EBV-Associated Cancer and Autoimmunity: Searching for Therapies
Abstract
:1. Introduction
2. Experimental Section
3. Results
3.1. Peptide Commonality between EBNA1 and the Human Proteome
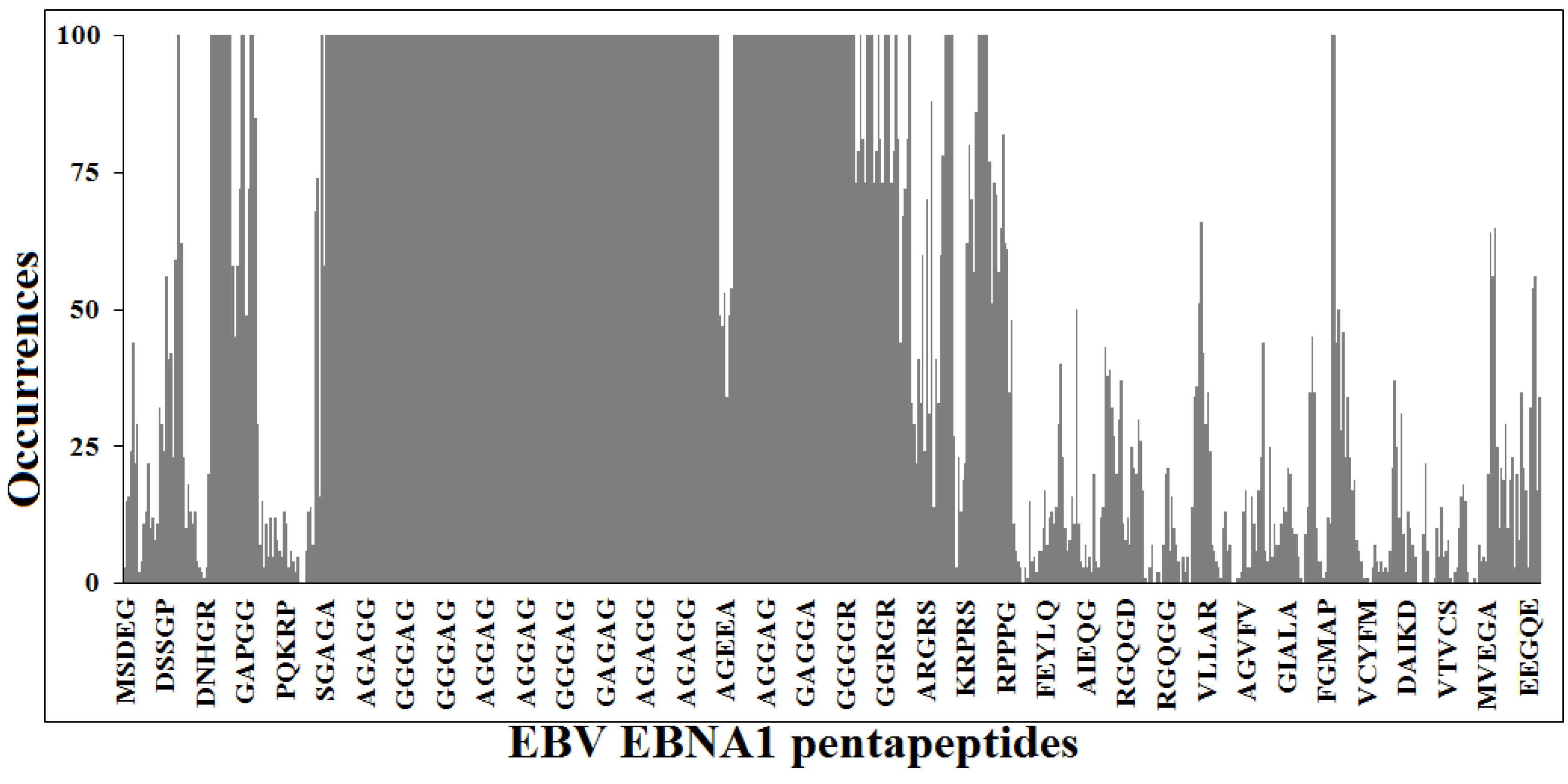
- (1)
- The EBNA154–62PGAPGGSGS nonapeptide is common to the human protein zyxin (UniProtKB/Swiss-Prot IDs: Q15942, ZYX_HUMAN), a component of a signal transduction pathway that mediates adhesion-stimulated changes in gene expression [57];
- (2)
- The AGAGGAGAG nonapeptide is repeated five times in EBNA1, and is shared with the human ubiquitin-conjugating enzyme E2 Q1 (UniProtKB/Swiss-Prot IDs: Q7Z7E8, UB2Q1_HUMAN) that catalyzes the covalent attachment of ubiquitin to other proteins [58];
- (3)
- The GGGAGGAGG nonapeptide is repeated five times in EBNA1, and is shared with the transcription factor jun-B (UniProtKB/Swiss-Prot IDs: P17275, JUNB_HUMAN). JUNB is a transcription factor involved in regulating gene activity following the primary growth factor response [59] and inhibits proliferation and transformation in B-lymphoid cells [60];
- (4)
- In addition, the GGGAGGAGG nonapeptide is also present in the human far upstream element-binding protein 2 (UniProtKB/Swiss-Prot IDs: Q92945, FUBP2_HUMAN). FUBP2 binds to the dendritic targeting element and may play a role in mRNA trafficking. FUBP2 also mediates exon inclusion in transcripts that are subject to tissue-specific alternative splicing [61];
- (5)
- The EBNA140–50GRGRGRGRGRGRGRG undecapeptide is also present in the human small nuclear ribonucleoprotein SmD1, a core component of the spliceosomal U1, U2, U4 and U5 small nuclear ribonucleoproteins (snRNPs), the building blocks of the spliceosome [62]. Importantly, this dipeptide Gly-Arg repeat crossreacts with Abs against an SmD-like epitope recognized by sera from SLE patients [63], thus possibly underlying the EBV-SLE association [10,11].
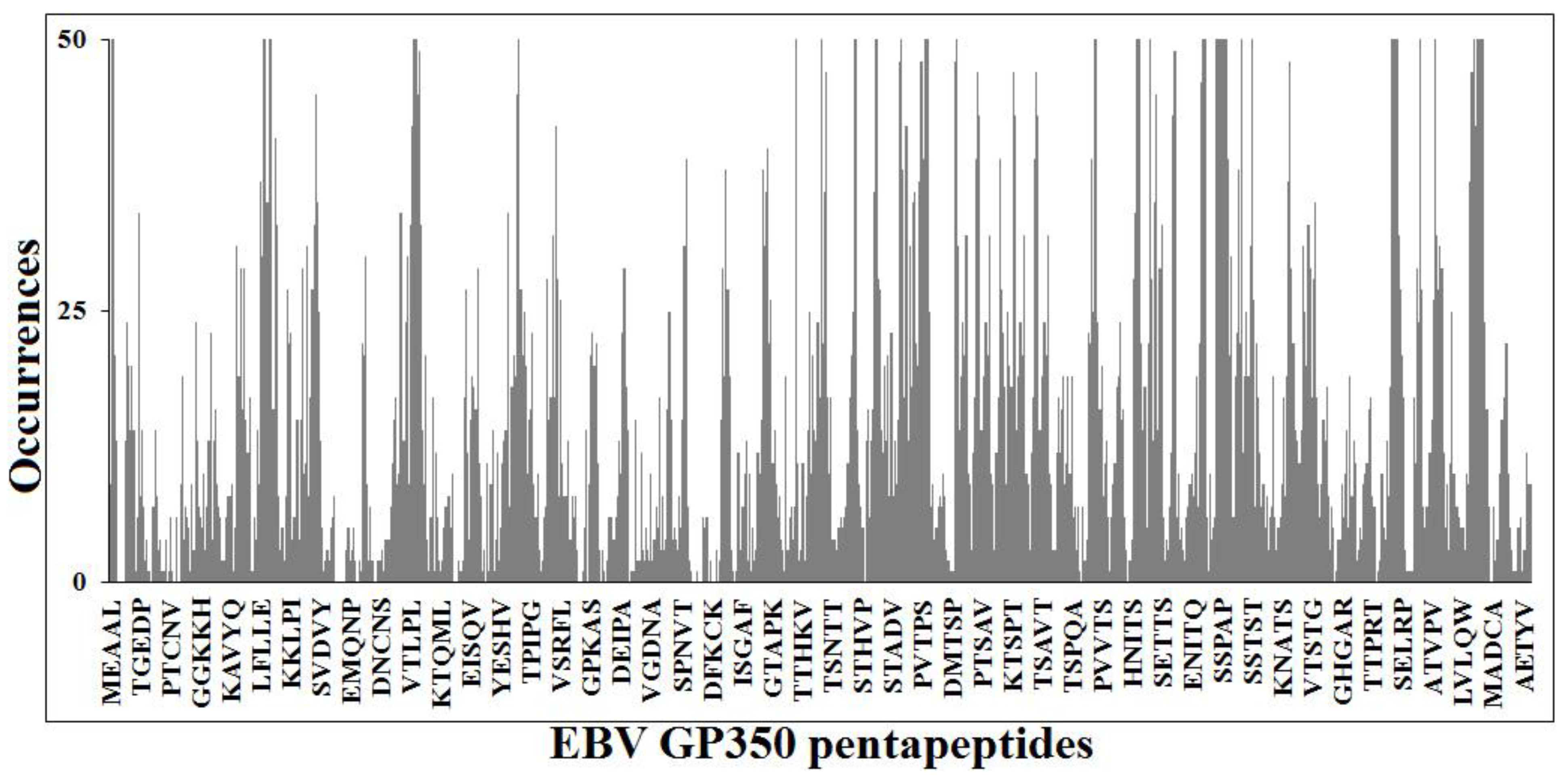
3.2. Searching for a Vaccine: Unique EBNA1 Sequences
| Position a | Sequence b,c | Position a | Sequence b,c | Position a | Sequence b,c |
|---|---|---|---|---|---|
| 80–84 | IGCKG | 467–471 | KHRGQ | 584–588 | MTKPA |
| 81–85 | GCKGA | 476–480 | PKFEN | 588–592 | APTCN |
| 82–86 | CKGAH | 499–503 | EEGNW | 589–593 | PTCNI |
| 461–465 | KGGWF | 500–504 | EGNWV | 598–602 | CSFDD |
| 464–468 | WFGKH | 561–565 | YFMVF | 609–613 | WFPPM |
| IEDB ID a | Epitope Sequence b,c | Immune Context | References |
|---|---|---|---|
| 1219 | aevlkdaikdlvMTKPAptc | B | [66] |
| 8395 | dggrrkKGGWFGKHr | T | [67,68] |
| 8397 | dggrrkKGGWFgrhr | T | [69] |
| 11651 | EEGNWVagvfvyggsktslynlrrg | T | [53] |
| 26761 | ikdlvMTKPAPTCNI | T | [70] |
| 30951 | KGGWFGKHRGQggs | B,T | [71,72] |
| 39079 | lresivcYFMVFlqthifae | T | [67] |
| 39080 | lresivcYFMVFlqthifaevlkda | T | [53] |
| 45378 | nPKFENiaeglrall | T | [67,68,69] |
| 45379 | nPKFENiaeglrallarshv | T | [55,73] |
| 45380 | nPKFENiaeglrallarshverttde | T | [74,75] |
| 48948 | ppWFPPMvegaaa | T | [76] |
| 49056 | pqpgplresivcYFMVFlqt | T | [53] |
| 49593 | PTCNIkatvCSFDDgvdlpp | T | [67,69] |
| 49594 | PTCNIkvtvCSFDDgvdlppWFPPM | T | [53] |
| 55299 | rpqkrpscIGCKGthggtga | B | [66] |
| 55336 | rpscIGCKGthggtg | T | [77] |
| 55684 | rrpqkrpscIGCKGt | T | [67,69] |
| 56433 | rvtvCSFDDgvdlppWFPPM | T | [67] |
| 59875 | snPKFENiaeglrvllarsh | T | [54,55] |
| 67891 | vcYFMVFlqthifae | T | [70] |
| 69559 | vlkdaikdlvMTKPAPTCNI | T | [67,69] |
| 73861 | YFMVFlqthifae | T | [76] |
| 73862 | YFMVFlqthifaevl | T | [77] |
| 93570 | PKFENiaeglr | T | [78] |
| 118828 | gsgprhrdgvrrpqkrpscIGCKGthggtg | B | [79] |
| EBV Strain | ID | Consensus Peptide Sequences | ||||
|---|---|---|---|---|---|---|
| GD1 | Q3KSS4 | IGCKG | GKHRG | APTCNI | CSFDD | WFPPM |
| B95-8 | P03211 | IGCKG | GKHRG | APTCNI | CSFDD | WFPPM |
| AG876 | Q1HVF7 | IGCKG | GKHRG | APTCNI | CSFDD | WFPPM |
4. Discussion
- EBNA1 targeting to dendritic cells to stimulate protective T-cell responses [80];
- EBV-specific cytotoxic T-lymphocytes to control EBV-related lymphoproliferation [81];
- a live recombinant virus, expressing under the 11K vaccinia promoter the major EBV membrane antigen BNLF-1 MA (GP 220-340) to protect against and/or delay EBV infection [82];
- EBV vaccines based on virus-like particles that mimic the structure of the parental virus but lack the viral genome [86];
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Appendix
References
- Saha, A.; Robertson, E.S. Epstein-Barr virus-associated B-cell lymphomas: Pathogenesis and clinical outcomes. Clin. Cancer. Res. 2011, 17, 3056–3063. [Google Scholar]
- Mueller, N.E.; Grufferman, S. Hodgkin lymphoma. In Cancer Epidemiology and Prevention, 3rd ed.; Schottenfeld, D., Fraumeni, J.F., Jr., Eds.; Oxford University Press: New York, NY, USA, 2006; pp. 872–897. [Google Scholar]
- Adam, P.; Bonzheim, I.; Fend, F.; Quintanilla-Martínez, L. Epstein-Barr virus-positive diffuse large B-cell lymphomas of the elderly. Adv. Anat. Pathol. 2011, 18, 349–355. [Google Scholar] [PubMed]
- Piccaluga, P.P.; Gazzola, A.; Agostinelli, C.; Bacci, F.; Sabattini, E.; Pileri, S.A. Pathobiology of Epstein-Barr virus-driven peripheral T-cell lymphomas. Semin. Diagn. Pathol. 2011, 28, 234–244. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.T.; Lin, C.W. EBV-encoded miR-BART20-5p and miR-BART8 inhibit the IFN-γ-STAT1 pathway associated with disease progression in nasal NK-cell lymphoma. Am. J. Pathol. 2014, 184, 1185–1197. [Google Scholar] [CrossRef]
- Kanazawa, T.; Hiramatsu, Y.; Iwata, S.; Siddiquey, M.N.; Sato, Y.; Suzuki, M.; Ito, Y.; Goshima, F.; Murata, T.; Kimura, H. Anti-CCR4 monoclonal antibody mogamulizumab for the treatment of EBV-associated T- and NK-Cell lymphoproliferative diseases. Clin. Cancer. Res. 2014. [Google Scholar] [CrossRef]
- Yen, C.Y.; Lu, M.C.; Tzeng, C.C.; Huang, J.Y.; Chang, H.W.; Chen, R.S.; Liu, S.Y.; Liu, S.T.; Shieh, B.; Li, C. Detection of EBV infection and gene expression in oral cancer from patients in Taiwan by microarray analysis. J. Biomed. Biotechnol. 2009. [Google Scholar] [CrossRef]
- Alibek, K.; Kakpenova, A.; Baiken, Y. Role of infectious agents in the carcinogenesis of brain and head and neck cancers. Infect. Agent. Cancer 2013. [Google Scholar] [CrossRef]
- Iizasa, H.; Nanbo, A.; Nishikawa, J.; Jinushi, M.; Yoshiyama, H. Epstein-Barr Virus (EBV)-associated Gastric Carcinoma. Viruses 2012, 4, 3420–3439. [Google Scholar] [CrossRef] [PubMed]
- Incaprera, M.; Rindi, L.; Bazzichi, A.; Garzelli, C. Potential role of the Epstein-Barr virus in systemic lupus erythematosus autoimmunity. Clin. Exp. Rheumatol. 1998, 16, 289–294. [Google Scholar]
- Poole, B.D.; Gross, T.; Maier, S.; Harley, J.B.; James, J.A. Lupus-like autoantibody development in rabbits and mice after immunization with EBNA-1 fragments. J. Autoimmun. 2008, 31, 362–371. [Google Scholar] [CrossRef] [PubMed]
- Youinou, P.; Buisson, M.; Berthelot, J.M.; Jamin, C.; Le Goff, P.; Genoulaz, O.; Lamour, A.; Lydyard, P.M.; Seigneurin, J.M. Anti-Epstein-Barr virus-nuclear antigen-1, -2A and -2B antibodies in rheumatoid arthritis patients and their relatives. Autoimmunity 1992, 13, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Toussirot, E.; Roudier, J. Pathophysiological links between rheumatoid arthritis and the Epstein-Barr virus: An update. Joint Bone Spine 2007, 74, 418–426. [Google Scholar] [PubMed]
- Csuka, D.; Simon, D.; Hóbor, R.; Uray, K.; Prohászka, Z.; Bánlaki, Z.; Jani, P.K.; Szilágyi, Á.; Hudecz, F.; Rajczy, K.; et al. Serum concentration of immunoglobulin G-type antibodies against the whole Epstein-Barr nuclear antigen 1 and its aa35–58 or aa398–404 fragments in the sera of patients with systemic lupus erythematosus and multiple sclerosis. Clin. Exp. Immunol. 2013, 171, 255–262. [Google Scholar] [CrossRef] [PubMed]
- Rand, K.H.; Houck, H.; Denslow, N.D.; Heilman, K.M. Epstein-Barr virus nuclear antigen-1 (EBNA-1) associated oligoclonal bands in patients with multiple sclerosis. J. Neurol. Sci. 2000, 173, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Hjalgrim, H.; Friborg, J.; Melbye, M. The epidemiology of EBV and its association with malignant disease. In Human Herpesviruses: Biology, Therapy, and Immunoprophylaxis; Arvin, A., Campadelli-Fiume, G., Mocarski, E., Moore, P.S., Roizman, B., Whitley, R., Yamanishiet, K., Eds.; Cambridge University Press: Cambridge, UK, 2007; Chapter 53. [Google Scholar]
- Capone, G.; Calabrò, M.; Lucchese, G.; Fasano, C.; Girardi, B.; Polimeno, L.; Kanduc, D. Peptide matching between Epstein-Barr virus and human proteins. Pathog. Dis. 2013, 69, 205–212. [Google Scholar] [CrossRef] [PubMed]
- Ferlay, J.; Soerjomataram, I.; Ervik, M.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. GLOBOCAN 2012 v1.0, Cancer Incidence and Mortality Worldwide: IARC CancerBase No. 11 (Internet). Available at: http://globocan.iarc.fr (accessed on 13 December 2013).
- Browne, P.; Chandraratna, D.; Angood, C.; Tremlett, H.; Baker, C.; Taylor, B.V.; Thompson, A.J. Atlas of multiple sclerosis 2013: A growing global problem with widespread inequity. Neurology 2014, 83, 1022–1024. [Google Scholar] [CrossRef] [PubMed]
- Uramoto, K.M.; Michet, C.J., Jr.; Thumboo, J.; Sunku, J.; O’Fallon, W.M.; Gabriel, S.E. Trends in the incidence and mortality of systemic lupus erythematosus, 1950–1992. Arthritis Rheum. 1999, 42, 46–50. [Google Scholar] [CrossRef] [PubMed]
- Gibofsky, A. Overview of epidemiology, pathophysiology, and diagnosis of rheumatoid arthritis. Am. J. Manag. Care 2012, 18, S295–S302. [Google Scholar]
- Kanduc, D. Immunogenicity in peptide-immunotherapy: From self/nonself to similar/dissimilar sequences. Adv. Exp. Med. Biol. 2008, 640, 198–207. [Google Scholar] [PubMed]
- Kanduc, D. “Self-nonself” peptides in the design of vaccines. Curr. Pharm. Des. 2009, 15, 3283–3289. [Google Scholar] [CrossRef] [PubMed]
- Kanduc, D. Protein information content resides in rare peptide segments. Peptides 2010, 31, 983–988. [Google Scholar] [CrossRef] [PubMed]
- Kanduc, D. The self/nonself issue: A confrontation between proteomes. SelfNonself 2010, 1, 255–258. [Google Scholar] [CrossRef]
- Zeng, M.S.; Li, D.J.; Liu, Q.L.; Song, L.B.; Li, M.Z.; Zhang, R.H.; Yu, X.J.; Wang, H.M.; Ernberg, I.; Zeng, Y.X. Genomic sequence analysis of Epstein-Barr virus strain GD1 from a nasopharyngeal carcinoma patient. J. Virol. 2005, 79, 15323–15330. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.H.; Yeh, L.S.; Huang, H.; Arminski, L.; Castro-Alvear, J.; Chen, Y.; Hu, Z.; Kourtesis, P.; Ledley, R.S.; Suzek, B.E.; et al. The protein information resource. Nucleic Acids Res. 2003, 31, 345–347. [Google Scholar] [CrossRef]
- Vita, R.; Zarebski, L.; Greenbaum, J.A.; Emami, H.; Hoof, I.; Salimi, N.; Damle, R.; Sette, A.; Peters, B. The immune epitope database 2.0. Nucleic Acids Res. 2010, 38, D854–D862. [Google Scholar] [CrossRef] [PubMed]
- Thompson, J.D.; Higgins, D.G.; Gibson, T.J. CLUSTAL W: Improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 1994, 22, 4673–4680. [Google Scholar] [CrossRef] [PubMed]
- Benjamin, D.C.; Berzofsky, J.A.; East, I.J.; Gurd, F.R.N.; Hannum, C.; Leach, S.J.; Margoliash, E.; Michael, J.G.; Miller, A.; Prager, E.M.; et al. The antigenic structure of proteins: a reappraisal. Annu. Rev. Immunol. 1984, 2, 67–101. [Google Scholar] [CrossRef] [PubMed]
- Lerner, R.A. Tapping the immunological repertoire to produce antibodies of predetermined specificity. Nature 1982. [Google Scholar] [CrossRef]
- Sutcliffe, J.G.; Shinnick, T.M.; Green, N.; Lerner, R.A. Antibodies that react with predetermined sites on proteins. Science 1983, 219, 660–666. [Google Scholar] [CrossRef] [PubMed]
- Shinnick, T.M.; Sutcliffe, J.G.; Green, N.; Lerner, R.A. Peptide-elicited protein-reactive antibodies in molecular biology and medicine. J. Invest. Dermatol. 1984, 83, 112S–115S. [Google Scholar] [CrossRef] [PubMed]
- Niman, H.L.; Houghten, R.A.; Walker, L.E.; Reisfeld, R.A.; Wilson, I.A.; Hogle, J.M.; Lerner, R.A. Generation of protein-reactive antibodies by short peptides is an event of high frequency: Implications for the structural basis of immune recognition. Proc. Natl. Acad. Sci. USA 1983, 80, 4949–4953. [Google Scholar] [CrossRef] [PubMed]
- Yao, B.; Zhang, L.; Liang, S.; Zhang, C. SVMTriP: A method to predict antigenic epitopes using support vector machine to integrate tri-peptide similarity and propensity. PLOS ONE 2012, 7, e45152. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.W.; Lin, Y.C.; Pai, T.W.; Chang, H.T. Prediction of B-cell linear epitopes with a combination of support vector machine classification and amino acid propensity identification. J. Biomed. Biotechnol. 2011. [Google Scholar] [CrossRef]
- Singh, H.; Ansari, H.R.; Raghava, G.P.S. Improved method for linear B-cell epitope prediction using antigen’s primary sequence. PLOS ONE 2013, 8, e62216. [Google Scholar] [CrossRef] [PubMed]
- Hemmer, B.; Kondo, T.; Gran, B.; Pinilla, C.; Cortese, I.; Pascal, J.; Tzou, A.; McFarland, H.F.; Houghten, R.; Martin, R. Minimal peptide length requirements for CD4+ T cell clones-implications for molecular mimicry and T cell survival. Int. Immunol. 2000, 12, 375–383. [Google Scholar] [CrossRef] [PubMed]
- Rothbard, J.B. Peptides and the cellular immune response. Ann. Inst. Pasteur. 1986, 137, 518–526. [Google Scholar] [CrossRef]
- Rothbard, J.B.; Taylor, W.R. A sequence pattern common to T cell epitopes. EMBO J. 1988, 7, 93–100. [Google Scholar] [PubMed]
- Mathews, J.H.; Allan, J.E.; Roehrig, J.T.; Brubaker, J.R.; Uren, M.F.; Hunt, A.R. T-helper cell and associated antibody response to synthetic peptides of the E glycoprotein of Murray Valley encephalitis virus. J. Virol. 1991, 65, 5141–5148. [Google Scholar]
- Reddehase, M.J.; Rothbard, J.B.; Koszinowski, U.H. A pentapeptide as minimal antigenic determinant for MHC class I-restricted T lymphocytes. Nature 1989, 337, 651–653. [Google Scholar] [CrossRef] [PubMed]
- Geluk, A.; van Meijgaarden, K.E.; Janson, A.A.; Drijfhout, J.W.; Meloen, R.H.; de Vries, R.R.; Ottenhoff, T.H. Functional analysis of DR17(DR3)-restricted mycobacterial T cell epitopes reveals DR17-binding motif and enables the design of allele-specific competitor peptides. J. Immunol. 1992, 149, 2864–2871. [Google Scholar]
- Zeng, W.; Pagnon, J.; Jackson, D.C. The C-terminal pentapeptide of LHRH is a dominant B cell epitope with antigenic and biological function. Mol. Immunol. 2007, 44, 3724–3731. [Google Scholar] [CrossRef] [PubMed]
- Rothbard, J.B.; Pemberton, R.M.; Bodmer, H.C.; Askonas, B.A.; Taylor, W.R. Identification of residues necessary for clonally specific recognition of a cytotoxic T cell determinant. EMBO J. 1989, 8, 2321–2328. [Google Scholar] [PubMed]
- Rothbard, J.B.; Gefter, M.L. Interactions between immunogenic peptides and MHC proteins. Annu. Rev. Immunol. 1991, 9, 527–565. [Google Scholar] [CrossRef] [PubMed]
- Sant’Angelo, D.B.; Robinson, E.; Janeway, C.A., Jr.; Denzin, L.K. Recognition of core and flanking amino acids of MHC class II-bound peptides by the T cell receptor. Eur. J. Immunol. 2002, 32, 2510–2520. [Google Scholar] [CrossRef] [PubMed]
- Willers, J.; Capone, G.; Lucchese, A. Peptides: An arrival point in cancer vaccinology. Front. Biosci. 2012, 4, 1381–1392. [Google Scholar] [CrossRef]
- Kanduc, D. Homology, similarity, and identity in peptide epitope immunodefinition. J. Pept. Sci. 2012, 18, 487–494. [Google Scholar] [CrossRef] [PubMed]
- Kanduc, D. Pentapeptides as minimal functional units in cell biology and immunology. Curr. Protein Pept. Sci. 2013, 14, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Rowe, M.; Rowe, D.T.; Gregory, C.D.; Young, L.S.; Fartell, P.J.; Rupani, H.; Rickinson, A.B. Differences in B cell growth phenotype reflect novel patterns of Epstein-Barr virus latent gene expression in Burkitt’s lymphoma cells. EMBO J. 1987, 6, 2743–2751. [Google Scholar] [PubMed]
- Sivachandran, N.; Dawson, C.W.; Young, L.S.; Liu, F.F.; Middeldorp, J.; Frappier, L. Contributions of the Epstein-Barr virus EBNA1 protein to gastric carcinoma. J. Virol. 2012, 86, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Tsang, C.W.; Lin, X.; Gudgeon, N.H.; Taylor, G.S.; Jia, H.; Hui, E.P.; Chan, A.T.; Lin, C.K.; Rickinson, A.B. CD4+ T-cell responses to Epstein-Barr virus nuclear antigen EBNA1 in Chinese populations are highly focused on novel C-terminal domain-derived epitopes. J. Virol. 2006, 80, 8263–8266. [Google Scholar] [CrossRef] [PubMed]
- Long, H.M.; Chagoury, O.L.; Leese, A.M.; Ryan, G.B.; James, E.; Morton, L.T.; Abbott, R.J.; Sabbah, S.; Kwok, W.; Rickinson, A.B. MHC II tetramers visualize human CD4+ T cell responses to Epstein-Barr virus infection and demonstrate atypical kinetics of the nuclear antigen EBNA1 response. J. Exp. Med. 2013, 210, 933–949. [Google Scholar] [CrossRef] [PubMed]
- Long, H.M.; Haigh, T.A.; Gudgeon, N.H.; Leen, A.M.; Tsang, C.W.; Brooks, J.; Landais, E.; Houssaint, E.; Lee, S.P.; Rickinson, A.B.; et al. CD4+ T-cell responses to Epstein-Barr virus (EBV) latent-cycle antigens and the recognition of EBV-transformed lymphoblastoid cell lines. J. Virol. 2005, 79, 4896–4907. [Google Scholar] [CrossRef] [PubMed]
- Capone, G.; Fasano, C.; Lucchese, G.; Calabrò, M.; Kanduc, D.; Department of Biosciences, Biotechnologies and Biopharmaceutics, University of Bari, Bari, Italy. Unpublished data. 2014.
- Macalma, T.; Otte, J.; Hensler, M.E.; Bockholt, S.M.; Louis, H.A.; Kalff-Suske, M.; Grzeschik, K.H.; von der Ahe, D.; Beckerle, M.C. Molecular characterization of human zyxin. J. Biol. Chem. 1996, 271, 31470–31478. [Google Scholar] [CrossRef] [PubMed]
- Sheng, Y.; Hong, J.H.; Doherty, R.; Srikumar, T.; Shloush, J.; Avvakumov, G.V.; Walker, J.R.; Xue, S.; Neculai, D.; Wan, J.W.; et al. A human ubiquitin conjugating enzyme (E2)-HECT E3 ligase structure-function screen. Mol. Cell. Proteomics 2012, 11, 329–341. [Google Scholar] [CrossRef] [PubMed]
- Schütte, J.; Viallet, J.; Nau, M.; Segal, S.; Fedorko, J.; Minna, J. jun-B inhibits and c-fos stimulates the transforming and trans-activating activities of c-jun. Cell 1989, 59, 987–997. [Google Scholar] [CrossRef] [PubMed]
- Szremska, A.P.; Kenner, L.; Weisz, E.; Ott, R.G.; Passegué, E.; Artwohl, M.; Freissmuth, M.; Stoxreiter, R.; Theussl, H.C.; Parzer, S.B.; et al. JunB inhibits proliferation and transformation in B-lymphoid cells. Blood 2003, 102, 4159–4165. [Google Scholar] [CrossRef] [PubMed]
- Davis-Smyth, T.; Duncan, R.C.; Zheng, T.; Michelotti, G.; Levens, D. The far upstream element-binding proteins comprise an ancient family of single-strand DNA-binding transactivators. J. Biol. Chem. 1996, 271, 31679–31687. [Google Scholar] [CrossRef] [PubMed]
- Chari, A.; Golas, M.M.; Klingenhäger, M.; Neuenkirchen, N.; Sander, B.; Englbrecht, C.; Sickmann, A.; Stark, H.; Fischer, U. An assembly chaperone collaborates with the SMNcomplex to generate spliceosomal SnRNPs. Cell 2008, 135, 497–509. [Google Scholar] [CrossRef] [PubMed]
- Rivkin, E.; Vella, M.J.; Lahita, R.G. A heterogeneous immune response to an SmD-like epitope by SLE patients. J. Autoimmun. 1994, 7, 119–132. [Google Scholar] [CrossRef] [PubMed]
- Levitskaya, J.; Sharipo, A.; Leonchiks, A.; Ciechanover, A.; Masucci, M.G. Inhibition of ubiquitin/proteasome-dependent protein degradation by the Gly-Ala repeat domain of the Epstein-Barr virus nuclear antigen 1. Proc. Natl. Acad. Sci. USA 1997, 94, 12616–12621. [Google Scholar] [CrossRef] [PubMed]
- Levitskaya, J.; Coram, M.; Levitsky, V.; Imreh, S.; Steigerwald-Mullen, P.M.; Klein, G.; Kurilla, M.G.; Masucci, M.G. Inhibition of antigen processing by the internal repeat region of the Epstein-Barr virus nuclear antigen-1. Nature 1995, 375, 685–688. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.M.; Foong, Y.T.; Sam, C.K.; Prasad, U.; Dillner, J. Epstein-Barr virus nuclear antigen 1 linear epitopes that are reactive with immunoglobulin A (IgA) or IgG in sera from nasopharyngeal carcinoma patients or from healthy donors. J. Clin. Microbiol. 1991, 29, 2180–2186. [Google Scholar] [PubMed]
- Leen, A.; Meij, P.; Redchenko, I.; Middeldorp, J.; Bloemena, E.; Rickinson, A.; Blake, N. Differential immunogenicity of Epstein-Barr virus latent-cycle proteins for human CD4+ T-helper 1 responses. J. Virol. 2001, 75, 8649–8659. [Google Scholar] [CrossRef] [PubMed]
- Taylor, G.S.; Haigh, T.A.; Gudgeon, N.H.; Phelps, R.J.; Lee, S.P.; Steven, N.M.; Rickinson, A.B. Dual stimulation of Epstein-Barr Virus (EBV)-specific CD4+- and CD8+-T-cell responses by a chimeric antigen construct: Potential therapeutic vaccine for EBV-positive nasopharyngeal carcinoma. J. Virol. 2004, 78, 768–778. [Google Scholar] [CrossRef] [PubMed]
- Woodberry, T.; Suscovich, T.J.; Henry, L.M.; Davis, J.K.; Frahm, N.; Walker, B.D.; Scadden, D.T.; Wang, F.; Brander, C. Differential targeting and shifts in the immunodominance of Epstein-Barr virus—Specific CD8 and CD4 T cell responses during acute and persistent infection. J. Infect. Dis. 2005, 192, 1513–1524. [Google Scholar] [CrossRef] [PubMed]
- Krüger, S.; Schroers, R.; Rooney, C.M.; Gahn, B.; Chen, S.Y. Identification of a naturally processed HLA-DR-restricted T-helper epitope in Epstein-Barr virus nuclear antigen type 1. J. Immunother. 2003, 26, 212–221. [Google Scholar] [CrossRef] [PubMed]
- Petersen, J.; Rhodes, G.; Patrick, K.; Roudier, J.; Vaughan, J.H. Human T cell responses to the Epstein-Barr nuclear antigen-1 (EBNA-1) as evaluated by synthetic peptides. Cell Immunol. 1989, 123, 325–333. [Google Scholar] [CrossRef] [PubMed]
- Petersen, J.; Rhodes, G.; Roudier, J.; Vaughan, J.H. Altered immune response to glycine-rich sequences of Epstein-Barr nuclear antigen-1 in patients with rheumatoid arthritis and systemic lupus erythematosus. Arthritis Rheum. 1990, 33, 993–1000. [Google Scholar] [CrossRef] [PubMed]
- Taylor, G.S.; Long, H.M.; Haigh, T.A.; Larsen, M.; Brooks, J.; Rickinson, A.B. A role for intercellular antigen transfer in the recognition of EBV-transformed B cell lines by EBV nuclear antigen-specific CD4+ T cells. J. Immunol. 2006, 177, 3746–3756. [Google Scholar] [CrossRef] [PubMed]
- Depil, S.; Moralès, O.; Castelli, F.A.; Delhem, N.; François, V.; Georges, B.; Dufossé, F.; Morschhauser, F.; Hammer, J.; Maillère, B.; et al. Determination of a HLA II promiscuous peptide cocktail as potential vaccine against EBV latency II malignancies. J. Immunother. 2007, 30, 215–226. [Google Scholar] [PubMed]
- Moralès, O.; Depil, S.; Mrizak, D.; Martin, N.; Ndour, P.A.; Dufosse, F.; Miroux, C.; Coll, J.; de Launoit, Y.; Auriault, C.; et al. EBV Latency II-derived peptides induce a specific CD4+ cytotoxic T-cell activity and not a CD4+ regulatory T-cell response. J. Immunother. 2012, 35, 254–266. [Google Scholar] [PubMed]
- Voo, K.S.; Peng, G.; Guo, Z.; Fu, T.; Li, Y.; Frappier, L.; Wang, R.F. Functional characterization of EBV-encoded nuclear antigen 1-specific CD4+ helper and regulatory T cells elicited by in vitro peptide stimulation. Cancer Res. 2005, 65, 1577–1586. [Google Scholar] [PubMed]
- Smith, C.; Cooper, L.; Burgess, M.; Rist, M.; Webb, N.; Lambley, E.; Tellam, J.; Marlton, P.; Seymour, J.F.; Gandhi, M.; et al. Functional reversion of antigen-specific CD8+ T cells from patients with Hodgkin lymphoma following in vitro stimulation with recombinant polyepitope. J. Immunol. 2006, 177, 4897–4906. [Google Scholar]
- Demachi-Okamura, A.; Ito, Y.; Akatsuka, Y.; Tsujimura, K.; Morishima, Y.; Takahashi, T.; Kuzushima, K. Epstein-Barr virus nuclear antigen 1-specific CD4+ T cells directly kill Epstein-Barr virus-carrying natural killer and T cells. Cancer Sci. 2008, 99, 1633–1642. [Google Scholar] [PubMed]
- Sundström, P.; Nyström, M.; Ruuth, K.; Lundgren, E. Antibodies to specific EBNA-1 domains and HLA DRB1*1501 interact as risk factors for multiple sclerosis. J. Neuroimmunol. 2009, 215, 102–107. [Google Scholar] [PubMed]
- Gurer, C.; Strowig, T.; Brilot, F.; Pack, M.; Trumpfheller, C.; Arrey, F.; Park, C.G.; Steinman, R.M.; Münz, C. Targeting the nuclear antigen 1 of Epstein-Barr virus to the human endocytic receptor DEC-205 stimulates protective T-cell responses. Blood 2008, 112, 1231–1239. [Google Scholar] [PubMed]
- Rooney, C.M.; Smith, C.A.; Ng, C.Y.; Loftin, S.; Li, C.; Krance, R.A.; Brenner, M.K.; Heslop, H.E. Use of gene-modified virus-specific T lymphocytes to control Epstein-Barr-virus-related lymphoproliferation. Lancet 1995, 345, 9–13. [Google Scholar] [PubMed]
- Gu, S.Y.; Huang, T.M.; Ruan, L.; Miao, Y.H.; Lu, H.; Chu, C.M.; Motz, M.; Wolf, H. First EBV vaccine trial in humans using recombinant vaccinia virus expressing the major membrane antigen. Dev. Biol. Stand. 1995, 84, 171–177. [Google Scholar] [PubMed]
- Moutschen, M.; Léonard, P.; Sokal, E.M.; Smets, F.; Haumont, M.; Mazzu, P.; Bollen, A.; Denamur, F.; Peeters, P.; Dubin, G.; et al. Phase I/II studies to evaluate safety and immunogenicity of a recombinant gp350 Epstein-Barr virus vaccine in healthy adults. Vaccine 2007, 25, 4697–4705. [Google Scholar] [CrossRef] [PubMed]
- Sokal, E.M.; Hoppenbrouwers, K.; Vandermeulen, C.; Moutschen, M.; Léonard, P.; Moreels, A.; Haumont, M.; Bollen, A.; Smets, F.; Denis, M. Recombinant gp350 vaccine for infectious mononucleosis: A phase 2, randomized, double-blind, placebo-controlled trial to evaluate the safety, immunogenicity, and efficacy of an Epstein-Barr virus vaccine in healthy young adults. J. Infect. Dis. 2007, 196, 1749–1753. [Google Scholar] [CrossRef] [PubMed]
- Cui, X.; Cao, Z.; Sen, G.; Chattopadhyay, G.; Fuller, D.H.; Fuller, J.T.; Snapper, D.M.; Snow, A.L.; Mond, J.J.; Snapper, C.M. A novel tetrameric gp350 1–470 as a potential Epstein-Barr virus vaccine. Vaccine 2013, 31, 3039–3045. [Google Scholar] [CrossRef] [PubMed]
- Ruiss, R.; Jochum, S.; Wanner, G.; Reisbach, G.; Hammerschmidt, W.; Zeidler, R. A virus-like particle-based Epstein-Barr virus vaccine. J. Virol. 2011, 85, 13105–13113. [Google Scholar] [CrossRef]
- Antsiferova, O.; Müller, A.; Rämer, P.C.; Chijioke, O.; Chatterjee, B.; Raykova, A.; Planas, R.; Sospedra, M.; Shumilov, A.; Tsai, M.H.; et al. Adoptive transfer of EBV specific CD8+ T sell clones can transiently control EBV infection in humanized mice. PLOS Pathog. 2014, 10, e1004333. [Google Scholar] [CrossRef] [PubMed]
- Lutzky, V.P.; Crooks, P.; Morrison, L.; Stevens, N.; Davis, J.E.; Corban, M.; Hall, D.; Panizza, B.; Coman, W.B.; Coman, S.; et al. Cytotoxic T cell adoptive immunotherapy as a treatment for nasopharyngeal carcinoma. Clin. Vaccine Immunol. 2014, 21, 256–259. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.K.; Perrine, S.P.; Faller, D.V. Advances in virus-directed therapeutics against Epstein-Barr Virus-associated malignancies. Adv. Virol. 2012. [Google Scholar] [CrossRef]
- Lünemann, J.D.; Tintoré, M.; Messmer, B.; Strowig, T.; Rovira, A.; Perkal, H.; Caballero, E.; Münz, C.; Montalban, X.; Comabella, M. Elevated Epstein-Barr virus-encoded nuclear antigen-1 immune responses predict conversion to multiple sclerosis. Ann. Neurol. 2010, 67, 159–169. [Google Scholar] [CrossRef] [PubMed]
- Angelini, D.F.; Serafini, B.; Piras, E.; Severa, M.; Coccia, E.M.; Rosicarelli, B.; Ruggieri, S.; Gasperini, C.; Buttari, F.; Centonze, D.; et al. Increased CD8+ T cell response to Epstein-Barr virus lytic antigens in the active phase of multiple sclerosis. PLOS Pathog. 2013, 9, e1003220. [Google Scholar] [CrossRef] [PubMed]
- Kannangai, R.; Sachithanandham, J.; Kandathil, A.J.; Ebenezer, D.L.; Danda, D.; Vasuki, Z.; Thomas, N.; Vasan, S.K.; Sridharan, G. Immune responses to Epstein-Barr virus in individuals with systemic and organ specific autoimmune disorders. Indian. J. Med. Microbiol. 2010, 28, 120–123. [Google Scholar] [CrossRef] [PubMed]
- Mahler, M.; Fritzler, M.J.; Blüthner, M. Identification of a SmD3 epitope with a single symmetrical dimethylation of an arginine residue as a specific target of a subpopulation of anti-Sm antibodies. Arthritis Res. Ther. 2005, 7, R19–R29. [Google Scholar] [CrossRef] [PubMed]
- Karin, N.; Binah, O.; Grabie, N.; Mitchell, D.J.; Felzen, B.; Solomon, M.D.; Conlon, P.; Gaur, A.; Ling, N.; Steinman, L. Short peptide-based tolerogens without self-antigenic or pathogenic activity reverse autoimmune disease. J. Immunol. 1998, 160, 5188–5194. [Google Scholar] [PubMed]
- Tselios, T.; Daliani, I.; Probert, L.; Deraos, S.; Matsoukas, E.; Roy, S.; Pires, J.; Moore, G.; Matsoukas, J. Treatment of experimental allergic encephalomyelitis (EAE) induced by guinea pig myelin basic protein epitope 72–85 with a human MBP(87–99) analogue and effects of cyclic peptides. Bioorg. Med. Chem. 2000, 8, 1903–1909. [Google Scholar] [CrossRef] [PubMed]
- Lucchese, A.; Mittelman, A.; Lin, M.S.; Kanduc, D.; Sinha, A.A. Epitope definition by proteomic similarity analysis: Identification of the linear determinant of the anti-Dsg3 MAb 5H10. J. Transl Med. 2004. [Google Scholar] [CrossRef]
- Lucchese, A.; Mittelman, A.; Tessitore, L.; Serpico, R.; Sinha, A.A.; Kanduc, D. Proteomic definition of a desmoglein linear determinant common to Pemphigus vulgaris and Pemphigus foliaceous. J. Transl. Med. 2006. [Google Scholar] [CrossRef]
- Salato, V.K.; Hacker-Foegen, M.K.; Lazarova, Z.; Fairley, J.A.; Lin, M.S. Role of intramolecular epitope spreading in pemphigus vulgaris. Clin Immunol. 2005, 116, 54–64. [Google Scholar] [CrossRef] [PubMed]
- Angelini, G.; Bonamonte, D.; Lin, M.S.; Lucchese, A.; Mittelman, A.; Serpico, R.; Simone, S.; Sinha, A.A.; Kanduc, D. Characterization of polyclonal antibodies raised against a linear peptide determinant of desmoglein-3. J. Exp. Ther. Oncol. 2005, 5, 1–7. [Google Scholar] [PubMed]
- Angelini, G.; Bonamonte, D.; Lucchese, A.; Favia, G.; Serpico, R.; Mittelman, A.; Simone, S.; Sinha, A.A.; Kanduc, D. Preliminary data on Pemphigus vulgaris treatment by a proteomics-defined peptide: A case report. J. Transl. Med. 2006. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Capone, G.; Fasano, C.; Lucchese, G.; Calabrò, M.; Kanduc, D. EBV-Associated Cancer and Autoimmunity: Searching for Therapies. Vaccines 2015, 3, 74-89. https://doi.org/10.3390/vaccines3010074
Capone G, Fasano C, Lucchese G, Calabrò M, Kanduc D. EBV-Associated Cancer and Autoimmunity: Searching for Therapies. Vaccines. 2015; 3(1):74-89. https://doi.org/10.3390/vaccines3010074
Chicago/Turabian StyleCapone, Giovanni, Candida Fasano, Guglielmo Lucchese, Michele Calabrò, and Darja Kanduc. 2015. "EBV-Associated Cancer and Autoimmunity: Searching for Therapies" Vaccines 3, no. 1: 74-89. https://doi.org/10.3390/vaccines3010074
APA StyleCapone, G., Fasano, C., Lucchese, G., Calabrò, M., & Kanduc, D. (2015). EBV-Associated Cancer and Autoimmunity: Searching for Therapies. Vaccines, 3(1), 74-89. https://doi.org/10.3390/vaccines3010074





