Humoral and Cellular Response after mRNA Vaccination in Nursing Homes: Influence of Age and of History of COVID-19
Abstract
:1. Introduction
2. Methods
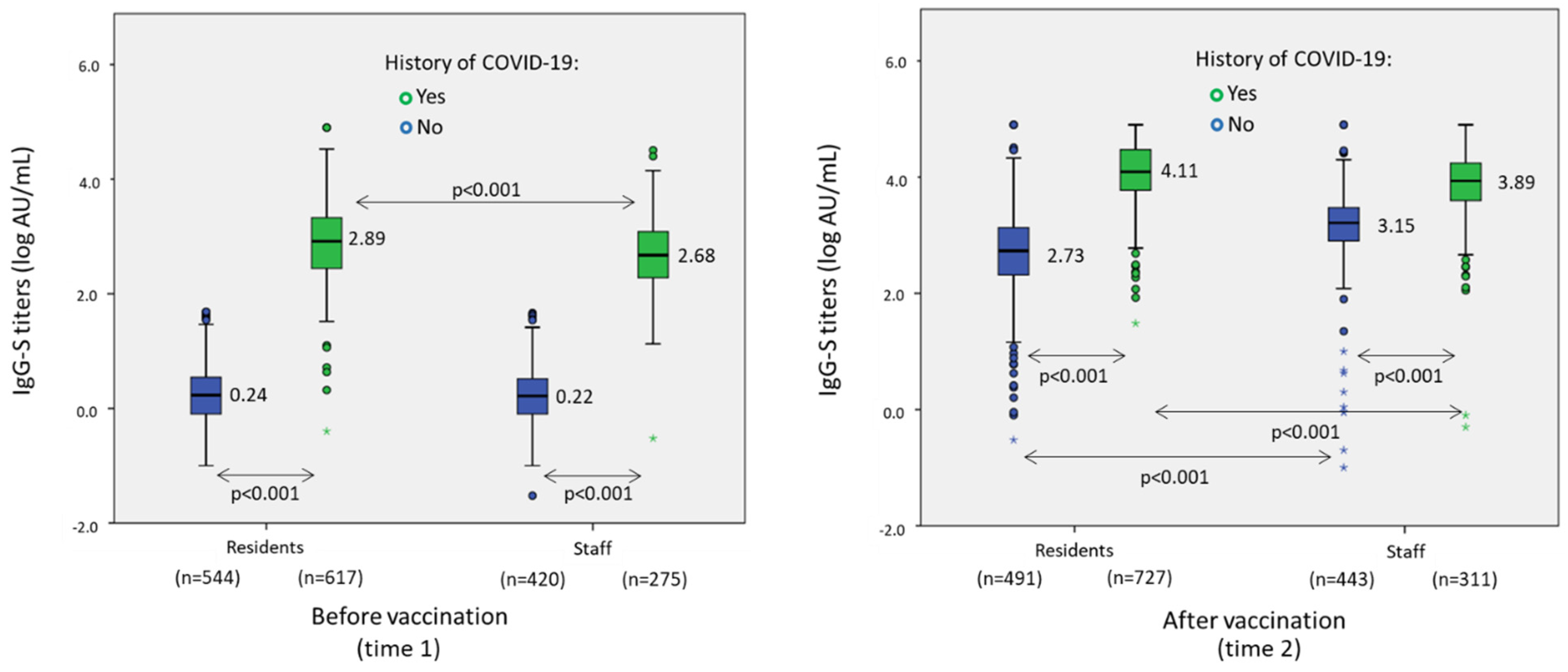
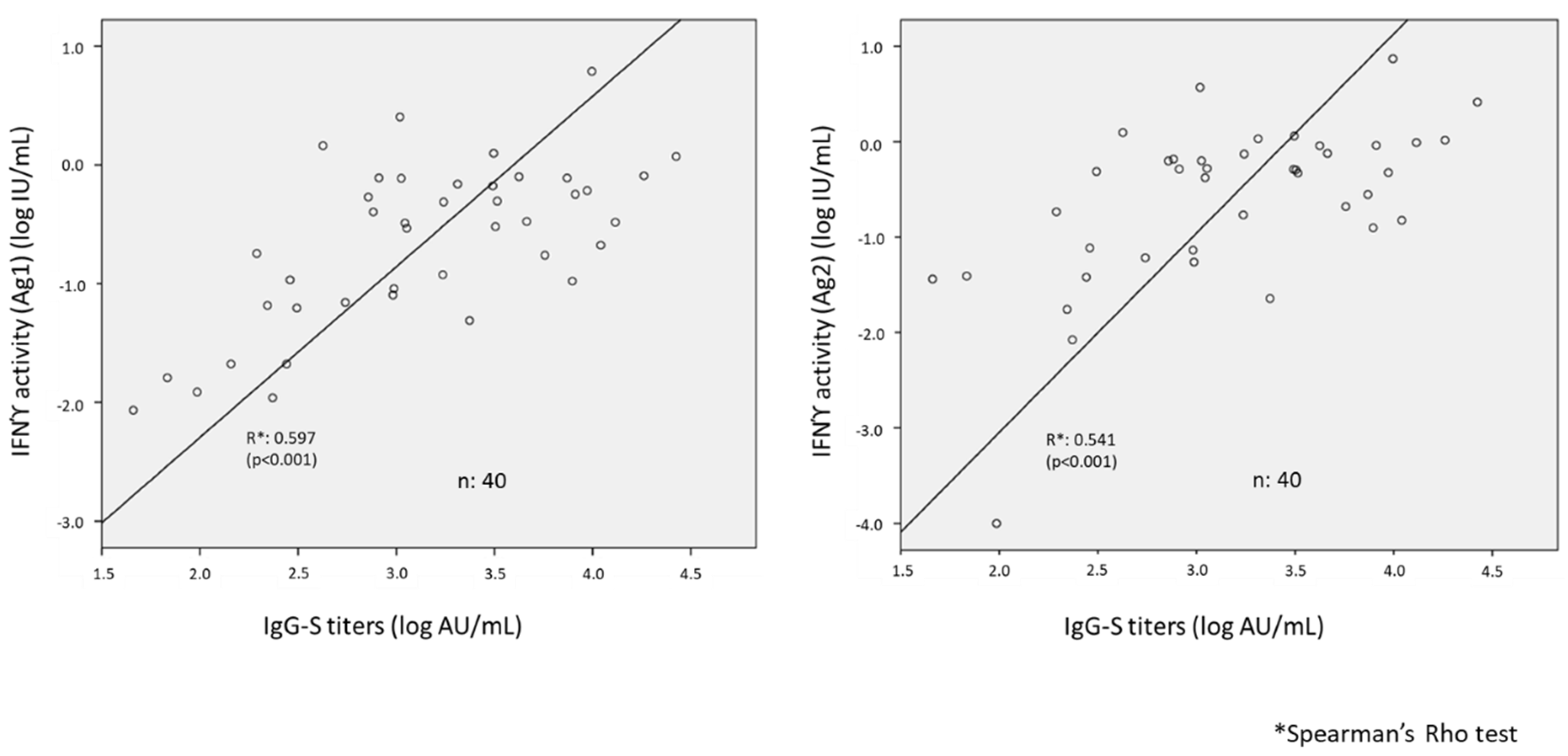
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Comas-Herrera, A.; Zalakaín, J.; Lemmon, E.; Henderson, D.; Litwin, C.; Hsu, A.T.; Schmidt, A.E.; Arling, G.; Kruse, F.; Fernández, J.L. Mortality Associated with COVID-19 in Care Homes: International Evidence. International Long-Term Care Policy Network. CPEC-LSE. 1 February 2021. Available online: https://ltccovid.org/2020/04/12/mortality-associated-with-covid-19-outbreaks-in-care-homes-early-international-evidence (accessed on 10 September 2021).
- Thompson, D.-C.; Barbu, M.-G.; Beiu, C.; Popa, L.G.; Mihai, M.M.; Berteanu, M.; Popescu, M.N. The Impact of COVID-19 pandemic on long-term care facilities worldwide: An overview on international issues. BioMed Res. Int. 2020, 2020, 8870249. [Google Scholar] [CrossRef] [PubMed]
- Cabezas, C.; Coma, E.; Mora-Fernández, N.; Li, X.; Martinez-Marcos, M.; Fina, F.; Fabregas, M.; Hermosilla, E.; Jover, A.; Contel, J.C.; et al. Associations of BNT162b2 vaccination with SARS-CoV-2 infection and hospital admission and death with covid-19 in nursing homes and healthcare workers in Catalonia: Prospective cohort study. BMJ 2021, 374, n1868. [Google Scholar] [CrossRef] [PubMed]
- Hansen, C.H.; Michlmayr, D.; Gubbels, S.M.; Molbak, K.; Ethelberg, S. Assessment of protection against reinfection with SARS-CoV-2 among 4 million PCR-tested individuals in Denmark in 2020: A population-level observational study. Lancet 2021, 397, 1204–1212. [Google Scholar] [CrossRef]
- Graham, M.; Sudre, C.; May, A.; Antonelli, M.; Murray, B.; Varsavsky, T.; Kläser, K.; Canas, L.S.; Molteni, E.; Modat, M.; et al. Changes in symptomatology, reinfection, and transmissibility associated with the SARS-CoV-2 variant B.1.1.7: An ecological study. Lancet Public Health 2021, 6, e335–e345. [Google Scholar] [CrossRef]
- Hanrath, A.; Payne, B.; Duncan, C. Prior SARS-CoV-2 infection is associated with protection against symptomatic reinfection. J. Infect. 2021, 82, 29–30. [Google Scholar] [CrossRef]
- Khoury, D.S.; Cromer, D.; Reynaldi, A.; Schlub, T.E.; Wheatley, A.K.; Juno, J.A.; Subbarao, K.; Kent, S.J.; Triccas, J.A.; Davenport, M.P. Neutralizing antibody levels are highly predictive of immune protection from symptomatic SARS-CoV-2 infection. Nat. Med. 2021, 27, 1205–1211. [Google Scholar] [CrossRef]
- Bar-On, Y.M.; Goldberg, Y.; Mandel, M.; Bodenheimer, O.; Freedman, L.; Kalkstein, N.; Mizrahi, B.; Alroy-Preis, S.; Ash, N.; Milo, R.; et al. Protection of BNT162b2 Vaccine Booster against Covid-19 in Israel. N. Engl. J. Med. 2021, 385, 1393–1400. [Google Scholar] [CrossRef]
- Canaday, D.H.; Carias, L.; Oyebanji, O.A.; Keresztesy, D.; Wilk, D.; Payne, M.; Aung, H.; Denis, K.S.; Lam, E.C.; Rowley, C.F.; et al. Reduced BNT162b2 messenger RNA vaccine response in severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2)–naive nursing home residents. Clin. Infect. Dis. 2021, 73, 2112–2115. [Google Scholar] [CrossRef]
- Candel, F.J.; Barreiro, P.; Román, J.S.; Carretero, M.d.; Sanz, J.C.; Pérez-Abeledo, M.; Ramos, B.; Viñuela-Prieto, J.M.; Canora, J.; Martínez-Peromingo, F.J.; et al. The demography and characteristics of SARS-CoV-2 seropositive residents and staff of nursing homes for older adults in the Community of Madrid: The SeroSOS study. Age Ageing 2021, 50, 1038–1047. [Google Scholar] [CrossRef]
- Jung, K.; Shin, S.; Nam, M.; Hong, Y.J.; Roh, E.Y.; Park, K.U.; Song, E.Y. Performance evaluation of three automated quantitative immunoassays and their correlation with a surrogate virus neutralization test in coronavirus disease 19 patients and pre-pandemic controls. J. Clin. Lab. Anal. 2021, 35, e23921. [Google Scholar] [CrossRef]
- DiaSorin®. LIAISON® QuantiFERON-TB Gold Plus (REF. 311010). Available online: https://www.accessdata.fda.gov/cdrh_docs/pdf18/P180047D.pdf (accessed on 12 December 2021).
- QIAGEN. QuantiFERON® SARS-CoV-2 Extended set Blood Collection Tubes Instructions for Use (Handbook). 2021. Available online: https://www.qiagen.com/us/resources/resourcedetail?id=60729925-a7b2-4cf3-8d28-52e4106af16b&lang=en (accessed on 12 December 2021).
- QIAGEN. QuantiFERON® Control Set Blood Collection Tubes Instructions for Use (Handbook). 2021. Available online: https://www.qiagen.com/us/resources/resourcedetail?id=80996d9a-d50c-40b5-940f-b62959d54ab4&lang=en (accessed on 12 December 2021).
- QIAGEN. QuantiFERON® SARSCoV-2 Starter Set Blood Collection Tubes Instructions for Use (Handbook). 2021. Available online: https://www.qiagen.com/nl/resources/resourcedetail?id=2a36a234-bfde-45f4-ae08-7c28ef95df7b&lang=en (accessed on 12 December 2021).
- Barreiro, P.; Sanz, J.C.; San Román, J.; Pérez-Abeledo, M.; Carretero, M.; Megías, G.; Viñuela-Prieto, J.M.; Ramos, B.; Canora, J.; Martínez-Peromingo, F.J.; et al. A pilot study for the evaluation of an interferon-gamma release assay (IGRA) to measure T-cell immune responses after SARS-CoV-2 infection or vaccination in a unique cloistered cohort. J. Clin. Microbiol. 2022. Preprint. [Google Scholar] [CrossRef]
- Prince, H.E.; Givens, T.S.; Lapé-Nixon, M.; Clarke, N.J.; Schwab, D.A.; Batterman, H.J.; Jones, R.S.; Meyer, W.A., III; Kapoor, H.; Rowland, C.M.; et al. Detection of SARS-CoV-2 IgG targeting nucleocapsid or spike protein by four high-throughput immunoassays authorized for emergency use. J. Clin. Microbiol. 2020, 58, e01742-20. [Google Scholar] [CrossRef] [PubMed]
- Grandjean, L.; Saso, A.; Ortiz, A.T.; Lam, T.; Hatcher, J.; Thistlethwayte, R.; Harris, M.; Best, T.; Johnson, M.; Wagstaffe, H.; et al. Long-Term persistence of spike protein antibody and predictive modeling of antibody dynamics after infection with severe acute respiratory syndrome coronavirus 2. Clin. Infect. Dis. 2021. Preprint. [Google Scholar] [CrossRef] [PubMed]
- García-Abellán, J.; Padilla, S.; Fernández-González, M.; García, J.A.; Agulló, V.; Andreo, M.; Ruiz, S.; Galiana, A.; Gutiérrez, F.; Masiá, M.; et al. Antibody response to SARS-CoV-2 is associated with long-term clinical outcome in patients with COVID-19: A longitudinal study. J. Clin. Immunol. 2021, 41, 1490–1501. [Google Scholar] [CrossRef] [PubMed]
- Jeffery-Smith, A.; Iyanger, N.; Williams, S.V.; Chow, J.Y.; Aiano, F.; Hoschler, K.; Lackenby, A.; Ellis, J.; Platt, S.; Miah, S.; et al. Antibodies to SARS-CoV-2 protect against re-infection during outbreaks in care homes, September and October 2020. Eurosurveillance 2021, 26, 2100092. [Google Scholar] [CrossRef]
- Cavanaugh, A.M.; Thoroughman, D.; Miranda, H.; Spicer, K. Suspected recurrent SARS-CoV-2 infections among residents of a skilled nursing facility during a second COVID-19 outbreak—Kentucky, July-November 2020. Morb. Mortal. Wkly. Rep. 2021, 70, 273–277. [Google Scholar] [CrossRef]
- Mateos-Nozal, J.; Montemayor, J.C.G.; Torres, S.L.; Barreiro, P.; Zamora, A.P.; Martínez, F.M.; Castelo, D.G.; Rodríguez-Domínguez, M.; Peromingo, F.J.M.; Cruz-Jentoft, A.J. SARS-CoV-2 B 1.1.7 Variant outbreak in a fully vaccinated nursing home—Madrid, June 2021. J. Am. Med. Dir. Assoc. 2021, 22, 2266–2268. [Google Scholar] [CrossRef]
- Van Praet, J.T.; Vandecasteele, S.; De Roo, A.; Vynck, M.; De Vriese, A.S.; Reynders, M. Dynamics of the cellular and humoral immune response after BNT162b2 messenger ribonucleic acid coronavirus disease 2019 (COVID-19) vaccination in COVID-19-naive nursing home residents. J. Infect. Dis. 2021, 224, 1690–1693. [Google Scholar] [CrossRef]
- Achiron, A.; Gurevich, M.; Falb, R.; Dreyer-Alster, S.; Sonis, P.; Mandel, M. SARS-CoV-2 antibody dynamics and B-cell memory response over time in COVID-19 convalescent subjects. Clin. Microbiol. Infect. 2021, 27, 1349.e1–1349.e6. [Google Scholar] [CrossRef]
- Goldberg, Y.; Mandel, M.; Bar-On, Y.M.; Bodenheimer, O.; Freedman, L.; Haas, E.J.; Milo, R.; Alroy-Preis, S.; Ash, N.; Huppert, A. Waning immunity of the BNT162b2 vaccine: A nationwide study from Israel. N. Engl. J. Med. 2021, 385, e85. [Google Scholar] [CrossRef]
- Blain, H.; Tuaillon, E.; Gamon, L.; Pisoni, A.; Miot, S.; Rolland, Y.; Picot, M.; Bousquet, J. Antibody response after one and two jabs of the BNT162b2 vaccine in nursing home residents: The CONsort-19 study. Allergy 2021, 77, 271–281. [Google Scholar] [CrossRef]
- Cevik, M.; Grubaugh, N.D.; Iwasaki, A.; Openshaw, P. COVID-19 vaccines: Keeping pace with SARS-CoV-2 variants. Cell 2021, 184, 5077–5081. [Google Scholar] [CrossRef] [PubMed]
- Jaganathan, S.; Stieber, F.; Rao, S.N.; Nikolayevskyy, V.; Manissero, D.; Allen, N.; Boyle, J.; Howard, J. Preliminary evaluation of QuantiFERON SARS-CoV-2 and QIAreach anti-SARS-CoV-2 total test in recently vaccinated individuals. Infect. Dis. Ther. 2021, 10, 2765–2776. [Google Scholar] [CrossRef] [PubMed]
- Goel, R.R.; Painter, M.M.; Apostolidis, S.A.; Mathew, D.; Meng, W.; Rosenfeld, A.M.; Lundgreen, K.A.; Reynaldi, A.; Khoury, D.S.; Pattekar, A.; et al. mRNA vaccines induce durable immune memory to SARS-CoV-2 and variants of concern. Science 2021, 374, abm0829. [Google Scholar] [CrossRef] [PubMed]
| Residents | Workers | p-Value | |
|---|---|---|---|
| Number (%) | 1218 | 755 | |
| Female sex (%) | 867 (71.2) | 644 (85.3) | |
| Mean age (SD) (years-old) | 83.7 (12.1) | 47.1 (11.4) | |
| Evidence of SARS-CoV-2 infection at time 1 (%) | |||
| 539 (44.2) + | 204 (27.1) * | <0.001 |
| 608 (50.0) + | 270 (35.9) * | <0.001 |
| 619 (50.8) | 277 (36.7) | <0.001 |
| Evidence of SARS-CoV-2 infection at time 2 (%) | |||
| 727 (59.7) | 311 (41.2) | <0.001 |
| Evolution of serology from time 1 to time 2 | |||
| 328 (26.9) | 171 (22.7) | 0.003 |
| 109 (8.9) | 35 (4.6) | <0.001 |
| 1180 (96.8) | 740 (98.1) | 0.09 |
| OR | 95% CI | p | |
|---|---|---|---|
| Age (per 10 years older) | −0.03 | −0.04 to −0.02 | <0.001 |
| History of COVID-19 (yes vs. no) | 1.14 | 1.08 to 1.20 | <0.001 |
| Female vs. male sex | −0.015 | −0.08 to 0.05 | 0.67 |
| Staff vs. residents | −0.01 | −0.10 to 0.12 | 0.87 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
San Román, J.; Candel, F.J.; Sanz, J.C.; López, P.; Menéndez-Colino, R.; Barreiro, P.; Carretero, M.d.M.; Pérez-Abeledo, M.; Viñuela-Prieto, J.M.; Ramos, B.; et al. Humoral and Cellular Response after mRNA Vaccination in Nursing Homes: Influence of Age and of History of COVID-19. Vaccines 2022, 10, 383. https://doi.org/10.3390/vaccines10030383
San Román J, Candel FJ, Sanz JC, López P, Menéndez-Colino R, Barreiro P, Carretero MdM, Pérez-Abeledo M, Viñuela-Prieto JM, Ramos B, et al. Humoral and Cellular Response after mRNA Vaccination in Nursing Homes: Influence of Age and of History of COVID-19. Vaccines. 2022; 10(3):383. https://doi.org/10.3390/vaccines10030383
Chicago/Turabian StyleSan Román, Jesús, Francisco Javier Candel, Juan Carlos Sanz, Paloma López, Rocío Menéndez-Colino, Pablo Barreiro, María del Mar Carretero, Marta Pérez-Abeledo, José Manuel Viñuela-Prieto, Belén Ramos, and et al. 2022. "Humoral and Cellular Response after mRNA Vaccination in Nursing Homes: Influence of Age and of History of COVID-19" Vaccines 10, no. 3: 383. https://doi.org/10.3390/vaccines10030383
APA StyleSan Román, J., Candel, F. J., Sanz, J. C., López, P., Menéndez-Colino, R., Barreiro, P., Carretero, M. d. M., Pérez-Abeledo, M., Viñuela-Prieto, J. M., Ramos, B., Canora, J., Barba, R., Zapatero-Gaviria, A., Martínez-Peromingo, F. J., & on behalf of the Investigators of the SeroVAC Study. (2022). Humoral and Cellular Response after mRNA Vaccination in Nursing Homes: Influence of Age and of History of COVID-19. Vaccines, 10(3), 383. https://doi.org/10.3390/vaccines10030383






