BNT162b2 COVID-19 Vaccines in Children, Adolescents and Young Adults with Cancer—A 1-Year Follow-Up
Abstract
:1. Introduction
2. Materials and Methods
3. Results
3.1. Global Population
3.2. Adverse Events
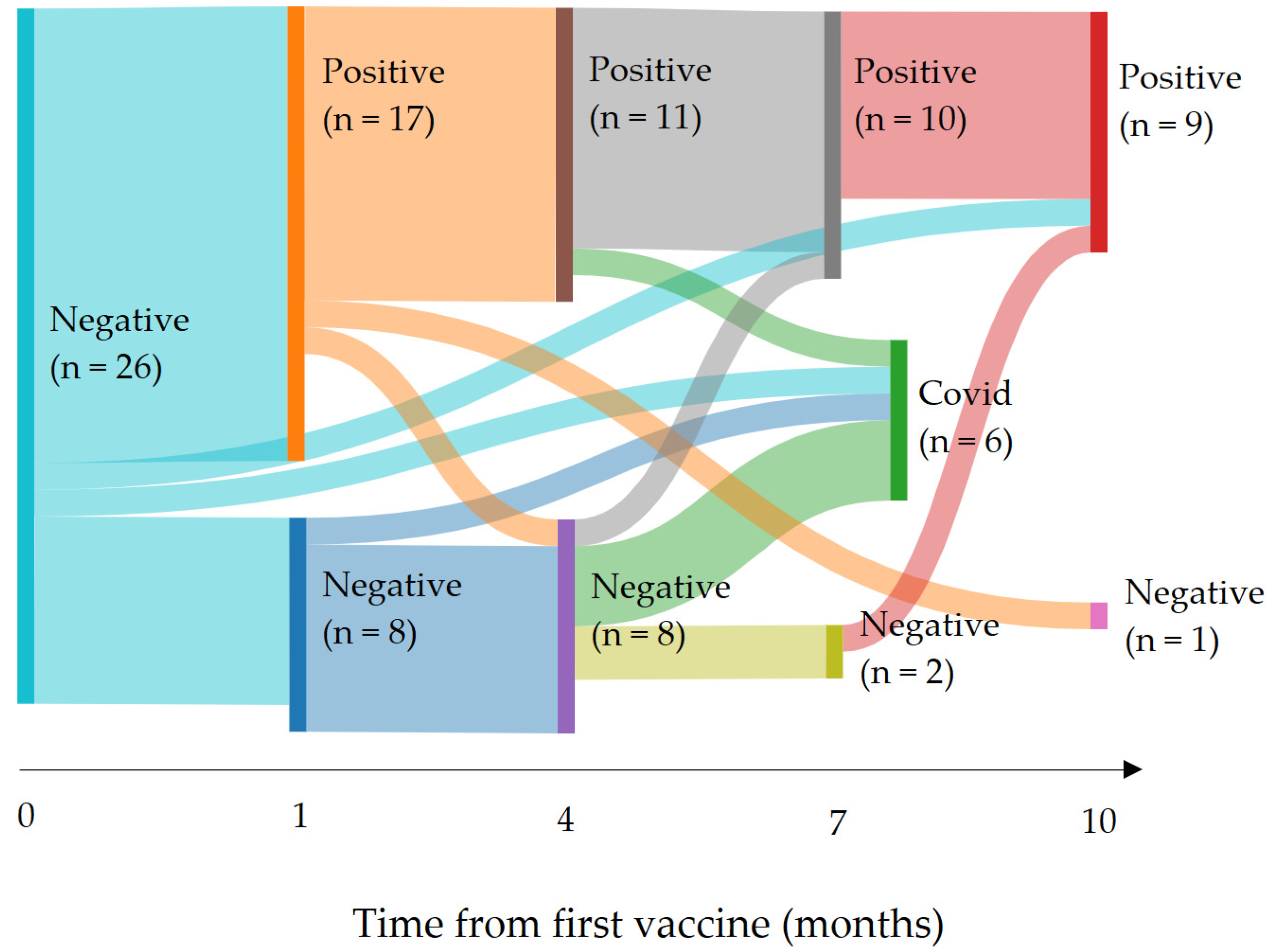
3.3. COVID-19 Infections
3.4. Immunity
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cheruiyot, I.; Kipkorir, V.; Ngure, B.; Misiani, M.; Munguti, J. Cancer is associated with coronavirus disease (COVID-19) severity and mortality: A pooled analysis. Am. J. Emerg. Med. 2021, 45, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Borrelli, M.; Corcione, A.; Castellano, F.; Fiori Nastro, F.; Santamaria, F. Coronavirus Disease 2019 in Children. Front. Pediatr. 2021, 9, 668484. [Google Scholar] [CrossRef] [PubMed]
- Mehta, N.S.; Mytton, O.T.; Mulli, E.W.S.; Fowler, T.A.; Falconer, C.L.; Murphy, O.B.; Langenberg, C.; Jayatunga, W.J.P.; Eddy, D.H.; Nguyen-Van-Tam, J.S. SARS-CoV-2 (COVID-19): What Do We Know About Children? A Systematic Review. Clin. Infect. Dis. 2020, 71, 2469–2479. [Google Scholar] [CrossRef] [PubMed]
- Yasuhara, J.; Kuno, T.; Takagi, H.; Sumitomo, N. Clinical characteristics of COVID-19 in children: A systematic review. Pediatr. Pulmonol. 2020, 55, 2565–2575. [Google Scholar] [CrossRef]
- André, N.; Rouger-Gaudichon, J.; Brethon, B.; Phulpin, A.; Thébault, É.; Pertuisel, S.; Gandemer, V. COVID-19 in pediatric oncology from French pediatric oncology and hematology centers: High risk of severe forms? Pediatr. Blood Cancer 2020, 67, e28392. [Google Scholar] [CrossRef]
- Mukkada, S.; Bhakta, N.; Chantada, G.L.; Chen, Y.; Vedaraju, Y.; Faughnan, L.; Homsi, M.R.; Muniz-Talavera, H.; Ranadive, R.; Metzger, M.; et al. Global characteristics and outcomes of SARS-CoV-2 infection in children and adolescents with cancer (GRCCC): A cohort study. Lancet Oncol. 2021, 22, 1416–1426. [Google Scholar] [CrossRef]
- Burki, T. Moving away from zero COVID in China. Lancet Respir. Med. 2022, 11, 132. [Google Scholar] [CrossRef]
- Waldhorn, I.; Holland, R.; Goshen-Lago, T.; Shirman, Y.; Szwarcwort-Cohen, M.; Reiner-Benaim, A.; Shachor-Meyouhas, Y.; Hussein, K.; Fahoum, L.; Peer, A.; et al. Six-Month Efficacy and Toxicity Profile of BNT162b2 Vaccine in Cancer Patients with Solid Tumors. Cancer Discov. 2021, 11, 2430–2435. [Google Scholar] [CrossRef]
- Sekkate, S.; Stoeklé, H.-C.; Mabro, M.; Billard, D.; Kennel, T.; Vasse, M.; Hervé, C.; Beuzeboc, P. Letter comments on: Efficacy and safety of BNT162b2 vaccination in solid cancer patients receiving anti-cancer therapy—A single-centre prospective study. Eur. J. Cancer 2022, 160, 282–284. [Google Scholar] [CrossRef]
- Mariatulqabtiah, A.R.; Buttigieg, K.R. COVID-19 vaccinations for children. Lancet Infect. Dis. 2022, 22, 1255–1256. [Google Scholar] [CrossRef]
- Revon-Riviere, G.; Ninove, L.; Min, V.; Rome, A.; Coze, C.; Verschuur, A.; de Lamballerie, X.; André, N. The BNT162b2 mRNA COVID-19 vaccine in adolescents and young adults with cancer: A monocentric experience. Eur. J. Cancer 2021, 154, 30–34. [Google Scholar] [CrossRef] [PubMed]
- Lehrnbecher, T.; Sack, U.; Speckmann, C.; Groll, A.H.; Boldt, A.; Siebald, B.; Hettmer, S.; Demmerath, E.M.; Schenk, B.; Ciesek, S.; et al. Longitudinal Immune Response to 3 Doses of Messenger RNA Vaccine Against Coronavirus Disease (COVID-19) in Pediatric Patients Receiving Chemotherapy for Cancer. Clin. Infect. Dis. 2022, 76, e510–e513. [Google Scholar] [CrossRef] [PubMed]
- Vaccination Dans le Cadre de la COVID-19. Available online: https://www.has-sante.fr/jcms/p_3178533/fr/vaccination-dans-le-cadre-de-la-covid-19 (accessed on 6 May 2023).
- Polack, F.P.; Thomas, S.J.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Pérez Marc, G.; Moreira, E.D.; Zerbini, C.; et al. Safety and Efficacy of the BNT162b2 mRNA COVID-19 Vaccine. N. Engl. J. Med. 2020, 383, 2603–2615. [Google Scholar] [CrossRef] [PubMed]
- Feng, S.; Phillips, D.J.; White, T.; Sayal, H.; Aley, P.K.; Bibi, S.; Dold, C.; Fuskova, M.; Gilbert, S.C.; Hirsch, I.; et al. Correlates of protection against symptomatic and asymptomatic SARS-CoV-2 infection. Nat. Med. 2021, 27, 2032–2040. [Google Scholar] [CrossRef] [PubMed]
- Gallian, P.; Pastorino, B.; Morel, P.; Chiaroni, J.; Ninove, L.; de Lamballerie, X. Lower prevalence of antibodies neutralizing SARS-CoV-2 in group O French blood donors. Antivir. Res. 2020, 181, 104880. [Google Scholar] [CrossRef] [PubMed]
- Kotecha, R.S. COVID-19 vaccination for children with cancer. Pediatr. Blood Cancer 2022, 69, e29340. [Google Scholar] [CrossRef] [PubMed]
- Walter, E.B.; Talaat, K.R.; Sabharwal, C.; Gurtman, A.; Lockhart, S.; Paulsen, G.C.; Barnett, E.D.; Muñoz, F.M.; Maldonado, Y.; Pahud, B.A.; et al. Evaluation of the BNT162b2 COVID-19 Vaccine in Children 5 to 11 Years of Age. N. Engl. J. Med. 2022, 386, 35–46. [Google Scholar] [CrossRef] [PubMed]
- Shmueli, E.S.; Itay, A.; Margalit, O.; Berger, R.; Halperin, S.; Jurkowicz, M.; Levin, E.G.; Levy, I.; Olmer, L.; Regev-Yochay, G.; et al. Efficacy and safety of BNT162b2 vaccination in patients with solid cancer receiving anticancer therapy—A single centre prospective study. Eur. J. Cancer 2021, 157, 124–131. [Google Scholar] [CrossRef]
- Della Pia, A.; Kim, G.Y.; Ip, A.; Ahn, J.; Liu, Y.; Kats, S.; Koropsak, M.; Lukasik, B.; Contractor, A.; Amin, K.; et al. Anti-spike antibody response to the COVID vaccine in lymphoma patients. PLoS ONE 2022, 17, e0266584. [Google Scholar] [CrossRef]
- Wu, Z.; McGoogan, J.M. Characteristics of and Important Lessons From the Coronavirus Disease 2019 (COVID-19) Outbreak in China: Summary of a Report of 72 314 Cases From the Chinese Center for Disease Control and Prevention. JAMA 2020, 323, 1239. [Google Scholar] [CrossRef]
- Gazit, S.; Shlezinger, R.; Perez, G.; Lotan, R.; Peretz, A.; Ben-Tov, A.; Herzel, E.; Alapi, H.; Cohen, D.; Muhsen, K.; et al. Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) Naturally Acquired Immunity versus Vaccine-induced Immunity, Reinfections versus Breakthrough Infections: A Retrospective Cohort Study. Clin. Infect. Dis. 2022, 75, e545–e551. [Google Scholar] [CrossRef] [PubMed]
- Lewis, N.; Chambers, L.C.; Chu, H.T.; Fortnam, T.; De Vito, R.; Gargano, L.M.; Chan, P.A.; McDonald, J.; Hogan, J.W. Effectiveness Associated With Vaccination After COVID-19 Recovery in Preventing Reinfection. JAMA Netw. Open 2022, 5, e2223917. [Google Scholar] [CrossRef] [PubMed]
- Hall, V.; Foulkes, S.; Insalata, F.; Kirwan, P.; Saei, A.; Atti, A.; Wellington, E.; Khawam, J.; Munro, K.; Cole, M.; et al. Protection against SARS-CoV-2 after COVID-19 Vaccination and Previous Infection. N. Engl. J. Med. 2022, 386, 1207–1220. [Google Scholar] [CrossRef] [PubMed]
- Hause, A.M.; Baggs, J.; Marquez, P.; Myers, T.R.; Gee, J.; Su, J.R.; Zhang, B.; Thompson, D.; Shimabukuro, T.T.; Shay, D.K. COVID-19 Vaccine Safety in Children Aged 5–11 Years—United States, November 3–December 19, 2021. MMWR Morb. Mortal. Wkly. Rep. 2021, 70, 1755–1760. [Google Scholar] [CrossRef] [PubMed]
- Al Khames Aga, Q.A.; Alkhaffaf, W.H.; Hatem, T.H.; Nassir, K.F.; Batineh, Y.; Dahham, A.T.; Shaban, D.; Al Khames Aga, L.A.; Agha, M.Y.R.; Traqchi, M. Safety of COVID-19 vaccines. J. Med. Virol. 2021, 93, 6588–6594. [Google Scholar] [CrossRef] [PubMed]
- Novembre, E.; Tosca, M.; Caffarelli, C.; Calvani, M.; Cardinale, F.; Castagnoli, R.; Chiappini, E.; Cravidi, C.; Del Giudice, M.M.; Duse, M.; et al. Management of BNT162b2 mRNA COVID-19 vaccine in children aged 5–11 years with allergies, asthma, and immunodeficiency: Consensus of the Italian Society of Pediatric Allergy and Immunology (SIAIP). Ital. J. Pediatr. 2022, 48, 76. [Google Scholar] [CrossRef] [PubMed]
- Maayan, H.; Kirgner, I.; Gutwein, O.; Herzog-Tzarfati, K.; Rahimi-Levene, N.; Koren-Michowitz, M.; Blickstein, D. Acquired thrombotic thrombocytopenic purpura: A rare disease associated with BNT162b2 vaccine. J. Thromb. Haemost 2021, 19, 2314–2317. [Google Scholar] [CrossRef] [PubMed]
- Dix, C.; McFadyen, J.; Huang, A.; Chunilal, S.; Chen, V.; Tran, H. Understanding vaccine-induced thrombotic thrombocytopenia (VITT). Intern. Med. J. 2022, 52, 717–723. [Google Scholar] [CrossRef]
- Rizk, J.G.; Gupta, A.; Sardar, P.; Henry, B.M.; Lewin, J.C.; Lippi, G.; Lavie, C.J. Clinical Characteristics and Pharmacological Management of COVID-19 Vaccine–Induced Immune Thrombotic Thrombocytopenia With Cerebral Venous Sinus Thrombosis: A Review. JAMA Cardiol. 2021, 6, 1451. [Google Scholar] [CrossRef]
- Aleem, A.; Nadeem, A.J. Coronavirus (COVID-19) Vaccine-Induced Immune Thrombotic Thrombocytopenia (VITT). In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. Available online: http://www.ncbi.nlm.nih.gov/books/NBK570605/ (accessed on 22 January 2023).
- Gerber, J.S.; Offit, P.A. COVID-19 vaccines for children. Science 2021, 374, 913. [Google Scholar] [CrossRef]
- van Kampen, K.; Laski, J.; Herman, G.; Chan, T.M. Investigating COVID-19 Vaccine Communication and Misinformation on TikTok: Cross-sectional Study. JMIR Infodemiol. 2022, 2, e38316. [Google Scholar] [CrossRef] [PubMed]
- Thorpe, A.; Fagerlin, A.; Butler, J.; Stevens, V.; Drews, F.A.; Shoemaker, H.; Riddoch, M.S.; Scherer, L.D. Communicating about COVID-19 vaccine development and safety. PLoS ONE 2022, 17, e0272426. [Google Scholar] [CrossRef] [PubMed]
- Kreps, S.; Kriner, D.L. Communication about vaccine efficacy and COVID-19 vaccine choice: Evidence from a survey experiment in the United States. PLoS ONE 2022, 17, e0265011. [Google Scholar] [CrossRef]
- Mahmud, S.; Mohsin, M.; Hossain, S.; Islam, M.M.; Muyeed, A. The acceptance of COVID-19 vaccine at early stage of development and approval: A global systematic review and meta-analysis. Heliyon 2022, 8, e10728. [Google Scholar] [CrossRef] [PubMed]
- Fendler, A.; de Vries, E.G.E.; GeurtsvanKessel, C.H.; Haanen, J.B.; Wörmann, B.; Turajlic, S.; von Lilienfeld-Toal, M. COVID-19 vaccines in patients with cancer: Immunogenicity, efficacy and safety. Nat. Rev. Clin. Oncol. 2022, 19, 385–401. [Google Scholar] [CrossRef] [PubMed]

| General Population N = 38 | |||
|---|---|---|---|
| N or Median [Q1; Q3] | Percentage | ||
| Sex | |||
| Female | 14 | 37% | |
| Male | 24 | 63% | |
| Number of administered vaccine doses | N | % | |
| 1 | 4 | N/A | |
| 2 | 17 | N/A | |
| 3 | 17 | N/A | |
| Age at the first vaccination | 16.0 [15.0; 18.0] | ||
| Death | |||
| Alive | 34 | 90% | |
| Deceased | 4 | 11% | |
| Type of cancer | |||
| Brain tumor | 9 | 24% | |
| Lymphoma | 7 | 18% | |
| Bone sarcoma | 6 | 16% | |
| Soft tissue sarcoma | 10 | 26% | |
| Other types | 6 | 16% | |
| Metastatic status | |||
| Localized | 24 | 63% | |
| Metastatic | 14 | 37% | |
| Types of treatment | |||
| Systemic | 37 | 97% | |
| Conventional chemotherapy | 36 | 95% | |
| High dose chemotherapy | 5 | 14% | |
| Immunotherapy | 11 | 29% | |
| Local | 29 | 76% | |
| Surgery | 23 | 61% | |
| Radiotherapy | 18 | 47% | |
| Radiofrequency | 1 | 3% | |
| Number of lines | |||
| 1 | 22 | N/A | |
| 2 | 9 | N/A | |
| >3 | 7 | N/A | |
| Treatment phase at the time of vaccination | |||
| Induction | 1 | N/A | |
| Adjuvant | 6 | N/A | |
| Maintenance | 16 | N/A | |
| Palliative chemotherapy | 6 | N/A | |
| Surveillance | 9 | N/A | |
| Vaccine 1 N = 35 | Vaccine 2 N = 32 | Vaccine 3 N = 16 | ||
|---|---|---|---|---|
| None | 13 (37%) | 14 (44%) | 5 (31%) | |
| Local pain | Grade 1 | 19 (54%) | 13 (41%) | 7 (23%) |
| Diffuse pain | ||||
| Grade 1 | 2 (6%) | 2 (6%) | 0 | |
| Grade 2 | 0 | 0 | 1 (6%) | |
| Fatigue | ||||
| Grade 1 | 5 (14%) | 5 (16%) | 3 (19%) | |
| Grade 2 | 1 (3%) | 2 (6%) | 2 (13%) | |
| Grade 3 | 1 (3%) | 3 (9%) | 3 (19%) | |
| Fever | Grade 1 | 0 | 1 (3%) | 1 (6%) |
| Vomiting | Grade 2 | 0 | 1 (3%) | 0 |
| Purpura | 0 | 0 | 1 (6%) | |
| Dizziness | 0 | 0 | 1 (6%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Donze, C.; Min, V.; Ninove, L.; de Lamballerie, X.; Revon Rivière, G.; Verschuur, A.; Saultier, P.; André, N. BNT162b2 COVID-19 Vaccines in Children, Adolescents and Young Adults with Cancer—A 1-Year Follow-Up. Vaccines 2023, 11, 989. https://doi.org/10.3390/vaccines11050989
Donze C, Min V, Ninove L, de Lamballerie X, Revon Rivière G, Verschuur A, Saultier P, André N. BNT162b2 COVID-19 Vaccines in Children, Adolescents and Young Adults with Cancer—A 1-Year Follow-Up. Vaccines. 2023; 11(5):989. https://doi.org/10.3390/vaccines11050989
Chicago/Turabian StyleDonze, Caroline, Victoria Min, Laetitia Ninove, Xavier de Lamballerie, Gabriel Revon Rivière, Arnauld Verschuur, Paul Saultier, and Nicolas André. 2023. "BNT162b2 COVID-19 Vaccines in Children, Adolescents and Young Adults with Cancer—A 1-Year Follow-Up" Vaccines 11, no. 5: 989. https://doi.org/10.3390/vaccines11050989
APA StyleDonze, C., Min, V., Ninove, L., de Lamballerie, X., Revon Rivière, G., Verschuur, A., Saultier, P., & André, N. (2023). BNT162b2 COVID-19 Vaccines in Children, Adolescents and Young Adults with Cancer—A 1-Year Follow-Up. Vaccines, 11(5), 989. https://doi.org/10.3390/vaccines11050989






