1. Introduction
Cervical cancer is a major cause of cancer death in women worldwide. Consistent evidence has indicated that human papillomavirus infection (HPV) is the main factor that induces cervical lesions and cervical cancer [
1]. It is worth noting that while the detection rate of HPV in cervical cancer tissue is as high as 99%, the HPV genotypes 16 and 18 are 73.8% and 16.4% in Southwest of China, respectively [
2,
3]. Traditional treatment methods such as surgery, radiotherapy, chemotherapy, etc., are still the preferred treatment at present for cervical cancer. However, the effect is not satisfactory for advanced stage, metastasis, and recurrent cervical cancer [
4,
5]. The HPV viral oncoproteins E6 and E7 are considered tumor-specific targets for immunotherapy, making them as the most effective vaccine for cervical cancer treatment.
With the development of molecular cell biology and immunology, basic study and clinical trials have been carried out to evaluate the efficiency and safety of immunotherapy in cervical cancer. In 2018, the National Comprehensive Cancer Network (NCCN) recommended Pembrolizumab, one of the immune checkpoint inhibitors targeting PD-1, as a new treatment for unsatisfactory advanced, metastatic, and recurrent cervical cancer [
6]. However, the inhibitor is effective only for patients with high expression of PD-L1, while has no benefit for patients who do not express or express low level of PD-L1 [
7]. Therefore, our attention was back on a tumor vaccine to stimulate HPV antigen specific T cells for the treatment of cervical cancer. Ferrara et al. reported a clinical trial for autologous DCs stimulated by recombinant HPV16E7 or HPV18E7 protein in the treatment of patients with advanced, metastatic, and recurrent cervical cancer, and found that the recombinant HPV E7 induced antitumor T cell responses in a portion of late stage cervical cancer patients [
8].
Due to their outstanding antigen presenting ability, DCs are the key mediator of T cell immune response. They capture and process antigens in the context of major histocompatibility complex (MHC) to naïve T cells, and trigger a specific adaptive immune response. However, the ex vivo DCs-based vaccine is difficult to standardize, therefore, in vivo induction of DCs with HPV antigen as vaccine for cervical cancer has great potential for clinical application. It was reported that the spliced exon encoding the type III repeat extra domain A (EDA) from fibronectin, which is produced in response to tissue injury and works as a damage-associated molecular pattern molecule [
9], is able to target antigens to DCs while inducing maturation through TLR4 ligation [
10,
11,
12,
13]. Moreover, scientists have amplified mouse origin EDA and constructed recombinant fusion protein EDA-E7 (HPV16E7), then evaluated the immune response in mouse condition and found that EDA-E7 could efficiently induce specific immune rejection of HPV16E7 infected TC-1 tumors [
14]. In the present work, we cloned human origin EDA and successfully constructed and purified EDA-HPV16E7 (EDA-E7), a fusion protein containing human EDA and part of HPV16E7. Further, EDA-E7 and Toll-like receptor (TLR) agonist were applied to induce human DCs cell maturation in vitro. Specific activation of human T cells by EDA-E7 matured DCs, and T cells mediated cell lysis of HPV16E7 infected cervical cancer cell were observed. Our research will fill the gap between bench study and clinical application in human for the treatment of HPV infected cervical cancer using EDA-E7 vaccine.
2. Materials and Methods
2.1. Cell Culture
SiHa, 293(HEK-293) and THP-1 were from Shanghai Cell Collection (Shanghai, China). SiHa and 293 were cultured in DMEM (Gibco, Beijing, China) and supplemented with 10% fetal bovine serum (FBS) and penicillin/streptomycin (Gibco, Beijing, China). THP-1 were cultured in RPMI 1640 (Gibco, Beijing, China) supplemented with 10% fetal bovine serum (FBS) and penicillin/streptomycin.
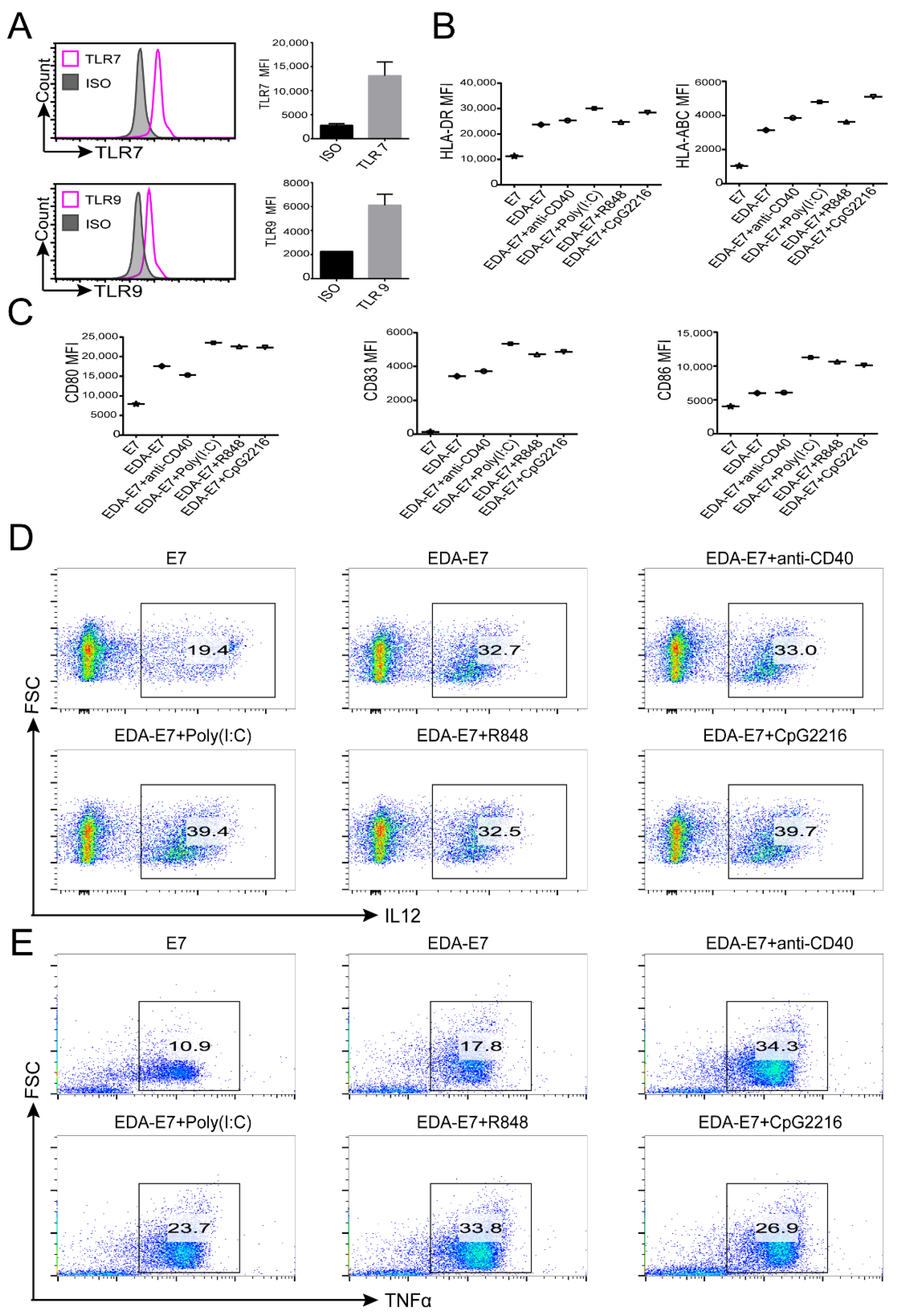
DCs culture: buffy coat is from healthy donors as approved by the ethics committee of Chongqing University Cancer Hospital. A total of 15 donors were used in this study. Each experiment was biologically replicated at least. Human PBMC were purified from buffy coat by density gradient centrifugation using Ficoll (Cytiva, Grens, Switzerland). PBMC were suspended in RPMI 1640 (Gibco, Beijing, China) basic medium with concentration of 2 × 106/mL. Seed PBMC in 24 well plate and culture in 37 °C, 5%CO2 for 90 min. To obtain DCs, the attached mononuclear cells were cultured for additional 5 days in RPMI 1640 (Gibco, Beijing, China) supplemented with 5% human serum, 100 U/mL penicillin and 100 µg/mL streptomycin (P/S), 2 mM L-glutamine, 800 IU/mL GM-CSF (Peprotech, Cranbury, NJ, USA) and 200 U/mL IL-4 (Peprotech, Cranbury, NJ, USA). For DCs maturation, 500 nM EDA-E7, 500 nM EDA-E7 + 10 ug/mL poly (I:C) (Invivogen, Toulouse, France), 500 nM EDA-E7 + 2 uM CPG (CPG2216, Invivogen, Toulouse, France), 500 nM EDA-E7 + 1 ug/mL R848 (Invivogen, Toulouse, France), or 500 nM EDA-E7 + 100 ng/mL anti-CD40 (Abcam, Cambridge, UK) were added into DCs culture medium on day 6 after DC stimulation.
For lymphocyte purification, we collected the unattached cell after incubating the PBMC in a 24 well plate for 90 min as described above, then purified T cells using human Pan T Cell Isolation Kit (Miltenyi Biotec, Gaithersburg, MD, USA) and cultured them in lymphocyte serum-free medium (Dayou, cat#:6111021, Hangzhou, China). For T cell activation, on day 7 of DCs culture, 1:1 (DCs:T cells) naïve pan T cells were added into DCs cells with addition of 200 U/mL IL-2, 30 ng/mL IL-21, 5 ng/mL IL-15. and 5 ng/mL IL7, then cultured in 37 °C, 5%CO2 for 10 days before analysis.
2.2. Human Recombinant EDA-E7 Fusion Protein Preparation
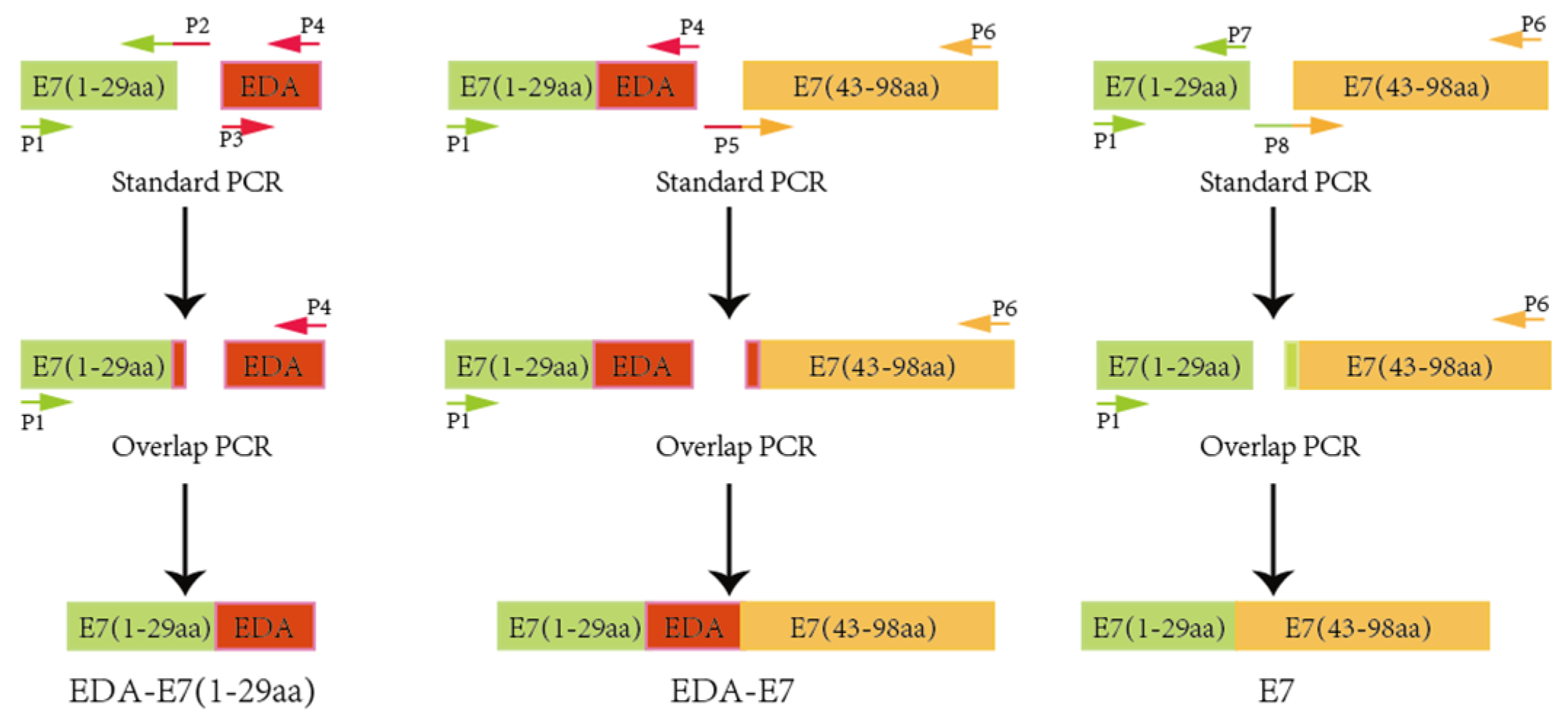
RNA from SiHa was isolated with RNA isolation kit (Omega, Atlanta, GA, USA) and reverse transcript into cDNA using PrimeScript RT reagent kit from Takara. We depleted pRB binding domain of HPVE7, then link E7 1-29 first amino acids (aa) to the N terminal of EDA and 43-98 aa to the C terminus of EDA to make target recombinant protein sequence: E7(1-29)aa + EDA + E7(43-98)aa (EDA + E7), while control protein without EDA as: E7(1-29)aa + E7(43-98)aa (E7). To construct the fusion protein, we used the over-lap PCR method. PCR primers are shown in
Table 1.
To construct E7(1-29)aa + EDA target DNA, primers 1 and 2 were used and SiHa cDNA was applied as a template to amplify E7(1-29)aa target sequence; while primer 3 and 4 were used and 293 cells cDNA were used as template to amplify human EDA target sequence. Purify E7(1-29)aa and EDA target DNA and link them to get E7(1-29)aa + EDA target DNA. E7(1-29)aa + EDA from previous step were further amplified using primer 1 and 4. E7(43-98)aa were amplified with primer 5 and 6, then linked to E7(1-29)aa + EDA. To get E7(1-29)aa + E7(43-98)aa DNA, product of primer 1 and 7 with template of SiHa cDNA, and product of primer 6 and 8 with template of EDA expression plasmid were linked together via the overlap sequence. For the construction of pET20b expressing EDA-E7 or E7, plasmid pET20b (Kindly provided by Prof. Jesu´s Prieto from Centro de Investigacio´n Me´dica Aplicada CIMA, Pamplona, Spain) and target DNA were digested with restriction enzyme NdeI and NotI, then ligated to get pET20b-EDA-E7 and pET20b-E7. Cloning protocol is outlined as below (
Figure 1):
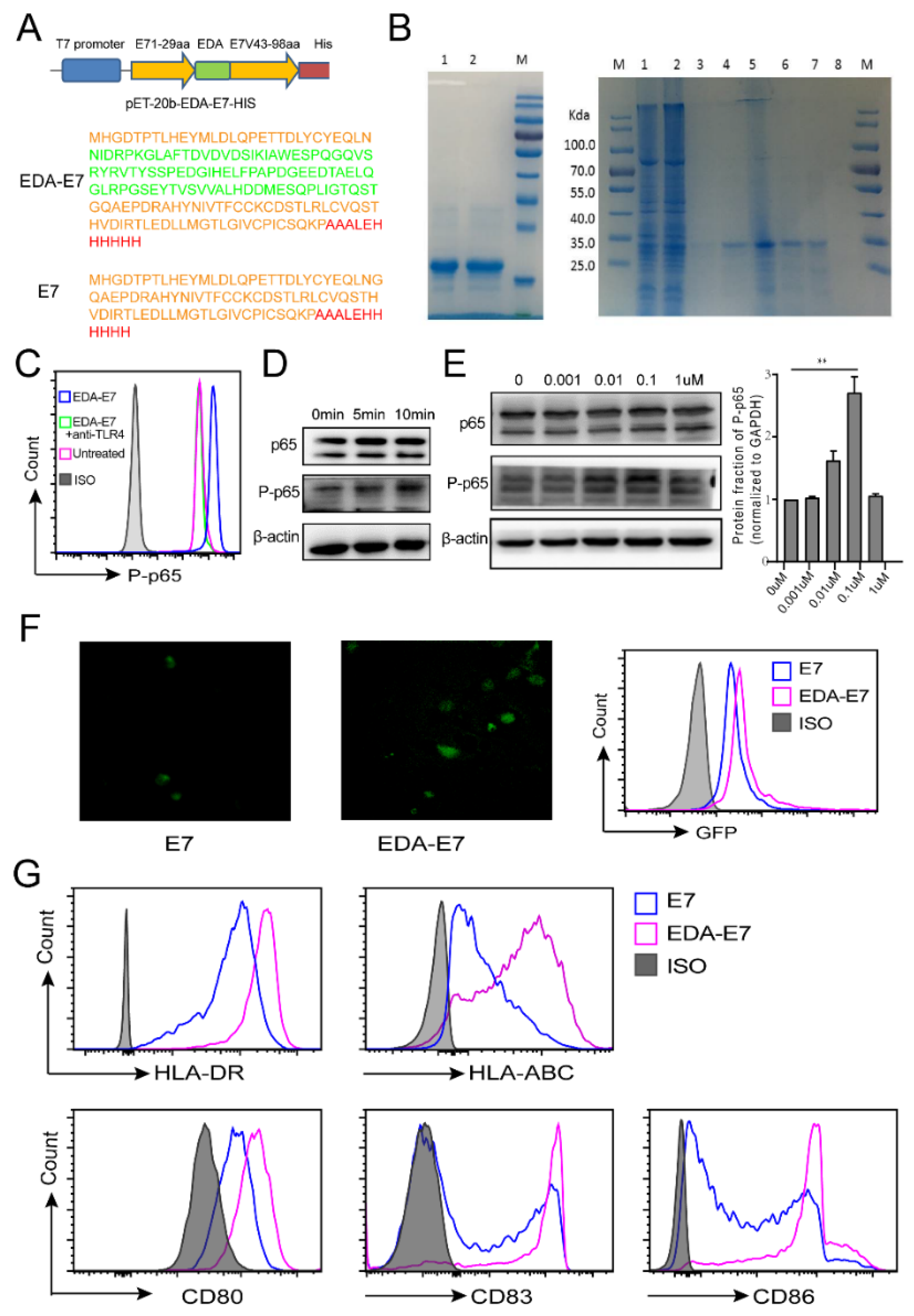
To obtain EDA-E7 or E7 recombinant protein, pET20b-EDA-HPVE7 and pET20b-E7 were transfected into BL21 (DE3) E-coli; incubate in 37 °C, 260 r/min shaking culture. Add 0.5 mM IPTG (isopropylthio-β-galactoside, purchased from Thermo Fisher Scientific, Shanghai, China) to the culture when the BL21 OD600 reached 0.5, then culture another 4h before protein purification. Affinity chromatography were applied to purify EDA-E7 and E7 recombinant protein. Then, elute the protein with different concentration of imidazole solution. SDS-PAGE and Coomassie brilliant blue staining were applied to confirm the target protein size as well as purity. Recombinant protein was purified with affinity chromatography as described previously [
14]. As the recombinant protein exists mainly in the inclusion body, protein renaturation using gentle removal of urea was applied as described previously [
15].
2.3. Analysis of E7, EDA-E7 Binding with DCs
E7 and EDA-E7 were labeled with LinKine™ FITC Labeling Kit (Abbkine, Wuhan, China), then 100 nM E7 or EDA-E7 was added into DCs culture. DCs binding with E7 or EDA-E7 was measured through FITC signal by a fluorescent microscope and flow cytometry after 24 h of incubation.
2.4. Flow Cytometry
For surface staining, cells were blocked with 2% normal rabbit serum and subsequently stained with fluorochrome-conjugated antibodies in FACS buffer (PBS + 2%FBS + P/S) at 4 °C for 30 min and analyzed with cytometer (Beckman coulter cytoflex). DCs activation were analyzed using anti-HLA-DR (Biolegend, San Diego, CA, USA), anti –HLA-ABC (BD Biosciences, New Jersey, USA), anti-CD80 (Biolegend, San Diego, CA, USA), anti-CD83 (Biolegend, San Diego, CA, USA), and anti-CD86 (Biolegend, San Diego, CA, USA). For T cells activation analysis, DC activated T cells were collected by centrifugation of the suspended pan T cells in 1000 rpm for 5 min, then stained with anti-CD3, anti-CD4, anti-CD8, anti-CD107a, anti-4-1BB, and anti-OX40 (Biolegend, San Diego, CA, USA) for flow analysis.
Intracellular staining for DCs IL12, TNFα: activated DCs were washed, fixed, and permeabilized using BD Cytofix/Cytoperm kit (BD Bioscience, Franklin Lakes, NJ, USA) at 4 °C for 20 min. The cells were then stained with anti-IL-12 and anti-TNFα (Biolegend, San Diego, CA, USA) in permeabilization solution following the protocol provided. Data was acquired on Beckman coulter cytoflex.
For the analysis of TLR7 and TLR9 expression on DCs, DCs were fixed and permeabilized as described above. Then, stained with anti-TLR7 and anti-TLR9 (Biolegend, San Diego, CA, USA) for flow analysis.
For caspase-3 staining, target cells SiHa were washed after coculture with T cells for 4 h to remove T cells, then digested to single cells with trypsin (Gibco, Beijing, China). The cells were fixed and permeabilized as described previously using BD cytofix/Cytoperm kit, and stained with anti-cleaved caspase-3 (BD Bioscience, Franklin Lakes, NJ, USA) before analysis.
2.5. Monocyte Activation Analysis
To analyze the function of TLR4 for EDA-E7 induced NF-KB activation, EDA-E7 was added to the THP-1 cells with or without addition of TLR4 blocking antibody (Biolegend, cat#: 312802, 20 μg/mL, San Diego, CA, USA). After incubation for 10 min, intracellular flow cytometry for P-p65 was performed to analyze the phosphorylation of p-65. Briefly, stimulated THP-1 cells were fixed and permeabilized as previously described, then stained with anti-P-p65 (CST, cat#: 3033S, Danvers, MA, USA) for 30 min at 4 °C. Washed and stained with secondary antibody, the anti-rabbit conjugated with Alexa fluor 647, then washed and analyzed with flow cytometer.
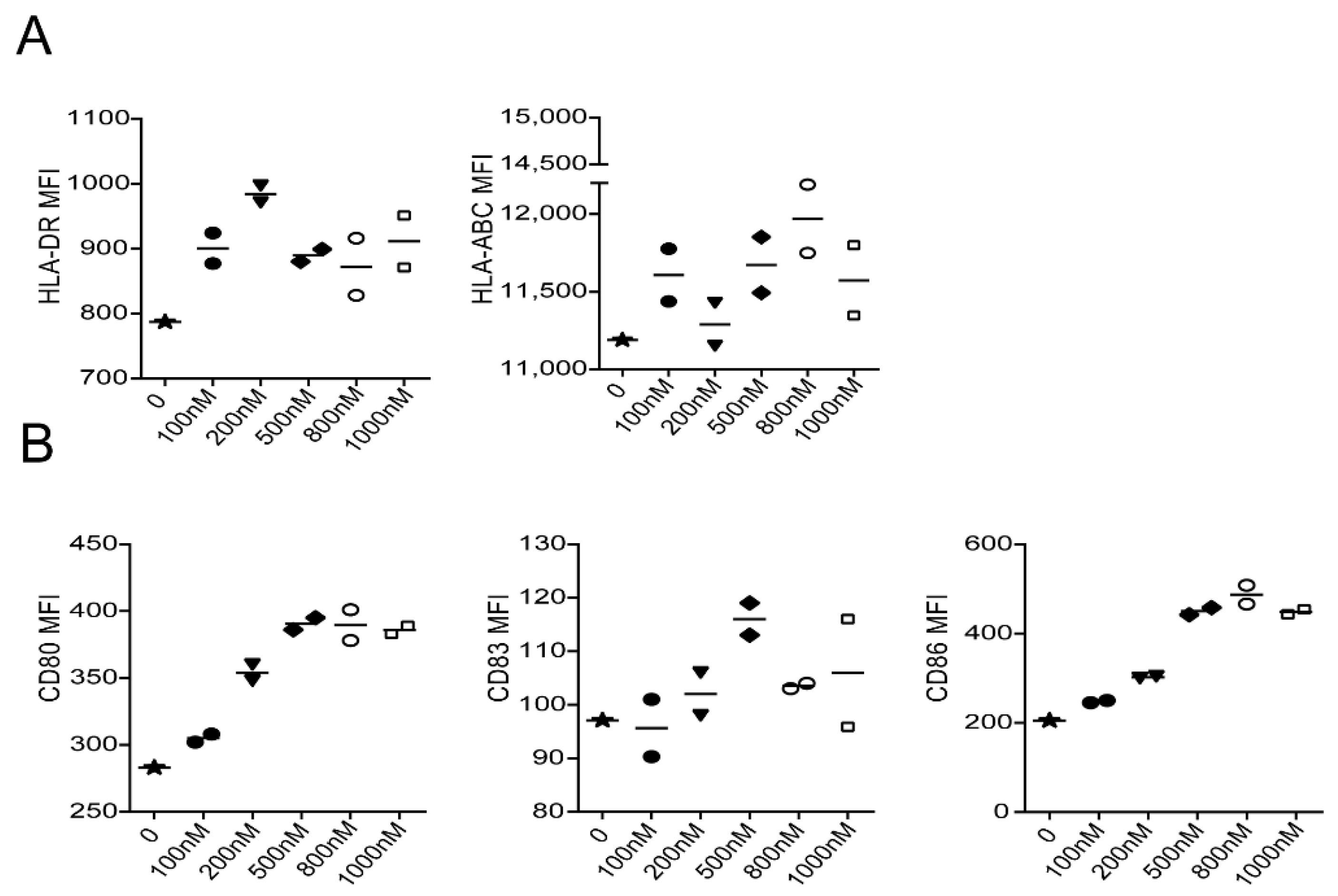
To determine which time point could achieve the best effect of THP-1 activation, THP-1 cells were cultured in a 12 well plate and 100 nM EDA-E7 were added to the culture. After coculture for 0 min, 5 min, 10 min, we collected the cells for analysis of p65 phosphorylation with Western blot. To test the dose effect of EDA-E7 on TLR4 pathway activation, THP-1 were treated with different doses of EDA-E7 and cultured in 37 °C, 5% CO2 for 10 min. Cells were collected and washed with PBS, then lysed with RIPA buffer supplemented with protease and phosphatase inhibitor cocktail (Roche, Switzerland). Purified and degenerated proteins were loaded onto 12% SDS-PAGE gels followed by electrophoretic transfer to nitrocellulose membranes. Primary antibodies for p65 and P-p65 were purchased from Abcam (Cambridge, UK); anti-β-actin antibody was from CST company (Danvers, MA, USA).
2.6. T Cell Proliferation Assay
For T cell proliferation, matured DCs (5 × 105) induced by different combinations of TLR agonists were cocultured with Naive Pan T (1:1) in 6-well plates in DCs culture medium. Cells were counted on day 7 and day 10 post coculture. There are 3 replicates for each group and the proliferation rate of each group was calculated based on the mean total cell number for each group. Since DCs could hardly proliferate during in vitro culture, it is difficult to separate T cells from DCs for cell counting. In addition, on day 7 and day 10 post coculture, total cell number of DCs and T cells reached more than 10 million. Therefore, we considered that the number of DCs would not affect the growth curve of T cells.
2.7. T Cell Receptor (TCR) Coupled Single Cell RNA Sequencing
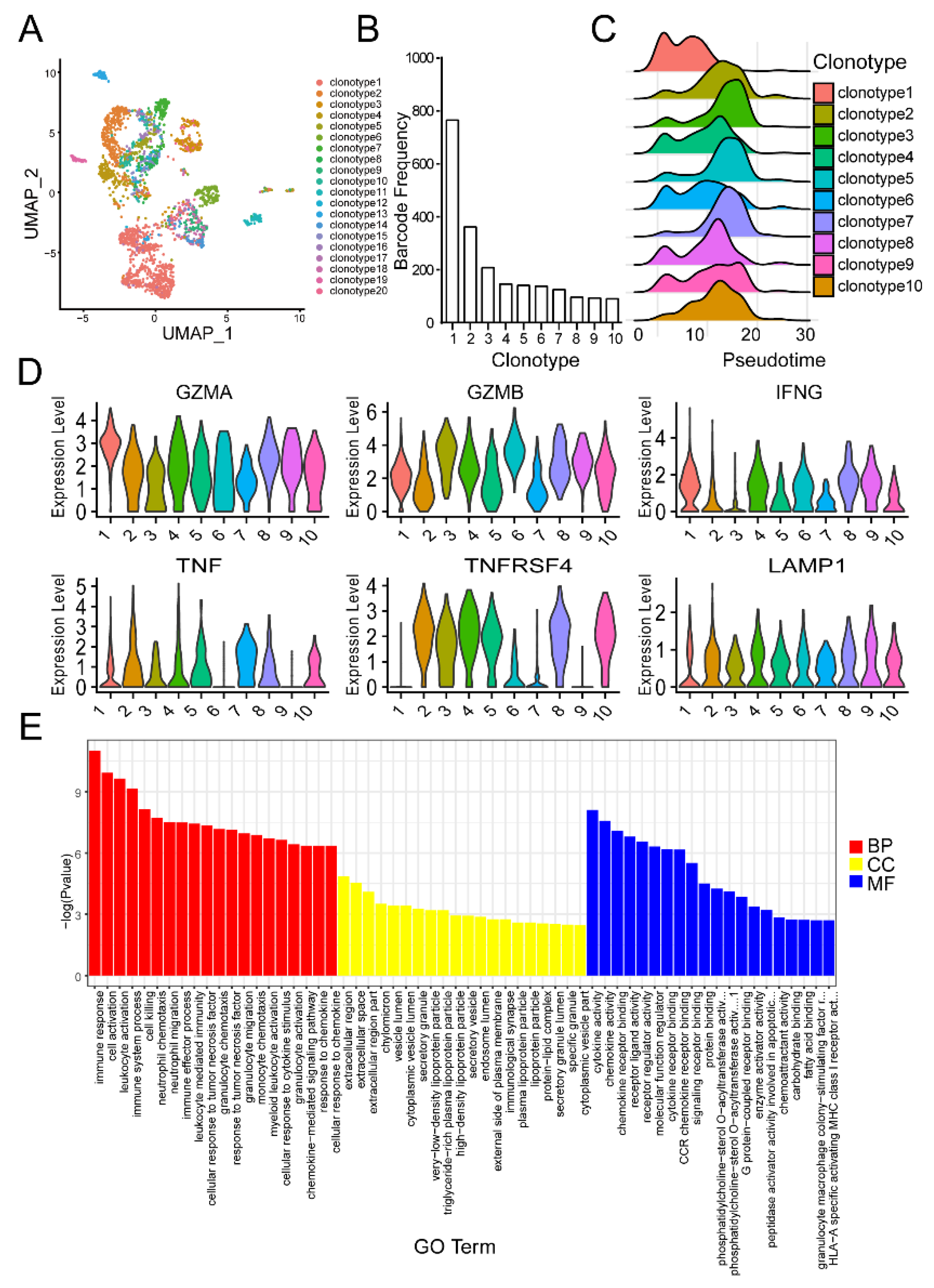
Collect T cells on day7 of T cells activation by EDA-E7 matured DCs for TCR coupled single cell RNA sequencing. Sequencing was completed by Beijing Genomics Institute (Beijing, China). To identify clonotypes, we used a 10×Genomics Cell Ranger pipeline with alignment and annotation according to the manufacturer’s instruction. TCR were aligned to GRCh38 reference genome. In-frame TCR alpha-beta pairs were considered as dominant TCR of a single cell.
2.8. T Cell In Vitro Cytotoxicity Assay
To overexpress HLA-A*02:01 in SiHa, HLA-A*02:01 expression sequence was synthesized in Gene Create company and constructed into a lentiviral plasmid with GFP as a transfection reporter gene. SiHa transfected with HLA-A*02:01 expressing lentivirus were sorted by GFP reporter using flow cytometry.
HPV16E7 11–19 episode specific TCR cloning and targeted cytotoxic assay were conducted accordingly to a previously published article [
16]. Briefly, PBMC derived T cells were activated using CD3 and CD28 activation beads. TCR overexpression lentivirus was added into the activated T cells at 36h post activation. Percentage of TCR expression T cells were confirmed by flow cytometry for reporter GFP expression. We could get around 80% GFP + T cells from each infection, which we consider sufficient for in vitro experiment. On day 7 post infection, TCR expression T cells were incubated with wild type SiHa or HLA-A*02:01 expressing SiHa with E:T ratio 5:1 in SiHa culture medium (DMEM + 10%FBS + P/S). As SiHa were labeled with luciferase, we used luciferase signal to measure the lysis efficiency of T cells. After incubation for 12 h, luciferase assay was conducted to evaluate T cell cytotoxicity.
For EDA-E7 induced T cell specific cytotoxicity assay, control E7 or EDA-E7 matured DCs were incubated with PBMC derived CD8+ T cells with ratio DCs:T as 1:4 to activate T cells. After incubation for 7 days, DCs activated T cells were purified with magnetics beads for 4-1BB. Then, 1 × 104 HLA-A*02:01 expressing and wild type SiHa were seeded in 96 well plate, after attachment overnight, and T cells were added into SiHa with indicated ratios. T cell lysis efficiency was analyzed by luciferase signal at 12 h post incubation. For apoptotic assay, T cells activated by E7 or EDA-E7 stimulated DCs were cocultured with HLA-A*02:01 expressing or wild type SiHa with E:T ratios 5:1 and 10:1 for 4 h before flow cytometry for caspase 3.
2.9. Statistical Analysis
Data is presented as mean ± SD. Statistical comparisons between groups were analyzed by a student’s t-test. A p value < 0.05 was considered statistically significant.
4. Discussion
Cervical cancer is one of the main malignant tumors that endanger the health of women worldwide. The fact that 99% of cervical cancer patients were positive for HPV while type 16 HPV in Southwest of China is as high as 73.8% [
3,
4] makes the vaccination of cervical cancer via HPV possible. Compared to checkpoint inhibitor immunotherapy such as anti-PD-1 or anti-PD-L1 therapy, which relies largely on the expression of PD-L1 in cancer cells, vaccination with HPV antigen seems more promising. Indeed, a preventive vaccine against HPV has already shown great potential to prevent 90% occurrence of cervical cancer [
22,
23]. However, tumor immunosuppressive microenvironment including the recruitment of regulatory T cells and myeloid derived suppressor cells, as well as hypoxia, high interstitial fluid pressure, and physical barriers, make the immune response not efficient for therapeutic vaccinations for established tumor. EDA-E7 as a therapeutic vaccine has already been shown to eradicate large tumor in mice by repeated intratumor injection, which could possibly be because EDA could not just activate DCs through TLR4 signaling pathway, but also create a pro-inflammatory microenvironment in tumor as reported by other groups, and attract the infiltration of activated T cells [
10]. In addition, in vitro generation of HPV vaccine is expensive and time consuming, as well as difficult to standardize each batch of product. Therefore, how to generate efficient therapeutic vaccine for HPV positive cervical cancer is of great interest.
The spliced exon encoding the type III repeat extra domain A (EDA) from fibronectin could target antigens to DCs and induce maturation through TLR4 [
13]. Furthermore, a mouse-derived EDA-E7 recombinant fused protein has been shown to induce maturation of DCs and was able to eradicate well-established tumors expressing HPVE7 protein in mouse system [
14]. In this study, we generated human derived EDA-HPVE7 fused protein and confirmed that this recombinant protein maintains the pro-inflammatory property of the EDA domain as well as to induce the maturation of DCs through binding of HPV16E7. From
Figure 2B we can see that EDA-E7 could bind to DCs and upregulate antigen presenting molecules and costimulatory molecules. Furthermore, 500 nM concentration of EDA-E7 was found to achieve the best activation of DCs (
Figure 3). TLR agonist was reported to work in synergy with EDA-E7 to eradicate established tumors through induction of pro-inflammatory cytokines such as TNFα and IL12. In this study, we also found that the recombinant protein EDA-E7 and TLR agonist could work in synergy to promote the secretion of cytokines from DCs to achieve functional maturity. Since it is difficult to use a humanized mouse model to mimic human immune system for eradicating established tumor in vivo, we used in vitro experiment to evaluate whether the DCs could induce antigen specific T cells. From a T cell activation marker as well as target killing experiments, we can conclude that naïve T cells were indeed activated after incubation with EDA-E7 and TLR agonist matured DCs. TCR-coupled single cell RNA sequencing indicated TCR clonal selection and amplification of T cells. In vitro T cell cytotoxic experiment indicated that EDA-E7 could induce HLA restricted antigen specific T cells response against HPV infected cervical cancer cell (
Figure 7). In the future, we will continue to use humanized mouse models to verify the effect of EDA-E7 in inducing antigen specific T cells and eradication efficiency of HPV infected cervical cancer in vivo.
In conclusion, we synthesized a human origin fusion protein EDA-E7, which could induce maturation of human DCs and activate anti-HPV infected cervical cancer immune responses in vitro. Moreover, combined use with a TLR agonist such as poly (I:C) will achieve better maturation of DCs and T cell activation. Our study found that EDA-E7 could activate human T cells to kill human cervical tumor cells arising from HPV infection, which indicates it is worthwhile to investigate these observations further in humanized mice.