A Systematic Review of T Cell Epitopes Defined from the Proteome of Hepatitis B Virus
Abstract
1. Introduction
2. Polymorphism of HLA Alleles and Association with HBV Infection
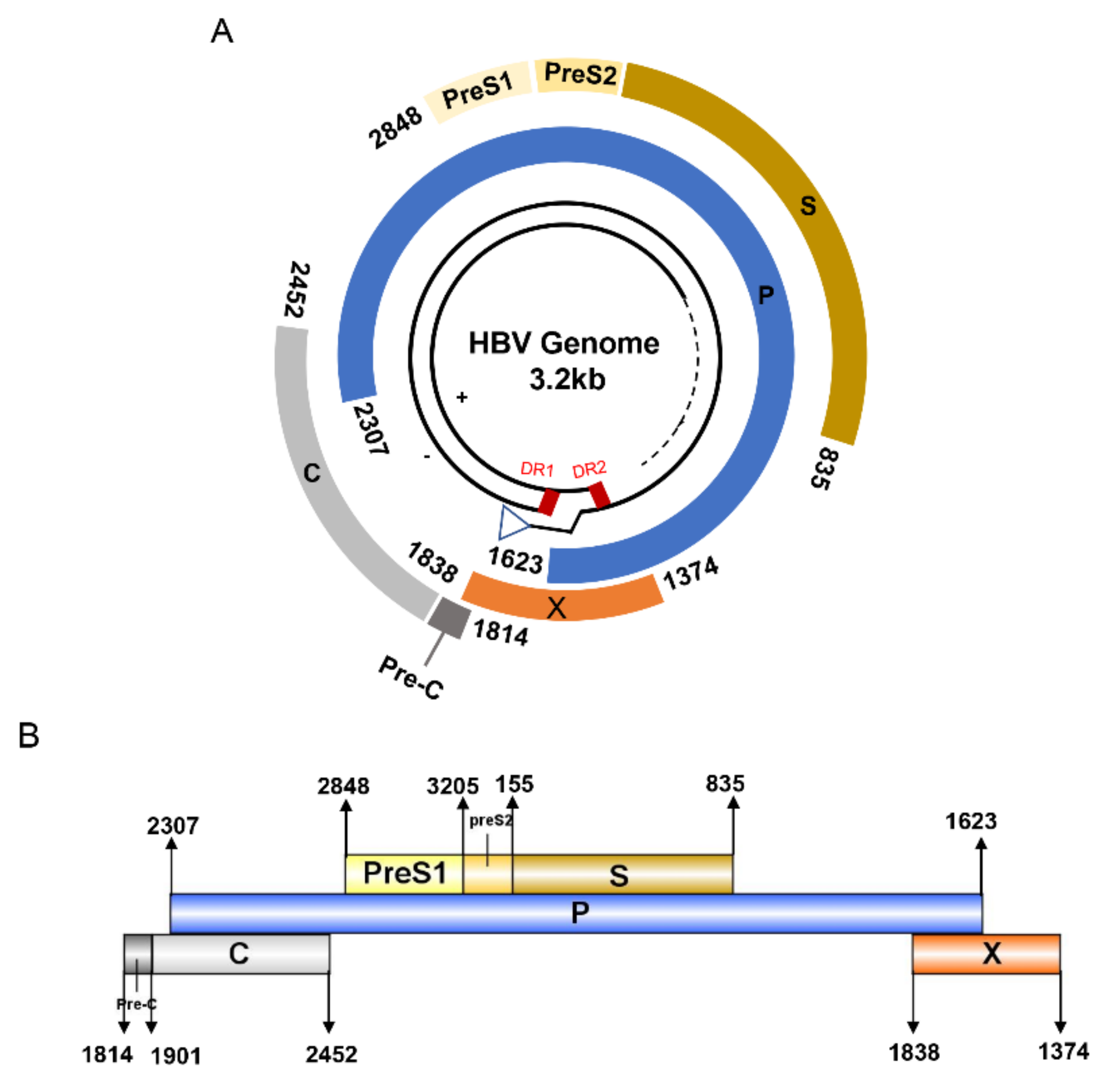
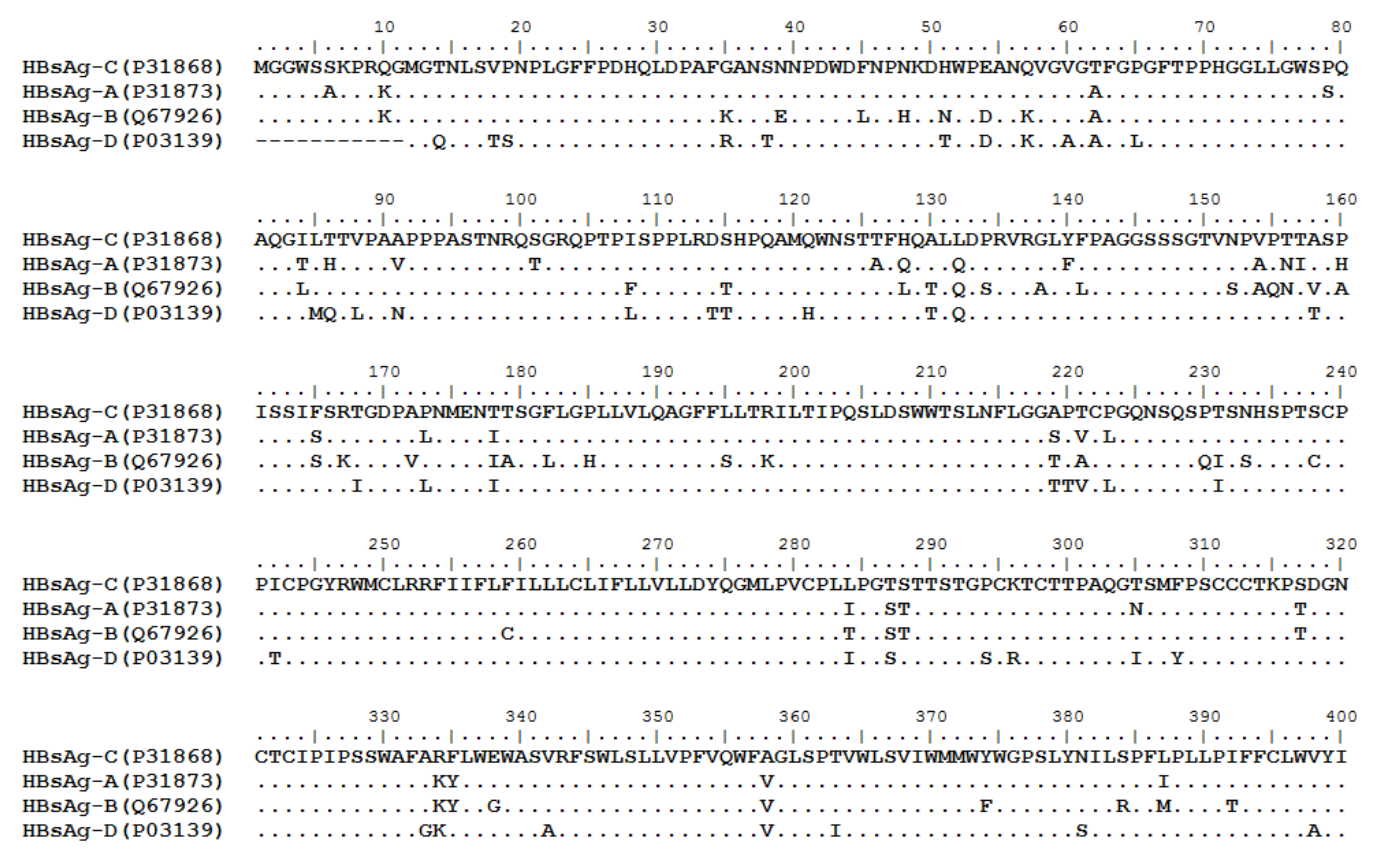
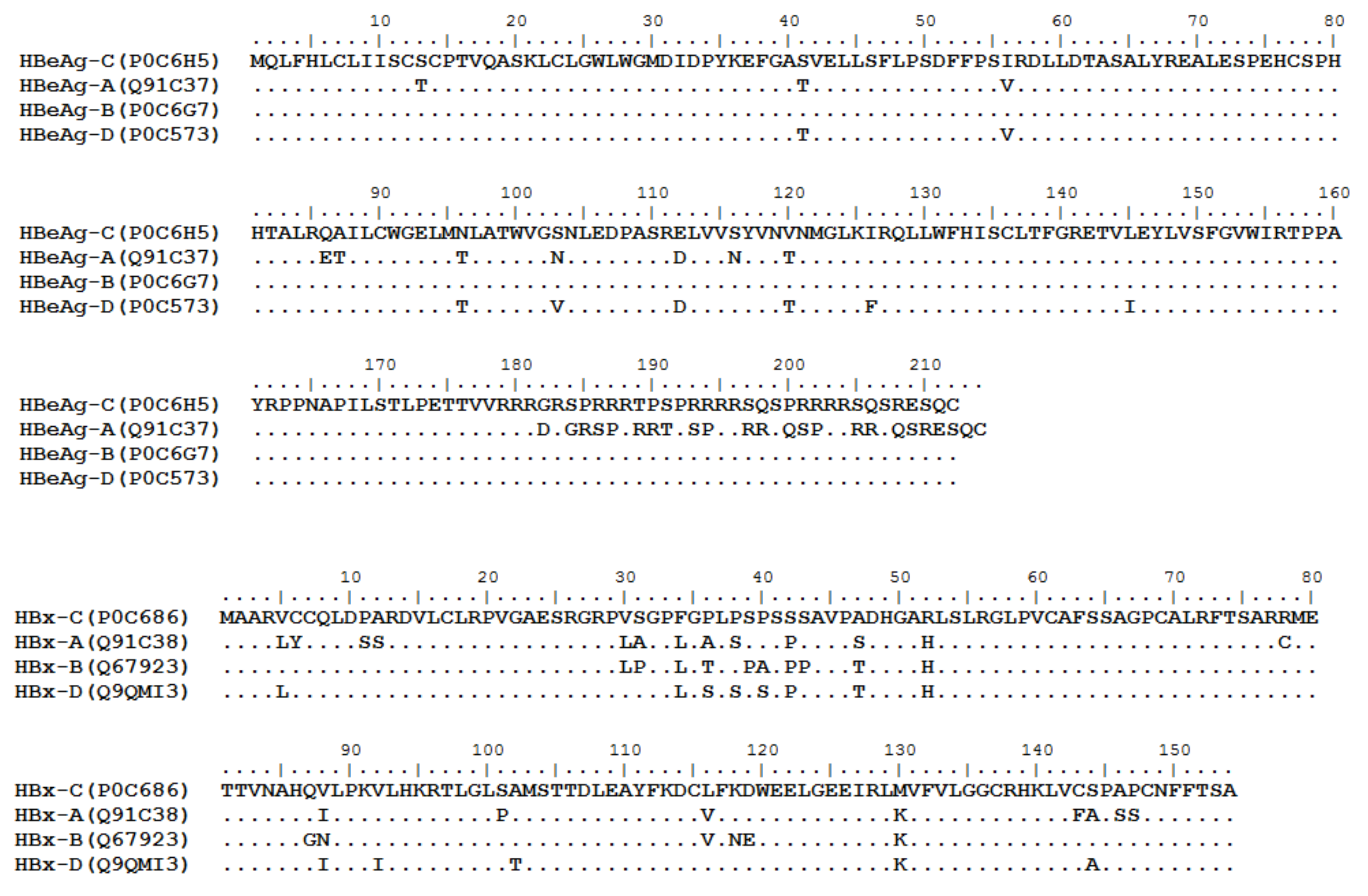
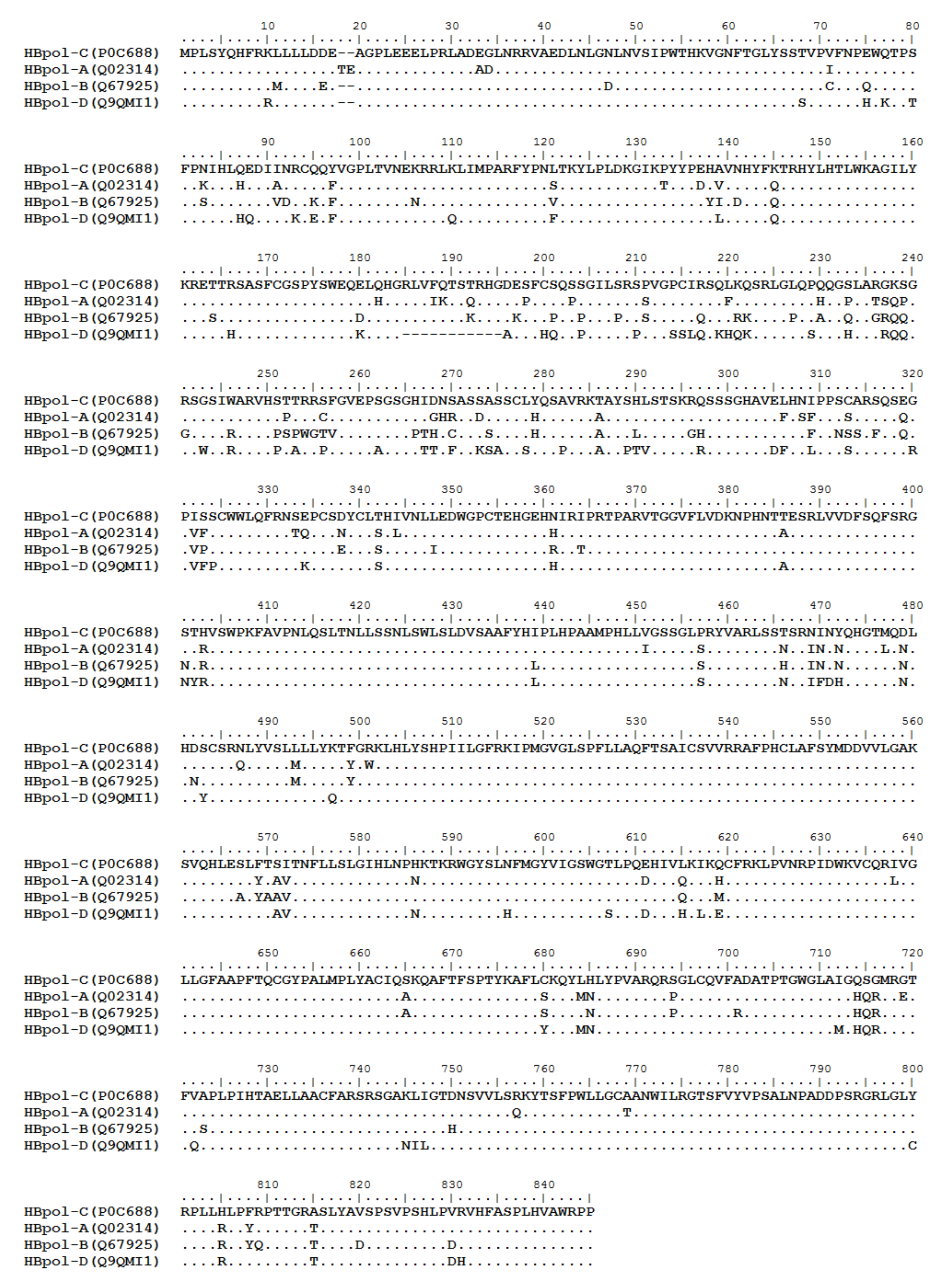
3. HBV Proteome and the Approaches Identifying T Cell Epitopes
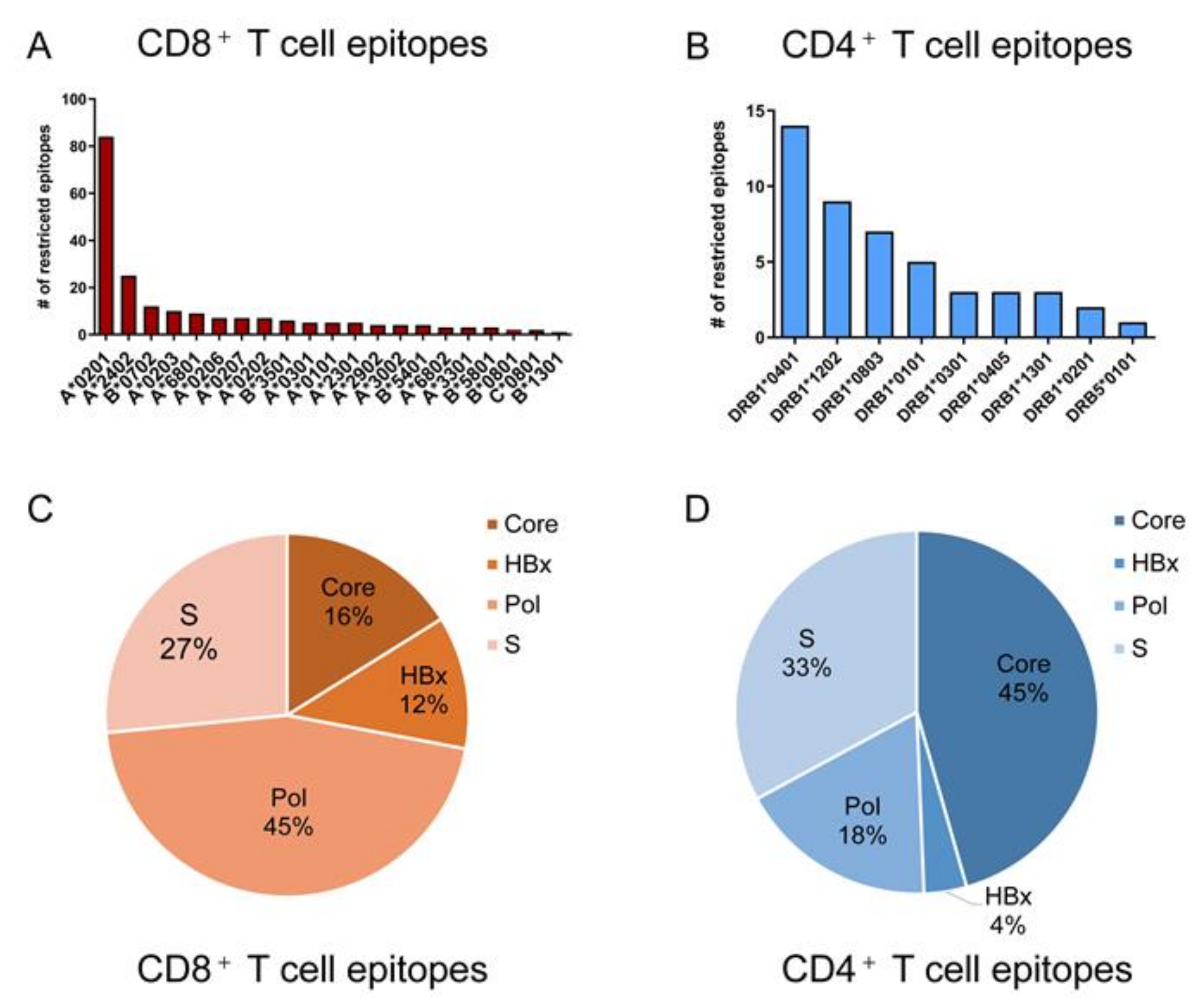
4. Defined T Cell Epitopes in HBV Proteins during the Past 33 Years
| Sequence | Protein | Position | Reference | HLA Restriction | Method to Screen Candidate Epitopes | Method to Validate the Candidate Epitopes |
|---|---|---|---|---|---|---|
| MQLFHLCLI | Core | 1–8 | [61] | A*0201 | Predicted | Binding assay; ELISpot; Cytotoxicity assay; CTL assay |
| KEFGASVEL(L) | Core | 7–15/16 | [62] | A*0206, B*4001 | Predicted | ELISpot; ICS; Binding assay |
| EFGASVELL | Core | 8–16 | [63] | A*0201, A*0207 | overlapping | ICS; ELISpot |
| FLPSDFFPS | Core | 18–26 | [64] | A*0201 | Predicted | ICS; Tetramer staining |
| FLPSDFFPSV | Core | 18–27 | [45,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79] | A*02, A*0201, A*0202, A*0203, A*0206, A*6802, A*0301, A*0207 | overlapping | Immunization of mice; Cytotoxicity assay; CTL assay; Binding assay; Tetramer staining; ELISA |
| LPSDFFPSV | Core | 19–27 | [74,80,81,82,83] | B*3501, B*51, B*5301, B*5401, B*07, B*51, A*0201 | overlapping | Binding assay; CTL assay; Cytotoxicity assay; Tetramer staining |
| FFPSIRDLL | Core | 23–31 | [84] | A*24 | Predicted | Tetramer staining |
| DLLDTASALY | Core | 39–48 | [81] | A*0101, A*2902, A*3002 | Predicted | Binding assay; Immunization of mice; ELISpot |
| DFFPSIRDL | Core | 51–59 | [85] | A*2402 | Predicted | ELISpot |
| LCWGELMNL | Core | 60–68 | [86] | A*0201 | Predicted | Stabilization assay; ELISpot assay |
| ELMNLATWV | Core | 64–72 | [87] | A*02 | Predicted | Binding assay; ELISpot; Cytotoxicity assay |
| SYVNMNMGL | Core | 87–95 | [88] | A*2402 | Predicted | Binding assay; CTL assay |
| SYVNTNMGL | Core | 87–95 | [89] | A*02 | Predicted | Tetramer staining |
| YVNVNMGLK | Core | 88–96 | [63] | A*1101 | overlapping | ICS; ELISpot |
| MGLKFRQL | Core | 93–100 | [90] | A*0201 | Predicted | Immunization of mice; FACS |
| LLWFHISCL | Core | 101–108 | [43] | A*0201 | Predicted | Proliferation assay; ICS; Cytotoxicity assay |
| LWFHISCLTF | Core | 101–110 | [85] | A*2402, A*2301 | Predicted | ELISpot |
| HISCLTFGR | Core | 104–112 | [91,92] | A*33 | Predicted | Cytotoxicity assay; ICS; Tetramer staining |
| CLTFGRETV | Core | 107–115 | [93] | A*02 | Predicted | Tetramer staining |
| EYLVSFGVW | Core | 117–125 | [81,84,88] | A*2402, A*2407, A*2301 | Predicted | Stabilization assay; CTL assay; Cytotoxicity assay; Tetramer staining; Binding assay; Immunization of mice; ELISpot |
| YLVSFGVWI | Core | 118–126 | [43] | A*0201 | Predicted | Proliferation assay; ICS; Cytotoxicity assay |
| LVSFGVWIR | Core | 119–127 | [91] | A*33 | Predicted | Stabilization assay; ELISpot; Cytotoxicity assay; Immunization of mice |
| GLKILQLL | Core | 123–130 | [82] | B*08 | overlapping | ICS; Tetramer staining |
| AYRPPNAPI | Core | 131–139 | [94] | A*0201 | Predicted | ELISpot; Cytotoxicity assay |
| LTFGRETVLEN | Core | 137–147 | [95] | A*0101, A*02, A*2902, A*3002 | Predicted | ELISpot |
| ILSTLPETTV | Core | 139–148 | [75] | A*02 | Predicted | CTL assay |
| STLPETTVVR | Core | 141–150 | [74,96] | A*11, A*6801, A*02 | overlapping | Binding assay; CTL assay; Cytotoxicity assay; ELISpot |
| STLPETTVVRR | Core | 141–151 | [17,76,81,92,97] | A*31, A*68, A*02, A*0201, A*6801, A*03, A*11, A*3101, A*0201 | overlapping | Cytotoxicity assay; Immunization of mice; CTL assay; Binding assay; ELISpot |
| TLPETTVVRR | Core | 142–151 | [63] | A*1101 | overlapping | ICS; ELISpot |
| GVWIRTPPA | Core | 152–160 | [98] | A*0201 | Predicted | ELISpot |
| STLPETAVVRR | Core | 170–180 | [9] | A*1101 | Predicted | Proliferation assay; Tetramer staining |
| RTQSPRRR | Core | 196–203 | [9] | A*1101 | Predicted | Proliferation assay; Tetramer staining |
| RTQSPRRRR | Core | 196–204 | [9] | A*1101 | Predicted | Proliferation assay; Tetramer staining |
| RSQSPRRRRSK | Core | 196–206 | [9] | A*1101 | Predicted | Proliferation assay; Tetramer staining |
| RLCCQLDPA | HBx | 4–12 | [99] | A*0201 | Predicted | Binding assay; ELISpot; Cytotoxicity assay |
| AYFKDCVFKDW | HBx | 6–16 | [45] | A*2402 | Predicted | ELISA |
| QLDPARDVL | HBx | 8–16 | [45,65,73,99,100,101] | A*0201 | Predicted | ELISpot |
| VLCLRPVGA | HBx | 15–23 | [45,99,102] | A*0201 | Predicted | ELISpot |
| RGRPVSGPF | HBx | 26–34 | [85] | A*2402 | Predicted | ELISpot |
| PVSGPFGPL | HBx | 29–37 | [100] | A*0201 | Predicted | Immunization of mice; CTL assay; Cytotoxicity assay |
| AVPADHGAHL | HBx | 44–53 | [100] | A*0201 | Predicted | Immunization of mice; CTL assay; Cytotoxicity assay |
| HLSLRGLPV | HBx | 52–60 | [65,99,100,101,102,103] | A*0201, A*02 | Predicted | Cytotoxicity assay; Immunization of mice; CTL assay; Binding assay; ELISpot |
| LPVCAFSSA | HBx | 58–66 | [45] | B*0702 | Predicted | ELISA |
| AFSSAGPCALRF | HBx | 62–73 | [45] | A*2402 | Predicted | ELISA |
| ALRFTSARR | HBx | 70–78 | [45] | A*0301 | Predicted | ELISA |
| ALRFTSARRM | HBx | 70–79 | [100] | A*0201 | Predicted | Immunization of mice; CTL assay; Cytotoxicity assay |
| NAHQILPKV | HBx | 84–92 | [99] | A*0201 | Predicted | Binding assay; ELISpot; Cytotoxicity assay |
| (K)VLHKRTLGL | HBx | 91/92–100 | [65,100,102] | A*0201 | Predicted | Cytotoxicity assay; Binding assay; ELISpot; Tetramer staining |
| VLHKRTLGL | HBx | 92–100 | [99,101,104] | A*0201, A*02 | Predicted | Binding assay; ELISpot; Cytotoxicity assay; Proliferation assay; ELISpot; ICS |
| TLGLAAMST | HBx | 97–105 | [100] | A*0201 | Predicted | Binding assay; ELISpot; Cytotoxicity assay |
| GLSAMSTTDL | HBx | 99–108 | [99,100,104] | A*0201, A*02 | Predicted | Binding assay; ELISpot; Cytotoxicity assay |
| AMSTTDLEA | HBx | 102–110 | [99] | A*0201 | Predicted | Binding assay; ELISpot; Cytotoxicity assay |
| STTDLEAYFK | HBx | 104–113 | [45] | A*1101 | Predicted | ELISA |
| DLEAYFKDCL | HBx | 107–116 | [100] | A*0201 | Predicted | Immunization of mice; CTL assay; Cytotoxicity assay |
| CLFKDWEEL | HBx | 115–123 | [99,100,102] | A*0201 | Predicted | Immunization of mice; CTL assay; Cytotoxicity assay; Binding assay; ELISpot |
| ELGEEIRLKV | HBx | 122–131 | [100] | A*0201 | Predicted | Immunization of mice; CTL assay; Cytotoxicity assay |
| EIRLKVFVL | HBx | 126–134 | [100] | A*0201 | Predicted | Immunization of mice; CTL assay; Cytotoxicity assay |
| VLGGCRHKL | HBx | 133–141 | [99,101] | A*0201, A*02 | Predicted | Binding assay; ELISpot; Cytotoxicity assay; ELISpot |
| VLGGCRHKL(V) | HBx | 133–141/142 | [98] | A*0201 | Predicted | Immunization of mice; CTL assay; Cytotoxicity assay |
| LLDDEAGPL | Pol | 13–21 | [105,106] | A*0201 | Predicted | Binding assay; Immunization of mice; CTL assay; Cytotoxicity assay |
| PLEEELPRL | Pol | 20–28 | [105,106] | A*0201 | Predicted | Binding assay; Immunization of mice; CTL assay; Cytotoxicity assay |
| DLNLGNLN | Pol | 40–48 | [106] | A*0201 | Predicted | Binding assay; Immunization of mice; CTL assay; Cytotoxicity assay |
| NLGNLNVSI | Pol | 42–50 | [106] | A*0201 | Predicted | Binding assay; Immunization of mice; CTL assay; Cytotoxicity assay |
| NVSIPWTHK | Pol | 47–55 | [9,74,81] | A*03, A*11, A*6801, A*0301, A*1101 | Predicted | Stabilization assay; ELISpot; Cytotoxicity assay; Immunization of mice; Proliferation assay; Tetramer staining; Binding assay |
| KVGNFTGLY | Pol | 55–63 | [45,74] | A*0301, A*03, A*11 | Predicted | Binding assay; CTL assay; Cytotoxicity assay; ELISA |
| GLYSSTVPV | Pol | 61–69 | [73,105,106] | A*0201 | Predicted | Binding assay; Immunization of mice; CTL assay; Cytotoxicity assay; Tetramer staining |
| LYSSTVPVF | Pol | 62–70 | [79] | A*24 | Predicted | ELISpot |
| STVPCFNPK | Pol | 65–73 | [9] | A*1101 | Predicted | Proliferation assay; Tetramer staining |
| TVPCFNPK | Pol | 66–73 | [9] | A*1101 | Predicted | Proliferation assay; Tetramer staining |
| PSFPHIHLK | Pol | 77–85 | [9] | A*1101 | Predicted | Proliferation assay; Tetramer staining |
| QYVGPLTVN | Pol | 94–102 | [85] | A*2402 | Predicted | ELISpot |
| YLHTLWKAGI | Pol | 147–156 | [65] | A*02 | Predicted | ELISpot assay; Tetramer staining |
| (H)TLWKAGILYK | Pol | 149/150–159 | [81] | A*03 | Predicted | Binding assay; Immunization of mice; ELISpot |
| HTLWKAGILYK | Pol | 149–159 | [74,76,98] | A*03, A*11, A*3101, A*3301, A*6801, A*02, A*11 | Predicted | Immunization of mice; Cytotoxicity assay; Binding assay; CTL assay |
| TLWKAGILY(K) | Pol | 150–158/159 | [74] | A*03, A*11 | Predicted | Binding assay; CTL assay; Cytotoxicity assay |
| RSASFCGSPY | Pol | 164–173 | [45] | A*1101 | Predicted | ELISA |
| ASFCGSPYSW | Pol | 166–175 | [45,62,63] | A*2402, B*5801 | overlapping | ELISA; ELISpot; ICS |
| SFCGSPYSW | Pol | 167–175 | [45] | A*2402 | Predicted | ELISA |
| ASFCGSPY | Pol | 166–173 | [81,95,107] | A*0101, A*2902, A*3002 | overlapping | Binding assay; Immunization of mice; ELISpot; Tetramer staining |
| SPYSWEQEL | Pol | 171–179 | [17] | A*0201, B*3501 | Predicted | Tetramer staining |
| QSSGILSR | Pol | 200–207 | [9] | A*1101 | Predicted | Proliferation assay; Tetramer staining |
| GILPRSSVGPR | Pol | 205–215 | [9] | A*1101 | Predicted | Proliferation assay; Tetramer staining |
| CLHQSAVRK | Pol | 274–282 | [45] | A*0301, A*1101 | Predicted | ELISA |
| KTAYSHLSTSK | Pol | 283–293 | [9] | A*1101 | Predicted | Proliferation assay; Tetramer staining |
| SSARSQSER | Pol | 310–318 | [9] | A*1101 | Predicted | Proliferation assay; Tetramer staining |
| CLSLIVNLL | Pol | 338–346 | [65] | A*02 | Predicted | ELISpot assay; Tetramer staining |
| TPARVTGGV | Pol | 354–362 | [45] | B*0702 | Predicted | ELISA |
| TPARVTGGVF | Pol | 354–363 | [45] | B*0702 | Predicted | ELISA |
| RVTGGVFLV | Pol | 357–365 | [45] | A*0201 | Predicted | ELISA |
| VTGGVFLVDK | Pol | 358–367 | [45] | A*1101, A*03 | Predicted | ELISA |
| RIPRTPSRV | Pol | 361–369 | [65] | A*02 | Predicted | ELISpot assay; Tetramer staining |
| TPARVTGGVF | Pol | 365–374 | [74,76,108] | B*0702, B*3501, A*03, B*07, A*02, B*51 | Predicted | Immunization of mice; Cytotoxicity assay; Binding assay; CTL assay |
| RVTGGVFLVDK | Pol | 368–378 | [74] | A*11 | Predicted | Binding assay; CTL assay; Cytotoxicity assay |
| VTGGVFLVDK | Pol | 369–378 | [74] | A*03, A*11 | Predicted | Binding assay; CTL assay; Cytotoxicity assay |
| FLVDKNPHNT | Pol | 374–383 | [62] | A*0203 | Predicted | ELISpot; ICS; Binding assay |
| LVVDFLHQFSR | Pol | 377–386 | [9] | A*1101, A*3301, A*6801 | Predicted | Proliferation assay; Tetramer staining; Binding assay; Immunization of mice; ELISpot; CTL assay; Cytotoxicity assay |
| SRLVVDFSQF | Pol | 386–395 | [63] | B*1301 | overlapping | ICS; ELISpot |
| VVDFSQFSR | Pol | 389–397 | [74,91] | A*11, A*6801, A*33 | Predicted | Stabilization assay; ELISpot; Cytotoxicity assay; Binding assay; Immunization of mice; CTL assay |
| SWPKFAVPNL | Pol | 392–401 | [45] | A*2402 | Predicted | ELISA |
| WPKFAVPNL | Pol | 393–401 | [45] | B*0702 | Predicted | ELISA |
| FAVPNLQSL | Pol | 396–404 | [45] | A*0201 | Predicted | ELISA |
| NLQSLTNLL | Pol | 411–419 | [105,106] | A*0201 | Predicted | Cytotoxicity assay; Immunization of mice; Binding assay; CTL assay |
| LLSSNLSWL | Pol | 418–426 | [65,105,106] | A*0201 | Predicted | Cytotoxicity assay; Immunization of mice; Binding assay; CTL assay; ELISpot; Tetramer staining |
| NLSWLSLDV | Pol | 422–430 | [101,105,106] | A*0201, A*02 | Predicted | Cytotoxicity assay; Immunization of mice; Binding assay; CTL assay; ELISpot |
| LSLDVSAAFY | Pol | 426–435 | [81] | A*0101, A*2902, A*3002 | Predicted | Binding assay; Immunization of mice; ELISpot |
| HPAAMPHLL | Pol | 440–448 | [74] | B*0702 | Predicted | Binding assay; CTL assay; Cytotoxicity assay |
| HLLVGSSGL | Pol | 446–454 | [105,106] | A*0201 | Predicted | Cytotoxicity assay; Immunization of mice; Binding assay; CTL assay |
| GLPRYVARL | Pol | 453–461 | [65,71,73,74,81,92,93,100,101,106,109,110,111] | A*0201, A*0202, A*0203, A*02, A*0207 | Predicted | Cytotoxicity assay; Immunization of mice; Binding assay; CTL assay; ELISpot; Tetramer staining |
| RIINNQHR | Pol | 466–473 | [9] | A*1101 | Predicted | Proliferation assay; Tetramer staining |
| RNLYVSLLL | Pol | 484–492 | [85] | A*2402 | Predicted | ELISpot |
| NLYVSLLLL | Pol | 485–493 | [65,106] | A*0201, A*02 | Predicted | Cytotoxicity assay; Immunization of mice; Binding assay; CTL assay; ELISpot; Tetramer staining |
| KLHLYSHPI | Pol | 500–508 | [45,62,93,101,106] | A*0201, A*02, A*0203, B*0801 | Predicted | Cytotoxicity assay; Immunization of mice; Binding assay; CTL assay; ELISpot; Tetramer staining; ELISA |
| HLYSHPIIL | Pol | 502–510 | [65,105,112,113,114] | A*0201, A*02, A*0203 | overlapping | Cytotoxicity assay; Immunization of mice; Binding assay; ELISpot; Tetramer staining |
| IPMGVGLSP | Pol | 504–512 | [45] | B*0702 | Predicted | ELISA |
| ILGFRKIPM | Pol | 509–517 | [45] | B*0801 | Predicted | ELISA |
| FLLAQFTSAI | Pol | 524–533 | [65,101] | A*0201, A*02 | Predicted | ELISpot; Tetramer staining |
| LLAQFTSAI | Pol | 525–533 | [65,101,106] | A*0201, A*02 | Predicted | Cytotoxicity assay; Immunization of mice; Binding assay; ELISpot; Tetramer staining |
| SAICSVVRR | Pol | 531–539 | [74] | A*11, A*3301, A*6801 | Predicted | Binding assay; CTL assay; Cytotoxicity assay |
| SVVRRAFPH | Pol | 535–542 | [9] | A*1101 | Predicted | Proliferation assay; Tetramer staining |
| FFPHCLAFSYM | Pol | 539–550 | [81] | B*07 | Predicted | Binding assay; Immunization of mice; ELISpot |
| FPHCLAFSYM | Pol | 540–550 | [74] | B*0702, B*3501, B*51, B*5301, B*5401 | Predicted | Binding assay; CTL assay; Cytotoxicity assay |
| YMDDVVLG | Pol | 549–556 | [81] | A*0201, A*0202, A*0203, A*0206, A*6802 | Predicted | Binding assay; Immunization of mice; ELISpot |
| YMDDVVLGA | Pol | 549–557 | [45,71,72,99,101,114,115,116] | A*0201, A*02, A*0101 | overlapping | Cytotoxicity assay; Immunization of mice; Binding assay; ELISpot; CTL assay; ELISA |
| YMDDVVLGAK | Pol | 549–558 | [74] | A*03 | Predicted | Binding assay; CTL assay; Cytotoxicity assay |
| FLLSLGIHL | Pol | 573–581 | [71,73,74,81,93,106,108,110,116,117,118,119,120] | A*02, A*0201, A*0206, A*0202 | Predicted | Cytotoxicity assay; Immunization of mice; Binding assay; ELISpot; CTL assay; Tetramer staining |
| SLNFMGYVI | Pol | 592–600 | [106] | A*0201 | Predicted | Binding assay; Immunization of mice; CTL assay; Cytotoxicity assay |
| PVNRPIDWK | Pol | 612–620 | [9] | A*1101 | Predicted | Proliferation assay; Tetramer staining |
| PVNRPIDWK | Pol | 623–631 | [74] | A*03, A*11 | Predicted | Binding assay; CTL assay; Cytotoxicity assay |
| CGYPALMPLY | Pol | 638–647 | [45] | A*2402 | Predicted | ELISA |
| GYPALMPLY | Pol | 639–647 | [45] | A*2402 | Predicted | ELISA |
| YPALMPLYA | Pol | 651–659 | [74] | B*0702, B*3501, B*51, B*5401 | Predicted | Binding assay; CTL assay; Cytotoxicity assay |
| YPALMPLSA | Pol | 651–659 | [62] | B*5401 | Predicted | ELISpot; ICS; Binding assay |
| ALMPLYACI | Pol | 653–661 | [71,74,93,106] | A*0201, A*0202, A*0203, A*0204, A*0206, A*02 | Predicted | Cytotoxicity assay; Immunization of mice; Binding assay; ELISpot; CTL assay; Tetramer staining |
| QAFTFSPTYK | Pol | 665–674 | [74,113] | A*03, A*11, A*6801 | Predicted | Cytotoxicity assay; Binding assay; CTL assay |
| VFADATPTGW | Pol | 686–695 | [45] | A*2402 | Predicted | ELISA |
| GLCQVFADA | Pol | 692–700 | [45] | A*0201 | Predicted | ELISA |
| LPIHTAELL | Pol | 712–720 | [45] | B*0702 | Predicted | ELISA |
| PLPIHTAEL | Pol | 722–730 | [106] | A*0201 | Predicted | Binding assay; Immunization of mice; CTL assay; Cytotoxicity assay |
| IIGTDNSVV | Pol | 744–752 | [65] | A*0201 | Predicted | ELISpot assay; Tetramer staining |
| RKYTSFPWLL | Pol | 744–753 | [45] | A*2402 | Predicted | ELISA |
| KYTSFPWLLG | Pol | 745–754 | [45] | A*2402 | Predicted | ELISA |
| GTDNSVVLSR | Pol | 746–755 | [74] | A*11 | Predicted | Binding assay; CTL assay; Cytotoxicity assay |
| KYTSFPWLL | Pol | 756–764 | [63,81,84,88,93] | A*24, A*2301, A*2402 | overlapping | Cytotoxicity assay; Immunization of mice; Binding assay; ELISpot; CTL assay; Tetramer staining; ICS; ELISA |
| LLGCAANWI | Pol | 763–771 | [65,106] | A*0201 | Predicted | Cytotoxicity assay; Immunization of mice; Binding assay; ELISpot; CTL assay; Tetramer staining |
| WILRGTSFV | Pol | 770–778 | [65,105] | A*0201, A*02 | Predicted | Immunization of mice; Binding assay; ELISpot; Tetramer staining |
| ILRGTSFVYV | Pol | 771–780 | [65,71] | A*0201, A*02 | Predicted | Cytotoxicity assay; ELISpot; Tetramer staining |
| DPSRGRLGL | Pol | 789–797 | [74] | B*0702 | Predicted | Binding assay; CTL assay; Cytotoxicity assay |
| RLGLSRPLL | Pol | 794–802 | [106] | A*0201 | Predicted | Binding assay; Immunization of mice; CTL assay; Cytotoxicity assay |
| GLSRPLLRL | Pol | 796–804 | [65] | A*02 | Predicted | ELISpot assay; Tetramer staining |
| LVYRPTTGR | Pol | 804–812 | [9] | A*1101 | Predicted | Proliferation assay; Tetramer staining |
| SLYADSPSV | Pol | 814–822 | [65,71,73,90,93,106,114,116] | A*0201, A*02 | Predicted | Cytotoxicity assay; Immunization of mice; Binding assay; ELISpot; CTL assay; Tetramer staining; FACS |
| FLLTRILTI | S | 20–28 | [66,67,68,77,100,121] | A*0201 | Predicted | ICS; Tetramer staining; Cytotoxicity assay; Degranulation assay |
| PLGFFPDH | S | 21–28 | [122] | A*11 | Predicted | ELISpot |
| NLLGWSPQA | S | 73–81 | [63] | A*0201, A*0207 | overlapping | ICS; ELISpot |
| LTTVPAASLLA | S | 85–95 | [95] | A*02 | Predicted | ELISpot |
| TTSTGPCK | S | 115–122 | [9] | A*1101 | Predicted | Proliferation assay; Tetramer staining |
| LLDPRVRGL | S | 131–139 | [75] | A*02 | Predicted | CTL assay |
| AILSKTGDPV | S | 160–169 | [116] | A*02 | Predicted | Tetramer staining |
| FLGPLLVLQA | S | 182–190 | [62,63,107] | C*0801 | overlapping | Cytotoxicity assay; Binding assay; ELISpot; Tetramer staining; |
| VLQAGFFL | S | 188–195 | [62] | C*0801 | Predicted | ELISpot; ICS; Binding assay |
| VLQAGFFLL | S | 188–196 | [65,73,101,116,123] | A*0201, A*02 | Predicted | Cytotoxicity assay; Immunization of mice; Binding assay; ELISpot; CTL assay; Tetramer staining |
| SWWTSLNFL | S | 192–200 | [85] | A*2402 | Predicted | ELISpot |
| FLLTRILTI | S | 194–202 | [54,74,76,81,90,93,94,101,108,111,114,116,119,120,123,124,125,126,127,128] | A*0201, A*0202, A*0203, A*0206, A*02 | overlapping | Cytotoxicity assay; Immunization of mice; Binding assay; ELISpot; CTL assay; Tetramer staining; ICS; FACS |
| IPQSLDSWWTSL | S | 202–213 | [129,130] | A*0201, A*02 | Predicted | Cytotoxicity assay; Immunization of mice; Binding assay; ELISpot |
| SILSPFLPLL | S | 207–216 | [131] | A*0201 | Predicted | Binding assay; ELISpot |
| NILSPFMPLL | S | 207–216 | [131] | A*0201 | Predicted | Binding assay; ELISpot |
| ILSPFMPLL | S | 208–216 | [131] | A*0201 | Predicted | Binding assay; ELISpot |
| TLSPFLPLL | S | 208–216 | [131] | A*0201 | Predicted | Binding assay; ELISpot |
| SWWTSLNFL | S | 208–216 | [84] | A*24 | Predicted | Tetramer staining |
| FLGGTPVCL | S | 215–223 | [95,116,123,125] | A*0201A*02, A*24 | Predicted | Cytotoxicity assay; Immunization of mice; Binding assay; ELISpot; CTL assay; Tetramer staining |
| SWLSLLVPF | S | 226–234 | [85] | A*2402 | Predicted | ELISpot |
| RWMCLRRFII | S | 236–245 | [85] | A*2402 | Predicted | ELISpot |
| CPGYRWMCL | S | 243–251 | [108] | B*07 | Predicted | Cytotoxicity assay |
| GYRWMCLRR | S | 245–253 | [91] | A*33 | Predicted | Stabilization assay; ELISpot; Cytotoxicity assay; Immunization of mice |
| RWMCLRRFII | S | 247–256 | [81] | A*2301, A*2402 | Predicted | Binding assay; Immunization of mice; ELISpot |
| ILLLCLIFL | S | 260–268 | [73,125] | A*0201 | Predicted | Cytotoxicity assay; Immunization of mice |
| LLLCLIFLL | S | 261–268 | [72] | A*02 | Predicted | Cytotoxicity assay |
| LLCLIFLLV | S | 262–269 | [65,115,123] | A*0201, A*02 | Predicted | Stabilization assay; ELISpot; Cytotoxicity assay; Tetramer staining; Immunization of mice |
| LCLIFLLVL | S | 263–271 | [85] | A*2402 | Predicted | ELISpot |
| (L)VLLDYQGML | S | 269/70–278 | [75] | A*0201 | Predicted | CTL assay |
| LLDYQGMLP | S | 271–279 | [123] | A*0201 | Predicted | Immunization of transgenic mice; Cytotoxicity assay; ELISpot; Binding assay |
| LLDYQGMLPV | S | 271–280 | [72,101,116,125] | A*02 | Predicted | ELISpot; Cytotoxicity assay; Binding assay; Tetramer staining |
| TSMFPSCCCTK | S | 305–315 | [9] | A*1101 | Predicted | Proliferation assay; Tetramer staining |
| IPIPSSWAF | S | 324–332 | [74,76,81,108] | B*0702, B*3501, B*51, B*5301, A*03, B*07, A*02, B*5101 | Predicted | ELISpot; Cytotoxicity assay; Immunization of mice; Binding assay; CTL assay |
| YLWEWASVR | S | 335–343 | [91] | A*33 | Predicted | Stabilization assay; ELISpot; Cytotoxicity assay; Immunization of mice |
| RFSWLSLLVPF | S | 343–353 | [81] | A*2301, A*2402 | Predicted | Binding assay; Immunization of mice; ELISpot |
| SWLSLLVPF | S | 345–353 | [84] | A*24 | Predicted | Tetramer staining |
| WLSLLVPFV | S | 346–354 | [71,72,73,74,75,76,99,105,108,117,118,120,123,132,133] | A*02, A*0201, A*0202, A*0203, A*0206, A*0207, A*04, A*6802 | Predicted | ELISpot; Cytotoxicity assay; Immunization of mice; Binding assay; Tetramer staining |
| LLVPFVQWFV | S | 349–358 | [93,101,111] | A*02 | Predicted | ICS; Degranulation assay; ELISpot; Tetramer staining |
| VGLSPTVWL | S | 358–366 | [85] | A*2402 | Predicted | ELISpot |
| GLSPTVWLS | S | 359–367 | [123] | A*0201 | Predicted | Immunization of transgenic mice; Cytotoxicity assay; ELISpot; Binding assay |
| GLSPTVWLSV | S | 359–368 | [72,73,90,93,105,111,114,116,124,125,128,130,134] | A*02, A*0201, A*0203, A*0207 | overlapping | Immunization of mice; FACS; CTL assay; ELISpot; Tetramer staining; Degranulation assay |
| VWLSVIWM | S | 364–371 | [90] | A*0201 | Predicted | Immunization of mice; FACS |
| (L)SVIWMMWYW | S | 366/367–375 | [62] | B*5801 | Predicted | ELISpot; ICS; Binding assay |
| SVIWMMWYW | S | 367–375 | [63,107] | B*5801 | overlapping | Tetramer staining; ICS; ELISpot |
| SIVSPFIPLL | S | 370–379 | [131] | A*0201 | Predicted | Binding assay; ELISpot |
| ILSPFLPLL | S | 371–379 | [131] | A*0201 | Predicted | Binding assay; ELISpot |
| MMWYWGPSLY | S | 371–380 | [74] | A*03 | Predicted | Binding assay; CTL assay; Cytotoxicity assay |
| NILSPFLPLL | S | 381–390 | [131] | A*0201 | Predicted | Binding assay; ELISpot |
| SILSPFLPLL | S | 381–390 | [77] | A*0201 | Predicted | ICS; Tetramer staining; |
| SIVSPFIPLL | S | 381–390 | [72,73,116,123] | A*02, A*0201 | Predicted | Immunization of mice; FACS; CTL assay; ELISpot; Tetramer staining |
| ILSPFLPLL | S | 382–390 | [75,90] | A*0201 | Predicted | Immunization of mice; FACS; CTL assay |
| IVSPFIPLL | S | 382–390 | [134] | A*0201 | Predicted | ELISA; Cytotoxicity assay |
| ILRSFIPLL | S | 382–390 | [95] | A*02, A*24 | Predicted | ELISpot |
| LLPIFFCLWV | S | 389–398 | [101] | A*02 | Predicted | ELISpot |
| DIDPYKEFGATVELL | Core | 2–16 | [135] | DRB1*0401 | overlapping | Proliferation assay; ICS |
| IDPYKEFGATVELLS | Core | 3–17 | [135] | DRB1*0401 | overlapping | Proliferation assay; ICS |
| DPYKEFGATVELLSF | Core | 4–18 | [135] | DRB1*0401 | overlapping | Proliferation assay; ICS |
| PYKEFGATVELLSFL | Core | 5–19 | [135] | DRB1*0401 | overlapping | Proliferation assay; ICS |
| YKEFGATVELLSFLP | Core | 6–20 | [135,136] | DRB1*0401, DRB1*1202 | overlapping | ICS; Proliferation assay |
| KEFGATVELLSFLPS | Core | 7–21 | [135] | DRB1*0401 | overlapping | Proliferation assay; ICS |
| EFGATVELLSFLPSD | Core | 8–22 | [135] | DRB1*0401 | overlapping | Proliferation assay; ICS |
| FGATVELLSFLPSDF | Core | 9–23 | [135] | DRB1*0401 | overlapping | Proliferation assay; ICS |
| GATVELLSFLPSDFF | Core | 10–24 | [135] | DRB1*0401 | overlapping | Proliferation assay; ICS |
| TVELLSFLPSDFFPS | Core | 12–26 | [135] | DRB1*0401 | overlapping | Proliferation assay; ICS |
| VELLSFLPSDFFPSV | Core | 13–27 | [135] | DRB1*0401 | overlapping | Proliferation assay; ICS |
| LLSFLPSDFFPSVRD | Core | 15–29 | [135] | DRB1*0401 | overlapping | Proliferation assay; ICS |
| LSFLPSDFFPSVRDL | Core | 16–30 | [135] | DRB1*0401 | overlapping | Proliferation assay; ICS |
| FLPSDFFPSVRD | Core | 18–29 | [137] | DPw4, DRB1*07 | Predicted | Cytotoxicity assay |
| RDLLDTASALYREALESPEH | Core | 28–47 | [138] | DRB1*07, DPw4 | overlapping | Proliferation assay |
| ALYREALESPEHCSP | Core | 36–50 | [136] | DRB1*1202 | overlapping | ICS |
| ALESPEHCSPHHTALRQAIL | Core | 41–60 | [139] | DRB1*13 | overlapping | Proliferation assay |
| EHCSPHHTALRQAIL | Core | 46–60 | [136] | DRB1*0803 | overlapping | ICS |
| PHHTALRQAILCWGELMTLA | Core | 50–69 | [81] | DRB1*07, DRB1*09, DRB1*11 | Predicted | Binding assay; Immunization of mice; ELISpot |
| HHTALRQAILCWGEL | Core | 51–65 | [136] | DRB1*1202 | overlapping | ICS |
| RQAILCWGELMNLAT | Core | 56–70 | [136] | DRB1*0803, DRB1*1202 | overlapping | ICS |
| LCWGELMTLATWVGVN | Core | 60–76 | [140] | DRB1*0101 | Predicted | Proliferation assay; ICS; Tetramer staining |
| MNLATWVGSNLEDPA | Core | 66–80 | [136] | DRB1*0803 | overlapping | ICS |
| LEDPASRELVVSYVN | Core | 76–90 | [136] | DRB1*1202 | overlapping | ICS |
| SRELVVSYVNVNMGL | Core | 81–95 | [136] | DRB1*0803 | overlapping | ICS |
| LEYLVSFGVWIRTPP | Core | 116–130 | [136] | DRB1*1202 | overlapping | ICS |
| EYLVSFGVWIRTPPA | Core | 117–131 | [138] | DRW52, DRB1*06 | overlapping | Proliferation assay |
| VSFGVWIRTPPAYRPPNAPI | Core | 120–139 | [81,138] | DRB1*01, DRB1*07, DRB1*11, DRB1*12, DRB1*13 | overlapping | Binding assay; Immunization of mice; ELISpot; Proliferation assay |
| NAPILSTLPETTVVR | Core | 136–150 | [136] | DRB1*0803 | overlapping | ICS |
| STLPETTVVRRRGRS | Core | 141–155 | [136] | DRB1*1202 | overlapping | ICS |
| STLPETTVVRRRGRSPRRRT | Core | 141–160 | [141] | DRB1*13 | Predicted | Proliferation assay; Cytotoxicity assay; ICS |
| PRRRTPSPRRRRSQS | Core | 156–170 | [136] | DRB1*0803 | overlapping | ICS |
| PPAYRPPNAPILSTL | Core | 158–172 | [135] | DRB1*0101 | overlapping | Proliferation assay; ICS |
| PAYRPPNAPIL | Core | 159–169 | [142] | DR52, DRw3 | overlapping | Proliferation assay; Cytotoxicity assay |
| PSPRRRRSQSPRRRR | Core | 161–175 | [136] | DRB1*0803 | overlapping | ICS |
| RRSQSPRRRRSQSRE | Core | 166–180 | [136] | DRB1*1202 | overlapping | ICS |
| YFKDCLFKDWEELGE | HBx | 111–125 | [143] | DRB1*1301 | overlapping | ELISpot; Binding assay; ICS |
| EIRLKVFVLGGCRHK | HBx | 126–140 | [143] | DRB1*0101, DRB1*0401, DRB1*1301, DRB5*0101 | overlapping | ELISpot; Binding assay; ICS |
| VFVLGGCRHKLVCAP | HBx | 131–145 | [143] | DRB1*1301 | overlapping | ELISpot; Binding assay; ICS |
| VGPLTVNEKRRLKLI | Pol | 96–111 | [113] | DRB1*0301 | Predicted | ELISpot; Cytotoxicity assay |
| RHYLHTLWKAGILYK | Pol | 145–160 | [113] | DRB1*0301, DRB1*07, DRB1*08, DRB1*09, DRB1*11, DRB1*12, DRB1*15 | Predicted | ELISpot; Cytotoxicity assay |
| ESRLVVDFSQFSRGN | Pol | 385–400 | [113] | DRB1*03, DRB1*04 | Predicted | ELISpot; Cytotoxicity assay |
| LQSLTNLLSSNLSWL | Pol | 412–427 | [113] | DRB1*01, DRB1*04, DRB1*07, DRB1*11, DRB1*12, DRB1*13, DRB1*15 | Predicted | ELISpot; Cytotoxicity assay |
| SSNLSWLSLDVSAAF | Pol | 420–435 | [113] | DRB1*01, DRB1*03, DRB1*04, DRB1*13 | Predicted | ELISpot; Cytotoxicity assay |
| LHLYSHPIILGFRKI | Pol | 501–516 | [113] | DRB1*01, DRB1*04, DRB1*11 | Predicted | ELISpot; Cytotoxicity assay |
| PFLLAQFTSAICSVV | Pol | 525–538 | [81] | DRB1*01, DRB1*04, DRB1*07, DRB1*08, DRB1*09, DRB1*11, DRB1*15, DRB5*01 | Predicted | Binding assay; Immunization of mice; ELISpot |
| KQCFRKLPVNRPIDW | Pol | 618–633 | [81,113] | DRB1*01, DRB1*04, DRB1*07, DRB1*13 | Predicted | Binding assay; Immunization of mice; ELISpot; Cytotoxicity assay |
| LCQVFADATPTGWGL | Pol | 649–664 | [81] | DRB1*03, DRB1*04, DRB1*07 | Predicted | Binding assay; Immunization of mice; ELISpot |
| KQAFTFSPTYKAFLC | Pol | 664–679 | [113] | DRB1*01, DRB1*04, DRB1*07, DRB1*08, DRB1*09, DRB1*11, DRB1*13, DRB1*15 | Predicted | ELISpot; Cytotoxicity assay |
| AANWILRGTSFVYVP | Pol | 676–691 | [81] | DRB1*07, DRB1*08, DRB1*09, DRB1*12, DRB1*13, DRB1*15 | Predicted | Binding assay; Immunization of mice; ELISpot |
| LCQVFADATPTGWGL | Pol | 694–709 | [113] | DRB1*03, DRB1*04 | Predicted | ELISpot; Cytotoxicity assay |
| AANWILRGTSFVYVP | Pol | 767–782 | [113] | DRB1*01, DRB1*07, DRB1*08, DRB1*09, DRB1*13, DRB1*15 | Predicted | ELISpot; Cytotoxicity assay |
| GTSFVYVPSALNPAD | Pol | 774–789 | [81] | DRB1*01, DRB1*04, DRB1*07, DRB1*08, DRB1*09, DRB1*11, DRB1*15, DRB5*01 | Predicted | Binding assay; Immunization of mice; ELISpot |
| AGFFLLTRILTIPQS | S | 17–31 | [144] | DRB1*07, DRB1*08, DRB1*11, DRB1*13 | Predicted | ELISpot; Proliferation assay |
| GFFPDHQLDPAF | S | 23–33 | [145] | DRB1*0405 | Predicted | Binding assay; FASC |
| TSLNFLGGSPVCLGQ | S | 37–51 | [144] | DRB1*01 | Predicted | ELISpot; Proliferation assay |
| GAFGPGFTPPHG | S | 61–72 | [145] | DRB1*0405 | Predicted | Binding assay; FASC |
| PICPGYRWMCLRRFI | S | 67–81 | [144] | DRB1*08, DRB1*11, DRB1*13 | Predicted | ELISpot; Proliferation assay |
| GWSPQAQGVLTT | S | 76–87 | [145] | DRB1*0405 | Predicted | ELISpot; Proliferation assay |
| MQWNSTTFHQTLQDPRVRGL | S | 109–134 | [47] | DRB1*01 | Predicted | Immunization of mice; Proliferation assay; ELISpot |
| TTFHQTLQDPRVRGL | S | 114–128 | [47] | DRB1*01 | Predicted | Immunization of mice; Proliferation assay; ELISpot |
| MQWNSTAFHQTLQDP | S | 109–123 | [146] | DRB1*02 | Predicted | Proliferation assay; Cytotoxicity assay |
| STLPETTVVRRRGRSPRRRT | S | 141–160 | [139] | DRB1*13 | overlapping | Proliferation assay |
| WASVRFSWLSLL | S | 165–176 | [147] | DRB1*11, DRB1*14 | Predicted | CTL assay; Proliferation assay |
| VPFVQWFVGLSPTVW | S | 177–191 | [144] | DRB1*11 | Predicted | ELISpot; Proliferation assay |
| QAGFFLLTRILTIPQS | S | 179–194 | [47] | DRB1*01 | Predicted | Immunization of mice; Proliferation assay; ELISpot |
| WLSVIWMMWYWGPSL | S | 191–205 | [136] | DRB1*1202 | overlapping | ICS |
| TSLNFLGGTTVCLGQ | S | 200–214 | [47] | DRB1*01 | Predicted | Immunization of mice; Proliferation assay; ELISpot |
| GPSLYSIVSPFIPLL | S | 202–216 | [144] | DRB1*07 | Predicted | ELISpot; Proliferation assay |
| LLPIFFCLWVYI | S | 215–226 | [147] | DRB1*07, DRB1*08, DRB1*14 | Predicted | CTL assay; Proliferation assay |
| PICPGYRWMCLRRFIIFL | S | 241–258 | [148] | DRB1*0201 | overlapping | Tetramer staining |
| FLLVLLDYQGMLP | S | 256–268 | [54] | DP4 | Predicted | Immunization of mice; Proliferation assay; ELISpot |
| WEWASARFSWLSL | S | 326–338 | [54] | DP4 | Predicted | Immunization of mice; Proliferation assay; ELISpot |
| WLSLLVPFVQWFVGL | S | 335–349 | [149] | DRB1*0101 | Predicted | Immunization of mice; Pentamer staining; ELISpot; ICS; Cytotoxicity assay |
| SLLVPFVQWFVGLSPTVWLSV | S | 337–357 | [47] | DRB1*01 | Predicted | Immunization of mice; Proliferation assay; ELISpot |
| SVRFSWLSLLVPFVQWF | S | 343–357 | [148] | DRB1*0201 | overlapping | Tetramer staining |
| VGLSPTVWLSVI | S | 347–358 | [54] | DP4 | Predicted | Immunization of mice; Proliferation assay; ELISpot |
| GLSPTVWLSVIW | S | 348–359 | [149] | DRB1*0101 | Predicted | Immunization of mice; Pentamer staining; ELISpot; ICS; Cytotoxicity assay |
| TVWLSVIWMMWYW | S | 352–364 | [54] | DP4 | Predicted | Immunization of mice; Proliferation assay; ELISpot |
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- World Health Organization. Global Hepatitis Report 2017; World Health Organization: Geneva, Switzerland, 2017. [Google Scholar]
- Liu, J.; Liang, W.; Jing, W.; Liu, M. Countdown to 2030: Eliminating hepatitis B disease, China. Bull. World Health Organ. 2019, 97, 230–238. [Google Scholar] [CrossRef]
- Rouviere, C.P.; Dousson, C.B.; Tavis, J.E. HBV replication inhibitors. Antivir. Res. 2020, 179, 104815. [Google Scholar] [CrossRef] [PubMed]
- Viganò, M.; Mangia, G.; Lampertico, P. HBeAg-negative chronic hepatitis B: Why do I treat my patients with nucleos(t)ide analogues? Liver Int. 2014, 34, 120–126. [Google Scholar] [CrossRef]
- Buti, M. HBeAg-positive chronic hepatitis B: Why do I treat my patients with Nucleos(t)ide Analogs? Liver Int. 2013, 34, 108–111. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Cameo, C.; Pons, M.; Esteban, R. New therapeutic perspectives in HBV: When to stop NAs. Liver Int. 2013, 34, 146–153. [Google Scholar] [CrossRef] [PubMed]
- Isogawa, M.; Tanaka, Y. Immunobiology of hepatitis B virus infection. Hepatol. Res. 2014, 45, 179–189. [Google Scholar] [CrossRef] [PubMed]
- Tseng, T.-C.; Huang, L.-R. Immunopathogenesis of Hepatitis B Virus. J. Infect. Dis. 2017, 216, S765–S770. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Zhu, Y.O.; Becht, E.; Aw, P.; Chen, J.; Poidinger, M.; de Sessions, P.F.; Hibberd, M.L.; Bertoletti, A.; Lim, S.G.; et al. Multifactorial heterogeneity of virus-specific T cells and association with the progression of human chronic hepatitis B infection. Sci. Immunol. 2019, 4, eaau6905. [Google Scholar] [CrossRef]
- Wang, X.; He, Q.; Shen, H.; Lu, X.-J.; Sun, B. Genetic and phenotypic difference in CD8+ T cell exhaustion between chronic hepatitis B infection and hepatocellular carcinoma. J. Med. Genet. 2018, 56, 18–21. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; Liang, T.J. Development of Direct-acting Antiviral and Host-targeting Agents for Treatment of Hepatitis B Virus Infection. Gastroenterology 2019, 156, 311–324. [Google Scholar] [CrossRef]
- Tang, T.J.; Kwekkeboom, J.; Mancham, S.; Binda, R.S.; de Man, R.A.; Schalm, S.W.; Kusters, J.G.; Janssen, H.L. Intrahepatic CD8+ T-lymphocyte response is important for therapy-induced viral clearance in chronic hepatitis B infection. J. Hepatol. 2005, 43, 45–52. [Google Scholar] [CrossRef]
- Papatheodoridis, G.; Vlachogiannakos, I.; Cholongitas, E.; Wursthorn, K.; Thomadakis, C.; Touloumi, G.; Petersen, J. Discontinuation of oral antivirals in chronic hepatitis B: A systematic review. Hepatology 2016, 63, 1481–1492. [Google Scholar] [CrossRef]
- Lampertico, P.; Agarwal, K.; Berg, T.; Buti, M.; Janssen, H.L.A.; Papatheodoridis, G.V.; Zoulim, F.; Tacke, F. EASL 2017 Clinical Practice Guidelines on the management of hepatitis B virus infection. J. Hepatol. 2017, 67, 370–398. [Google Scholar] [CrossRef] [PubMed]
- Rivino, L.; Le Bert, N.; Gill, U.S.; Kunasegaran, K.; Cheng, Y.; Tan, D.Z.; Becht, E.; Hansi, N.K.; Foster, G.R.; Su, T.-H.; et al. Hepatitis B virus–specific T cells associate with viral control upon nucleos(t)ide-analogue therapy discontinuation. J. Clin. Investig. 2018, 128, 668–681. [Google Scholar] [CrossRef] [PubMed]
- Schuch, A.; Alizei, E.S.; Heim, K.; Wieland, D.; Kiraithe, M.M.; Kemming, J.; Llewellyn-Lacey, S.; Sogukpinar, Ö.; Ni, Y.; Urban, S.; et al. Phenotypic and functional differences of HBV core-specific versus HBV polymerase-specific CD8+ T cells in chronically HBV-infected patients with low viral load. Gut 2019, 68, 905–915. [Google Scholar] [CrossRef] [PubMed]
- Lang-Meli, J.; Neumann-Haefelin, C.; Thimme, R. Immunotherapy and therapeutic vaccines for chronic HBV infection. Curr. Opin. Virol. 2021, 51, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Loomba, R.; Liang, T.J. Hepatitis B Reactivation Associated With Immune Suppressive and Biological Modifier Therapies: Current Concepts, Management Strategies, and Future Directions. Gastroenterology 2017, 152, 1297–1309. [Google Scholar] [CrossRef]
- Desmond, C.P.; Bartholomeusz, A.; Gaudieri, S.; Revill, P.; Lewin, S.R. A systematic review of T-cell epitopes in hepatitis B virus: Identification, genotypic variation and relevance to antiviral therapeutics. Antivir. Ther. 2008, 13, 161–175. [Google Scholar]
- Nitschke, K.; Luxenburger, H.; Neumann-Haefelin, C.; Kiraithe, M.M.; Thimme, R. CD8+ T-Cell Responses in Hepatitis B and C: The (HLA-) A, B, and C of Hepatitis B and C. Dig. Dis. 2016, 34, 396–409. [Google Scholar] [CrossRef]
- Roche, P.A.; Furuta, K. The ins and outs of MHC class II-mediated antigen processing and presentation. Nat. Rev. Immunol. 2015, 15, 203–216. [Google Scholar] [CrossRef] [PubMed]
- Elahi, S.; Horton, H. Association of HLA-alleles with the immune regulation of chronic viral infections. Int. J. Biochem. Cell Biol. 2012, 44, 1361–1365. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zou, Z.-Q.; Wang, K. Clinical Relevance of HLA Gene Variants in HBV Infection. J. Immunol. Res. 2016, 2016, 9069375. [Google Scholar] [CrossRef] [PubMed]
- Boeijen, L.L.; Hoogeveen, R.C.; Boonstra, A.; Lauer, G.M. Hepatitis B virus infection and the immune response: The big questions. Best Pract. Res. Clin. Gastroenterol. 2017, 31, 265–272. [Google Scholar] [CrossRef] [PubMed]
- Medzhitov, R. Recognition of microorganisms and activation of the immune response. Nature 2007, 449, 819–826. [Google Scholar] [CrossRef]
- Thursz, M.; Kwiatkowski, D.; Allsopp, C.E.; Greenwood, B.M.; Thomas, H.C.; Hill, A.V. Association between an MHC Class II Allele and Clearance of Hepatitis B Virus in the Gambia. N. Engl. J. Med. 1995, 332, 1065–1069. [Google Scholar] [CrossRef]
- Cotrina, M.; Buti, M.; Jardí, R.; Rodríguez-Frías, F.; Campins, M.; Esteban, R.; Guardia, J. [Study of HLA-II antigens in chronic hepatitis C and B and in acute hepatitis B]. Gastroenterología Hepatología 1997, 20, 115–118. [Google Scholar]
- Ahn, S.H.; Han, K.-H.; Park, J.Y.; Lee, C.K.; Kang, S.-W.; Chon, C.Y.; Kim, Y.S.; Park, K.; Kim, D.K.; Moon, Y.M. Association between hepatitis B virus infection and HLA-DR type in Korea. Hepatology 2000, 31, 1371–1373. [Google Scholar] [CrossRef]
- Yang, G.; Liu, J.; Han, S.; Xie, H.; Du, R.; Yan, Y.; Xu, D.; Fan, D. Association between hepatitis B virus infection and HLA-DRB1 genotyping in Shaanxi Han patients in northwestern China. Tissue Antigens 2007, 69, 170–175. [Google Scholar] [CrossRef]
- Ramezani, A.; Roshan, M.R.H.; Kalantar, E.; Eslamifar, A.; Banifazl, M.; Taeb, J.; Aghakhani, A.; Gachkar, L.; Velayati, A.A. Association of human leukocyte antigen polymorphism with outcomes of hepatitis B virus infection. J. Gastroenterol. Hepatol. 2008, 23, 1716–1721. [Google Scholar] [CrossRef]
- Thio, C.L.; Thomas, D.L.; Karacki, P.; Gao, X.; Marti, D.; Kaslow, R.A.; Goedert, J.J.; Hilgartner, M.; Strathdee, S.A.; Duggal, P.; et al. Comprehensive Analysis of Class I and Class II HLA Antigens and Chronic Hepatitis B Virus Infection. J. Virol. 2003, 77, 12083–12087. [Google Scholar] [CrossRef]
- Fattovich, G.; Bortolotti, F.; Donato, F. Natural history of chronic hepatitis B: Special emphasis on disease progression and prognostic factors. J. Hepatol. 2008, 48, 335–352. [Google Scholar] [CrossRef]
- Rashidi, S.; Farhadi, L.; Ghasemi, F.; Sheikhesmaeili, F.; Mohammadi, A. The potential role of HLA-G in the pathogenesis of HBV infection: Immunosuppressive or immunoprotective? Infect. Genet. Evol. 2020, 85, 104580. [Google Scholar] [CrossRef]
- Xu, R.; Hu, P.; Li, Y.; Tian, A.; Li, J.; Zhu, C. Advances in HBV infection and replication systems in vitro. Virol. J. 2021, 18, 105. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.M.; Ahn, S.H. Quantification of HBsAg: Basic virology for clinical practice. World J. Gastroenterol. 2011, 17, 283–289. [Google Scholar] [CrossRef] [PubMed]
- Buti, M.; Frías, F.R.; Esteban, R. Cuantificación del antígeno de superficie del virus de la hepatitis B: Implicaciones clínicas. Med. Clin. 2012, 138, 483–488. [Google Scholar] [CrossRef] [PubMed]
- Alexopoulou, A. HBeAg negative variants and their role in the natural history of chronic hepatitis B virus infection. World J. Gastroenterol. 2014, 20, 7644–7652. [Google Scholar] [CrossRef]
- Ou, J.-H. Molecular biology of hepatitis B virus e antigen. J. Gastroenterol. Hepatol. 1997, 12, S178–S187. [Google Scholar] [CrossRef]
- Mak, L.-Y.; Wong, D.K.-H.; Cheung, K.-S.; Seto, W.-K.; Lai, C.-L.; Yuen, M.-F. Review article: Hepatitis B core-related antigen (HBcrAg): An emerging marker for chronic hepatitis B virus infection. Aliment. Pharmacol. Ther. 2017, 47, 43–54. [Google Scholar] [CrossRef]
- Ali, A.; Abdel-Hafiz, H.; Suhail, M.; Al-Mars, A.; Zakaria, M.K.; Fatima, K.; Ahmad, S.; Azhar, E.; Chaudhary, A.; Qadri, I. Hepatitis B virus, HBx mutants and their role in hepatocellular carcinoma. World J. Gastroenterol. 2014, 20, 10238–10248. [Google Scholar] [CrossRef]
- Qi, Y.; Gao, Z.; Xu, G.; Peng, B.; Liu, C.; Yan, H.; Yao, Q.; Sun, G.; Liu, Y.; Tang, D.; et al. DNA Polymerase κ Is a Key Cellular Factor for the Formation of Covalently Closed Circular DNA of Hepatitis B Virus. PLoS Pathog. 2016, 12, e1005893. [Google Scholar] [CrossRef]
- Gerlich, W.H.; Glebe, D.; Kramvis, A.; Magnius, L.O. Peculiarities in the designations of hepatitis B virus genes, their products, and their antigenic specificities: A potential source of misunderstandings. Virus Genes 2020, 56, 109–119. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Yang, D.; Li, S.; Gao, Y.; Jiang, R.; Deng, L.; Frankel, F.R.; Sun, B. Development of a Listeria monocytogenes-based vaccine against hepatocellular carcinoma. Oncogene 2011, 31, 2140–2152. [Google Scholar] [CrossRef] [PubMed]
- Brinck-Jensen, N.-S.; Vorup-Jensen, T.; Leutscher, P.D.C.; Erikstrup, C.; Petersen, E. Immunogenicity of twenty peptides representing epitopes of the hepatitis B core and surface antigens by IFN-γ response in chronic and resolved HBV. BMC Immunol. 2015, 16, 65. [Google Scholar] [CrossRef] [PubMed]
- de Beijer, M.T.A.; Jansen, D.T.S.L.; Dou, Y.; van Esch, W.J.E.; Mok, J.Y.; Maas, M.J.P.; Brasser, G.; de Man, R.A.; Woltman, A.M.; Buschow, S.I. Discovery and Selection of Hepatitis B Virus-Derived T Cell Epitopes for Global Immunotherapy Based on Viral Indispensability, Conservation, and HLA-Binding Strength. J. Virol. 2020, 94, e01663-19. [Google Scholar] [CrossRef]
- Shafer-Weaver, K.; Sayers, T.; Strobl, S.; Derby, E.; Ulderich, T.; Baseler, M.; Malyguine, A. The Granzyme B ELISPOT assay: An alternative to the 51Cr-release assay for monitoring cell-mediated cytotoxicity. J. Transl. Med. 2003, 1, 14. [Google Scholar] [CrossRef]
- Pajot, A.; Michel, M.-L.; Bourgine, M.; Ungeheuer, M.-N.; Ojcius, D.; Deng, Q.; Lemonnier, F.A.; Lone, Y.-C. Identification of novel HLA-DR1-restricted epitopes from the hepatitis B virus envelope protein in mice expressing HLA-DR1 and vaccinated human subjects. Microbes Infect. 2006, 8, 2783–2790. [Google Scholar] [CrossRef]
- Freer, G.; Rindi, L. Intracellular cytokine detection by fluorescence-activated flow cytometry: Basic principles and recent advances. Methods 2013, 61, 30–38. [Google Scholar] [CrossRef]
- Ji, N.; Forsthuber, T.G. ELISPOT Techniques. In Multiple Sclerosis; Humana Press: New York, NY, USA, 2014; Volume 1304, pp. 63–71. [Google Scholar]
- Reynolds, T.D.; Moshkani, S.; Robek, M.D. An ELISPOT-Based Assay to Measure HBV-Specific CD8+ T Cell Responses in Immunocompetent Mice. In Hepatitis B Virus; Methods in Molecular Biology; Humana Press: New York, NY, USA, 2017; Volume 1540, pp. 237–247. [Google Scholar]
- Dolton, G.; Tungatt, K.; Lloyd, A.; Bianchi, V.; Theaker, S.M.; Trimby, A.; Holland, C.J.; Donia, M.; Godkin, A.; Cole, D.; et al. More tricks with tetramers: A practical guide to staining T cells with peptide-MHC multimers. Immunology 2015, 146, 11–22. [Google Scholar] [CrossRef]
- Savage, P.; Millrain, M.; Dimakou, S.; Stebbing, J.; Dyson, J. Expansion of CD8+ Cytotoxic T Cells in vitro and in vivo Using MHC Class I Tetramers. Tumor Biol. 2007, 28, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Bertoletti, A.; Ferrari, C. Adaptive immunity in HBV infection. J. Hepatol. 2016, 64, S71–S83. [Google Scholar] [CrossRef]
- Ru, Z.; Xiao, W.; Pajot, A.; Kou, Z.; Sun, S.; Maillere, B.; Zhao, G.; Ojcius, D.; Lone, Y.-C.; Zhou, Y. Development of a Humanized HLA-A2.1/DP4 Transgenic Mouse Model and the Use of This Model to Map HLA-DP4-Restricted Epitopes of HBV Envelope Protein. PLoS ONE 2012, 7, e32247. [Google Scholar] [CrossRef] [PubMed]
- Ferretti, A.P.; Kula, T.; Wang, Y.; Nguyen, D.M.; Weinheimer, A.; Dunlap, G.S.; Xu, Q.; Nabilsi, N.; Perullo, C.R.; Cristofaro, A.W.; et al. Unbiased Screens Show CD8+ T Cells of COVID-19 Patients Recognize Shared Epitopes in SARS-CoV-2 that Largely Reside outside the Spike Protein. Immunity 2020, 53, 1095–1107.e3. [Google Scholar] [CrossRef] [PubMed]
- Chikata, T.; Paes, W.; Akahoshi, T.; Partridge, T.; Murakoshi, H.; Gatanaga, H.; Ternette, N.; Oka, S.; Borrow, P.; Takiguchi, M. Identification of Immunodominant HIV-1 Epitopes Presented by HLA-C*12:02, a Protective Allele, Using an Immunopeptidomics Approach. J. Virol. 2019, 93, e00634-19. [Google Scholar] [CrossRef] [PubMed]
- Paul, S.; Croft, N.P.; Purcell, A.W.; Tscharke, D.C.; Sette, A.; Nielsen, M.; Peters, B. Benchmarking predictions of MHC class I restricted T cell epitopes in a comprehensively studied model system. PLoS Comput. Biol. 2020, 16, e1007757. [Google Scholar] [CrossRef]
- Palucka, K.; Banchereau, J. Dendritic-Cell-Based Therapeutic Cancer Vaccines. Immunity 2013, 39, 38–48. [Google Scholar] [CrossRef] [PubMed]
- Lumley, S.; Noble, H.; Hadley, M.J.; Callow, L.; Malik, A.; Chua, Y.Y.; Duffey, O.J.; Grolmusova, N.; Kumar, A.; Ravenscroft, S.; et al. Hepitopes: A live interactive database of HLA class I epitopes in hepatitis B virus. Wellcome Open Res. 2016, 1, 9. [Google Scholar] [CrossRef]
- Nayagam, S.; Thursz, M.; Sicuri, E.; Conteh, L.; Wiktor, S.; Low-Beer, D.; Hallett, T. Requirements for global elimination of hepatitis B: A modelling study. Lancet Infect. Dis. 2016, 16, 1399–1408. [Google Scholar] [CrossRef]
- Zheng, J.; Ou, Z.; Lin, X.; Wang, L.; Liu, Y.; Jin, S.; Wu, J. Analysis of epitope-based vaccine candidates against the E antigen of the hepatitis B virus based on the B genotype sequence: An in silico and in vitro approach. Cell. Immunol. 2018, 329, 56–65. [Google Scholar] [CrossRef]
- Rivino, L.; Tan, A.T.; Chia, A.; Kumaran, E.A.P.; Grotenbreg, G.M.; MacAry, P.; Bertoletti, A. Defining CD8+ T Cell Determinants during Human Viral Infection in Populations of Asian Ethnicity. J. Immunol. 2013, 191, 4010–4019. [Google Scholar] [CrossRef]
- Tan, A.T.; Sodsai, P.; Chia, A.; Moreau, E.; Chng, M.H.Y.; Tham, C.Y.L.; Ho, Z.Z.; Banu, N.; Hirankarn, N.; Bertoletti, A. Immunoprevalence and Immunodominance of HLA-Cw*0801-Restricted T Cell Response Targeting the Hepatitis B Virus Envelope Transmembrane Region. J. Virol. 2013, 88, 1332–1341. [Google Scholar] [CrossRef]
- Hoogeveen, R.C.; Robidoux, M.P.; Schwarz, T.; Heydmann, L.; A Cheney, J.; Kvistad, D.; Aneja, J.; Melgaço, J.G.; A Fernandes, C.; Chung, R.T.; et al. Phenotype and function of HBV-specific T cells is determined by the targeted epitope in addition to the stage of infection. Gut 2019, 68, 893–904. [Google Scholar] [CrossRef] [PubMed]
- Urbani, S.; Boni, C.; Amadei, B.; Fisicaro, P.; Cerioni, S.; Valli, M.A.; Missale, G.; Ferrari, C. Acute phase HBV-specific T cell responses associated with HBV persistence after HBV/HCV coinfection. Hepatology 2005, 41, 826–831. [Google Scholar] [CrossRef] [PubMed]
- Shen, C.; Xu, T.; Wu, Y.; Li, X.; Xia, L.; Wang, W.; Shahzad, K.A.; Zhang, L.; Wan, X.; Qiu, J. Frequency and reactivity of antigen-specific T cells were concurrently measured through the combination of artificial antigen-presenting cell, MACS and ELISPOT. Sci. Rep. 2017, 7, 16400. [Google Scholar] [CrossRef]
- Tan, A.T.; Yang, N.; Krishnamoorthy, T.; Oei, V.; Chua, A.; Zhao, X.; Tan, H.S.; Chia, A.; Le Bert, N.; Low, D.; et al. Use of Expression Profiles of HBV-DNA Integrated Into Genomes of Hepatocellular Carcinoma Cells to Select T Cells for Immunotherapy. Gastroenterology 2019, 156, 1862–1876.e9. [Google Scholar] [CrossRef] [PubMed]
- Khakpoor, A.; Ni, Y.; Chen, A.; Ho, Z.Z.; Oei, V.; Yang, N.; Giri, R.; Chow, J.X.; Tan, A.T.; Kennedy, P.T.; et al. Spatiotemporal Differences in Presentation of CD8 T Cell Epitopes during Hepatitis B Virus Infection. J. Virol. 2019, 93, e01457-18. [Google Scholar] [CrossRef] [PubMed]
- Bertoletti, A.; Chisari, F.V.; Penna, A.; Guilhot, S.; Galati, L.; Missale, G.; Fowler, P.; Schlicht, H.J.; Vitiello, A.; Chesnut, R.C. Definition of a minimal optimal cytotoxic T-cell epitope within the hepatitis B virus nucleocapsid protein. J. Virol. 1993, 67, 2376–2380. [Google Scholar] [CrossRef]
- Bertoletti, A.; Costanzo, A.; Chisari, F.; Levrero, M.; Artini, M.; Sette, A.; Penna, A.; Giuberti, T.; Fiaccadori, F.; Ferrari, C. Cytotoxic T lymphocyte response to a wild type hepatitis B virus epitope in patients chronically infected by variant viruses carrying substitutions within the epitope. J. Exp. Med. 1994, 180, 933–943. [Google Scholar] [CrossRef]
- Rehermann, B.; Fowler, P.; Sidney, J.; Person, J.; Redeker, A.; Brown, M.; Moss, B.; Sette, A.; Chisari, F. The cytotoxic T lymphocyte response to multiple hepatitis B virus polymerase epitopes during and after acute viral hepatitis. J. Exp. Med. 1995, 181, 1047–1058. [Google Scholar] [CrossRef]
- Rehermann, B.; Pasquinelli, C.; Mosier, S.M.; Chisari, F.V. Hepatitis B virus (HBV) sequence variation of cytotoxic T lymphocyte epitopes is not common in patients with chronic HBV infection. J. Clin. Investig. 1995, 96, 1527–1534. [Google Scholar] [CrossRef]
- Sette, A.; Vitiello, A.; Reherman, B.; Fowler, P.; Nayersina, R.; Kast, W.M.; Melief, C.J.; Oseroff, C.; Yuan, L.; Ruppert, J.; et al. The relationship between class I binding affinity and immunogenicity of potential cytotoxic T cell epitopes. J. Immunol. 1994, 153, 5586–5592. [Google Scholar]
- Bertoni, R.; Sidney, J.; Fowler, P.; Chesnut, R.W.; Chisari, F.V.; Sette, A. Human histocompatibility leukocyte antigen-binding supermotifs predict broadly cross-reactive cytotoxic T lymphocyte responses in patients with acute hepatitis. J. Clin. Investig. 1997, 100, 503–513. [Google Scholar] [CrossRef][Green Version]
- Lee, H.G.; Lim, J.-S.; Lee, K.-Y.; Choi, Y.-K.; Choe, I.-S.; Chung, T.-W.; Kim, K. Peptide-specific CTL induction in HBV-seropositive PBMC by stimulation with peptides in vitro: Novel epitopes identified from chronic carriers. Virus Res. 1997, 50, 185–194. [Google Scholar] [CrossRef]
- Oseroff, C.; Sette, A.; Wentworth, P.; Celis, E.; Maewal, A.; Dahlberg, C.; Fikes, J.; Kubo, R.T.; Chesnut, R.W.; Grey, H.M.; et al. Pools of lipidated HTL-CTL constructs prime for multiple HBV and HCV CTL epitope responses. Vaccine 1998, 16, 823–833. [Google Scholar] [CrossRef]
- Tham, C.Y.; Kah, J.; Tan, A.T.; Volz, T.; Chia, A.; Giersch, K.; Ladiges, Y.; Loglio, A.; Borghi, M.; Sureau, C.; et al. Hepatitis Delta Virus Acts as an Immunogenic Adjuvant in Hepatitis B Virus-Infected Hepatocytes. Cell Rep. Med. 2020, 1, 100060. [Google Scholar] [CrossRef] [PubMed]
- Dou, Y.; Jansen, D.T.; Bosch, A.V.D.; de Man, R.A.; van Montfoort, N.; Araman, C.; van Kasteren, S.I.; Zom, G.G.; Krebber, W.-J.; Melief, C.J.; et al. Design of TLR2-ligand-synthetic long peptide conjugates for therapeutic vaccination of chronic HBV patients. Antivir. Res. 2020, 178, 104746. [Google Scholar] [CrossRef]
- Bihl, F.K.; Loggi, E.; Chisholm, J.V.; Hewitt, H.S.; Henry, L.M.; Linde, C.; Suscovich, T.J.; Wong, J.T.; Frahm, N.; Andreone, P.; et al. Simultaneous assessment of cytotoxic T lymphocyte responses against multiple viral infections by combined usage of optimal epitope matrices, anti- CD3 mAb T-cell expansion and “RecycleSpot”. J. Transl. Med. 2005, 3, 20. [Google Scholar] [CrossRef] [PubMed]
- Thimme, R.; Chang, K.-M.; Pemberton, J.; Sette, A.; Chisari, F.V. Degenerate Immunogenicity of an HLA-A2-Restricted Hepatitis B Virus Nucleocapsid Cytotoxic T-Lymphocyte Epitope That Is Also Presented by HLA-B. J. Virol. 2001, 75, 3984–3987. [Google Scholar] [CrossRef]
- Depla, E.; Van der Aa, A.; Livingston, B.D.; Crimi, C.; Allosery, K.; De Brabandere, V.; Krakover, J.; Murthy, S.; Huang, M.; Power, S.; et al. Rational Design of a Multiepitope Vaccine Encoding T-Lymphocyte Epitopes for Treatment of Chronic Hepatitis B Virus Infections. J. Virol. 2008, 82, 435–450. [Google Scholar] [CrossRef] [PubMed]
- Kefalakes, H.; Jochum, C.; Hilgard, G.; Kahraman, A.; Bohrer, A.M.; El Hindy, N.; Heinemann, F.M.; Verheyen, J.; Gerken, G.; Roggendorf, M.; et al. Decades after recovery from hepatitis B and HBsAg clearance the CD8+ T cell response against HBV core is nearly undetectable. J. Hepatol. 2015, 63, 13–19. [Google Scholar] [CrossRef]
- Pacella, I.; Cammarata, I.; Martire, C.; Brancaccio, G.; Gaeta, G.B.; Barnaba, V.; Piconese, S. CD8+ T cells specific to apoptosis-associated epitopes are expanded in patients with chronic HBV infection and fibrosis. Liver Int. 2021, 41, 470–481. [Google Scholar] [CrossRef]
- Kondo, Y.; Asabe, S.; Kobayashi, K.; Shiina, M.; Niitsuma, H.; Ueno, Y.; Kobayashi, T.; Shimosegawa, T. Recovery of functional cytotoxic T lymphocytes during lamivudine therapy by acquiring multi-specificity. J. Med. Virol. 2004, 74, 425–433. [Google Scholar] [CrossRef]
- Yamamiya, D.; Mizukoshi, E.; Kaji, K.; Terashima, T.; Kitahara, M.; Yamashita, T.; Arai, K.; Fushimi, K.; Honda, M.; Kaneko, S. Immune responses of human T lymphocytes to novel hepatitis B virus-derived peptides. PLoS ONE 2018, 13, e0198264. [Google Scholar] [CrossRef]
- Zhang, Y.; Ren, Y.; Wu, Y.; Zhao, B.; Qiu, L.; Li, X.; Xu, D.; Liu, J.; Gao, G.F.; Meng, S. The L60V Variation in Hepatitis B Virus Core Protein Elicits New Epitope-Specific Cytotoxic T Lymphocytes and Enhances Viral Replication. J. Virol. 2013, 87, 8075–8084. [Google Scholar] [CrossRef]
- Liu, Q.; Zheng, Y.; Yu, Y.; Tan, Q.; Huang, X. Identification of HLA-A*0201-restricted CD8+ T-cell epitope C64–72 from hepatitis B virus core protein. Int. Immunopharmacol. 2012, 13, 141–147. [Google Scholar] [CrossRef]
- Sobao, Y.; Sugi, K.; Tomiyama, H.; Saito, S.; Fujiyama, S.; Morimoto, M.; Hasuike, S.; Tsubouchi, H.; Tanaka, K.; Takiguchi, M. Identification of hepatitis B virus-specific CTL epitopes presented by HLA-A*2402, the most common HLA class I allele in East Asia. J. Hepatol. 2001, 34, 922–929. [Google Scholar] [CrossRef]
- Zhou, M.; Xu, Y.; Lou, Z.; Cole, D.K.; Li, X.; Liu, Y.; Tien, P.; Rao, Z.; Gao, G.F. Complex assembly, crystallization and preliminary X-ray crystallographic studies of MHC H-2Kdcomplexed with an HBV-core nonapeptide. Acta Crystallogr. Sect. D Biol. Crystallogr. 2004, 60, 1473–1475. [Google Scholar] [CrossRef] [PubMed]
- Riedl, P.; Wieland, A.; Lamberth, K.; Buus, S.; Lemonnier, F.; Reifenberg, K.; Reimann, J.; Schirmbeck, R. Elimination of Immunodominant Epitopes from Multispecific DNA-Based Vaccines Allows Induction of CD8 T Cells That Have a Striking Antiviral Potential. J. Immunol. 2009, 183, 370–380. [Google Scholar] [CrossRef] [PubMed]
- Pan, X.; Ding, H.; Zhou, X.; Tien, P. Identification of hepatitis B virus-specific CTL epitopes presented by HLA-A*33:03 in peripheral blood mononuclear cells from patients and transgenic mice. Biochem. Biophys. Res. Commun. 2014, 449, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Khakoo, S.I.; Ling, R.; Scott, I.; Dodi, A.I.; Harrison, T.J.; Dusheiko, G.M.; A Madrigal, J. Cytotoxic T lymphocyte responses and CTL epitope escape mutation in HBsAg, anti-HBe positive individuals. Gut 2000, 47, 137–143. [Google Scholar] [CrossRef]
- Webster, G.J.M.; Reignat, S.; Brown, D.; Ogg, G.S.; Jones, L.; Seneviratne, S.L.; Williams, R.; Dusheiko, G.; Bertoletti, A. Longitudinal Analysis of CD8+ T Cells Specific for Structural and Nonstructural Hepatitis B Virus Proteins in Patients with Chronic Hepatitis B: Implications for Immunotherapy. J. Virol. 2004, 78, 5707–5719. [Google Scholar] [CrossRef]
- Chen, A.; Wang, L.; Zhang, J.; Zou, L.; Jia, Z.; Zhou, W.; Wan, Y.; Wu, Y. H-2 Kd-Restricted Hepatitis B Virus-Derived Epitope Whose Specific CD8 + T Lymphocytes Can Produce Gamma Interferon without Cytotoxicity. J. Virol. 2005, 79, 5568–5576. [Google Scholar] [CrossRef]
- Comber, J.D.; Karabudak, A.; Shetty, V.; Testa, J.S.; Huang, X.; Philip, R. MHC Class I Presented T Cell Epitopes as Potential Antigens for Therapeutic Vaccine against HBV Chronic Infection. Hepat. Res. Treat. 2014, 2014, 860562. [Google Scholar] [CrossRef]
- Sun, L.; Zhang, Y.; Zhao, B.; Deng, M.; Liu, J.; Li, X.; Hou, J.; Gui, M.; Zhang, S.; Li, X.; et al. A new unconventional HLA-A2-restricted epitope from HBV core protein elicits antiviral cytotoxic T lymphocytes. Protein Cell 2014, 5, 317–327. [Google Scholar] [CrossRef] [PubMed]
- Missale, G.; Redeker, A.; Person, J.; Fowler, P.; Guilhot, S.; Schlicht, H.J.; Ferrari, C.; Chisari, F. HLA-A31- and HLA-Aw68-restricted cytotoxic T cell responses to a single hepatitis B virus nucleocapsid epitope during acute viral hepatitis. J. Exp. Med. 1993, 177, 751–762. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wu, Y.; Deng, M.; Xu, D.; Li, X.; Xu, Z.; Hu, J.; Zhang, H.; Liu, K.; Zhao, Y.; et al. CD8 + T-Cell Response-Associated Evolution of Hepatitis B Virus Core Protein and Disease Progress. J. Virol. 2018, 92, e02120-17. [Google Scholar] [CrossRef] [PubMed]
- Malmassari, S.; Lone, Y.C.; Zhang, M.; Transy, C.; Michel, M.-L. In vivo hierarchy of immunodominant and subdominant HLA-A*0201-restricted T-cell epitopes of HBx antigen of hepatitis B virus. Microbes Infect. 2005, 7, 626–634. [Google Scholar] [CrossRef]
- Ishioka, G.Y.; Fikes, J.; Hermanson, G.; Livingston, B.; Crimi, C.; Qin, M.; Del Guercio, M.F.; Oseroff, C.; Dahlberg, C.; Alexander, J.; et al. Utilization of MHC class I transgenic mice for development of minigene DNA vaccines encoding multiple HLA-restricted CTL epitopes. J. Immunol. 1999, 162, 3915–3925. [Google Scholar]
- Gehring, A.; Ho, Z.Z.; Tan, A.T.; Aung, M.O.; Lee, K.H.; Tan, K.C.; Lim, S.G.; Bertoletti, A. Profile of Tumor Antigen-Specific CD8 T Cells in Patients With Hepatitis B Virus-Related Hepatocellular Carcinoma. Gastroenterology 2009, 137, 682–690. [Google Scholar] [CrossRef]
- Guo, Y.J.; Zhu, Y.; Sun, S.H. Identification and functional studies of HLA-A0201 restricted CTL epitopes in the X protein of hepatitis B virus. Acta Virol. 2011, 55, 107–115. [Google Scholar] [CrossRef][Green Version]
- Ding, F.-X.; Wang, F.; Lu, Y.-M.; Li, K.; Wang, K.-H.; He, X.-W.; Sun, S.-H. Multiepitope peptide-loaded virus-like particles as a vaccine against hepatitis B virus-related hepatocellular carcinoma. Hepatology 2009, 49, 1492–1502. [Google Scholar] [CrossRef]
- King, T.H.; Kemmler, C.B.; Guo, Z.; Mann, D.; Lu, Y.; Coeshott, C.; Gehring, A.J.; Bertoletti, A.; Ho, Z.Z.; Delaney, W.; et al. A Whole Recombinant Yeast-Based Therapeutic Vaccine Elicits HBV X, S and Core Specific T Cells in Mice and Activates Human T Cells Recognizing Epitopes Linked to Viral Clearance. PLoS ONE 2014, 9, e101904. [Google Scholar] [CrossRef]
- Van Der Burg, S.H.; Visseren, M.J.; Brandt, R.M.; Kast, W.M.; Melief, C.J. Immunogenicity of peptides bound to MHC class I molecules depends on the MHC-peptide complex stability. J. Immunol. 1996, 156, 3308–3314. [Google Scholar]
- Vitiello, A.; Sette, A.; Yuan, L.; Farness, P.; Southwood, S.; Sidney, J.; Chesnut, R.W.; Grey, H.M.; Livingston, B. Comparison of cytotoxic T lymphocyte responses induced by peptide or DNA immunization: Implications on immunogenicity and immunodominance. Eur. J. Immunol. 1997, 27, 671–678. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.X.L.; Tan, A.T.; Or, M.Y.; Toh, K.Y.; Lim, P.Y.; Chia, A.S.E.; Froesig, T.M.; Nadua, K.D.; Oh, H.J.; Leong, H.N.; et al. Conditional ligands for A sian HLA variants facilitate the definition of CD8+ T-cell responses in acute and chronic viral diseases. Eur. J. Immunol. 2013, 43, 1109–1120. [Google Scholar] [CrossRef] [PubMed]
- Bertoni, R.; Sette, A.; Sidney, J.; Guidotti, L.G.; Shapiro, M.; Purcell, R.; Chisari, F. Human class I supertypes and CTL repertoires extend to chimpanzees. J. Immunol. 1998, 161, 4447–4455. [Google Scholar] [PubMed]
- Tangri, S.; Ishioka, G.Y.; Huang, X.; Sidney, J.; Southwood, S.; Fikes, J.; Sette, A. Structural Features of Peptide Analogs of Human Histocompatibility Leukocyte Antigen Class I Epitopes That Are More Potent and Immunogenic than Wild-Type Peptide. J. Exp. Med. 2001, 194, 833–846. [Google Scholar] [CrossRef] [PubMed]
- Sing, G.K.; Ladhams, A.; Arnold, S.; Parmar, H.; Chen, X.; Cooper, J.; Butterworth, L.; Stuart, K.; D’Arcy, D.; Cooksley, W.G.E. A longitudinal analysis of cytotoxic T lymphocyte precursor frequencies to the hepatitis B virus in chronically infected patients. J. Viral Hepat. 2001, 8, 19–29. [Google Scholar] [CrossRef]
- Tan, A.T.; Loggi, E.; Boni, C.; Chia, A.; Gehring, A.; Sastry, K.S.R.; Goh, V.; Fisicaro, P.; Andreone, P.; Brander, C.; et al. Host Ethnicity and Virus Genotype Shape the Hepatitis B Virus-Specific T-Cell Repertoire. J. Virol. 2008, 82, 10986–10997. [Google Scholar] [CrossRef]
- Zheng, J.; Xia, Z.; Xu, Y.; Ou, Z.; Lin, X.; Jin, S.; Liu, Y.; Wu, J. Identification of novel hepatitis B virus therapeutic vaccine candidates derived from polymerase protein. Aging 2021, 13, 14372–14384. [Google Scholar] [CrossRef] [PubMed]
- Mizukoshi, E.; Sidney, J.; Livingston, B.; Ghany, M.; Hoofnagle, J.H.; Sette, A.; Rehermann, B. Cellular Immune Responses to the Hepatitis B Virus Polymerase. J. Immunol. 2004, 173, 5863–5871. [Google Scholar] [CrossRef] [PubMed]
- Schirmbeck, R.; Riedl, P.; Fissolo, N.M.; Lemonnier, F.A.; Bertoletti, A.; Reimann, J. Translation from Cryptic Reading Frames of DNA Vaccines Generates an Extended Repertoire of Immunogenic, MHC Class I-Restricted Epitopes. J. Immunol. 2005, 174, 4647–4656. [Google Scholar] [CrossRef]
- Livingston, B.D.; Newman, M.; Crimi, C.; McKinney, D.; Chesnut, R.; Sette, A. Optimization of epitope processing enhances immunogenicity of multiepitope DNA vaccines. Vaccine 2001, 19, 4652–4660. [Google Scholar] [CrossRef]
- Lin, C.-L.; Tsai, S.-L.; Lee, T.-H.; Chien, R.-N.; Liao, S.-K.; Liaw, Y.-F. High frequency of functional anti-YMDD and -mutant cytotoxic T lymphocytes after in vitro expansion correlates with successful response to lamivudine therapy for chronic hepatitis B. Gut 2005, 54, 152–161. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Maini, M.; Boni, C.; Ogg, G.S.; King, A.S.; Reignat, S.; Lee, C.K.; Larrubia, J.R.; Webster, G.J.; McMichael, A.J.; Ferrari, C.; et al. Direct ex vivo analysis of hepatitis B virus-specific CD8+ T cells associated with the control of infection. Gastroenterology 1999, 117, 1386–1396. [Google Scholar] [CrossRef]
- Wu, Y.; Zhang, J.; Chen, S.; Chen, A.; Wang, L.; Li, J.; Zhao, T.; Zou, L.; Tang, Y.; Tingrong, L.; et al. Frequencies of epitope-specific cytotoxic T lymphocytes in active chronic viral hepatitis B infection by using MHC class I peptide tetramers. Immunol. Lett. 2004, 92, 253–258. [Google Scholar] [CrossRef] [PubMed]
- Shi, M.; Qian, S.; Chen, W.-W.; Zhang, H.; Zhang, B.; Tang, Z.-R.; Zhang, Z.; Wang, F.-S. Hepatitis B virus (HBV) antigen-pulsed monocyte-derived dendritic cells from HBV-associated hepatocellular carcinoma patients significantly enhance specific T cell responses in vitro. Clin. Exp. Immunol. 2006, 147, 277–286. [Google Scholar] [CrossRef] [PubMed]
- Rehermann, B.; Lau, D.; Hoofnagle, J.H.; Chisari, F.V. Cytotoxic T lymphocyte responsiveness after resolution of chronic hepatitis B virus infection. J. Clin. Investig. 1996, 97, 1655–1665. [Google Scholar] [CrossRef]
- Zhao, X.; Sankaran, S.; Yap, J.; Too, C.T.; Ho, Z.Z.; Dolton, G.; Legut, M.; Ren, E.C.; Sewell, A.K.; Bertoletti, A.; et al. Nonstimulatory peptide-MHC enhances human T-cell antigen-specific responses by amplifying proximal TCR signaling. Nat. Commun. 2018, 9, 2716. [Google Scholar] [CrossRef]
- Jin, Y.; Shih, W.K.; Berkower, I. Human T cell response to the surface antigen of hepatitis B virus (HBsAg). Endosomal and nonendosomal processing pathways are accessible to both endogenous and exogenous antigen. J. Exp. Med. 1988, 168, 293–306. [Google Scholar] [CrossRef] [PubMed]
- Loirat, D.; Lemonnier, F.A.; Michel, M.-L. Multiepitopic HLA-A*0201-Restricted Immune Response Against Hepatitis B Surface Antigen After DNA-Based Immunization. J. Immunol. 2000, 165, 4748–4755. [Google Scholar] [CrossRef] [PubMed]
- Loirat, D.; Mancini-Bourgine, M.; Abastado, J.; Michel, M. HBsAg/HLA-A2 transgenic mice: A model for T cell tolerance to hepatitis B surface antigen in chronic hepatitis B virus infection. Int. Immunol. 2003, 15, 1125–1136. [Google Scholar] [CrossRef]
- Vandepapelière, P.; Rehermann, B.; Koutsoukos, M.; Moris, P.; Garçon, N.; Wettendorff, M.; Leroux-Roels, G. Potent enhancement of cellular and humoral immune responses against recombinant hepatitis B antigens using AS02A adjuvant in healthy adults. Vaccine 2005, 23, 2591–2601. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.-G.; Fan, Z.-P.; Chen, W.-W.; Yang, H.-Y.; Liu, Q.-F.; Zhang, H.; Tien, P.; Wang, F.-S. A mutant HBs antigen (HBsAg)183–191 epitope elicits specific cytotoxic T lymphocytes in acute hepatitis B patients. Clin. Exp. Immunol. 2008, 151, 441–447. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Jin, B.; Zhang, J.-Y.; Xu, B.; Wang, H.; Shi, M.; Wherry, E.J.; Lau, G.K.; Wang, F.-S. Dynamic decrease in PD-1 expression correlates with HBV-specific memory CD8 T-cell development in acute self-limited hepatitis B patients. J. Hepatol. 2009, 50, 1163–1173. [Google Scholar] [CrossRef] [PubMed]
- Boni, C.; Laccabue, D.; Lampertico, P.; Giuberti, T.; Viganò, M.; Schivazappa, S.; Alfieri, A.; Pesci, M.; Gaeta, G.B.; Brancaccio, G.; et al. Restored Function of HBV-Specific T Cells After Long-term Effective Therapy With Nucleos(t)ide Analogues. Gastroenterology 2012, 143, 963–973.e9. [Google Scholar] [CrossRef] [PubMed]
- Schirmbeck, R.; Melber, K.; Mertens, T.; Reimann, J. Selective stimulation of murine cytotoxic T cell and antibody responses by particulate or monomeric hepatitis B virus surface (S) antigen. Eur. J. Immunol. 1994, 24, 1088–1096. [Google Scholar] [CrossRef] [PubMed]
- Marsac, D.; Puaux, A.-L.; Rivière, Y.; Michel, M.-L. In vivo induction of cellular and humoral immune responses by hybrid DNA vectors encoding simian/human immunodeficiency virus/hepatitis B surface antigen virus particles in BALB/c and HLA-A2-transgenic mice. Immunobiology 2005, 210, 305–319. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Tian, Y.; Li, Y.; Zhang, W.; Cai, W.; Liu, Y.; Ren, Y.; Liang, Z.; Zhou, P.; Zhang, Y.; et al. In vivo therapeutic effects of affinity-improved-TCR engineered T-cells on HBV-related hepatocellular carcinoma. J. Immunother. Cancer 2020, 8, e001748. [Google Scholar] [CrossRef] [PubMed]
- Bertoletti, A.; Sette, A.; Chisari, F.; Penna, A.; Levrero, M.; De Carli, M.; Fiaccadori, F.; Ferrari, C. Natural variants of cytotoxic epitopes are T-cell receptor antagonists for antiviral cytotoxic T cells. Nature 1994, 369, 407–410. [Google Scholar] [CrossRef]
- Roy, M.J.; Wu, M.S.; Barr, L.J.; Fuller, J.T.; Tussey, L.G.; Speller, S.; Culp, J.; Burkholder, J.K.; Swain, W.F.; Dixon, R.M.; et al. Induction of antigen-specific CD8+ T cells, T helper cells, and protective levels of antibody in humans by particle-mediated administration of a hepatitis B virus DNA vaccine. Vaccine 2000, 19, 764–778. [Google Scholar] [CrossRef]
- Riedl, P.; Bertoletti, A.; Lopes, R.; Lemonnier, F.; Reimann, J.; Schirmbeck, R. Distinct, Cross-Reactive Epitope Specificities of CD8 T Cell Responses Are Induced by Natural Hepatitis B Surface Antigen Variants of Different Hepatitis B Virus Genotypes. J. Immunol. 2006, 176, 4003–4011. [Google Scholar] [CrossRef] [PubMed]
- Feng, I.-C.; Koay, L.-B.; Sheu, M.-J.; Kuo, H.-T.; Sun, C.-S.; Lee, C.; Chuang, W.-L.; Liao, S.-K.; Wang, S.-L.; Tang, L.-Y.; et al. HBcAg-specific CD4+CD25+regulatory T cells modulate immune tolerance and acute exacerbation on the natural history of chronic hepatitis B virus infection. J. Biomed. Sci. 2007, 14, 43–57. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Luo, H.; Wan, X.; Fu, X.; Mao, Q.; Xiang, X.; Zhou, Y.; He, W.; Zhang, J.; Guo, Y.; et al. TNF-α/IFN-γ profile of HBV-specific CD4 T cells is associated with liver damage and viral clearance in chronic HBV infection. J. Hepatol. 2020, 72, 45–56. [Google Scholar] [CrossRef]
- Desombere, I.; Gijbels, Y.; Verwulgen, A.; Leroux-Roels, G. Characterization of the T cell recognition of hepatitis B surface antigen (HBsAg) by good and poor responders to hepatitis B vaccines. Clin. Exp. Immunol. 2000, 122, 390–399. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, C.; Bertoletti, A.; Penna, A.; Cavalli, A.; Valli, A.; Missale, G.; Pilli, M.; Fowler, P.; Giuberti, T.; Chisari, F. Identification of immunodominant T cell epitopes of the hepatitis B virus nucleocapsid antigen. J. Clin. Investig. 1991, 88, 214–222. [Google Scholar] [CrossRef] [PubMed]
- Cao, T.; Desombere, I.; Vanlandschoot, P.; Sällberg, M.; Leroux-Roels, G. Characterization of HLA DR13-restricted CD4+ T cell epitopes of hepatitis B core antigen associated with self-limited, acute hepatitis B. J. Gen. Virol. 2002, 83, 3023–3033. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Raziorrouh, B.; Heeg, M.; Kurktschiev, P.; Schraut, W.; Zachoval, R.; Wendtner, C.; Wächtler, M.; Spannagl, M.; Denk, G.; Ulsenheimer, A.; et al. Inhibitory Phenotype of HBV-Specific CD4+ T-Cells Is Characterized by High PD-1 Expression but Absent Coregulation of Multiple Inhibitory Molecules. PLoS ONE 2014, 9, e105703. [Google Scholar] [CrossRef]
- Cao, T.; Meuleman, P.; Desombere, I.; Sällberg, M.; Leroux-Roels, G. In Vivo Inhibition of Anti-Hepatitis B Virus Core Antigen (HBcAg) Immunoglobulin G Production by HBcAg-Specific CD4 + Th1-Type T-Cell Clones in a hu-PBL-NOD/SCID Mouse Model. J. Virol. 2001, 75, 11449–11456. [Google Scholar] [CrossRef]
- Tsai, S.-L.; Chen, P.-J.; Yang, P.-M.; Liao, T.-H.; Sung, J.-L.; Lai, M.-Y.; Huang, J.-H.; Chang, T.-H.; Chen, D.-S. Characterization of T cell clones specific to a determinant of hepatitis B virus core and e antigens in chronic type B hepatitis: Implication for a T cell mechanism of HBV immunopathogenesis. J. Biomed. Sci. 1994, 1, 105–118. [Google Scholar] [CrossRef] [PubMed]
- Malmassari, S.L.; Deng, Q.; Fontaine, H.; Houitte, D.; Rimlinger, F.; Thiers, V.; Maillere, B.; Pol, S.; Michel, M.-L. Impact of hepatitis B virus basic core promoter mutations on T cell response to an immunodominant HBx-derived epitope. Hepatology 2007, 45, 1199–1209. [Google Scholar] [CrossRef] [PubMed]
- Kruger, A.; Adams, P.; Hammer, J.; Böcher, W.O.; Schneider, P.M.; Rittner, C.; Hoehler, T. Hepatitis B surface antigen presentation and HLA-DRB1*—Lessons from twins and peptide binding studies. Clin. Exp. Immunol. 2005, 140, 325–332. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-H.; Park, J.-H.; Lee, Y.-J.; Cho, E.-W.; Bae, Y.-S.; Kim, K.L. In Vitro Binding Analysis of Hepatitis B Virus preS-derived Putative Helper T-cell Epitopes to MHC Class II Molecules Using Stable HLA-DRB1*0405/-DRA*0101 Transfected Cells. IUBMB Life 2000, 50, 379–384. [Google Scholar] [CrossRef] [PubMed]
- Barnaba, V.; Franco, A.; Alberti, A.; Balsano, C.; Benvenuto, R.; Balsano, F. Recognition of hepatitis B virus envelope proteins by liver-infiltrating T lymphocytes in chronic HBV infection. J. Immunol. 1989, 143, 2650–2655. [Google Scholar]
- Honorati, M.C.; Dolzani, P.; Mariani, E.; Piacentini, A.; Lisignoli, G.; Ferrari, C.; Facchini, A. Epitope specificity of Th0/Th2 CD4+ T-lymphocyte clones induced by vaccination with rHBsAg vaccine. Gastroenterology 1997, 112, 2017–2027. [Google Scholar] [CrossRef]
- Chow, I.-T.; James, E.A.; Tan, V.; Moustakas, A.K.; Papadopoulos, G.K.; Kwok, W.W. DRB1*12:01 presents a unique subset of epitopes by preferring aromatics in pocket. Mol. Immunol. 2012, 50, 26–34. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bayard, F.; Malmassari, S.; Deng, Q.; Lone, Y.-C.; Michel, M.-L. Hepatitis B virus (HBV)-derived DRB1*0101-restricted CD4 T-cell epitopes help in the development of HBV-specific CD8+ T cells in vivo. Vaccine 2010, 28, 3818–3826. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, Y.; Ding, Y.; Shen, C. A Systematic Review of T Cell Epitopes Defined from the Proteome of Hepatitis B Virus. Vaccines 2022, 10, 257. https://doi.org/10.3390/vaccines10020257
Wu Y, Ding Y, Shen C. A Systematic Review of T Cell Epitopes Defined from the Proteome of Hepatitis B Virus. Vaccines. 2022; 10(2):257. https://doi.org/10.3390/vaccines10020257
Chicago/Turabian StyleWu, Yandan, Yan Ding, and Chuanlai Shen. 2022. "A Systematic Review of T Cell Epitopes Defined from the Proteome of Hepatitis B Virus" Vaccines 10, no. 2: 257. https://doi.org/10.3390/vaccines10020257
APA StyleWu, Y., Ding, Y., & Shen, C. (2022). A Systematic Review of T Cell Epitopes Defined from the Proteome of Hepatitis B Virus. Vaccines, 10(2), 257. https://doi.org/10.3390/vaccines10020257








