Public Reaction towards the Potential Side Effects of a COVID-19 Vaccine: An Italian Cross-Sectional Study
Abstract
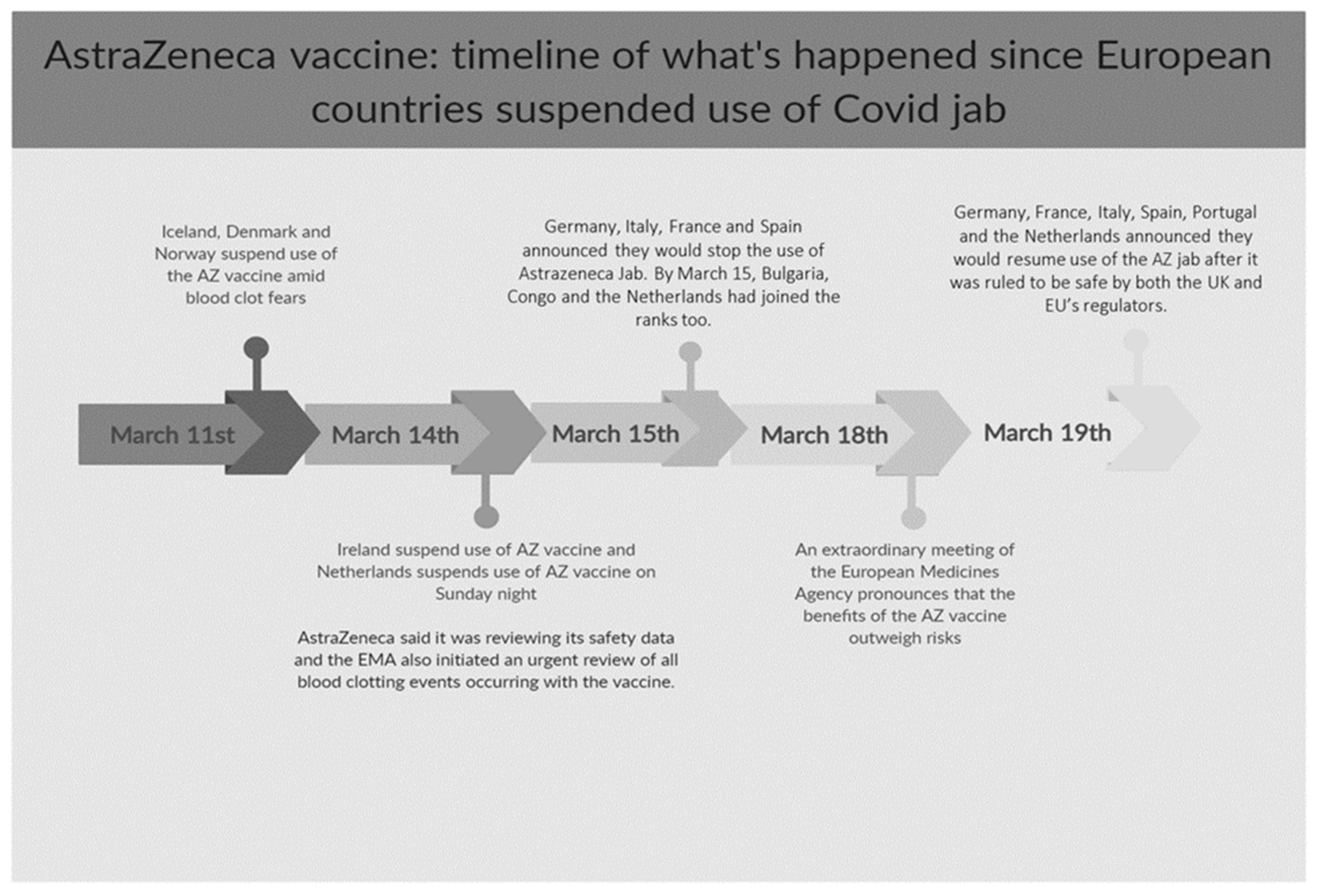
:1. Introduction
2. Materials and Methods
2.1. Participants and Procedure
2.2. Measures
- Gender;
- Age;
- Italian region of residence, which was then recoded to divide the sample into four main geographical areas (north-west, north-east, center, and south and islands);
- Education: participants were asked their higher-ranked school degree, and were then grouped into three clusters (middle school or lower, high school degree, university degree or higher);
- Average family net income on a monthly basis, participants were then divided in two groups (above and below the median value of 1800 €/month);
- Previous vaccinal behavior (“have you ever been vaccinated against influenza?”) answered in a yes/no fashion;
- Closeness to COVID-19: participants were asked three questions (“have you ever been diagnosed with COVID-19, confirmed by a positive test?”, “has one of your relatives been diagnosed with COVID-19, confirmed by a positive test?”, and “have you been quarantined due to a close contact with a suspect COVID-19 case?”) and were asked to answer yes/no to each one.
2.3. Data Analysis
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Puri, N.; Coomes, E.A.; Haghbayan, H.; Gunaratne, K. Social Media and Vaccine Hesitancy: New Updates for the Era of COVID-19 and Globalized Infectious Diseases. Hum. Vaccines Immunother. 2020, 16, 2586–2593. [Google Scholar] [CrossRef] [PubMed]
- Peretti-Watel, P.; Seror, V.; Cortaredona, S.; Launay, O.; Raude, J.; Verger, P.; Fressard, L.; Beck, F.; Legleye, S.; L’Haridon, O.; et al. A Future Vaccination Campaign against COVID-19 at Risk of Vaccine Hesitancy and Politicisation. Lancet Infect. Dis. 2020, 20, 769–770. [Google Scholar] [CrossRef]
- Palamenghi, L.; Barello, S.; Boccia, S.; Graffigna, G. Mistrust in Biomedical Research and Vaccine Hesitancy: The Forefront Challenge in the Battle against COVID-19 in Italy. Eur. J. Epidemiol. 2020, 35, 785–788. [Google Scholar] [CrossRef] [PubMed]
- Sallam, M. COVID-19 Vaccine Hesitancy Worldwide: A Concise Systematic Review of Vaccine Acceptance Rates. Vaccines 2021, 9, 160. [Google Scholar] [CrossRef] [PubMed]
- Mesch, G.S.; Schwirian, K.P. Social and Political Determinants of Vaccine Hesitancy: Lessons Learned from the H1N1 Pandemic of 2009–2010. Am. J. Infect. Control 2015, 43, 1161–1165. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, F.P.; Tafuri, S. A Public Health Perspective on the Responsibility of Mass Media for the Outcome of the Anti-COVID-19 Vaccination Campaign: The AstraZeneca Case. Ann. Ig. 2022. [Google Scholar] [CrossRef]
- World Health Organization. Managing the COVID-19 Infodemic: Promoting Healthy Behaviours and Mitigating the Harm from Misinformation and Disinformation. Available online: https://www.who.int/news/item/23-09-2020-managing-the-covid-19-infodemic-promoting-healthy-behaviours-and-mitigating-the-harm-from-misinformation-and-disinformation (accessed on 27 July 2021).
- Hyland-Wood, B.; Gardner, J.; Leask, J.; Ecker, U.K.H. Toward Effective Government Communication Strategies in the Era of COVID-19. Humanit. Soc. Sci. Commun. 2021, 8, 30. [Google Scholar] [CrossRef]
- Betsch, C. How Behavioural Science Data Helps Mitigate the COVID-19 Crisis. Nat. Hum. Behav. 2020, 4, 438. [Google Scholar] [CrossRef] [Green Version]
- Van Bavel, J.J.; Baicker, K.; Boggio, P.S.; Capraro, V.; Cichocka, A.; Cikara, M.; Crockett, M.J.; Crum, A.J.; Douglas, K.M.; Druckman, J.N.; et al. Using Social and Behavioural Science to Support COVID-19 Pandemic Response. Nat. Hum. Behav. 2020, 4, 460–471. [Google Scholar] [CrossRef]
- Krpan, D.; Makki, F.; Saleh, N.; Brink, S.I.; Klauznicer, H.V. When Behavioural Science Can Make a Difference in Times of COVID-19. Behav. Public Policy 2021, 5, 153–179. [Google Scholar] [CrossRef]
- Haviland, A.M.; Elliott, M.N.; Hambarsoomian, K.; Lurie, N. Immunization Disparities by Hispanic Ethnicity and Language Preference. Arch. Intern. Med. 2011, 171, 158–165. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hebert, P.L.; Frick, K.D.; Kane, R.L.; McBean, A.M. The Causes of Racial and Ethnic Differences in Influenza Vaccination Rates among Elderly Medicare Beneficiaries. Health Serv. Res. 2005, 40, 517–538. [Google Scholar] [CrossRef] [Green Version]
- Ojha, R.P.; Stallings-Smith, S.; Flynn, P.M.; Adderson, E.E.; Offutt-Powell, T.N.; Gaur, A.H. The Impact of Vaccine Concerns on Racial/Ethnic Disparities in Influenza Vaccine Uptake among Health Care Workers. Am. J. Public Health 2015, 105, e35–e41. [Google Scholar] [CrossRef] [PubMed]
- Rangel, M.C.; Shoenbach, V.J.; Weigle, K.A.; Hogan, V.K.; Strauss, R.P.; Bangdiwala, S.I. Racial and Ethnic Disparities in Influenza Vaccination among Elderly Adults. J. Gen. Intern. Med. 2005, 20, 426–431. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Straits-Tröster, K.A.; Kahwati, L.C.; Kinsinger, L.S.; Orelien, J.; Burdick, M.B.; Yevich, S.J. Racial/Ethnic Differences in Influenza Vaccination in the Veterans Affairs Healthcare System. Am. J. Prev. Med. 2006, 31, 375–382. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.; Stevens, G.D. Vulnerability and Unmet Health Care Needs: The Influence of Multiple Risk Factors. J. Gen. Intern. Med. 2005, 20, 148–154. [Google Scholar] [CrossRef]
- Shavers, V.L.; Fagan, P.; Jones, D.; Klein, W.M.P.; Boyington, J.; Moten, C.; Rorie, E. The State of Research on Racial/Ethnic Discrimination in The Receipt of Health Care. Am. J. Public Health 2012, 102, 953–966. [Google Scholar] [CrossRef] [Green Version]
- Shui, I.M.; Weintraub, E.S.; Gust, D.A. Parents Concerned About Vaccine Safety. Am. J. Prev. Med. 2006, 31, 244–251. [Google Scholar] [CrossRef]
- Leask, J.; Willaby, H.W.; Kaufman, J. The Big Picture in Addressing Vaccine Hesitancy. Hum. Vaccines Immunother. 2014, 10, 2600–2602. [Google Scholar] [CrossRef] [Green Version]
- Hornsey, M.J.; Harris, E.A.; Fielding, K.S. The Psychological Roots of Anti-Vaccination Attitudes: A 24-Nation Investigation. Health Psychol. 2018, 37, 307–315. [Google Scholar] [CrossRef]
- Rossen, I.; Hurlstone, M.J.; Dunlop, P.D.; Lawrence, C. Accepters, Fence Sitters, or Rejecters: Moral Profiles of Vaccination Attitudes. Soc. Sci. Med. 2019, 224, 23–27. [Google Scholar] [CrossRef] [PubMed]
- Rieger, M. Triggering Altruism Increases the Willingness to Get Vaccinated against COVID-19. Soc. Health Behav. 2020, 3, 78. [Google Scholar] [CrossRef]
- Johnson, M.O. Personality Correlates of HIV Vaccine Trial Participation. Personal. Individ. Differ. 2000, 29, 459–467. [Google Scholar] [CrossRef]
- Aharon, A.A.; Nehama, H.; Rishpon, S.; Baron-Epel, O. Human Vaccines & Immunotherapeutics A Path Analysis Model Suggesting the Association between Health Locus of Control and Compliance with Childhood Vaccinations Anat Amit Aharon, Haim Nehama, Shmuel Rishpon & Orna Baron-Epel A Path Analysis Model Suggestin. Taylor Fr. 2018, 14, 1618–1625. [Google Scholar] [CrossRef]
- Damnjanović, K.; Graeber, J.; Ilić, S.; Lam, W.Y.; Lep, Ž.; Morales, S.; Pulkkinen, T.; Vingerhoets, L. Parental Decision-Making on Childhood Vaccination. Front. Psychol. 2018, 9, 735. [Google Scholar] [CrossRef] [PubMed]
- Murakami, H.; Kobayashi, M.; Hachiya, M.; Khan, Z.S.; Hassan, S.Q.; Sakurada, S. Refusal of Oral Polio Vaccine in Northwestern Pakistan: A Qualitative and Quantitative Study. Vaccine 2014, 32, 1382–1387. [Google Scholar] [CrossRef] [PubMed]
- Jamison, A.M.; Quinn, S.C.; Freimuth, V.S. “You Don’t Trust a Government Vaccine”: Narratives of Institutional Trust and Influenza Vaccination among African American and White Adults. Soc. Sci. Med. 2019, 221, 87–94. [Google Scholar] [CrossRef]
- Kennedy, J. Populist Politics and Vaccine Hesitancy in Western Europe: An Analysis of National-Level Data. Eur. J. Public Health 2019, 29, 512–516. [Google Scholar] [CrossRef]
- Nihlén Fahlquist, J. Vaccine Hesitancy and Trust. Ethical Aspects of Risk Communication. Scand. J. Public Health 2018, 46, 182–188. [Google Scholar] [CrossRef]
- Suk, J.E.; Lopalco, P.; Celentano, L.P. Hesitancy, Trust and Individualism in Vaccination Decision-Making. PLoS Curr. 2015, 7. [Google Scholar] [CrossRef]
- Graffigna, G.; Barello, S.; Savarese, M.; Palamenghi, L.; Castellini, G.; Bonanomi, A.; Lozza, E. Measuring Italian Citizens′ Engagement in the First Wave of the COVID-19 Pandemic Containment Measures A Cross-Sectional Study. PLoS ONE 2020, 15, e0238613. [Google Scholar] [CrossRef] [PubMed]
- Valliant, R.; Dever, J.A.; Kreuter, F. (Eds.) Basic Steps in Weighting. In Practical Tools for Designing and Weighting Survey Samples; Statistics for Social and Behavioral Sciences; Springer: New York, NY, USA, 2013; pp. 307–348. ISBN 978-1-4614-6449-5. [Google Scholar]
- Bruder, M.; Haffke, P.; Neave, N.; Nouripanah, N.; Imhoff, R. Measuring Individual Differences in Generic Beliefs in Conspiracy Theories Across Cultures: Conspiracy Mentality Questionnaire. Front. Psychol. 2013, 4, 225. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Betsch, C.; Schmid, P.; Heinemeier, D.; Korn, L.; Holtmann, C.; Böhm, R. Beyond Confidence: Development of a Measure Assessing the 5C Psychological Antecedents of Vaccination. PLoS ONE 2018, 13, e0208601. [Google Scholar] [CrossRef] [Green Version]
- Knoll, M.D.; Wonodi, C. Oxford–AstraZeneca COVID-19 Vaccine Efficacy. Lancet 2021, 397, 72–74. [Google Scholar] [CrossRef]
- Leask, J. Restoring Confidence after the Covid-19 Oxford/AstraZeneca Vaccine Pause Will Be an Uphill Battle. Available online: https://blogs.bmj.com/bmj/2021/03/18/restoring-confidence-after-the-covid-19-oxford-astrazeneca-vaccine-pause-will-be-an-uphill-battle/ (accessed on 25 July 2021).
- Paris, C.; Bénézit, F.; Geslin, M.; Polard, E.; Baldeyrou, M.; Turmel, V.; Tadié, É.; Garlantezec, R.; Tattevin, P. COVID-19 Vaccine Hesitancy among Healthcare Workers. Infect. Dis. Now 2021, 51, 484–487. [Google Scholar] [CrossRef] [PubMed]
- Cascini, F.; Pantovic, A.; Al-Ajlouni, Y.; Failla, G.; Ricciardi, W. Attitudes, Acceptance and Hesitancy among the General Population Worldwide to Receive the COVID-19 Vaccines and Their Contributing Factors: A Systematic Review. EClinicalMedicine 2021, 40, 101113. [Google Scholar] [CrossRef]
- Biswas, M.R.; Alzubaidi, M.S.; Shah, U.; Abd-Alrazaq, A.A.; Shah, Z. A Scoping Review to Find Out Worldwide COVID-19 Vaccine Hesitancy and Its Underlying Determinants. Vaccines 2021, 9, 1243. [Google Scholar] [CrossRef]
- Reno, C.; Maietti, E.; Fantini, M.P.; Savoia, E.; Manzoli, L.; Montalti, M.; Gori, D. Enhancing COVID-19 Vaccines Acceptance: Results from a Survey on Vaccine Hesitancy in Northern Italy. Vaccines 2021, 9, 378. [Google Scholar] [CrossRef]
- Schmid, P.; Rauber, D.; Betsch, C.; Lidolt, G.; Denker, M.-L. Barriers of Influenza Vaccination Intention and Behavior—A Systematic Review of Influenza Vaccine Hesitancy, 2005–2016. PLoS ONE 2017, 12, e0170550. [Google Scholar] [CrossRef]
- Bouyer, M.; Bagdassarian, S.; Chaabanne, S.; Mullet, E. Personality Correlates of Risk Perception. Risk Anal. 2001, 21, 457–466. [Google Scholar] [CrossRef]
- Dubé, E.; Gagnon, D.; MacDonald, N.; Bocquier, A.; Peretti-Watel, P.; Verger, P. Underlying Factors Impacting Vaccine Hesitancy in High Income Countries: A Review of Qualitative Studies. Expert Rev. Vaccines 2018, 17, 989–1004. [Google Scholar] [CrossRef] [PubMed]
- Ihlen, Ø. Science Communication, Strategic Communication and Rhetoric: The Case of Health Authorities, Vaccine Hesitancy, Trust and Credibility. J. Commun. Manag. 2020, 24, 163–167. [Google Scholar] [CrossRef]
- Taylor, S.; Alecu, L.S.; Taylor, S. The Psychology of Pandemics: Preparing for the Next Global Outbreak of Infectious Disease; Cambridge Scholars Publishing: Newcastle upon Tyne, UK, 2019; ISBN 1-5275-3959-8. [Google Scholar]
| Variable | N | % |
|---|---|---|
| Gender | ||
| Female | 426 | 50.8 |
| Male | 440 | 49.2 |
| Geographical area | ||
| N-W | 226 | 26.1 |
| N-E | 151 | 17.5 |
| Center | 174 | 20.1 |
| South and islands | 314 | 36.3 |
| Education | ||
| Middle school or lower | 144 | 16.6 |
| High school | 503 | 58.1 |
| University or higher | 219 | 25.3 |
| Family income | ||
| Below median (≤1800 €/month) | 475 | 54.9 |
| Above median (>1800 €/month) | 391 | 45.1 |
| Previously vaccinated for influence | ||
| Yes | 293 | 33.8 |
| No | 573 | 66.2 |
| Confirmed (by test) COVID-19 diagnosis in the past | ||
| Yes | 109 | 12.5 |
| No | 757 | 87.5 |
| Confirmed (by test) COVID-19 diagnosis for a relative in the past | ||
| Yes | 204 | 23.6 |
| No | 661 | 76.4 |
| Quarantined due to close contact with a suspect COVID-19 case | ||
| Yes | 181 | 21 |
| No | 684 | 79 |
| I am willing to wait to receive a vaccine which I think is better | ||
| Agree | 398 | 46 |
| Disagree/neutral | 467 | 54 |
| Variable | Mean (std. dev.) | Min–Max | Skewness | Kurtosis | Cronbach’s α |
|---|---|---|---|---|---|
| Age | 46 (13) | 18–70 | −0.07 | −0.93 | N/A |
| Risk perception | 3.79 (0.72) | 1–5 | −0.80 | 1.19 | 0.92 |
| Conspiracy beliefs | 66.15 (20.07) | 0–100 | −0.49 | 0.05 | 0.89 |
| Confidence | 5.00 (1.50) | 1–7 | −0.80 | 0.13 | 0.89 |
| Complacency | 3.30 (1.59) | 1–7 | 0.22 | −0.84 | 0.80 |
| Constrains | 2.85 (1.63) | 1–7 | 0.43 | −0.86 | 0.85 |
| Calculation | 5.04 (1.39) | 1–7 | −0.62 | 0.01 | 0.82 |
| Collective responsibility | 5.38 (1.39) | 1–7 | −0.56 | −0.22 | 0.76 |
| Variable | B | S.E. | Wald | p-Value | Odds Ratio * |
|---|---|---|---|---|---|
| Education | 7.478 | 0.024 | |||
| Education (high School) | 0.559 | 0.206 | 7.348 | 0.007 | 1.749 |
| Education (university) | 0.384 | 0.237 | 2.616 | 0.106 | 1.468 |
| Previously vaccinated | −0.435 | 0.158 | 7.586 | 0.006 | 0.647 |
| Risk perception (z score) | 0.390 | 0.080 | 24.064 | <0.001 | 1.477 |
| Conspiracy mentality (z score) | 0.426 | 0.078 | 29.529 | <0.001 | 1.531 |
| Calculation (z score) | 0.351 | 0.078 | 20.123 | <0.001 | 1.420 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barello, S.; Palamenghi, L.; Graffigna, G. Public Reaction towards the Potential Side Effects of a COVID-19 Vaccine: An Italian Cross-Sectional Study. Vaccines 2022, 10, 429. https://doi.org/10.3390/vaccines10030429
Barello S, Palamenghi L, Graffigna G. Public Reaction towards the Potential Side Effects of a COVID-19 Vaccine: An Italian Cross-Sectional Study. Vaccines. 2022; 10(3):429. https://doi.org/10.3390/vaccines10030429
Chicago/Turabian StyleBarello, Serena, Lorenzo Palamenghi, and Guendalina Graffigna. 2022. "Public Reaction towards the Potential Side Effects of a COVID-19 Vaccine: An Italian Cross-Sectional Study" Vaccines 10, no. 3: 429. https://doi.org/10.3390/vaccines10030429
APA StyleBarello, S., Palamenghi, L., & Graffigna, G. (2022). Public Reaction towards the Potential Side Effects of a COVID-19 Vaccine: An Italian Cross-Sectional Study. Vaccines, 10(3), 429. https://doi.org/10.3390/vaccines10030429






