Does Measles, Mumps, and Rubella (MMR) Vaccination Protect against COVID-19 Outcomes: A Nationwide Cohort Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Sources
2.2. Study Population
2.3. Exposure to the MMR Vaccine
2.4. Outcomes
2.4.1. Main Outcome: Hospitalization for COVID-19
2.4.2. Secondary Outcome: Hospitalization for COVID-19 or PIMS
2.5. Co-Variates
2.6. Statistical Analyses
3. Results
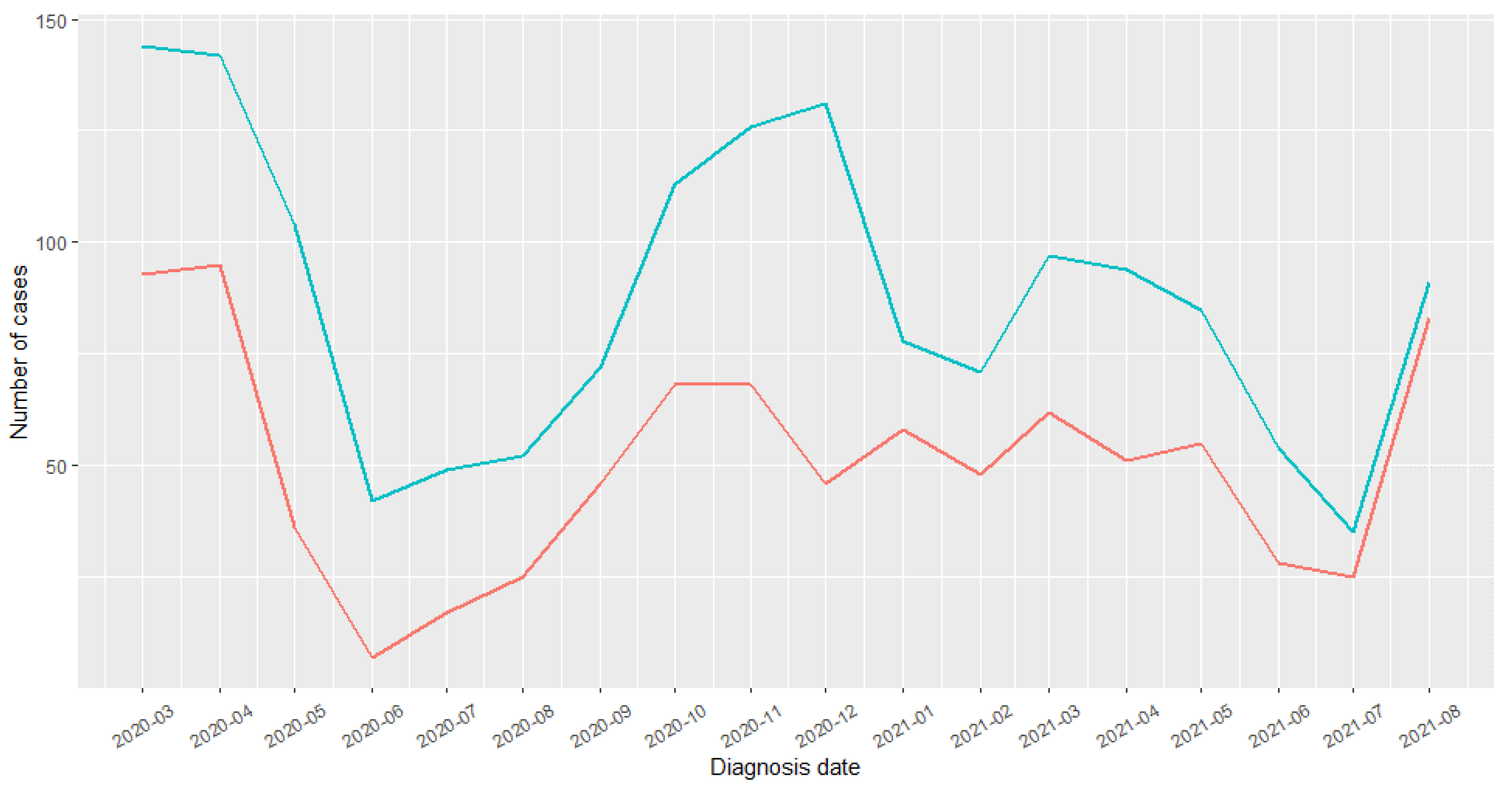
Descriptive and Analytic Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wu, P.; Hao, X.; Lau, E.H.Y.; Wong, J.Y.; Leung, K.S.M.; Wu, J.T.; Cowling, B.J.; Leung, G.M. Real-time tentative assessment of the epidemiological characteristics of novel coronavirus infections in Wuhan, China, as at 22 January 2020. Eurosurveillance 2020, 25, 2000044. [Google Scholar] [CrossRef] [PubMed]
- Semenzato, L.; Botton, J.; Drouin, J.; Cuenot, F.; Dray-Spira, R.; Weill, A.; Zureik, M. Chronic diseases, health conditions and risk of COVID-19-related hospitalization and in-hospital mortality during the first wave of the epidemic in France: A cohort study of 66 million people. Lancet Reg. Health Eur. 2021, 8, 100158. [Google Scholar] [CrossRef] [PubMed]
- Felsenstein, S.; Hedrich, C.M. SARS-CoV-2 infections in children and young people. Clin. Immunol. 2020, 220, 108588. [Google Scholar] [CrossRef] [PubMed]
- de Lusignan, S.; Dorward, J.; Correa, A.; Jones, N.; Akinyeme, O.; Amirthalingam, G.; Andrews, N.; Byford, R.; Dabrera, B.; Elliot, A.; et al. Risk factors for SARS-CoV-2 among patients in the Oxford Royal College of General Practitioners Research and Surveillance Centre primary care network: A cross-sectional study. Lancet Infect Dis. 2020, 20, 1034–1042. [Google Scholar] [CrossRef]
- Viner, R.M.; Mytton, O.T.; Bonell, C.; Melendez-Torres, G.J.; Ward, J.; Hudson, L.; Waddington, C.; Thomas, J.; Russell, S.; van der Klis, F.; et al. Susceptibility to SARS-CoV-2 Infection Among Children and Adolescents Compared With Adults:: A systematic review and meta-analysis. JAMA Pediatr. 2021, 175, 143–156. [Google Scholar] [CrossRef]
- Yonker, L.M.; Neilan, A.M.; Bartsch, Y.; Patel, A.B.; Regan, J.; Arya, P.; Gootkind, E.; Park, G.; Hardcastle, M.; St John, A.; et al. Pediatric SARS-CoV-2: Clinical Presentation, Infectivity, and Immune Responses. J. Pediatr. [CrossRef]
- Bunyavanich, S.; Do, A.; Vicencio, A. Nasal Gene Expression of Angiotensin-Converting Enzyme 2 in Children and Adults. JAMA 2020, 323, 2427–2429. [Google Scholar] [CrossRef]
- Pittet, L.F.; Messina, N.L.; Gardiner, K.; Orsini, F.; Abruzzo, V.; Bannister, S.; Bonten, M.; Campbell, J.L.; Croda, J.; Dalcolmo, M.; et al. BCG vaccination to reduce the impact of COVID-19 in healthcare workers: Protocol for a randomised controlled trial (BRACE trial). BMJ Open 2021, 11, e052101. [Google Scholar] [CrossRef]
- Gujar, N.; Tambe, M.; Parande, M.; Salunke, N.; Jagdale, G.; Anderson, S.G.; Dharmadhikari, A.; Lakhkar, A.; Kulkarni, P.S. A case control study to assess effectiveness of measles containing vaccines in preventing severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection in children. Hum. Vaccines Immunother. 2021, 17, 3316–3321. [Google Scholar] [CrossRef]
- Carryn, S.; Feyssaguet, M.; Povey, M.; Di Paolo, E. Long-term immunogenicity of measles, mumps and rubella-containing vaccines in healthy young children: A 10-year follow-up. Vaccine 2019, 37, 5323–5331. [Google Scholar] [CrossRef]
- Moorlag, S.J.; van Deuren, R.C.; van Werkhoven, C.H.; Jaeger, M.; Debisarun, P.; Taks, E.; Mourits, V.P.; Koeken, V.A.; de Bree, L.C.J.; Doesschate, T.T.; et al. Safety and COVID-19 Symptoms in Individuals Recently Vaccinated with BCG: A Retrospective Cohort Study. Cell Rep. Med. 2020, 1, 100073. [Google Scholar] [CrossRef] [PubMed]
- Anbarasu, A.; Ramaiah, S.; Livingstone, P. Vaccine repurposing approach for preventing COVID 19: Can MMR vaccines reduce morbidity and mortality? Hum. Vaccines Immunother. 2020, 16, 2217–2218. [Google Scholar] [CrossRef] [PubMed]
- Netea, M.G.; Giamarellos-Bourboulis, E.J.; Domínguez-Andrés, J.; Curtis, N.; van Crevel, R.; van de Veerdonk, F.L.; Bonten, M. Trained Immunity: A Tool for Reducing Susceptibility to and the Severity of SARS-CoV-2 Infection. Cell 2020, 181, 969–977. [Google Scholar] [CrossRef] [PubMed]
- Ashford, J.W.; Gold, J.E.; Huenergardt, M.A.; Katz, R.B.; Strand, S.E.; Bolanos, J.; Wheeler, C.J.; Perry, G.; Smith, C.J.; Steinman, L.; et al. MMR Vaccination: A Potential Strategy to Reduce Severity and Mortality of COVID-19 Illness. Am. J. Med. 2020, 134, 153–155. [Google Scholar] [CrossRef]
- Lundberg, L.; Bygdell, M.; von Feilitzen, G.S.; Woxenius, S.; Ohlsson, C.; Kindblom, J.M.; Leach, S. Recent MMR vaccination in health care workers and Covid-19: A test negative case-control study. Vaccine 2021, 39, 4414–4418. [Google Scholar] [CrossRef]
- Synthèse des couvertures vaccinales chez l’enfant de 2 ans. Available online: https://www.santepubliquefrance.fr/determinants-de-sante/vaccination/articles/synthese-des-couvertures-vaccinales-chez-l-enfant-de-2-ans (accessed on 23 February 2021).
- Tuppin, P.; Rudant, J.; Constantinou, P.; Gastaldi-Ménager, C.; Rachas, A.; de Roquefeuil, L.; Maura, G.; Caillol, H.; Tajahmady, A.; Coste, J.; et al. Value of a national administrative database to guide public decisions: From the système national d’information interrégimes de l’Assurance Maladie (SNIIRAM) to the système national des données de santé (SNDS) in France. Rev. Epidemiol. Sante Publique 2017, 65 (Suppl. S4), S149–S167. [Google Scholar] [CrossRef]
- Bezin, J.; Duong, M.; Lassalle, R.; Droz, C.; Pariente, A.; Blin, P.; Moore, N. The national healthcare system claims databases in France, SNIIRAM and EGB: Powerful tools for pharmacoepidemiology. Pharmacoepidemiol. Drug Saf. 2017, 26, 954–962. [Google Scholar] [CrossRef]
- Moulis, G.; Lapeyre-Mestre, M.; Palmaro, A.; Pugnet, G.; Montastruc, J.-L.; Sailler, L. French health insurance databases: What interest for medical research? La Rev. De Médecine Interne 2015, 36, 411–417. [Google Scholar] [CrossRef]
- Weill, A.; Dalichampt, M.; Raguideau, F.; Ricordeau, P.; Blotière, P.-O.; Rudant, J.; Alla, F.; Zureik, M. Low dose oestrogen combined oral contraception and risk of pulmonary embolism, stroke, and myocardial infarction in five million French women: Cohort study. BMJ 2016, 353, i2002. [Google Scholar] [CrossRef]
- Palmaro, A.; Moulis, G.; Despas, F.; Dupouy, J.; Lapeyre-Mestre, M. Overview of drug data within French health insurance databases and implications for pharmacoepidemiological studies. Fundam. Clin. Pharmacol. 2016, 30, 616–624. [Google Scholar] [CrossRef]
- Laanani, M.; Coste, J.; Blotière, P.-O.; Carbonnel, F.; Weill, A. Patient, Procedure, and Endoscopist Risk Factors for Perforation, Bleeding, and Splenic Injury After Colonoscopies. Clin. Gastroenterol. Hepatol. 2019, 17, 719–727.e13. [Google Scholar] [CrossRef] [PubMed]
- Tubiana, S.; Blotière, P.-O.; Hoen, B.; Lesclous, P.; Millot, S.; Rudant, J.; Weill, A.; Coste, J.; Alla, F.; Duval, X. Dental procedures, antibiotic prophylaxis, and endocarditis among people with prosthetic heart valves: Nationwide population based cohort and a case crossover study. BMJ 2017, 358, j3776. [Google Scholar] [CrossRef] [PubMed]
- Meyer, A.; Rudant, J.; Drouin, J.; Weill, A.; Carbonnel, F.; Coste, J. Effectiveness and Safety of Reference Infliximab and Biosimilar in Crohn Disease: A French Equivalence Study. Ann. Intern. Med. 2018, 170, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Penso, L.; Dray-Spira, R.; Weill, A.; Zureik, M.; Sbidian, E. Psoriasis-related treatment exposure and hospitalization or in-hospital mortality due to COVID-19 during the first and second wave of the pandemic: Cohort study of 1 326 312 patients in France. Br. J. Dermatol. 2021, 186, 59–68. [Google Scholar] [CrossRef]
- Roland, N.; Drouin, J.; Desplas, D.; Cuenot, F.; Dray-Spira, R.; Weill, A.; Zureik, M. Effects of the Coronavirus Disease 2019 (COVID-19) Lockdown on the Use of Contraceptives and Ovulation Inductors in France. Obstet. Gynecol. 2021, 137, 415–417. [Google Scholar] [CrossRef]
- Toubiana, J.; Poirault, C.; Corsia, A.; Bajolle, F.; Fourgeaud, J.; Angoulvant, F.; Debray, A.; Basmaci, R.; Salvador, E.; Biscardi, S.; et al. Kawasaki-like multisystem inflammatory syndrome in children during the covid-19 pandemic in Paris, France: Prospective observational study. BMJ 2020, 369, m2094. [Google Scholar] [CrossRef]
- Belot, A.; Antona, D.; Renolleau, S.; Javouhey, E.; Hentgen, V.; Angoulvant, F.; Delacourt, C.; Iriart, X.; Ovaert, C.; Bader-Meunier, B.; et al. SARS-CoV-2-related paediatric inflammatory multisystem syndrome, an epidemiological study, France, 1 March to 17 May 2020. Eurosurveillance 2020, 25, 2001010. [Google Scholar] [CrossRef]
- Bellino, S.; Punzo, O.; Rota, M.C.; Del Manso, M.; Urdiales, A.M.; Andrianou, X.; Fabiani, M.; Boros, S.; Vescio, F.; Riccardo, F.; et al. COVID-19 Disease Severity Risk Factors for Pediatric Patients in Italy. Pediatrics 2020, 146. [Google Scholar] [CrossRef]
- Shekerdemian, L.S.; Mahmood, N.R.; Wolfe, K.K.; Riggs, B.J.; Ross, C.E.; McKiernan, C.A.; Heidemann, S.M.; Kleinman, L.C.; Sen, A.I.; Hall, M.W.; et al. Characteristics and Outcomes of Children With Coronavirus Disease 2019 (COVID-19) Infection Admitted to US and Canadian Pediatric Intensive Care Units. JAMA Pediatr. 2020, 174, 868–873. [Google Scholar] [CrossRef]
- Rey, G.; Jougla, E.; Fouillet, A.; Hémon, D. Ecological association between a deprivation index and mortality in France over the period 1997–2001: Variations with spatial scale, degree of urbanicity, age, gender and cause of death. BMC Public Health 2009, 9, 33. [Google Scholar] [CrossRef]
- Feudtner, C.; Feinstein, J.A.; Zhong, W.; Hall, M.; Dai, D. Pediatric complex chronic conditions classification system version 2: Updated for ICD-10 and complex medical technology dependence and transplantation. BMC Pediatr. 2014, 14, 199. [Google Scholar] [CrossRef] [PubMed]
- Rachas, A.; Gastaldi-Ménager, C.; Denis, P.; Barthélémy, P.; Constantinou, P.; Drouin, J.; Lastier, D.; Lesuffleur, T.; Mette, C.; Nicolas, M.; et al. The Economic Burden of Disease in France From the National Health Insurance Perspective: The Healthcare Expenditures and Conditions Mapping Used to Prepare the French Social Security Funding Act and the Public Health Act. Med Care 2022, 60, 655–664. [Google Scholar] [CrossRef] [PubMed]
- Leslie, S.; Thiebaud, P.T.; Using Propensity Scores to Adjust For Treatment Selection Bias. Available online Published online 2007. Available online: https://support.sas.com/resources/papers/proceedings/proceedings/forum2007/184-2007.pdf (accessed on 17 August 2021).
- Gold, J.E.; Baumgartl, W.H.; Okyay, R.A.; Licht, W.E.; Fidel, P.L.; Noverr, M.C.; Tilley, L.P.; Hurley, D.J.; Rada, B.; Ashford, J.W. Analysis of Measles-Mumps-Rubella (MMR) Titers of Recovered COVID-19 Patients. mBio 2020, 11, e02628-20. [Google Scholar] [CrossRef] [PubMed]
- Götzinger, F.; Santiago-García, B.; Noguera-Julián, A.; Lanaspa, M.; Lancella, L.; Carducci, F.I.C.; Gabrovska, N.; Velizarova, S.; Prunk, P.; Osterman, V.; et al. COVID-19 in children and adolescents in Europe: A multinational, multicentre cohort study. Lancet Child Adolesc. Health 2020, 4, 653–661. [Google Scholar] [CrossRef]
- Premier bilan annuel des obligations vaccinales du nourrisson. Vie publique.fr. Available online: https://www.vie-publique.fr/rapport/274980-premier-bilan-annuel-des-obligations-vaccinales-du-nourrisson (accessed on 23 October 2021).
- Feinstein, J.A.; Hall, M.; Antoon, J.W.; Thomson, J.; Flores, J.C.; Goodman, D.M.; Cohen, E.; Azuine, R.; Agrawal, R.; Houtrow, A.J.; et al. Chronic Medication Use in Children Insured by Medicaid: A Multistate Retrospective Cohort Study. Pediatrics 2019, 143, e20183397. [Google Scholar] [CrossRef]
- Caridade, G.; Vallier, N.; Jousselme, C.; Tardieu, M.; Bouyer, J.; Weill, A.; Mikaeloff, Y. Les Maladies Chroniques Psychiatriques et Neurologiques Des Enfants et Adolescents En France: Prévalence Des Affections de Longue Durée En 2004. 2008. Available online: https://assurance-maladie.ameli.fr/etudes-et-donnees/2008-ald-psychiatriques-neurologiques-enfants-ado-2004 (accessed on 12 June 2021).
- Cortes, J.B.; Fernández, C.S.; de Oliveira, M.B.; Lagos, C.M.; Martínez, M.T.B.; Hernández, C.L.; González, I.D.C. Chronic diseases in the paediatric population: Comorbidities and use of primary care services. An. De Pediatría Engl. Ed. 2020, 93, 183–193. [Google Scholar] [CrossRef]
- DeBiasi, R.L.; Song, X.; Delaney, M.; Bell, M.; Smith, K.; Pershad, J.; Ansusinha, E.; Hahn, A.; Hamdy, R.; Harik, N.; et al. Severe Coronavirus Disease-2019 in Children and Young Adults in the Washington, DC, Metropolitan Region. J. Pediatr. 2020, 223, 199–203. [Google Scholar] [CrossRef]
- Swann, O.V.; Holden, K.A.; Turtle, L.; Pollock, L.; Fairfield, C.J.; Drake, T.M.; Seth, S.; Egan, C.; Hardwick, H.E.; Halpin, S.; et al. Clinical characteristics of children and young people admitted to hospital with covid-19 in United Kingdom: Prospective multicentre observational cohort study. BMJ 2020, 370, m3249. [Google Scholar] [CrossRef]
- Dong, Y.; Mo, X.; Hu, Y.; Qi, X.; Jiang, F.; Jiang, Z.; Tong, S. Epidemiology of COVID-19 Among Children in China. Pediatrics 2020, 145, e20200702. [Google Scholar] [CrossRef]
- Lu, X.; Zhang, L.; Du, H.; Zhang, J.; Li, Y.Y.; Qu, J.; Zhang, W.; Wang, Y.; Bao, S.; Li, Y.; et al. SARS-CoV-2 Infection in Children. N. Engl. J. Med. 2020, 382, 1663–1665. [Google Scholar] [CrossRef]
- Guan, W.J.; Ni, Z.Y.; Hu, Y.; Liang, W.H.; Qu, C.Q.; He, J.X.; Liu, L.; Shan, H.; Lei, C.L.; Hui, D.S.C.; et al. Clinical Characteristics of coronavirus disease 2019 in China. N. Engl. J. Med. 2020, 382, 1708–1720. [Google Scholar] [CrossRef] [PubMed]
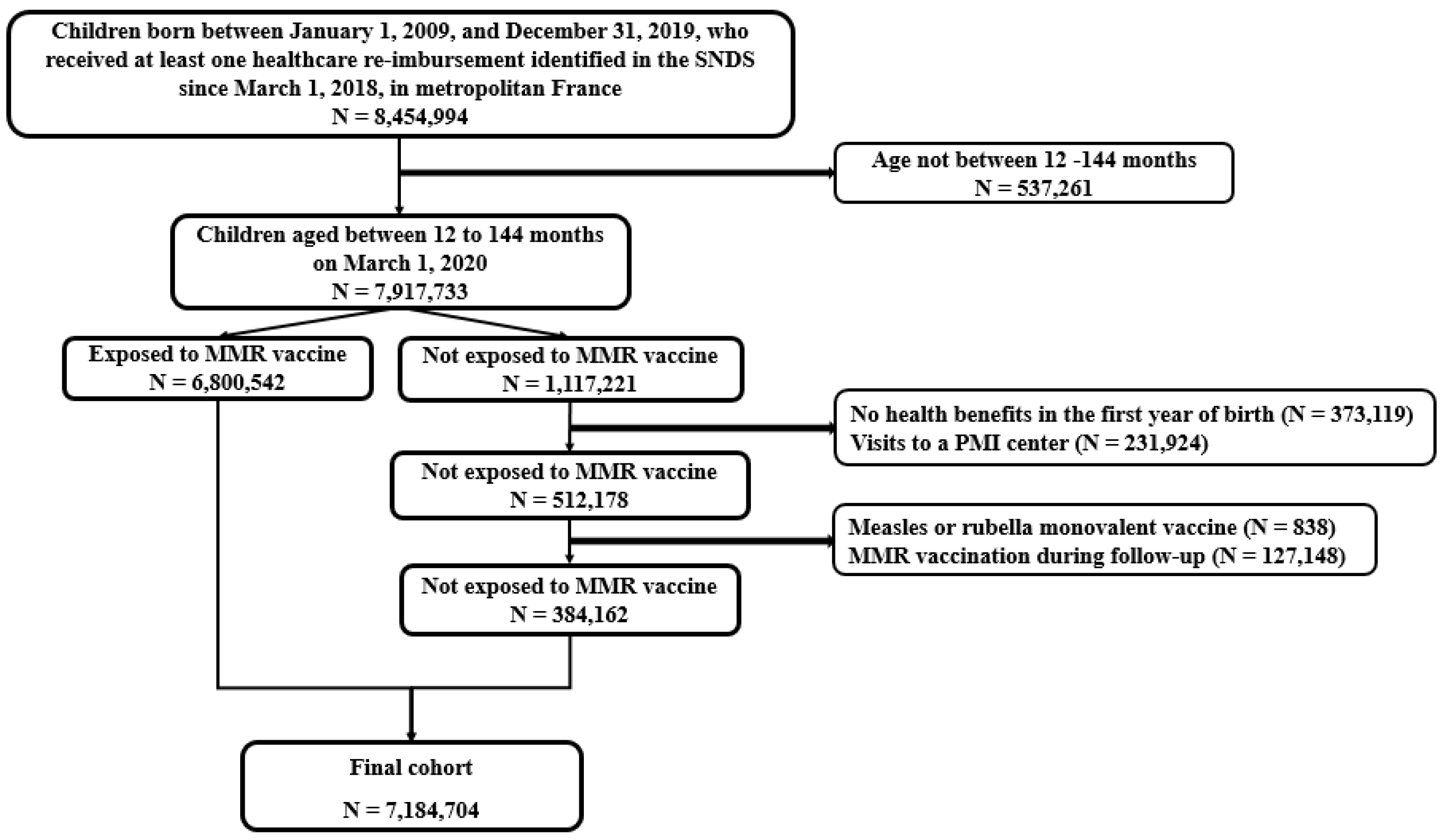
| Exposed to MMR Vaccine N = 6,800,542 | Not Exposed to MMR Vaccine N = 384,162 | Whole Population N = 7,184,704 | |
|---|---|---|---|
| Age (months)—Median (IIQ) | 75 [45–105] | 81 [47–111] | 75 [45–105] |
| Age (years)—Median (IIQ) | 6 [3–8] | 6 [3–9] | 6 [3–8] |
| Age—n (%) | |||
| 1–4 | 2,510,919 (37.0) | 125,755 (32.7) | 2,636,674 (36.7) |
| 5–9 | 3,443,691 (50.6) | 188,939 (49.2) | 3,632,630 (50.6) |
| 10–12 | 845,932 (12.4) | 69,468 (18.1) | 915,400 (12.7) |
| Female sex—n (%) | 3,316,762 (48.8) | 189,234 (49.2) | 3,505,996 (48.8) |
| CMU beneficiaries—n (%) | 1,077,298 (15.8) | 65,721 (17.1) | 1,143,019 (15.9) |
| Region—n (%) | |||
| Île-de-France | 1,338,383 (19.7) | 75,330 (19.6) | 1,413,713 (19.7) |
| Northwest | 1,377,208 (20.3) | 67,072 (17.5) | 1,444,280 (20.1) |
| Northeast | 1,524,092 (22.4) | 67,977 (17.7) | 1,592,069 (22.2) |
| Southeast | 1,686,970 (24.8) | 118,071 (30.7) | 1,805,041 (25.1) |
| Southwest | 851,461 (12.5) | 53,981 (14.1) | 905,442 (12.6) |
| Unknown | 22,428 (0.3) | 1731 (0.4) | 24,159 (0.3) |
| Social deprivation index (quintiles)—n (%) | |||
| 1 (the least deprivation) | 1,362,638 (20.0) | 72,457 (18.9) | 1,435,095 (20.0) |
| 2 | 1,376,843 (20.2) | 76,104 (19.8) | 1,452,947 (20.2) |
| 3 | 1,323,056 (19.5) | 78,690 (20.4) | 1,401,396 (19.5) |
| 4 | 1,291,105 (19.0) | 78,690 (20.5) | 1,369,795 (19.1) |
| 5 (the most deprivation) | 1,320,012 (19.4) | 73,171 (19.0) | 1,393,183 (19.4) |
| Unknown | 126,888 (1.9) | 5400 (1.4) | 132,288 (1.8) |
| Exposed to other vaccines—n (%) | 6,752,998 (99.3) | 313,940 (81.7) | 7,066,938 (98.4) |
| Number of previous consultations with a GP or pediatrician—n (%) | |||
| No consultation | 447,194 (6.6) | 112,181 (29.2) | 559,375 (7.8) |
| 1–3 | 1,345,416 (19.8) | 105,901 (27.6) | 1,451,317 (20.2) |
| 4–7 | 1,913,406 (28.1) | 86,873 (22.6) | 2,000,279 (27.8) |
| 8–12 | 1,512,152 (22.2) | 48,526 (12.6) | 1,560,678 (21.7) |
| 13 or more | 1,582,374 (23.3) | 30,681 (8.0) | 1,613,055 (22.5) |
| At least one hospitalization—n (%) | 316,120 (4.6) | 14,054 (3.6) | 330,174 (4.6) |
| Co-morbidities—n (%) | |||
| Neurological/neuromuscular | 52,843 (0.8) | 2889 (0.7) | 55,732 (0.8) |
| Respiratory | 562,916 (8.3) | 18,268 (4.7) | 581,184 (8.1) |
| Cardio-vascular | 220,806 (0.3) | 995 (0.2) | 21,801 (0.3) |
| Psychiatric | 87,895 (1.3) | 4340 (1.1) | 92,235 (1.3) |
| Metabolic | 13,897 (0.2) | 800 (0.2) | 14,697 (0.2) |
| Gastro-intestinal | 8169 (0.1) | 528 (0.1) | 8697 (0.1) |
| Renal/urological | 12,985 (0.2) | 539 (0.1) | 13,524 (0.2) |
| Hematological/immunological | 14,454 (0.2) | 819 (0.2) | 15,273 (0.2) |
| Genetic/congenital defect | 19,376 (0.3) | 976 (0.2) | 20,352 (0.3) |
| Cancer | 5007 (0.1) | 301 (0.1) | 5308 (0.1) |
| COVID-19 | COVID-19 or PIMS | |
|---|---|---|
| No. of events/no. of individuals | 911/7,184,704 | 1580/7,184,704 |
| Crude analysis—OR (95%CI) | 1.29 [0.93–1.79] | 1.16 [0.92–1.47] |
| Multi-variate logistic regression model *—aOR (95%CI) | 1.19 [0.85–1.65] | 1.05 [0.83–1.34] |
| Propensity score approach | ||
| Inverse probability of treatment weighting (IPTW)—aOR (95%CI) | 1.09 [0.81–1.48] | 1.03 [0.82–1.29] |
| No. of Events/No of Individuals | aOR (95%CI) * | |
|---|---|---|
| Sensitivity analysis | ||
| Exposed to MMR vaccine before 1 January 2018 | 626/5,659,103 | 1.10 [0.79–1.54] |
| Exposed to at least two doses of MMR vaccine | 717/6,034,653 | 1.07 [0.78–1.46] |
| Exposed to up to two doses of MMR vaccine | 623/5,447,486 | 1.06 [0.77–1.46] |
| Exclusion of children aged one year | 776/6,617,726 | 0.98 [0.72–1.35] |
| Exclusion of children with at least one comorbidity | 614/6,414,384 | 1.30 [0.88–1.93] |
| Stratified analysis | ||
| Stratification by sex | ||
| Male | 501/3,678,708 | 0.96 [0.65–1.42] |
| Female | 410/3,505,996 | 1.31 [0.80–2.14] |
| Stratification by age | ||
| 1 to 4 years | 397/2,636,674 | 1.03 [0.64–1.66] |
| 5 to 9 years | 399/3,632,630 | 1.38 [0.82–2.31] |
| 10 to 12 years | 115/915,400 | 1.11 [0.54–2.29] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kolla, E.; Weill, A.; Desplas, D.; Semenzato, L.; Zureik, M.; Grimaldi, L. Does Measles, Mumps, and Rubella (MMR) Vaccination Protect against COVID-19 Outcomes: A Nationwide Cohort Study. Vaccines 2022, 10, 1938. https://doi.org/10.3390/vaccines10111938
Kolla E, Weill A, Desplas D, Semenzato L, Zureik M, Grimaldi L. Does Measles, Mumps, and Rubella (MMR) Vaccination Protect against COVID-19 Outcomes: A Nationwide Cohort Study. Vaccines. 2022; 10(11):1938. https://doi.org/10.3390/vaccines10111938
Chicago/Turabian StyleKolla, Epiphane, Alain Weill, David Desplas, Laura Semenzato, Mahmoud Zureik, and Lamiae Grimaldi. 2022. "Does Measles, Mumps, and Rubella (MMR) Vaccination Protect against COVID-19 Outcomes: A Nationwide Cohort Study" Vaccines 10, no. 11: 1938. https://doi.org/10.3390/vaccines10111938
APA StyleKolla, E., Weill, A., Desplas, D., Semenzato, L., Zureik, M., & Grimaldi, L. (2022). Does Measles, Mumps, and Rubella (MMR) Vaccination Protect against COVID-19 Outcomes: A Nationwide Cohort Study. Vaccines, 10(11), 1938. https://doi.org/10.3390/vaccines10111938






