The Role of Glutathione in Protecting against the Severe Inflammatory Response Triggered by COVID-19
Abstract
1. Introduction
2. Cellular GSH Homeostasis
3. The Protective Actions of GSH
- GSH protects cells by neutralizing (i.e., reducing) ROS, which are key signaling molecules that play an important role in the progression of inflammatory disorders. The relationship between ROS production and proinflammatory cytokine activation is well established [22]. An enhanced ROS generation by polymorphonuclear neutrophils at the site of inflammation causes endothelial dysfunction and tissue injury [23].
- The conjugation of GSH to xenobiotics is particularly abundant; glutathione S-transferase enzymes catalyze GSH conjugation to lipophilic xenobiotics, facilitating the excretion or further metabolism of many drugs. The conjugation process is illustrated by the metabolism of N-acetyl-p-benzoquinone imine (NAPQI). NAPQI is a reactive metabolite formed by the action of cytochrome P450 on paracetamol (acetaminophen). Glutathione conjugates to NAPQI and the resulting product is excreted [24].
- Many enzymes use GSH as a cofactor or substrate. For example reactions requiring GSH as a key cofactor are catalyzed by the prostaglandin H synthase, the rate-limiting enzyme in the production of prostaglandins and thromboxane [25], which are essential regulators of vascular function. Moreover, the enzyme leukotriene C(4) synthase conjugates LTA(4) with GSH to form the leukotriene LTC(4), the parent compound of the cysteinyl leukotrienes [26]; these molecules are potent mediators of airway narrowing.
- GSH is used to synthesize S-nitrosoglutathione (GSNO), an endogenous S-nitrosothiol that plays a critical role in nitric oxide (NO) signaling and is a source of bioavailable NO. The generation of GSNO can serve as a stable NO pool which can properly transduce NO signaling [27]. NO produced by nitric oxide synthase eNOS and nNOS, in the presence of GSH, can effectively modulate vessels and neuronal functions, regulating the blood flow according to the local calcium influx.
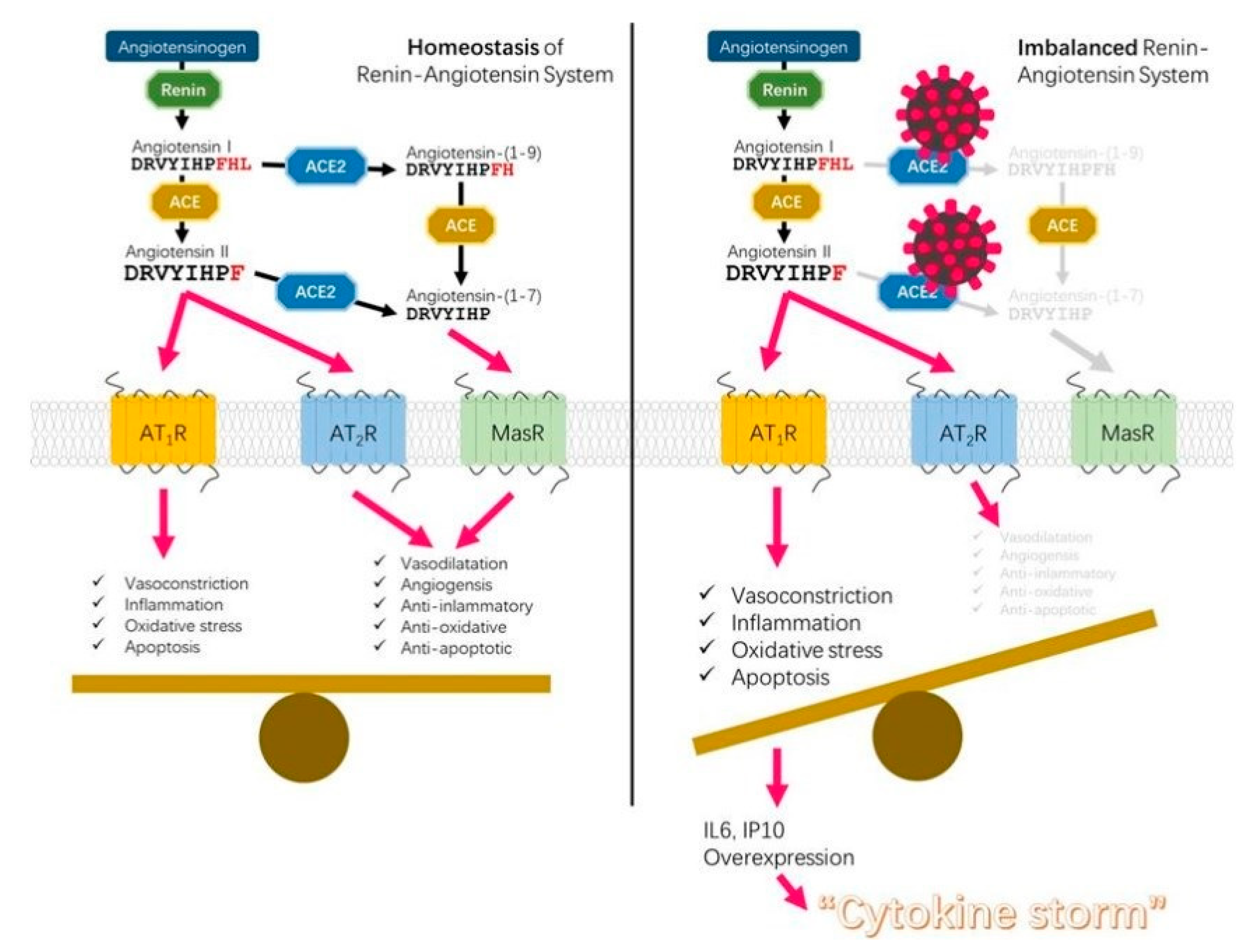
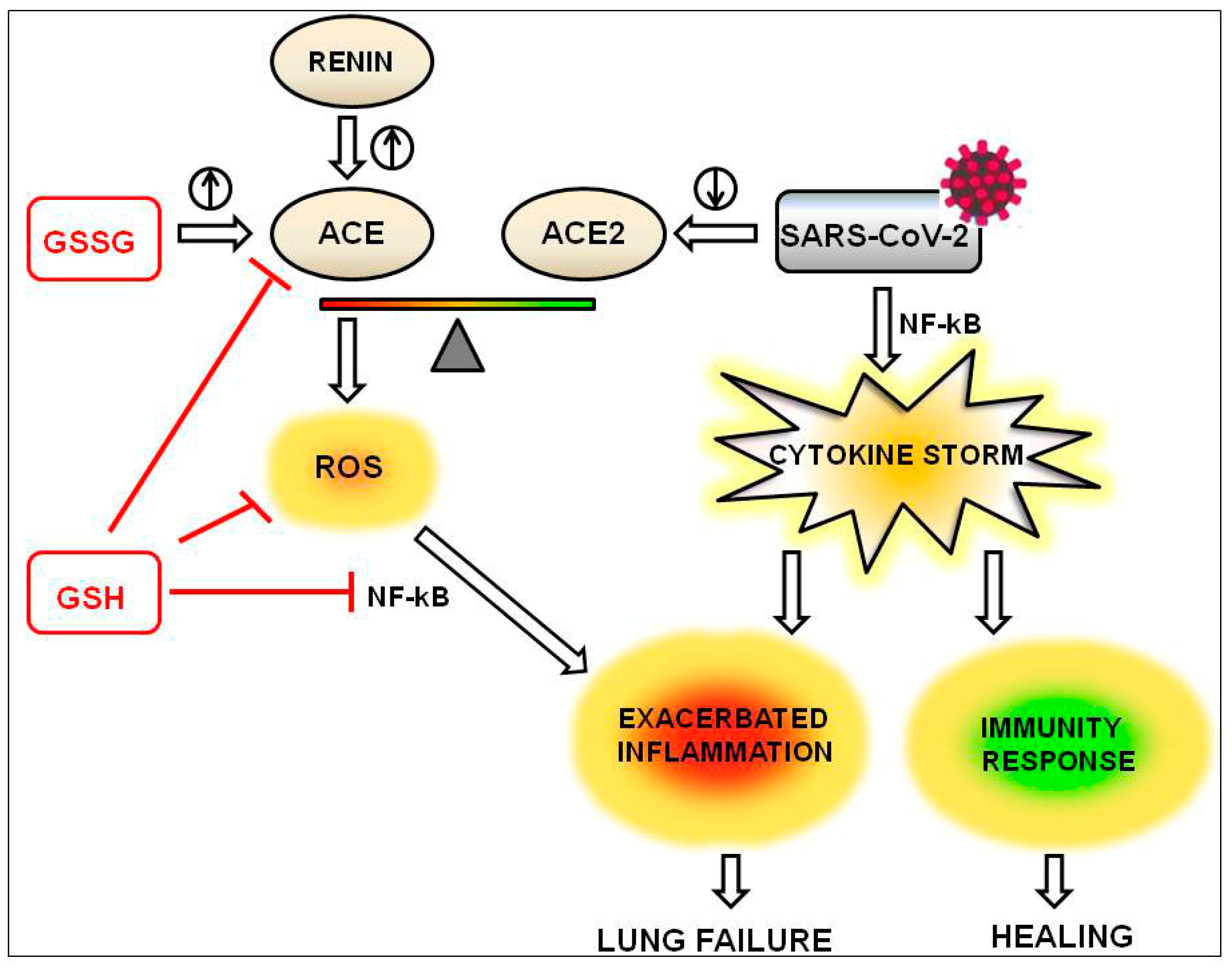
4. The Beneficial Effects of GSH on the Inflammation Driven by the Imbalance of ACE/ACE2
5. Conditions Associated with Low GSH
6. Treatment with GSH and Thiols
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| GSH | glutathione |
| SARS-CoV-2 | severe acute respiratory syndrome coronavirus 2 |
| ACE | angiotensin-converting enzyme |
| RAS | renin-angiotensin system |
| ROS | reactive oxygen species |
| GST | glutathione S-transferase |
| GPX | glutathione peroxidase |
| NAPQI | N-acetyl-p-benzoquinone imine |
| LT | leukotriene |
| GSNO | S-nitrosoglutathione |
| NO | nitric oxide |
| NOS | nitric oxide synthase |
| ANGII | angiotensin II |
| AT1R | angiotensin II receptor type 1 |
| NF-kB | nuclear factor kappa-light-chain-enhancer of activated B cells |
| IL-6 | interleukin-6 |
| CQ | chloroquine |
| PEDV | porcine epidemic diarrhea virus |
| NAC | N-acetyl-l-cysteine |
| COPD | chronic obstructive pulmonary disease |
References
- Capettini, L.S.A.; Montecucco, F.; Mach, F.; Stergiopulos, N.; Santos, R.A.S.; da Silva, R.F. Role of renin-angiotensin system in inflammation, immunity and aging. Curr. Pharm. Des. 2012, 18, 963–970. [Google Scholar] [CrossRef] [PubMed]
- Kuba, K.; Imai, Y.; Rao, S.; Gao, H.; Guo, F.; Guan, B.; Huan, Y.; Yang, P.; Zhang, Y.; Deng, W.; et al. A crucial role of angiotensin converting enzyme 2 (ACE2) in SARS coronavirus-induced lung injury. Nat. Med. 2005, 11, 875–879. [Google Scholar] [CrossRef] [PubMed]
- Ji, X.; Zhang, C.; Zhai, Y.; Zhang, Z.; Xue, Y.; Zhang, C.; Tan, G.; Niu, G. TWIRLS, an Automated Topic-wise Inference Method Based on Massive Literature, Suggests a Possible Mechanism via ACE2 for the Pathological Changes in the Human Host after Coronavirus Infection. medRxiv 2020. [Google Scholar] [CrossRef]
- Hanff, T.C.; Harhay, M.O.; Brown, T.S.; Cohen, J.B.; Mohareb, A.M. Is There an Association Between COVID-19 Mortality and the Renin-Angiotensin System—A Call for Epidemiologic Investigations. Clin. Infect. Dis. 2020, ciaa329. [Google Scholar] [CrossRef]
- Vaduganathan, M.; Vardeny, O.; Michel, T.; McMurray, J.J.V.; Pfeffer, M.A.; Solomon, S.D. Renin–Angiotensin–Aldosterone System Inhibitors in Patients with Covid-19. N. Engl. J. Med. 2020, 382, 1653–1659. [Google Scholar] [CrossRef]
- Naik, E.; Dixit, V.M. Mitochondrial reactive oxygen species drive proinflammatory cytokine production. J. Exp. Med. 2011, 208, 417–420. [Google Scholar] [CrossRef]
- Zherebiatiev, A.; Kamyshnyi, A. Expression Levels of Proinflammatory Cytokines and NLRP3 Inflammasome in an Experimental Model of Oxazolone-induced Colitis. Iran. J. Allergy Asthma. Immunol. 2016, 15, 39–45. [Google Scholar]
- Forman, H.J.; Zhang, H.; Rinna, A. Glutathione: Overview of its protective roles, measurement, and biosynthesis. Mol. Aspects Med. 2009, 30, 1–12. [Google Scholar] [CrossRef]
- Liu, R.-M.; Vayalil, P.K.; Ballinger, C.; Dickinson, D.A.; Huang, W.-T.; Wang, S.; Kavanagh, T.J.; Matthews, Q.L.; Postlethwait, E.M. Transforming growth factor β suppresses glutamate-cysteine ligase gene expression and induces oxidative stress in a lung fibrosis model. Free Radic. Biol. Med. 2012, 53, 554–563. [Google Scholar] [CrossRef]
- Gorowiec, M.R.; Borthwick, L.A.; Parker, S.M.; Kirby, J.A.; Saretzki, G.C.; Fisher, A.J. Free radical generation induces epithelial-to-mesenchymal transition in lung epithelium via a TGF-β1-dependent mechanism. Free Radic. Biol. Med. 2012, 52, 1024–1032. [Google Scholar] [CrossRef]
- Barcellos-Hoff, M.H.; Dix, T.A. Redox-mediated activation of latent transforming growth factor-beta 1. Mol. Endocrinol. 1996, 10, 1077–1083. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.-M.; Gaston Pravia, K.A. Oxidative stress and glutathione in TGF-beta-mediated fibrogenesis. Free Radic. Biol. Med. 2010, 48, 1–15. [Google Scholar] [CrossRef]
- Jackson, R.M.; Gupta, C. Hypoxia and kinase activity regulate lung epithelial cell glutathione. Exp. Lung Res. 2010, 36, 45–56. [Google Scholar] [CrossRef] [PubMed]
- Lei, G.-S.; Zhang, C.; Cheng, B.-H.; Lee, C.-H. Mechanisms of Action of Vitamin D as Supplemental Therapy for Pneumocystis Pneumonia. Antimicrob. Agents Chemother. 2017, 61. [Google Scholar] [CrossRef] [PubMed]
- Biolo, G.; Antonione, R.; De Cicco, M. Glutathione metabolism in sepsis. Crit. Care Med. 2007, 35, S591–S595. [Google Scholar] [CrossRef]
- Dobrakowski, M.; Pawlas, N.; Hudziec, E.; Kozłowska, A.; Mikołajczyk, A.; Birkner, E.; Kasperczyk, S. Glutathione, glutathione-related enzymes, and oxidative stress in individuals with subacute occupational exposure to lead. Environ. Toxicol. Pharmacol. 2016, 45, 235–240. [Google Scholar] [CrossRef]
- Wang, W.; Ballatori, N. Endogenous glutathione conjugates: Occurrence and biological functions. Pharmacol. Rev. 1998, 50, 335–356. [Google Scholar]
- Ballatori, N.; Krance, S.M.; Marchan, R.; Hammond, C.L. Plasma membrane glutathione transporters and their roles in cell physiology and pathophysiology. Mol. Aspects Med. 2009, 30, 13–28. [Google Scholar] [CrossRef]
- Reid, M.; Jahoor, F. Glutathione in disease. Curr. Opin. Clin. Nutr. Metab. Care 2001, 4, 65–71. [Google Scholar] [CrossRef]
- Wu, G.; Fang, Y.-Z.; Yang, S.; Lupton, J.R.; Turner, N.D. Glutathione metabolism and its implications for health. J. Nutr. 2004, 134, 489–492. [Google Scholar] [CrossRef]
- Townsend, D.M.; Tew, K.D.; Tapiero, H. The importance of glutathione in human disease. Biomed. Pharmacother. 2003, 57, 145–155. [Google Scholar] [CrossRef]
- Agita, A.; Alsagaff, M.T. Inflammation, Immunity, and Hypertension. Acta Med. Indones 2017, 49, 158–165. [Google Scholar] [PubMed]
- Mittal, M.; Siddiqui, M.R.; Tran, K.; Reddy, S.P.; Malik, A.B. Reactive oxygen species in inflammation and tissue injury. Antioxid. Redox Signal. 2014, 20, 1126–1167. [Google Scholar] [CrossRef] [PubMed]
- Hedgpeth, B.; Missall, R.; Bambaci, A.; Smolen, M.; Yavuz, S.; Cottrell, J.; Chu, T.; Chang, S.L. A Review of Bioinformatics Tools to Understand Acetaminophen-Alcohol Interaction. Medicines 2019, 6, 79. [Google Scholar] [CrossRef] [PubMed]
- Burch, J.W.; Services, P.T. Glutathione disulfide production during arachidonic acid oxygenation in human platelets. Prostaglandins 1990, 39, 123–134. [Google Scholar] [CrossRef]
- Lam, B.K. Leukotriene C(4) synthase. Prostaglandins Leukot. Essent. Fatty Acids 2003, 69, 111–116. [Google Scholar] [CrossRef]
- Balazy, M.; Kaminski, P.M.; Mao, K.; Tan, J.; Wolin, M.S. S-Nitroglutathione, a product of the reaction between peroxynitrite and glutathione that generates nitric oxide. J. Biol. Chem. 1998, 273, 32009–32015. [Google Scholar] [CrossRef]
- Basi, Z.; Turkoglu, V. In vitro effect of oxidized and reduced glutathione peptides on angiotensin converting enzyme purified from human plasma. J. Chromatogr. B 2019, 1104, 190–195. [Google Scholar] [CrossRef]
- Zhang, H.; Penninger, J.M.; Li, Y.; Zhong, N.; Slutsky, A.S. Angiotensin-converting enzyme 2 (ACE2) as a SARS-CoV-2 receptor: Molecular mechanisms and potential therapeutic target. Intensive. Care Med. 2020, 46, 586–590. [Google Scholar] [CrossRef]
- Walls, A.C.; Park, Y.-J.; Tortorici, M.A.; Wall, A.; McGuire, A.T.; Veesler, D. Structure, Function, and Antigenicity of the SARS-CoV-2 Spike Glycoprotein. Cell 2020, 181, 281–292. [Google Scholar] [CrossRef]
- Glowacka, I.; Bertram, S.; Herzog, P.; Pfefferle, S.; Steffen, I.; Muench, M.O.; Simmons, G.; Hofmann, H.; Kuri, T.; Weber, F.; et al. Differential downregulation of ACE2 by the spike proteins of severe acute respiratory syndrome coronavirus and human coronavirus NL63. J. Virol. 2010, 84, 1198–1205. [Google Scholar] [CrossRef]
- Banu, N.; Panikar, S.S.; Leal, L.R.; Leal, A.R. Protective role of ACE2 and its downregulation in SARS-CoV-2 infection leading to Macrophage Activation Syndrome: Therapeutic implications. Life Sci. 2020, 256, 117905. [Google Scholar] [CrossRef]
- Vajapey, R.; Rini, D.; Walston, J.; Abadir, P. The impact of age-related dysregulation of the angiotensin system on mitochondrial redox balance. Front. Physiol. 2014, 5. [Google Scholar] [CrossRef]
- DeDiego, M.L.; Nieto-Torres, J.L.; Regla-Nava, J.A.; Jimenez-Guardeno, J.M.; Fernandez-Delgado, R.; Fett, C.; Castano-Rodriguez, C.; Perlman, S.; Enjuanes, L. Inhibition of NF- B-Mediated Inflammation in Severe Acute Respiratory Syndrome Coronavirus-Infected Mice Increases Survival. J. Virol. 2014, 88, 913–924. [Google Scholar] [CrossRef]
- Zhang, X.; Wu, K.; Wang, D.; Yue, X.; Song, D.; Zhu, Y.; Wu, J. Nucleocapsid protein of SARS-CoV activates interleukin-6 expression through cellular transcription factor NF-κB. Virology 2007, 365, 324–335. [Google Scholar] [CrossRef]
- Henry, B.M.; de Oliveira, M.H.S.; Benoit, S.; Plebani, M.; Lippi, G. Hematologic, biochemical and immune biomarker abnormalities associated with severe illness and mortality in coronavirus disease 2019 (COVID-19): A meta-analysis. Clin. Chem. Lab. Med. 2020, 58, 1021–1028. [Google Scholar] [CrossRef]
- Rahman, I.; MacNee, W. Oxidative stress and regulation of glutathione in lung inflammation. Eur. Respir. J. 2000, 16, 534–554. [Google Scholar] [CrossRef]
- Park, H.J.; Lee, J.-Y.; Chung, M.-Y.; Park, Y.-K.; Bower, A.M.; Koo, S.I.; Giardina, C.; Bruno, R.S. Green Tea Extract Suppresses NFκB Activation and Inflammatory Responses in Diet-Induced Obese Rats with Nonalcoholic Steatohepatitis. J. Nutr. 2012, 142, 57–63. [Google Scholar] [CrossRef]
- Franco, R.; Schoneveld, O.J.; Pappa, A.; Panayiotidis, M.I. The central role of glutathione in the pathophysiology of human diseases. Arch. Physiol. Biochem. 2007, 113, 234–258. [Google Scholar] [CrossRef]
- Teskey, G.; Abrahem, R.; Cao, R.; Gyurjian, K.; Islamoglu, H.; Lucero, M.; Martinez, A.; Paredes, E.; Salaiz, O.; Robinson, B.; et al. Glutathione as a Marker for Human Disease. Adv. Clin. Chem. 2018, 87, 141–159. [Google Scholar] [CrossRef]
- Du, Y.; Tu, L.; Zhu, P.; Mu, M.; Wang, R.; Yang, P.; Wang, X.; Hu, C.; Ping, R.; Hu, P.; et al. Clinical Features of 85 Fatal Cases of COVID-19 from Wuhan. A Retrospective Observational Study. Am. J. Respir. Crit. Care Med. 2020, 201, 1372–1379. [Google Scholar] [CrossRef]
- Guan, W.-J.; Liang, W.-H.; Zhao, Y.; Liang, H.-R.; Chen, Z.-S.; Li, Y.-M.; Liu, X.-Q.; Chen, R.-C.; Tang, C.-L.; Wang, T.; et al. Comorbidity and its impact on 1590 patients with COVID-19 in China: A nationwide analysis. Eur. Respir. J. 2020, 55. [Google Scholar] [CrossRef]
- Wang, B.; Li, R.; Lu, Z.; Huang, Y. Does comorbidity increase the risk of patients with COVID-19: Evidence from meta-analysis. Aging (Albany NY) 2020, 12, 6049–6057. [Google Scholar] [CrossRef]
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef]
- Palaiodimos, L.; Kokkinidis, D.G.; Li, W.; Karamanis, D.; Ognibene, J.; Arora, S.; Southern, W.N.; Mantzoros, C.S. Severe obesity, increasing age and male sex are independently associated with worse in-hospital outcomes, and higher in-hospital mortality, in a cohort of patients with COVID-19 in the Bronx, New York. Metab. Clin. Exp. 2020, 108, 154262. [Google Scholar] [CrossRef]
- Amatore, D.; Celestino, I.; Brundu, S.; Galluzzi, L.; Coluccio, P.; Checconi, P.; Magnani, M.; Palamara, A.T.; Fraternale, A.; Nencioni, L. Glutathione increase by the n-butanoyl glutathione derivative (GSH-C4) inhibits viral replication and induces a predominant Th1 immune profile in old mice infected with influenza virus. FASEB Bioadv. 2019, 1, 296–305. [Google Scholar] [CrossRef]
- Erden-Inal, M.; Sunal, E.; Kanbak, G. Age-related changes in the glutathione redox system. Cell Biochem. Funct. 2002, 20, 61–66. [Google Scholar] [CrossRef]
- Matsubara, L.S.; Machado, P.E. Age-related changes of glutathione content, glutathione reductase and glutathione peroxidase activity of human erythrocytes. Braz. J. Med. Biol. Res. 1991, 24, 449–454. [Google Scholar]
- Loguercio, C.; Taranto, D.; Vitale, L.M.; Beneduce, F.; Del Vecchio Blanco, C. Effect of liver cirrhosis and age on the glutathione concentration in the plasma, erythrocytes, and gastric mucosa of man. Free Radic. Biol. Med. 1996, 20, 483–488. [Google Scholar] [CrossRef]
- Suh, J.H.; Wang, H.; Liu, R.-M.; Liu, J.; Hagen, T.M. (R)-alpha-lipoic acid reverses the age-related loss in GSH redox status in post-mitotic tissues: Evidence for increased cysteine requirement for GSH synthesis. Arch. Biochem. Biophys. 2004, 423, 126–135. [Google Scholar] [CrossRef]
- Lang, C.A.; Naryshkin, S.; Schneider, D.L.; Mills, B.J.; Lindeman, R.D. Low blood glutathione levels in healthy aging adults. J. Lab. Clin. Med. 1992, 120, 720–725. [Google Scholar] [PubMed]
- Al-Turk, W.A.; Stohs, S.J.; el-Rashidy, F.H.; Othman, S. Changes in glutathione and its metabolizing enzymes in human erythrocytes and lymphocytes with age. J. Pharm. Pharmacol. 1987, 39, 13–16. [Google Scholar] [CrossRef] [PubMed]
- Jin, J.-M.; Bai, P.; He, W.; Wu, F.; Liu, X.-F.; Han, D.-M.; Liu, S.; Yang, J.-K. Gender Differences in Patients With COVID-19: Focus on Severity and Mortality. Front. Public Health 2020, 8, 152. [Google Scholar] [CrossRef] [PubMed]
- Dincer, Y.; Ozen, E.; Kadioglu, P.; Hatemi, H.; Akçay, T. Effect of sex hormones on lipid peroxidation in women with polycystic ovary syndrome, healthy women, and men. Endocr. Res. 2001, 27, 309–316. [Google Scholar] [CrossRef] [PubMed]
- Lavoie, J.C.; Chessex, P. Gender and maturation affect glutathione status in human neonatal tissues. Free Radic. Biol. Med. 1997, 23, 648–657. [Google Scholar] [CrossRef]
- Goldfarb, A.H.; McKenzie, M.J.; Bloomer, R.J. Gender comparisons of exercise-induced oxidative stress: Influence of antioxidant supplementation. Appl. Physiol. Nutr. Metab. 2007, 32, 1124–1131. [Google Scholar] [CrossRef]
- Bukowska, A.; Spiller, L.; Wolke, C.; Lendeckel, U.; Weinert, S.; Hoffmann, J.; Bornfleth, P.; Kutschka, I.; Gardemann, A.; Isermann, B.; et al. Protective regulation of the ACE2/ACE gene expression by estrogen in human atrial tissue from elderly men. Exp. Biol. Med. 2017, 242, 1412–1423. [Google Scholar] [CrossRef]
- Liu, J.; Ji, H.; Zheng, W.; Wu, X.; Zhu, J.J.; Arnold, A.P.; Sandberg, K. Sex differences in renal angiotensin converting enzyme 2 (ACE2) activity are 17β-oestradiol-dependent and sex chromosome-independent. Biol. Sex Differ. 2010, 1, 6. [Google Scholar] [CrossRef] [PubMed]
- Stelzig, K.E.; Canepa-Escaro, F.; Schiliro, M.; Berdnikovs, S.; Prakash, Y.S.; Chiarella, S.E. Estrogen regulates the expression of SARS-CoV-2 receptor ACE2 in differentiated airway epithelial cells. Am. J. Physiol. Lung Cell Mol. Physiol. 2020, 318, L1280–L1281. [Google Scholar] [CrossRef] [PubMed]
- Landazuri, P.; Granobles, C.; Loango, N. Gender differences in serum angiotensin-converting enzyme activity and blood pressure in children: An observational study. Arq. Bras. Cardiol. 2008, 91, 352–357. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ejima, K.; Nanri, H.; Araki, M.; Uchida, K.; Kashimura, M.; Ikeda, M. 17beta-estradiol induces protein thiol/disulfide oxidoreductases and protects cultured bovine aortic endothelial cells from oxidative stress. Eur. J. Endocrinol. 1999, 140, 608–613. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Walther, U.I. Changes in the glutathione system of lung cell lines after treatment with hydrocortisone. Arch. Toxicol. 2004, 78, 402–409. [Google Scholar] [CrossRef] [PubMed]
- Barton, R.N.; Horan, M.A.; Weijers, J.W.; Sakkee, A.N.; Roberts, N.A.; van Bezooijen, C.F. Cortisol production rate and the urinary excretion of 17-hydroxycorticosteroids, free cortisol, and 6 beta-hydroxycortisol in healthy elderly men and women. J. Gerontol. 1993, 48, M213–M218. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y. Sex differences in oxidative damage in ddY mouse kidney treated with a renal carcinogen, iron nitrilotriacetate. Carcinogenesis 1998, 19, 1983–1988. [Google Scholar] [CrossRef]
- Hu, J.J.; Lee, M.J.; Vapiwala, M.; Reuhl, K.; Thomas, P.E.; Yang, C.S. Sex-Related Differences in Mouse Renal Metabolism and Toxicity of Acetaminophen. Toxicol. Appl. Pharmacol. 1993, 122, 16–26. [Google Scholar] [CrossRef]
- Basile, M.; Ciardi, L.; Crespi, I.; Saliva, E.; Bellomo, G.; Vidali, M. Assessing Serum Concentrations of 25-Hydroxy-Vitamin D in North-Western Italy. J. Frailty Aging 2013, 2, 174–178. [Google Scholar] [CrossRef]
- Yoshida, K.; Hirokawa, J.; Tagami, S.; Kawakami, Y.; Urata, Y.; Kondo, T. Weakened cellular scavenging activity against oxidative stress in diabetes mellitus: Regulation of glutathione synthesis and efflux. Diabetologia 1995, 38, 201–210. [Google Scholar] [CrossRef]
- Thornalley, P.J.; McLellan, A.C.; Lo, T.W.C.; Benn, J.; Sönksen, P.H. Negative Association between Erythrocyte Reduced Glutathione Concentration and Diabetic Complications. Clin. Sci. 1996, 91, 575–582. [Google Scholar] [CrossRef]
- Arnalich, F.; Hernanz, A.; López-Maderuelo, D.; de la Fuente, M.; Arnalich, F.M.; Andrés-Mateos, E.; Fernández-Capitán, C.; Montiel, C. Intracellular glutathione deficiency is associated with enhanced nuclear factor-κB activation in older noninsulin dependent diabetic patients. Free Radic. Res. 2001, 35, 873–884. [Google Scholar] [CrossRef]
- Samiec, P.S.; Drews-Botsch, C.; Flagg, E.W.; Kurtz, J.C.; Sternberg, P.; Reed, R.L.; Jones, D.P. Glutathione in human plasma: Decline in association with aging, age-related macular degeneration, and diabetes. Free Radic. Biol. Med. 1998, 24, 699–704. [Google Scholar] [CrossRef]
- Petrie, J.R.; Ueda, S.; Webb, D.J.; Elliott, H.L.; Connell, J.M.C. Endothelial Nitric Oxide Production and Insulin Sensitivity: A Physiological Link With Implications for Pathogenesis of Cardiovascular Disease. Circulation 1996, 93, 1331–1333. [Google Scholar] [CrossRef] [PubMed]
- McGrowdera, D.; Ragoobirsingh, D.; Brown, P. Modulation of glucose uptake in adipose tissue by nitric oxide-generating compounds. J. Biosci. 2006, 31, 347–354. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, A.B.; Guarino, M.P.; Macedo, M.P. Understanding the in-vivo relevance of S -nitrosothiols in insulin action. Can. J. Physiol. Pharmacol. 2012, 90, 887–894. [Google Scholar] [CrossRef] [PubMed]
- Fang, L.; Karakiulakis, G.; Roth, M. Are patients with hypertension and diabetes mellitus at increased risk for COVID-19 infection? Lancet Respir. Med. 2020, 8, e21. [Google Scholar] [CrossRef]
- Vaziri, N.D.; Wang, X.Q.; Oveisi, F.; Rad, B. Induction of oxidative stress by glutathione depletion causes severe hypertension in normal rats. Hypertension 2000, 36, 142–146. [Google Scholar] [CrossRef] [PubMed]
- Guo, D.; Gu, J.; Jiang, H.; Ahmed, A.; Zhang, Z.; Gu, Y. Inhibition of pyruvate kinase M2 by reactive oxygen species contributes to the development of pulmonary arterial hypertension. J. Mol. Cell. Cardiol. 2016, 91, 179–187. [Google Scholar] [CrossRef]
- Zhou, X.J.; Vaziri, N.D.; Wang, X.Q.; Silva, F.G.; Laszik, Z. Nitric oxide synthase expression in hypertension induced by inhibition of glutathione synthase. J. Pharmacol. Exp. Ther. 2002, 300, 762–767. [Google Scholar] [CrossRef]
- Richardson, S.; Hirsch, J.S.; Narasimhan, M.; Crawford, J.M.; McGinn, T.; Davidson, K.W.; The Northwell COVID-19 Research Consortium; Barnaby, D.P.; Becker, L.B.; Chelico, J.D.; et al. Presenting Characteristics, Comorbidities, and Outcomes Among 5700 Patients Hospitalized With COVID-19 in the New York City Area. JAMA 2020. [Google Scholar] [CrossRef]
- Habib, S.A.; Saad, E.A.; Elsharkawy, A.A.; Attia, Z.R. Pro-inflammatory adipocytokines, oxidative stress, insulin, Zn and Cu: Interrelations with obesity in Egyptian non-diabetic obese children and adolescents. Adv. Med. Sci. 2015, 60, 179–185. [Google Scholar] [CrossRef]
- Uzun, H.; Konukoglu, D.; Gelisgen, R.; Zengin, K.; Taskin, M. Plasma protein carbonyl and thiol stress before and after laparoscopic gastric banding in morbidly obese patients. Obes. Surg. 2007, 17, 1367–1373. [Google Scholar] [CrossRef]
- Zamora-Mendoza, R.; Rosas-Vargas, H.; Ramos-Cervantes, M.T.; Garcia-Zuniga, P.; Perez-Lorenzana, H.; Mendoza-Lorenzo, P.; Perez-Ortiz, A.C.; Estrada-Mena, F.J.; Miliar-Garcia, A.; Lara-Padilla, E.; et al. Dysregulation of mitochondrial function and biogenesis modulators in adipose tissue of obese children. Int. J. Obes. 2018, 42, 618–624. [Google Scholar] [CrossRef]
- Parsanathan, R.; Jain, S.K. Glutathione deficiency induces epigenetic alterations of vitamin D metabolism genes in the livers of high-fat diet-fed obese mice. Sci. Rep. 2019, 9. [Google Scholar] [CrossRef] [PubMed]
- Andrich, D.E.; Melbouci, L.; Ou, Y.; Auclair, N.; Mercier, J.; Grenier, J.-C.; Lira, F.S.; Barreiro, L.B.; Danialou, G.; Comtois, A.-S.; et al. A Short-Term High-Fat Diet Alters Glutathione Levels and IL-6 Gene Expression in Oxidative Skeletal Muscles of Young Rats. Front. Physiol. 2019, 10, 372. [Google Scholar] [CrossRef] [PubMed]
- Ponsoda, X.; Jover, R.; Gómez-Lechón, M.J.; Fabra, R.; Trullenque, R.; Castell, J. Intracellular glutathione in human hepatocytes incubated with S-adenosyl-L-methionine and GSH-depleting drugs. Toxicology 1991, 70, 293–302. [Google Scholar] [CrossRef]
- Post, A.; Crochemore, C.; Uhr, M.; Holsboer, F.; Behl, C. Differential induction of NF-kappaB activity and neural cell death by antidepressants in vitro. Eur. J. Neurosci. 2000, 12, 4331–4337. [Google Scholar] [CrossRef]
- Bhattacharyya, B.; Chatterjee, T.K.; Ghosh, J.J. Effects of chloroquine on lysosomal enzymes, NADPH-induced lipid peroxidation, and antioxidant enzymes of rat retina. Biochem. Pharmacol. 1983, 32, 2965–2968. [Google Scholar] [CrossRef]
- Tacconelli, E.; Tumbarello, M.; Camilli, G.; Bertagnolio, S. Case report: Retinopathy after malaria prophylaxis with chloroquine. Am. J. Trop. Med. Hyg. 2001, 65, 637–638. [Google Scholar] [CrossRef][Green Version]
- Vera, M.; Torramade-Moix, S.; Martin-Rodriguez, S.; Cases, A.; Cruzado, J.M.; Rivera, J.; Escolar, G.; Palomo, M.; Diaz-Ricart, M. Antioxidant and Anti-Inflammatory Strategies Based on the Potentiation of Glutathione Peroxidase Activity Prevent Endothelial Dysfunction in Chronic Kidney Disease. Cell. Physiol. Biochem. 2018, 51, 1287–1300. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; Gao, Y.; Guo, X.; Zhang, M.; Gong, L. Blackberry and Blueberry Anthocyanin Supplementation Counteract High-Fat-Diet-Induced Obesity by Alleviating Oxidative Stress and Inflammation and Accelerating Energy Expenditure. Oxid. Med. Cell Longev. 2018, 2018, 4051232. [Google Scholar] [CrossRef]
- He, Y.; Yue, Y.; Zheng, X.; Zhang, K.; Chen, S.; Du, Z. Curcumin, inflammation, and chronic diseases: How are they linked? Molecules 2015, 20, 9183–9213. [Google Scholar] [CrossRef]
- Zinellu, A.; Zinellu, E.; Sotgiu, E.; Fois, A.G.; Paliogiannis, P.; Scano, V.; Piras, B.; Sotgia, S.; Mangoni, A.A.; Carru, C.; et al. Systemic transsulfuration pathway thiol concentrations in chronic obstructive pulmonary disease patients. Eur. J. Clin. Investig. 2020, e13267. [Google Scholar] [CrossRef]
- Bourgonje, A.R.; Feelisch, M.; Faber, K.N.; Pasch, A.; Dijkstra, G.; van Goor, H. Oxidative Stress and Redox-Modulating Therapeutics in Inflammatory Bowel Disease. Trends. Mol. Med. 2020. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; Yao, L.; Shi, J.; Li, J.; Xu, C. Protective effects of N-acetylcysteine on a chemical-induced murine model of asthma. J. Asthma 2020, 1–8. [Google Scholar] [CrossRef]
- Nadeem, A.; Siddiqui, N.; Alharbi, N.O.; Alharbi, M.M.; Imam, F.; Sayed-Ahmed, M.M. Glutathione modulation during sensitization as well as challenge phase regulates airway reactivity and inflammation in mouse model of allergic asthma. Biochimie 2014, 103, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Wasyanto, T.; Yasa’, A.; Jalaludinsyah, A. Effect of Oral N-Acetylcysteine Supplementation on the Immunity System in Patients with Acute Myocardial Infarction. Acta Med. Indones 2019, 51, 311–317. [Google Scholar] [PubMed]
- Ceriello, A.; Giugliano, D.; Quatraro, A.; Lefebvre, P.J. Anti-oxidants show an anti-hypertensive effect in diabetic and hypertensive subjects. Clin. Sci. 1991, 81, 739–742. [Google Scholar] [CrossRef] [PubMed]
- Janssen-Heininger, Y.; Reynaert, N.L.; van der Vliet, A.; Anathy, V. Endoplasmic reticulum stress and glutathione therapeutics in chronic lung diseases. Redox Biol. 2020, 33, 101516. [Google Scholar] [CrossRef]
- Sanguinetti, C.M. N-acetylcysteine in COPD: Why, how, and when? Multidiscip. Respir. Med. 2016, 11. [Google Scholar] [CrossRef]
- Sgarbanti, R.; Nencioni, L.; Amatore, D.; Coluccio, P.; Fraternale, A.; Sale, P.; Mammola, C.L.; Carpino, G.; Gaudio, E.; Magnani, M.; et al. Redox regulation of the influenza hemagglutinin maturation process: A new cell-mediated strategy for anti-influenza therapy. Antioxid. Redox Signal. 2011, 15, 593–606. [Google Scholar] [CrossRef]
- Checconi, P.; De Angelis, M.; Marcocci, M.E.; Fraternale, A.; Magnani, M.; Palamara, A.T.; Nencioni, L. Redox-Modulating Agents in the Treatment of Viral Infections. Int. J. Mol. Sci. 2020, 21. [Google Scholar] [CrossRef]
- Lamson, D.W.; Brignall, M.S. The use of nebulized glutathione in the treatment of emphysema: A case report. Altern. Med. Rev. 2000, 5, 429–431. [Google Scholar] [PubMed]
- Horowitz, R.I.; Freeman, P.R.; Bruzzese, J. Efficacy of glutathione therapy in relieving dyspnea associated with COVID-19 pneumonia: A report of 2 cases. Respir. Med. Case Rep. 2020, 101063. [Google Scholar] [CrossRef] [PubMed]
- Sekhar, R.V.; Patel, S.G.; Guthikonda, A.P.; Reid, M.; Balasubramanyam, A.; Taffet, G.E.; Jahoor, F. Deficient synthesis of glutathione underlies oxidative stress in aging and can be corrected by dietary cysteine and glycine supplementation. Am. J. Clin. Nutr. 2011, 94, 847–853. [Google Scholar] [CrossRef] [PubMed]
- Cazzola, M.; Calzetta, L.; Page, C.; Jardim, J.; Chuchalin, A.G.; Rogliani, P.; Gabriella Matera, M. Influence of N-acetylcysteine on chronic bronchitis or COPD exacerbations: A meta-analysis. Eur. Respir. Rev. 2015, 24, 451–461. [Google Scholar] [CrossRef] [PubMed]
- Silvagno, F.; Vernone, A.; Pescarmona, G.P. COVID-19: Can glutathione (GSH) help to reduce severe symptoms? Available online: https://covid-19.conacyt.mx/jspui/bitstream/1000/2366/1/1101532.pdf (accessed on 27 June 2020).
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Silvagno, F.; Vernone, A.; Pescarmona, G.P. The Role of Glutathione in Protecting against the Severe Inflammatory Response Triggered by COVID-19. Antioxidants 2020, 9, 624. https://doi.org/10.3390/antiox9070624
Silvagno F, Vernone A, Pescarmona GP. The Role of Glutathione in Protecting against the Severe Inflammatory Response Triggered by COVID-19. Antioxidants. 2020; 9(7):624. https://doi.org/10.3390/antiox9070624
Chicago/Turabian StyleSilvagno, Francesca, Annamaria Vernone, and Gian Piero Pescarmona. 2020. "The Role of Glutathione in Protecting against the Severe Inflammatory Response Triggered by COVID-19" Antioxidants 9, no. 7: 624. https://doi.org/10.3390/antiox9070624
APA StyleSilvagno, F., Vernone, A., & Pescarmona, G. P. (2020). The Role of Glutathione in Protecting against the Severe Inflammatory Response Triggered by COVID-19. Antioxidants, 9(7), 624. https://doi.org/10.3390/antiox9070624





