IgG and IgA Antibodies Post SARS-CoV-2 Vaccine in the Breast Milk and Sera of Breastfeeding Women
Abstract
:1. Introduction
2. Materials and Methods
2.1. Data Collection Method
2.2. Statistical Analysis
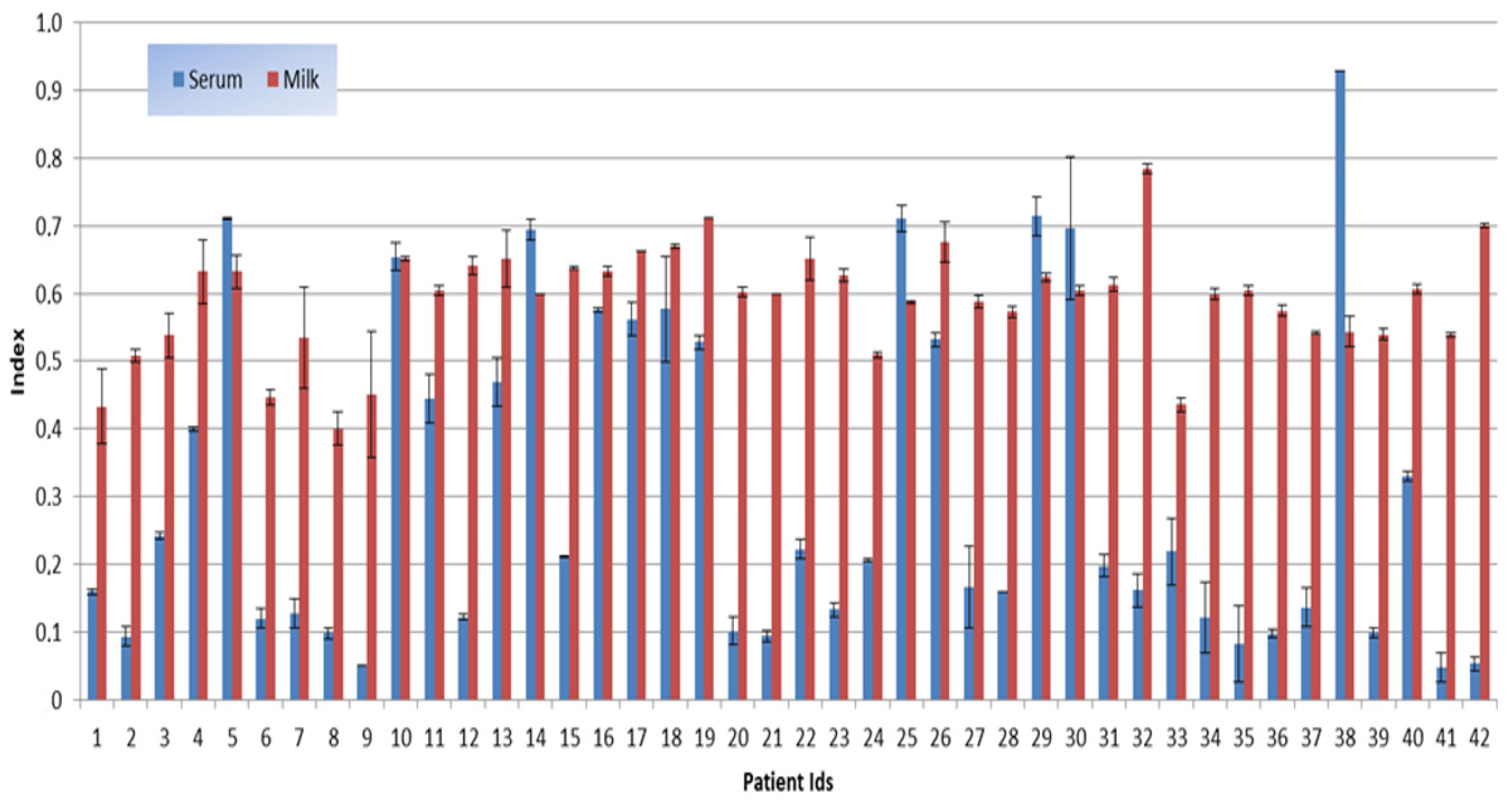
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hodgson, S.H.; Mansatta, K.; Mallett, G.; Harris, V.; Emary, K.R.W.; Pollard, A.J. What defines an efficacious COVID-19 vaccine? A review of the challenges assessing the clinical efficacy of vaccines against SARS-CoV-2. Lancet Infect. Dis. 2020, 21, e26–e35. [Google Scholar] [CrossRef]
- Sutton, D.; D’Alton, M.; Zhang, Y.; Kahe, K.; Cepin, A.; Goffman, D.; Staniczenko, A.; Yates, H.; Burgansky, A.; Coletta, J.; et al. COVID-19 vaccine acceptance among pregnant, breastfeeding, and nonpregnant reproductive-aged women. Am. J. Obstet. Gynecol. MFM 2021, 3, 100403. [Google Scholar] [CrossRef] [PubMed]
- HBelisa SARS-CoV-2 Neutralizing Antibody Detection Kit. Available online: http://hbhealthcare.net/?page_id=525&lang=en (accessed on 14 July 2021).
- Oddy, W.H. Breastfeeding protects against illness and infection in infants and children: A review of the evidence. Breastfeed. Rev. Prof. Publ. Nurs. Mothers’ Assoc. Aust. 2001, 9, 11–18. [Google Scholar]
- Rad, H.S.; Röhl, J.; Stylianou, N.; Allenby, M.C.; Bazaz, S.R.; Warkiani, M.E.; Guimaraes, F.S.F.; Clifton, V.L.; Kulasinghe, A. The Effects of COVID-19 on the Placenta During Pregnancy. Front. Immunol. 2021, 12. [Google Scholar] [CrossRef] [PubMed]
- Ballard, O.; Morrow, A.L. Human milk composition: Nutrients and bioactive factors. Pediatr. Clin. N. Am. 2013, 60, 49–74. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sterlin, D.; Mathian, A.; Miyara, M.; Mohr, A.; Anna, F.; Claër, L.; Quentric, P.; Fadlallah, J.; Devilliers, H.; Ghillani, P.; et al. IgA dominates the early neutralizing antibody response to SARS-CoV-2. Sci. Transl. Med. 2021, 13. [Google Scholar] [CrossRef]
- Wisnewski, A.V.; Campillo Luna, J.; Redlich, C.A. Human IgG and IgA responses to COVID-19 mRNA vaccines. PLoS ONE 2021, 16, e0249499. [Google Scholar] [CrossRef]
- Juncker, H.G.; Romijn, M.; Loth, V.N.; Caniels, T.G.; de Groot, C.J.; Pajkrt, D.; van Gils, M.J.; van Goudoever, J.B.; van Keulen, B.J. Human Milk Antibodies Against SARS-CoV-2: A Longitudinal Follow-Up Study. J. Hum. Lact. 2021, 37, 485–491. [Google Scholar] [CrossRef]
- Pullen, K.M.; Atyeo, C.; Collier, A.-R.Y.; Gray, K.J.; Belfort, M.B.; Lauffenburger, D.A.; Edlow, A.G.; Alter, G. Selective functional antibody transfer into the breastmilk after SARS-CoV-2 infection. Cell Rep. 2021. [Google Scholar] [CrossRef]
- Juncker, H.G.; Mulleners, S.J.; van Gils, M.J.; de Groot, C.J.M.; Pajkrt, D.; Korosi, A.; van Goudoever, J.B.; van Keulen, B.J. The Levels of SARS-CoV-2 Specific Antibodies in Human Milk Following Vaccination. J. Hum. Lact. 2021, 37, 477–484. [Google Scholar] [CrossRef]
- Perl, S.H.; Uzan-Yulzari, A.; Klainer, H.; Asiskovich, L.; Youngster, M.; Rinott, E.; Youngster, I. SARS-CoV-2–Specific Antibodies in Breast Milk After COVID-19 Vaccination of Breastfeeding Women. JAMA 2021, 325, 2013. [Google Scholar] [CrossRef]
- Schlaudecker, E.; Steinhoff, M.C.; Omer, S.B.; McNeal, M.M.; Roy, E.; Arifeen, S.; Dodd, C.N.; Raqib, R.; Breiman, R.F.; Zaman, K. IgA and Neutralizing Antibodies to Influenza A Virus in Human Milk: A Randomized Trial of Antenatal Influenza Immunization. PLoS ONE 2013, 8, e70867. [Google Scholar] [CrossRef]
- Gray, K.J.; Bordt, E.A.; Atyeo, C.; Deriso, E.; Akinwunmi, B.; Young, N.; Baez, A.M.; Shook, L.L.; Cvrk, D.; James, K.; et al. Coronavirus disease 2019 vaccine response in pregnant and lactating women: A cohort study. Am. J. Obstet. Gynecol. 2021, 225, 303.e1–303.e17. [Google Scholar] [CrossRef]
- Young, B.E.; Seppo, A.E.; Diaz, N.; Rosen-Carole, C.; Nowak-Wegrzyn, A.; Vasquez, J.M.C.; Ferri-Huerta, R.; Nguyen-Contant, P.; Fitzgerald, T.; Sangster, M.Y.; et al. Association of Human Milk Antibody Induction, Persistence, and Neutralizing Capacity With SARS-CoV-2 Infection vs mRNA Vaccination. JAMA Pediatr. 2021. [Google Scholar] [CrossRef]
- Charepe, N.; Gonçalves, J.; Juliano, A.M.; Lopes, D.G.; Canhão, H.; Soares, H.; Serrano, E.F. COVID-19 mRNA vaccine and antibody response in lactating women: A prospective cohort study. BMC Pregnancy Childbirth 2021, 21, 1–9. [Google Scholar] [CrossRef]
- Czosnykowska-Łukacka, M.; Lis-Kuberka, J.; Królak-Olejnik, B.; Orczyk-Pawiłowicz, M. Changes in Human Milk Immunoglobulin Profile During Prolonged Lactation. Front. Pediatr. 2020, 8, 428. [Google Scholar] [CrossRef] [PubMed]
- Esteve-Palau, E.; Gonzalez-Cuevas, A.; Guerrero, M.E.; Garcia-Terol, C.; Alvarez, M.C.; Casadevall, D.; Diaz-Brito, V. Quantification of Specific Antibodies Against SARS-CoV-2 in Breast Milk of Lactating Women Vaccinated With an mRNA Vaccine. JAMA Netw. Open 2021, 4, e2120575. [Google Scholar] [CrossRef] [PubMed]
- Jakuszko, K.; Kościelska-Kasprzak, K.; Żabińska, M.; Bartoszek, D.; Poznański, P.; Rukasz, D.; Kłak, R.; Królak-Olejnik, B.; Krajewska, M. Immune Response to Vaccination against COVID-19 in Breastfeeding Health Workers. Vaccines 2021, 9, 663. [Google Scholar] [CrossRef]
- Mazur, N.; Horsley, N.M.; A Englund, J.; Nederend, M.; Magaret, A.; Kumar, A.; Jacobino, S.R.; de Haan, C.; Khatry, S.K.; LeClerq, S.C.; et al. Breast Milk Prefusion F Immunoglobulin G as a Correlate of Protection Against Respiratory Syncytial Virus Acute Respiratory Illness. J. Infect. Dis. 2018, 219, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Guida, M.; Terracciano, D.; Cennamo, M.; Aiello, F.; La Civita, E.; Esposito, G.; Gargiulo, V.; Maruotti, G.; Portella, G.; Sarno, L. COVID-19 Vaccine mRNABNT162b2 Elicits Human Antibody Response in Milk of Breastfeeding Women. Vaccines 2021, 9, 785. [Google Scholar] [CrossRef] [PubMed]
- Academy of Breastfeeding Medicine (ABM). ABM Statement. Consideration for COVID-19 Vaccination in Lactation. Available online: www.bfmed.org/abm-statement-considerations-for-covid-19-vaccination-in-lactation (accessed on 14 December 2020).
- ACOG. COVID-19 Vaccination Considerations for Obstetric-Gynecologic Care. Available online: www.acog.org/clinical/clinical-guidance/practice-advisory/articles/2020/12/covid-19-vaccination-considerations-for-obstetric-gynecologic-care (accessed on 9 June 2021).
- Davanzo, R.; Agosti, M.; Cetin, I.; Chiantera, A.; Corsello, G.; Ramenghi, L.A.; Staiano, A.; Tavio, M.; Villani, A.; Viora, E.; et al. Breastfeeding and COVID-19 vaccination: Position statement of the Italian scientific societies. Ital. J. Pediatr. 2021, 47, 1–4. [Google Scholar] [CrossRef] [PubMed]
- WHO. COVID-19 Advice for the Public: Getting Vaccinated. Available online: www.who.int/emergencies/diseases/novel-coronavirus-2019/covid-19-vaccines/advice (accessed on 14 July 2021).
- COVID-19: Pregnancy, Childbirth and Breastfeeding. Available online: www.epicentro.iss.it/coronavirus/sars-cov-2-gravidanza-parto-allattamentostudio-prospettico-itoss (accessed on 14 July 2021).
- CDC V-Safe COVID-19 Vaccine Pregnancy Registry Updated. Available online: www.cdc.gov/coronavirus/2019-ncov/vaccines/safety/vsafepregnancyregistry.html (accessed on 25 August 2021).
- Collier, A.-R.Y.; McMahan, K.; Yu, J.; Tostanoski, L.H.; Aguayo, R.; Ansel, J.; Chandrashekar, A.; Patel, S.; Bondzie, E.A.; Sellers, D.; et al. Immunogenicity of COVID-19 mRNA Vaccines in Pregnant and Lactating Women. JAMA 2021, 325, 2370. [Google Scholar] [CrossRef] [PubMed]
- Drugs and Lactation Database (LactMed) [Internet]; National Library of Medicine (US): Bethesda, MD, USA, 2006; COVID-19 Vaccines. Available online: https://www.ncbi.nlm.nih.gov/books/NBK565969/ (accessed on 19 July 2021).
| Variable | n = 42 |
|---|---|
| Age, Median (IQR) | 34.5 (33.0–39.0) |
| Education, n (%) | |
| High school diploma | 4 (9.5) |
| Degree | 33 (78.6) |
| Postgraduate | 1 (2.4) |
| PhD | 4 (9.5) |
| Occupation, n (%) | |
| Medical doctor | 16 (38.0) |
| Nurse-midwife | 8 (19.1) |
| Teacher | 8 (19.1) |
| Other | 10 (23.8) |
| Comorbidity, n (%) | 7 (16.7) |
| Basedow disease | 1 (2.4) |
| Behcet disease | 1 (2.4) |
| Hypothyroidism | 2 (4.8) |
| Hashimoto’s thyroiditis | 3 (7.2) |
| Drugs in the last 10 days, n (%) | 6 (14.3) |
| Antihistamines | 2 (4.8) |
| Anti-inflammatory | 1 (2.4) |
| Other | 3 (7.2) |
| Previous COVID-19 infection, n (%) | 2 (4.8) |
| Vaccine type | |
| Pfizer/BioNtech | 35 (83.3) |
| Astrazeneca | 4 (9.5) |
| Moderna | 2 (4.8) |
| Mixed | 1 (2.4) |
| Days between collection and the 2nd dose of vaccine, median (IQR) | 22 (16–46) |
| At least one adverse event, n (%) | 20 (47.6) |
| Injection site pain | 11 (26.2) |
| Headache | 4 (9.5) |
| Pyrexia | 9 (45.0) |
| Myalgia | 9 (45.0) |
| Ostealgia | 2 (4.8) |
| Arthralgia | 2 (4.8) |
| Allergic reaction | 0 (0.0) |
| Asthenia | 12 (28.6) |
| Chills | 3 (7.2) |
| Vaccine influence on breastfeeding, n (%) | 8 (19.1) |
| Number of milk feed, median (IQR) | 5 (4–7) |
| OD milk, median (IQR) | 0.8 (0.7–1.2) |
| OD serum, median (IQR) | 0.3 (0.2–0.8) |
| Index milk, median (IQR) | 0.6 (0.5–0.6) |
| Index serum, median (IQR) | 0.2 (0.1–0.5) |
| New-born age in months, median (IQR) | 11.5 (8–17) |
| Days between delivery and vaccine, median (IQR) | 319.5 (111.0–486.0) |
| Adverse events in the child within 7 days after the vaccine | |
| Restlessness | 1 (2.4) |
| Cold | 1 (2.4) |
| Skin rash | 1 (2.4) |
| Infant’s attendance of kindergarten, n (%) | 7 (16.7) |
| N | Min | 25% | Median | 75% | Max | Wilcoxon Rank Sign Test p-Value | |
|---|---|---|---|---|---|---|---|
| OD milk | 42 | 0.37 | 0.67 | 0.81 | 1.20 | 1.60 | |
| OD serum | 42 | 0.05 | 0.15 | 0.27 | 0.78 | 1.68 | |
| Diff OD | 42 | −1.26 | −0.71 | −0.46 | −0.20 | 0.58 | <0.0001 |
| Index milk | 42 | 0.40 | 0.54 | 0.60 | 0.64 | 0.78 | |
| Index serum | 42 | 0.05 | 0.12 | 0.20 | 0.53 | 0.93 | |
| Diff Index | 42 | −0.65 | −0.44 | −0.32 | −0.14 | 0.39 | <0.0001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Scrimin, F.; Campisciano, G.; Comar, M.; Ragazzon, C.; Davanzo, R.; Quadrifoglio, M.; Giangreco, M.; Stabile, G.; Ricci, G. IgG and IgA Antibodies Post SARS-CoV-2 Vaccine in the Breast Milk and Sera of Breastfeeding Women. Vaccines 2022, 10, 125. https://doi.org/10.3390/vaccines10010125
Scrimin F, Campisciano G, Comar M, Ragazzon C, Davanzo R, Quadrifoglio M, Giangreco M, Stabile G, Ricci G. IgG and IgA Antibodies Post SARS-CoV-2 Vaccine in the Breast Milk and Sera of Breastfeeding Women. Vaccines. 2022; 10(1):125. https://doi.org/10.3390/vaccines10010125
Chicago/Turabian StyleScrimin, Federica, Giuseppina Campisciano, Manola Comar, Chiara Ragazzon, Riccardo Davanzo, Mariachiara Quadrifoglio, Manuela Giangreco, Guglielmo Stabile, and Giuseppe Ricci. 2022. "IgG and IgA Antibodies Post SARS-CoV-2 Vaccine in the Breast Milk and Sera of Breastfeeding Women" Vaccines 10, no. 1: 125. https://doi.org/10.3390/vaccines10010125
APA StyleScrimin, F., Campisciano, G., Comar, M., Ragazzon, C., Davanzo, R., Quadrifoglio, M., Giangreco, M., Stabile, G., & Ricci, G. (2022). IgG and IgA Antibodies Post SARS-CoV-2 Vaccine in the Breast Milk and Sera of Breastfeeding Women. Vaccines, 10(1), 125. https://doi.org/10.3390/vaccines10010125








