Effectiveness of Monovalent Rotavirus Vaccine in Mozambique, a Country with a High Burden of Chronic Malnutrition
Abstract
:1. Introduction
2. Materials and Methods
2.1. Enrolment and Eligibility Criteria
2.2. Sample Collection and Laboratory Procedure
2.3. Exposure and Outcomes Definition
2.4. Statistical Analysis
2.5. Definitions of Covariates
2.6. Ethical Statement
3. Results
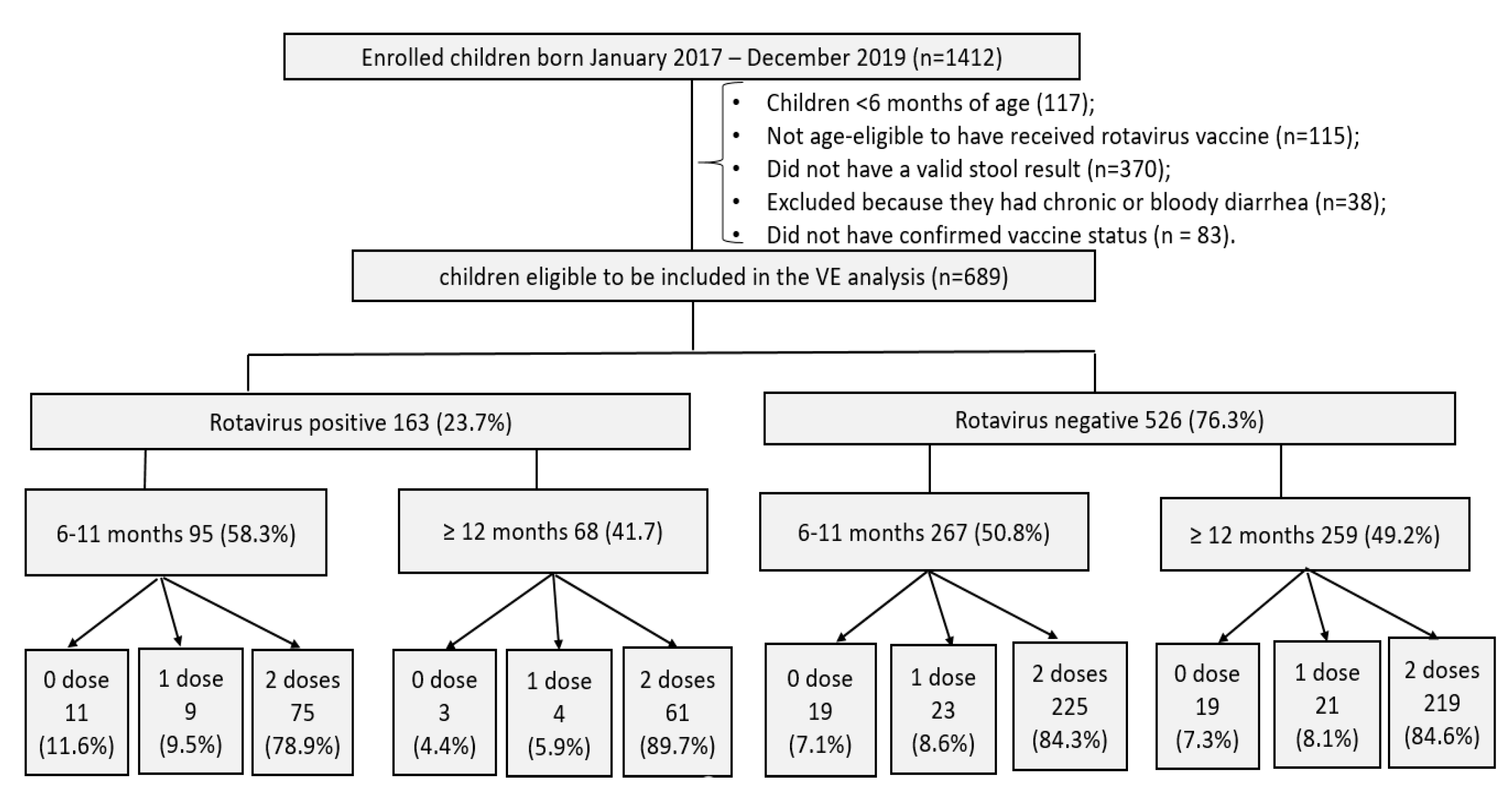
3.1. Rotavirus Vaccine Coverage
3.2. Vaccine Effectiveness
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Troeger, C.; Khalil, I.A.; Rao, P.C.; Cao, S.; Blacker, B.F.; Ahmed, T.; Armah, G.; Bines, J.E.; Brewer, T.G.; Colombara, D.V.; et al. Rotavirus vaccination and the global burden of rotavirus diarrhea among children younger than 5 years. JAMA Pediatrics 2018, 172, 958–965. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- GBD. Results Tool|GHDx: Mozambique: Global, Both Sexes, Under 5, All Causes, Etiology: Rotavirus. 2019. Available online: http://ghdx.healthdata.org/gbd-results-tool (accessed on 24 October 2021).
- Aliabadi, N.; Antoni, S.; Mwenda, J.M.; Weldegebriel, G.; Biey, J.N.M.; Cheikh, D.; Fahmy, K.; Teleb, N.; Ashmony, H.A.; Ahmed, H.; et al. Global impact of rotavirus vaccine introduction on rotavirus hospitalisations among children under 5 years of age, 2008–2016: Findings from the global rotavirus surveillance network. Lancet Glob. Health 2019, 7, e893–e903. [Google Scholar] [CrossRef] [Green Version]
- Burnett, E.; Parashar, U.D.; Tate, J.E. Real-world effectiveness of rotavirus vaccines, 2006–2019: A literature review and meta-analysis. Lancet Glob. Health 2020, 8, e1195–e1202. [Google Scholar] [CrossRef]
- Cochrane. Update of a Systematic Review and Meta-Analysis of the Safety, Effectiveness and Efficacy of Childhood Schedules using Rotavirus Vaccines. 2017. Available online: https://www.who.int/vaccine_safety/publications/WHO_Rotavirus_vaccines_systematic_review_Cochrane.pdf (accessed on 1 March 2022).
- Burnett, E.; Parashar, U.D.; Tate, J.E. Rotavirus infection, illness, and vaccine performance in malnourished children: A review of the literature. Pediatric Infect. Dis. J. 2021, 40, 930–936. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization; United Nations Children's Fund (UNICEF); The World Bank. Levels and Trends in Child Malnutrition: UNICEF; World Health Organization: Geneva, Switzerland, 2021; ISBN 978-92-4-002525-7. [Google Scholar]
- IDS. Moçambique Inquérito Demográfico e de Saúde 2011; MISAU, INE e ICFI: Calverton, MD, USA, 2011; p. 412. [Google Scholar]
- Gastañaduy, P.A.; Steenhoff, A.P.; Mokomane, M.; Esona, M.D.; Bowen, M.D.; Jibril, H.; Pernica, J.M.; Mazhani, L.; Smieja, M.; Tate, J.E.; et al. Effectiveness of monovalent rotavirus vaccine after programmatic implementation in Botswana: A multisite prospective case-control study. Clin. Infect. Dis. 2016, 62 (Suppl. 2), S161–S167. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mujuru, H.A.; Burnett, E.; Nathoo, K.J.; Ticklay, I.; Gonah, N.A.; Mukaratirwa, A.; Berejena, C.; Manangazira, P.; Rupfutse, M.; Weldegebriel, G.G.; et al. Monovalent rotavirus vaccine effectiveness against rotavirus hospitalizations among children in Zimbabwe. Clin. Infect. Dis. 2019, 69, 1339–1344. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bar-Zeev, N.; Jere, K.C.; Bennett, A.; Pollock, L.; Tate, J.E.; Nakagomi, O.; Iturriza-Gomara, M.; Costello, A.; Mwansambo, C.; Parashar, U.D.; et al. Population Impact and effectiveness of monovalent rotavirus vaccination in urban malawian children 3 years after vaccine introduction: Ecological and case-control analyses. Clin. Infect. Dis. 2016, 62 (Suppl. 2), S213–S219. [Google Scholar] [CrossRef] [PubMed]
- Khagayi, S.; Omore, R.; Otieno, G.P.; Ogwel, B.; Ochieng, J.B.; Juma, J.; Apondi, E.; Bigogo, G.; Onyango, C.; Ngama, M.; et al. Effectiveness of monovalent rotavirus vaccine against hospitalization with acute rotavirus gastroenteritis in Kenyan children. Clin. Infect. Dis. 2020, 70, 2298–2305. [Google Scholar] [CrossRef] [Green Version]
- de Deus, N.; Chilaúle, J.J.; Cassocera, M.; Bambo, M.; Langa, J.S.; Sitoe, E.; Chissaque, A.; Anapakala, E.; Sambo, J.; Guimarães, E.L.; et al. Early impact of rotavirus vaccination in children less than five years of age in Mozambique. Vaccine 2018, 36, 7205–7209. [Google Scholar] [CrossRef]
- Bauhofer, A.F.L.; Cossa-Moiane, I.; Marques, S.; Guimarães, E.L.; Munlela, B.; Anapakala, E.; Chilaúle, J.J.; Cassocera, M.; Langa, J.S.; Chissaque, A.; et al. Intestinal protozoan infections among children 0–168 months with diarrhea in Mozambique: June 2014–January 2018. PLOS Negl. Trop. Dis. 2020, 14, e0008195. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chissaque, A.; Cassocera, M.; Gasparinho, C.; Langa, J.S.; Bauhofer, A.F.L.; Chilaúle, J.J.; João, E.D.; Munlela, B.A.; Sambo, J.A.M.; Boene, S.S.; et al. Rotavirus a infection in children under five years old with a double health problem: Undernutrition and diarrhoea—A cross-sectional study in four provinces of Mozambique. BMC Infect. Dis. 2021, 21, 18. [Google Scholar] [CrossRef] [PubMed]
- Nhacolo, A.; Jamisse, E.; Augusto, O.; Matsena, T.; Hunguana, A.; Mandomando, I.; Arnaldo, C.; Munguambe, K.; Macete, E.; Alonso, P.; et al. Cohort profile update: Manhiça Health and Demographic Surveillance System (HDSS) of the Manhiça Health Research Centre (CISM). Int. J. Epidemiol. 2021, 50, 395. [Google Scholar] [CrossRef] [PubMed]
- Kotloff, K.L.; Nataro, J.P.; Blackwelder, W.C.; Nasrin, D.; Farag, T.H.; Panchalingam, S.; Wu, Y.; Sow, S.O.; Sur, D.; Breiman, R.F.; et al. Burden and aetiology of diarrhoeal disease in infants and young children in developing countries (the Global Enteric Multicenter Study, GEMS): A prospective, case-control study. Lancet 2013, 382, 209–222. [Google Scholar] [CrossRef]
- WHO Diarrhoeal Disease. [Internet]. 2017 [Cited 20 November 2021]. Available online: https://www.who.int/news-room/factsheets/detail/diarrhoeal-dis (accessed on 7 March 2020).
- Gentsch, J.R.; Glass, R.I.; Woods, P.; Gouvea, V.; Gorziglia, M.; Flores, J.; Das, B.K.; Bhan, M.K. Identification of group a rotavirus gene 4 types by polymerase chain reaction. J. Clin. Microbiol. 1992, 30, 1365–1373. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gouvea, V.; Glass, R.I.; Woods, P.; Taniguchi, K.; Clark, H.F.; Forrester, B.; Fang, Z.Y. Polymerase chain reaction amplification and typing of rotavirus nucleic acid from stool specimens. J. Clin. Microbiol. 1990, 28, 276–282. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- João, E.D.; Munlela, B.; Chissaque, A.; Chilaúle, J.; Langa, J.; Augusto, O.; Boene, S.S.; Anapakala, E.; Sambo, J.; Guimarães, E.; et al. Molecular epidemiology of rotavirus a strains pre- and post-vaccine (Rotarix®) introduction in Mozambique, 2012–2019: Emergence of genotypes G3P[4] and G3P[8]. Pathogens 2020, 9, 671. [Google Scholar] [CrossRef]
- World Health Organization. WHO Anthro Survey Analyser. 2019. Available online: https://www.who.int/tools/child-growth-standards/software (accessed on 1 March 2022).
- Groome, M.J.; Page, N.; Cortese, M.M.; Moyes, J.; Zar, H.J.; Kapongo, C.N.; Mulligan, C.; Diedericks, R.; Cohen, C.; Fleming, J.A.; et al. Effectiveness of monovalent human rotavirus vaccine against admission to hospital for acute rotavirus diarrhoea in South African children: A case-control study. Lancet Infect. Dis. 2014, 14, 1096–1104. [Google Scholar] [CrossRef]
- Abeid, K.A.; Jani, B.; Cortese, M.M.; Kamugisha, C.; Mwenda, J.M.; Pandu, A.S.; Msaada, K.A.; Mohamed, A.S.; Khamis, A.U.; Parashar, U.D.; et al. Monovalent rotavirus vaccine effectiveness and impact on rotavirus hospitalizations in Zanzibar, Tanzania: Data from the first 3 years after introduction. J. Infect. Dis. 2017, 215, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Madhi, S.A.; Cunliffe, N.A.; Steele, D.; Witte, D.; Kirsten, M.; Louw, C.; Ngwira, B.; Victor, J.C.; Gillard, P.H.; Cheuvart, B.B.; et al. Effect of human rotavirus vaccine on severe diarrhea in african infants. Malawi Med. J. 2016, 28, 108–114. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Armah, G.E.; Sow, S.O.; Breiman, R.F.; Dallas, M.J.; Tapia, M.D.; Feikin, D.R.; Binka, F.N.; Steele, A.D.; Laserson, K.F.; Ansah, N.A.; et al. Efficacy of pentavalent rotavirus vaccine against severe rotavirus gastroenteritis in infants in developing countries in sub-saharan africa: A randomised, double-blind, placebo-controlled trial. Lancet 2010, 376, 606–614. [Google Scholar] [CrossRef]
- Lopman, B.A.; Pitzer, V.E.; Sarkar, R.; Gladstone, B.; Patel, M.; Glasser, J.; Gambhir, M.; Atchison, C.; Grenfell, B.T.; Edmunds, W.J.; et al. Understanding reduced rotavirus vaccine efficacy in low socio-economic settings. PLoS ONE 2012, 7, e41720. [Google Scholar] [CrossRef]
- Baker, J.M.; Tate, J.E.; Leon, J.; Haber, M.J.; Lopman, B.A. Antirotavirus IgA seroconversion rates in children who receive concomitant oral poliovirus vaccine: A secondary, pooled analysis of phase II and III trial data from 33 countries. PLoS Med. 2019, 16, e1003005. [Google Scholar] [CrossRef] [Green Version]
- Otero, C.E.; Langel, S.N.; Blasi, M.; Permar, S.R. Maternal antibody interference contributes to reduced rotavirus vaccine efficacy in developing countries. PLOS Pathog. 2020, 16, e1009010. [Google Scholar] [CrossRef] [PubMed]
- Cates, J.E.; Amin, A.B.; Tate, J.E.; Lopman, B.; Parashar, U. Do rotavirus strains affect vaccine effectiveness? A systematic review and meta-analysis. Pediatric Infect. Dis. J. 2021, 40, 1135–1143. [Google Scholar] [CrossRef]
- Jonesteller, C.L.; Burnett, E.; Yen, C.; Tate, J.E.; Parashar, U.D. Effectiveness of rotavirus vaccination: A systematic review of the first decade of global postlicensure data, 2006–2016. Clin. Infect. Dis. 2017, 65, 840–850. [Google Scholar] [CrossRef] [PubMed]
- Clark, A.; van Zandvoort, K.; Flasche, S.; Sanderson, C.; Bines, J.; Tate, J.; Parashar, U.; Jit, M. Efficacy of live oral rotavirus vaccines by duration of follow-up: A meta-regression of randomised controlled trials. Lancet Infect. Dis. 2019, 19, 717–727. [Google Scholar] [CrossRef] [Green Version]
- Pitzer, V.E.; Bennett, A.; Bar-Zeev, N.; Jere, K.C.; Lopman, B.A.; Lewnard, J.A.; Parashar, U.D.; Cunliffe, N.A. Evaluating strategies to improve rotavirus vaccine impact during the second year of life in Malawi. Sci. Transl. Med. 2019, 11, eaav6419. [Google Scholar] [CrossRef] [PubMed]
- Prendergast, A.J. Malnutrition and vaccination in developing countries. Philos. Trans. R. Soc. B Biol. Sci. 2015, 370, 20140141. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hoest, C.; Seidman, J.C.; Pan, W.; Ambikapathi, R.; Kang, G.; Kosek, M.; Knobler, S.; Mason, C.J.; Miller, M. MAL-ED Network investigators evaluating associations between vaccine response and malnutrition, gut function, and enteric infections in the MAL-ED cohort study: Methods and challenges. Clin. Infect. Dis. 2014, 59 (Suppl. 4), S273–S279. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bosomprah, S.; Beach, L.B.; Beres, L.K.; Newman, J.; Kapasa, K.; Rudd, C.; Njobvu, L.; Guffey, B.; Hubbard, S.; Foo, K.; et al. Findings from a comprehensive diarrhoea prevention and treatment programme in Lusaka, Zambia. BMC Public Health 2016, 16, 475. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Colgate, E.R.; Haque, R.; Dickson, D.M.; Carmolli, M.P.; Mychaleckyj, J.C.; Nayak, U.; Qadri, F.; Alam, M.; Walsh, M.C.; Diehl, S.A.; et al. Delayed dosing of oral rotavirus vaccine demonstrates decreased risk of rotavirus gastroenteritis associated with serum zinc: A randomized controlled trial. Clin. Infect. Dis. 2016, 63, 634–641. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- National Department of Health (NDoH), Statistics South Africa (Stats SA), South African Medical Research Council (SAMRC), and ICF South Africa Demographic and Health Survey 2016. 2019. [Cited 18 November 2021]. Available online: https://dhsprogram.com/pubs/pdf/FR337/FR337.pdf (accessed on 1 March 2022).
- Zimbabwe National Statistics Agency and ICF International Zimbabwe Demographic and Health Survey 2015: Final Report. 2016. [Cited 18 November 2021]. Available online: https://dhsprogram.com/pubs/pdf/FR322/FR322.pdf (accessed on 1 March 2022).
- Ministry of Health, Community Development, Gender, Elderly and Children (MoHCDGEC) [Tanzania Mainland], Ministry of Health (MoH) [Zanzibar], National Bureau of Statistics (NBS), Office of the Chief Government Statistician (OCGS), and ICF Tanzania Demographic and Health Survey and Malaria Indicator Survey (TDHS-MIS) 2015–2016. 2016. [Cited 20 November 2021]. Available online: https://dhsprogram.com/pubs/pdf/fr321/fr321.pdf (accessed on 1 March 2022).
- Chissaque, A.; Bauhofer, A.F.L.; Cossa-Moiane, I.; Sitoe, E.; Munlela, B.; João, E.D.; Langa, J.S.; Chilaúle, J.J.; Boene, S.S.; Cassocera, M.; et al. Rotavirus a infection in pre- and post-vaccine period: Risk factors, genotypes distribution by vaccination status and age of children in Nampula Province, Northern Mozambique (2015–2019). PLoS ONE 2021, 16, e0255720. [Google Scholar] [CrossRef] [PubMed]
- Acácio, S.; Mandomando, I.; Nhampossa, T.; Quintó, L.; Vubil, D.; Sacoor, C.; Kotloff, K.; Farag, T.; Nasrin, D.; Macete, E.; et al. Risk factors for death among children 0–59 months of age with moderate-to-severe diarrhea in Manhiça District, Southern Mozambique. BMC Infect. Dis. 2019, 19, 322. [Google Scholar] [CrossRef] [PubMed]
| Characteristic | Cases (n = 163) | Controls (n = 526) | p Value * |
|---|---|---|---|
| RV Vaccination Status | |||
| Unvaccinated | 14 (8.6%) | 38 (7.2%) | 0.84 |
| Partially Vaccinated | 13 (8%) | 44 (8.4%) | - |
| Fully Vaccinated | 136 (83.4%) | 444 (84.4%) | - |
| Year of Admission | |||
| 2017 | 91 (55.8%) | 216 (41.1%) | 0.003 |
| 2018 | 40 (24.5%) | 193 (36.7%) | - |
| 2019 | 32 (19.6%) | 117 (22.2%) | - |
| Sex | |||
| Female | 73 (44.8%) | 196 (37.3%) | 0.10 |
| Male | 90 (55.2%) | 330 (62.7%) | - |
| Age Group | |||
| 6–8 months | 42 (25.8%) | 114 (21.7%) | 0.45 |
| 9–11 months | 53 (32.5%) | 153 (29.1%) | - |
| 12–17 months | 43 (26.4%) | 153 (29.1%) | - |
| 18–23 months | 17 (10.4%) | 59 (11.2%) | - |
| 24–35 months | 6 (3.7%) | 40 (7.6%) | - |
| 36–47 months | 2 (1.2%) | 7 (1.3%) | - |
| Number Persons in HH (median) | 5.0 (4.0, 7.0) | 4.0 (3.0, 6.0) | 0.001 |
| Number Children in HH (median) | 2.0 (1.0, 2.0) | 1.0 (1.0, 2.0) | 0.003 |
| Anthropometrics | |||
| Normal height for age | 75 (63.6%) | 234 (63.8%) | 1.00 |
| Stunted (HAZ < −2) | 43 (36.4%) | 133 (36.2%) | - |
| Missing | 45 | 159 | - |
| Surveillance Site | <0.001 | ||
| Centro de Saude da Manhica | 27 (16.6%) | 80 (15.2%) | |
| Hospital Central da Beira | 5 (3.1%) | 34 (6.5%) | - |
| Hospital Central de Maputo | 13 (8.0%) | 79 (15.0%) | - |
| Hospital Central de Nampula | 59 (36.2%) | 112 (21.3%) | - |
| Hospital Gera Jose Macamo | 17 (10.4%) | 59 (11.2%) | - |
| Hospital Geral de Mavalane | 25 (15.3%) | 137 (26.0%) | - |
| Hospital Geral de Qualimane | 17 (10.4%) | 25 (4.8%) | - |
| Electricity | |||
| Yes | 108 (66.7%) | 422 (80.7%) | <0.001 |
| No | 54 (33.3%) | 101 (19.3%) | - |
| Unknown | 1 | 3 | - |
| House Type | |||
| Reed house | 12 (7.4%) | 31 (6%) | <0.001 |
| Mud house | 57 (35%) | 96 (18.4%) | - |
| Brick house | 94 (57.7%) | 394 (75.6%) | - |
| Missing | 0 | 5 | - |
| Fridge | |||
| Yes | 67 (41.1%) | 286 (54.5%) | 0.004 |
| No | 96 (58.9%) | 239 (45.5%) | - |
| Unknown | 0 | 1 | - |
| Cell Phone | |||
| Yes | 124 (76.5%) | 445 (84.8%) | 0.021 |
| No | 38 (23.5%) | 80 (15.2%) | - |
| Unknown | 1 | 1 | - |
| Model | Cases | Controls | Crude | Adjusted * | ||||
|---|---|---|---|---|---|---|---|---|
| Vaccinated/Total (%) | Vaccinated/Total (%) | VE | 95% CI | p-Value | VE | 95% CI | p-Value | |
| 6 to 59 months | 148/162 (91.4%) | 488/526 (92.8%) | 18 | (−61, 56) | 0.55 | 35 | (−30, 66) | 0.20 |
| 6 to 8 months | 37/42 (88.1%) | 107/114 (93.9%) | 52 | (−72, 85) | 0.24 | 56 | (−65, 88) | 0.21 |
| 9 to 11 months | 47/53 (88.7%) | 141/153 (92.2%) | 33 | (−100, 76) | 0.44 | 46 | (−70, 82) | 0.27 |
| 6 to 11 months | 84/95 (88.4%) | 248/267 (92.9%) | 41 | (−32, 73) | 0.18 | 52 | (−11, 79) | 0.078 |
| 12 to 23 months | 56/59 (94.9%) | 195/212 (92%) | −63 | (−615, 48) | 0.45 | −24 | (−459, 62) | 0.74 |
| Severe: Modified Vesikari Score ≥ 11 | 51/58 (87.9%) | 73/81 (90.1%) | 20 | (−141, 73) | 0.68 | 54 | (−58, 87) | 0.21 |
| Less severe: Modified Vesikari Score < 11 | 25/28 (89.3%) | 69/78 (88.5%) | −9 | (−419, 70) | 0.91 | −18 | (−510, 71) | 0.82 |
| Stunted (HAZ < −2) | 36/43 (83.7%) | 114/133 (85.7%) | 14 | (−134, 65) | 0.75 | 14 | (−138, 66) | 0.76 |
| Not Stunted (HAZ ≥ −2) | 72/75 (96%) | 226/234 (96.6%) | 15 | (−296, 76) | 0.81 | 59 | (−125, 91) | 0.27 |
| G1P[8] Rotavirus | 21/23 (91.3%) | 488/526 (92.8%) | 18 | (−423, 77) | 0.79 | 30 | (−355, 81) | 0.64 |
| Non-G1P[8] Rotavirus # | 127/139 (91.4%) | 488/526 (92.8%) | 18 | (−69, 57) | 0.58 | 35 | (−35, 67) | 0.22 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chissaque, A.; Burke, R.M.; Guimarães, E.L.; Manjate, F.; Nhacolo, A.; Chilaúle, J.; Munlela, B.; Chirinda, P.; Langa, J.S.; Cossa-Moiane, I.; et al. Effectiveness of Monovalent Rotavirus Vaccine in Mozambique, a Country with a High Burden of Chronic Malnutrition. Vaccines 2022, 10, 449. https://doi.org/10.3390/vaccines10030449
Chissaque A, Burke RM, Guimarães EL, Manjate F, Nhacolo A, Chilaúle J, Munlela B, Chirinda P, Langa JS, Cossa-Moiane I, et al. Effectiveness of Monovalent Rotavirus Vaccine in Mozambique, a Country with a High Burden of Chronic Malnutrition. Vaccines. 2022; 10(3):449. https://doi.org/10.3390/vaccines10030449
Chicago/Turabian StyleChissaque, Assucênio, Rachel M. Burke, Esperança L. Guimarães, Filomena Manjate, Arsénio Nhacolo, Jorfélia Chilaúle, Benilde Munlela, Percina Chirinda, Jerónimo S. Langa, Idalécia Cossa-Moiane, and et al. 2022. "Effectiveness of Monovalent Rotavirus Vaccine in Mozambique, a Country with a High Burden of Chronic Malnutrition" Vaccines 10, no. 3: 449. https://doi.org/10.3390/vaccines10030449
APA StyleChissaque, A., Burke, R. M., Guimarães, E. L., Manjate, F., Nhacolo, A., Chilaúle, J., Munlela, B., Chirinda, P., Langa, J. S., Cossa-Moiane, I., Anapakala, E., Bauhofer, A. F. L., Garrine, M., João, E. D., Sambo, J., Gonçalves, L., Weldegebriel, G., Shaba, K., Bello, I. M., ... de Deus, N. (2022). Effectiveness of Monovalent Rotavirus Vaccine in Mozambique, a Country with a High Burden of Chronic Malnutrition. Vaccines, 10(3), 449. https://doi.org/10.3390/vaccines10030449








