Effect of COVID-19 Pandemic on Influenza Vaccination Intention: A Meta-Analysis and Systematic Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Inclusion and Exclusion Criteria
2.3. Study Selection and Data Extraction
2.4. Statistical Analysis and Quality Assessment
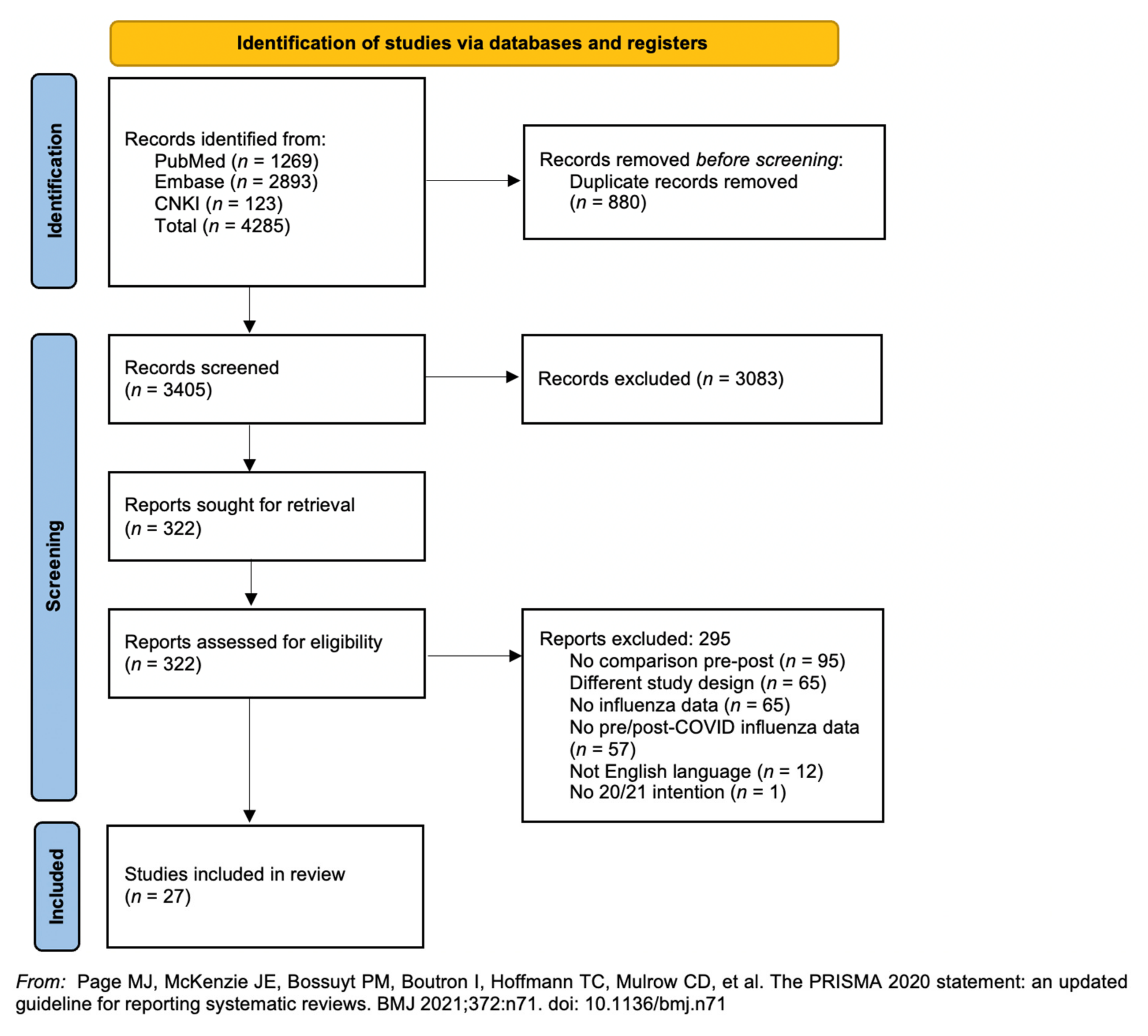
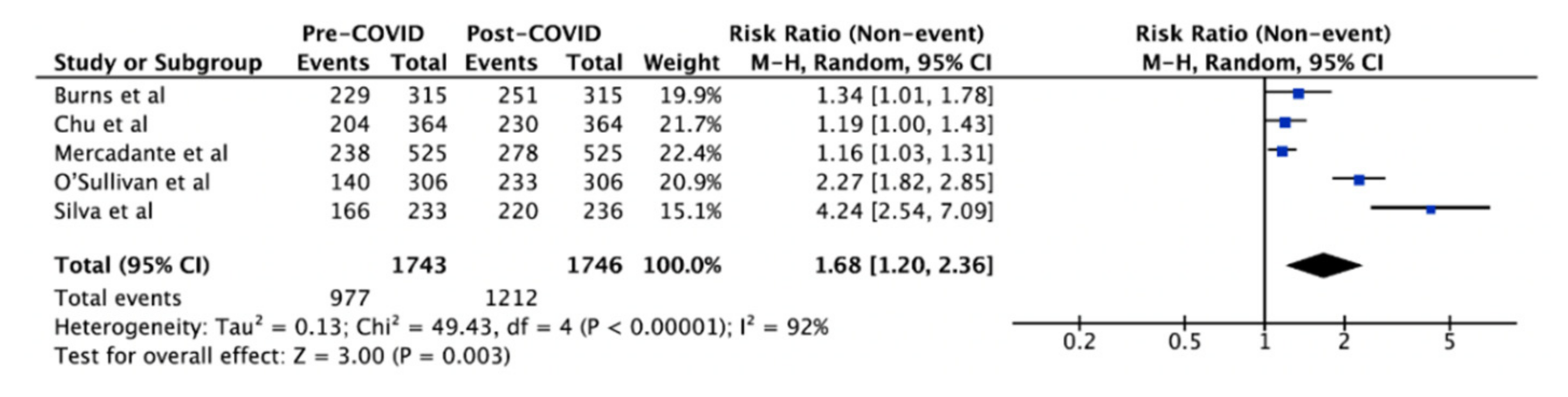
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Registration
References
- World Health Organization. Top Ten Causes of Death. Available online: https://www.who.int/news-room/fact-sheets/detail/the-top-10-causes-of-death (accessed on 7 August 2021).
- Paget, J.; Spreeuwenberg, P.; Charu, V.; Taylor, R.J.; Iuliano, A.D.; Bresee, J.; Simonsen, L.; Viboud, C. Global mortality associated with seasonal influenza epidemics: New burden estimates and predictors from the GLaMOR Project. J. Glob. Health 2019, 9, 020421. [Google Scholar] [CrossRef] [PubMed]
- Molinari, N.-A.M.; Ortega-Sanchez, I.R.; Messonnier, M.L.; Thompson, W.W.; Wortley, P.M.; Weintraub, E.; Bridges, C.B. The annual impact of seasonal influenza in the US: Measuring disease burden and costs. Vaccine 2007, 25, 5086–5096. [Google Scholar] [CrossRef] [PubMed]
- Chow, E.J.; Rolfes, M.A.; O’Halloran, A.; Anderson, E.J.; Bennett, N.M.; Billing, L.; Chai, S.; Dufort, E.; Herlihy, R.; Kim, S. Acute cardiovascular events associated with influenza in hospitalized adults: A cross-sectional study. Ann. Intern. Med. 2020, 173, 605–613. [Google Scholar] [CrossRef] [PubMed]
- Iuliano, A.D.; Roguski, K.M.; Chang, H.H.; Muscatello, D.J.; Palekar, R.; Tempia, S.; Cohen, C.; Gran, J.M.; Schanzer, D.; Cowling, B.J. Estimates of global seasonal influenza-associated respiratory mortality: A modelling study. Lancet 2018, 391, 1285–1300. [Google Scholar] [CrossRef]
- Lin, J.C.; Nichol, K.L. Excess mortality due to pneumonia or influenza during influenza seasons among persons with acquired immunodeficiency syndrome. Arch. Intern. Med. 2001, 161, 441–446. [Google Scholar] [CrossRef]
- Morgan, O.W.; Bramley, A.; Fowlkes, A.; Freedman, D.S.; Taylor, T.H.; Gargiullo, P.; Belay, B.; Jain, S.; Cox, C.; Kamimoto, L. Morbid obesity as a risk factor for hospitalization and death due to 2009 pandemic influenza A (H1N1) disease. PLoS ONE 2010, 5, e9694. [Google Scholar] [CrossRef]
- Siston, A.M.; Rasmussen, S.A.; Honein, M.A.; Fry, A.M.; Seib, K.; Callaghan, W.M.; Louie, J.; Doyle, T.J.; Crockett, M.; Lynfield, R. Pandemic 2009 influenza A (H1N1) virus illness among pregnant women in the United States. JAMA 2010, 303, 1517–1525. [Google Scholar] [CrossRef]
- Centers for Disease Control Prevention. Prevention and control of seasonal influenza with vaccines. Recommendations of the Advisory Committee on Immunization Practices—United States, 2013–2014. MMWR Recomm. Rep 2013, 62, 1–43. [Google Scholar]
- Nichol, K.; Margolis, K.; Wuorenma, J.; Von Sternberg, T. The efficacy and cost effectiveness of vaccination against influenza among elderly persons living in the community. N. Engl. J. Med. 1994, 331, 778–784. [Google Scholar] [CrossRef]
- Campbell, D.S.; Rumley, M.H. Cost-effectiveness of the influenza vaccine in a healthy, working-age population. J. Occup. Environ. Med. 1997, 39, 408–414. [Google Scholar] [CrossRef]
- Bridges, C.B.; Thompson, W.W.; Meltzer, M.I.; Reeve, G.R.; Talamonti, W.J.; Cox, N.J.; Lilac, H.A.; Hall, H.; Klimov, A.; Fukuda, K. Effectiveness and cost-benefit of influenza vaccination of healthy working adults: A randomized controlled trial. JAMA 2000, 284, 1655–1663. [Google Scholar] [CrossRef]
- Organization for Economic Co-Operation and Development (OECD). Influenza Vaccination Rates (Indicator). Available online: https://data.oecd.org/healthcare/influenza-vaccination-rates.htm (accessed on 19 July 2021).
- European Centre for Disease Prevention and Control (ECDC). Seasonal Influenza Vaccination and Antiviral Use in EU/EEA Member States. Available online: https://www.ecdc.europa.eu/sites/default/files/documents/seasonal-influenza-antiviral-use-2018.pdf (accessed on 19 July 2021).
- Endrich, M.M.; Blank, P.R.; Szucs, T.D. Influenza vaccination uptake and socioeconomic determinants in 11 European countries. Vaccine 2009, 27, 4018–4024. [Google Scholar] [CrossRef] [PubMed]
- Gualano, M.R.; Corradi, A.; Voglino, G.; Catozzi, D.; Olivero, E.; Corezzi, M.; Bert, F.; Siliquini, R. Healthcare Workers’(HCWs) attitudes towards mandatory influenza vaccination: A systematic review and meta-analysis. Vaccine 2021, 39, 901–914. [Google Scholar] [CrossRef] [PubMed]
- Yue, H.; Zhang, M.; Xing, L.; Wang, K.; Rao, X.; Liu, H.; Tian, J.; Zhou, P.; Deng, Y.; Shang, J. The epidemiology and clinical characteristics of co-infection of SARS-CoV-2 and influenza viruses in patients during COVID-19 outbreak. J. Med. Virol. 2020, 92, 2870–2873. [Google Scholar] [CrossRef] [PubMed]
- Amato, M.; Werba, J.P.; Frigerio, B.; Coggi, D.; Sansaro, D.; Ravani, A.; Ferrante, P.; Veglia, F.; Tremoli, E.; Baldassarre, D. Relationship between influenza vaccination coverage rate and COVID-19 outbreak: An Italian ecological study. Vaccines 2020, 8, 535. [Google Scholar] [CrossRef]
- World Health Organisation. Coronavirus Disease (COVID-19): Similarities and Differences with Influenza. Available online: https://www.who.int/news-room/q-a-detail/coronavirus-disease-covid-19-similarities-and-differences-with-influenza (accessed on 24 July 2021).
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Shallal, A.; Vahia, A.T.; Kenney, R.; Weinmann, A.J. 11. Missed Vaccine Opportunities During the COVID-19 Pandemic. Open Forum Infect. Dis. 2020, 7, S28–S29. [Google Scholar] [CrossRef]
- Zhou, Y.; Tang, J.; Zhang, J.; Wu, Q. Impact of the coronavirus disease 2019 epidemic and a free influenza vaccine strategy on the willingness of residents to receive influenza vaccines in Shanghai, China. Hum. Vaccines Immunother. 2021, 17, 2289–2292. [Google Scholar] [CrossRef]
- Perrone, P.M.; Biganzoli, G.; Lecce, M.; Campagnoli, E.M.; Castrofino, A.; Cinnirella, A.; Fornaro, F.; Gallana, C.; Grosso, F.M.; Maffeo, M. Influenza vaccination campaign during the covid-19 pandemic: The experience of a research and teaching hospital in Milan. Int. J. Environ. Res. Public Health 2021, 18, 5874. [Google Scholar] [CrossRef]
- Kang, M.; Clark, S.; Mendoza, S.; Arocha, D.; Cutrell, J.B.; Perl, T.M.; Trivedi, J. Influenza vaccination among healthcare personnel during the coronavirus disease 2019 (COVID-19) pandemic. Infect. Control Hosp. Epidemiol. 2021, 1–2, online ahead of print. [Google Scholar] [CrossRef]
- Fragoulis, G.E.; Grigoropoulos, I.; Mavrea, E.; Arida, A.; Bournia, V.-K.; Evangelatos, G.; Fragiadaki, K.; Karamanakos, A.; Kravvariti, E.; Panopoulos, S. Increased influenza vaccination rates in patients with autoimmune rheumatic diseases during the Covid-19 pandemic: A cross-sectional study. Rheumatol. Int. 2021, 41, 895–902. [Google Scholar] [CrossRef] [PubMed]
- Gerussi, V.; Peghin, M.; Palese, A.; Bressan, V.; Visintini, E.; Bontempo, G.; Graziano, E.; De Martino, M.; Isola, M.; Tascini, C. Vaccine hesitancy among Italian patients recovered from COVID-19 infection towards influenza and Sars-Cov-2 vaccination. Vaccines 2021, 9, 172. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Wong, E.L.Y.; Ho, K.F.; Cheung, A.W.L.; Chan, E.Y.Y.; Yeoh, E.K.; Wong, S.Y.S. Intention of nurses to accept coronavirus disease 2019 vaccination and change of intention to accept seasonal influenza vaccination during the coronavirus disease 2019 pandemic: A cross-sectional survey. Vaccine 2020, 38, 7049–7056. [Google Scholar] [CrossRef] [PubMed]
- Grochowska, M.; Ratajczak, A.; Zdunek, G.; Adamiec, A.; Waszkiewicz, P.; Feleszko, W. A Comparison of the Level of Acceptance and Hesitancy towards the Influenza Vaccine and the Forthcoming COVID-19 Vaccine in the Medical Community. Vaccines 2021, 9, 475. [Google Scholar] [CrossRef]
- La Vecchia, C.; Negri, E.; Alicandro, G.; Scarpino, V. Attitudes towards influenza vaccine and a potential COVID-19 vaccine in Italy and differences across occupational groups, September 2020. Med. Lav. 2020, 111, 445–448. [Google Scholar]
- Bonet-Esteve, A.; Muñoz-Miralles, R.; Gonzalez-Claramunt, C.; Rufas, A.M.; Cruz, X.P.; Vidal-Alaball, J. Influenza vaccination during the coronavirus pandemic: Intention to vaccinate among the at-risk population in the Central Catalonia Health Region (VAGCOVID). BMC Fam. Pract. 2021, 22, 84. [Google Scholar] [CrossRef]
- Cheung, M.W.L.; Ho, R.C.; Lim, Y.; Mak, A. Conducting a meta-analysis: Basics and good practices. Int. J. Rheum. Dis. 2012, 15, 129–135. [Google Scholar] [CrossRef]
- Higgins, J.P.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef]
- Wells, G.A.; Shea, B.; O’Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses. Available online: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed on 15 September 2021).
- AlHajri, B.; Alenezi, D.; Alfouzan, H.; Altamimi, S.; Alzalzalah, S.; Almansouri, W.; Alqudeimat, Y.; Almokhaizeem, Z.; Ziyab, A.H. Willingness of parents to vaccinate their children against influenza and the novel coronavirus disease-2019. J. Pediatr. 2021, 231, 298–299. [Google Scholar] [CrossRef]
- Bachtiger, P.; Adamson, A.; Chow, J.-J.; Sisodia, R.; Quint, J.K.; Peters, N.S. The impact of the COVID-19 pandemic on the uptake of influenza vaccine: UK-wide observational study. JMIR Public Health Surveill. 2021, 7, e26734. [Google Scholar] [CrossRef]
- Burns, V.M.; Castillo, F.M.; Coldren, C.R.L.; Prosser, T.; Howell, L.P.R.L.; Kabbur, M.B. Perceptions of seasonal influenza vaccine among US Army civilians and dependents in the Kaiserslautern Military Community: A mixed-methods survey. Mil. Med. 2021, 187, e394–e403. [Google Scholar] [CrossRef]
- Chu, A.; Gupta, V.; Unni, E.J. Utilizing the Theory of Planned Behavior to determine the intentions to receive the influenza vaccine during COVID-19: A cross-sectional survey of US adults. Prev. Med. Rep. 2021, 23, 101417. [Google Scholar] [CrossRef] [PubMed]
- Cuschieri, S.; Grech, V. A comparative assessment of attitudes and hesitancy for influenza vis-à-vis COVID-19 vaccination among healthcare students and professionals in Malta. J. Public Health 2021, 1–8, online ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Domnich, A.; Cambiaggi, M.; Vasco, A.; Maraniello, L.; Ansaldi, F.; Baldo, V.; Bonanni, P.; Calabrò, G.E.; Costantino, C.; de Waure, C. Attitudes and beliefs on influenza vaccination during the COVID-19 pandemic: Results from a representative Italian survey. Vaccines 2020, 8, 711. [Google Scholar] [CrossRef] [PubMed]
- Gagneux-Brunon, A.; Detoc, M.; Bruel, S.; Tardy, B.; Rozaire, O.; Frappe, P.; Botelho-Nevers, E. Intention to get vaccinations against COVID-19 in French healthcare workers during the first pandemic wave: A cross-sectional survey. J. Hosp. Infect. 2021, 108, 168–173. [Google Scholar] [CrossRef] [PubMed]
- Gatwood, J.; McKnight, M.; Fiscus, M.; Hohmeier, K.C.; Chisholm-Burns, M. Factors influencing likelihood of COVID-19 vaccination: A survey of Tennessee adults. Am. J. Health Syst. Pharm. 2021, 78, 879–889. [Google Scholar] [CrossRef]
- Goldman, R.D.; McGregor, S.; Marneni, S.R.; Katsuta, T.; Griffiths, M.A.; Hall, J.E.; Seiler, M.; Klein, E.J.; Cotanda, C.P.; Gelernter, R. Willingness to vaccinate children against influenza after the Coronavirus disease 2019 pandemic. J. Pediatr. 2021, 228, 87–93.e82. [Google Scholar] [CrossRef]
- Gönüllü, E.; Soysal, A.; Atıcı, S.; Engin, M.; Yeşilbaş, O.; Kasap, T.; Fedakar, A.; Bilgiç, E.; Tavil, E.B.; Tutak, E. Pediatricians’ COVID-19 experiences and views on the willingness to receive COVID-19 vaccines: A cross-sectional survey in Turkey. Hum. Vaccines Immunother. 2021, 17, 2389–2396. [Google Scholar] [CrossRef]
- Hou, Z.; Song, S.; Du, F.; Shi, L.; Zhang, D.; Lin, L.; Yu, H. The Influence of the COVID-19 Epidemic on Prevention and Vaccination Behaviors Among Chinese Children and Adolescents: Cross-sectional Online Survey Study. JMIR Public Health Surveill. 2021, 7, e26372. [Google Scholar] [CrossRef]
- Jiang, M.; Feng, L.; Wang, W.; Gong, Y.; Ming, W.-K.; Hayat, K.; Li, P.; Gillani, A.H.; Yao, X.; Fang, Y. Knowledge, attitudes, and practices towards influenza among Chinese adults during the epidemic of COVID-19: A cross-sectional online survey. Hum. Vaccines Immunother. 2021, 17, 1412–1419. [Google Scholar] [CrossRef]
- Maltezou, H.C.; Pavli, A.; Dedoukou, X.; Georgakopoulou, T.; Raftopoulos, V.; Drositis, I.; Bolikas, E.; Ledda, C.; Adamis, G.; Spyrou, A. Determinants of intention to get vaccinated against COVID-19 among healthcare personnel in hospitals in Greece. Infect. Dis. Health 2021, 26, 189–197. [Google Scholar] [CrossRef]
- Mercadante, A.R.; Law, A.V. Will they, or Won’t they? Examining patients’ vaccine intention for flu and COVID-19 using the Health Belief Model. Res. Soc. Adm. Pharm. 2020, 17, 1596–1605. [Google Scholar] [CrossRef] [PubMed]
- O’Sullivan, N.; O’Sullivan, G.; Van Harten, M. The Impact of the COVID-19 Pandemic on the Uptake of the Seasonal Influenza Vaccine. Ir. Med. J. 2021, 114, 269. [Google Scholar]
- Raftopoulos, V.; Iordanou, S.; Katsapi, A.; Dedoukou, X.; Maltezou, H.C. A comparative online survey on the intention to get COVID-19 vaccine between Greek and Cypriot healthcare personnel: Is the country a predictor? Hum. Vaccines Immunother. 2021, 17, 2397–2404. [Google Scholar] [CrossRef] [PubMed]
- Silva, J.; Bratberg, J.; Lemay, V. COVID-19 and influenza vaccine hesitancy among college students. J. Am. Pharm. Assoc. 2021, 61, 709–714.e1. [Google Scholar] [CrossRef] [PubMed]
- Sturm, L.; Kasting, M.L.; Head, K.J.; Hartsock, J.A.; Zimet, G.D. Influenza vaccination in the time of COVID-19: A national US survey of adults. Vaccine 2021, 39, 1921–1928. [Google Scholar] [CrossRef]
- Di Gennaro, F.; Murri, R.; Segala, F.V.; Cerruti, L.; Abdulle, A.; Saracino, A.; Bavaro, D.F.; Fantoni, M. Attitudes towards anti-sars-cov2 vaccination among healthcare workers: Results from a national survey in Italy. Viruses 2021, 13, 371. [Google Scholar] [CrossRef]
- Di Giuseppe, G.; Pelullo, C.P.; Paolantonio, A.; Della Polla, G.; Pavia, M. Healthcare workers’ willingness to receive influenza vaccination in the context of the covid-19 pandemic: A survey in southern Italy. Vaccines 2021, 9, 766. [Google Scholar] [CrossRef]
- Kopsidas, I.; Chorianopoulou, E.; Kourkouni, E.; Triantafyllou, C.; Molocha, N.M.; Koniordou, M.; Maistreli, S.; Tsopela, C.G.; Maroudi-Manta, S.; Filippou, D.; et al. Covid-19 pandemic impact on seasonal flu vaccination: A cross-sectional study. Pneumon 2021, 34, 1–9. [Google Scholar] [CrossRef]
- Kosaka, M.; Kotera, Y.; Tsuda, K.; Takahashi, K.; Hamaki, T.; Kusumi, E.; Kami, M.; Tanimoto, T. Influenza vaccination uptake and attitudes among adult cancer patients in Japan: A web-based questionnaire survey before the 2020/2021 season. Hum. Vaccines Immunother. 2021, 17, 5509–5513. [Google Scholar] [CrossRef]
- Alosaimi, B.; Naeem, A.; Hamed, M.E.; Alkadi, H.S.; Alanazi, T.; Al Rehily, S.S.; Almutairi, A.Z.; Zafar, A. Influenza co-infection associated with severity and mortality in COVID-19 patients. Virol. J. 2021, 18, 127. [Google Scholar] [CrossRef] [PubMed]
- Bish, A.; Yardley, L.; Nicoll, A.; Michie, S. Factors associated with uptake of vaccination against pandemic influenza: A systematic review. Vaccine 2011, 29, 6472–6484. [Google Scholar] [CrossRef] [PubMed]
- Poland, G.A.; Jacobson, R.M. The age-old struggle against the antivaccinationists. N. Engl. J. Med. 2011, 364, 97–99. [Google Scholar] [CrossRef] [PubMed]
- MacDonald, N.E. Vaccine hesitancy: Definition, scope and determinants. Vaccine 2015, 33, 4161–4164. [Google Scholar] [CrossRef] [PubMed]
- Brewer, N.T.; Chapman, G.B.; Gibbons, F.X.; Gerrard, M.; McCaul, K.D.; Weinstein, N.D. Meta-analysis of the relationship between risk perception and health behavior: The example of vaccination. Health Psychol. 2007, 26, 136–145. [Google Scholar] [CrossRef]
- Soo, R.J.J.; Chiew, C.J.; Ma, S.; Pung, R.; Lee, V. Decreased influenza incidence under COVID-19 control measures, Singapore. Emerg. Infect. Dis. 2020, 26, 1933. [Google Scholar] [CrossRef]
- Cowling, B.J.; Ali, S.T.; Ng, T.W.; Tsang, T.K.; Li, J.C.; Fong, M.W.; Liao, Q.; Kwan, M.Y.; Lee, S.L.; Chiu, S.S. Impact assessment of non-pharmaceutical interventions against coronavirus disease 2019 and influenza in Hong Kong: An observational study. Lancet Public Health 2020, 5, e279–e288. [Google Scholar] [CrossRef]
- Sakamoto, H.; Ishikane, M.; Ueda, P. Seasonal influenza activity during the SARS-CoV-2 outbreak in Japan. JAMA 2020, 323, 1969–1971. [Google Scholar] [CrossRef]
- Groves, H.E.; Piché-Renaud, P.P.; Peci, A.; Farrar, D.S.; Buckrell, S.; Bancej, C.; Sevenhuysen, C.; Campigotto, A.; Gubbay, J.B.; Morris, S.K. The impact of the COVID-19 pandemic on influenza, respiratory syncytial virus, and other seasonal respiratory virus circulation in Canada: A population-based study. Lancet Reg. Health Am. 2021, 1, 100015. [Google Scholar] [CrossRef]
- Lee, K.; Jalal, H.; Raviotta, J.M.; Krauland, M.G.; Zimmerman, R.; Burke, D.S.; Roberts, M.S. Predicting the impact of low influenza activity in 2020 on population immunity and future influenza season in the United States. MedRxiv 2021. [Google Scholar] [CrossRef]
- Puri, N.; Coomes, E.A.; Haghbayan, H.; Gunaratne, K. Social media and vaccine hesitancy: New updates for the era of COVID-19 and globalized infectious diseases. Hum. Vaccines Immunother. 2020, 16, 2586–2593. [Google Scholar] [CrossRef] [PubMed]
- Seeman, N.; Rizo, C. Assessing and responding in real time to online anti-vaccine sentiment during a flu pandemic. Healthc. Q. 2010, 13, 8–15. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kata, A. A postmodern Pandora’s box: Anti-vaccination misinformation on the Internet. Vaccine 2010, 28, 1709–1716. [Google Scholar] [CrossRef] [PubMed]
- Karafillakis, E.; Dinca, I.; Apfel, F.; Cecconi, S.; Wűrz, A.; Takacs, J.; Suk, J.; Celentano, L.P.; Kramarz, P.; Larson, H.J. Vaccine hesitancy among healthcare workers in Europe: A qualitative study. Vaccine 2016, 34, 5013–5020. [Google Scholar] [CrossRef] [PubMed]
- Sturm, L.A.; Mays, R.M.; Zimet, G.D. Parental beliefs and decision making about child and adolescent immunization: From polio to sexually transmitted infections. J. Dev. Behav. Pediatr. 2005, 26, 441–452. [Google Scholar] [CrossRef]
- Lo, N.C.; Hotez, P.J. Public health and economic consequences of vaccine hesitancy for measles in the United States. JAMA Pediatr. 2017, 171, 887–892. [Google Scholar] [CrossRef]
- Lehmann, B.A.; Ruiter, R.A.; Chapman, G.; Kok, G. The intention to get vaccinated against influenza and actual vaccination uptake of Dutch healthcare personnel. Vaccine 2014, 32, 6986–6991. [Google Scholar] [CrossRef]
- Van den Dool, C.; Van Strien, A.; Looijmans-Van Den Akker, I.; Bonten, M.; Sanders, E.; Hak, E. Attitude of Dutch hospital personnel towards influenza vaccination. Vaccine 2008, 26, 1297–1302. [Google Scholar] [CrossRef]
- Jiménez-Corona, M.; Ponce-de-León-Rosales, S. Influenza vaccine and healthcare workers. Arch. Med. Res. 2011, 42, 652–657. [Google Scholar]
- Fall, E.; Izaute, M.; Chakroun-Baggioni, N. How can the health belief model and self-determination theory predict both influenza vaccination and vaccination intention? A longitudinal study among university students. Psychol. Health 2018, 33, 746–764. [Google Scholar] [CrossRef]
- Gollwitzer, P.M.; Sheeran, P. Implementation intentions and goal achievement: A meta-analysis of effects and processes. Adv. Exp. Soc. Psychol. 2006, 38, 69–119. [Google Scholar]
- Sheeran, P.; Stroebe, W.; Hewstone, M. Intention–behavior relations: A conceptual and empirical review. Eur. Rev. Soc. Psychol. 2002, 12, 1–36. [Google Scholar] [CrossRef]
- Sniehotta, F.F.; Scholz, U.; Schwarzer, R. Bridging the intention–behaviour gap: Planning, self-efficacy, and action control in the adoption and maintenance of physical exercise. Psychol. Health 2005, 20, 143–160. [Google Scholar] [CrossRef]
- Ernsting, A.; Gellert, P.; Schneider, M.; Lippke, S. A mediator model to predict workplace influenza vaccination behaviour—An application of the health action process approach. Psychol. Health 2013, 28, 579–592. [Google Scholar] [CrossRef] [PubMed]
- Roman, P.C.; Kirtland, K.; Zell, E.R.; Jones-Jack, N.; Shaw, L.; Shrader, L.; Sprague, C.; Schultz, J.; Le, Q.; Nalla, A. Influenza Vaccinations During the COVID-19 Pandemic—11 US Jurisdictions, September–December 2020. Morb. Mortal. Wkly. Rep. 2021, 70, 1575. [Google Scholar] [CrossRef]
| Authors | Country | Study Population | Outcome Studied in 2020–2021 Season | Age (Years) * | Female | Healthcare Worker | 2019/2020 Influenza Vaccine Uptake | 2020/2021 Influenza Vaccine Intention and/or Uptake |
|---|---|---|---|---|---|---|---|---|
| Domnich et al. (2020) [39] | Italy | n = 2543 Italian adults | Intention | 46.7 ± 15.5 | 45.5% | - | 27.4% | 44.0% |
| Jiang et al. (2020) [45] | China | n = 4822 Chinese adults | Intention | 18–40: 66.1% 41–60: 30.2% >60: 3.7% | 61.2% | 38.0% | 15.9% | 62.5% |
| La Vecchia et al. (2020) [29] | Italy | n = 1055 Italian population | Intention | 15–34: 23.8% 35–54: 34.1% ≥55: 42.1% | 51.8% | - | 24.4% | 40.8% |
| Wang et al. (2020) [27] | Hong Kong, China | n = 806 Association of Hong Kong Nursing Staff | Intention | 18–29: 21.6% 30–39: 31.1% 40–49: 27.1% ≥50: 20.2% | 87.5% | 100.0% | 47.5% | 44.7% |
| AlHajri et al. (2020) [34] | Kuwait | n = 1038 Kuwaiti parents and their children | Intention | <18: 100% | - | - | 17.6% | 32.9% |
| Bachtiger et al. (2020) [35] | United Kingdom | n = 5664 Registrants of the Care Information Exchange (CIE) of Imperial College Healthcare NHS Foundation Trust | Intention | - | 50.0% | 14.3% | 71.3% | 82.1% |
| Gagneux-Brunon et al. (2020) [40] | France | n = 2047 Healthcare workers | Intention | <30: 22.7% 30–49: 47.3% 50–64: 26.8% ≥65: 3.1% | 74% | 100.0% | 57.3% | 54.5% |
| Gatwood et al. (2020) [41] | United States | n = 1000 Tennessee adults | Intention | 18–24: 17.0% 25–34: 21.7% 35–44: 24.8% 45–54: 18.1% 55–64: 18.4% | 52.8% | - | 36.4% | 49.3% |
| Gerussi et al. (2021) [26] | Italy | n = 599 Italian patients recovered from COVID-19 | Intention | 53 ± 15.8 | 53.40% | 22.2% | 26.2% | 45.4% |
| Goldman et al. (2021) [42] | US, Canada, Israel, Japan, Spain, and Switzerland | n = 2422 Parents and caregivers at paediatric emergency departments | Intention | 8.6 ± 4.6 | 48.1% | - | 39.0% | 54.3% |
| Raftopoulos et al. (2021) [49] | Greece and Cypriot | n = 2238 Healthcare workers | Intention | Greece 40.6 ± 9.6 Rep Cyprus 35.5 ± 8.8 | - | 94.0% | 42.2% | 52.7% |
| Sturm et al. (2021) [51] | United States | n = 3502 Dynata database comprising North American survey respondents | Intention | Non-vaccinators 42.9 ± 15.2 Vaccinators 48.2 ± 17.9 | 51.9% | - | 53.0% | 60.6% |
| Bonet-Esteve et al. (2021) [30] | Spain | n = 434 Individuals registered at the Primary Care Teams of the Catalan Institute of Health of Central Catalonia | Intention | <60: 35.0% 60–70: 23.0% >70: 41.9% | 59.4% | - | 35.7% | 43.3% |
| Cuschieri et al. (2021) [38] | Malta | n = 1802 Healthcare workers | Intention | 18–24: 33.7% 25–34: 25.0% 35–44: 14.7% 45–54: 14.0% 55–64: 11.2% ≥65: 1.5% | 65.2% | 100.0% | 48.1% | 68.9% |
| Gönüllü et al. (2021) [43] | Turkey | n = 506 Turkish Paediatric Atelier | Intention | 41 ± 8 26–35: 33.0% 36–44: 33.0% 45–60: 30.0% >60: 4.0% | 58.0% | 100.0% | 39.1% | 70.0% |
| Grochowska et al. (2021) [28] | Poland | n = 419 Doctors, nurses, physiotherapists, dieticians, medical students | Intention | 19–25: 60.4% 26–30: 22.9% 31–40: 8.1% 41–50: 4.8% >50: 3.8% | 79.0% | 100.0% | 32.9% | 61.6% |
| Hou et al. (2021) [44] | China | n = 1655 Parents of children 3–17 years | Intention | 3–5: 19.4% 6–9: 26.1% 10–14: 21.7% 15–17: 32.8% | 49.9% | - | 54.7% | 80.9% |
| Maltezou et al. (2021) [46] | Greece | n = 1591 Healthcare workers | Intention | ≤30: 17.7% 31–40: 22.8% 41–50: 28.3% >50: 31.2% | 65.0% | 82.7% | 54% | 65% |
| Di Gennaro et al. (2021) [52] | Italy | n = 1723 Healthcare workers | Intention | 35.5 ± 11.8 | 53.0% | 100.0% | 47.0% | 89.1% |
| Di Giuseppe et al. (2021) [53] | Italy | n = 490 Healthcare workers | Intention | 50.7 ± 10.5 | 54.5% | 100.0% | 40.8% | 68.0% |
| Kopsidas et al. (2021) [54] | Greece | n = 1004 Greek adult population | Intention | 41.7 ± 17.7 | 50.2% | - | 33.0% | 52.8% |
| Kosaka et al. (2021) [55] | Japan | n = 163 Cancer patients | Intention | 55.0 ± 12.4 | 60.1% | - | 61.9% | 72.4% |
| Chu et al. (2021) [37] | United States | n = 364 US adults above 18 years | Uptake and intention | 18–29: 26.3% 30–44: 29.9% 45–60: 29.4% >60: 14.4% | 59.1% | - | 56.0% | 63.2% |
| O’Sullivan et al. (2021) [48] | Ireland | n = 307 Patients at GP practice during the 2020 “flu season” | Uptake and intention | 2–12: 13.0% 13–18: 3.9% 19–30: 13.0% 31–50: 30.3% 51–70: 29.3% >70: 9.8% | 57.7% | - | 45.6% | 76.1% |
| Burns et al. (2020) [36] | United States | n = 315 Enrolled non-active duty patients at Landstuhl Regional Medical Center (LRMC) | Uptake and intention | 21–59: 65.1% ≥60: 34.9% | 42.9% | 14.0% | 72.7% | 79.7% |
| Mercadante et al. (2020) [47] | United States | n = 525 United States adults | Uptake and intention | 18–29: 21.0% 30–49: 32.8% 50–69: 32.0% ≥70: 14.1% | 49.0% | - | 45.3% | 53.0% |
| Silva et al. (2021) [50] | United States | n = 237 US students | Uptake and intention | 18–19: 43.0% 20–29: 54.0% 30–39: 2.0% | 65.0% | 17.0% | 70.0% | 93.2% |
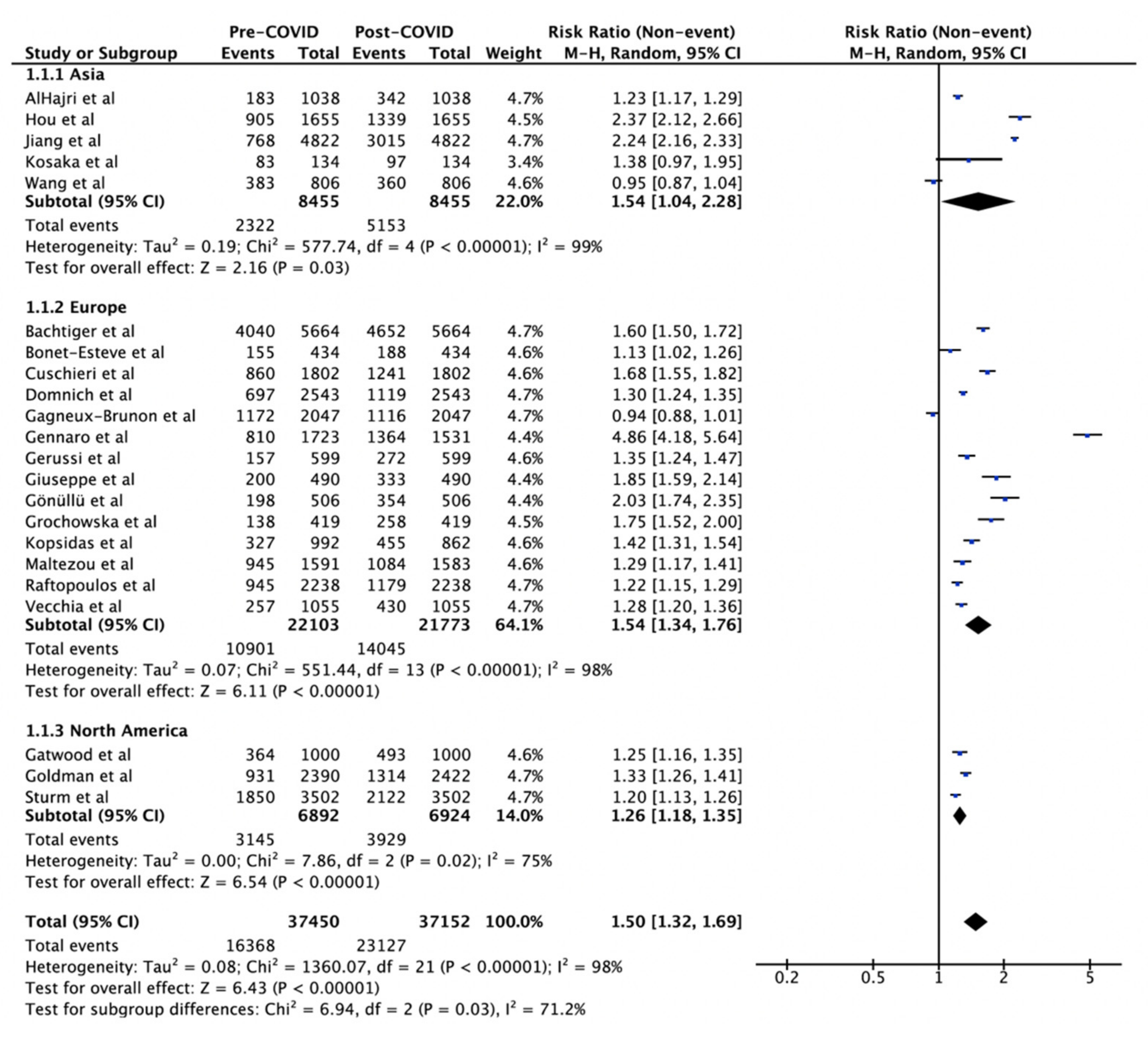
| Comparison | Number of Studies | Sample Size | Risk Ratio (95% CI) | I2 | p-Value |
|---|---|---|---|---|---|
| Region | 98% | 0.03 * | |||
| Asia | 5 | 8455 | 1.54 (1.04–2.28) | 99% | 0.03 |
| Europe | 14 | 22,103/21,773 | 1.54 (1.34–1.76) | 98% | <0.001 |
| North America | 3 | 6892/6924 | 1.26 (1.18–1.35) | 75% | <0.001 |
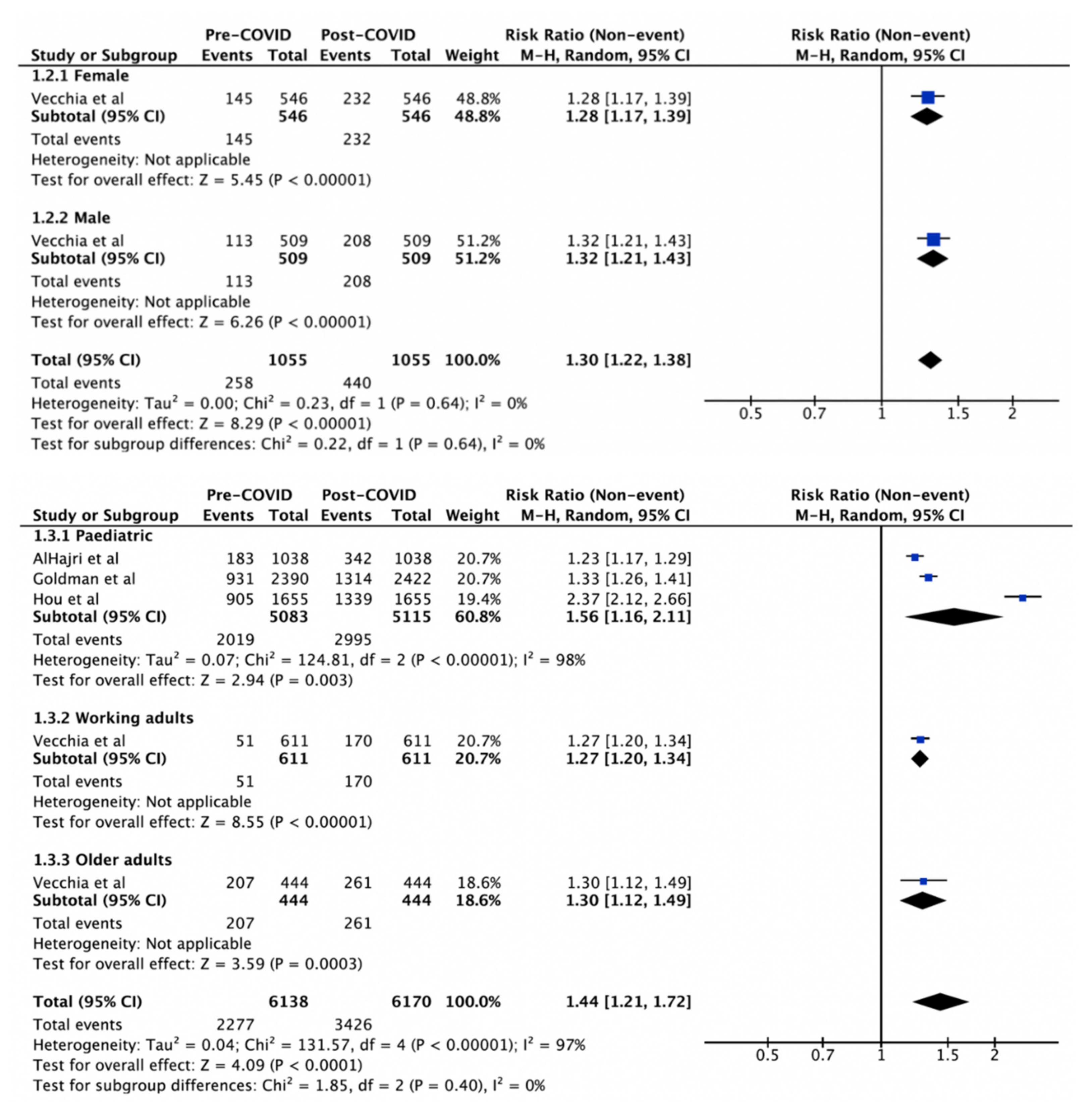
| Gender | 0% | 0.64 * | |||
| Female | 1 | 546 | 1.28 (1.17–1.39) | - | <0.001 |
| Male | 1 | 509 | 1.32 (1.21–1.43) | - | <0.001 |
| Age | 97% | 0.40 * | |||
| Paediatric | 3 | 5083/5115 | 1.56 (1.16–2.11) | 98% | 0.003 |
| Working Adults | 1 | 611 | 1.27 (1.20–1.34) | - | <0.001 |
| Older Adults | 1 | 444 | 1.30 (1.12–1.49) | - | <0.001 |
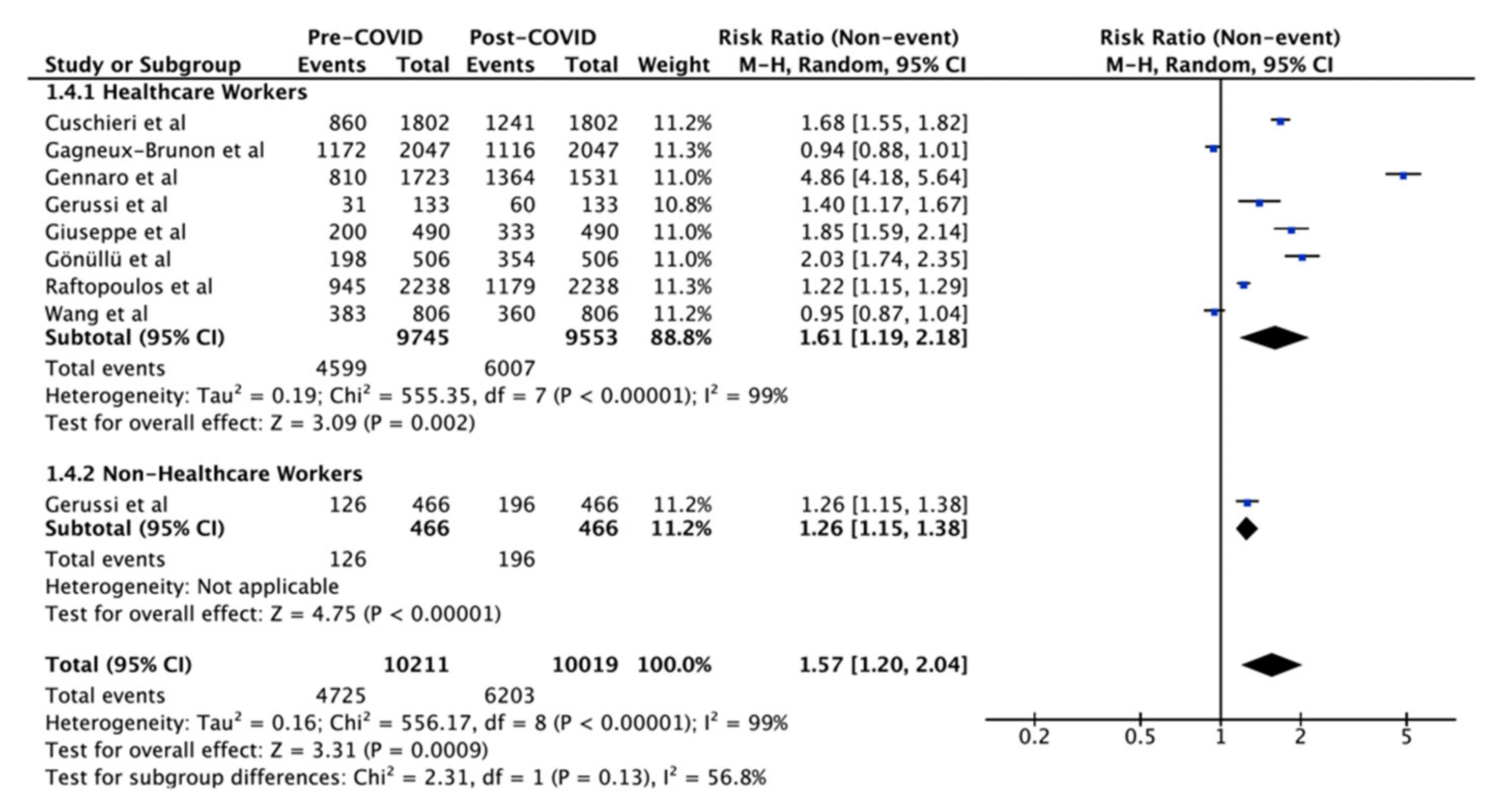
| Occupation | 99% | 0.13 * | |||
| HCW | 8 | 9745/9553 | 1.61 (1.19–2.18) | 99% | 0.002 |
| Non-HCW | 1 | 466 | 1.26 (1.15–1.38) | - | <0.001 |
| Overall | 22 | 37,450/37,152 | 1.50 (1.32–1.69) | 98% | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kong, G.; Lim, N.-A.; Chin, Y.H.; Ng, Y.P.M.; Amin, Z. Effect of COVID-19 Pandemic on Influenza Vaccination Intention: A Meta-Analysis and Systematic Review. Vaccines 2022, 10, 606. https://doi.org/10.3390/vaccines10040606
Kong G, Lim N-A, Chin YH, Ng YPM, Amin Z. Effect of COVID-19 Pandemic on Influenza Vaccination Intention: A Meta-Analysis and Systematic Review. Vaccines. 2022; 10(4):606. https://doi.org/10.3390/vaccines10040606
Chicago/Turabian StyleKong, Gwyneth, Nicole-Ann Lim, Yip Han Chin, Yvonne Peng Mei Ng, and Zubair Amin. 2022. "Effect of COVID-19 Pandemic on Influenza Vaccination Intention: A Meta-Analysis and Systematic Review" Vaccines 10, no. 4: 606. https://doi.org/10.3390/vaccines10040606
APA StyleKong, G., Lim, N.-A., Chin, Y. H., Ng, Y. P. M., & Amin, Z. (2022). Effect of COVID-19 Pandemic on Influenza Vaccination Intention: A Meta-Analysis and Systematic Review. Vaccines, 10(4), 606. https://doi.org/10.3390/vaccines10040606






