.W. World Health Organization approaches to evaluating the potential use and quality of Hepatitis E vaccine. Open Forum Infect. Dis. 2014. [Google Scholar] [CrossRef] [PubMed]
- Nelson, K.E.; Shih, J.W.; Zhang, J.; Zhao, Q.; Xia, N.; Ticehurst, J.R.; Labrique, A.B. Hepatitis e vaccine to prevent morbidity and mortality during epidemics. Open Forum Infect. Dis. 2014. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Zhou, Z.; Wei, F.X.; Huang, S.J.; Tan, Z.; Fang, Y.; Zhu, F.C.; Wu, T.; Zhang, J.; Xia, N.S. Modeling the long-term antibody response of a hepatitis E vaccine. Vaccine 2015, 33, 4124–4129. [Google Scholar] [CrossRef] [PubMed]
- Joshi, S.S.; Arankalle, V.A. Enhanced humoral response in pregnant mice immunized with liposome encapsulated recombinant neutralizing epitope protein of Hepatitis-E virus. Virol. J. 2015. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Zhang, J.; Xia, N. Lessons from hepatitis E vaccine design. Curr. Opin. Virol. 2015, 11, 130–136. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zeng, H.; Liu, P.; Liu, L.; Xia, J.; Wang, L.; Zou, Q.; Wang, L.; Zhuang, H. Hepatitis E vaccine immunization for rabbits to prevent animal HEV infection and zoonotic transmission. Vaccine 2015, 33, 4922–4928. [Google Scholar] [CrossRef] [PubMed]
| Individuals traveling to or working in countries with high or intermediate rates of hepatitis A |
| Individuals with chronic liver disease |
| Individuals with clotting factor disorders |
| Men who have sex with men |
| Illicit drug users (injection and non-injection) |
| Individuals with close personal contact with an international adoptee from a country of high or intermediate endemicity during the first 60 days following arrival in the United States |
| Individuals working with HAV-infected primates or with HAV in a research laboratory |
| Individuals with recent exposure for post-exposure prophylaxis |
| People at Risk of Infection by Sexual Exposure |
| Sex partners of HBsAg-positive people |
| People with more than one sex partner in the past six months |
| People seeking evaluation or treatment for a sexually transmitted infection |
| Men who have sex with men |
| People at Risk of Infection by Percutaneous or Mucosal Exposure to Blood |
| Current or recent injection drug users |
| Household contacts of HBsAg-positive people |
| Residents and staff of facilities for people with developmental disabilities |
| Health-care workers with risk of exposure to blood-contaminated body fluids |
| People with end-stage renal disease |
| People with diabetes mellitus 19 through 59 years of age |
| Others |
| International travelers to regions with increased HBV prevalence |
| People with chronic liver disease |
| People with human immunodeficiency (HIV) infection |
| Study Type | Vaccine biology | Study Number | Individual type | Outcome |
|---|---|---|---|---|
| Preventative | AdCh3NSmut1 MVA-NSmut | NCT01436357 | Healthy at risk population | Pending/Jan 2016 |
| Preventative | AdCh3NSmut1 MVA-NSmut | NCT02362217 | Healthy subjects/HIV + | Pending/Oct 2016 |
| Therapeutic/peptide antigens | PEV2A PEV2B | NCT00445419 | Hep C+ | Updated 2/2010 – no results |
| Therapeutic | MRKAd5 HIV-1 gag vaccine | NCT00857311 | Hep C+ | Canceled - non-efficacious results of previous study using similar agent |
| Therapeutic | Recombinant IMPs Ad6NSmut and MVA-NSmu | NCT01701336 | Chronic Hep C+ | Last updated 2013, no results |
| Therapeutic | TG4040 | NCT00449124 | Chronic Hep C+ | Withdrawn |
| Therapeutic | GI-5005 | NCT00124215 | Chronic Hep C+ | Completed 2010 – no results |
| Therapeutic | IC41 | NCT00601770 | Chronic Hep C+ | Phase 2 – no results 2014 |
| Therapeutic – Genotype 1a | Autologous dendritic cells transduced with Ad encoding NS3 | NCT02309086 | Chronic Hep C+ | Study complete 2015 – no results reported |
| Shrestha et al. NEJM–March 2007 [74] | Zhu et al. Lancet–August 2010 [77] | |
|---|---|---|
| Phase study | Phase II trial | Phase III trial |
| Type of study | Randomized, double-blind, placebo controlled | Randomized, double-blind, placebo controlled |
| Company | GlaxoSmithKline Biologicals | Xiamen Innovax Biotech |
| Country | Nepal | Jiangsu province, China |
| Recombinant protein ORF2 | Baculovirus expressed 56 kDa | E. coli expressed HEV 239 |
| Number of vaccines | 1794 healthy subjects | 112 604 healthy subjects |
| Randomization | HEV vaccine vs. placebo | HEV vs. HBV vaccine |
| Population | Mostly males (99.6%) Young (mean age 25 years) | Males and females, 16–65 years |
| HEV genotypes | Genotype 1 prevalent | Genotypes 1 and 4 prevalent with predominance |
| Doses | 20 µg | 30 µg |
| Route of administration | Intramuscularly | Intramuscularly |
| Intervals between doses | 0, 1, 6 months | 0, 1, 6 months |
| Primary end-point | Prevention of clinically overt HEV disease | Prevention of clinically overt HEV disease |
| Follow-up period | 2 years post-vaccination | 13 month post-vaccination |
| Efficacy | ||
| After 1st dose | 87.5% | 95.5% |
| After 2nd dose | 85.7% | 100% |
| After 3rd dose | 95.5% | 100% |
| Side effects Commercialization | Increased injection-site pain Not further developed | No side effects Hecolin® (Innovax) |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ogholikhan, S.; Schwarz, K.B. Hepatitis Vaccines. Vaccines 2016, 4, 6. https://doi.org/10.3390/vaccines4010006
Ogholikhan S, Schwarz KB. Hepatitis Vaccines. Vaccines. 2016; 4(1):6. https://doi.org/10.3390/vaccines4010006
Chicago/Turabian StyleOgholikhan, Sina, and Kathleen B. Schwarz. 2016. "Hepatitis Vaccines" Vaccines 4, no. 1: 6. https://doi.org/10.3390/vaccines4010006
APA StyleOgholikhan, S., & Schwarz, K. B. (2016). Hepatitis Vaccines. Vaccines, 4(1), 6. https://doi.org/10.3390/vaccines4010006





