Kinome Analysis to Define Mechanisms of Adjuvant Action: PCEP Induces Unique Signaling at the Injection Site and Lymph Nodes
Abstract
1. Introduction
2. Materials and Methods
2.1. Adjuvants
2.2. Mouse Trials
2.3. Quantitative Real-Time PCR (qRT-PCR)
2.4. Isolation of Recruited Cells from Site of Injection and Draining Lymph Node
2.5. Flow Cytometry
2.6. Peptide Arrays for Kinome Analysis
2.7. Analysis of Kinome Data
2.8. Pathway Over-Representation Analysis
2.9. Statistical Analysis
3. Results
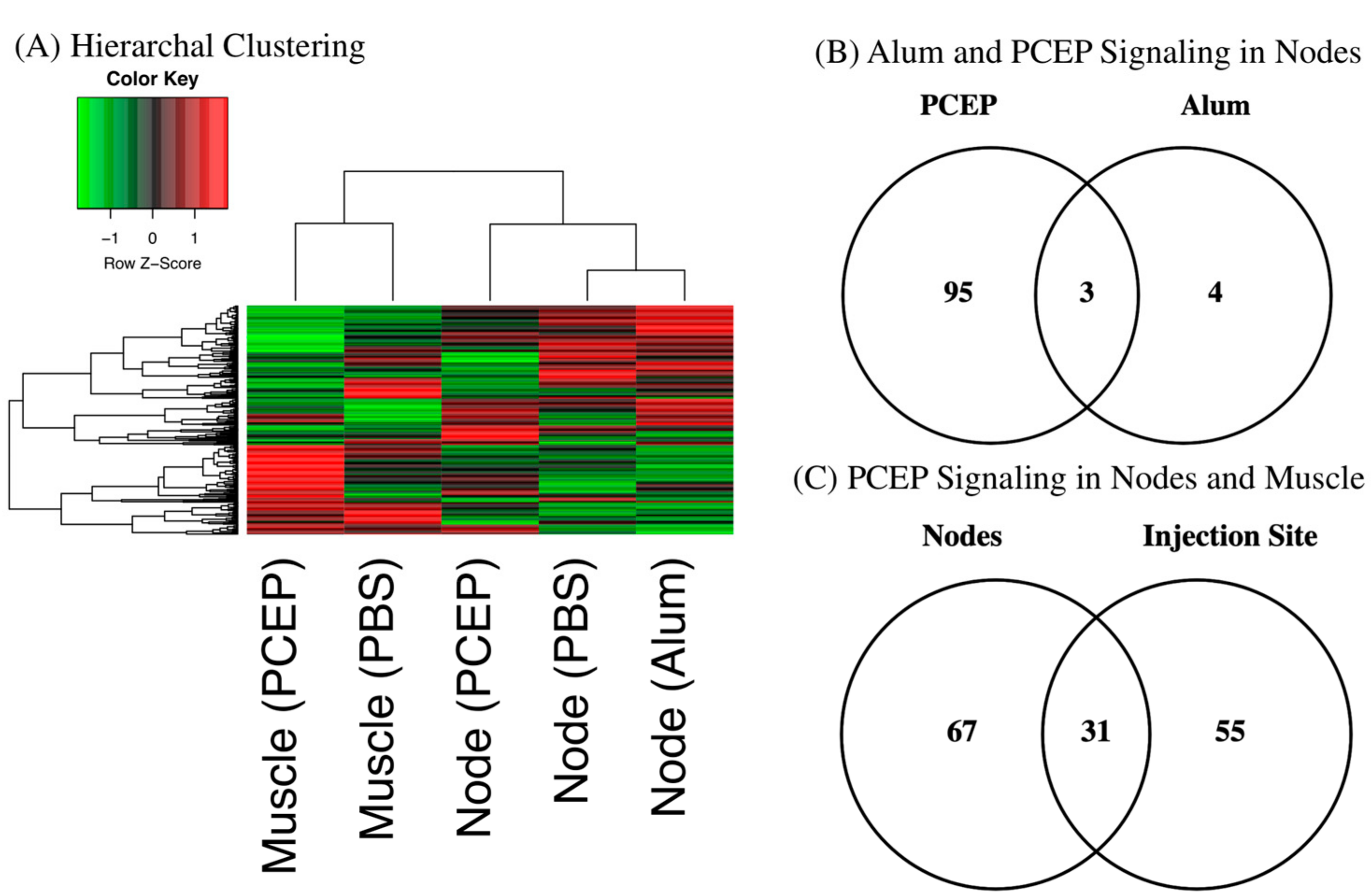
3.1. Kinome Analysis
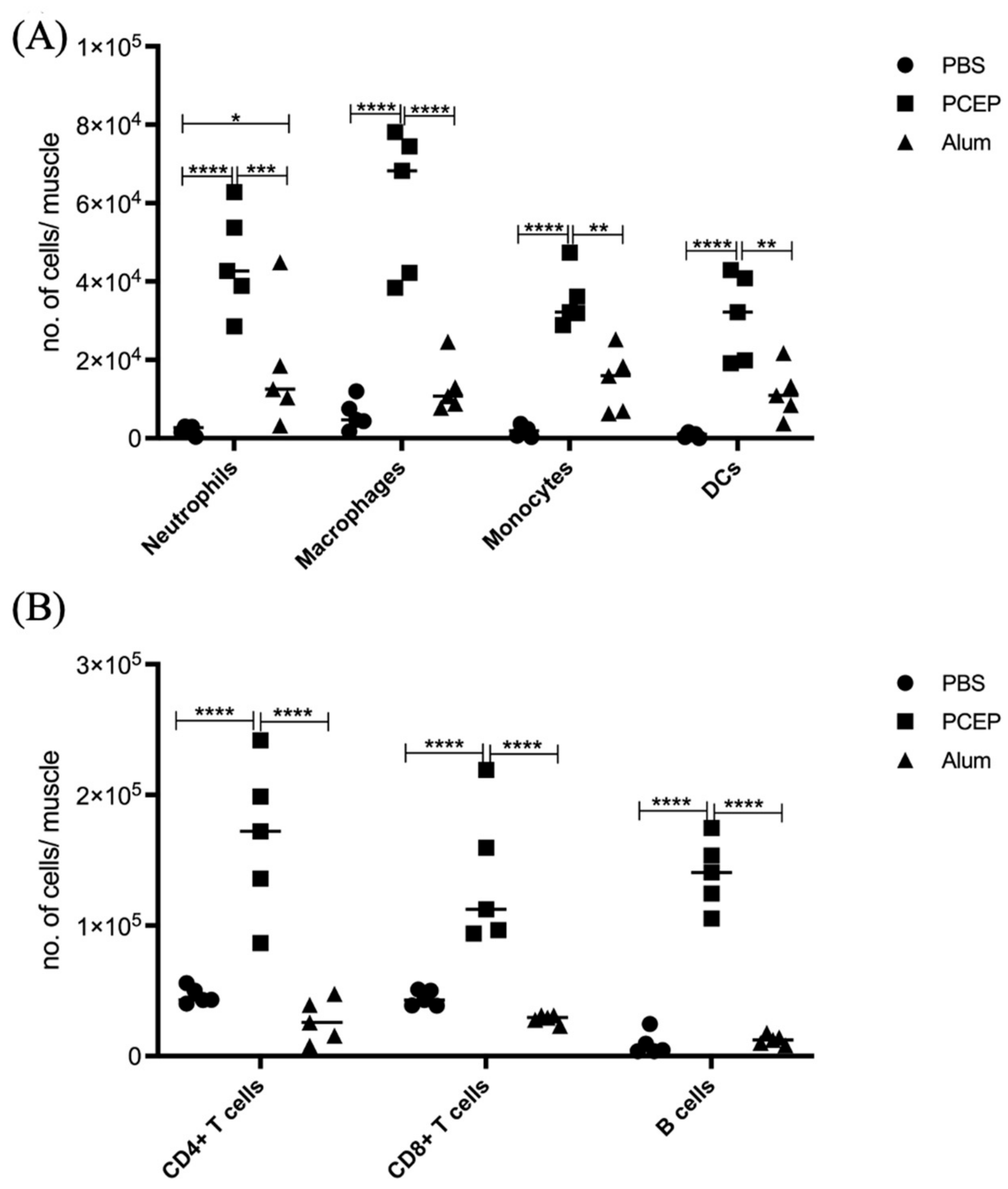
3.2. Signaling Events at the Injection Site
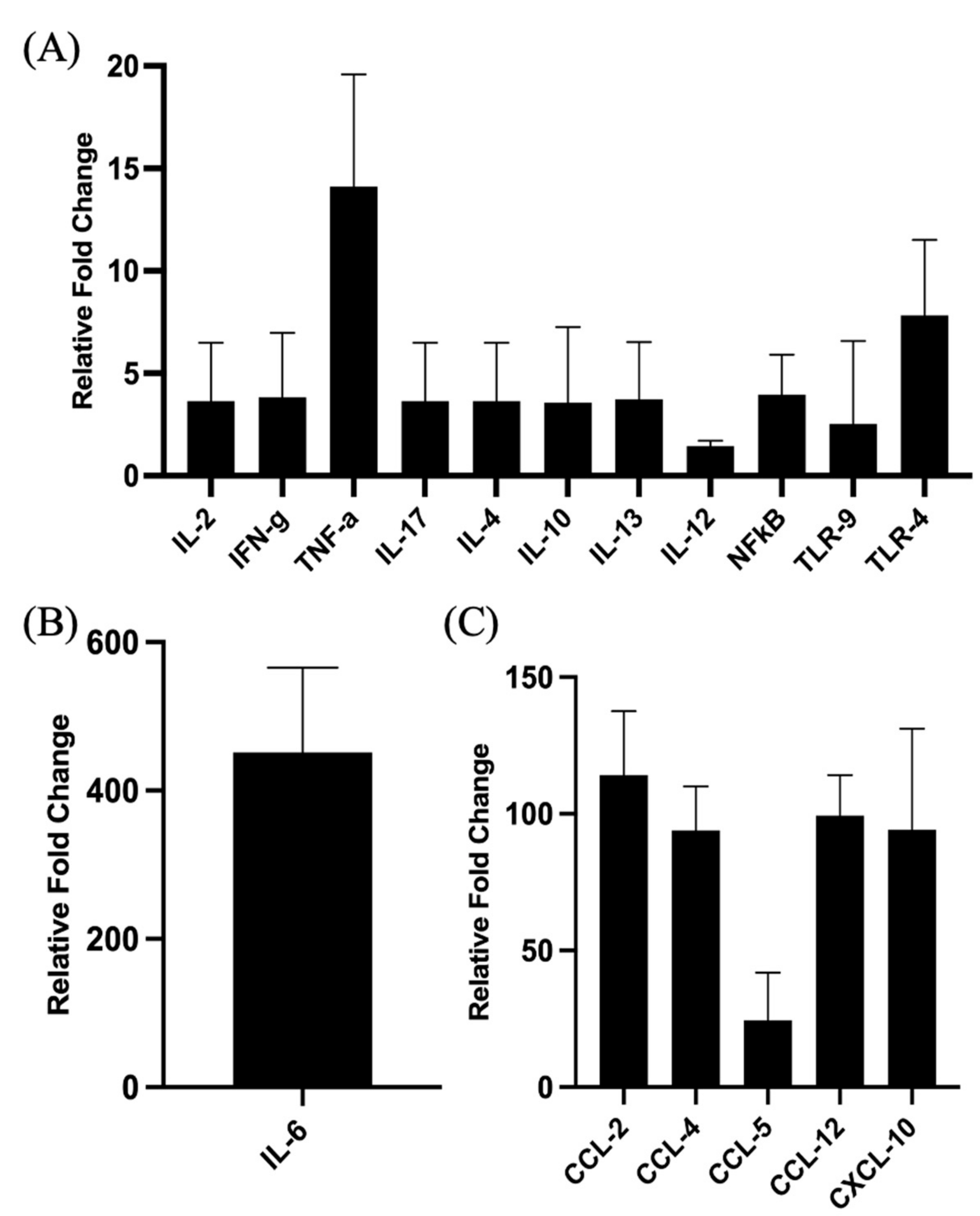
3.3. Validation of Signaling Events at Site of Injection
3.4. Signaling Events at the Draining Lymph Nodes
3.5. Validation of Signaling Events at Draining Lymph Nodes
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Acknowledgments
Conflicts of Interest
References
- Reed, S.G.; Orr, M.T.; Fox, C.B. Key roles of adjuvants in modern vaccines. Nat. Med. 2013, 19, 1597–1608. [Google Scholar] [CrossRef] [PubMed]
- Nanishi, E.; Dowling, D.J.; Levy, O. Toward precision adjuvants: Optimizing science and safety. Curr. Opin. Pediatr. 2020, 32, 125–138. [Google Scholar] [CrossRef] [PubMed]
- Morefield, G.L.; Sokolovska, A.; Jiang, D.; HogenEsch, H.; Robinson, J.P.; Hem, S.L. Role of aluminum-containing adjuvants in antigen internalization by dendritic cells in vitro. Vaccine 2005, 23, 1588–1595. [Google Scholar] [CrossRef] [PubMed]
- Seubert, A.; Monaci, E.; Pizza, M.; O’Hagan, D.T.; Wack, A. The adjuvants aluminum hydroxide and MF59 induce monocyte and granulocyte chemoattrac- tants and enhance monocyte differentiation toward dendritic cells. J. Immunol. 2008, 180, 5402–5412. [Google Scholar] [CrossRef]
- Wilson-Welder, J.H.; Torres, M.P.; Kipper, M.J.; Mallapragada, S.K.; Wannemuehler, M.J.; Narasimhan, B. Vaccine adjuvants: Current challenges and future approaches. J. Pharm. Sci. 2009, 98, 1278–1316. [Google Scholar] [CrossRef]
- Mutwiri, G.; Benjamin, P.; Soita, H.; Babiuk, L.A. Co-administration of polyphosphazenes with CpG oligodeoxynucleotides strongly enhances immune responses in mice immunized with Hepatitis B virus surface antigen. Vaccine 2008, 26, 2680–2688. [Google Scholar] [CrossRef]
- Li, H.; Nookala, S.; Re, F. Aluminum hydroxide adjuvants activate caspase-1 and induce IL-1beta and IL-18 release. J. Immunol. 2007, 178, 5271–5276. [Google Scholar] [CrossRef]
- Mosca, F.; Tritto, E.; Muzzi, A.; Monaci, E.; Bagnoli, F.; Iavarone, C.; O’Hagan, D.; Rappuoli, R.; De Gregorio, E. Molecular and cellular signatures of human vaccine adjuvants. Proc. Natl. Acad. Sci. USA 2008, 105, 10501–10506. [Google Scholar] [CrossRef]
- Calabro, S.; Tortoli, M.; Baudner, B.C.; Pacitto, A.; Cortese, M.; O’Hagan, D.T.; De Gregorio, E.; Seubert, A.; Wack, A. Vaccine adjuvants alum and MF59 induce rapid recruitment of neutrophils and monocytes that participate in antigen transport to draining lymph nodes. Vaccine 2011, 29, 1812–1823. [Google Scholar] [CrossRef]
- Caproni, E.; Tritto, E.; Cortese, M.; Muzzi, A.; Mosca, F.; Monaci, E.; Baudner, B.; Seubert, A.; Gregorio, D.G. MF59 and Pam3CSK4 boost adaptive responses to influenza subunit vaccine through an IFN type I-independent mechanism of action. J. Immunol. 2012, 188, 3088–3098. [Google Scholar] [CrossRef]
- Santoro, F.; Pettini, E.; Kazmin, D.; Ciabattini, A.; Fiorino, F.; Gilfillan, G.D.; Evenroed, I.M.; Andersen, P.; Pozzi, G.; Medaglini, D. Transcriptomics of the Vaccine Immune Response: Priming with Adjuvant Modulates Recall Innate Responses After Boosting. Front. Immunol. 2018, 9, 1248. [Google Scholar] [CrossRef] [PubMed]
- Lindqvist, M.; Nookaew, I.; Brinkenberg, I.; Samuelson, E.; Thörn, K.; Nielsen, J.; Harandi, A.M. Unraveling molecular signatures of immunostimulatory adjuvants in the female genital tract through systems biology. PLoS ONE 2011, 6, e20448. [Google Scholar] [CrossRef] [PubMed]
- Morel, S.; Didierlaurent, A.; Bourguignon, P.; Delhaye, S.; Baras, B.; Jacob, V.; Planty, C.; Elouahabi, A.; Harvengt, P.; Carlsen, H.; et al. Adjuvant System AS03 containing α-tocopherol modulates innate immune response and leads to improved adaptive immunity. Vaccine 2011, 29, 2461–2473. [Google Scholar] [CrossRef] [PubMed]
- Olafsdottir, T.; Lindqvist, M.; Harandi, A.M. Molecular signatures of vaccine adjuvants. Vaccine 2015, 33, 5302–5307. [Google Scholar] [CrossRef] [PubMed]
- Cohen, P. Protein kinases--the major drug targets of the twenty-first century? Nat. Rev. Drug Discov. 2002, 1, 309–315. [Google Scholar] [CrossRef] [PubMed]
- Facciuolo, A.; Denomy, C.; Lipsit, S.; Kusalik, A.; Napper, S. From Beef to Bees: High-Throughput Kinome Analysis to Understand Host Responses of Livestock Species to Infectious Diseases and Industry-Associated Stress. Front. Immun. 2020, 11, 765. [Google Scholar] [CrossRef]
- Kindrachuk, J.; Arsenault, R.; Kusalik, A.; Kindrachuk, K.; Trost, B.; Napper, S.; Jahrling, P.; Blaney, J. Systems Kinomics Demonstrates Congo Basin Monkeypox Virus Infection Selectively Modulates Host Cell Signaling Responses as Compared to West African Monkeypox Virus. Mol. Cell. Proteomics. 2012, 11, M111.015701. [Google Scholar] [CrossRef]
- Arsenault, R.J.; Li, Y.; Bell, K.; Doig, K.; Potter, A.; Griebel, P.J.; Kusalik, A. Mycobacterium avium subsp. paratuberculosis inhibits gamma interferon-induced signaling in bovine monocytes: Insights into the cellular mechanisms of Johne’s disease. Infect. Immun. 2012, 80, 3039–3048. [Google Scholar] [CrossRef]
- Arsenault, R.J.; Napper, S.; Kogut, M.H. Salmonella enterica Typhimurium infection causes metabolic changes in chicken muscle involving AMPK, fatty acid and insulin/mTOR signaling. Vet. Res. 2013, 44, 35. [Google Scholar] [CrossRef]
- Lipsit, S.W.L.; Wilkinson, J.; Scruten, E.; Facciuolo, A.; Denomy, C.; Griebel, P.J.; Kausalik, A.; Plastow, G.; Napper, S. Kinome profiling of peripheral blood mononuclear cells collected prior to vaccination reveals biomarkers and potential mechanisms of vaccine unresponsiveness in pigs. Sci. Rep. 2020, 10, 11546. [Google Scholar] [CrossRef]
- Diks, S.H.; Kok, K.; O’Toole, T.; Hommes, D.W.; van Dijken, P.; Joore, J.; Peppelenbosch, M.P. Kinome profiling for studying lipopolysaccharide signal transduction in human peripheral blood mononuclear cells. J. Biol. Chem. 2004, 279, 49206–49213. [Google Scholar] [CrossRef] [PubMed]
- Arsenault, R.; Jalal, S.; Babiuk, L.A.; Potter, A.; Griebel, P.; Napper, S. Kinome analysis of Toll-like receptor signaling in bovine monocytes, J. Recept. Signal Transduct. Res. 2009, 29, 299–311. [Google Scholar] [CrossRef] [PubMed]
- Trost, B.; Kindrachuk, J.; Scruten, E.; Griebel, P.J.; Kausalik, A.; Napper, S. Kinotypes: Stable species- and individual-specific profiles of cellular kinase activity. BMC Genom. 2013, 14, 854. [Google Scholar] [CrossRef] [PubMed]
- Määttänen, P.; Trost, B.; Scruten, E.; Potter, A.; Kusalik, A.; Griebel, P.J.; Napper, S. Divergent immune responses to Mycobacterium avium subsp. paratuberculosis infection correlate with kinome responses at the site of intestinal infection. Infect. Immun. 2013, 81, 2861–2872. [Google Scholar] [CrossRef]
- Robertson, A.; Trost, B.; Scruten, E.; Robertson, T.; Mostajeran, M.; Connor, W.; Kusalik, A.; Griebel, P.; Napper, S. Identification of developmentally-specific kinotypes and mechanisms of Varroa mite resistance through whole-organism, kinome analysis of honeybee. Front. Genet. 2014, 5, 139. [Google Scholar] [CrossRef]
- Mutwiri, G.; Gerdts, V.; Lopez, M.; Babiuk, L.A. Innate immunity and new adjuvants. Rev. Sci. Tech. 2007, 1, 147–156. [Google Scholar] [CrossRef]
- Eng, N.F.; Garlapati, S.; Gerdts, V.; Babiuk, L.A.; Mutwiri, G.K. PCEP enhances IgA mucosal immune responses in mice following different immunization routes with influenza virus antigens. J. Immune Based Ther. Vaccines 2010, 8, 4. [Google Scholar] [CrossRef]
- Andrianov, A.K.; Svirkin, Y.Y.; LeGolvan, M.P. Synthesis and biologically relevant properties of polyphosphazene polyacids. Biomacromolecules 2004, 5, 1999–2006. [Google Scholar] [CrossRef]
- Andrianov, A.K.; Marin, A.; Chen, J. Synthesis, properties, and biological activity of poly[di(sodium carboxylatoethylphenoxy)phosphazene]. Biomacromolecules 2006, 7, 394–399. [Google Scholar] [CrossRef]
- Payne, L.G.; Jenkins, S.A.; Woods, A.L.; Grund, E.M.; Geribo, W.E.; Loebelenz, J.R.; Andrianov, A.K.; Roberts, B.E. Poly[di(carboxylatophenoxy)phosphazene] (PCPP) is a potent immunoadjuvant for an influenza vaccine. Vaccine 1998, 16, 92–98. [Google Scholar] [CrossRef]
- Payne, L.G.; Van Nest, G.; Barchfeld, G.L.; Siber, G.R.; Gupta, R.K.; Jenkins, S.A. PCPP as a parenteral adjuvant for diverse antigens. Dev. Biol. Stand. 1998, 92, 79–87. [Google Scholar] [PubMed]
- Chaffey, A.; Hamonic, G.; Chand, D.; Mutwiri, G.K.; Wilson, H.L. The Adjuvants Polyphosphazene (PCEP) and a Combination of Curdlan Plus Leptin Promote a Th17-Type Immune Response to an Intramuscular Vaccine in Mice. Vaccines 2021, 9, 507. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Taylor, R.K.; Wade, W.F. Anti-class II monoclonal antibody-targeted Vibrio cholerae TcpA pilin: Modulation of serologic response, epitope specificity, and isotype. Infect. Immun. 2001, 69, 7679–7686. [Google Scholar] [CrossRef] [PubMed]
- Chand, D.J.; Magiri, R.B.; Wilson, H.L.; Mutwiri, G.K. Polyphosphazenes as Adjuvants for Animal Vaccines and Other Medical Applications. Front. Bioeng. Biotechnol. 2021, 9, 625482. [Google Scholar] [CrossRef]
- Andrianov, A.K.; Marin, A.; Roberts, B.E. Polyphosphazene polyelectrolytes: A link between the formation of noncovalent complexes with antigenic proteins and immunostimulating activity. Biomacromolecules 2005, 6, 1375–1379. [Google Scholar] [CrossRef]
- Awate, S.; Eng, N.F.; Gerdts, V.; Babiuk, L.A.; Mutwiri, G. Caspase-1 Dependent IL-1β Secretion and Antigen-Specific T-Cell Activation by the Novel Adjuvant, PCEP. Vaccines 2014, 2, 500–514. [Google Scholar] [CrossRef]
- Awate, S.; Wilson, H.L.; Singh, B.; Babiuk, L.A.; Mutwiri, G. The adjuvant PCEP induces recruitment of myeloid and lymphoid cells at the injection site and draining lymph node. Vaccine 2014, 32, 2420–2427. [Google Scholar] [CrossRef]
- Awate, S.; Wilson, H.L.; Lai, K.; Babiuk, L.A.; Mutwiri, G. Activation of adjuvant core response genes by the novel adjuvant PCEP. Mol. Immunol. 2012, 51, 292–303. [Google Scholar] [CrossRef]
- Trost, B.; Kindrachuk, J.; Määttänen, P.; Napper, S.; Kusalik, A. PIIKA 2: An expanded, web-based platform for analysis of kinome microarray data. PLoS ONE 2013, 8, e80837. [Google Scholar] [CrossRef]
- Breuer, K.; Foroushani, A.K.; Laird, M.R.; Chen, C.; Sribnaia, A.; Lo, R.; Winsor, G.L.; Hancock, R.E.W.; Brinkman, F.S.L.; Lynn, D.J. InnateDB: Systems biology of innate immunity and beyond-recent updates and continuing curation. Nucleic Acids Res. 2013, 41, D1228–D1233. [Google Scholar] [CrossRef]
- Régnier, M.; Gourbeyre, P.; Pinton, P.; Napper, S.; Laffite, J.; Cossalter, A.-M.; Bailly, J.-D.; Lippi, Y.; Bertrand-Michel, J.; Bracarense, P.F.R.L.; et al. Identification of Signaling Pathways Targeted by the Food Contaminant FB1: Transcriptome and Kinome Analysis of Samples from Pig Liver and Intestine. Mol. Nutr. Food Res. 2017, 61, 1700433. [Google Scholar] [CrossRef] [PubMed]
- O’Hagan, D.T.; Fox, C.B. Are we entering a new age for human vaccine adjuvants? Expert Rev. Vaccines 2015, 14, 909–911. [Google Scholar] [CrossRef] [PubMed][Green Version]
- O’Hagan, D.; Lodaya, R.; Lofano, G. The continued advance of vaccine adjuvants—“we can work it out”. Semin. Immunol. 2020, 50, 101426. [Google Scholar] [CrossRef] [PubMed]
- Pulendran, B.; Arunachalam, P.S.; O’Hagan, D.T. Emerging concepts in the science of vaccine adjuvants. Nat. Rev. Drug Discov. 2021, 20, 454–475. [Google Scholar] [CrossRef]
- Katz, O.; Gil, L.; Lifshitz, L.; Prutchi-Sagiv, S.; Gassmann, M.; Mittelman, M.; Neumann, D. Erythropoietin enhances immune responses in mice. Eur. J. Immunol. 2007, 37, 1584–1593. [Google Scholar] [CrossRef]
- Hassan, K.; Shternberg, L.; Alhaj, M.; Giron, R.; Reshef, R.; Barak, M.; Kristal, B. The effect of erythropoietin therapy and hemoglobin levels on the immune response to Engerix-B vaccination in chronic kidney disease. Ren. Fail. 2003, 25, 471–478. [Google Scholar] [CrossRef]
- Oster, H.S.; Prutchi-Sagiv, S.; Halutz, O.; Shabtai, E.; Hoffman, M.; Neumann, D.; Mittelman, M. Erythropoietin treatment is associated with an augmented immune response to the influenza vaccine in hematologic patients. Exp. Hematol. 2013, 41, 167–171. [Google Scholar] [CrossRef]

| S. No | Gene Symbol | Forward Primer | Reverse Primer |
|---|---|---|---|
| 1 | IL-2 | CCTGGAGCAGCTGTTGATGG | CAGAACATGCCGCAGAGGTC |
| 2 | IL-4 | ATGGGTCTCAACCCCCAGC | GCTCTTTAGGCTTTCCAGG |
| 3 | IL-6 | TGTCTATACCACTTCACAAGTC | GCACAACTCTTTTCTCATTTCCA |
| 4 | IL-10 | TAGTTCCCAGAAGCCATGTG | AGAGGGAGCAGTTTGTAAGC |
| 5 | IL-12 | TGCCAGCCTGCCTTATATTG | TCCACCAGGACCACTAAATG |
| 6 | IL-13 | CAGCAGCTTGAGCACATTTC | CATAGGCAGCAAACCATGTC |
| 7 | IL-17 | ACCTCAACCGTTCCACGTCA | CAGGGTCTTCATTGCGGTG |
| 8 | IFN-γ | TGAACGCTACACACTGCAT | CGACTCCTTTTCCGCTTCCT |
| 9 | TNF-α | GACCCTCACACTCAGATCATCT | CCACTTGGTGGTTTGCTACGA |
| 10 | NFκB | AGAAGACACGAGGCTACAAC | TCACAGACGCTGTCACTATC |
| 11 | TLR-4 | TCCCAGTGATGGCTGATTAG | GCACCCAACATTGTGTTACC |
| 12 | TLR-9 | GAAGGGACAGCAATGGAAAG | GCCAAGTGCTACCATTAACC |
| 13 | CCL-2 | TCACCTGCTGCTACTCATTC | TCTGGACCCATTCCTTCTTG |
| 14 | CCL-4 | CCAGCTGTGGTATTCCTGAC | GAGCTGCTCAGTTCAACTCC |
| 15 | CCL-5 | CTCCCTGCTGCTTTGCCTAC | CACACTTGGCGGTTCCTTCG |
| 16 | CCL-12 | TGCCTCCTGCTCATAGCTAC | GGCTGCTTGTGATTCTCCTG |
| 17 | CXCL-10 | GTCACATCAGCTGCTACTCC | CGCACCTCCACATAGCTTAC |
| Increased Phosphorylation | Decreased Phosphorylation | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| ID | P Site | Accession | FC | p | ID | P Site | Accession | FC | p |
| Shc1 | Y439 | P29353 | 1.59 | 0.02 | Lyn | Y396 | P07948 | −1.5 | 0.05 |
| PLCG2 | Y759 | P16885 | 1.49 | 0.05 | IRF-3 | S402 | Q14653 | −1.37 | 0.04 |
| Smad3 | S423 | P84022 | 1.47 | 0.03 | IKK-g | S43 | Q9Y6K9 | −1.35 | 0.01 |
| Syk | Y352 | P43405 | 1.44 | 0.01 | IKK-b | Y188 | O14920 | −1.3 | 0.03 |
| Syk | Y525 | P43405 | 1.43 | 0.01 | MDM2 | S166 | Q00987 | −1.3 | 0.004 |
| Ck2-B | S228 | Q5SRQ6 | 1.35 | 0.04 | ACTA1 | Y55 | P68133 | −1.29 | 0.02 |
| Cdk4 | S150 | P11802 | 1.34 | 0.02 | MAVS | S233 | Q7Z434 | −1.28 | 0.04 |
| p300 | S2279 | Q09472 | 1.34 | 0.05 | Keap1 | S293 | Q14145 | −1.28 | 0.02 |
| EP300 | S2366 | Q09472 | 1.33 | 0.0001 | MK2 | Y132 | P16389 | −1.27 | 0.05 |
| CTNNB1 | Y654 | P35222 | 1.31 | 0.005 | IKK-a | S180 | O15111 | −1.26 | 0.0002 |
| TAK1 | T178 | O43318 | 1.3 | 0.02 | p38-a | Y322 | Q16539 | −1.25 | 0.04 |
| SEK1 | T261 | P45985 | 1.3 | 0.04 | IRAK1 | T100 | P51617 | −1.25 | 0.02 |
| TAK1 | T187 | O43318 | 1.29 | 0.04 | IL7R | Y449 | P16871 | −1.25 | 0.004 |
| Grb10 | S150 | Q13322 | 1.28 | 0.01 | PDK1 | S241 | O15530 | −1.24 | 0.001 |
| K8 | S74 | P05787 | 1.27 | 0.02 | Fos | S362 | P01100 | −1.23 | 0.05 |
| Sek1 | S80 | P45985 | 1.27 | 0.03 | MEK1 | Y385 | Q02750 | −1.23 | 0.03 |
| EGFR | T693 | P00533 | 1.26 | 0.007 | CREB | S133 | P16220 | −1.23 | 0.02 |
| SOC3 | Y221 | O14543 | 1.26 | 0.02 | Jun | S63 | P05412 | −1.22 | 0.05 |
| TBK1 | S172 | Q9UHD2 | 1.25 | 0.02 | HSP70 | Y525 | P08107 | −1.22 | 0.04 |
| Smad6 | S435 | O43541 | 1.24 | 0.02 | Met | Y1003 | P08581 | −1.22 | 0.01 |
| Cdc42 | Y32 | P60953 | 1.24 | 0.03 | ACC1 | S29 | Q13085 | −1.22 | 0.004 |
| XIAP | S87 | P98170 | 1.23 | 0.02 | Akt1 | T308 | P31749 | −1.21 | 0.05 |
| IRAK4 | T208 | P51617 | 1.21 | 0.01 | JNK2 | T183 | P45984 | −1.2 | 0.03 |
| SMAD3 | S204 | Q15796 | 1.21 | 0.03 | PDK1 | Y373 | O15530 | −1.2 | 0.01 |
| STMN1 | S24 | P16949 | 1.21 | 0.05 | Casp3 | S150 | P42574 | −1.19 | 0.05 |
| CDK2 | Y14 | P24941 | 1.2 | 0.006 | Mapk14 | T122 | Q16539 | −1.19 | 0.03 |
| TrKA | Y496 | P04629 | 1.2 | 0.02 | Aura | T287 | O14965 | −1.18 | 0.04 |
| P27kip1 | Y74 | P46527 | 1.2 | 0.03 | JNK1 | T183 | P45983 | −1.18 | 0.02 |
| Crk | Y221 | P46108 | 1.19 | 0.02 | Bim | S69 | O43521 | −1.17 | 0.02 |
| IRAK1 | T387 | P51617 | 1.18 | 0.01 | p70S6K | S447 | P23443 | −1.16 | 0.005 |
| TNIK | T181 | Q9UKE5 | 1.18 | 0.02 | Mnk1 | T255 | Q9BUB5 | −1.15 | 0.05 |
| Tyk2 | Y1054 | P29597 | 1.18 | 0.03 | CHOP | S79 | P35638 | −1.15 | 0.05 |
| EGFR | Y869 | P00533 | 1.18 | 0.05 | Mek1 | S217 | Q02750 | −1.15 | 0.02 |
| TrKA | Y757 | P04629 | 1.17 | 0.008 | CDK2 | T160 | P24941 | −1.15 | 0.02 |
| gp130 | Y767 | P40189 | 1.17 | 0.02 | MSK2 | S360 | O75676 | −1.14 | 0.02 |
| CREB | S117 | P16220 | 1.17 | 0.02 | NFkB p65 | S536 | Q04206 | −1.14 | 0.01 |
| SHC3 | Y341 | Q92529 | 1.17 | 0.03 | CREB | S111 | P16220 | −1.13 | 0.05 |
| PI3K p85 | Y605 | O00459 | 1.17 | 0.05 | Fyn | Y420 | P06241 | −1.13 | 0.02 |
| Smad3 | T179 | P84022 | 1.17 | 0.05 | Met | Y1234 | P08581 | −1.11 | 0.02 |
| IFNAR1 | Y466 | P17181 | 1.15 | 0.03 | |||||
| STAT6 | Y641 | P42226 | 1.14 | 0.03 | |||||
| HSP60 | S70 | P10809 | 1.14 | 0.03 | |||||
| GIT2 | Y592 | Q14161 | 1.13 | 0.01 | |||||
| STMN1 | S37 | P16949 | 1.12 | 0.03 | |||||
| IFNGR1 | S495 | P15260 | 1.11 | 0.02 | |||||
| SOC3 | Y204 | O14543 | 1.09 | 0.003 | |||||
| CTNNB1 | S33 | P35222 | 1.07 | 0.02 | |||||
| Pathway Name | Pathway ID | Source Name | Pathway Uploaded | Pathway p-Value |
|---|---|---|---|---|
| JAK STAT pathway and regulation | 16125 | INOH | 27 | 5.04 × 1023 |
| RANKL | 15925 | NETPATH | 19 | 1.20 × 1022 |
| Fc epsilon receptor signaling | 17802 | REACTOME | 23 | 2.07 × 1022 |
| Innate Immune System | 17476 | REACTOME | 33 | 3.51 × 1022 |
| IL-7 signaling | 16106 | INOH | 23 | 3.54 × 1022 |
| Pathways in cancer | 4397 | KEGG | 27 | 1.98 × 1021 |
| EPO signaling pathway | 16151 | INOH | 22 | 8.75 × 1021 |
| VEGF signaling pathway | 16190 | INOH | 22 | 1.01 × 1020 |
| Immune System | 18444 | REACTOME | 40 | 2.63 × 1020 |
| BCR signaling pathway | 15384 | PID NCI | 16 | 3.06 × 1020 |
| IL2 | 15918 | NETPATH | 17 | 4.59 × 1020 |
| Toll-like receptor signaling pathway | 564 | KEGG | 18 | 8.14 × 1020 |
| Increased Phosphorylation | Decreased Phosphorylation | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| ID | P Site | Accession | FC | p | ID | P Site | Accession | FC | p |
| p47phox | S370 | P14598 | 1.71 | 0.003 | PKACa | S10 | P17612 | −1.74 | 0.03 |
| NFAT3 | S676 | Q14934 | 1.69 | 0.009 | P300 | S89 | Q09472 | −1.61 | 0.007 |
| P27kip1 | T157 | P46527 | 1.64 | 0.004 | PKACa | S338 | P17612 | −1.54 | 0.04 |
| SHC3 | Y341 | Q92529 | 1.62 | 0.004 | IKK-beta | Y188 | O14920 | −1.54 | 0.03 |
| K8 | S74 | P05787 | 1.58 | 0.02 | Lyn | Y396 | P07948 | −1.53 | 0.01 |
| STAT1 | S708 | P42224 | 1.57 | 0.006 | p70S6K | S447 | P23443 | −1.5 | 0.02 |
| Mek2 | S226 | P36507 | 1.57 | 0.04 | MyD88 | Y257 | Q99836 | −1.49 | 0.04 |
| IKK-alpha | S473 | O15111 | 1.55 | 0.02 | Jak2 | Y813 | O60674 | −1.48 | 0.02 |
| Rack1 | Y194 | P63244 | 1.52 | 0.0004 | PKACa | T197 | P17612 | −1.46 | 0.005 |
| CHOP | S79 | P35638 | 1.51 | 0.02 | p67phox | S208 | P19878 | −1.46 | 0.0008 |
| STAT1 | S727 | P42224 | 1.49 | 0.02 | MSK2 | S360 | O75676 | −1.43 | 0.01 |
| MK2 | Y415 | P16389 | 1.48 | 0.01 | IKK-a | S180 | O15111 | −1.42 | 0.02 |
| Rab5A | S123 | P20339 | 1.47 | 0.01 | Lyn | Y507 | P07948 | −1.42 | 0.02 |
| DVL1 | S679 | O14640 | 1.46 | 0.0002 | PPARG | S112 | P37231 | −1.41 | 0.04 |
| PI3K p85 b | Y464 | O00459 | 1.46 | 0.05 | Jak2 | Y119 | O60674 | −1.41 | 0.04 |
| ACC1 | S80 | Q13085 | 1.45 | 0.02 | Mnk1 | T250 | Q9BUB5 | −1.41 | 0.03 |
| Smad6 | S435 | O43541 | 1.41 | 0.02 | p67phox | T233 | P19878 | −1.41 | 0.01 |
| EP300 | S2366 | Q09472 | 1.4 | 0.007 | MAPK14 | T179 | Q16539 | −1.41 | 0.007 |
| 4E-BP1 | T46 | Q13541 | 1.39 | 0.04 | PPP2CA | T304 | P67775 | −1.4 | 0.05 |
| Flt3 | Y842 | P36888 | 1.37 | 0.05 | Pyk2 | S213 | Q14289 | −1.38 | 0.01 |
| Rab5A | Y205 | P20339 | 1.36 | 0.01 | Mnk1 | T255 | Q9BUB5 | −1.37 | 0.008 |
| IRAK4 | T208 | P51617 | 1.34 | 0.01 | NFAT1 | S326 | Q13469 | −1.36 | 0.01 |
| Crk | Y221 | P46108 | 1.34 | 0.04 | STMN1 | S15 | P16949 | −1.34 | 0.02 |
| IFNGR1 | S495 | P15260 | 1.33 | 0.006 | NFAT1 | S110 | Q13469 | −1.32 | 0.01 |
| Grb10 | S150 | Q13322 | 1.33 | 0.02 | IL4R | Y713 | P24394 | −1.32 | 0.01 |
| Cdc42 | Y64 | P60953 | 1.32 | 0.02 | MEK1 | T385 | Q02750 | −1.31 | 0.04 |
| Met | Y1003 | P08581 | 1.32 | 0.02 | STAT5B | S731 | P51692 | −1.3 | 0.01 |
| TGFBR1 | T204 | P36897 | 1.31 | 0.03 | IKK-g | S43 | Q9Y6K9 | −1.29 | 0.03 |
| Sek1 | S80 | P45985 | 1.3 | 0.02 | SMAD3 | S416 | Q15796 | −1.28 | 0.01 |
| CTNNB1 | Y654 | P35222 | 1.3 | 0.05 | Tgfbr2 | S409 | P37173 | −1.27 | 0.005 |
| Calmodulin | Y99 | P62158 | 1.27 | 0.03 | Keap1 | S293 | Q14145 | −1.26 | 0.03 |
| Cdc2 | T161 | P06493 | 1.27 | 0.05 | STAT4 | S722 | Q14765 | −1.25 | 0.05 |
| BRAF1 | S579 | P15056 | 1.26 | 0.02 | Jun | S63 | P05412 | −1.25 | 0.04 |
| IKK-alpha | T23 | O15111 | 1.25 | 0.02 | PIK3R1 | Y528 | P27986 | −1.2 | 0.05 |
| Cdk4 | S150 | P11802 | 1.25 | 0.02 | PDGFRb | Y686 | P09619 | −1.2 | 0.004 |
| NFkB-p65 | S276 | Q04206 | 1.24 | 0.01 | PDK1 | Y376 | O15530 | −1.19 | 0.05 |
| Rab4 | Y189 | P20338 | 1.24 | 0.03 | Pyk2 | S399 | Q14289 | −1.19 | 0.05 |
| Rack1 | Y52 | P63244 | 1.21 | 0.03 | PDGFRb | Y740 | P09619 | −1.17 | 0.004 |
| TBK1 | S172 | Q9UHD2 | 1.2 | 0.02 | Jak1 | Y220 | P23458 | −1.16 | 0.04 |
| TrKA | Y680 | P04629 | 1.19 | 0.04 | PDK1 | S241 | O15530 | −1.15 | 0.02 |
| Grb2 | Y37 | P62993 | 1.18 | 0.02 | MAPK14 | T122 | Q16539 | −1.14 | 0.04 |
| caveolin-1 | Y6 | Q03135 | 1.18 | 0.03 | TAB1 | S423 | Q15750 | −1.12 | 0.05 |
| Shc1 | Y349 | P29353 | 1.18 | 0.03 | ACC1 | S1263 | Q13085_ | −1.11 | 0.04 |
| TRAF6 | Y353 | Q9Y4K3 | 1.18 | 0.03 | Rab4 | S199 | P20338 | −1.06 | 0.03 |
| EGFR | T693 | P00533 | 1.16 | 0.03 | |||||
| PI3Kp85 B | Y605 | O00459 | 1.16 | 0.04 | |||||
| PAK4 | S474 | O96013 | 1.16 | 0.04 | |||||
| p38 delta | Y182 | O15264 | 1.15 | 0.01 | |||||
| IRAK4 | T235 | P51617 | 1.15 | 0.03 | |||||
| gp130 | Y676 | P40189 | 1.15 | 0.04 | |||||
| Kit | Y568 | P10721 | 1.15 | 0.05 | |||||
| MEK1 | S297 | Q02750 | 1.14 | 0.03 | |||||
| 4E-BP1 | S64 | Q13541 | 1.1 | 0.004 | |||||
| Mlk3 | T277 | Q16584 | 1.1 | 0.03 | |||||
| PTEN | Y315 | P60484 | 1.1 | 0.04 | |||||
| TAK1 | T187 | O43318 | 1.03 | 0.04 | |||||
| Increased Phosphorylation | Decreased Phosphorylation | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| ID | P Site | Accession | FC | p | ID | P Site | Accession | FC | p |
| ACC1 | S29 | Q13085 | 1.07 | 0.0007 | SHC3 | Y341 | Q92529 | −1.19 | 0.04 |
| NFkB-p65 | S536 | Q04206 | −1.17 | 0.02 | |||||
| NFAT3 | S676 | Q14934 | −1.16 | 0.02 | |||||
| IKK-alpha | S180 | O15111 | −1.15 | 0.02 | |||||
| TAK1 | S192 | O43318 | −1.1 | 0.04 | |||||
| P27kip1 | T1576 | P46527 | −1.08 | 0.01 | |||||
| Pathway Name | Pathway ID | Source Name | Gene Count | p-Value (Corrected) |
|---|---|---|---|---|
| EPO signaling pathway | 16151 | INOH | 32 | 1.12 × 1033 |
| IL-7 signaling | 16106 | INOH | 31 | 1.45 × 1032 |
| JAK STAT pathway | 16125 | INOH | 35 | 1.60 × 1032 |
| VEGF signaling pathway | 16190 | INOH | 29 | 3.32 × 1029 |
| Pathways in cancer | 4397 | KEGG | 33 | 3.74 × 1027 |
| Signaling by Interleukins | 18744 | REACTOME | 23 | 1.14 × 1025 |
| TGF_beta_Receptor | 15911 | NETPATH | 27 | 4.49 × 1024 |
| IL2 | 15918 | NETPATH | 20 | 9.23 × 1024 |
| BCR | 15916 | NETPATH | 24 | 1.41 × 1023 |
| IL6 | 15922 | NETPATH | 20 | 2.16 × 1023 |
| Signaling by NGF | 16818 | REACTOME | 28 | 3.77 × 1023 |
| MAPK signaling pathway | 487 | KEGG | 27 | 1.87 × 1022 |
| Osteoclast differentiation | 10367 | KEGG | 22 | 2.19 × 1022 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Awate, S.; Scruten, E.; Mutwiri, G.; Napper, S. Kinome Analysis to Define Mechanisms of Adjuvant Action: PCEP Induces Unique Signaling at the Injection Site and Lymph Nodes. Vaccines 2022, 10, 927. https://doi.org/10.3390/vaccines10060927
Awate S, Scruten E, Mutwiri G, Napper S. Kinome Analysis to Define Mechanisms of Adjuvant Action: PCEP Induces Unique Signaling at the Injection Site and Lymph Nodes. Vaccines. 2022; 10(6):927. https://doi.org/10.3390/vaccines10060927
Chicago/Turabian StyleAwate, Sunita, Erin Scruten, George Mutwiri, and Scott Napper. 2022. "Kinome Analysis to Define Mechanisms of Adjuvant Action: PCEP Induces Unique Signaling at the Injection Site and Lymph Nodes" Vaccines 10, no. 6: 927. https://doi.org/10.3390/vaccines10060927
APA StyleAwate, S., Scruten, E., Mutwiri, G., & Napper, S. (2022). Kinome Analysis to Define Mechanisms of Adjuvant Action: PCEP Induces Unique Signaling at the Injection Site and Lymph Nodes. Vaccines, 10(6), 927. https://doi.org/10.3390/vaccines10060927







