Willingness to Accept the COVID-19 Vaccine and Related Factors among Indian Adults: A Cross-Sectional Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Participants and Design
2.2. Definitions and Measures
2.2.1. Vaccine Intentions
2.2.2. Vaccine Attitudes
2.2.3. Predictor Variables
2.3. Statistical Analysis
3. Results
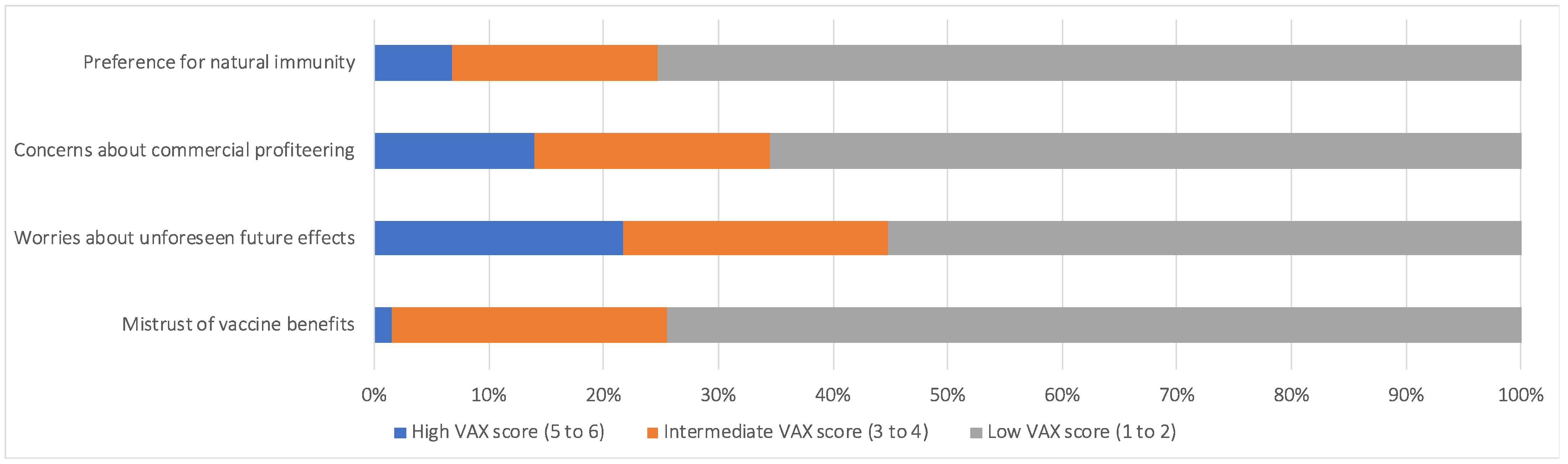
3.1. Perception of Vaccines
3.2. Type of COVID-19 Vaccine Preferred
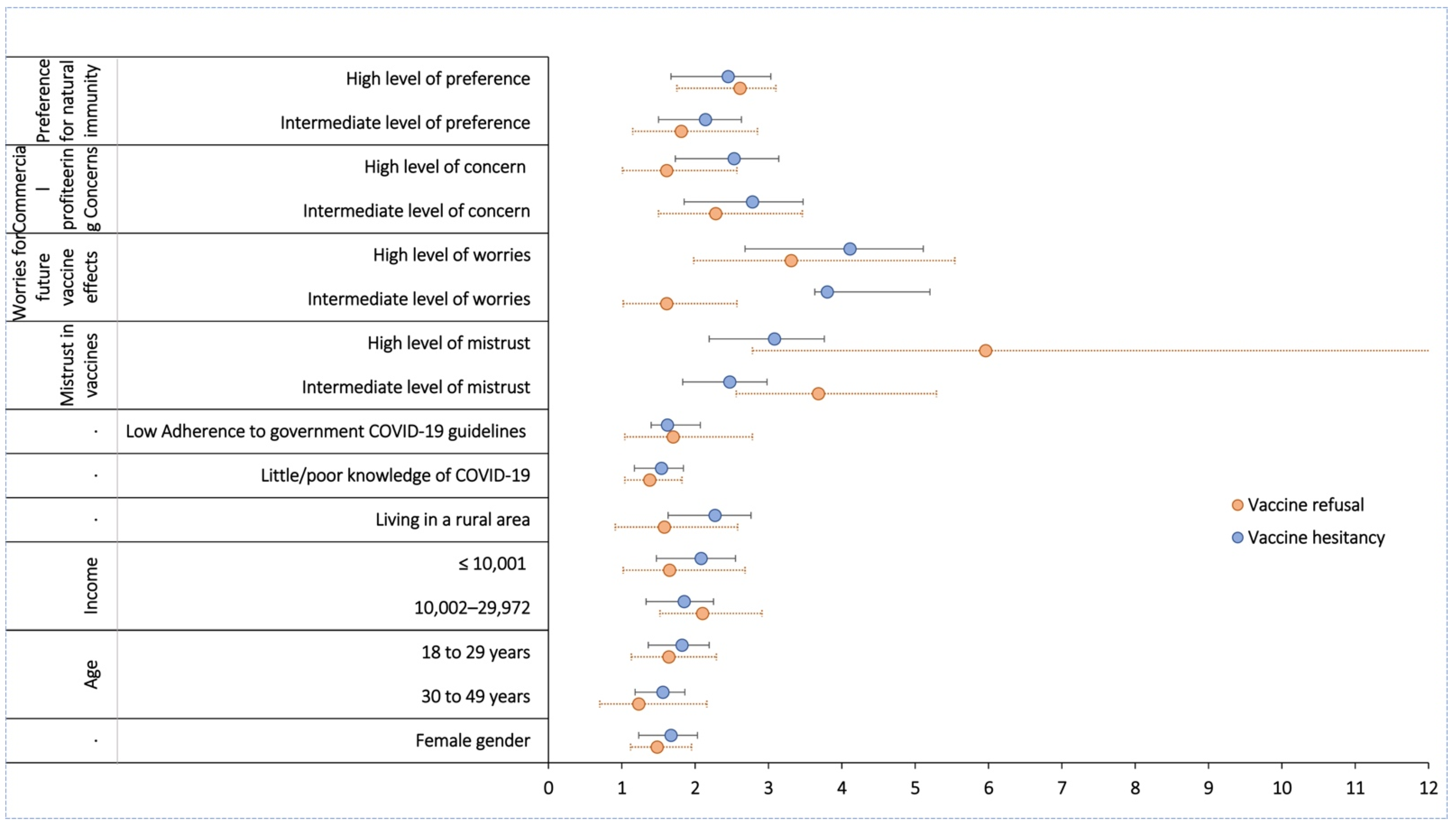
3.3. Predictors of Refusal to Vaccinate against COVID-19
3.4. Predictors of Hesitancy to Vaccinate against COVID-19
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lu, H.; Stratton, C.W.; Tang, Y.-W. Outbreak of Pneumonia of Unknown Etiology in Wuhan, China: The Mystery and the Miracle. J. Med. Virol. 2020, 92, 401–402. [Google Scholar] [CrossRef]
- COVID-19 Coronavirus Pandemic. Available online: https://www.worldometers.info/coronavirus/ (accessed on 19 March 2021).
- Nussbaumer-Streit, B.; Mayr, V.; Dobrescu, A.I.; Chapman, A.; Persad, E.; Klerings, I.; Wagner, G.; Siebert, U.; Christof, C.; Zachariah, C.; et al. Quarantine Alone or in Combination with Other Public Health Measures to Control COVID-19: A Rapid Review. Cochrane Database Syst. Rev. 2020, 4, CD013574. [Google Scholar] [CrossRef]
- Ayouni, I.; Maatoug, J.; Dhouib, W.; Zammit, N.; Fredj, S.B.; Ghammam, R.; Ghannem, H. Effective Public Health Measures to Mitigate the Spread of COVID-19: A Systematic Review. BMC Public Health 2021, 21, 1015. [Google Scholar] [CrossRef] [PubMed]
- Talic, S.; Shah, S.; Wild, H.; Gasevic, D.; Maharaj, A.; Ademi, Z.; Li, X.; Xu, W.; Mesa-Eguiagaray, I.; Rostron, J.; et al. Effectiveness of Public Health Measures in Reducing the Incidence of COVID-19, SARS-CoV-2 Transmission, and COVID-19 Mortality: Systematic Review and Meta-Analysis. BMJ 2021, 375, e068302. [Google Scholar] [CrossRef] [PubMed]
- Randolph, H.E.; Barreiro, L.B. Herd Immunity: Understanding COVID-19. Immunity 2020, 52, 737–741. [Google Scholar] [CrossRef] [PubMed]
- AstraZeneca. AstraZeneca and Oxford University Announce Landmark Agreement for COVID-19 Vaccine. 2020. Available online: https://www.astrazeneca.com/media-centre/press-releases/2020/astrazeneca-and-oxford-university-announce-landmark-agreement-for-covid-19-vaccine.html#! (accessed on 7 May 2021).
- Ministry of Health and Family Welfare. Government of India. Available online: https://main.mohfw.gov.in/ (accessed on 19 March 2021).
- D’Souza, G.; Dowdy, D. What Is Herd Immunity and How Do We Achive It. Available online: https://publichealth.jhu.edu/2020/what-is-herd-immunity-and-how-can-we-achieve-it-with-covid-19 (accessed on 18 April 2021).
- India’s Unusual Covid Vaccine Problem: Shots in Abundance, But Few Takers. Available online: https://www.hindustantimes.com/india-news/indias-unusual-covid-vaccine-problem-shots-in-abundance-but-few-takers-101611724716011.html (accessed on 18 April 2021).
- Neumann-Böhme, S.; Varghese, N.E.; Sabat, I.; Barros, P.P.; Brouwer, W.; van Exel, J.; Schreyögg, J.; Stargardt, T. Once We Have It, Will We Use It? A European Survey on Willingness to Be Vaccinated against COVID-19. Eur. J. Health Econ. 2020, 21, 977–982. [Google Scholar] [CrossRef] [PubMed]
- Rhodes, A.; Hoq, M.; Measey, M.-A.; Danchin, M. Intention to Vaccinate against COVID-19 in Australia. Lancet Infect. Dis. 2020, 21, e110. [Google Scholar] [CrossRef]
- Williams, L.; Gallant, A.J.; Rasmussen, S.; Brown Nicholls, L.A.; Cogan, N.; Deakin, K.; Young, D.; Flowers, P. Towards Intervention Development to Increase the Uptake of COVID-19 Vaccination among Those at High Risk: Outlining Evidence-based and Theoretically Informed Future Intervention Content. Br. J. Health Psychol. 2020, 25, 1039–1054. [Google Scholar] [CrossRef]
- Sharma, R. Over 23 Lakhs Covid-19 Vaccine Doses Wasted in India: Why It Happened and How It Can Be Fixed. Available online: https://www.news18.com/news/india/over-23-lakhs-covid-19-vaccine-doses-wasted-in-india-why-it-happened-and-how-it-can-be-fixed-3549464.html (accessed on 19 March 2021).
- Peretti-Watel, P.; Seror, V.; Cortaredona, S.; Launay, O.; Raude, J.; Verger, P.; Fressard, L.; Beck, F.; Legleye, S.; L’Haridon, O.; et al. A Future Vaccination Campaign against COVID-19 at Risk of Vaccine Hesitancy and Politicisation. Lancet Infect. Dis. 2020, 20, 769–770. [Google Scholar] [CrossRef]
- Thunstrom, L.; Ashworth, M.; Finnoff, D.; Newbold, S. Hesitancy Towards a COVID-19 Vaccine and Prospects for Herd Immunity. SSRN Electron. J. 2020. [Google Scholar] [CrossRef]
- Martin, L.R.; Petrie, K.J. Understanding the Dimensions of Anti-Vaccination Attitudes: The Vaccination Attitudes Examination (VAX) Scale. Ann. Behav. Med. 2017, 51, 652–660. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.A. Coronavirus Anxiety Scale: A Brief Mental Health Screener for COVID-19 Related Anxiety. Death Stud. 2020, 44, 393–401. [Google Scholar] [CrossRef] [PubMed]
- Zhong, B.-L.; Luo, W.; Li, H.-M.; Zhang, Q.-Q.; Liu, X.-G.; Li, W.-T.; Li, Y. Knowledge, Attitudes, and Practices towards COVID-19 among Chinese Residents during the Rapid Rise Period of the COVID-19 Outbreak: A Quick Online Cross-Sectional Survey. Int. J. Biol. Sci. 2020, 16, 1745–1752. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.; Kang, B.-A.; You, M. Knowledge, Attitudes, and Practices (KAP) toward COVID-19: A Cross-Sectional Study in South Korea. BMC Public Health 2021, 21, 295. [Google Scholar] [CrossRef] [PubMed]
- Clements, J.M. Knowledge and Behaviors Toward COVID-19 among US Residents during the Early Days of the Pandemic: Cross-Sectional Online Questionnaire. JMIR Public Health Surveill. 2020, 6, e19161. [Google Scholar] [CrossRef]
- Kang, H. The Prevention and Handling of the Missing Data. Korean J. Anesthesiol. 2013, 64, 402–406. [Google Scholar] [CrossRef]
- De Figueiredo, A.; Simas, C.; Karafillakis, E.; Paterson, P.; Larson, H.J. Mapping Global Trends in Vaccine Confidence and Investigating Barriers to Vaccine Uptake: A Large-Scale Retrospective Temporal Modelling Study. Lancet 2020, 396, 898–908. [Google Scholar] [CrossRef]
- Robinson, E.; Jones, A.; Lesser, I.; Daly, M. International Estimates of Intended Uptake and Refusal of COVID-19 Vaccines: A Rapid Systematic Review and Meta-Analysis of Large Nationally Representative Samples. Vaccine 2021, 39, 2024–2034. [Google Scholar] [CrossRef]
- Lazarus, J.V.; Ratzan, S.C.; Palayew, A.; Gostin, L.O.; Larson, H.J.; Rabin, K.; Kimball, S.; El-Mohandes, A. A Global Survey of Potential Acceptance of a COVID-19 Vaccine. Nat. Med. 2021, 27, 225–228. [Google Scholar] [CrossRef]
- Parthasarathi, A.; Puvvada, R.K.; Basappa, S.; Krishna, M.T.; Pa, M. Global Intention of the General Public to Undergo COVID-19 Vaccination: Time Trends and Risk Factors for Vaccine Refusal, a Systematic Review and Meta-Analysis. Wellcome Open Res. 2022, 7, 17. [Google Scholar] [CrossRef]
- Patwary, M.M.; Alam, M.A.; Bardhan, M.; Disha, A.S.; Haque, M.Z.; Billah, S.M.; Kabir, M.P.; Browning, M.H.E.M.; Rahman, M.M.; Parsa, A.D.; et al. COVID-19 Vaccine Acceptance among Low- and Lower-Middle-Income Countries: A Rapid Systematic Review and Meta-Analysis. Vaccines 2022, 10, 427. [Google Scholar] [CrossRef] [PubMed]
- Dangerfeild, K. Why Women Are Bearing the Brunt of COVID-19 Vaccine Side Effects. Available online: https://globalnews.ca/news/7762679/covid-vaccine-side-effects-women/ (accessed on 18 April 2021).
- Stock, N. Some People Are Reporting Abnormal Periods after a COVID-19 Vaccine. U. of I. Professor Is Looking for Answers. Chic. Trib. 2021. Available online: https://www.chicagotribune.com/coronavirus/vaccine/ct-vaccine-side-effects-period-menstruation-20210420-i4fy7a7tnnbchi654z3o64a7my-story.html/ (accessed on 5 May 2021).
- Schmid, P.; MacDonald, N.E.; Habersaat, K.; Butler, R. Commentary to: How to Respond to Vocal Vaccine Deniers in Public. Vaccine 2018, 36, 196–198. [Google Scholar] [CrossRef] [PubMed]
- Wise, J. COVID-19: European Countries Suspend Use of Oxford-AstraZeneca Vaccine after Reports of Blood Clots. BMJ 2021, 372, n699. [Google Scholar] [CrossRef] [PubMed]
- Hippisley-Cox, J.; Patone, M.; Mei, X.W.; Saatci, D.; Dixon, S.; Khunti, K.; Zaccardi, F.; Watkinson, P.; Shankar-Hari, M.; Doidge, J.; et al. Risk of Thrombocytopenia and Thromboembolism after COVID-19 Vaccination and SARS-CoV-2 Positive Testing: Self-Controlled Case Series Study. BMJ 2021, 374, n1931. [Google Scholar] [CrossRef]
- Opinion: 7 Former FDA Commissioners: The Trump Administration Is Undermining the Credibility of the FDA. Available online: https://www.washingtonpost.com/opinions/2020/09/29/former-fda-commissioners-coronavirus-vaccine-trump/ (accessed on 19 March 2021).
- Burki, T.K. The Russian Vaccine for COVID-19. Lancet Respir. Med. 2020, 8, e85–e86. [Google Scholar] [CrossRef]
- Loomba, S.; de Figueiredo, A.; Piatek, S.J.; de Graaf, K.; Larson, H.J. Measuring the Impact of COVID-19 Vaccine Misinformation on Vaccination Intent in the UK and USA. Nat. Hum. Behav. 2021, 5, 337–348. [Google Scholar] [CrossRef]
- Khan, N. Trends in COVID-19 Misinformation in India. Available online: https://www.ha-asia.com/trends-in-covid-19-misinformation-in-india/ (accessed on 19 March 2021).
- Abena, P.M.; Decloedt, E.H.; Bottieau, E.; Suleman, F.; Adejumo, P.; Sam-Agudu, N.A.; Muyembe TamFum, J.-J.; Seydi, M.; Eholie, S.P.; Mills, E.J.; et al. Chloroquine and Hydroxychloroquine for the Prevention or Treatment of COVID-19 in Africa: Caution for Inappropriate Off-Label Use in Healthcare Settings. Am. J. Trop. Med. Hyg. 2020, 102, 1184–1188. [Google Scholar] [CrossRef]
- Schellack, N.; Strydom, M.; Pepper, M.S.; Herd, C.L.; Hendricks, C.L.; Bronkhorst, E.; Meyer, J.C.; Padayachee, N.; Bangalee, V.; Truter, I.; et al. Social Media and COVID-19-Perceptions and Public Deceptions of Ivermectin, Colchicine and Hydroxychloroquine: Lessons for Future Pandemics. Antibiotics 2022, 11, 445. [Google Scholar] [CrossRef]
- Dutta, S.; Kaur, R.J.; Bhardwaj, P.; Ambwani, S.; Godman, B.; Jha, P.A.; Sukhija, S.; Venkatesh, S.S.; Lugova, H.; Islam, S.; et al. Demand of COVID-19 Medicines without Prescription among Community Pharmacies in Jodhpur, India: Findings and Implications. J. Fam. Med. Prim. Care 2022, 11, 503–511. [Google Scholar] [CrossRef]
- Paul, E.; Steptoe, A.; Fancourt, D. Attitudes towards Vaccines and Intention to Vaccinate against COVID-19: Implications for Public Health Communications. Lancet Reg. Health Eur. 2021, 1, 100012. [Google Scholar] [CrossRef]
- Chen, M.; Li, Y.; Chen, J.; Wen, Z.; Feng, F.; Zou, H.; Fu, C.; Chen, L.; Shu, Y.; Sun, C. An Online Survey of the Attitude and Willingness of Chinese Adults to Receive COVID-19 Vaccination. Hum. Vaccines Immunother. 2021, 17, 2279–2288. [Google Scholar] [CrossRef] [PubMed]
- Craig, B.M. United States COVID-19 Vaccination Preferences (CVP): 2020 Hindsight. Patient-Patient-Cent. Outcomes Res. 2021, 14, 309–318. [Google Scholar] [CrossRef] [PubMed]
- Verger, P.; Scronias, D.; Dauby, N.; Adedzi, K.A.; Gobert, C.; Bergeat, M.; Gagneur, A.; Dubé, E. Attitudes of Healthcare Workers towards COVID-19 Vaccination: A Survey in France and French-Speaking Parts of Belgium and Canada, 2020. Eurosurveillance 2021, 26, 2002047. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Li, X.; Su, X.; Xiao, T.; Wang, Y.; Hu, P.; Li, H.; Guan, J.; Tian, H.; Wang, P.; et al. A Study on Willingness and Influencing Factors to Receive COVID-19 Vaccination among Qingdao Residents. Hum. Vaccines Immunother. 2021, 17, 408–413. [Google Scholar] [CrossRef]
- Rose, J. The Mortal Coil of Covid-19, Fake News, and Negative Epistemic Postdigital Inculcation. Postdigital Sci. Educ. 2020, 2, 812–829. [Google Scholar] [CrossRef]
- Neil, S.J.D.; Campbell, E.M. Fake Science: XMRV, COVID-19, and the Toxic Legacy of Dr. Judy Mikovits. AIDS Res. Hum. Retrovir. 2020, 36, 545–549. [Google Scholar] [CrossRef]
- Carrion-Alvarez, D.; Tijerina-Salina, P.X. Fake News in COVID-19: A Perspective. Health Promot. Perspect. 2020, 10, 290–291. [Google Scholar] [CrossRef]
- Siegrist, M.; Zingg, A. The Role of Public Trust during Pandemics: Implications for Crisis Communication. Eur. Psychol. 2014, 19, 23–32. [Google Scholar] [CrossRef]
- Abedin, M.; Islam, M.A.; Rahman, F.N.; Reza, H.M.; Hossain, M.Z.; Hossain, M.A.; Arefin, A.; Hossain, A. Willingness to vaccinate against COVID-19 among Bangladeshi adults: Understanding the strategies to optimize vaccination coverage. PLoS ONE 2021, 16, e0250495. [Google Scholar] [CrossRef]
- Acheampong, T.; Akorsikumah, E.A.; Osae-Kwapong, J.; Khalid, M.; Appiah, A.; Amuasi, J.H. Examining Vaccine Hesitancy in Sub-Saharan Africa: A Survey of the Knowledge and Attitudes among Adults to Receive COVID-19 Vaccines in Ghana. Vaccines 2021, 9, 814. [Google Scholar] [CrossRef]
- Adebisi, Y.A.; Alaran, A.J.; Bolarinwa, O.A.; Akande-Sholabi, W.; Lucero-Prisno, D.E. When it is available, will we take it? Social media users’ perception of hypothetical COVID-19 vaccine in Nigeria. Pan Afr. Med. J. 2021, 38, 230. [Google Scholar] [CrossRef]
- Ahmed, M.A.M.; Colebunders, R.; Gele, A.A.; Farah, A.A.; Osman, S.; Guled, I.A.; Abdullahi, A.A.; Hussein, A.M.; Ali, A.M.; Siewe Fodjo, J.N. COVID-19 Vaccine Acceptability and Adherence to Preventive Measures in Somalia: Results of an Online Survey. Vaccines 2021, 9, 543. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, T.F.; Ahmed, A.; Ahmed, S.; Ahmed, H.U. Understanding COVID-19 vaccine acceptance in Pakistan: An echo of previous immunizations or prospect of change? Expert Rev. Vaccines 2021, 20, 1185–1193. [Google Scholar] [CrossRef] [PubMed]
- Akiful Haque, M.M.; Rahman, M.L.; Hossian, M.; Matin, K.F.; Nabi, M.H.; Saha, S.; Hasan, M.; Manna, R.M.; Barsha, S.Y.; Hasan, S.M.R.; et al. Acceptance of COVID-19 vaccine and its determinants: Evidence from a large sample study in Bangladesh. Heliyon 2021, 7, e07376. [Google Scholar] [CrossRef]
- Al-Qerem, W.A.; Jarab, A.S. COVID-19 Vaccination Acceptance and Its Associated Factors among a Middle Eastern Population. Front. Public Health 2021, 9, 632914. [Google Scholar] [CrossRef]
- Udoakang, A.J.; Djomkam Zune, A.L.; Tapela, K.; Owoicho, O.; Fagbohun, I.K.; Anyigba, C.A.; Lowe, M.; Nganyewo, N.N.; Keneme, B.; Olisaka, F.N.; et al. Knowledge, attitude and perception of West Africans towards COVID-19: A survey to inform public health intervention. BMC Public Health 2022, 22, 445. [Google Scholar] [CrossRef] [PubMed]
- Arshad, M.S.; Hussain, I.; Mahmood, T.; Hayat, K.; Majeed, A.; Imran, I.; Saeed, H.; Iqbal, M.O.; Uzair, M.; Rehman, A.U.; et al. A National Survey to Assess the COVID-19 Vaccine-Related Conspiracy Beliefs, Acceptability, Preference, and Willingness to Pay among the General Population of Pakistan. Vaccines 2021, 9, 720. [Google Scholar] [CrossRef]
- Bongomin, F.; Olum, R.; Andia-Biraro, I.; Nakwagala, F.N.; Hassan, K.H.; Nassozi, D.R.; Kaddumukasa, M.; Byakika-Kibwika, P.; Kiguli, S.; Kirenga, B.J. COVID-19 vaccine acceptance among high-risk populations in Uganda. Ther. Adv. Infect. Dis. 2021, 8, 20499361211024376. [Google Scholar] [CrossRef]
- Bono, S.A.; Faria de Moura Villela, E.; Siau, C.S.; Chen, W.S.; Pengpid, S.; Hasan, M.T.; Sessou, P.; Ditekemena, J.D.; Amodan, B.O.; Hosseinipour, M.C.; et al. Factors Affecting COVID-19 Vaccine Acceptance: An International Survey among Low- and Middle-Income Countries. Vaccines 2021, 9, 515. [Google Scholar] [CrossRef]
- Chaudhary, F.A.; Ahmad, B.; Khalid, M.D.; Fazal, A.; Javaid, M.M.; Butt, D.Q. Factors influencing COVID-19 vaccine hesitancy and acceptance among the Pakistani population. Hum. Vaccines Immunother. 2021, 17, 3365–3370. [Google Scholar] [CrossRef]
- Moore, D.C.B.C.; Nehab, M.F.; Camacho, K.G.; Reis, A.T.; de Fátima Junqueira-Marinho, M.; Abramov, D.M.; de Azevedo, Z.M.A.; de Menezes, L.A.; dos Santos Salú, M.; da Silva Figueiredo, C.E.; et al. Low COVID-19 vaccine hesitancy in Brazil. Vaccine 2021, 39, 6262–6268. [Google Scholar] [CrossRef] [PubMed]
- Dambadarjaa, D.; Altankhuyag, G.-E.; Chandaga, U.; Khuyag, S.-O.; Batkhorol, B.; Khaidav, N.; Dulamsuren, O.; Gombodorj, N.; Dorjsuren, A.; Singh, P.; et al. Factors Associated with COVID-19 Vaccine Hesitancy in Mongolia: A Web-Based Cross-Sectional Survey. Int. J. Environ. Res. Public Health 2021, 18, 12903. [Google Scholar] [CrossRef] [PubMed]
- Harapan, H.; Wagner, A.L.; Yufika, A.; Winardi, W.; Anwar, S.; Gan, A.K.; Setiawan, A.M.; Rajamoorthy, Y.; Sofyan, H.; Mudatsir, M. Acceptance of a COVID-19 Vaccine in Southeast Asia: A Cross-Sectional Study in Indonesia. Front. Public Health 2020, 8, 381. [Google Scholar] [CrossRef] [PubMed]
- Amo-Adjei, J.; Nurzhynska, A.; Essuman, R.; Lohiniva, A.-L. Trust and willingness towards COVID-19 vaccine uptake: A mixed-method study in Ghana, 2021. Arch. Public Health 2022, 80, 64. [Google Scholar] [CrossRef]
- Shareef, L.G.; Fawzi Al-Hussainy, A.; Majeed Hameed, S. COVID-19 vaccination hesitancy among Iraqi general population between beliefs and barriers: An observational study. F1000Research 2022, 11, 334. [Google Scholar] [CrossRef]
- Tlale, L.B.; Gabaitiri, L.; Totolo, L.K.; Smith, G.; Puswane-Katse, O.; Ramonna, E.; Mothowaeng, B.; Tlhakanelo, J.; Masupe, T.; Rankgoane-Pono, G.; et al. Acceptance rate and risk perception towards the COVID-19 vaccine in Botswana. PLoS ONE 2022, 17, e0263375. [Google Scholar] [CrossRef]
- Bou Hamdan, M.; Singh, S.; Polavarapu, M.; Jordan, T.R.; Melhem, N.M. COVID-19 vaccine hesitancy among university students in Lebanon. Epidemiol. Infect. 2021, 149, e242. [Google Scholar] [CrossRef]
- Gaur, P.; Agrawat, H.; Shukla, A. COVID-19 vaccine hesitancy in patients with systemic autoimmune rheumatic disease: An interview-based survey. Rheumatol. Int. 2021, 41, 1601–1605. [Google Scholar] [CrossRef]
- Salali, G.D.; Uysal, M.S. COVID-19 vaccine hesitancy is associated with beliefs on the origin of the novel coronavirus in the UK and Turkey. Psychol. Med. 2020, 1–3. [Google Scholar] [CrossRef]
- Sallam, M.; Dababseh, D.; Eid, H.; Al-Mahzoum, K.; Al-Haidar, A.; Taim, D.; Yaseen, A.; Ababneh, N.A.; Bakri, F.G.; Mahafzah, A. High Rates of COVID-19 Vaccine Hesitancy and Its Association with Conspiracy Beliefs: A Study in Jordan and Kuwait among Other Arab Countries. Vaccines 2021, 9, 42. [Google Scholar] [CrossRef]
- Syed Alwi, S.A.R.; Rafidah, E.; Zurraini, A.; Juslina, O.; Brohi, I.B.; Lukas, S. A survey on COVID-19 vaccine acceptance and concern among Malaysians. BMC Public Health 2021, 21, 1129. [Google Scholar] [CrossRef] [PubMed]
- Solís Arce, J.S.; Warren, S.S.; Meriggi, N.F.; Scacco, A.; McMurry, N.; Voors, M.; Syunyaev, G.; Malik, A.A.; Aboutajdine, S.; Adeojo, O.; et al. COVID-19 vaccine acceptance and hesitancy in low- and middle-income countries. Nat. Med. 2021, 27, 1385–1394. [Google Scholar] [CrossRef] [PubMed]
- Wong, L.P.; Alias, H.; Wong, P.-F.; Lee, H.Y.; AbuBakar, S. The use of the health belief model to assess predictors of intent to receive the COVID-19 vaccine and willingness to pay. Hum. Vaccines Immunother. 2020, 16, 2204–2214. [Google Scholar] [CrossRef] [PubMed]
- Qunaibi, E.A.; Helmy, M.; Basheti, I.; Sultan, I. A high rate of COVID-19 vaccine hesitancy in a large-scale survey on Arabs. Elife 2021, 10, e68038. [Google Scholar] [CrossRef] [PubMed]

| Variables | Number | Percentage |
|---|---|---|
| Gender | ||
| Male | 945 | 59.7% |
| Female | 637 | 40.3% |
| Geographic quadrants | ||
| North | 427 | 26.4% |
| South | 538 | 34.6% |
| West | 259 | 16.4% |
| East | 358 | 22.6% |
| Age (years) | ||
| 18–24 | 461 | 29.1% |
| 25–44 | 622 | 39.3% |
| 45–64 | 437 | 27.6% |
| 65+ | 62 | 3.9% |
| Educational attainment | ||
| Postgraduate education | 431 | 27.2% |
| Undergraduate education | 573 | 36.2% |
| Secondary school education | 291 | 18.4% |
| Not willing to disclose | 287 | 18.1% |
| Approximate family income | ||
| ≥199,862 | 151 | 9.5% |
| 99,931–199,861 | 229 | 14.5% |
| 74,756–99,930 | 346 | 21.9% |
| 49,962–74,755 | 277 | 17.5% |
| 29,973–49,961 | 64 | 4.0% |
| 10,002–29,972 | 16 | 1.0% |
| ≤10,001 | 83 | 5.2% |
| Not willing to disclose | 416 | 26.3% |
| Living area | ||
| Urban | 1087 | 68.7% |
| Rural | 495 | 31.3% |
| Knowledge of COVID-19 * | ||
| Good knowledge | 1211 | 76.5% |
| Poor knowledge | 371 | 23.5% |
| Adherence to government COVID-19 guidelines | ||
| Very much following | 964 | 60.9% |
| Following less | 618 | 39.1% |
| Have had COVID-19 | ||
| Have not had COVID-19 | 1017 | 64.3% |
| Have had COVID-19 | 565 | 35.7% |
| Coronavirus Anxiety Symptoms (CAS) ** | ||
| Ever ≥ 1 CAS symptom | 201 | 12.7% |
| Never CAS symptoms | 1381 | 87.3% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Parthasarathi, A.; Puvvada, R.K.; Shankar, M.; Siddaiah, J.B.; Ganguly, K.; Upadhyay, S.; Mahesh, P.A. Willingness to Accept the COVID-19 Vaccine and Related Factors among Indian Adults: A Cross-Sectional Study. Vaccines 2022, 10, 1095. https://doi.org/10.3390/vaccines10071095
Parthasarathi A, Puvvada RK, Shankar M, Siddaiah JB, Ganguly K, Upadhyay S, Mahesh PA. Willingness to Accept the COVID-19 Vaccine and Related Factors among Indian Adults: A Cross-Sectional Study. Vaccines. 2022; 10(7):1095. https://doi.org/10.3390/vaccines10071095
Chicago/Turabian StyleParthasarathi, Ashwaghosha, Rahul Krishna Puvvada, Malavika Shankar, Jayaraj Biligere Siddaiah, Koustav Ganguly, Swapna Upadhyay, and Padukudru Anand Mahesh. 2022. "Willingness to Accept the COVID-19 Vaccine and Related Factors among Indian Adults: A Cross-Sectional Study" Vaccines 10, no. 7: 1095. https://doi.org/10.3390/vaccines10071095
APA StyleParthasarathi, A., Puvvada, R. K., Shankar, M., Siddaiah, J. B., Ganguly, K., Upadhyay, S., & Mahesh, P. A. (2022). Willingness to Accept the COVID-19 Vaccine and Related Factors among Indian Adults: A Cross-Sectional Study. Vaccines, 10(7), 1095. https://doi.org/10.3390/vaccines10071095









