Appraisal of COVID-19 Vaccination Acceptance in the Romanian Pregnant Population
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design and Participants
2.2. Surveys and Variables
2.3. Statistical Analysis
3. Results
3.1. Background and Obstetrical Characteristics
3.2. Standardized and Unstandardized Questionnaires
3.3. Factors for Acceptance
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhao, Z.-Y.; Niu, Y.; Luo, L.; Hu, Q.-Q.; Yang, T.-L.; Chu, M.-J.; Chen, Q.-P.; Lei, Z.; Rui, J.; Song, C.-L.; et al. The optimal vaccination strategy to control COVID-19: A modeling study in Wuhan City, China. Infect. Dis. Poverty 2021, 10, 140. [Google Scholar] [CrossRef]
- Ayouni, I.; Maatoug, J.; Dhouib, W.; Zammit, N.; Fredj, S.B.; Ghammam, R.; Ghannem, H. Effective public health measures to mitigate the spread of COVID-19: A systematic review. BMC Public Health 2021, 21, 1015. [Google Scholar] [CrossRef]
- Nicola, M.; Alsafi, Z.; Sohrabi, C.; Kerwan, A.; Al-Jabir, A.; Iosifidis, C.; Agha, M.; Agha, R. The socio-economic implications of the coronavirus pandemic (COVID-19): A review. Int. J. Surg. 2020, 78, 185–193. [Google Scholar] [CrossRef]
- Citu, I.M.; Citu, C.; Gorun, F.; Sas, I.; Tomescu, L.; Neamtu, R.; Motoc, A.; Gorun, O.M.; Burlea, B.; Bratosin, F.; et al. Immunogenicity Following Administration of BNT162b2 and Ad26.COV2.S COVID-19 Vaccines in the Pregnant Population during the Third Trimester. Viruses 2022, 14, 307. [Google Scholar] [CrossRef]
- Bryson, H.; Mensah, F.; Price, A.; Gold, L.; Mudiyanselage, S.B.; Kenny, B.; Dakin, P.; Bruce, T.; Noble, K.; Kemp, L.; et al. Clinical, financial and social impacts of COVID-19 and their associations with mental health for mothers and children experiencing adversity in Australia. PLoS ONE 2021, 16, e0257357. [Google Scholar] [CrossRef]
- Bogdan, I.; Citu, C.; Bratosin, F.; Malita, D.; Romosan, I.; Gurban, C.V.; Bota, A.V.; Turaiche, M.; Bratu, M.L.; Pilut, C.N.; et al. The Impact of Multiplex PCR in Diagnosing and Managing Bacterial Infections in COVID-19 Patients Self-Medicated with Antibiotics. Antibiotics 2022, 11, 437. [Google Scholar] [CrossRef]
- Garegnani, L.I.; Madrid, E.; Meza, N. Misleading clinical evidence and systematic reviews on ivermectin for COVID-19. BMJ Evid. Based Med. 2021, 27, 156–158. [Google Scholar] [CrossRef]
- Tirnea, L.; Bratosin, F.; Vidican, I.; Cerbu, B.; Turaiche, M.; Timircan, M.; Margan, M.-M.; Marincu, I. The Efficacy of Convalescent Plasma Use in Critically Ill COVID-19 Patients. Medicina 2021, 57, 257. [Google Scholar] [CrossRef]
- Han, F.; Liu, Y.; Mo, M.; Chen, J.; Wang, C.; Yang, Y.; Wu, J. Current treatment strategies for COVID-19 (Review). Mol. Med. Rep. 2021, 24, 858. [Google Scholar] [CrossRef]
- Yang, E.A.L.; Chai, P.; Yu, J.; Fan, X. Effects of cancer on patients with COVID-19: A systematic review and meta-analysis of 63,019 participants. Cancer Biol. Med. 2021, 18, 298–307. [Google Scholar] [CrossRef]
- Marincu, I.; Bratosin, F.; Vidican, I.; Bostanaru, A.-C.; Frent, S.; Cerbu, B.; Turaiche, M.; Tirnea, L.; Timircan, M. Predictive Value of Comorbid Conditions for COVID-19 Mortality. J. Clin. Med. 2021, 10, 2652. [Google Scholar] [CrossRef] [PubMed]
- Cerbu, B.; Pantea, S.; Bratosin, F.; Vidican, I.; Turaiche, M.; Frent, S.; Borsi, E.; Marincu, I. Liver Impairment and Hematological Changes in Patients with Chronic Hepatitis C and COVID-19: A Retrospective Study after One Year of Pandemic. Medicina 2021, 57, 597. [Google Scholar] [CrossRef] [PubMed]
- Citu, I.M.; Citu, C.; Margan, M.-M.; Craina, M.; Neamtu, R.; Gorun, O.M.; Burlea, B.; Bratosin, F.; Rosca, O.; Grigoras, M.L.; et al. Calcium, Magnesium, and Zinc Supplementation during Pregnancy: The Additive Value of Micronutrients on Maternal Immune Response after SARS-CoV-2 Infection. Nutrients 2022, 14, 1445. [Google Scholar] [CrossRef]
- Du, Z.; Wang, L.; Pandey, A.; Lim, W.W.; Chinazzi, M.; Piontti, A.P.Y.; Lau, E.H.Y.; Wu, P.; Malani, A.; Cobey, S.; et al. Modeling comparative cost-effectiveness of SARS-CoV-2 vaccine dose fractionation in India. Nat. Med. 2022, 28, 934–938. [Google Scholar] [CrossRef]
- Heinz, F.X.; Stiasny, K. Distinguishing features of current COVID-19 vaccines: Knowns and unknowns of antigen presentation and modes of action. NPJ Vaccines 2021, 6, 104. [Google Scholar] [CrossRef] [PubMed]
- Citu, I.M.; Citu, C.; Gorun, F.; Motoc, A.; Gorun, O.M.; Burlea, B.; Bratosin, F.; Tudorache, E.; Margan, M.-M.; Hosin, S.; et al. Determinants of COVID-19 Vaccination Hesitancy among Romanian Pregnant Women. Vaccines 2022, 10, 275. [Google Scholar] [CrossRef]
- Honora, A.; Wang, K.-Y.; Chih, W.-H. How does information overload about COVID-19 vaccines influence individuals’ vaccination intentions? The roles of cyberchondria, perceived risk, and vaccine skepticism. Comput. Hum. Behav. 2022, 130, 107176. [Google Scholar] [CrossRef]
- Citu, I.M.; Citu, C.; Gorun, F.; Sas, I.; Bratosin, F.; Motoc, A.; Burlea, B.; Rosca, O.; Malita, D.; Gorun, O.M. The Risk of Spontaneous Abortion Does Not Increase Following First Trimester mRNA COVID-19 Vaccination. J. Clin. Med. 2022, 11, 1698. [Google Scholar] [CrossRef]
- Tolia, V.; Singh, R.R.; Deshpande, S.; Dave, A.; Rathod, R.M. Understanding Factors to COVID-19 Vaccine Adoption in Gujarat, India. Int. J. Environ. Res. Public Health 2022, 19, 2707. [Google Scholar] [CrossRef]
- Ritchie, H.; Mathieu, E.; Rodés-Guirao, L.; Appel, C.; Giattino, C.; Ortiz-Ospina, E.; Hasell, J.; Macdonald, B.; Beltekian, D.; Roser, M. Coronavirus Pandemic (COVID-19). Published Online at OurWorldInData.org. 2020. Available online: https://ourworldindata.org/coronavirus (accessed on 20 April 2022).
- Litaker, J.R.; Tamez, N.; Bray, C.L.; Durkalski, W.; Taylor, R. Sociodemographic Factors Associated with Vaccine Hesitancy in Central Texas Immediately Prior to COVID-19 Vaccine Availability. Int. J. Environ. Res. Public Health 2021, 19, 368. [Google Scholar] [CrossRef]
- Fekadu, G.; Bekele, F.; Tolossa, T.; Fetensa, G.; Turi, E.; Getachew, M.; Abdisa, E.; Assefa, L.; Afeta, M.; Demisew, W.; et al. Impact of COVID-19 pandemic on chronic diseases care follow-up and current perspectives in low resource settings: A narrative review. Int. J. Physiol. Pathophysiol. Pharm. 2021, 13, 86–93. [Google Scholar]
- Khairat, S.; Zou, B.; Adler-Milstein, J. Factors and reasons associated with low COVID-19 vaccine uptake among highly hesitant communities in the US. Am. J. Infect. Control 2022, 50, 262–267. [Google Scholar] [CrossRef] [PubMed]
- Citu, I.M.; Citu, C.; Gorun, F.; Neamtu, R.; Motoc, A.; Burlea, B.; Rosca, O.; Bratosin, F.; Hosin, S.; Manolescu, D.; et al. Using the NYHA Classification as Forecasting Tool for Hospital Readmission and Mortality in Heart Failure Patients with COVID-19. J. Clin. Med. 2022, 11, 1382. [Google Scholar] [CrossRef]
- Allen, J.D.; Feng, W.; Corlin, L.; Porteny, T.; Acevedo, A.; Schildkraut, D.; King, E.; Ladin, K.; Fu, Q.; Stopka, T.J. Why are some people reluctant to be vaccinated for COVID-19? A cross-sectional survey among U.S. Adults in May–June 2020. Prev. Med. Rep. 2021, 24, 101494. [Google Scholar] [CrossRef] [PubMed]
- Alqudeimat, Y.; Alenezi, D.; AlHajri, B.; Alfouzan, H.; Almokhaizeem, Z.; Altamimi, S.; Almansouri, W.; Alzalzalah, S.; Ziyab, A.H. Acceptance of a COVID-19 Vaccine and Its Related Determinants among the General Adult Population in Kuwait. Med. Princ. Pract. 2021, 30, 262–271. [Google Scholar] [CrossRef]
- Saied, S.M.; Saied, E.M.; Kabbash, I.A.; Abdo, S.A.E. Vaccine hesitancy: Beliefs and barriers associated with COVID-19 vaccination among Egyptian medical students. J. Med. Virol. 2021, 93, 4280–4291. [Google Scholar] [CrossRef]
- Sekizawa, Y.; Hashimoto, S.; Denda, K.; Ochi, S.; So, M. Association between COVID-19 vaccine hesitancy and generalized trust, depression, generalized anxiety, and fear of COVID-19. BMC Public Health 2022, 22, 126. [Google Scholar] [CrossRef]
- Timircan, M.; Bratosin, F.; Vidican, I.; Suciu, O.; Turaiche, M.; Bota, A.V.; Mitrescu, S.; Marincu, I. Coping Strategies and Health-Related Quality of Life in Pregnant Women with SARS-CoV-2 Infection. Medicina 2021, 57, 1113. [Google Scholar] [CrossRef]
- Galanis, P.; Vraka, I.; Siskou, O.; Konstantakopoulou, O.; Katsiroumpa, A.; Kaitelidou, D. Uptake of COVID-19 Vaccines among Pregnant Women: A Systematic Review and Meta-Analysis. Vaccines 2022, 10, 766. [Google Scholar] [CrossRef]
- Timircan, M.; Bratosin, F.; Vidican, I.; Suciu, O.; Tirnea, L.; Avram, V.; Marincu, I. Exploring Pregnancy Outcomes Associated with SARS-CoV-2 Infection. Medicina 2021, 57, 796. [Google Scholar] [CrossRef]
- Romanian Government. Daily Update (13/03)—Records of People Vaccinated against COVID-19. 2022. Available online: https://vaccinare-covid.gov.ro/actualizare-zilnica-13-03-evidenta-persoanelor-vaccinate-impotriva-covid-19-3/ (accessed on 12 June 2022).
- Zigmond, A.S.; Snaith, R.P. The Hospital Anxiety and Depression Scale. Acta Psychiatr. Scand. 1983, 67, 361–370. [Google Scholar] [CrossRef] [Green Version]
- Ware, J.E.; Kosinski, M.; Keller, S.D. A 12-item short-form health survey. Med. Care 1996, 34, 220–223. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Skre, I.; Friborg, O.; Elgarøy, S.; Evans, C.; Myklebust, L.H.; Lillevoll, K.; Sørgaard, K.; Hansen, V. The factor structure and psychometric properties of the Clinical Outcomes in Routine Evaluation—Outcome Measure (CORE-OM) in Norwegian clin-ical and non-clinical samples. BMC Psychiatry 2013, 13, 99. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Statista. Number of COVID-19 Vaccine Doses Administered in Europe as of 12 May 2022, by Country. 2022. Available online: https://www.statista.com/statistics/1196071/covid-19-vaccination-rate-in-europe-by-country/ (accessed on 12 June 2022).
- Lamptey, E. Overcoming barriers to COVID-19 vaccination of pregnant women. Gynecol. Obstet. Clin. Med. 2022, 2, 29–33. [Google Scholar] [CrossRef]
- Smith, D.D.; Pippen, J.L.; Adesomo, A.A.; Rood, K.M.; Landon, M.B.; Costantine, M.M. Exclusion of Pregnant Women from Clinical Trials during the Coronavirus Disease 2019 Pandemic: A Review of International Registries. Am. J. Perinatol. 2020, 37, 792–799. [Google Scholar] [CrossRef]
- Beigi, R.H.; Krubiner, C.; Jamieson, D.J.; Lyerly, A.D.; Hughes, B.; Riley, L.; Faden, R.; Karron, R. The need for inclusion of pregnant women in COVID-19 vaccine trials. Vaccine 2021, 39, 868–870. [Google Scholar] [CrossRef] [PubMed]
- Skjefte, M.; Ngirbabul, M.; Akeju, O.; Escudero, D.; Hernandez-Diaz, S.; Wyszynski, D.F.; Wu, J.W. COVID-19 vaccine acceptance among pregnant women and mothers of young children: Results of a survey in 16 countries. Eur. J. Epidemiol. 2021, 36, 197–211. [Google Scholar] [CrossRef]
- Iacobucci, G. COVID-19 and pregnancy: Vaccine hesitancy and how to overcome it. BMJ 2021, 375, n2862. [Google Scholar] [CrossRef] [PubMed]
- Kachikis, A.; Englund, J.A.; Singleton, M.; Covelli, I.; Drake, A.L.; Eckert, L.O. Short-term Reactions Among Pregnant and Lactating Individuals in the First Wave of the COVID-19 Vaccine Rollout. JAMA Netw. Open 2021, 4, e2121310. [Google Scholar] [CrossRef]
| Variables * | Vaccinated (n = 184) | Unvaccinated (n = 161) | p |
|---|---|---|---|
| Age range (years) | 0.772 | ||
| <25 | 38 (20.7%) | 35 (21.7%) | |
| 25–34 | 95 (51.6%) | 77 (47.8%) | |
| >34 | 51 (27.7%) | 49 (30.4%) | |
| Area of Residence | 0.022 | ||
| Rural | 57 (31.0%) | 69 (42.9%) | |
| Urban | 127 (69.0%) | 92 (57.1%) | |
| Relationship Status | 0.182 | ||
| Married/concubinage | 171 (92.9%) | 143 (88.8%) | |
| Single/divorced/widowed | 13 (7.1%) | 18 (11.2%) | |
| Income | 0.042 | ||
| Low | 53 (28.8%) | 67 (41.6%) | |
| Medium | 98 (53.3%) | 72 (44.7%) | |
| High | 33 (17.9%) | 22 (13.7%) | |
| Education | 0.001 | ||
| Primary education | 10 (5.4%) | 16 (9.9%) | |
| High school | 82 (44.6%) | 95 (59.0%) | |
| Higher education | 92 (50.0%) | 50 (31.1%) | |
| Occupation | 0.740 | ||
| Employed/self-employed | 153 (83.2%) | 136 (84.5%) | |
| Unemployed | 31 (16.8%) | 25 (15.5%) | |
| Behavior | 0.026 | ||
| Frequent alcohol consumption | 6 (3.3%) | 8 (5.0%) | |
| Frequent smoker | 18 (9.8%) | 29 (18.0%) | |
| COVID-19 vaccine | |||
| BNT162b2 | 163 (88.6%) | - | - |
| mRNA-1273 | 18 (9.8%) | - | - |
| Ad26.COV2.S | 3 (1.6%) | - | - |
| Variables * | Vaccinated (n = 184) | Unvaccinated (n = 161) | p |
|---|---|---|---|
| Gravidity | 0.754 | ||
| 1 | 94 (51.1%) | 88 (54.7%) | |
| 2 | 72 (39.1%) | 60 (37.3%) | |
| ≥3 | 18 (9.8%) | 13 (8.1%) | |
| Parity | 0.881 | ||
| 1 | 99 (53.8%) | 90 (55.9%) | |
| 2 | 74 (40.2%) | 63 (39.1%) | |
| ≥3 | 11 (6.0%) | 8 (5.0%) | |
| Trimester of pregnancy | 0.002 | ||
| 1 | 32 (17.4%) | 40 (24.8%) | |
| 2 | 64 (34.8%) | 73 (45.3%) | |
| 3 | 88 (47.8%) | 48 (29.8%) | |
| Pregnancy-associated complications ** | 0.362 | ||
| 0 | 147 (79.9%) | 138 (85.7%) | |
| 1 | 32 (17.4%) | 20 (12.4%) | |
| ≥ 2 | 5 (2.7%) | 3 (1.9%) | |
| Body mass index *** | 0.780 | ||
| Normal weight | 136 (73.9%) | 124 (77.0%) | |
| Overweight | 30 (16.3%) | 24 (14.9%) | |
| Obese | 18 (9.8%) | 13 (8.1%) | |
| History of pregnancy loss | 0.371 | ||
| None | 146 (79.3%) | 135 (83.9%) | |
| Medical abortion | 6 (3.3%) | 8 (5.0%) | |
| Stillbirth (<20 weeks) | 13 (7.1%) | 7 (4.3%) | |
| Miscarriage (>20 weeks) | 19 (10.3%) | 11 (6.8%) | |
| Comorbidities | |||
| Cardiovascular | 7 (3.8%) | 4 (2.5%) | 0.486 |
| Metabolic | 4 (2.2%) | 4 (2.5%) | 0.848 |
| Autoimmune | 3 (1.6%) | 1 (0.6%) | 0.382 |
| Respiratory | 9 (4.9%) | 7 (4.3%) | 0.810 |
| Other | 4 (2.2%) | 2 (1.2%) | 0.508 |
| History of depression | 6 (3.3%) | 6 (3.7%) | 0.813 |
| Variables * | Vaccinated (n = 184) | Unvaccinated (n = 161) | p |
|---|---|---|---|
| HADS | |||
| Anxiety | 7.3 ± 4.1 | 6.2 ± 4.0 | 0.012 |
| Depression | 6.4 ± 3.5 | 5.1 ± 2.9 | <0.001 |
| Total score | 12.5 ± 5.4 | 10.2 ± 4.6 | <0.001 |
| SF-12 | |||
| Physical | 56.3 ± 7.5 | 55.1 ± 7.0 | 0.127 |
| Mental | 53.8 ± 9.1 | 52.4 ± 8.8 | 0.148 |
| Total score | 55.2 ± 8.3 | 53.7 ± 7.6 | 0.082 |
| CORE-OM | |||
| CORE-W | 0.89 ± 0.58 | 0.93 ± 0.62 | 0.536 |
| CORE-P | 0.71 ± 0.57 | 0.77 ± 0.58 | 0.334 |
| CORE-F | 0.63 ± 0.53 | 0.71 ± 0.56 | 0.174 |
| CORE-R | 0.26 ± 0.14 | 0.28 ± 0.11 | 0.145 |
| Total score | 0.80 ± 0.52 | 0.84 ± 0.55 | 0.488 |
| Variables * | Vaccinated (n = 184) | Unvaccinated (n = 161) | p |
|---|---|---|---|
| Influencing factors | |||
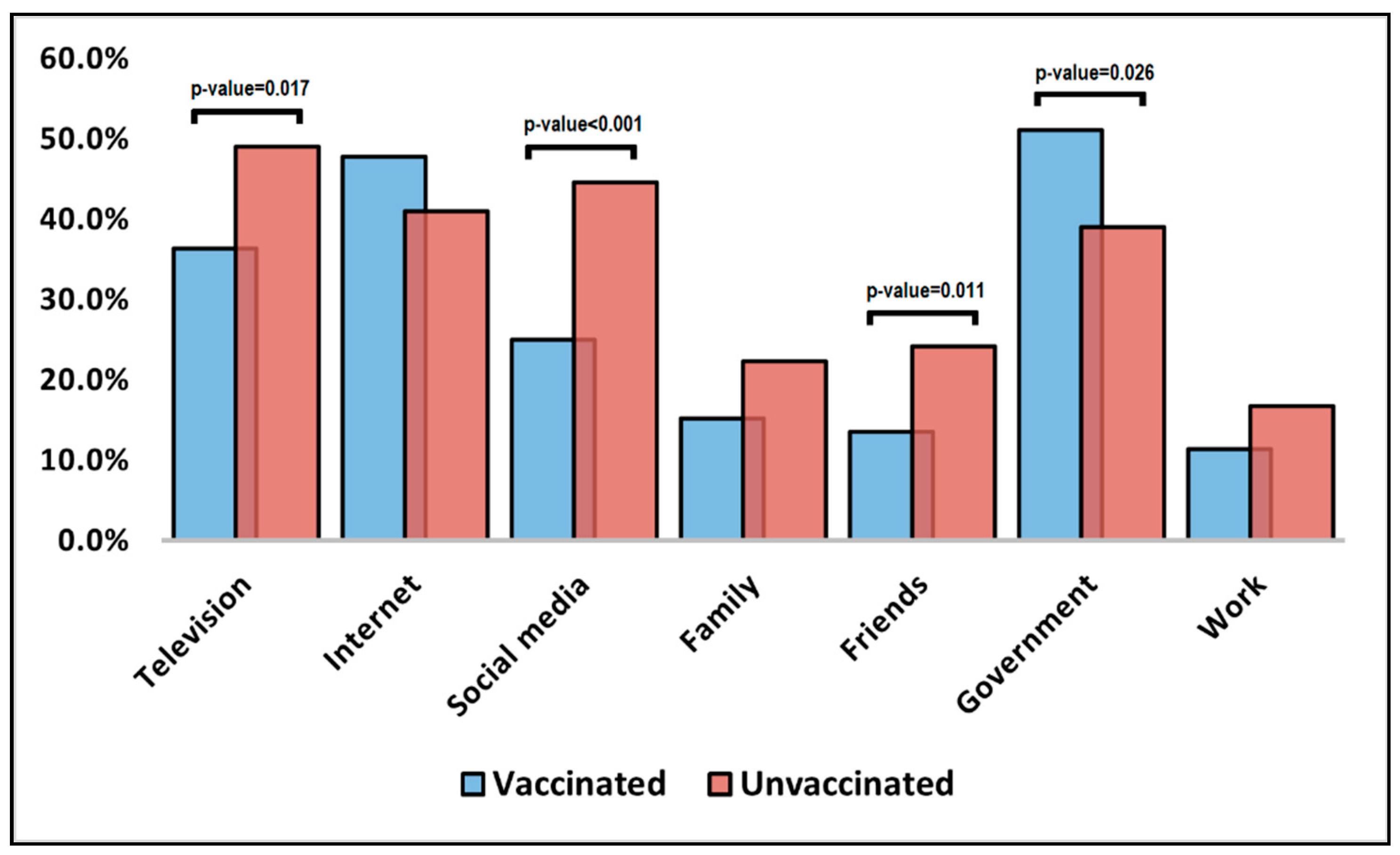
| Television | 67 (36.4%) | 79 (49.1%) | 0.017 |
| Internet | 88 (47.8%) | 66 (41.0%) | 0.202 |
| Social media | 46 (25.0%) | 72 (44.7%) | <0.001 |
| Family | 28 (15.2%) | 36 (22.4%) | 0.088 |
| Friends | 25 (13.6%) | 39 (24.2%) | 0.011 |
| Government | 94 (51.1%) | 63 (39.1%) | 0.026 |
| Work | 21 (11.4%) | 27 (16.8%) | 0.151 |
| Beliefs over factors that can prevent COVID-19 | |||
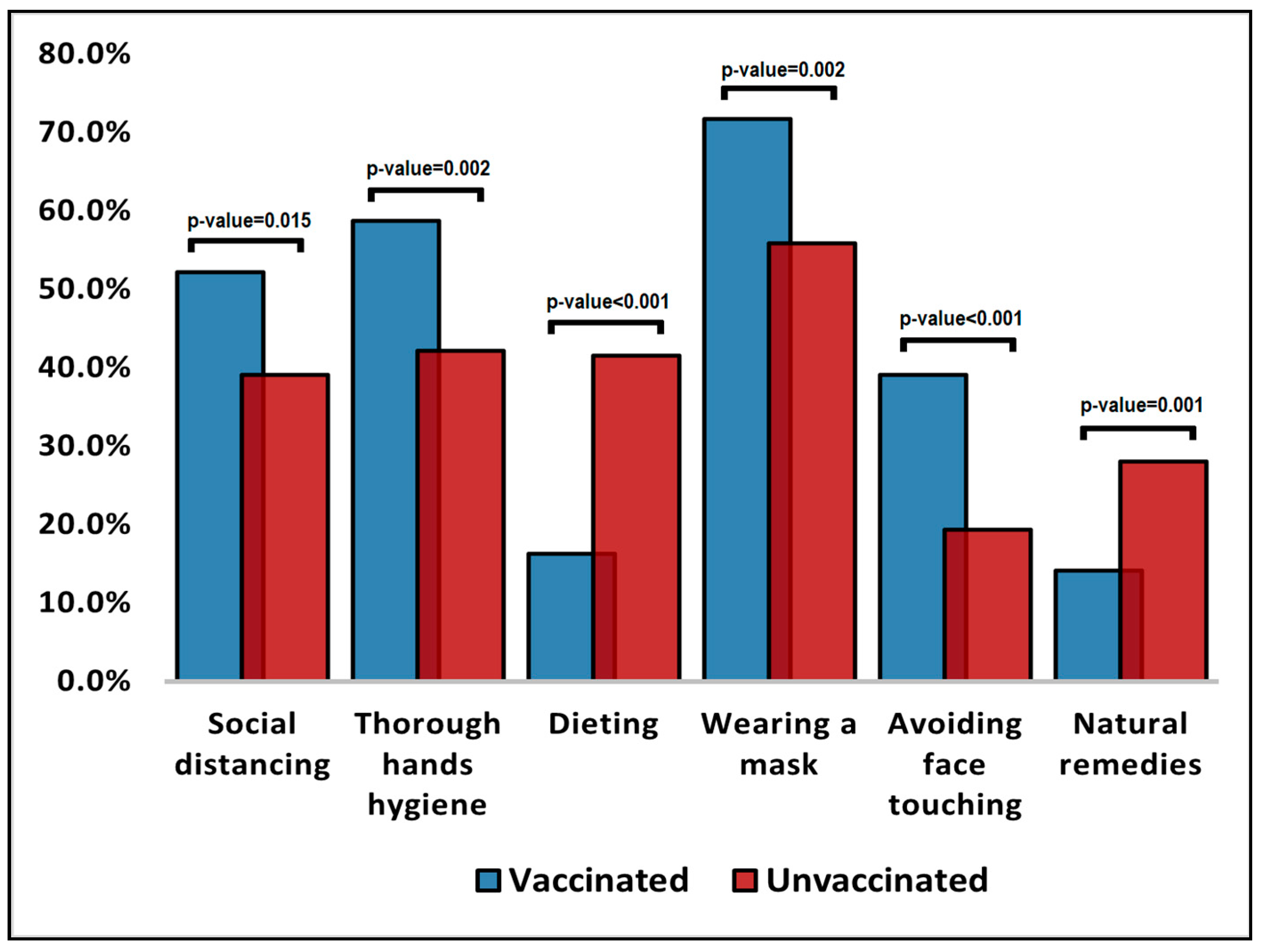
| Social distancing | 96 (52.2%) | 63 (39.1%) | 0.015 |
| Thorough hand hygiene | 108 (58.7%) | 68 (42.2%) | 0.002 |
| Dieting | 30 (16.3%) | 67 (41.6%) | <0.001 |
| Wearing a mask | 132 (71.7%) | 90 (55.9%) | 0.002 |
| Avoiding face touching | 72 (39.1%) | 31 (19.3%) | <0.001 |
| Natural remedies | 26 (14.1%) | 45 (28.0%) | 0.001 |
| Questions | Vaccinated (n = 184) | Unvaccinated (n = 161) | p |
|---|---|---|---|
| “I care about” | |||
| Putting an end to the pandemic | 166 (90.2%) | 129 (80.1%) | 0.007 |
| Allowing life to return to normal | 124 (67.4%) | 97 (60.2%) | 0.167 |
| Travelling without restrictions | 109 (59.2%) | 62 (38.5%) | <0.001 |
| The severity and complications of COVID-19 | 127 (69.0%) | 56 (34.8%) | <0.001 |
| COVID-19 vaccines becoming mandatory | 88 (47.8%) | 50 (31.1%) | 0.001 |
| What doctors recommend | 70 (38.0%) | 44 (27.3%) | 0.034 |
| “I am concerned about” | |||
| Vaccination side effects | 51 (27.7%) | 84 (52.2%) | <0.001 |
| Vaccination efficacy | 48 (26.1%) | 74 (46.0%) | <0.001 |
| COVID-19 being a conspiracy | 11 (6.0%) | 37 (23.0%) | <0.001 |
| Vaccination being against my religion | 8 (4.3%) | 12 (7.5%) | 0.218 |
| The quality of vaccines received by my country | 15 (8.2%) | 31 (19.3%) | 0.002 |
| Vaccine efficacy against new SARS-CoV-2 strains | 19 (10.3%) | 24 (14.9%) | 0.198 |
| The technology of COVID-19 vaccines | 60 (32.6%) | 87 (54.0%) | |
| “I might be influenced by” | |||
| Availability of vaccines near me | 41 (22.3%) | 68 (42.2%) | <0.001 |
| Seeing better results against new SARS-CoV-2 infections | 45 (24.5%) | 53 (32.9%) | 0.082 |
| Seeing more people getting vaccinated | 26 (14.1%) | 57 (35.4%) | <0.001 |
| Clinical trials’ results | 12 (6.5%) | 9 (5.6%) | 0.718 |
| Determinants | OR * | 95% CI | p-Value |
|---|---|---|---|
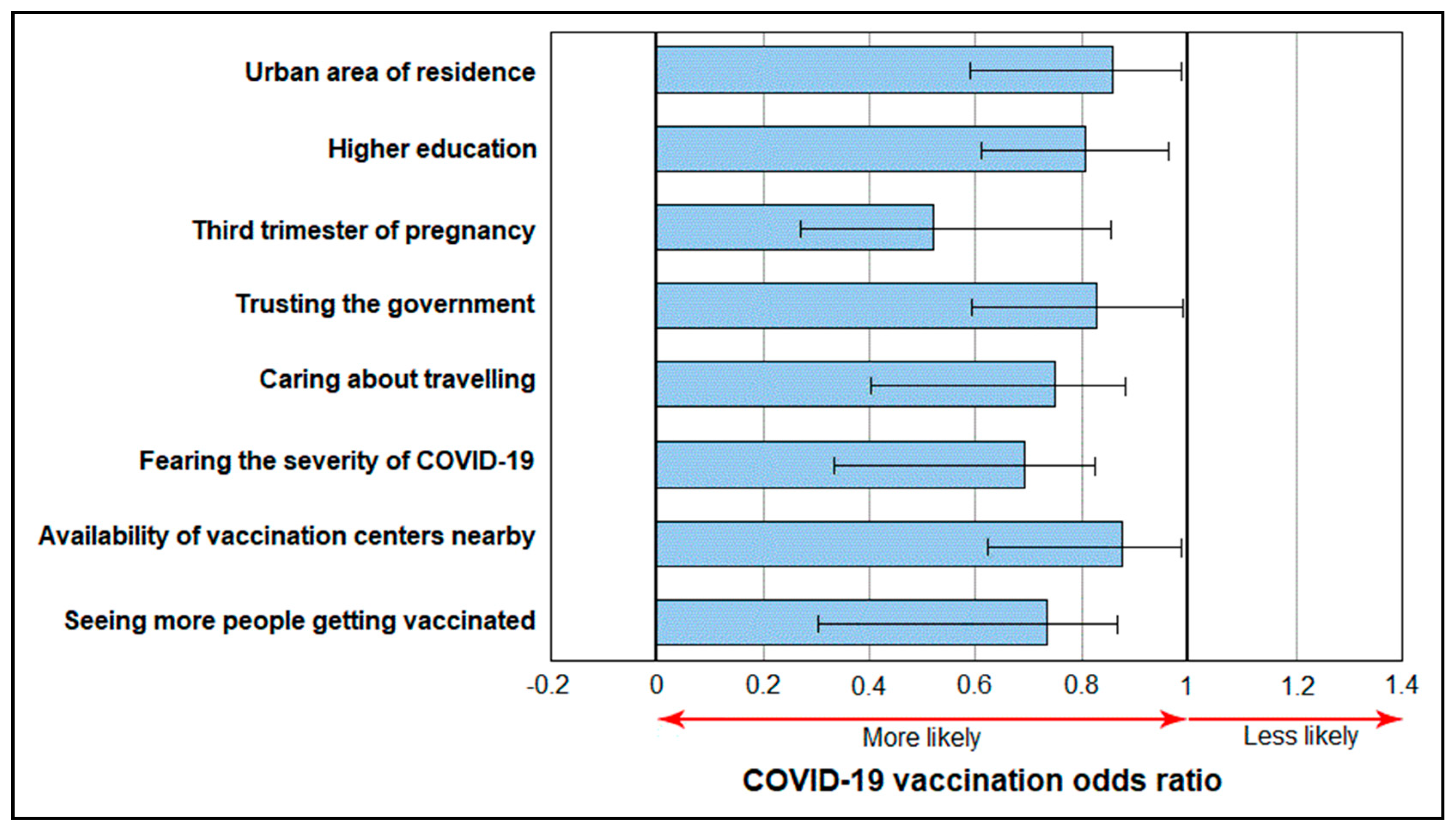
| Urban area of residence | 0.86 | 0.59–0.98 | 0.043 |
| Higher education | 0.81 | 0.62–0.95 | 0.030 |
| Third trimester of pregnancy | 0.54 | 0.28–0.86 | <0.001 |
| Trusting the government | 0.83 | 0.59–0.99 | 0.047 |
| Caring about travelling | 0.76 | 0.40–0.87 | 0.005 |
| Fearing the severity of COVID-19 | 0.68 | 0.34–0.82 | 0.001 |
| Availability of vaccination centers nearby | 0.87 | 0.63–0.99 | 0.045 |
| Seeing more people getting vaccinated | 0.75 | 0.33–0.88 | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Citu, C.; Chiriac, V.D.; Citu, I.M.; Gorun, O.M.; Burlea, B.; Bratosin, F.; Popescu, D.-E.; Ratiu, A.; Buca, O.; Gorun, F. Appraisal of COVID-19 Vaccination Acceptance in the Romanian Pregnant Population. Vaccines 2022, 10, 952. https://doi.org/10.3390/vaccines10060952
Citu C, Chiriac VD, Citu IM, Gorun OM, Burlea B, Bratosin F, Popescu D-E, Ratiu A, Buca O, Gorun F. Appraisal of COVID-19 Vaccination Acceptance in the Romanian Pregnant Population. Vaccines. 2022; 10(6):952. https://doi.org/10.3390/vaccines10060952
Chicago/Turabian StyleCitu, Cosmin, Veronica Daniela Chiriac, Ioana Mihaela Citu, Oana Maria Gorun, Bogdan Burlea, Felix Bratosin, Daniela-Eugenia Popescu, Adrian Ratiu, Oana Buca, and Florin Gorun. 2022. "Appraisal of COVID-19 Vaccination Acceptance in the Romanian Pregnant Population" Vaccines 10, no. 6: 952. https://doi.org/10.3390/vaccines10060952
APA StyleCitu, C., Chiriac, V. D., Citu, I. M., Gorun, O. M., Burlea, B., Bratosin, F., Popescu, D.-E., Ratiu, A., Buca, O., & Gorun, F. (2022). Appraisal of COVID-19 Vaccination Acceptance in the Romanian Pregnant Population. Vaccines, 10(6), 952. https://doi.org/10.3390/vaccines10060952









