SARS-CoV-2 Breakthrough Infections: Incidence and Risk Factors in a Large European Multicentric Cohort of Health Workers
Abstract
:1. Introduction
2. Materials and Methods
2.1. Design and Setting
2.2. Vaccination, Case Definition, and Inclusion Criteria
2.3. Outcome and Data Collection
2.4. Statistical Analysis
2.5. Ethical Approval
3. Results
3.1. Primary Analysis on Individual Data
3.2. Secondary Analyses on Aggregate Data
4. Discussion
Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhu, N.; Zhang, D.; Wang, W.; Li, X.; Yang, B.; Song, J.; Zhao, X.; Huang, B.; Shi, W.; Lu, R.; et al. A Novel Coronavirus from Patients with Pneumonia in China, 2019. N. Engl. J. Med. 2020, 382, 727–733. [Google Scholar] [CrossRef]
- Global Excess Deaths Associated with COVID-19 (Modelled Estimates). Available online: https://www.who.int/data/sets/global-excess-deaths-associated-with-COVID-19-modelled-estimates (accessed on 9 May 2022).
- Baden, L.R.; El Sahly, H.M.; Essink, B.; Kotloff, K.; Frey, S.; Novak, R.; Diemert, D.; Spector, S.A.; Rouphael, N.; Creech, C.B.; et al. Efficacy and Safety of the MRNA-1273 SARS-CoV-2 Vaccine. N. Engl. J. Med. 2021, 384, 403–416. [Google Scholar] [CrossRef]
- Sadoff, J.; Gray, G.; Vandebosch, A.; Cárdenas, V.; Shukarev, G.; Grinsztejn, B.; Goepfert, P.A.A.; Truyers, C.; Fennema, H.; Spiessens, B.; et al. Safety and Efficacy of Single-Dose Ad26.COV2.S Vaccine against COVID-19. N. Engl. J. Med. 2021, 384, 2187–2201. [Google Scholar] [CrossRef]
- Angel, Y.; Spitzer, A.; Henig, O.; Saiag, E.; Sprecher, E.; Padova, H.; Ben-Ami, R. Association Between Vaccination with BNT162b2 and Incidence of Symptomatic and Asymptomatic SARS-CoV-2 Infections Among Health Care Workers. JAMA 2021, 325, 2457. [Google Scholar] [CrossRef] [PubMed]
- Klompas, M. Understanding Breakthrough Infections Following MRNA SARS-CoV-2 Vaccination. JAMA 2021, 326, 2018. [Google Scholar] [CrossRef]
- CDC. Coronavirus Disease 2019 (COVID-19) [Internet]. Centers for Disease Control and Prevention. 2020. Available online: https://www.cdc.gov/coronavirus/2019-ncov/variants/variant-classifications.html (accessed on 20 July 2022).
- Lippi, G.; Mattiuzzi, C.; Henry, B.M. Updated Picture of SARS-CoV-2 Variants and Mutations. Diagnosis 2021, 9, 11–17, Tracking SARS-CoV-2 Variants [Internet]. Available online: https://www.who.int/activities/tracking-SARS-CoV-2-variants) (accessed on 20 July 2022). [CrossRef] [PubMed]
- WHO Tracking SARS-CoV-2 Variants. Available online: https://www.who.int/health-topics/typhoid/tracking-SARS-CoV-2-variants (accessed on 19 April 2022).
- Planas, D.; Saunders, N.; Maes, P.; Guivel-Benhassine, F.; Planchais, C.; Buchrieser, J.; Bolland, W.-H.; Porrot, F.; Staropoli, I.; Lemoine, F.; et al. Considerable Escape of SARS-CoV-2 Omicron to Antibody Neutralization. Nature 2022, 602, 671–675. [Google Scholar] [CrossRef]
- Andrews, N.; Stowe, J.; Kirsebom, F.; Toffa, S.; Rickeard, T.; Gallagher, E.; Gower, C.; Kall, M.; Groves, N.; O’Connell, A.M.; et al. COVID-19 Vaccine Effectiveness against the Omicron (B.1.1.529) Variant. N. Engl. J. Med. 2022, 386, 1532–1546. [Google Scholar] [CrossRef] [PubMed]
- Bergwerk, M.; Gonen, T.; Lustig, Y.; Amit, S.; Lipsitch, M.; Cohen, C.; Mandelboim, M.; Levin, E.G.; Rubin, C.; Indenbaum, V.; et al. COVID-19 Breakthrough Infections in Vaccinated Health Care Workers. N. Engl. J. Med. 2021, 385, 1474–1484. [Google Scholar] [CrossRef] [PubMed]
- Menni, C.; Klaser, K.; May, A.; Polidori, L.; Capdevila, J.; Louca, P.; Sudre, C.H.; Nguyen, L.H.; Drew, D.A.; Merino, J.; et al. Vaccine Side-Effects and SARS-CoV-2 Infection after Vaccination in Users of the COVID Symptom Study App in the UK: A Prospective Observational Study. Lancet Infect. Dis. 2021, 21, 939–949. [Google Scholar] [CrossRef]
- Yamamoto, S.; Maeda, K.; Matsuda, K.; Tanaka, A.; Horii, K.; Okudera, K.; Takeuchi, J.S.; Mizoue, T.; Konishi, M.; Ozeki, M.; et al. COVID-19 Breakthrough Infection and Post-Vaccination Neutralizing Antibody among Healthcare Workers in a Referral Hospital in Tokyo: A Case-Control Matching Study. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2021. [Google Scholar] [CrossRef]
- Thomas, S.J.; Moreira, E.D.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Pérez Marc, G.; Polack, F.P.; Zerbini, C.; et al. Safety and Efficacy of the BNT162b2 MRNA COVID-19 Vaccine through 6 Months. N. Engl. J. Med. 2021, 385, 1761–1773. [Google Scholar] [CrossRef]
- Shrotri, M.; Navaratnam, A.M.D.; Nguyen, V.; Byrne, T.; Geismar, C.; Fragaszy, E.; Beale, S.; Fong, W.L.E.; Patel, P.; Kovar, J.; et al. Spike-Antibody Waning after Second Dose of BNT162b2 or ChAdOx1. Lancet 2021, 398, 385–387. [Google Scholar] [CrossRef]
- Gaebler, C.; Wang, Z.; Lorenzi, J.C.C.; Muecksch, F.; Finkin, S.; Tokuyama, M.; Cho, A.; Jankovic, M.; Schaefer-Babajew, D.; Oliveira, T.Y.; et al. Evolution of Antibody Immunity to SARS-CoV-2. Nature 2021, 591, 639–644. [Google Scholar] [CrossRef]
- Mizrahi, B.; Lotan, R.; Kalkstein, N.; Peretz, A.; Perez, G.; Ben-Tov, A.; Chodick, G.; Gazit, S.; Patalon, T. Correlation of SARS-CoV-2-Breakthrough Infections to Time-from-Vaccine. Nat. Commun. 2021, 12, 6379. [Google Scholar] [CrossRef] [PubMed]
- Seow, J.; Graham, C.; Merrick, B.; Acors, S.; Pickering, S.; Steel, K.J.A.; Hemmings, O.; O’Byrne, A.; Kouphou, N.; Galao, R.P.; et al. Longitudinal Observation and Decline of Neutralizing Antibody Responses in the Three Months Following SARS-CoV-2 Infection in Humans. Nat. Microbiol. 2020, 5, 1598–1607. [Google Scholar] [CrossRef] [PubMed]
- Goldberg, Y.; Mandel, M.; Bar-On, Y.M.; Bodenheimer, O.; Freedman, L.; Haas, E.J.; Milo, R.; Alroy-Preis, S.; Ash, N.; Huppert, A. Waning Immunity after the BNT162b2 Vaccine in Israel. N. Engl. J. Med. 2021, 385, e85. [Google Scholar] [CrossRef]
- Edelstein, M.; Beiruti, K.W.; Ben-Amram, H.; Bar-Zeev, N.; Sussan, C.; Asulin, H.; Strauss, D.; Bathish, Y.; Zarka, S.; Abu Jabal, K. Antibody-Mediated Immunogenicity against SARS-CoV-2 Following Priming, Boosting and Hybrid Immunity: Insights from 11 Months of Follow-up of a Healthcare Worker Cohort in Israel, December 2020–October 2021. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2022. [Google Scholar] [CrossRef]
- Oster, Y.; Benenson, S.; Nir-Paz, R.; Buda, I.; Cohen, M.J. The Effect of a Third BNT162b2 Vaccine on Breakthrough Infections in Health Care Workers: A Cohort Analysis. Clin. Microbiol. Infect. 2022, S1198743X2200043X. [Google Scholar] [CrossRef]
- Falsey, A.R.; Frenck, R.W.; Walsh, E.E.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Bailey, R.; Swanson, K.A.; Xu, X.; et al. SARS-CoV-2 Neutralisation with BNT162b2 Vaccine Dose 3. N. Engl. J. Med. 2021, 385, 1627–1629. [Google Scholar] [CrossRef]
- Eliakim-Raz, N.; Leibovici-Weisman, Y.; Stemmer, A.; Ness, A.; Awwad, M.; Ghantous, N.; Stemmer, S.M. Antibody Titers Before and After a Third Dose of the SARS-CoV-2 BNT162b2 Vaccine in Adults Aged ≥ 60 Years. JAMA 2021, 326, 2203. [Google Scholar] [CrossRef]
- Garcia-Beltran, W.F.; St. Denis, K.J.; Hoelzemer, A.; Lam, E.C.; Nitido, A.D.; Sheehan, M.L.; Berrios, C.; Ofoman, O.; Chang, C.C.; Hauser, B.M.; et al. MRNA-Based COVID-19 Vaccine Boosters Induce Neutralising Immunity against SARS-CoV-2 Omicron Variant. Cell 2022, 185, 457–466.e4. [Google Scholar] [CrossRef]
- Terreri, S.; Piano Mortari, E.; Vinci, M.R.; Russo, C.; Alteri, C.; Albano, C.; Colavita, F.; Gramigna, G.; Agrati, C.; Linardos, G.; et al. Persistent B Cell Memory after SARS-CoV-2 Vaccination Is Functional during Breakthrough Infections. Cell Host Microbe 2022, S1931312822000397. [Google Scholar] [CrossRef]
- Lipsitch, M.; Krammer, F.; Regev-Yochay, G.; Lustig, Y.; Balicer, R.D. SARS-CoV-2 Breakthrough Infections in Vaccinated Individuals: Measurement, Causes and Impact. Nat. Rev. Immunol. 2022, 22, 57–65. [Google Scholar] [CrossRef]
- Chou, R.; Dana, T.; Buckley, D.I.; Selph, S.; Fu, R.; Totten, A.M. Update Alert 10: Epidemiology of and Risk Factors for Coronavirus Infection in Health Care Workers. Ann. Intern. Med. 2022, 175, W8–W9. [Google Scholar] [CrossRef]
- von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gøtzsche, P.C.; Vandenbroucke, J.P. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement: Guidelines for Reporting Observational Studies. Lancet Lond. Engl. 2007, 370, 1453–1457. [Google Scholar] [CrossRef]
- CDC COVID-19 Vaccination. Available online: https://www.cdc.gov/coronavirus/2019-ncov/vaccines/effectiveness/why-measure-effectiveness/breakthrough-cases.html (accessed on 10 April 2022).
- Higgins, J.P.T.; Thompson, S.G. Quantifying Heterogeneity in a Meta-Analysis. Stat. Med. 2002, 21, 1539–1558. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.T.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring Inconsistency in Meta-Analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- DerSimonian, R.; Laird, N. Meta-Analysis in Clinical Trials. Control. Clin. Trials 1986, 7, 177–188. [Google Scholar] [CrossRef]
- Hellewell, J.; Russell, T.W.; Beale, R.; Kelly, G.; Houlihan, C.; Nastouli, E.; Kucharski, A.J. Estimating the Effectiveness of Routine Asymptomatic PCR Testing at Different Frequencies for the Detection of SARS-CoV-2 Infections. BMC Med. 2021, 19, 106. [Google Scholar] [CrossRef]
- Al Youha, S.; Alowaish, O.; Ibrahim, I.K.; Alghounaim, M.; Abu-Sheasha, G.A.; Fakhra, Z.; Al Hendi, S.; AlQabandi, Y.; Almazeedi, S.; Al Asoomi, F.; et al. Factors Associated with SARS-CoV-2 Infection amongst Healthcare Workers in a COVID-19 Designated Hospital. J. Infect. Public Health 2021, 14, 1226–1232. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Ochoa, S.A.; Franco, O.H.; Rojas, L.Z.; Raguindin, P.F.; Roa-Díaz, Z.M.; Wyssmann, B.M.; Guevara, S.L.R.; Echeverría, L.E.; Glisic, M.; Muka, T. COVID-19 in Health-Care Workers: A Living Systematic Review and Meta-Analysis of Prevalence, Risk Factors, Clinical Characteristics, and Outcomes. Am. J. Epidemiol. 2021, 190, 161–175. [Google Scholar] [CrossRef] [PubMed]
- Porru, S.; Carta, A.; Monaco, M.G.L.; Verlato, G.; Battaggia, A.; Parpaiola, M.; Lo Cascio, G.; Pegoraro, M.; Militello, V.; Moretti, F.; et al. Health Surveillance and Response to SARS-CoV-2 Mass Testing in Health Workers of a Large Italian Hospital in Verona, Veneto. Int. J. Environ. Res. Public. Health 2020, 17, 5104. [Google Scholar] [CrossRef]
- Boffetta, P.; Violante, F.; Durando, P.; De Palma, G.; Pira, E.; Vimercati, L.; Cristaudo, A.; Icardi, G.; Sala, E.; Coggiola, M.; et al. Determinants of SARS-CoV-2 Infection in Italian Healthcare Workers: A Multicenter Study. Sci. Rep. 2021, 11, 5788. [Google Scholar] [CrossRef] [PubMed]
- Chou, R.; Dana, T.; Selph, S.; Totten, A.M.; Buckley, D.I.; Fu, R. Update Alert 6: Epidemiology of and Risk Factors for Coronavirus Infection in Health Care Workers. Ann. Intern. Med. 2021, 174, W18–W19. [Google Scholar] [CrossRef]
- Basso, P.; Negro, C.; Cegolon, L.; Larese Filon, F. Risk of Vaccine Breakthrough SARS-CoV-2 Infection and Associated Factors in Healthcare Workers of Trieste Teaching Hospitals (North-Eastern Italy). Viruses 2022, 14, 336. [Google Scholar] [CrossRef]
- Nguyen, L.H.; Drew, D.A.; Graham, M.S.; Joshi, A.D.; Guo, C.-G.; Ma, W.; Mehta, R.S.; Warner, E.T.; Sikavi, D.R.; Lo, C.-H.; et al. Risk of COVID-19 among Front-Line Health-Care Workers and the General Community: A Prospective Cohort Study. Lancet Public Health 2020, 5, e475–e483. [Google Scholar] [CrossRef]
- Hall, V.; Foulkes, S.; Insalata, F.; Kirwan, P.; Saei, A.; Atti, A.; Wellington, E.; Khawam, J.; Munro, K.; Cole, M.; et al. Protection against SARS-CoV-2 after COVID-19 Vaccination and Previous Infection. N. Engl. J. Med. 2022, 386, 1207–1220. [Google Scholar] [CrossRef]
| Positive | Sex (%) | Job Title (%) | Age (10 Years %) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Centre | N | Cases (%) | Male | Female | Administr. | Technician | Nurse | Physician | Other HW | <30 | 30–39 | 40–49 | ≥50 |
| Individual data available | |||||||||||||
| Verona | 6404 | 97 (1.5) | 1979 (30.9) | 4425 (69.1) | 514 (8.0) | 579 (9.0) | 2176 (34.0) | 2167 (33.8) | 968 (15.1) | 1068 (16.7) | 1635 (25.5) | 1241 (19.4) | 2460 (38.4) |
| Padua | 6208 | 92 (1.5) | 1901 (30.6) | 4307 (69.4) | 542 (8.8) | 428 (7.0) | 2370 (38.5) | 1844 (30.0) | 973 (15.8) | 850 (13.7) | 1214 (19.6) | 1400 (22.6) | 2744 (44.2) |
| Trieste | 3559 | 59 (1.7) | 1013 (31.8) | 2169 (68.2) | 166 (5.2) | 144 (4.5) | 1313 (41.2) | 527 (16.5) | 1038 (32.6) | 220 (6.9) | 554 (17.4) | 854 (26.8) | 1554 (48.8) |
| Modena | 5250 | 90 (1.7) | 1550 (29.5) | 3699 (70.5) | 268 (5.2) | 179 (3.5) | 1846 (36.0) | 1603 (31.2) | 1239 (24.1) | 946 (18.0) | 1409 (26.8) | 1140 (21.7) | 1755 (33.4) |
| Perugia | 2364 | 30 (1.3) | 789 (33.4) | 1575 (66.6) | 170 (7.2) | 327 (13.8) | 1002 (42.4) | 514 (21.7) | 351 (14.9) | 31 (1.3) | 468 (19.8) | 543 (23.0) | 1322 (55.9) |
| Bari | 5923 | 38 (0.6) | 2330 (39.3) | 3593 (60.1) | 379 (6.4) | 200 (3.4) | 1612 (27.2) | 2884 (48.7) | 848 (14.3) | 901 (15.2) | 1356 (22.9) | 1081 (18.3) | 2585 (43.6) |
| Slovakia | 671 | 9 (1.3) | 106 (15.8) | 565 (84.2) | 74 (11.1) | 36 (5.4) | 227 (34.0) | 83 (12.4) | 247 (37.0) | 75 (11.2) | 96 (14.3) | 220 (32.8) | 280 (41.7) |
| Romania | 1458 | 11 (0.8) | 276 (18.9) | 1182 (81.1) | 69 (4.7) | 13 (0.9) | 172 (11.8) | 1080 (74.1) | 124 (8.5) | 89 (6.1) | 179 (12.3) | 440 (30.2) | 750 (51.4) |
| Subtotal | 31,837 | 426 (1.3) | 9944 (31.6) | 21,515 (68.4) | 2182 (7.0) | 1906 (6.1) | 10,718 (34.3) | 10,702 (34.2) | 5788 (18.5) | 4180 (13.3) | 6911 (22.0) | 6919 (22.0) | 13,450 (42.8) |
| Aggregated data only | |||||||||||||
| Turin | 8787 | 75 (0.9) | 2427 (27.6) | 6360 (72.4) | 1225 (13.9) | 1169 (13.3) | 3219 (36.6) | 1841 (20.9) | 1333 (15.2) | 1063 (12.1) | 1197 (13.6) | 2033 (23.1) | 4491 (51.1) |
| Brescia | 8903 | 134 (1.5) | 2446 (27.5) | 6457 (72.5) | 985 (11.1) | 702 (7.9) | 2855 (32.1) | 2642 (29.7) | 1719 (19.3) | 1386 (15.6) | 1968 (22.1) | 2015 (22.6) | 3534 (39.7) |
| Bologna | 7229 | 95 (1.3) | 2417 (33.4) | 4812 (66.6) | 274 (3.9) | 705 (10.0) | 2474 (34.9) | 1998 (28.2) | 1631 (23.0) | 1395 (19.3) | 1934 (26.7) | 1474 (20.4) | 2426 (33.6) |
| Oviedo | 7416 | 67 (0.9) | 1569 (21.2) | 5847 (78.8) | 691 (9.3) | 413 (5.6) | 3494 (47.1) | 1615 (21.8) | 1203 (16.2) | 582 (7.8) | 1290 (17.4) | 1760 (23.7) | 3784 (51.1) |
| Total | 64,172 | 797 (1.2) | 18,803 (29.5) | 44,991 (70.5) | 5357 (8.4) | 4895 (7.7) | 22,760 (35.9) | 18,798 (29.6) | 11674 (18.4) | 8606 (13.5) | 13,300 (20.8) | 14,201 (22.3) | 27,685 (43.4) |
| Hazard Ratio (95% CI) | p-Value | |
|---|---|---|
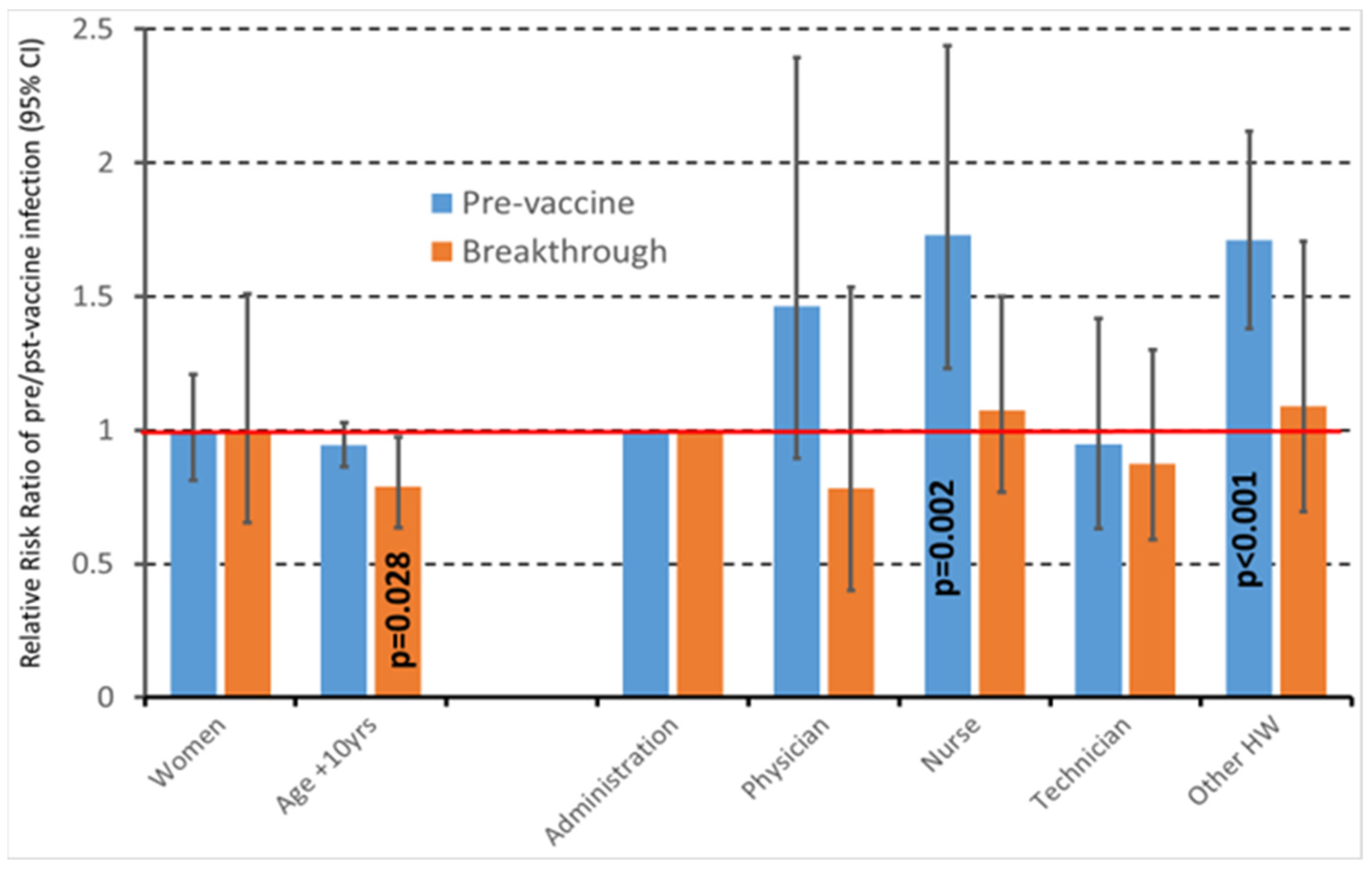
| Sex (Women vs. men) | 0.98 (0.79–1.23) | 0.899 |
| Age (increase by 10 years) | 0.81 (0.74–0.88) | <0.001 |
| Job title | ||
| Administrative | 1 (reference) | |
| Physician | 0.82 (0.53–1.27) | 0.374 |
| Nurse | 0.98 (0.64–1.49) | 0.920 |
| Technician | 0.75 (0.41–1.36) | 0.341 |
| Other HW | 1.03 (0.66–1.61) | 0.910 |
| Previous infection (Yes vs. No) | 0.23 (0.12–0.45) | <0.001 |
| Centres | Pooled OR (95% CI) | p-Value | I-Squared | |
|---|---|---|---|---|
| Gender: women vs. men | 11 | 0.91 (0.70–1.19) | 0.488 | 51.6% (p = 0.024) |
| Age (increased by 10 years) | 12 | 0.91 (0.80–1.05) | 0.188 | 45.5% (p = 0.043) |
| Job title (ref. administration) | ||||
| Physician | 11 | 1.05 (0.62–1.79) | 0.858 | 51.6% (p = 0.024) |
| Nurse | 12 | 1.29 (0.93–1.80) | 0.130 | 0.0% (p = 0.562) |
| Technician | 10 | 1.37 (0.86–2.18) | 0.191 | 0.0% (p = 0.777) |
| Other HW | 11 | 1.20 (0.82–1.74) | 0.353 | 0.0% (p = 0.516) |
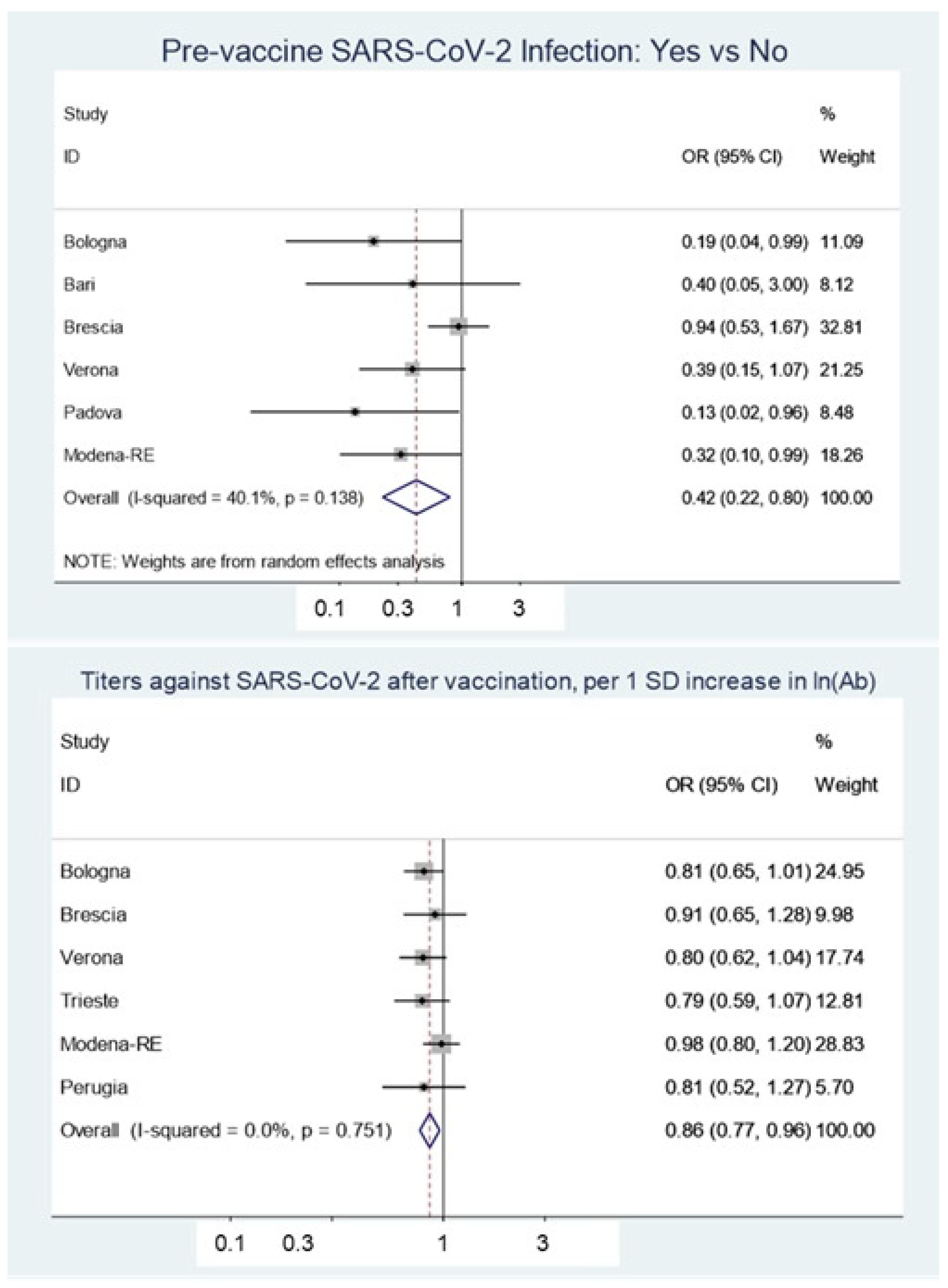
| Previous COVID-19 | 6 | 0.425 (0.225–0.80) | 0.008 | 40.1% (p = 0.138) |
| Ln (Antibody titre) (+1 sd) | 6 | 0.86 (0.775–0.96) | 0.007 | 0% (p = 0.751) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Porru, S.; Monaco, M.G.L.; Spiteri, G.; Carta, A.; Pezzani, M.D.; Lippi, G.; Gibellini, D.; Tacconelli, E.; Dalla Vecchia, I.; Sala, E.; et al. SARS-CoV-2 Breakthrough Infections: Incidence and Risk Factors in a Large European Multicentric Cohort of Health Workers. Vaccines 2022, 10, 1193. https://doi.org/10.3390/vaccines10081193
Porru S, Monaco MGL, Spiteri G, Carta A, Pezzani MD, Lippi G, Gibellini D, Tacconelli E, Dalla Vecchia I, Sala E, et al. SARS-CoV-2 Breakthrough Infections: Incidence and Risk Factors in a Large European Multicentric Cohort of Health Workers. Vaccines. 2022; 10(8):1193. https://doi.org/10.3390/vaccines10081193
Chicago/Turabian StylePorru, Stefano, Maria Grazia Lourdes Monaco, Gianluca Spiteri, Angela Carta, Maria Diletta Pezzani, Giuseppe Lippi, Davide Gibellini, Evelina Tacconelli, Ilaria Dalla Vecchia, Emma Sala, and et al. 2022. "SARS-CoV-2 Breakthrough Infections: Incidence and Risk Factors in a Large European Multicentric Cohort of Health Workers" Vaccines 10, no. 8: 1193. https://doi.org/10.3390/vaccines10081193
APA StylePorru, S., Monaco, M. G. L., Spiteri, G., Carta, A., Pezzani, M. D., Lippi, G., Gibellini, D., Tacconelli, E., Dalla Vecchia, I., Sala, E., Sansone, E., De Palma, G., Bonfanti, C., Lombardo, M., Terlenghi, L., Pira, E., Mansour, I., Coggiola, M., Ciocan, C., ... Verlato, G. (2022). SARS-CoV-2 Breakthrough Infections: Incidence and Risk Factors in a Large European Multicentric Cohort of Health Workers. Vaccines, 10(8), 1193. https://doi.org/10.3390/vaccines10081193




















