The Fluctuation Trend of Serum Anti-SARS-CoV-2 IgM/IgG Antibodies Seroprevalence in the Non-COVID-19 Infected Population and Correlation with Peripheral Blood Leukocyte Parameters in Beijing, China, 2021: A Real-World Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Sample Collection and Processing
2.2.1. Nasal or Mouth Throat Swab Specimen Collection
2.2.2. The Peripheral Blood Sample Collection
2.3. Analytical Procedures and Instruments
2.3.1. SARS-CoV-2 RNA Determination
2.3.2. The Semi-Quantitative Analysis of Serum SARS-CoV-2 IgM/IgG Antibodies
2.3.3. The Peripheral Blood Leukocyte Analysis
2.4. Statistical Analysis
3. Results
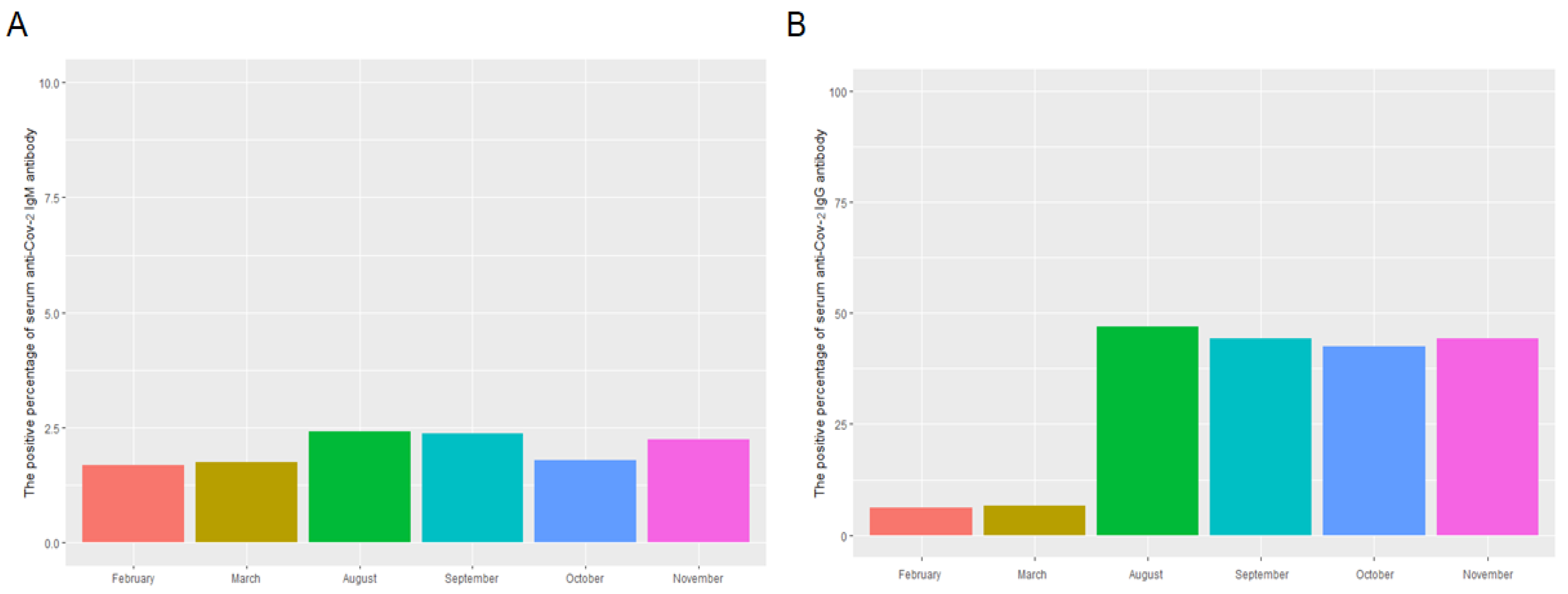
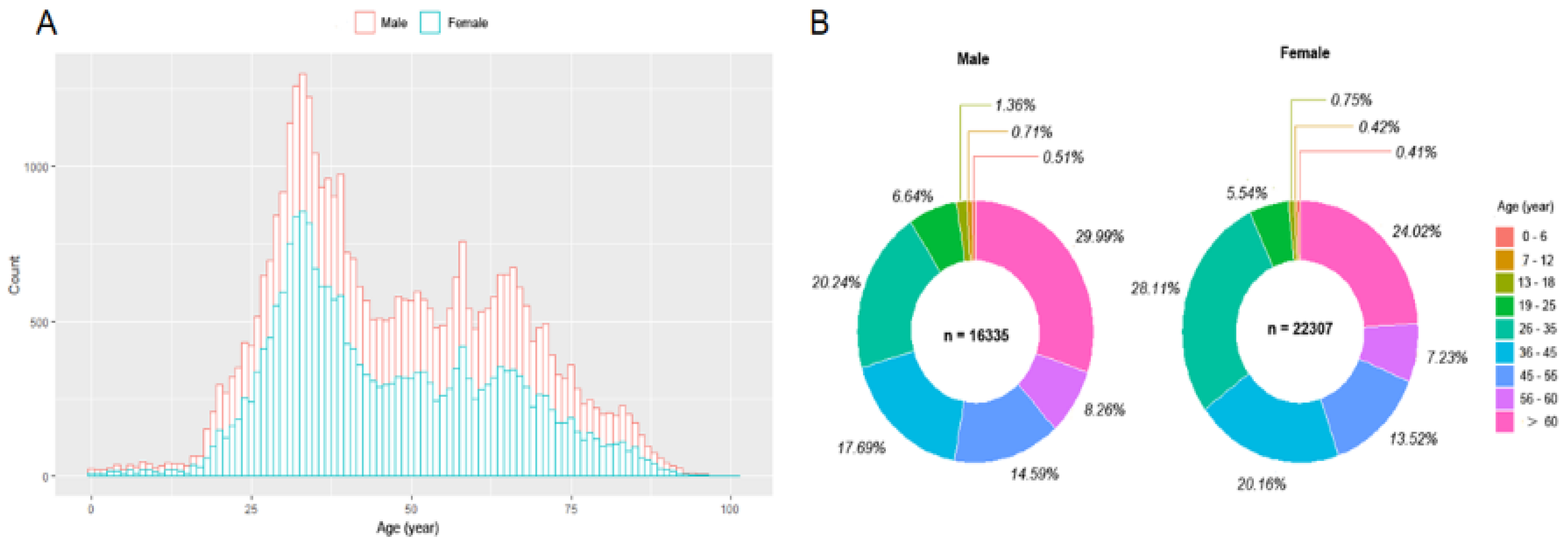
3.1. The Distribution, Levels, and Monthly Positive Percentage of Serum Anti-SARS-CoV-2 IgM/IgG and Peripheral Blood Leukocyte Analysis in the Study Population
3.2. The Levels of Serum SARS-CoV-2 IgM and IgG in the Different Age Group
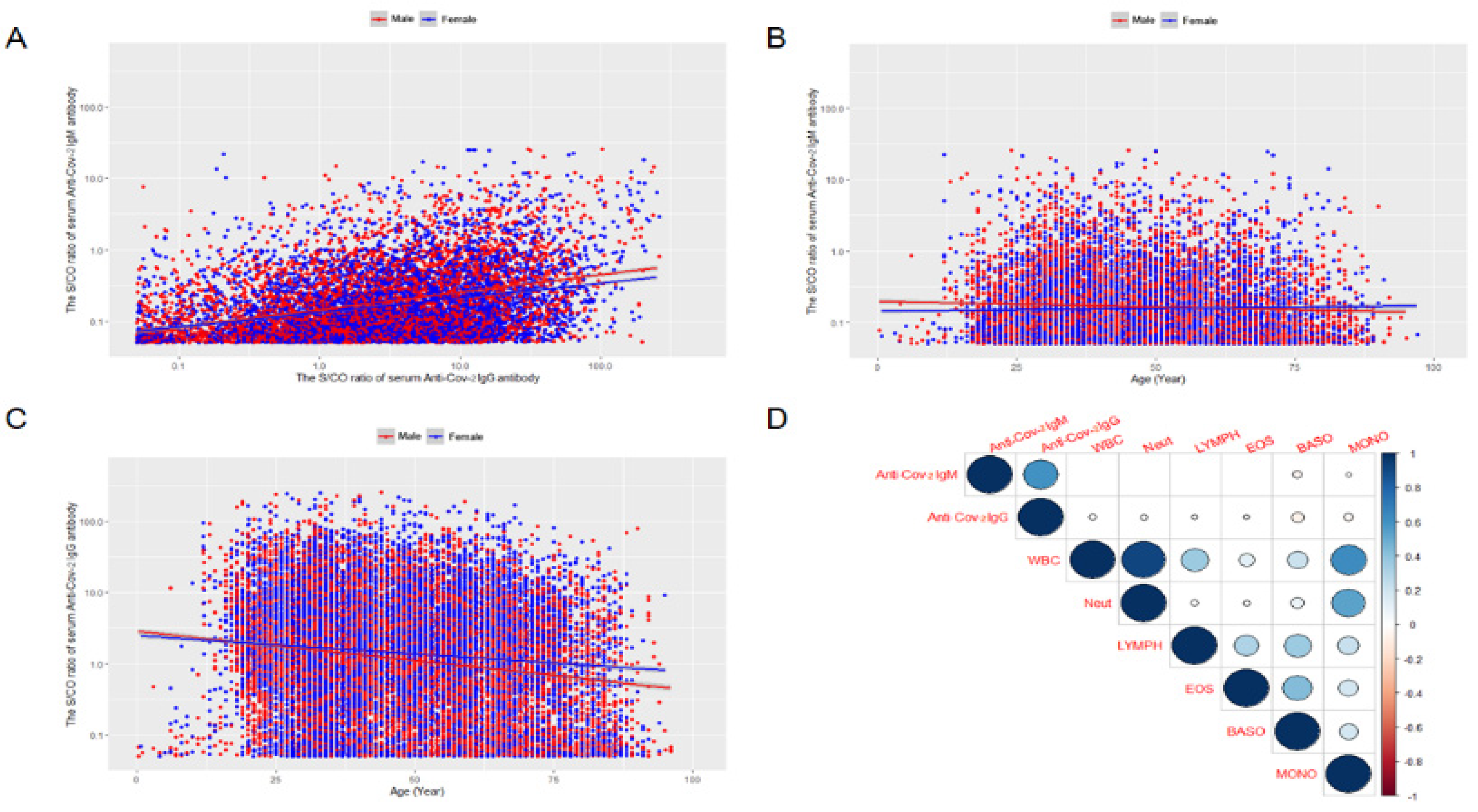
3.3. The Correlation between the Serum Anti-SARS-CoV-2 IgM and IgG Antibodies
3.4. The Relationship between the Serum Anti-SARS-CoV-2 IgM/IgG Antibodies Levels and Age
3.5. The Relationship Serum Anti-SARS-CoV-2 IgM/IgG Antibodies Levels and Peripheral Blood Neutrophils or Lymphocytes by Generalized Linear Mixed Model (GLMMs) Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Harvey, R.A.; Rassen, J.A.; Kabelac, C.A.; Turenne, W.; Leonard, S.; Klesh, R.; Meyer, W.A.; Kaufman, H.W.; Anderson, S.; Cohen, V.I.; et al. Real-World Data Suggest Antibody Positivity to SARS-CoV-2 Is Associated with a Decreased Risk of Future Infection. medRxiv 2020. [Google Scholar] [CrossRef]
- Zheng, W.; Yan, X.; Zhao, Z.; Yang, J.; Yu, H. COVID-19 vaccination program in the mainland of China: A subnational descriptive analysis on target population size and current progress. Infect. Dis. Poverty 2021, 10, 1–10. [Google Scholar] [CrossRef]
- Plebani, M.; Cosma, C.; Padoan, A. SARS-CoV-2 Antibody Assay after Vaccination: One Size Does Not Fit All. Clin. Chem. Lab. Med. 2021, 59, e380–e381. [Google Scholar] [CrossRef] [PubMed]
- He, Z.; Ren, L.; Yang, J.; Guo, L.; Feng, L.; Ma, C.; Wang, X.; Leng, Z.; Tong, X.; Zhou, W.; et al. Seroprevalence and Humoral Immune Durability of Anti-SARS-CoV-2 Antibodies in Wuhan, China: A Longitudinal, Population-Level, Cross-Sectional Study. Lancet 2021, 397, 1075–1084. [Google Scholar] [CrossRef]
- Yang, M.; Cao, S.; Liu, Y.; Zhang, Z.; Zheng, R.; Li, Y.; Zhou, J.; Zong, C.; Cao, D.; Qin, X. Performance Verification of Five Commercial Rt-Qpcr Diagnostic Kits for SARS-CoV-2. Clin. Chim. Acta 2022, 525, 46–53. [Google Scholar] [CrossRef] [PubMed]
- Xu, Q.Y.; Xue, J.H.; Xiao, Y.; Jia, Z.J.; Wu, M.J.; Liu, Y.Y.; Li, W.L.; Liang, X.M.; Yang, T.C. Response and Duration of Serum Anti-SARS-CoV-2 Antibodies after Inactivated Vaccination within 160 Days. Front. Immunol. 2021, 12, 786554. [Google Scholar] [CrossRef] [PubMed]
- Strugnell, R.; Wang, N. Sustained Neutralising Antibodies in the Wuhan Population Suggest Durable Protection against SARS-CoV-2. Lancet 2021, 397, 1037–1039. [Google Scholar] [CrossRef]
- Ferrari, D.; Mangia, A.; Spanò, M.S.; Zaffarano, L.; Viganò, M.; Di Resta, C.; Locatelli, M.; Ciceri, F.; De Vecchi, E. Quantitative serological evaluation as a valuable tool in the COVID-19 vaccination campaign. Clin. Chem. Lab. Med. 2021, 59, 2019–2026. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Lian, R.; Zhang, G.; Hou, B.; Wang, C.; Dong, J.; Yang, L.; Wang, J.; Dai, S.; Chen, L.; et al. Changes in serum virus-specific IgM/IgG antibody in asymptomatic and discharged patients with reoccurring positive COVID-19 nucleic acid test (RPNAT). Ann. Med. 2020, 53, 34–42. [Google Scholar] [CrossRef]
- Peroni, L.A.; Toscaro, J.M.; Canateli, C.; Tonoli, C.C.C.; de Olivera, R.R.; Benedetti, C.E.; Coimbra, L.D.; Pereira, A.B.; Marques, R.E.; Proença-Modena, J.L.; et al. Serological Testing for COVID-19, Immunological Surveillance, and Exploration of Protective Antibodies. Front. Immunol. 2021, 12, 635701. [Google Scholar] [CrossRef]
- Danese, E.; Montagnana, M.; Salvagno, G.L.; Gelati, M.; Peserico, D.; Pighi, L.; de Nitto, S.; Henry, B.M.; Porru, S.; Lippi, G. Comparison of Five Commercial Anti-SARS-CoV-2 Total Antibodies and Igg Immunoassays after Vaccination with Bnt162b2 Mrna. J. Med. Biochem. 2021, 40, 335–340. [Google Scholar] [CrossRef] [PubMed]
- Premkumar, L.; Segovia-Chumbez, B.; Jadi, R.; Martinez, D.R.; Raut, R.; Markmann, A.; Cornaby, C.; Bartelt, L.; Weiss, S.; Park, Y.; et al. The Receptor Binding Domain of the Viral Spike Protein Is an Immunodominant and Highly Specific Target of Antibodies in SARS-CoV-2 Patients. Sci. Immunol. 2020, 5, eabc8413. [Google Scholar] [CrossRef]
- Malipiero, G.; D’Agaro, P.; Segat, L.; Moratto, A.; Villalta, D. Long-Term Decay of Anti-Rbd Igg Titers after Bnt162b2 Vaccination Is Not Mirrored by Loss of Neutralizing Bioactivity against SARS-CoV-2. Clin. Chim. Acta 2022, 524, 11–17. [Google Scholar] [CrossRef]
- Robinson, A.V.; Weaving, G.; Philips, B.J.; Eziefula, A.C.; Shipman, K.E.; Chevassut, T. Real-World Experience of SARS-CoV-2 Antibody Assays in Uk Healthcare Workers. Clin. Med. 2021, 21, e300. [Google Scholar] [CrossRef] [PubMed]
- Irsara, C.; Egger, A.E.; Prokop, W.; Nairz, M.; Loacker, L.; Sahanic, S.; Pizzini, A.; Sonnweber, T.; Holzer, B.; Mayer, W.; et al. Clinical Validation of the Siemens Quantitative SARS-CoV-2 Spike Igg Assay (Scovg) Reveals Improved Sensitivity and a Good Correlation with Virus Neutralization Titers. Clin. Chem. Lab. Med. 2021, 59, 1453–1462. [Google Scholar] [CrossRef]
- Charepe, N.; Gonçalves, J.; Juliano, A.M.; Lopes, D.G.; Canhão, H.; Soares, H.; Serrano, E.F. COVID-19 mRNA vaccine and antibody response in lactating women: A prospective cohort study. BMC Pregnancy Childbirth 2021, 21, 632. [Google Scholar] [CrossRef]
- Kiefer, M.K.; Allen, K.D.; Russo, J.R.; Ma’ayeh, M.; Gee, S.E.; Kniss, D.; Cackovic, M.; Costantine, M.M.; Rood, K.M. Decline in SARS-CoV-2 Antibodies over 6-Month Follow-up in Obstetrical Healthcare Workers. Am. J. Reprod. Immunol. 2021, 86, e13490. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Jin, F.; Cao, S.; Yuan, H.; Qu, J.; Zhang, J.; Li, Y.; Chen, X.; Song, W.; Xie, Z. Seroprevalence of SARS-CoV-2 Infections among Children Visiting a Hospital. Pediatr. Investig. 2020, 4, 236–241. [Google Scholar] [CrossRef] [PubMed]
- Tang, M.S.; Case, J.B.; Franks, C.E.; Chen, R.E.; Anderson, N.W.; Henderson, J.P.; Diamond, M.S.; Gronowski, A.M.; Farnsworth, C.W. Association between SARS-CoV-2 Neutralizing Antibodies and Commercial Serological Assays. Clin. Chem. 2020, 66, 1538–1547. [Google Scholar] [CrossRef]
- Ou, J.; Tan, M.; He, H.; Tan, H.; Mai, J.; Long, Y.; Jiang, X.; He, Q.; Huang, Y.; Li, Y.; et al. SARS-CoV-2 Antibodies and Associated Factors at Different Hospitalization Time Points in 192 COVID-19 Cases. J. Appl. Lab. Med. 2021, 6, 1133–1142. [Google Scholar] [CrossRef]
- Banga Ndzouboukou, J.L.; Zhang, Y.D.; Lei, Q.; Lin, X.S.; Yao, Z.J.; Fu, H.; Yuan, L.Y.; Fan, X.L. Human Igm and Igg Responses to an Inactivated SARS-CoV-2 Vaccine. Curr. Med. Sci. 2021, 41, 1081–1086. [Google Scholar] [CrossRef] [PubMed]
- Mitani, A.; Hamada, K.; Yoshikawa, N.; Morita, Y.; Horie, T.; Inoue, Y.; Saito, M.; Ishii, T.; Sunohara, M.; Takahashi, R.; et al. Epidemiological Study Using Igm and Igg Antibody Titers against SARS-CoV-2 in the University of Tokyo, Japan (Ut-Cats). J. Infect. Chemother. 2021, 27, 1342–1349. [Google Scholar] [CrossRef] [PubMed]
- Raposo, F.; Lippi, G. Antibody response induced by the boost overdose during COVID-19 heterologous prime-boost vaccination strategy. Clin. Chim. Acta 2021, 523, 201–204. [Google Scholar] [CrossRef] [PubMed]
- Müller, L.; Andrée, M.; Moskorz, W.; Drexler, I.; Walotka, L.; Grothmann, R.; Ptok, J.; Hillebrandt, J.; Ritchie, A.; Rabl, D.; et al. Age-dependent Immune Response to the Biontech/Pfizer BNT162b2 Coronavirus Disease 2019 Vaccination. Clin. Infect. Dis. 2021, 73, 2065–2072. [Google Scholar] [CrossRef] [PubMed]
- Al-Saadi, E.; Abdulnabi, M.A. Hematological Changes Associated with COVID-19 Infection. J. Clin. Lab. Anal. 2022, 36, e24064. [Google Scholar] [CrossRef]
- Rezaei, M.; Mahmoudi, S.; Mortaz, E.; Marjani, M. Immune cell profiling and antibody responses in patients with COVID-19. BMC Infect. Dis. 2021, 21, 646. [Google Scholar] [CrossRef]
- Santotoribio, J.D.; Jurado, D.N.; Lepe-Balsalobre, E. Evaluation of Routine Blood Tests for Diagnosis of Suspected Coronavirus Disease 2019. Clin. Lab. 2020, 66. [Google Scholar] [CrossRef]
- Li, Z.; Xiang, T.; Liang, B.; Deng, H.; Wang, H.; Feng, X.; Quan, X.; Wang, X.; Li, S.; Lu, S.; et al. Characterization of SARS-CoV-2-Specific Humoral and Cellular Immune Responses Induced by Inactivated COVID-19 Vaccines in a Real-World Setting. Front. Immunol. 2021, 12, 802858. [Google Scholar] [CrossRef]
- Zhang, J.; Xing, S.; Liang, D.; Hu, W.; Ke, C.; He, J.; Yuan, R.; Huang, Y.; Li, Y.; Liu, D.; et al. Differential Antibody Response to Inactivated COVID-19 Vaccines in Healthy Subjects. Front. Cell. Infect. Microbiol. 2021, 11, 791660. [Google Scholar] [CrossRef]


| Male (n = 16,335) | Female (n = 22,302) | ALL (n = 38,637) | ||
|---|---|---|---|---|
| Unit | Mean ± SD/Median (Range) | |||
| Age | Year | 48.71 ± 18.45/48.00 (98.00) | 46.16 ± 17.24/42.00 (101.00) | 47.24 ± 17.81/44.00 (101.00) |
| SARS-CoV-2 nucleic acid | Negative | |||
| Serum Anti-SARS-CoV-2 antibody | ||||
| IgM | S/CO | 0.16 ± 0.75/0.03 (25.78) | 0.13 ± 0.72/0.03 (25.29) | 0.14 ± 0.73/0.03 (25.78) |
| IgG | S/CO | 3.00 ± 10.82/0.08 (255.77) | 3.20 ± 10.89/0.06 (254.10) | 3.11 ± 10.86/0.07 (255.77) |
| The parameters of peripheral blood leukocytes | ||||
| Leukocyte | (×109/L) | 7.06 ± 2.37/6.67 (67.93) | 6.86 ± 2.36/6.45 (42.45) | 6.95 ± 2.37/6.55 (68.37) |
| Neutrophils | (×109/L) | 4.44 ± 2.19/3.99 (65.56) | 4.42 ± 2.19/3.94 (41.72) | 4.43 ± 2.18/3.96 (65.57) |
| % | 61.28 ± 10.74/60.70 (92.20) | 62.58 ± 10.73/62.20 (91.95) | 62.03 ± 10.76/61.60 (93.00) | |
| Eosinophils | (×109/L) | 0.15 ± 0.16/0.11 (6.03) | 0.11 ± 0.19/0.08 (21.16) | 0.13 ± 0.18/0.90 (21.16) |
| % | 2.20 ± 2.09/1.70 (46.00) | 1.68 ± 1.82/1.20 (73.20) | 1.90 ± 1.96/1.40 (73.20) | |
| Basophils | (×109/L) | 0.04 ± 0.02/0.03 (0.54) | 0.03 ± 0.02/0.03 (0.65) | 0.03 ± 0.02/0.03 (0.65) |
| % | 0.54 ± 0.31/0.50 (7.60) | 0.49 ± 0.29/0.50 (7.20) | 0.52 ± 0.30/0.50 (7.60) | |
| Lymphocytes | (×109/L) | 2.00 ± 0.75/1.95 (12.87) | 1.92 ± 0.67/1.87 (15.91) | 1.95 ± 0.71/1.90 (15.91) |
| % | 29.68 ± 9.96/30.10 (87.00) | 29.60 ± 9.82/29.80 (88.90) | 29.63 ± 9.88/29.90 (89.00) | |
| Monocytes | (×109/L) | 0.43 ± 0.16/0.40 (3.49) | 0.38 ± 0.15/0.35 (10.95) | 0.40 ± 0.16/0.37 (10.95) |
| % | 6.29 ± 1.88/6.00 (34.50) | 5.64 ± 1.69/5.40 (42.60) | 5.92 ± 1.81/5.70 (42.60) | |
| Model Term | Coefficient | Std. Error | t | p | 95% CI | |
|---|---|---|---|---|---|---|
| IgM | Intercept | −4.119 | 0.669 | −6.161 | <0.001 | −5.430–−2.809 |
| Age | −0.001 | 0.000 | −4.181 | <0.001 | −0.001–−0.000 | |
| Sex | 0.036 | 0.008 | 4.637 | <0.001 | 0.021–0.051 | |
| Neutrophils | −0.005 | 0.002 | −2.433 | 0.015 | −0.009–−0.001 | |
| Eosinophils | 0.012 | 0.022 | 1.426 | 0.583 | −0.031–0.056 | |
| Basophils | 0.059 | 0.199 | 0.477 | 0.634 | −0.296–0.486 | |
| Lymphocytes | 0.008 | 0.006 | 1.426 | 0.154 | −0.003–0.020 | |
| Monocytes | −0.049 | 0.029 | −1.698 | 0.090 | −0.105–0.008 | |
| IgG | Intercept | −312.912 | 9.782 | −31.99 | <0.001 | −332.084–−293.739 |
| Age | -0.038 | 0.003 | −12.29 | <0.001 | −0.045–−0.032 | |
| Sex | 0.008 | 0.113 | 0.067 | 0.947 | −0.214–0.230 | |
| Neutrophils | −0.103 | 0.030 | −3.459 | 0.001 | −0.162–−0.045 | |
| Eosinophils | −0.149 | 0.325 | −0.459 | 0.647 | −0.787–0.488 | |
| Basophils | −1.956 | 2.918 | −0.670 | 0.503 | −7.676–3.764 | |
| Lymphocytes | 0.274 | 0.086 | 3.179 | 0.001 | 0.105–0.443 | |
| Monocytes | −0.660 | 0.422 | −1.564 | 0.118 | −1.487–0.167 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, P.; Yang, N.; Xue, Y.; Zhou, J.; Wu, Y.; Wang, T.; Cui, L. The Fluctuation Trend of Serum Anti-SARS-CoV-2 IgM/IgG Antibodies Seroprevalence in the Non-COVID-19 Infected Population and Correlation with Peripheral Blood Leukocyte Parameters in Beijing, China, 2021: A Real-World Study. Vaccines 2022, 10, 571. https://doi.org/10.3390/vaccines10040571
Wang P, Yang N, Xue Y, Zhou J, Wu Y, Wang T, Cui L. The Fluctuation Trend of Serum Anti-SARS-CoV-2 IgM/IgG Antibodies Seroprevalence in the Non-COVID-19 Infected Population and Correlation with Peripheral Blood Leukocyte Parameters in Beijing, China, 2021: A Real-World Study. Vaccines. 2022; 10(4):571. https://doi.org/10.3390/vaccines10040571
Chicago/Turabian StyleWang, Pan, Nan Yang, Yuting Xue, Jiansuo Zhou, Yonghua Wu, Tiancheng Wang, and Liyuan Cui. 2022. "The Fluctuation Trend of Serum Anti-SARS-CoV-2 IgM/IgG Antibodies Seroprevalence in the Non-COVID-19 Infected Population and Correlation with Peripheral Blood Leukocyte Parameters in Beijing, China, 2021: A Real-World Study" Vaccines 10, no. 4: 571. https://doi.org/10.3390/vaccines10040571
APA StyleWang, P., Yang, N., Xue, Y., Zhou, J., Wu, Y., Wang, T., & Cui, L. (2022). The Fluctuation Trend of Serum Anti-SARS-CoV-2 IgM/IgG Antibodies Seroprevalence in the Non-COVID-19 Infected Population and Correlation with Peripheral Blood Leukocyte Parameters in Beijing, China, 2021: A Real-World Study. Vaccines, 10(4), 571. https://doi.org/10.3390/vaccines10040571






