The Immunogenicity of a Foot-and-Mouth Disease Virus Serotype O Vaccine in Commercial and Subsistence Cattle Herds in Zambia
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Vaccine
2.3. Serological Testing
2.4. Data Analysis
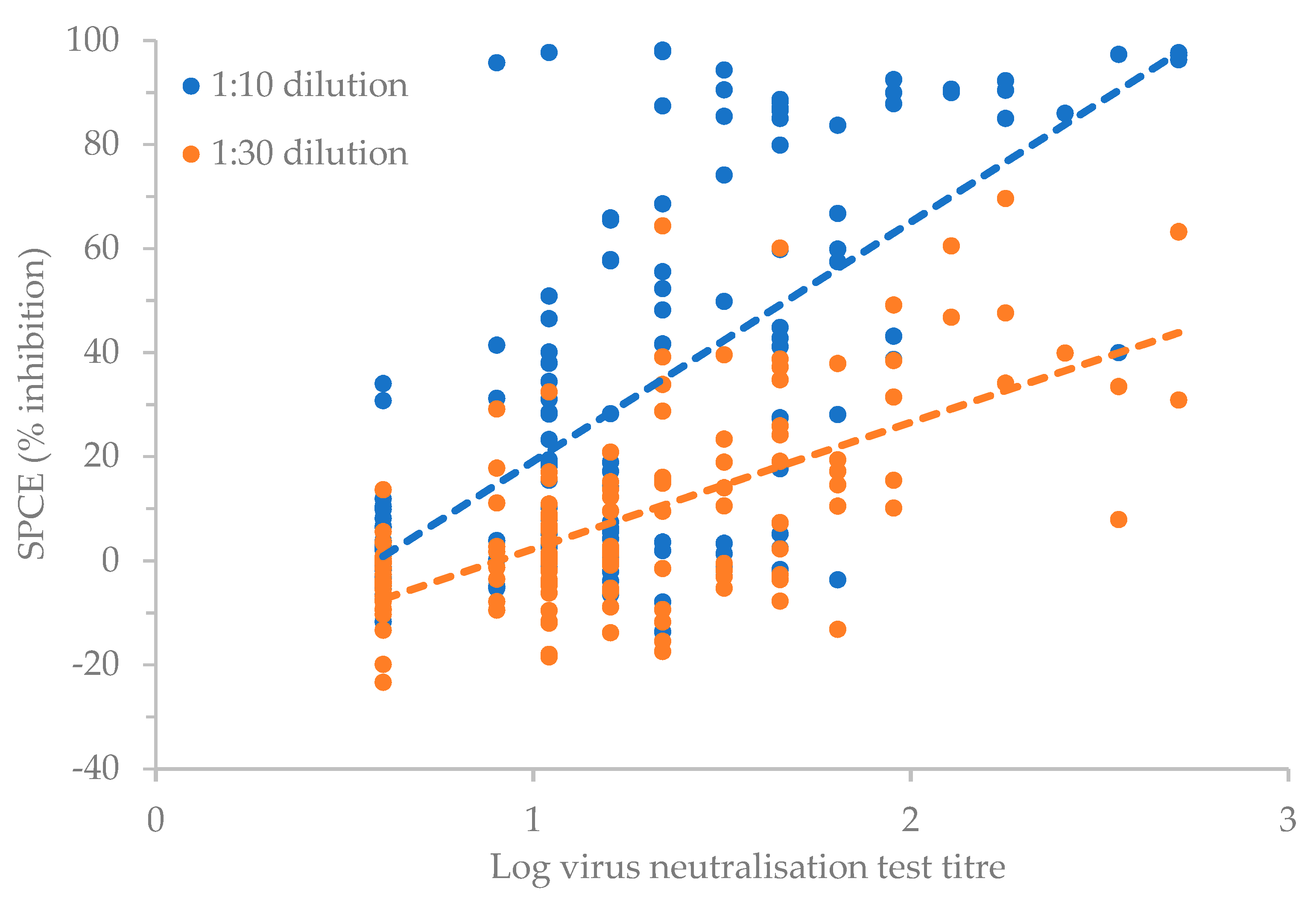
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Woldemariyam, F.T.; Kariuki, C.K.; Kamau, J.; De Vleeschauwer, A.; De Clercq, K.; Lefebvre, D.J.; Paeshuyse, J. Epidemiological Dynamics of Foot-and-Mouth Disease in the Horn of Africa: The Role of Virus Diversity and Animal Movement. Viruses 2023, 15, 969. [Google Scholar] [CrossRef] [PubMed]
- Chitray, M.; Grazioli, S.; Willems, T.; Tshabalala, T.; De Vleeschauwer, A.; Esterhuysen, J.J.; Brocchi, E.; De Clercq, K.; Maree, F.F. Development and validation of a foot-and-mouth disease virus SAT serotype-specific 3ABC assay to differentiate infected from vaccinated animals. J. Virol. Methods 2018, 255, 44–51. [Google Scholar] [CrossRef] [PubMed]
- Banda, F.; Shilongo, A.; Hikufe, E.H.; Khaiseb, S.; Kabajani, J.; Shikongo, B.; Set, P.; Kapapero, J.K.; Shoombe, K.K.; Zaire, G.; et al. The first detection of a serotype O foot-and-mouth disease virus in Namibia. Transbound. Emerg. Dis. 2022, 69, 3261–3267. [Google Scholar] [CrossRef]
- Overby, E.; Zyambo, G.C.N. Foot and Mouth Disease outbreaks in Zambia. Rev. Sci. Tech. Off. Int. Epizotics 1983, 2, 189–197. [Google Scholar] [CrossRef] [PubMed]
- Chilonda, P.; Woodford, J.D.; Ahmadu, B.; Samui, K.L.; Syakalima, M.; Mlangwa, J.E. Foot and mouth disease in Zambia: A review of the aetiology and epidemiology and recommendations for possible control. Rev. Sci. Tech. Off. Int. Epizotics 1999, 18, 585–592. [Google Scholar] [CrossRef]
- Mweene, A.S.; Pandey, G.S.; Sinyangwe, P.; Nambota, A.; Samui, K.; Kida, H. Viral diseases of livestock in Zambia. Jpn. J. Vet. Res. 1996, 44, 89–105. [Google Scholar]
- WRLFMD. World Reference Laboratory for FMD Quarterly Report for April to June 2022. P 12,25 and 26. Available online: https://www.wrlfmd.org/ref-lab-reports (accessed on 8 October 2023).
- FAO. Quarterly Report. 2023. Available online: https://www.fao.org/3/cc4292en/cc4292en.pdf (accessed on 25 November 2023).
- Bergmann, I.E.; Malirat, V.; Neitzert, E.; Beck, E.; Panizzutti, N.; Sanchez, C.; Falczuk, A. Improvement of a serodiagnostic strategy for foot-and-mouth disease virus surveillance in cattle under systematic vaccination: A combined system of an indirect ELISA-3ABC with an enzyme-linked immunoelectrotransfer blot assay. Arch. Virol. 2000, 145, 473–489. [Google Scholar] [CrossRef]
- Leforban, Y.; Gerbier, G. Review of the status of foot and mouth disease and approach to control/eradication in Europe and Central Asia. Rev. Sci. Tech. Off. Int. Epizotics 2002, 21, 477–492. [Google Scholar] [CrossRef]
- Hunter, P. Vaccination as a means of control of foot-and-mouth disease in sub-saharan Africa. Vaccine 1998, 16, 261–264. [Google Scholar] [CrossRef]
- FAO-OIE. Foot and Mouth Disease Vaccination and Post-Vaccination Monitoring Guidelines. 2016. Available online: https://www.fao.org/3/i5975e/i5975e.pdf (accessed on 23 December 2018).
- Fana, E.M.; Mpoloka, S.W.; Leteane, M.; Seoke, L.; Masoba, K.; Mokopasetso, M.; Rapharing, A.; Kabelo, T.; Made, P.; Hyera, J. A Five-Year Retrospective Study of Foot-and-Mouth Disease Outbreaks in Southern Africa, 2014 to 2018. Vet. Med. Int. 2021, 2021, 7438809. [Google Scholar] [CrossRef]
- Doel, T.R. Optimisation of the immune response to foot-and-mouth disease vaccines. Vaccine 1999, 17, 1767–1771. [Google Scholar] [CrossRef] [PubMed]
- WOAH-Manual. Foot and Mouth Disease Chapter 3.1.8. 2022. Manual of Diagnostic Tests and Vaccines for Terrestrial Animals, Twelfth Edition WOAH. 2021. Available online: https://www.woah.org/en/what-we-do/standards/codes-and-manuals/terrestrial-manual-online-access/ (accessed on 30 June 2021).
- Barnett, P.V.; Geale, D.W.; Clarke, G.; Davis, J.; Kasari, T.R. A Review of OIE Country Status Recovery Using Vaccinate-to-Live Versus Vaccinate-to-Die Foot-and-Mouth Disease Response Policies I: Benefits of Higher Potency Vaccines and Associated NSP DIVA Test Systems in Post-Outbreak Surveillance. Transbound. Emerg. Dis. 2015, 62, 367–387. [Google Scholar] [CrossRef]
- Colling, A.; Morrissy, C.; Barr, J.; Meehan, G.; Wright, L.; Goff, W.; Gleeson, L.J.; van der Heide, B.; Riddell, S.; Yu, M.; et al. Development and validation of a 3ABC antibody ELISA in Australia for foot and mouth disease. Aust. Vet. J. 2014, 92, 192–199. [Google Scholar] [CrossRef] [PubMed]
- Bruderer, U.; Swam, H.; Haas, B.; Visser, N.; Brocchi, E.; Grazioli, S.; Esterhuysen, J.J.; Vosloo, W.; Forsyth, M.; Aggarwal, N.; et al. Differentiating infection from vaccination in foot-and-mouth-disease: Evaluation of an ELISA based on recombinant 3ABC. Vet. Microbiol. 2004, 101, 187–197. [Google Scholar] [CrossRef]
- Chen, T.H.; Lee, F.; Lin, Y.L.; Dekker, A.; Chung, W.B.; Pan, C.H.; Jong, M.H.; Huang, C.C.; Lee, M.C.; Tsai, H.J. Differentiation of foot-and-mouth disease-infected pigs from vaccinated pigs using antibody-detecting sandwich ELISA. J. Vet. Med. Sci. 2011, 73, 977–984. [Google Scholar] [CrossRef] [PubMed]
- WRLFMD. FMD Vaccine Matching Strain Differeciation Report. 2021. Available online: https://www.wrlfmd.org/sites/world/files/quick_media/WRLFMD-2021-00008-ZAM-VMR-O-multiple_001.pdf (accessed on 15 November 2021).
- FAO-WRLFMD. Recommendation to AgResults on Using Serological Indicators (“Valency Testing”) of Cross-Protection for FMD Vaccines. 2022. Available online: https://www.wrlfmd.org/sites/world/files/quick_media/Cross-neutralisation%20measure%20AgResults%20Final%20v2.1.pdf (accessed on 22 January 2023).
- Hammond, J.M.; Maulidi, B.; Henning, N. Targeted FMD Vaccines for Eastern Africa: The AgResults Foot and Mouth Disease Vaccine Challenge Project. Viruses 2021, 13, 1830. [Google Scholar] [CrossRef]
- Perry, B.D.; Mwanaumo, B.; Schels, H.F.; Eicher, E.; Zaman, M.R.; Perry, B.D.; Mwanaumo, B.; Schels, H.F.; Eicher, E.; Zaman, M.R. A study of health and productivity of traditionally managed cattle in Zambia. Prev. Vet. Med. 1984, 2, 633–653. [Google Scholar] [CrossRef]
- Landis, J.R.; Koch, G.G. The measurement of observer agreement for categorical data. Biometrics 1977, 33, 159–174. [Google Scholar] [CrossRef]
- Altman, D.G. Statistics in the medical literature: 3. Stat. Med. 1999, 18, 487–490. [Google Scholar] [CrossRef]
- Gobiye, M.; Fosgate, G.T.; Heath, L.; Lazarus, D.D.; Seoke, L.; Opperman, P.A. Preliminary validation of a single-spot version of a solid-phase competition ELISA for the detection of southern African territories foot-and-mouth disease serotype exposure in goats. Small Rumin. Res. 2023, 224, 106982. [Google Scholar] [CrossRef]
- Gubbins, S.; Paton, D.J.; Dekker, A.; Ludi, A.B.; Wilsden, G.; Browning, C.F.J.; Eschbaumer, M.; Barnabei, J.; Duque, H.; Pauszek, L.L.; et al. Predicting cross-protection against foot-and-mouth disease virus strains by serology after vaccination. Front. Vet. Sci. 2022, 9, 106982. [Google Scholar] [CrossRef]
- Brehm, K.E.; Kumar, N.; Thulke, H.H.; Haas, B.; Brehm, K.E.; Kumar, N.; Thulke, H.H.; Haas, B. High potency vaccines induce protection against heterologous challenge with foot-and-mouth disease virus. Vaccine 2008, 26, 1681–1687. [Google Scholar] [CrossRef] [PubMed]
- Barnett, P.V.; Statham, R.J.; Vosloo, W.; Haydon, D.T.; Barnett, P.V.; Statham, R.J.; Vosloo, W.; Haydon, D.T. Foot-and-mouth disease vaccine potency testing: Determination and statistical validation of a model using a serological approach. Vaccine 2003, 21, 3240–3248. [Google Scholar] [CrossRef]
- van Bekkum, J.G.; Fish, R.C.; Nathans, I. Immunologic responses in Dutch cattle vaccinated with foot-and-mouth disease vaccines under field conditions: Neutralizing antibody responses and immunity to O, A, and C types. Am. J. Vet. Res. 1969, 30, 2125–2129. [Google Scholar] [PubMed]
- Robiolo, B.; La Torre, J.; Maradei, E.; Beascoechea, C.P.; Perez, A.; Seki, C.; Smitsaart, E.; Fondevila, N.; Palma, E.; Goris, N.; et al. Confidence in indirect assessment of foot-and-mouth disease vaccine potency and vaccine matching carried out by liquid phase ELISA and virus neutralization tests. Vaccine 2010, 28, 6235. [Google Scholar] [CrossRef] [PubMed]
- Paton, D.J.; Reeve, R.; Capozzo, A.V.; Ludi, A. Estimating the protection afforded by foot-and-mouth disease vaccines in the laboratory. Vaccine 2019, 37, 5515–5524. [Google Scholar] [CrossRef]
- Ludi, A.B.; Morris, A.; Gubbins, S.; Asfor, A.; Afzal, M.; Browning, C.F.; Grazioli, S.; Foglia, E.A.; Wilsden, G.; Burman, A.; et al. Cross-Serotype Reactivity of ELISAs Used to Detect Antibodies to the Structural Proteins of Foot-and-Mouth Disease Virus. Viruses 2022, 14, 1495. [Google Scholar] [CrossRef]
- Chénard, G.; Miedema, K.; Moonen, P.; Schrijver, R.S.; Dekker, A. A solid-phase blocking ELISA for detection of type O foot-and-mouth disease virus antibodies suitable for mass serology. J. Virol. Methods 2003, 107, 89–98. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Banda, F.; Ludi, A.B.; Wilsden, G.; Browning, C.; Kangwa, H.L.; Mooya, L.; Ngoma, M.; Muuka, G.M.; Mundia, C.; Fandamu, P.; et al. The Immunogenicity of a Foot-and-Mouth Disease Virus Serotype O Vaccine in Commercial and Subsistence Cattle Herds in Zambia. Vaccines 2023, 11, 1818. https://doi.org/10.3390/vaccines11121818
Banda F, Ludi AB, Wilsden G, Browning C, Kangwa HL, Mooya L, Ngoma M, Muuka GM, Mundia C, Fandamu P, et al. The Immunogenicity of a Foot-and-Mouth Disease Virus Serotype O Vaccine in Commercial and Subsistence Cattle Herds in Zambia. Vaccines. 2023; 11(12):1818. https://doi.org/10.3390/vaccines11121818
Chicago/Turabian StyleBanda, Frank, Anna B. Ludi, Ginette Wilsden, Clare Browning, Henry L. Kangwa, Lynnfield Mooya, Masuzyo Ngoma, Geoffrey M. Muuka, Cornelius Mundia, Paul Fandamu, and et al. 2023. "The Immunogenicity of a Foot-and-Mouth Disease Virus Serotype O Vaccine in Commercial and Subsistence Cattle Herds in Zambia" Vaccines 11, no. 12: 1818. https://doi.org/10.3390/vaccines11121818
APA StyleBanda, F., Ludi, A. B., Wilsden, G., Browning, C., Kangwa, H. L., Mooya, L., Ngoma, M., Muuka, G. M., Mundia, C., Fandamu, P., Paton, D. J., King, D. P., & Quan, M. (2023). The Immunogenicity of a Foot-and-Mouth Disease Virus Serotype O Vaccine in Commercial and Subsistence Cattle Herds in Zambia. Vaccines, 11(12), 1818. https://doi.org/10.3390/vaccines11121818






