Neurocognitive Profile of the Post-COVID Condition in Adults in Catalonia—A Mixed Method Prospective Cohort and Nested Case–Control Study: Study Protocol
Abstract
:1. Introduction
- Analyze the structural and functional changes in the brain, the eye, and the vestibular system in COVID-19 patients, employing functional and structural magnetic resonance imaging (MRI) studies, retinography, and posturography;
- Analyze the neuropsychological status, emotional state, and quality of life using neuropsychological questionnaires;
- Analyze different inflammatory and immune biomarkers produced as a response to SARS-CoV-2;
- Investigate the relationship between factors and biomarkers acquired in 1–3.
- Describe the impact of persistent symptoms in the context of daily life through individualized interviews and four focus groups (the same ones as for the quantitative part).
2. Materials and Methods
2.1. Design
- -
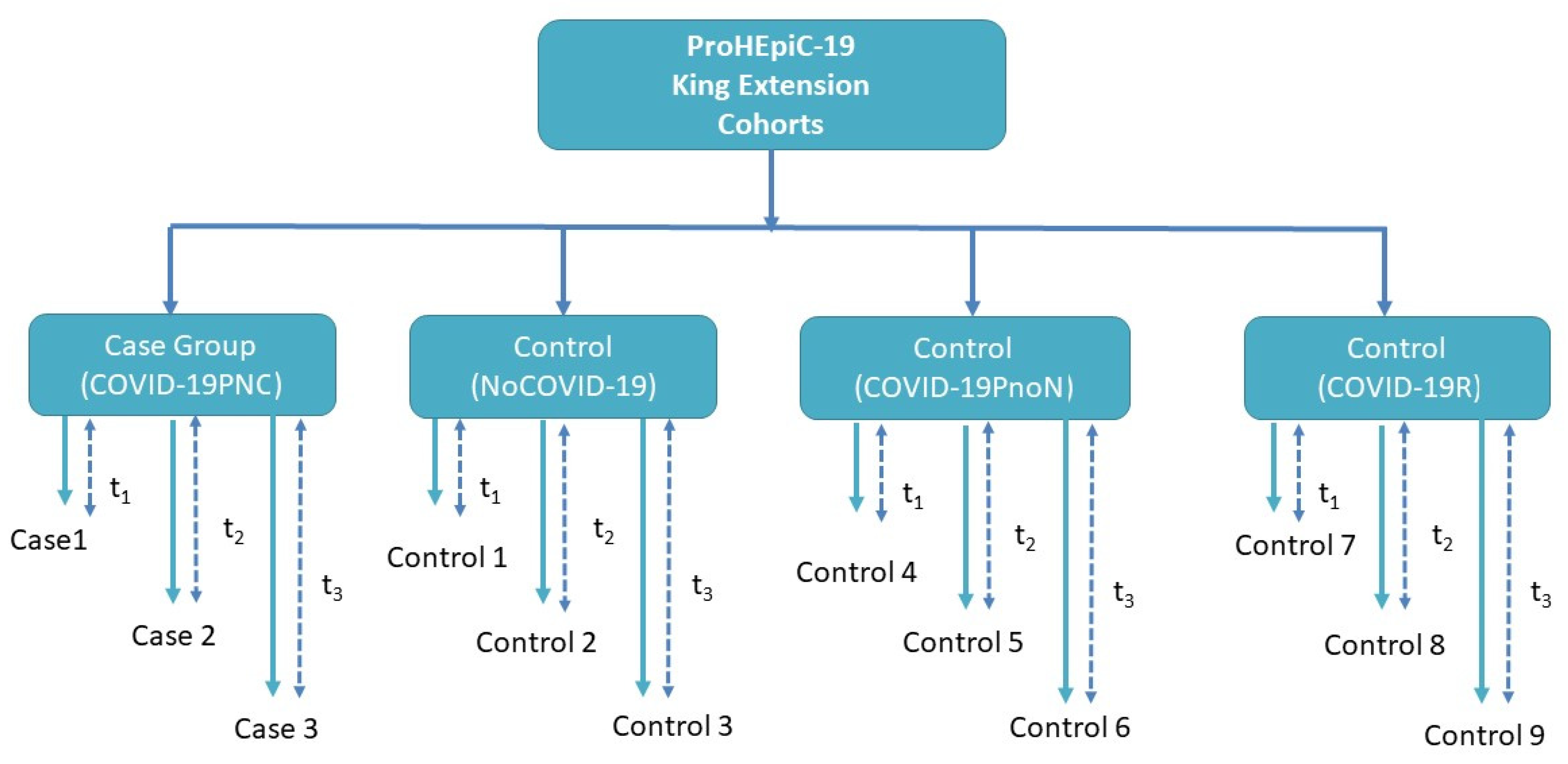
- Control group 0 (NoCOVID-19) includes 40 people who are not infected with SARS-CoV2.
- -
- Control group 1 (COVID-19PnoN) includes 40 people suffering from PCC but without any neurocognitive deficit, according to the WHO definition, 2021.
- -
- Control group 2 (COVID-19R) includes 40 people who have recovered from SARS-CoV-2 infection, without cognitive impairment or psychiatric symptoms prior to COVID-19 infection (See Figure 1).
2.2. Participants, Exposure
2.3. Recruitment
2.4. Sample Size and Sampling Procedure
2.5. Variables
2.5.1. Demographical and Clinical Data
2.5.2. Clinical Variables
- Neurological symptomatology include headache, change in vision, change in hearing, ageusia, anosmia, tremor, fatigue, myalgia (presence or absence of the sign or symptom).
- Neuropsychiatric symptomatology will be measured using the following tests: Hospital Anxiety Depression Scale (HADS); [64] Geriatric Depression Scale 15-item version (GDS); [65] Pittsburgh Sleep Quality Index (PSQI); [66] Stress Disorder Symptom Severity Scale according to the DSM-V (SDSSS); [67] obsessive-compulsive disorder according to DSM-V (OCD), [67].
2.5.3. Neurocognitive Variables
- To measure cognitive performance, all participants will complete an extensive neuropsychological examination with the following tests, which provide measures of multiple cognitive functions. Abstract reasoning and fluid intelligence (Intelligence Vocabulary and Matrix Reasoning, WAIS-III [68]; attention (forward span, WAIS-III [68]; working memory (backward, WAIS-III [68]; visuospatial speed (Symbol Search, WAIS-III; [68] Symbol Coding, WAIS-III [68]; Trail Making Test-A [69]; copy time Rey–Osterrieth Complex Figure [70]; visuospatial and visuoconstructive function (copy accuracy Rey–Osterrieth Complex Figure [70]; verbal memory (total learning and recall-II Rey Auditory Verbal Learning Test [71]; visual memory (memory accuracy, Rey–Osterrieth Complex Figure [70]; language (Boston Naming Test-15 [72]); flexibility (Trail Making Test B-A time); [69] fluency (letter and category fluency); [73] inhibition (interference, Stroop Test [74].
- Emotional status will be measured using the following tests: the Emotion Regulation Questionnaire [75], which is a 10-item scale designed to measure respondents’ tendency to regulate their emotions in the following two ways: (1) cognitive reappraisal and (2) expressive suppression. Respondents answer each item on a 7-point Likert-type scale ranging from 1 (strongly disagree) to 7 (strongly agree). The Impact of Event Scale-Revised [76] is a self-report questionnaire with 22 questions to capture the DSM-IV criteria for PTSD with a special focus on intrusion, avoidance, hyperarousal, and total subjective stress.
2.5.4. Neuroimaging and Neurophysiological Variables
- Brain structural changes will be measured with structural MRI, including volumetry of different anatomical regions, cortical thickness, and structural connectivity of principal white matter tracts.
- Brain functional changes will be measured with functional MRI, including the activation level of functional networks and whole-brain functional connectivity as measured with GraphVar.
- For retinal microcirculation disorders, retinography with an amidriatic camera (TRC-NW8) and retinal microvascular analysis (SIRIUS software) to assess the ratio of artery/vessel and the tortuosity will be used.
- For balance and gait disorders, gait, gait speed, and balance as measured with posturography and dynamometric platform (Dinascan/IBVP600) will be considered.
2.5.5. Inflammatory and Immunology Markers
- C-reactive protein.
- Serum antibodies against SARS-CoV-2 (IgM, IgG (Spike), IgG (Nucleocapsid)).
- Plasma cytokines IL-6, IL-8, IL-12, TNF-α, IFN-α, MCP-1, TGF-β1, and IL-15 measured Simoa multiplex immunoassay platform and analyzed using HD1 analyzer (Quanterix, Lexington, MA, USA). This technology is sensitive enough to overcome the limitation that some of these cytokines are poorly released in case of impaired functionality and are, therefore, difficult to detect and quantify in plasma. Light plasma neurofilament (NfL) and glial fibrillar acid protein (GFAP) will be measured using Neurology 2-Plex B, 103520.
2.5.6. Lifestyle-Related Variables
- The Mediterranean diet assessment tool (PREDIMED) [77] is a brief dietary assessment instrument that consists of 14 short questions whose evaluation aims to offer information about adherence to the Mediterranean diet.
- For physical activity, the International Physical Activity Questionnaire (IPAQ) [78] will be used. It is a 27-item self-reported measure of physical activity for use with individual adult patients aged 15–69 years old.
- The Sedentary Behavior Questionary (SBQ) [79] is a summary measure of total sedentary time.
- Quality of life (EuroQol-5D) [80].
- For impairment in everyday life, narratives emerging from the 30 semi-structured interviews, identifying specific aspects of the experience and delving into their details, will be used. Contingent repertoires of sociolinguistic resources will be shared within the 4-focus group, allowing participants to give meaning to their experiences.
2.6. Statistical Analysis Plan
2.6.1. Quantitative Methodology
Data Preprocessing
Statistical and Qualitative Analysis per Aims
2.6.2. Integration of Quantitative and Qualitative Approaches
2.7. Ethical Considerations
3. Discussion
3.1. Expected Results
3.2. Impact
3.2.1. Public Health Policies
3.2.2. Scientific Impact
3.2.3. Economic Impact
3.2.4. Innovation
3.3. Strengths and Limitations
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Moghadas, S.M.; Vilches, T.N.; Zhang, K.; Wells, C.R.; Shoukat, A.; Singer, B.H.; Meyers, L.A.; Neuzil, K.M.; Langley, J.M.; Fitzpatrick, M.C.; et al. The Impact of Vaccination on Coronavirus Disease 2019 (COVID-19) Outbreaks in the United States. Clin. Infect. Dis. 2021, 73, 2257–2264. [Google Scholar] [CrossRef]
- Soleimanpour, S.; Yaghoubi, A. COVID-19 vaccine: Where are we now and where should we go? Expert Rev. Vaccines 2021, 20, 23–44. [Google Scholar] [CrossRef]
- Murchu, E.O.; Byrne, P.; Carty, P.G.; De Gascun, C.; Keogan, M.; O’Neill, M.; Harrington, P.; Ryan, M. Quantifying the risk of SARS-CoV-2 reinfection over time. Rev. Med. Virol. 2022, 32, e2260. [Google Scholar] [CrossRef]
- Soriano, J.B.; Murthy, S.; Marshall, J.C.; Relan, P.; Diaz, J.V. A clinical case definition of post-COVID-19 condition by a Delphi consensus. Lancet Infect. Dis. 2021, 22, E102–E107. [Google Scholar] [CrossRef]
- López-Sampalo, A.; Bernal-López, M.R.; Gómez-Huelgas, R. Síndrome de COVID-19 persistente. Una revisión narrativa. Rev. Clín. Esp. 2021, 222, 141–150. [Google Scholar] [CrossRef]
- Torjesen, I. COVID-19: Middle aged women face greater risk of debilitating long term symptoms. BMJ 2021, 372, n829. [Google Scholar] [CrossRef]
- Vivanco-Hidalgo, R.M.; Pons Ràfols, J.M.V.; Moharra, M. Símptomes de Llarga Durada o Persistents en Pacients amb Infecció Lleumoderada per SARS-CoV-2 (COVID-19): Revisió Ràpida de la Literatura i Discussió de L’evidència Disponible; Agència de Qualitat i Avaluació Sanitàries de Catalunya: Barcelona, Spain, 2020.
- Oliveira, R.; Sotero, F.D.; Teodoro, T. NeuroCOVID: Critical review of neuropsychiatric manifestations of SARS-CoV-2 infection. Irish J. Med. Sci. 2021, 190, 851–852. [Google Scholar] [CrossRef]
- Almeria, M.; Cejudo, J.C.; Sotoca, J.; Deus, J.; Krupinski, J. Cognitive profile following COVID-19 infection: Clinical predictors leading to neuropsychological impairment. Brain Behav. Immun.-Health 2020, 9, 100163. [Google Scholar] [CrossRef]
- Søraas, A.; Bø, R.; Kalleberg, K.T.; Støer, N.C.; Ellingjord-Dale, M.; Landrø, N.I. Self-reported Memory Problems 8 Months after COVID-19 Infection. JAMA Netw. Open 2021, 4, e2118717. [Google Scholar] [CrossRef]
- Whiteside, D.M.; Oleynick, V.; Holker, E.; Waldron, E.J.; Porter, J.; Kasprzak, M. Neurocognitive deficits in severe COVID-19 infection: Case series and proposed model. Clin. Neuropsychol. 2021, 35, 799–818. [Google Scholar] [CrossRef]
- Negrini, F.; Ferrario, I.; Mazziotti, D.; Berchicci, M.; Bonazzi, M.; de Sire, A.; Negrini, S.; Zapparoli, L. Neuropsychological Features of Severe Hospitalized Coronavirus Disease 2019 Patients at Clinical Stability and Clues for Postacute Rehabilitation. Arch. Phys. Med. Rehabil. 2021, 102, 155–158. [Google Scholar] [CrossRef] [PubMed]
- Hellmuth, J.; Barnett, T.A.; Asken, B.M.; Kelly, J.D.; Torres, L.; Stephens, M.L.; Greenhouse, B.; Martin, J.N.; Chow, F.C.; Deeks, S.G.; et al. Persistent COVID-19-associated neurocognitive symptoms in non-hospitalized patients. J. Neurovirol. 2021, 27, 191–195. [Google Scholar] [CrossRef] [PubMed]
- Méndez, R.; Balanzá-Martínez, V.; Luperdi, S.C.; Estrada, I.; Latorre, A.; González-Jiménez, P.; Bouzas, L.; Yépez, K.; Ferrando, A.; Reyes, S.; et al. Long-term neuropsychiatric outcomes in COVID-19 survivors: A 1-year longitudinal study. J. Intern. Med. 2021, 291, 247–251. [Google Scholar] [CrossRef]
- Zhou, H.; Lu, S.; Chen, J.; Wei, N.; Wang, D.; Lyu, H.; Shi, C.; Hu, S. The landscape of cognitive function in recovered COVID-19 patients. J. Psychiatr. Res. 2020, 129, 98–102. [Google Scholar] [CrossRef]
- Orrù, G.; Bertelloni, D.; Diolaiuti, F.; Mucci, F.; Di Giuseppe, M.; Biella, M.; Gemignani, A.; Ciacchini, R.; Conversano, C. Long-COVID Syndrome? A Study on the Persistence of Neurological, Psychological and Physiological Symptoms. Healthcare 2021, 9, 575. [Google Scholar] [CrossRef]
- Hampshire, A.; Trender, W.; Chamberlain, S.R.; Jolly, A.E.; Grant, J.E.; Patrick, F.; Mazibuko, N.; Williams, S.C.; Barnby, J.M.; Hellyer, P.; et al. Cognitive deficits in people who have recovered from COVID-19. EClinicalMedicine 2021, 39, 101044. [Google Scholar] [CrossRef]
- García-Sánchez, C.; Calabria, M.; Grunden, N.; Pons, C.; Arroyo, J.A.; Gómez-Anson, B.; Lleó, A.; Alcolea, D.; Belvís, R.; Morollón, N.; et al. Neuropsychological deficits in patients with cognitive complaints after COVID-19. Brain Behav. 2022, 12, e2508. [Google Scholar] [CrossRef]
- Xu, Z.; Shi, L.; Wang, Y.; Zhang, J.; Huang, L.; Zhang, C.; Liu, S.; Zhao, P.; Liu, H.; Zhu, L.; et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir. Med. 2020, 8, 420–422. [Google Scholar] [CrossRef]
- Albu, S.; Zozaya, N.R.; Murillo, N.; García-Molina, A.; Chacón, C.A.F.; Kumru, H. What’s going on following acute COVID-19? Clinical characteristics of patients in an out-patient rehabilitation program. NeuroRehabilitation 2021, 48, 469–480. [Google Scholar] [CrossRef]
- Graham, E.L.; Clark, J.R.; Orban, Z.S.; Lim, P.H.; Szymanski, A.L.; Taylor, C.; DiBiase, R.M.; Jia, D.T.; Balabanov, R.; Ho, S.U.; et al. Persistent neurologic symptoms and cognitive dysfunction in non-hospitalized COVID-19 “long haulers”. Ann. Clin. Transl. Neurol. 2021, 8, 1073–1085. [Google Scholar] [CrossRef]
- Dressing, A.; Bormann, T.; Blazhenets, G.; Schroeter, N.; Walter, L.I.; Thurow, J.; August, D.; Hilger, H.; Stete, K.; Gerstacker, K.; et al. Neuropsychological profiles and cerebral glucose metabolism in neurocognitive Long COVID-syndrome. J. Nucl. Med. 2021, 63, jnumed.121.262677. [Google Scholar] [CrossRef] [PubMed]
- Boldrini, M.; Canoll, P.D.; Klein, R.S. How COVID-19 Affects the Brain. JAMA Psychiatry 2021, 78, 682. [Google Scholar] [CrossRef] [PubMed]
- Ellul, M.A.; Benjamin, L.; Singh, B.; Lant, S.; Michael, B.D.; Easton, A.; Kneen, R.; Defres, S.; Sejvar, J.; Solomon, T. Neurological associations of COVID-19. Lancet Neurol. 2020, 19, 767–783. [Google Scholar] [CrossRef]
- Reddy, S.T.; Garg, T.; Shah, C.; Nascimento, F.A.; Imran, R.; Kan, P.; Bowry, R.; Gonzales, N.; Barreto, A.; Kumar, A.; et al. Cerebrovascular Disease in Patients with COVID-19: A Review of the Literature and Case Series. Case Rep. Neurol. 2020, 12, 199–209. [Google Scholar] [CrossRef] [PubMed]
- Dickman, M.S. Von Economo Encephalitis. Arch. Neurol. 2001, 58, 1696. [Google Scholar] [CrossRef]
- Wright, D.; Rowley, R.; Halks-Wellstead, P.; Anderson, T.; Wu, T.Y. Abnormal Saccadic Oscillations Associated with Severe Acute Respiratory Syndrome Coronavirus 2 Encephalopathy and Ataxia. Mov. Disord. Clin. Pract. 2020, 7, 980–982. [Google Scholar] [CrossRef] [PubMed]
- Schellekens, M.M.I.; Bleeker-Rovers, C.P.; Keurlings, P.A.J.; Mummery, C.J.; Bloem, B.R. Reversible Myoclonus-Ataxia as a Postinfectious Manifestation of COVID-19. Mov. Disord. Clin. Pract. 2020, 7, 977–979. [Google Scholar] [CrossRef]
- Zaffina, S.; Lanteri, P.; Gilardi, F.; Garbarino, S.; Santoro, A.; Vinci, M.R.; Carsetti, R.; Scorpecci, A.; Raponi, M.; Magnavita, N.; et al. Recurrence, Reactivation, or Inflammatory Rebound of SARS-CoV-2 Infection with Acute Vestibular Symptoms: A Case Report and Revision of Literature. Front. Hum. Neurosci. 2021, 15, 444. [Google Scholar] [CrossRef]
- Fancello, V.; Hatzopoulos, S.; Corazzi, V.; Bianchini, C.; Skarżyńska, M.B.; Pelucchi, S.; Skarżyński, P.H.; Ciorba, A. SARS-CoV-2 (COVID-19) and audio-vestibular disorders. Int. J. Immunopathol. Pharmacol. 2021, 35, 205873842110273. [Google Scholar] [CrossRef]
- Invernizzi, A.; Torre, A.; Parrulli, S.; Zicarelli, F.; Schiuma, M.; Colombo, V.; Giacomelli, A.; Cigada, M.; Milazzo, L.; Ridolfo, A.; et al. Retinal findings in patients with COVID-19: Results from the SERPICO-19 study. EClinicalMedicine 2020, 27, 100550. [Google Scholar] [CrossRef]
- Finn, A.P.; Khurana, R.N.; Chang, L.K. Hemi-retinal vein occlusion in a young patient with COVID-19. Am. J. Ophthalmol. Case Rep. 2021, 22, 101046. [Google Scholar] [CrossRef]
- Rehak, M.; Wiedemann, P. Retinal vein thrombosis: Pathogenesis and management. J. Thromb. Haemost. 2010, 8, 1886–1894. [Google Scholar] [CrossRef]
- Virgo, J.; Mohamed, M. Paracentral acute middle maculopathy and acute macular neuroretinopathy following SARS-CoV-2 infection. Eye 2020, 34, 2352–2353. [Google Scholar] [CrossRef] [PubMed]
- Forés, R.; Manresa, J.M.; López-Lifante, V.M.; Heras, A.; Delgado, P.; Vázquez, X.; Ruiz, S.; Alzamora, M.T.; Toran, P. Relationship between Retinal Microvasculature, Cardiovascular Risk and Silent Brain Infarction in Hypertensive Patients. Diagnostics 2021, 11, 937. [Google Scholar] [CrossRef]
- Lopez-Leon, S.; Wegman-Ostrosky, T.; Perelman, C.; Sepulveda, R.; Rebolledo, P.A.; Cuapio, A.; Villapol, S. More than 50 long-term effects of COVID-19: A systematic review and meta-analysis. Sci. Rep. 2021, 11, 16144. [Google Scholar] [CrossRef] [PubMed]
- Carfì, A.; Bernabei, R.; Landi, F. Persistent Symptoms in Patients after Acute COVID-19. JAMA 2020, 324, 603. [Google Scholar] [CrossRef]
- Deng, J.; Zhou, F.; Hou, W.; Silver, Z.; Wong, C.Y.; Chang, O.; Huang, E.; Zuo, Q.K. The prevalence of depression, anxiety, and sleep disturbances in COVID-19 patients: A meta-analysis. Ann. N. Y. Acad. Sci. 2021, 1486, 90–111. [Google Scholar] [CrossRef] [PubMed]
- Mandal, S.; Barnett, J.; Brill, S.E.; Brown, J.S.; Denneny, E.K.; Hare, S.S.; Heightman, M.; Hillman, T.E.; Jacob, J.; Jarvis, H.C.; et al. ‘Long-COVID’: A cross-sectional study of persisting symptoms, biomarker and imaging abnormalities following hospitalisation for COVID-19. Thorax 2021, 76, 396–398. [Google Scholar] [CrossRef] [PubMed]
- Varatharaj, A.; Thomas, N.; Ellul, M.A.; Davies, N.W.S.; Pollak, T.A.; Tenorio, E.L.; Sultan, M.; Easton, A.; Breen, G.; Zandi, M.; et al. Neurological and neuropsychiatric complications of COVID-19 in 153 patients: A UK-wide surveillance study. Lancet Psychiatry 2020, 7, 875–882. [Google Scholar] [CrossRef]
- Carruthers, B.M.; van de Sande, M.I.; De Meirleir, K.L.; Klimas, N.G.; Broderick, G.; Mitchell, T.; Staines, D.; Powles, A.C.P.; Speight, N.; Vallings, R.; et al. Myalgic encephalomyelitis: International Consensus Criteria. J. Intern. Med. 2011, 270, 327–338. [Google Scholar] [CrossRef] [Green Version]
- Nath, A. Long-Haul COVID. Neurology 2020, 95, 559–560. [Google Scholar] [CrossRef] [PubMed]
- Yazdanpanah, F.; Garg, A.; Shadman, S.; Asmarz, H.Y. Literature Review of COVID-19, Pulmonary and Extrapulmonary Disease. Am. J. Med. Sci. 2021, 361, 567–574. [Google Scholar] [CrossRef] [PubMed]
- Taquet, M.; Geddes, J.R.; Husain, M.; Luciano, S.; Harrison, P.J. 6-month neurological and psychiatric outcomes in 236,379 survivors of COVID-19: A retrospective cohort study using electronic health records. Lancet Psychiatry 2021, 8, 416–427. [Google Scholar] [CrossRef]
- Taquet, M.; Dercon, Q.; Luciano, S.; Geddes, J.R.; Husain, M.; Harrison, P.J. Incidence, co-occurrence, and evolution of long-COVID features: A 6-month retrospective cohort study of 273,618 survivors of COVID-19. PLoS Med. 2021, 18, e1003773. [Google Scholar] [CrossRef]
- Zolnikov, T.R.; Clark, T.; Zolnikov, T. Likely Exacerbation of Psychological Disorders from COVID-19 Response. J. Prim. Care Community Health 2021, 12, 215013272110167. [Google Scholar] [CrossRef]
- Taquet, M.; Luciano, S.; Geddes, J.R.; Harrison, P.J. Bidirectional associations between COVID-19 and psychiatric disorder: Retrospective cohort studies of 62,354 COVID-19 cases in the USA. Lancet Psychiatry 2021, 8, 130–140. [Google Scholar] [CrossRef]
- Troyer, E.A.; Kohn, J.N.; Hong, S. Are we facing a crashing wave of neuropsychiatric sequelae of COVID-19? Neuropsychiatric symptoms and potential immunologic mechanisms. Brain Behav. Immun. 2020, 87, 34–39. [Google Scholar] [CrossRef]
- Wijeratne, T.; Crewther, S. COVID-19 and long-term neurological problems: Challenges ahead with Post-COVID-19 Neurological Syndrome. Aust. J. Gen. Pract. 2021, 50. [Google Scholar] [CrossRef]
- Puchner, B.; Sahanic, S.; Kirchmair, R.; Pizzini, A.; Sonnweber, B.; Wöll, E.; Mühlbacher, A.; Garimorth, K.; Dareb, B.; Ehling, R.; et al. Beneficial effects of multi-disciplinary rehabilitation in postacute COVID-19: An observational cohort study. Eur. J. Phys. Rehabil. Med. 2021, 57, S1973–S9087. [Google Scholar] [CrossRef]
- Rogers, J.P.; Chesney, E.; Oliver, D.; Pollak, T.A.; McGuire, P.; Fusar-Poli, P.; Zandi, M.S.; Lewis, G.; David, A.S. Psychiatric and neuropsychiatric presentations associated with severe coronavirus infections: A systematic review and meta-analysis with comparison to the COVID-19 pandemic. Lancet Psychiatry 2020, 7, 611–627. [Google Scholar] [CrossRef]
- Zanin, L.; Saraceno, G.; Panciani, P.P.; Renisi, G.; Signorini, L.; Migliorati, K.; Fontanella, M.M. SARS-CoV-2 can induce brain and spine demyelinating lesions. Acta Neurochir. 2020, 162, 1491–1494. [Google Scholar] [CrossRef]
- Kuba, K.; Yamaguchi, T.; Penninger, J.M. Angiotensin-Converting Enzyme 2 (ACE2) in the Pathogenesis of ARDS in COVID-19. Front. Immunol. 2021, 12, 732690. [Google Scholar] [CrossRef]
- Cosarderelioglu, C.; Nidadavolu, L.S.; George, C.J.; Oh, E.S.; Bennett, D.A.; Walston, J.D.; Abadir, P.M. Brain Renin–Angiotensin System at the Intersect of Physical and Cognitive Frailty. Front. Neurosci. 2020, 14, 981. [Google Scholar] [CrossRef] [PubMed]
- Merino, J.J.; Macho-González, A.; Benedi, J.; González, M.P. Neurological manifestations of COVID-19 in patients: From path physiology to therapy. Neurol. Sci. 2021, 42, 4867–4879. [Google Scholar] [CrossRef] [PubMed]
- Desai, I.; Manchanda, R.; Kumar, N.; Tiwari, A.; Kumar, M. Neurological manifestations of coronavirus disease 2019: Exploring past to understand present. Neurol. Sci. 2021, 42, 773–785. [Google Scholar] [CrossRef] [PubMed]
- Hugon, J. Long-COVID: Cognitive Deficits (Brain Fog) and Brain Lesions in Non-Hospitalized Patients. Presse Med. 2021, 51, 104090. [Google Scholar] [CrossRef] [PubMed]
- Lichenstein, S.D.; Verstynen, T.; Forbes, E.E. Adolescent brain development and depression: A case for the importance of connectivity of the anterior cingulate cortex. Neurosci. Biobehav. Rev. 2016, 70, 271–287. [Google Scholar] [CrossRef] [Green Version]
- Ma, X.; Reynolds, S.L.; Baker, B.J.; Li, X.; Benveniste, E.N.; Qin, H. IL-17 Enhancement of the IL-6 Signaling Cascade in Astrocytes. J. Immunol. 2010, 184, 4898–4906. [Google Scholar] [CrossRef] [Green Version]
- Lindqvist, D.; Dhabhar, F.S.; Mellon, S.H.; Yehuda, R.; Grenon, S.M.; Flory, J.D.; Bierer, L.M.; Abu-Amara, D.; Coy, M.; Makotkine, I.; et al. Increased pro-inflammatory milieu in combat related PTSD—A new cohort replication study. Brain Behav. Immun. 2017, 59, 260–264. [Google Scholar] [CrossRef] [Green Version]
- Karagüzel, E.Ö.; Arslan, F.C.; Uysal, E.K.; Demir, S.; Aykut, D.S.; Tat, M.; Karahan, S.C. Blood levels of interleukin-1 beta, interleukin-6 and tumor necrosis factor-alpha and cognitive functions in patients with obsessive compulsive disorder. Compr. Psychiatry 2019, 89, 61–66. [Google Scholar] [CrossRef]
- Parker, A.M.; Sricharoenchai, T.; Raparla, S.; Schneck, K.W.; Bienvenu, O.J.; Needham, D.M. Posttraumatic Stress Disorder in Critical Illness Survivors. Crit. Care Med. 2015, 43, 1121–1129. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Q.; Xu, M.; Li, J.; Liu, Y.; Zhang, J.; Xu, Y.; Dong, W. Clinical sequelae of COVID-19 survivors in Wuhan, China: A single-centre longitudinal study. Clin. Microbiol. Infect. 2021, 27, 89–95. [Google Scholar] [CrossRef]
- Zigmond, A.S.; Snaith, R.P. The Hospital Anxiety and Depression Scale. Acta Psychiatr. Scand. 1983, 67, 361–370. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yesavage, J.A.; Sheikh, J.I. 9/Geriatric Depression Scale (GDS). Clin. Gerontol. 1986, 5, 165–173. [Google Scholar] [CrossRef]
- Buysse, D.J.; Reynolds, C.F.; Monk, T.H.; Berman, S.R.; Kupfer, D.J. The Pittsburgh sleep quality index: A new instrument for psychiatric practice and research. Psychiatry Res. 1989, 28, 193–213. [Google Scholar] [CrossRef]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; APA: Washington, DC, USA, 2013. [Google Scholar]
- Wechsler, D. WAIS-III. Escala de Inteligencia de Wechsler Para Adultos; Ediciones, T., Ed.; TEA Ediciones: Madrid, Spain, 2001. [Google Scholar]
- Tombaugh, T. Trail Making Test A and B: Normative data stratified by age and education. Arch. Clin. Neuropsychol. 2004, 19, 203–214. [Google Scholar] [CrossRef]
- Rey, A. REY. Test de Copia de una Figura Compleja; TEA Ediciones: Madrid, Spain, 2009. [Google Scholar]
- Schmidt, M. Rey Auditory Verbal Learning Test: A Handbook; Western Psychological Services: Los Angeles, CA, USA, 1996. [Google Scholar]
- Goodglass, H.; Kaplan, E.; Barresi, B. Test de Boston Para el Diagnóstico de la Afasia, 3rd ed.; Editorial Médica Panamericana: Madrid, Spain, 2001. [Google Scholar]
- Pena-Casanova, J.; Quinones-Ubeda, S.; Gramunt-Fombuena, N.; Quintana-Aparicio, M.; Aguilar, M.; Badenes, D.; Cerulla, N.; Molinuevo, J.L.; Ruiz, E.; Robles, A.; et al. Spanish Multicenter Normative Studies (NEURONORMA Project): Norms for Verbal Fluency Tests. Arch. Clin. Neuropsychol. 2009, 24, 395–411. [Google Scholar] [CrossRef] [Green Version]
- Golden, C.J. Stroop. Test de Colores y Palabras, 3rd ed.; TEA Ediciones: Madrid, Spain, 2001. [Google Scholar]
- Gross, J.J.; John, O.P. Individual differences in two emotion regulation processes: Implications for affect, relationships, and well-being. J. Pers. Soc. Psychol. 2003, 85, 348–362. [Google Scholar] [CrossRef]
- Weiss, D.S. The impact of event scale—Revised. In Assessing Psychological Trauma and PTSD; Wilson, J.P., Tang, S.-K., Eds.; Guilford Press: New York, NY, USA, 1997; pp. 399–411. [Google Scholar]
- Ros, E. The PREDIMED study. Endocrinol. Diabetes Nutr. (Engl. Ed.) 2017, 64, 63–66. [Google Scholar] [CrossRef]
- Craig, C.L.; Marshall, A.L.; SJÖSTRÖM, M.; Bauman, A.E.; Booth, M.L.; Ainsworth, B.E.; Pratt, M.; Ekelund, U.; Yngve, A.; Sallis, J.F.; et al. International Physical Activity Questionnaire: 12-Country Reliability and Validity. Med. Sci. Sport. Exerc. 2003, 35, 1381–1395. [Google Scholar] [CrossRef] [Green Version]
- Rosenberg, D.E.; Norman, G.J.; Wagner, N.; Patrick, K.; Calfas, K.J.; Sallis, J.F. Reliability and validity of the sedentary behavior questionnaire (SBQ) for adults. J. Phys. Act. Health 2010, 7, 697–705. [Google Scholar] [CrossRef] [PubMed]
- Herdman, M.; Badia, X.; Berra, S. El EuroQol-5D: Una alternativa sencilla para la medición de la calidad de vida relacionada con la salud en atención primaria. Atención Primaria 2001, 28, 425–429. [Google Scholar] [CrossRef] [Green Version]
- Smith, J.A.; Shinebourne, P. Research designs: Quantitative, qualitative, neuropsychological, and biological. In Interpretative Phenomenological Analysis; Cooper, P.M., Camic, D.L., Long, A.T., Panter, D., Rindskopf, K.J.S., Eds.; APA: Washington, DC, USA, 2012; pp. 73–82. [Google Scholar]
- Ladds, E.; Rushforth, A.; Wieringa, S.; Taylor, S.; Rayner, C.; Husain, L.; Greenhalgh, T. Persistent symptoms after COVID-19: Qualitative study of 114 “long COVID” patients and draft quality principles for services. BMC Health Serv. Res. 2020, 20, 1144. [Google Scholar] [CrossRef] [PubMed]
- Nalbandian, A.; Sehgal, K.; Gupta, A.; Madhavan, M.V.; McGroder, C.; Stevens, J.S.; Cook, J.R.; Nordvig, A.S.; Shalev, D.; Sehrawat, T.S.; et al. Post-acute COVID-19 syndrome. Nat. Med. 2021, 27, 601–615. [Google Scholar] [CrossRef] [PubMed]
- Living with COVID-19—Second Review. 2021. Available online: https://evidence.nihr.ac.uk/themedreview/living-with-covid19-second-review/ (accessed on 24 April 2021).
- Verguet, S.; Hailu, A.; Eregata, G.T.; Memirie, S.T.; Johansson, K.A.; Norheim, O.F. Toward universal health coverage in the post-COVID-19 era. Nat. Med. 2021, 27, 380–387. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dacosta-Aguayo, R.; Lamonja-Vicente, N.; Chacón, C.; Carrasco-Ribelles, L.A.; Montero-Alia, P.; Costa-Garrido, A.; García-Sierra, R.; López-Lifante, V.M.; Moreno-Gabriel, E.; Massanella, M.; et al. Neurocognitive Profile of the Post-COVID Condition in Adults in Catalonia—A Mixed Method Prospective Cohort and Nested Case–Control Study: Study Protocol. Vaccines 2022, 10, 849. https://doi.org/10.3390/vaccines10060849
Dacosta-Aguayo R, Lamonja-Vicente N, Chacón C, Carrasco-Ribelles LA, Montero-Alia P, Costa-Garrido A, García-Sierra R, López-Lifante VM, Moreno-Gabriel E, Massanella M, et al. Neurocognitive Profile of the Post-COVID Condition in Adults in Catalonia—A Mixed Method Prospective Cohort and Nested Case–Control Study: Study Protocol. Vaccines. 2022; 10(6):849. https://doi.org/10.3390/vaccines10060849
Chicago/Turabian StyleDacosta-Aguayo, Rosalia, Noemí Lamonja-Vicente, Carla Chacón, Lucia Amalía Carrasco-Ribelles, Pilar Montero-Alia, Anna Costa-Garrido, Rosa García-Sierra, Victor M. López-Lifante, Eduard Moreno-Gabriel, Marta Massanella, and et al. 2022. "Neurocognitive Profile of the Post-COVID Condition in Adults in Catalonia—A Mixed Method Prospective Cohort and Nested Case–Control Study: Study Protocol" Vaccines 10, no. 6: 849. https://doi.org/10.3390/vaccines10060849
APA StyleDacosta-Aguayo, R., Lamonja-Vicente, N., Chacón, C., Carrasco-Ribelles, L. A., Montero-Alia, P., Costa-Garrido, A., García-Sierra, R., López-Lifante, V. M., Moreno-Gabriel, E., Massanella, M., Puig, J., Muñoz-Moreno, J. A., Mateu, L., Prats, A., Rodríguez, C., Mataró, M., Prado, J. G., Martínez-Cáceres, E., Violán, C., & Torán-Monserrat, P. (2022). Neurocognitive Profile of the Post-COVID Condition in Adults in Catalonia—A Mixed Method Prospective Cohort and Nested Case–Control Study: Study Protocol. Vaccines, 10(6), 849. https://doi.org/10.3390/vaccines10060849








