Neuroprotective Activity of Polyphenol-Rich Ribes diacanthum Pall against Oxidative Stress in Glutamate-Stimulated HT-22 Cells and a Scopolamine-Induced Amnesia Animal Model
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
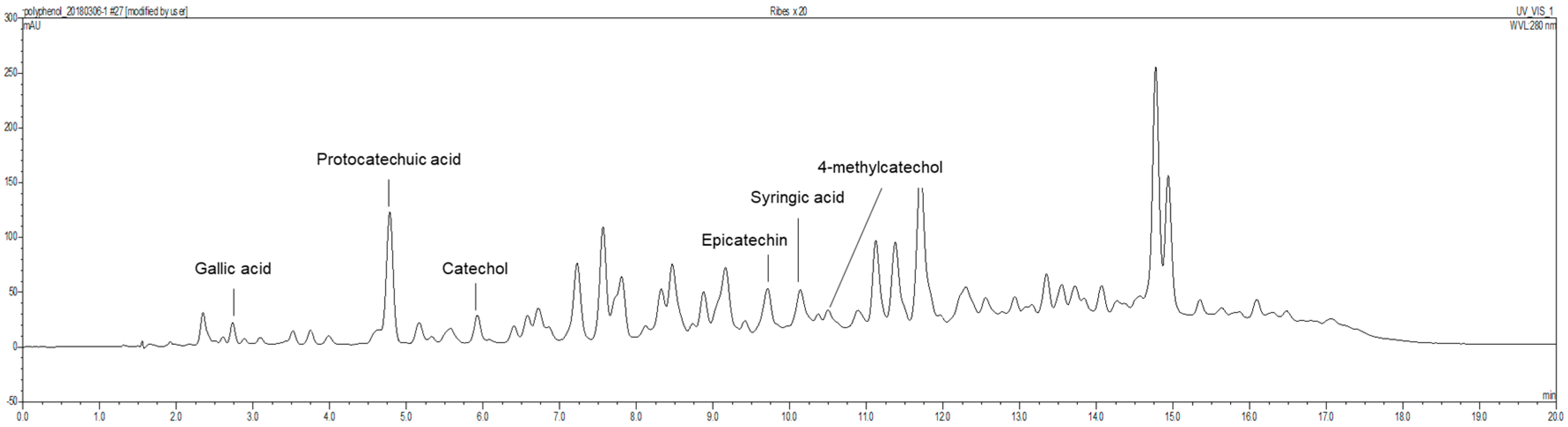
2.2. Preparation of Ribes diacanthum Pall (RDP) Extract
2.3. Cell Culture
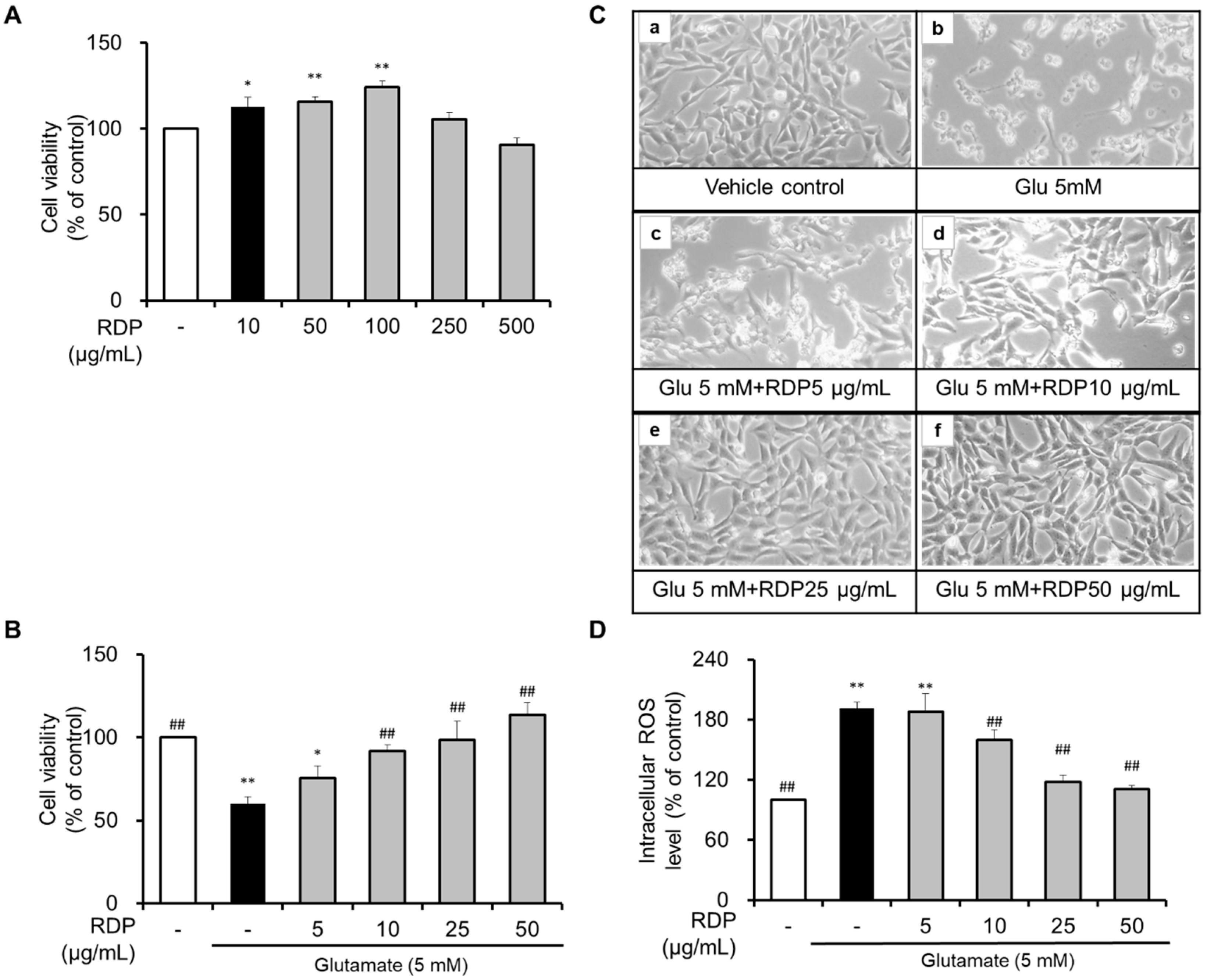
2.4. Measurement of Cell Viability
2.5. Measurement of Intracellular ROS Levels
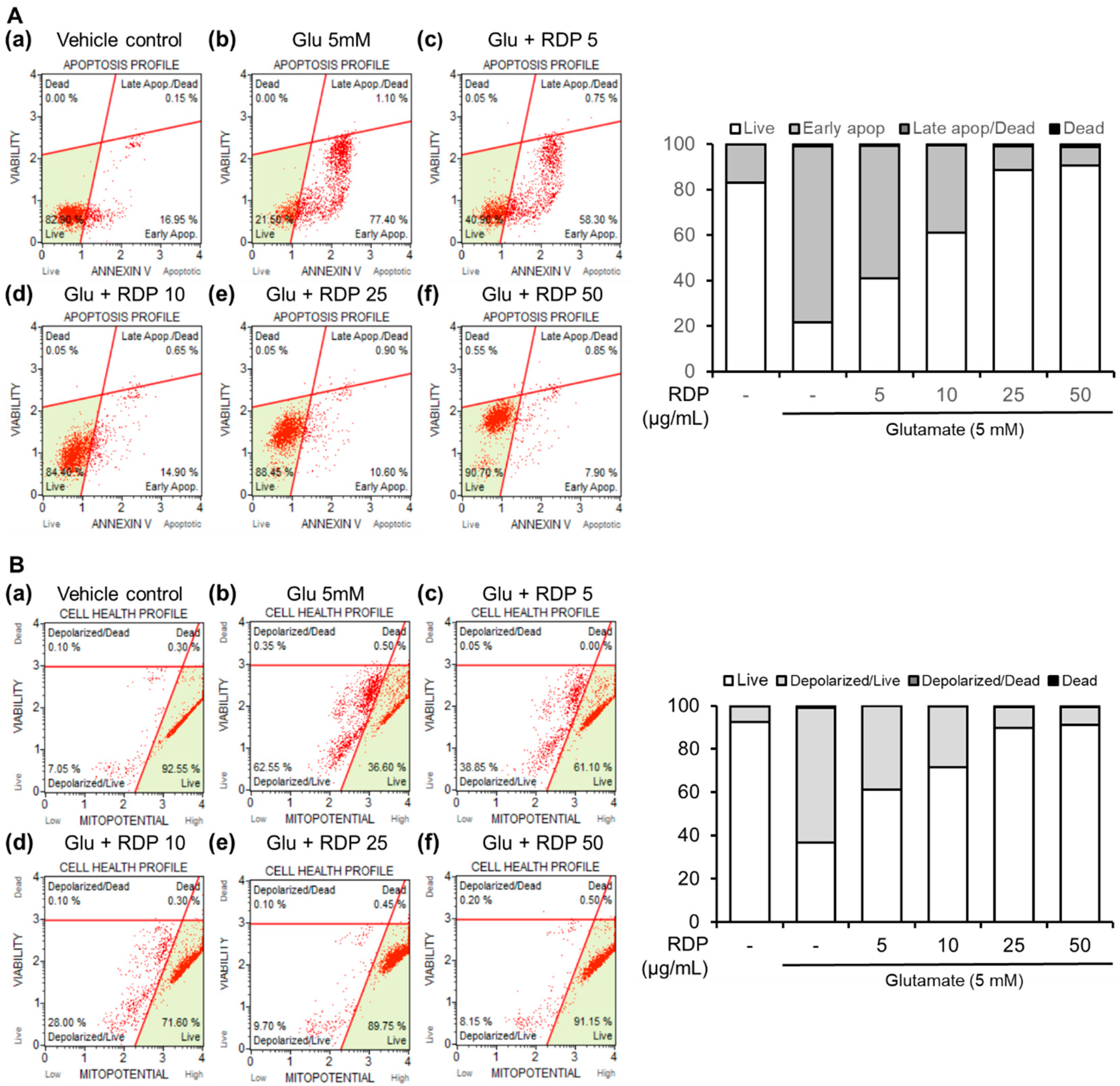
2.6. Cell Death Assessment by Flow Cytometry
2.7. Measurement of Total GSH (Glutathione) Content
2.8. Nuclear and Cytosolic Protein Extraction
2.9. Western Blot Analyses
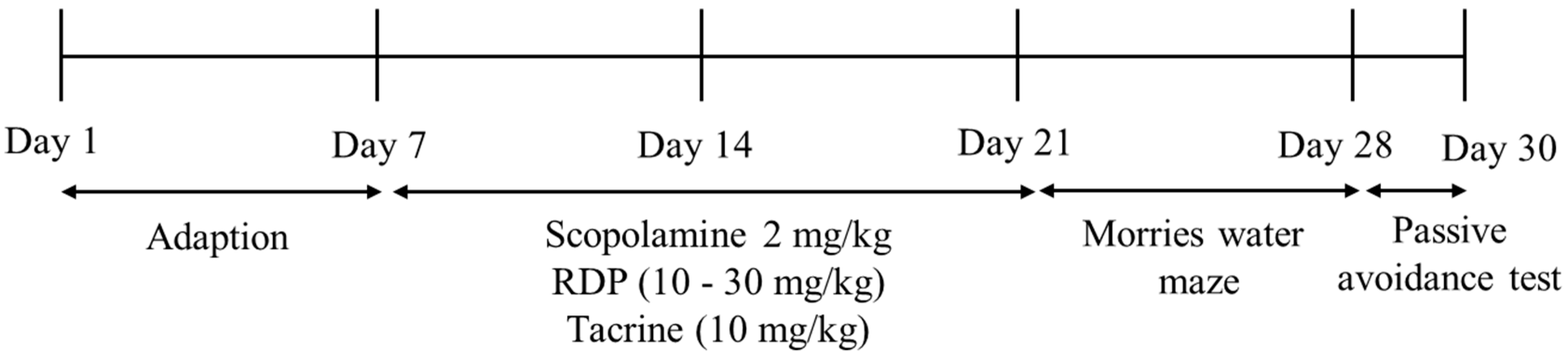
2.10. Animals
2.11. Morris Water Maze Test
2.12. Passive Avoidance Test
2.13. Statistical Analyses
3. Results
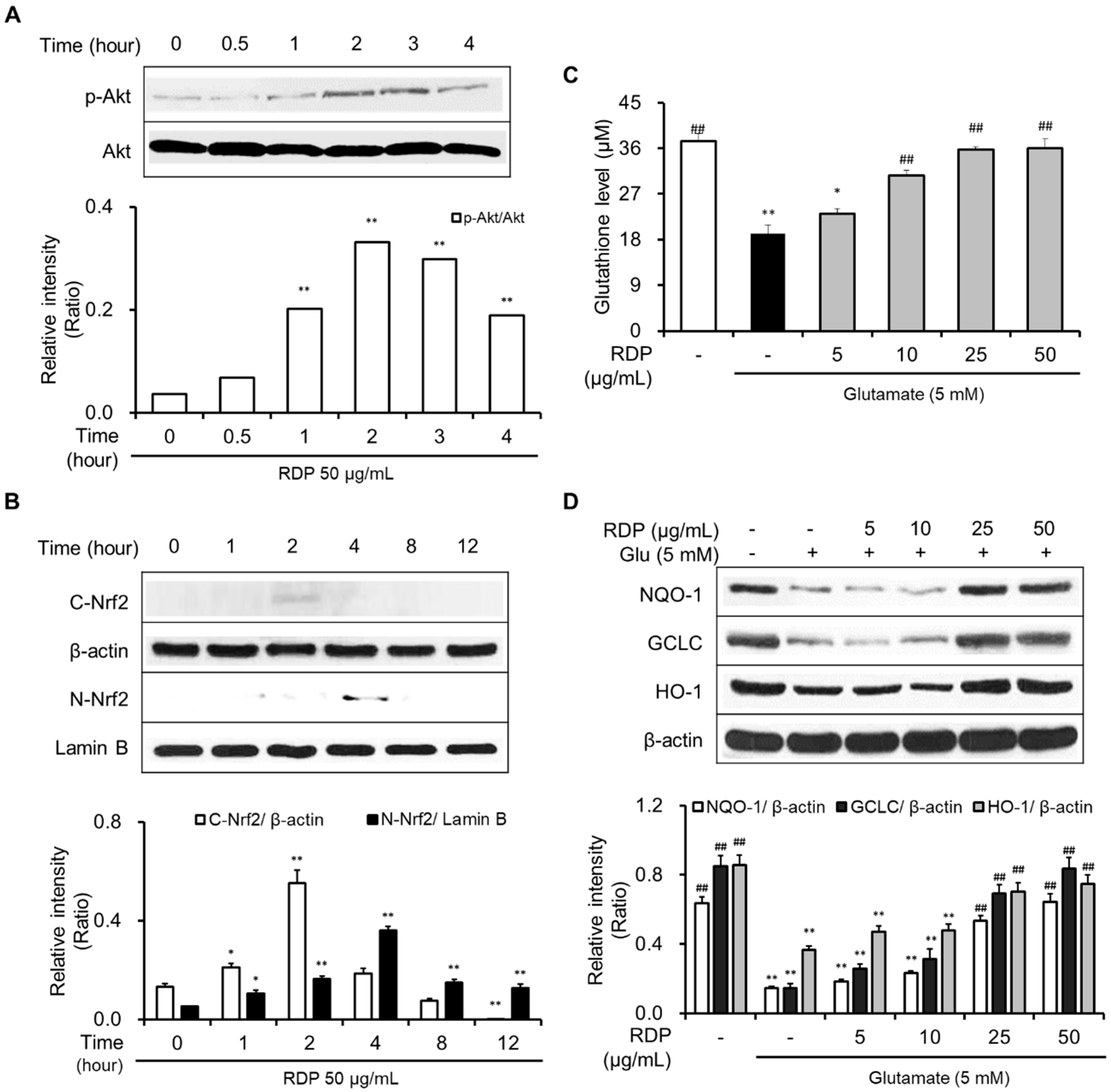
3.1. Neuroprotective Effect of RDP on Glutamate-Induced Oxidative Stress in HT-22 Cells
3.2. Preventative Effect of RDP on Apoptosis in HT-22 Cells by Flow Cytometry
3.3. Inhibitory Effect of RDP on Mitochondrial Dysfunction in HT-22 Cells
3.4. Anti-Apoptotic Effect of RDP on Oxidative Stress-Induced Apoptosis
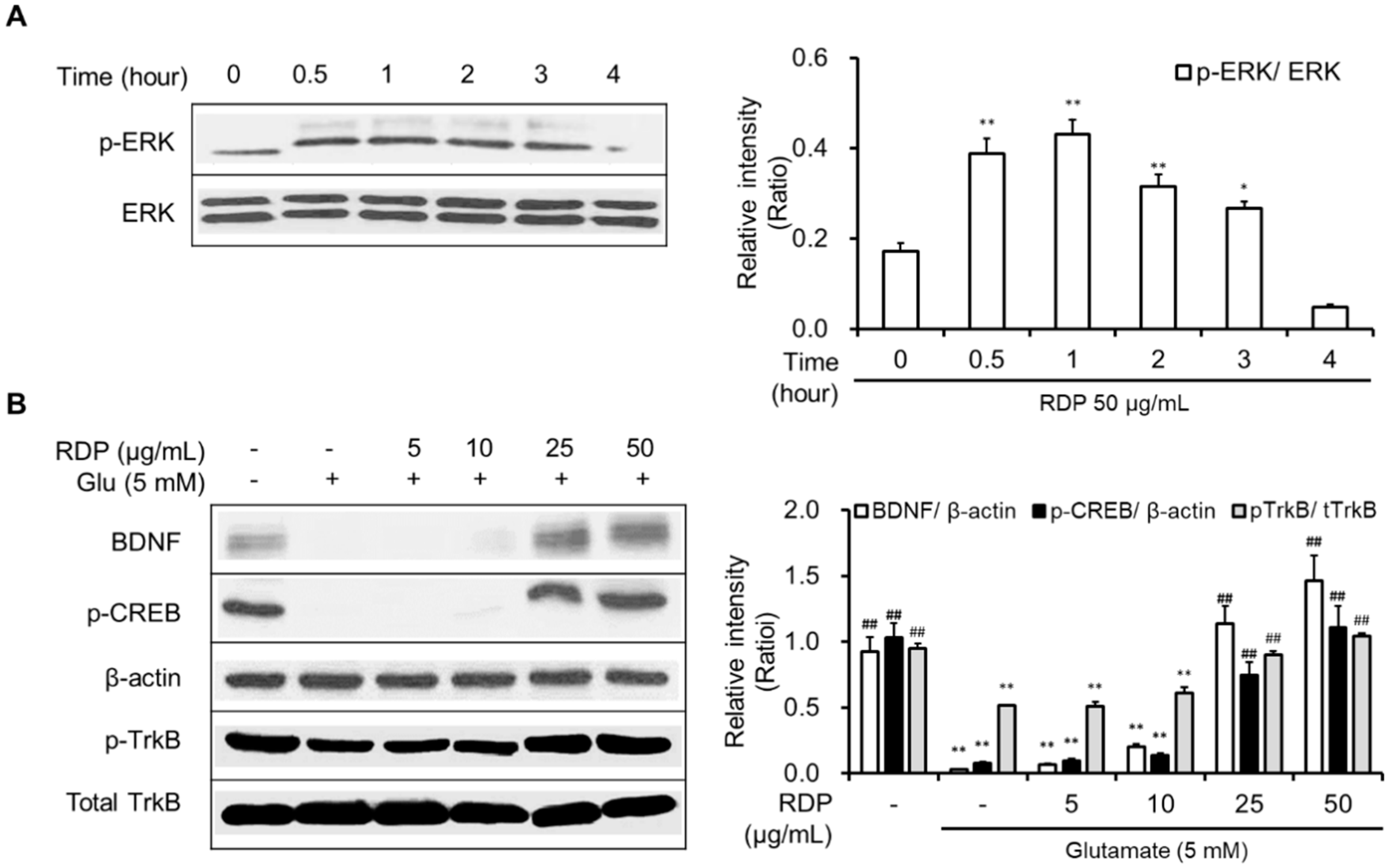
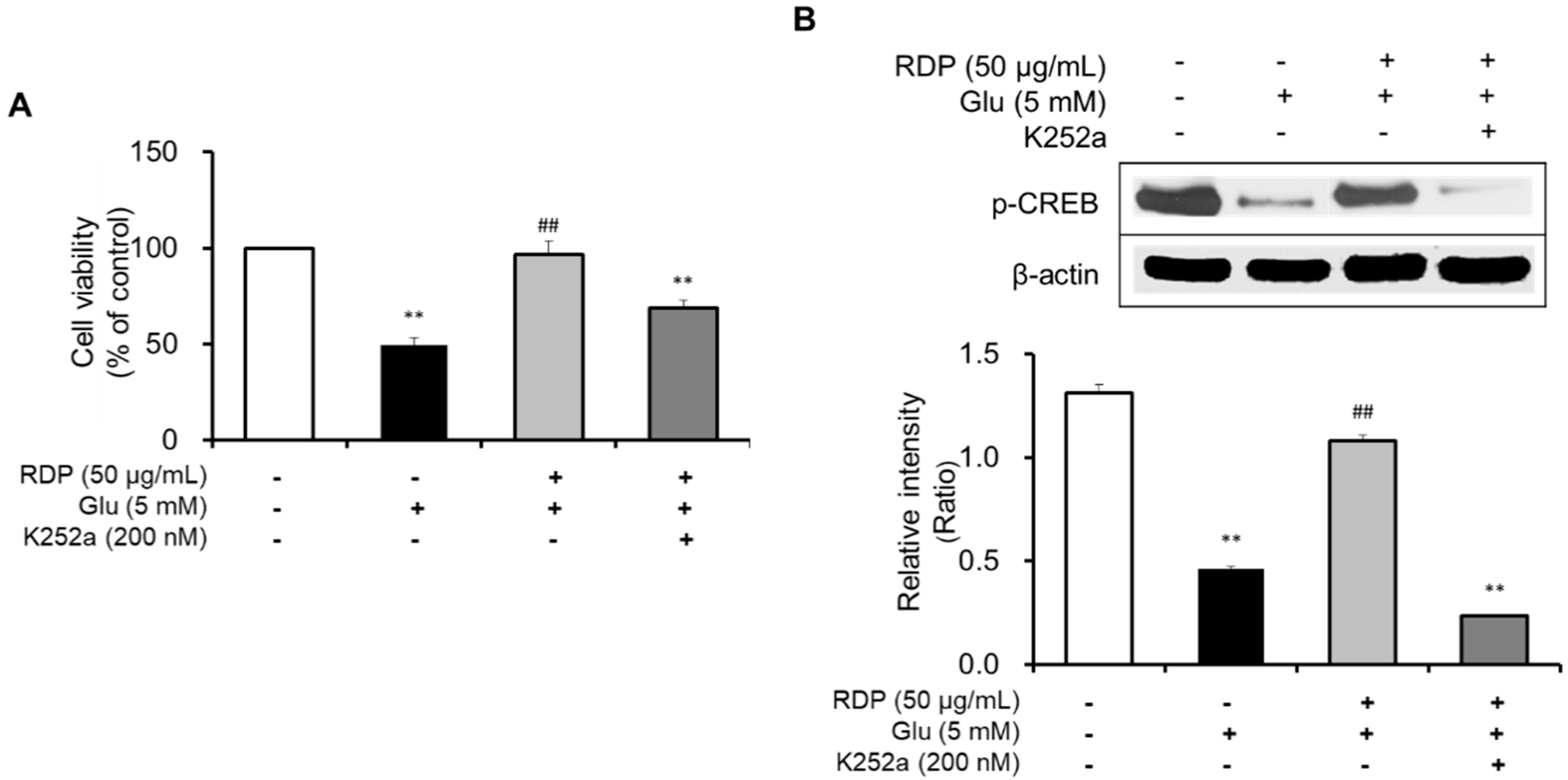
3.5. Activation of Antioxidant Defense System and BDNF/CREB Pathway by RDP
3.6. Changes of Body Weight in RDP-Treated Mice
3.7. RDP Improves Scopolamine-Induced Memory Impairment in Morris Water Maze Test
3.8. Enhancing Action of RDP on Memory Failure in Passive Avoidance Test
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AIF | apoptosis-inducing factor; |
| Akt | protein kinase B; |
| Bax | Bcl-2-associated X protein; |
| Bcl-2 | B-cell lymphoma 2; |
| BDNF | brain-derived neurotrophic factor; |
| CREB | cAMP response element-binding protein; |
| ERK | extracellular signal-regulated kinase; |
| GCLC | glutamate-cysteine ligase catalytic subunit; |
| HO-1 | heme oxygenase-1; |
| NQO-1 | NAD(P)H quinine oxidoreductase-1; |
| Nrf2 | NF-E2-related factor-2; |
| ROS | reactive oxygen species; |
| TrkB | Tropomyosin-related kinase receptor B |
References
- Ross, C.A.; Poirier, M.A. Protein aggregation and neurodegenerative disease. Nat. Med. 2004, 10, 10–17. [Google Scholar] [CrossRef]
- Yang, E.J.; Kim, G.S.; Kim, J.A.; Song, K.S. Protective effects of onion derived quercetin on glutamate mediated hippocampal neuronal cell death. Pharmacogn. Mag. 2013, 9, 302–308. [Google Scholar] [PubMed] [Green Version]
- Tan, M.; Ouyang, Y.; Jin, M.; Chen, M.; Liu, P.; Chao, X.; Chen, Z.; Chen, X.; Ramassamy, C.; Gao, Y.; et al. Downregulation of Nrf2/HO-1 pathway and activation of JNK/c-Jun pathway are involved in homocysteic acid-induced cytotoxicity in HT-22 cells. Toxicol. Lett. 2013, 223, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Gliyazova, N.S.; Huh, E.Y.; Ibeanu, G.C. A novel phenoxy thiophene sulphonamide molecule protects against glutamate evoked oxidative injury in a neuronal cell model. BMC. Neurosci. 2013, 14, 93–105. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, M.T.; Beal, M.F. Mitochondrial dysfunction and oxidative stress in neurodegenerative diseases. Nature 2006, 443, 787–795. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Li, H.; Li, X.J. Intracellular degradation of misfolded proteins in polyglutamine neurodegenerative diseases. Brain Res. Rev. 2008, 59, 245–252. [Google Scholar] [CrossRef] [Green Version]
- Calabrese, V.; Boyd-kimball, D.; Scapagnini, G.; Butterfield, A. Nitric oxide and cellular stress response in brain aging and neurodegenerative disorders: The role of vitagenes. In Vivo 2004, 18, 245–268. [Google Scholar]
- Scapagnini, G.; Vasto, S.; Abraham, N.G.; Caruso, C.; Zella, D.; Fabio, G. Modulation of Nrf2/ARE pathway by food polyphenol: A nutritional neuroprotective strategy for cognitive and neurodegenerative disorders. Mol. Neurobiol. 2011, 44, 192–201. [Google Scholar] [CrossRef] [Green Version]
- Lee, B.D.; Yoo, J.M.; Baek, S.Y.; Li, F.Y.; Sok, D.E.; Kim, M.R. 3, 3′-Diindolylmethane promotes BDNF and antioxidant enzyme formation via TrkB/Akt pathway activation for neuroprotection against oxidative stress-induced apoptosis in hippocampal neuronal cells. Antioxidants 2020, 9, 3. [Google Scholar] [CrossRef] [Green Version]
- Baek, S.Y.; Kim, M.R. Neuroprotective Effect of Carotenoid-Rich Enteromorpha prolifera Extract via TrkB/Akt Pathway against Oxidative Stress in Hippocampal Neuronal Cells. Mar. Drugs 2020, 18, 372. [Google Scholar] [CrossRef]
- Johnson, J.A.; Johnson, D.A.; Kraft, A.D.; Calkins, M.J.; Jakel, R.J.; Vargas, M.R.; Chen, P.C. The Nrf2-ARE pathway: An indicator and modulator of oxidative stress in neurodegeneration. Ann. N. Y. Acad. Sci. 2008, 1147, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.K.; Yoo, J.M.; Li, F.Y.; Baek, S.Y.; Kim, M.R. Mulberry fruit improves memory in scopolamine-treated mice: Role of cholinergic function, antioxidant system, and TrkB/Akt signaling. Nutr. Neurosci. 2019, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, C.N.; Kim, H.E.; Lee, S.G. Caffeolyserotonin protects human keratinocyte HaCaT cells against H2O2-induced oxidative stress and apoptosis through upregulation of HO-1 expression via activation of the PI3K/Akt/Nrf2 pathway. Phytother. Res. 2013, 27, 1810–1818. [Google Scholar] [CrossRef]
- Kim, C.R.; Jeon, H.L.; Shin, S.K.; Kim, H.J.; Ahn, C.W.; Jung, S.U.; Park, S.H.; Kim, M.R. Neuroprotective action of deer bone extract against glutamate or Aβ 1–42-induced oxidative stress in mouse hippocampal Cells. J. Med. Food 2014, 17, 226–235. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.X.; Song, J.H.; Song, D.K.; Yong, V.W.; Shuaib, A.; Hao, C. Cyclin-dependent kinase-5 prevents neuronal apoptosis through ERK-mediated upregulation of Bcl-2. Cell Death Differ. 2006, 13, 1203–1212. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, R.; Li, Y.B.; Li, Y.H.; Xu, Y.; Wu, H.L.; Li, X.J. Curcumin protects against glutamate excitotoxicity in rat cerebral cortical neurons by increasing brain-derived neurotrophic factor level and activating TrkB. Brain Res. 2008, 1210, 84–91. [Google Scholar] [CrossRef]
- Yoo, J.M.; Lee, B.D.; Sok, D.E.; Ma, J.Y.; Kim, M.R. Neuroprotective action of n-acetyl serotonin in oxidative stress-induced apoptosis through the activation of both trkb/creb/bdnf pathway and akt/nrf2/antioxidant enzyme in neuronal cells. Redox Biol. 2017, 11, 592–599. [Google Scholar] [CrossRef]
- Min, A.Y.; Doo, C.N.; Son, E.J.; Sung, N.Y.; Lee, K.J.; Sok, D.E.; Kim, M.R. N-palmitoyl serotonin alleviates scopolamine-induced memory impairment via regulation of cholinergic and antioxidant systems, and expression of BDNF and p-CREB in mice. Chem. Biol. Interact. 2015, 242, 153–162. [Google Scholar] [CrossRef]
- Numakawa, T.; Suzuki, S.; Kumamaru, E.; Adachi, N.; Richards, M.; Kunugi, H. BDNF function and intracellular signaling in neurons. Histol. Histopathol. 2010, 25, 2. [Google Scholar]
- Zhang, F.; Kang, Z.; Li, W.; Xiao, Z.-C.; Zhou, X.-F. Roles of brain-derived neurotrophic factor/tropomyosin-related kinase B (BDNF/TrkB) signalling in Alzheimer’s disease. J. Clin. Neurosci. 2012, 19, 946–949. [Google Scholar] [CrossRef]
- Ji, J.F.; Ji, S.J.; Sun, R.; Li, K.; Zhang, Y.; Zhang, L.Y.; Tian, Y. Forced running exercise attenuates hippocampal neurogenesis impairment and the neurocognitive deficits induced by whole-brain irradiation via the BDNF-mediated pathway. Biochem. Biophys. Res. Common. 2014, 443, 646–651. [Google Scholar] [CrossRef]
- Yan, L.; Xu, X.; He, Z.; Wang, S.; Zhao, L.; Qiu, J.; Wang, D.; Gong, Z.; Qiu, X.; Huang, H. Antidepressant-like effects and cognitive enhancement of coadministration of chaihu shugan san and fluoxetine: Dependent on the BDNF-ERK-CREB signaling pathway in the hippocampus and frontal cortex. BioMed Res. Int. 2020, 2020, 2794263. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baek, S.Y.; Li, F.Y.; Kim, D.H.; Kim, S.J.; Kim, M.R. Enteromorpha prolifera extract improves memory in scopolamine-treated mice via downregulating amyloid-β expression and upregulating BDNF/TrkB pathway. Antioxidants 2020, 9, 620. [Google Scholar] [CrossRef] [PubMed]
- Du, C.N.; Min, A.Y.; Kim, H.J.; Shin, S.K.; Yu, H.N.; Sohn, E.J.; Park, S.H.; Kim, M.R. Deer bone extract prevents against scopolamine-induced memory impairment in mice. J. Med. Food 2015, 18, 157–165. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, F.Y.; Kim, M.R. Effect of Aged Garlic Ethyl Acetate Extract on Oxidative Stress and Cholinergic Function of Scopolamine-Induced Cognitive Impairment in Mice. Prev. Nutr. Food Sci. 2019, 24, 165. [Google Scholar] [CrossRef]
- Birasuren, B.; Oh, H.L.; Kim, C.R.; Kim, N.Y.; Jeon, H.R.; Kim, M.R. Antioxidant activities of Ribes diacanthum Pall extracts in the northern region of mongolia. Prev. Nutr. Food Sci. 2012, 17, 261–268. [Google Scholar] [CrossRef] [Green Version]
- Ligaa, U.; Davaasuren, B.; Ninjil, N. Medicinal Plants of Mongolia Used in Western and Eastern Medicine; JKL Printing: Ulaanbaatar, Mongolia, 2006; p. 374. [Google Scholar]
- Khasbagan, S.; Xing-he, G.E.N.G.; Orgil, J.I.N.; Shan, C.H.E.N. Nutritional contents of the fruits of Ribes diacanthum Pall. and its evaluation of edible value. J. Inner Mong. Norm. Univ. 2007, 4, 23. [Google Scholar]
- Kim, E.S.; Birasuren, B.; Kim, M.R. Preventive action of Ribes diacanthum Pall. against high blood glucose level in alloxan-induced diabetic mice. J. East Asian Soc. Diet. Life 2013, 23, 53–60. [Google Scholar]
- Jin, M.C.; Yoo, J.M.; Sok, D.E.; Kim, M.R. Neuroprotective effect of N-acyl 5-hydroxytryptamines on glutamate-induced cytotoxicity in HT-22 cells. Neurochem. Res. 2014, 39, 2440–2451. [Google Scholar] [CrossRef]
- Morris, R. Developments of a water-maze procedure for studying spatial learning in the rat. J. Neurosci. Method 1984, 11, 47–60. [Google Scholar] [CrossRef]
- Lorenzini, C.G.A.; Baldi, E.; Bucherelli, C.; Sacchetti, B.; Tassoni, G. Role of ventral hippocampus in acquisition, consolidation and retrieval of rat’s passive avoidance response memory trace. Brain Res. 1997, 768, 242–248. [Google Scholar] [CrossRef]
- Loh, K.P.; Huang, S.H.; De Silva, R.; Tan, B.K.; Zhu, Y.Z. Oxidative stress: Apoptosis in neuronal injury. Curr. Alzheimer Res. 2006, 3, 327–337. [Google Scholar] [CrossRef]
- Fukui, M.; Song, J.H.; Choi, J.; Choi, H.J.; Zhu, B.T. Mechanism of glutamate induced neurotoxicity in HT 22 mouse hippocampal cells. Eur. J. Pharmacol. 2009, 617, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Su, B.; Wang, X.; Smith, M.A.; Perry, G. Causes of oxidative stress in Alzheimer disease. Cell. Mol. Life Sci. 2007, 64, 2202–2210. [Google Scholar] [CrossRef] [PubMed]
- Blass, J.P. The mitochondrial spiral: An adequate cause of dementia in the Alzheimers syndrome. Ann. N. Y. Acad. Sci. 2000, 924, 170–183. [Google Scholar] [CrossRef] [PubMed]
- Gelain, D.P.; Antonio-Behr, G.; De Oliveira-Birnfeld, R.; Trujillo, M. Antioxidant therapies for neurodegenerative diseases: Mechanisms, current trends, and perspectives. Oxid. Med. Cell. Longev. 2012, 2012, 895153. [Google Scholar] [CrossRef]
- Choi, D.W. Glutamate neurotoxicity in cortical cell culture is calcium dependent. Neurosci. Lett. 1985, 58, 689–695. [Google Scholar] [CrossRef]
- Coyle, J.T.; Puttfarcken, P. Oxidative stress, glutamate, and neurodegenerative disorders. Science 1993, 262, 689–695. [Google Scholar] [CrossRef]
- Pastorino, J.G.; Chen, S.T.; Tafani, M.; Snyder, J.W.; Farber, J.L. The overexpression of Bax produces cell death upon induction of the mitochondrial permeability transition. J. Biol. Chem. 1998, 273, 7770–7775. [Google Scholar] [CrossRef] [Green Version]
- Kumar, H.; Kim, I.S.; More, S.V.; Kim, B.W.; Choi, D.K. Natural product derived pharmacological modulators of Nrf2/ARE pathway for chronic diseases. Nat. Prod. Rep. 2014, 31, 109–139. [Google Scholar] [CrossRef]
- Almeida, R.D.; Manadas, B.J.; Melo, C.V.; Gomes, J.R.; Mendes, C.S.; Graos, M.M.; Carvalho, R.F.; Carvalho, A.P.; Duarte, C.B. Neuroprotection by bdnf against glutamate-induced apoptotic cell death is mediated by erk and pi3-kinase pathways. Cell Death Differ. 2005, 12, 1329–1343. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, N.Y.; Cheong, S.H.; Lee, K.J.; Sok, D.E.; Kim, M.R. Anti-inflammatory effects of Ribes diacanthum Pall mediated via regulation of Nrf2/HO-1 and NF-κB signaling pathways in LPS-stimulated RAW 264.7 macrophages and a TPA-induced dermatitis animal model. Antioxidants 2020, 9, 622. [Google Scholar] [CrossRef] [PubMed]
- Bhullar, K.S.; Rupasinghe, H.P. Polyphenols: Multipotent therapeutic agents in neurodegenerative diseases. Oxid. Med. Cell. Longev. 2013, 2013, 891748. [Google Scholar] [CrossRef] [PubMed] [Green Version]


| Group | Con | Sco | Tac | RDP10 | RDP20 | RDP30 | |
|---|---|---|---|---|---|---|---|
| Weight of Body (g) | 1 day | 21.73 ± 0.08 | |||||
| 7 days | 29.80 ± 0.35 | 30.43 ± 0.35 | 30.03 ± 0.42 | 30.63 ± 0.45 | 29.82 ± 0.39 | 30.87 ± 0.44 | |
| 14 days | 32.73 ± 0.50 | 33.00 ± 0.41 | 32.52 ± 0.60 | 33.07 ± 0.56 | 32.85 ± 0.62 | 33.40 ± 0.31 | |
| 21 days | 34.78 ± 0.68 | 34.90 ± 0.87 | 34.57 ± 0.80 | 34.45 ± 0.58 | 36.15 ± 0.62 | 35.47 ± 0.86 | |
| 28 days | 36.35 ± 0.94 | 35.42 ± 0.81 | 35.00 ± 0.77 | 35.60 ± 0.52 | 36.73 ± 0.76 | 36.45 ± 0.70 | |
| 31 days | 36.83 ± 0.79 | 35.77 ± 0.71 | 35.17 ± 0.86 | 35.70 ± 0.55 | 36.50 ± 0.76 | 37.92 ± 0.84 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, H.J.; Baek, S.Y.; Sok, D.-E.; Lee, K.J.; Kim, Y.-J.; Kim, M.R. Neuroprotective Activity of Polyphenol-Rich Ribes diacanthum Pall against Oxidative Stress in Glutamate-Stimulated HT-22 Cells and a Scopolamine-Induced Amnesia Animal Model. Antioxidants 2020, 9, 895. https://doi.org/10.3390/antiox9090895
Kim HJ, Baek SY, Sok D-E, Lee KJ, Kim Y-J, Kim MR. Neuroprotective Activity of Polyphenol-Rich Ribes diacanthum Pall against Oxidative Stress in Glutamate-Stimulated HT-22 Cells and a Scopolamine-Induced Amnesia Animal Model. Antioxidants. 2020; 9(9):895. https://doi.org/10.3390/antiox9090895
Chicago/Turabian StyleKim, Hyun Jeong, Seung Yeon Baek, Dai-Eun Sok, Kun Jong Lee, Young-Jun Kim, and Mee Ree Kim. 2020. "Neuroprotective Activity of Polyphenol-Rich Ribes diacanthum Pall against Oxidative Stress in Glutamate-Stimulated HT-22 Cells and a Scopolamine-Induced Amnesia Animal Model" Antioxidants 9, no. 9: 895. https://doi.org/10.3390/antiox9090895






