Carotenoids and Chlorophylls as Antioxidants
Abstract
1. Introduction
2. Methods Used to Determine the Antioxidant Capacity of Lipophilic Pigments
2.1. Hydrogen-Atom-Transfer-Based (HAT) Assays
2.2. Single-Electron-Transfer-Based Assays
2.3. Mixed-Mode Assays
2.4. Antioxidant Biomarkers Assessed by In Vivo and Ex Vivo Assays
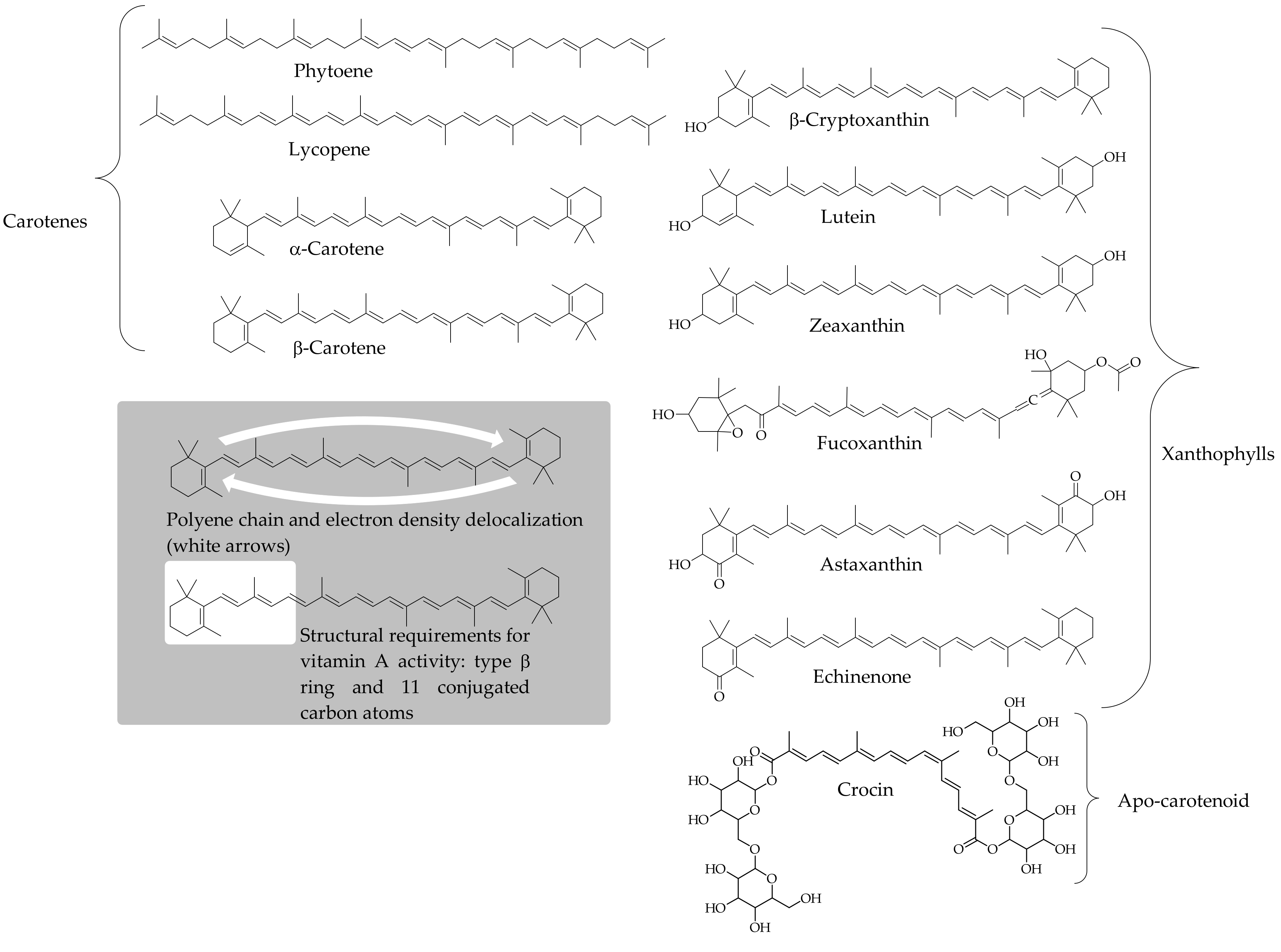
3. Carotenoids as Antioxidants: From Magic Bullets to Dietary Trends
3.1. A Conceivable Basis to Consider Carotenoids as Dietary Antioxidants
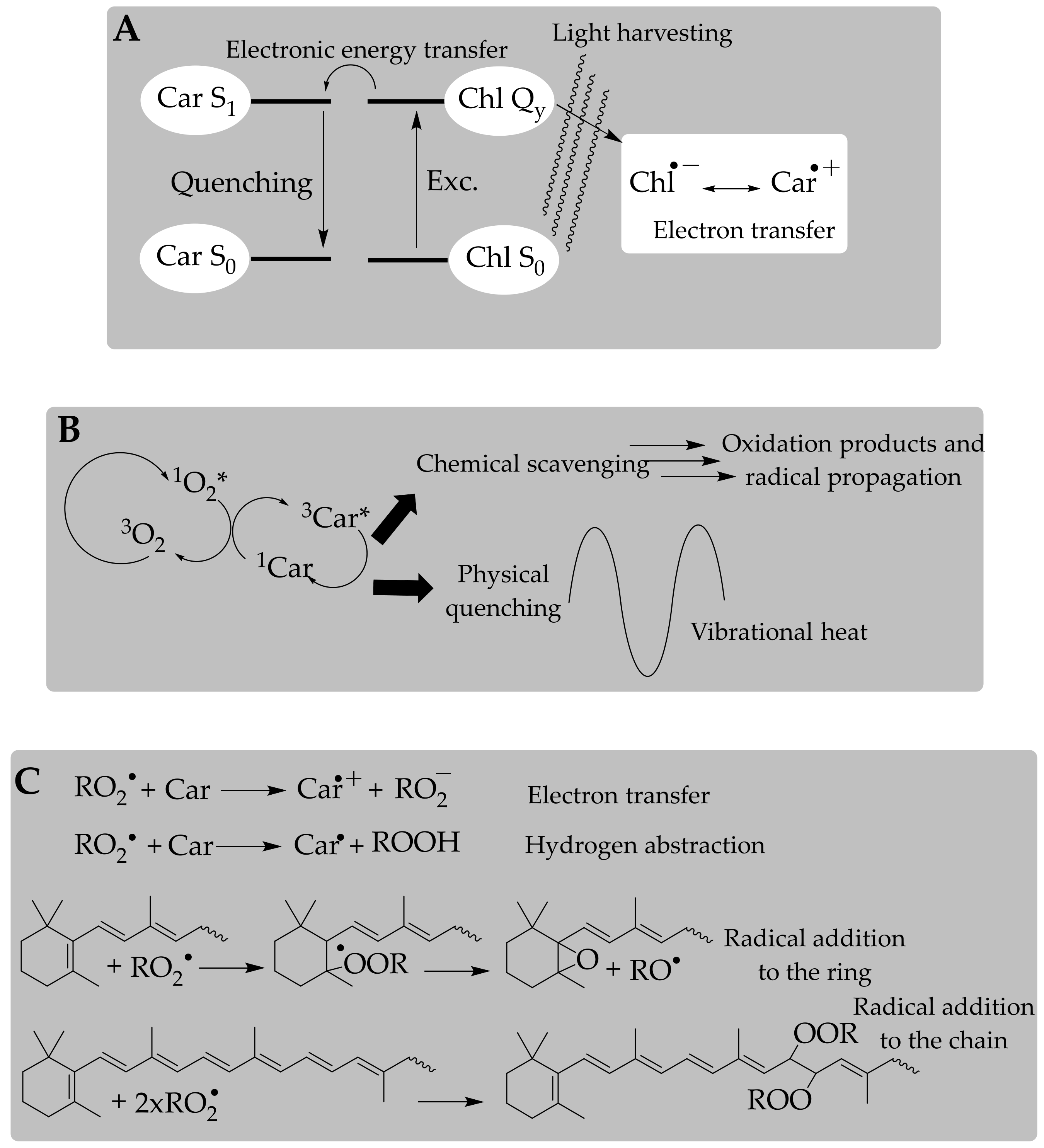
3.2. Antioxidant Activity of Carotenoids: In Vitro Approach of a Chemical Process
3.3. Antioxidant Capacity of Carotenoids: A Further Step to Estimate the In Vivo Antioxidant Action
3.3.1. Measurement of the Antioxidant Capacity in Liposomes
3.3.2. Measurement of the Antioxidant Capacity in LDLs
3.3.3. Measurement of the Antioxidant Capacity in Cell Models: Carotenoid Oxidation Products
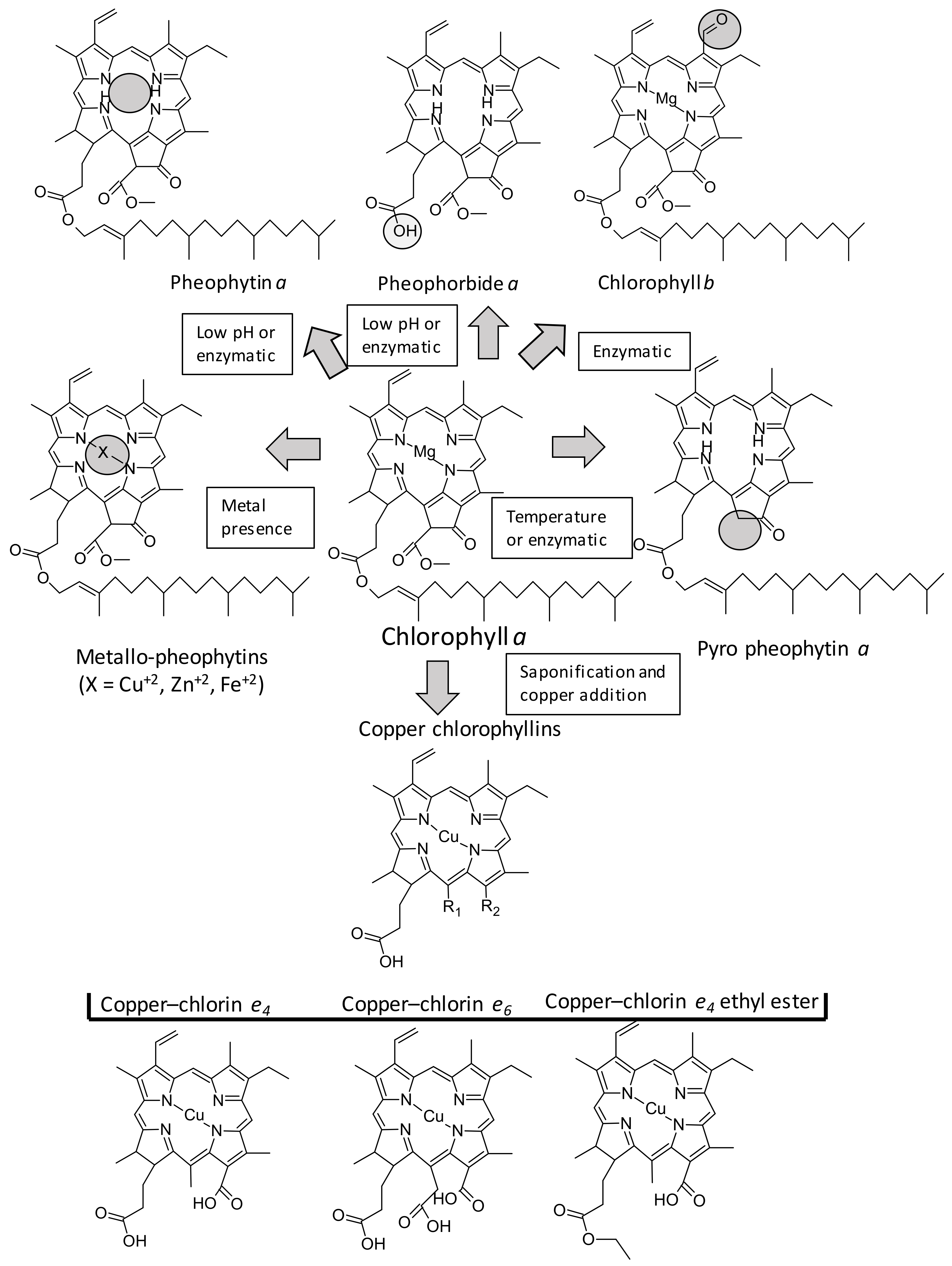
4. Antioxidant Capacity of Chlorophylls
4.1. Different Chlorophyll Standards
4.2. Antioxidant Activity of Chlorophyll Extracts from Different Sources
4.3. In Vivo Antioxidant Activity of Chlorophylls
4.3.1. In Vivo Free-Radical-Scavenging Properties of Chlorophylls
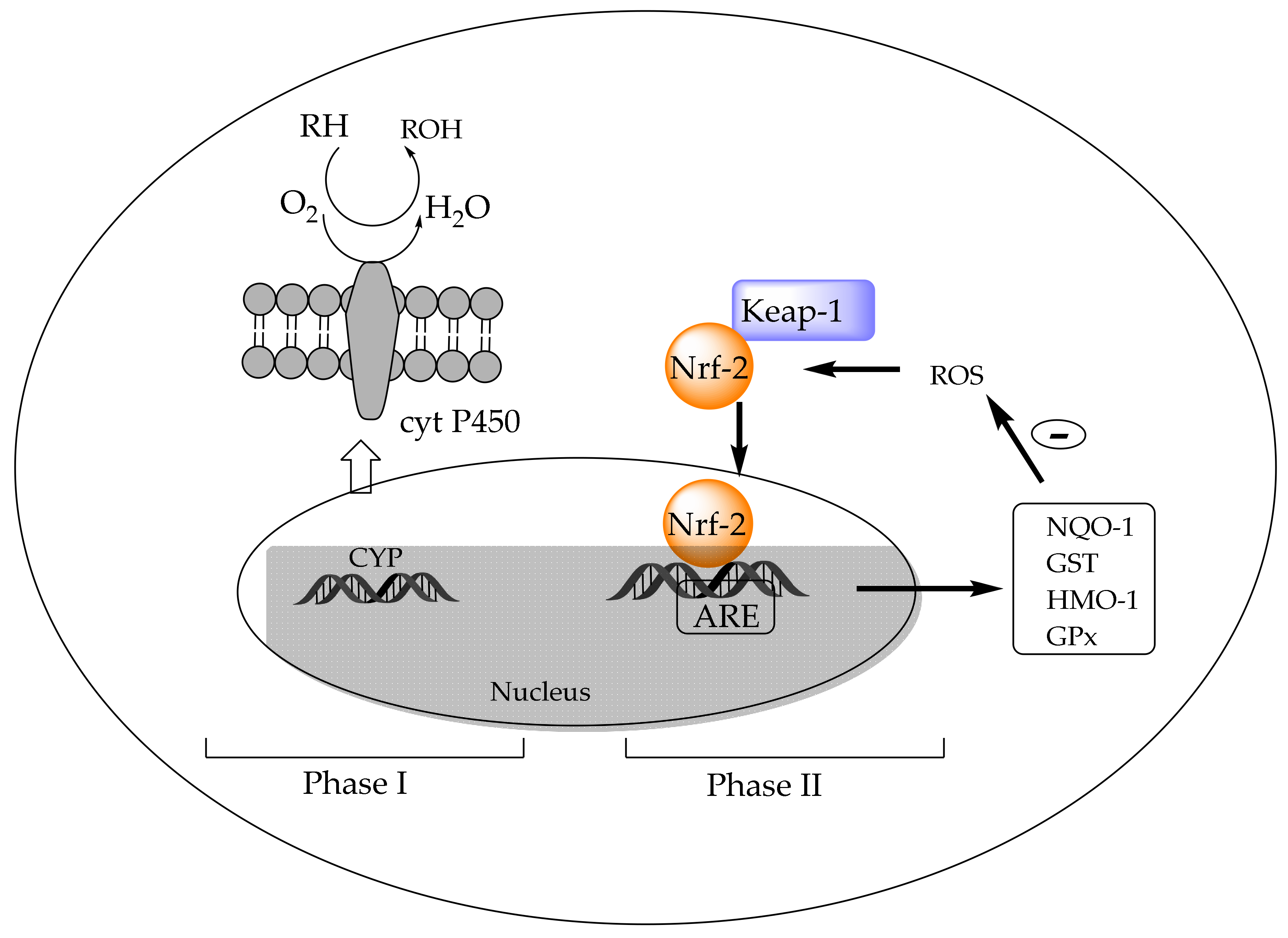
4.3.2. In Vivo Activation of Detoxification Pathways by Chlorophylls
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Sun, Y.; Yang, C.; Tsao, R. Nomenclature and general classification of antioxidant activity/capacity assays. In Measurement of Antioxidant Activity & Capacity: Recent Trends and Applications; Apak, R., Capanoglu, E., Shahidi, F., Eds.; John Wiley & Sons Ltd.: Hoboken, NJ, USA, 2018; Chapter 1; pp. 1–19. [Google Scholar]
- Capanoglu, E.; Kamiloglu, S.; Ozkan, G.; Apak, R. Evaluation of antioxidant activity/capacity measurement methods for food products. In Measurement of Antioxidant Activity & Capacity: Recent Trends and Applications; Apak, R., Capanoglu, E., Shahidi, F., Eds.; John Wiley & Sons Ltd.: Hoboken, NJ, USA, 2018; Chapter 13; pp. 273–286. [Google Scholar]
- Halliwell, B. How to Characterize a Biological Antioxidant. Free Radic. Res. Commun. 1990, 9, 1–32. [Google Scholar] [CrossRef]
- Apak, R.; Özyürek, M.; Güçlü, K.; Çapanoğlu, E. Antioxidant Activity/Capacity Measurement. 1. Classification, Physicochemical Principles, Mechanisms, and Electron Transfer (ET)-Based Assays. J. Agric. Food Chem. 2016, 64, 997–1027. [Google Scholar] [CrossRef]
- Pellegrini, N.; Vitaglione, P.; Granato, D.; Fogliano, V. Twenty-five years of total antioxidant capacity measurement of foods and biological fluids: Merits and limitations. J. Sci. Food Agric. 2018. [Google Scholar] [CrossRef] [PubMed]
- Munialo, C.D.; Naumovski, N.; Sergi, D.; Stewart, D.; Mellor, D.D. Critical evaluation of the extrapolation of data relative to antioxidant function from the laboratory and their implications on food production and human health: A review. Int. J. Food Sci. Technol. 2019, 54, 1448–1459. [Google Scholar] [CrossRef]
- Cao, G.; Alessio, H.M.; Cutler, R.G. Oxygen-radical absorbance capacity assay for antioxidants. Free Rad. Biol. Med. 1993, 14, 303–311. [Google Scholar] [CrossRef]
- Cao, G.; Prior, R.L. Measurement of oxygen radical absorbance capacity in biological samples. Methods Enzymol. 1999, 299, 50–62. [Google Scholar]
- Frankel, E.N.; Meyer, A.S. The problem of using one-dimensional methods to evaluate multifunctional food and biological antioxidants. J. Sci. Food Agric. 2000, 80, 1925–1941. [Google Scholar] [CrossRef]
- Karadag, A.; Ozcelik, B.; Saner, S. Review of Methods to Determine Antioxidant Capacities. Food Anal. Methods 2009, 2, 41–60. [Google Scholar] [CrossRef]
- Ou, B.; Hampsch-Woodill, M.; Prior, R.L. Development and Validation of an Improved Oxygen Radical Absorbance Capacity Assay Using Fluorescein as the Fluorescent Probe. J. Agric. Food Chem. 2001, 49, 4619–4626. [Google Scholar] [CrossRef]
- Prior, R.L.; Hoang, H.; Gu, L.; Wu, X.; Bacchioca, M.; Howard, L.; Hampsch-Woodill, M.; Huang, D.; Ou, B.; Jacob, R. Assays for hydrophilic and lipophilic antioxidant capacity (oxygen radical absorbance capacity (ORACFL)) of plasma and other biological and food samples. J. Agric. Food Chem. 2003, 51, 3273–3279. [Google Scholar] [CrossRef]
- Anderson, S.M.; Krinsky, N.I. Protective action of carotenoid pigments against photodynamic damage to liposomes. Photochem. Photobiol. 1973, 18, 403–408. [Google Scholar] [CrossRef] [PubMed]
- Krinsky, N.I.; Deneke, S.M. Interaction of oxygen and oxy-radicals with carotenoids. J. Natl. Cancer Inst. 1982, 69, 205–210. [Google Scholar]
- Miki, W. Biological functions and activities of animal carotenoids. Pure App. Chem. 1991, 63, 141–146. [Google Scholar] [CrossRef]
- Hill, J.; Clarke, J.D.; Vargesson, N.; Jowett, T.; Holder, N. Exogenous retinoic acid causes specific alterations in the development of the midbrain and hindbrain of the zebrafish embryo including positional respecification of the Mauthner neuron. Mech. Dev. 1995, 50, 3–16. [Google Scholar] [CrossRef]
- Rengel, D.; Díez-Navajas, A.; Serna-Rico, A.; Veiga, P.; Muga, A.; Milicua, J.C. Exogenously incorporated ketocarotenoids in large unilamellar vesicles. Protective activity against peroxidation. Biochim. Biophys. Acta 2000, 1463, 179–187. [Google Scholar] [CrossRef]
- Matsushita, Y.; Suzuki, R.; Nara, E.; Yokoyama, A.; Miyashita, K. Antioxidant activity of polar carotenoids including astaxanthin-β-glucoside from marine bacterium on PC liposomes. Fisheries Sci. 2000, 66, 980–985. [Google Scholar] [CrossRef]
- Cvetković, D.J.; Stanojević, J.; Cakić, M.; Marković, D. Investigation into the potential chemical mechanism of carotenoids pro-oxidant activity with liposomes under UV-irradiation. J. Serb. Chem. Soc. 2017, 82, 1009–1023. [Google Scholar] [CrossRef]
- Porter, W.L. Recent trends in food applications of antioxidants. In Autoxidation in Food and Biological Systems; Simic, M.G., Karel, M., Eds.; Plenum Press: New York, NY, USA, 1980; pp. 295–365. [Google Scholar]
- Frankel, E.N.; Huang, S.-W.; Kanner, J.; German, J.B. Interfacial phenomena in the evaluation of antioxidants: Bulk oils vs emulsion. J. Agric. Food Chem. 1994, 42, 1054–1059. [Google Scholar] [CrossRef]
- Laguerre, M.; Bayrasy, C.; Panya, A.; Weiss, J.; McClements, D.J.; Lecomte, J.; Decker, E.A.; Villeneuve, P. What makes good antioxidants in lipid-based systems? The next theories beyond the polar paradox. Crit. Rev. Food Sci. Nutr. 2015, 55, 183–201. [Google Scholar] [CrossRef]
- Kevers, C.; Falkowski, M.; Tabart, J.; Defraigne, J.-O.; Dommes, J.; Pincemail, J. Evolution of Antioxidant Capacity during Storage of Selected Fruits and Vegetables. J. Agric. Food Chem. 2007, 55, 8596–8603. [Google Scholar] [CrossRef]
- Wayner, D.D.M.; Burton, G.W.; Ingold, K.U.; Locke, S. Quantitative measurement of the total, peroxyl radical-trapping antioxidant capability of human blood plasma by controlled peroxidation: The important contribution made by plasma proteins. FEBS Lett. 1985, 187, 33–37. [Google Scholar] [CrossRef]
- Rice-Evans, C.; Miller, N.J. [241 Total antioxidant status in plasma and body fluids. In Methods in Enzymology; Elsevier: Amsterdam, The Netherlands, 1994; Volume 234, pp. 279–293. ISBN 9780121821357. [Google Scholar]
- Mueller, L.; Boehm, V. Antioxidant Activity of β-Carotene Compounds in Different In Vitro Assays. Molecules 2011, 16, 1055–1069. [Google Scholar] [CrossRef] [PubMed]
- Wright, J.S.; Johnson, E.R.; DiLabio, G.A.; DiLabio, G.A. Predicting the activity of phenolic antioxidants: Theoretical method, analysis of substituent effects, and application to major families of antioxidants. J. Am. Chem. Soc. 2001, 123, 1173–1183. [Google Scholar] [CrossRef] [PubMed]
- Benzie, I.F.F.; Strain, J.J. The Ferric Reducing Ability of Plasma (FRAP) as a Measure of “Antioxidant Power”: The FRAP Assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef]
- Benzie, I.F.; Strain, J.J. Ferric reducing/antioxidant power assay: Direct measure of total antioxidant activity of biological fluids and modified version for simultaneous measurement of total antioxidant power and ascorbic acid concentration. Methods Enzymol. 1999, 299, 15–27. [Google Scholar]
- Halvorsen, B.L.; Holte, K.; Myhrstad, M.C.W.; Barikmo, I.; Hvattum, E.; Remberg, S.F.; Wold, A.-B.; Haffner, K.; Baugerød, H.; Andersen, L.F.; et al. A Systematic Screening of Total Antioxidants in Dietary Plants. Nutr. J. 2002, 132, 461–471. [Google Scholar] [CrossRef]
- Payne, A.C.; Mazzer, A.; Clarkson, G.J.J.; Taylor, G. Antioxidant assays—Consistent findings from FRAP and ORAC reveal a negative impact of organic cultivation on antioxidant potential in spinach but not watercress or rocket leaves. Food Sci. Nutr. 2013, 1, 439–444. [Google Scholar] [CrossRef]
- Apak, R.; Güçlü, K.; Özyürek, M.; Karademir, S.E. Novel Total Antioxidant Capacity Index for Dietary Polyphenols and Vitamins C and E, Using Their Cupric Ion Reducing Capability in the Presence of Neocuproine: CUPRAC Method. J. Agric. Food Chem. 2004, 52, 7970–7981. [Google Scholar] [CrossRef]
- Serafini, M.; Del Rio, D. Understanding the association between dietary antioxidants, redox status and disease: Is the Total Antioxidant Capacity the right tool? Redox Rep. 2004, 9, 145–152. [Google Scholar] [CrossRef]
- Apak, R.; Güçlü, K.; Demirata, B.; Özyürek, M.; Çelik, S.; Bektaşoğlu, B.; Berker, K.; Özyurt, D. Comparative Evaluation of Various Total Antioxidant Capacity Assays Applied to Phenolic Compounds with the CUPRAC Assay. Molecules 2007, 12, 1496–1547. [Google Scholar] [CrossRef]
- Prior, R.L.; Wu, X.; Schaich, K. Standardized methods for the determination of antioxidant capacity and phenolics in foods and dietary supplements. J. Agric. Food Chem. 2005, 18, 4290–4302. [Google Scholar] [CrossRef]
- Nilsson, J.; Pillai, D.; Önning, G.; Persson, C.; Nilsson, Å.; Åkesson, B. Comparison of the 2,2′-azinobis-3-ethylbenzotiazo-line-6-sulfonic acid (ABTS) and ferric reducing anti-oxidant power (FRAP) methods to asses the total antioxidant capacity in extracts of fruit and vegetables. Mol. Nutr. Food Res. 2005, 49, 239–246. [Google Scholar] [CrossRef]
- Sözgen Başkan, K.; Tütem, E.; Özer, N.; Apak, R. Spectrophotometric and Chromatographic Assessment of Contributions of Carotenoids and Chlorophylls to the Total Antioxidant Capacities of Plant Foods. J. Agric. Food Chem. 2013, 61, 11371–11381. [Google Scholar] [CrossRef]
- Shalaby, E.A.; Sanaa, S.M.M. Comparison of DPPH and ABTS assays for determining antioxidant potential of water and methanol extracts of Spirulina platensis. Indian J. Mar. Sci. 2013, 42, 556–564. [Google Scholar]
- Jiménez-Escrig, A.; Jiménez-Jiménez, I.; Pulido, R.; Saura-Calixto, F. Antioxidant activity of fresh and processed edible seaweeds: Antioxidant activity of seaweeds. J. Sci. Food Agric. 2001, 81, 530–534. [Google Scholar] [CrossRef]
- Arnao, M.B. Some methodological problems in the determination of antioxidant activity using chromogen radicals: A practical case. Trends Food Sci. Technol. 2000, 11, 419–421. [Google Scholar] [CrossRef]
- Böhm, V.; Puspitasari-Nienaber, N.L.; Ferruzzi, M.G.; Schwartz, S.J. Trolox Equivalent Antioxidant Capacity of Different Geometrical Isomers of α-Carotene, β-Carotene, Lycopene, and Zeaxanthin. J. Agric. Food Chem. 2002, 50, 221–226. [Google Scholar] [CrossRef]
- Awika, J.M.; Rooney, L.W.; Wu, X.; Prior, R.L.; Cisneros-Zevallos, L. Screening Methods to Measure Antioxidant Activity of Sorghum (Sorghum bicolor) and Sorghum Products. J. Agric. Food Chem. 2003, 51, 6657–6662. [Google Scholar] [CrossRef]
- Arnao, M.B.; Cano, A.; Acosta, M. The hydrophilic and lipophilic contribution to total antioxidant activity. Food Chem. 2001, 73, 239–244. [Google Scholar] [CrossRef]
- Blois, M.S. Antioxidant Determinations by the Use of a Stable Free Radical. Nature 1958, 181, 1199–1200. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Plank, D.W.; Szpylka, J.; Sapirstein, H.; Woollard, D.; Zapf, C.M.; Lee, V.; Chen, C.-Y.O.; Liu, R.H.; Tsao, R.; Düsterloh, A.; et al. Determination of Antioxidant Activity in Foods and Beverages by Reaction with 2,2′-Diphenyl-1-Picrylhydrazyl (DPPH): Collaborative Study First Action 2012.04. J. AOAC Int. 2012, 95, 1562–1569. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decoloration assay. Free Rad. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Roginsky, V.; Lissi, E. Review of methods to determine chain-breaking antioxidant activity in food. Food Chem. 2005, 92, 235–254. [Google Scholar] [CrossRef]
- Ghiselli, A.; Serafini, M.; Maiani, G.; Azzini, E.; Ferro-Luzzi, A. A fluorescence-based method for measuring total plasma antioxidant capability. Free Radic. Biol. Med. 1995, 18, 29–36. [Google Scholar] [CrossRef]
- Miller, N.J.; Rice-Evans, C.; Davies, M.J.; Gopinathan, V.; Milner, A. A Novel Method for Measuring Antioxidant Capacity and its Application to Monitoring the Antioxidant Status in Premature Neonates. Clin. Sci. 1993, 84, 407–412. [Google Scholar] [CrossRef]
- Adom, K.K.; Liu, R.H. Rapid Peroxyl Radical Scavenging Capacity (PSC) Assay for Assessing both Hydrophilic and Lipophilic Antioxidants. J. Agric. Food Chem. 2005, 53, 6572–6580. [Google Scholar] [CrossRef]
- Huang, D.; Ou, B.; Prior, R.L. The Chemistry behind Antioxidant Capacity Assays. J. Agric. Food Chem. 2005, 53, 1841–1856. [Google Scholar] [CrossRef]
- Wolfe, K.L.; Liu, R.H. Cellular Antioxidant Activity (CAA) Assay for Assessing Antioxidants, Foods, and Dietary Supplements. J. Agric. Food Chem. 2007, 55, 8896–8907. [Google Scholar] [CrossRef]
- Yang, C.; Shahidi, F.; Tsao, R. Biomarkers of oxidative stress and cellular-based assays of indirect antioxidant measurement. In Measurement of Antioxidant Activity & Capacity: Recent Trends and Applications; Apak, R., Capanoglu, E., Shahidi, F., Eds.; John Wiley & Sons Ltd.: Hoboken, NJ, USA, 2018; pp. 165–186. [Google Scholar]
- FDA. Nutrient Content Claims for “Good Source”, “High”, “More”, and “High Potency”. Available online: https://www.ecfr.gov/cgi-bin/text-idx?SID=f157cbe6acc56dd4486f7ff998e27aa8&mc=true&node=se21.2.101_154&rgn=div8. (accessed on 20 May 2020).
- Turck, D.; Bresson, J.L.; Burlingame, B.; Dean, T.; Fairweather-Tait, S.; Heinonen, M.; Hirsch-Ernst, K.; Mangelsdorf, I.; Mcardle, H.; Naska, A.; et al. Guidance for the scientific requirements for health claims related to antioxidants, oxidative damage and cardiovascular health. EFSA J. 2018, 16, e05136. [Google Scholar] [CrossRef]
- Giovannucci, E.; Ascherio, A.; Rimm, E.B.; Stampfer, M.J.; Colditz, G.A.; Willett, W.C. Intake of carotenoids and retinol in relation to risk of prostate cancer. J. Natl. Cancer Inst. 1995, 87, 1767–1776. [Google Scholar] [CrossRef] [PubMed]
- Cooper, D.A. Carotenoids in health and disease: Recent scientific evaluations, research recommendations and the consumer. J. Nutr. 2004, 134, 221S–224S. [Google Scholar] [CrossRef] [PubMed]
- Tapiero, H.; Townsend, D.M.; Tew, K.D. The role of carotenoids in the prevention of human pathologies. Biomed. Pharmacother. 2004, 58, 100–110. [Google Scholar] [CrossRef] [PubMed]
- Hu, F.; Wang, Y.B.; Zhang, W.; Liang, J.; Lin, C.; Li, D.; Wang, F.; Pang, D.; Zhao, Y. Carotenoids and breast cancer risk: A meta-analysis and meta-regression. Breast Canc. Res. Treat. 2012, 131, 239–253. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Chung, S.J.; McCullough, M.L.; Song, W.O.; Fernandez, M.L.; Koo, S.I.; Chun, O.K. Dietary carotenoids are associated with cardiovascular disease risk biomarkers mediated by serum carotenoid concentrations. J. Nutr. 2014, 144, 1067–1074. [Google Scholar] [CrossRef]
- Arathi, B.P.; Sowmya, P.R.R.; Vijay, K.; Baskaran, V.; Lakshminarayana, R. Metabolomics of carotenoids: The challenges and prospects—A review. Trends Food Sci. Technol. 2015, 45, 105–117. [Google Scholar] [CrossRef]
- World Cancer Research Fund, Continuous Update Project. Cancer Prevention Recommendations. Available online: https://www.wcrf.org/int/continuous-update-project (accessed on 20 March 2020).
- Linnewiel, K.; Ernst, H.; Caris-Veyrat, C.; Ben-Dor, A.; Kampf, A.; Salman, H.; Danilenko, M.; Levy, J.; Sharoni, Y. Structure activity relationship of carotenoid derivatives in activation of the electrophile/antioxidant response element transcription system. Free Radic. Biol. Med. 2009, 47, 659–667. [Google Scholar] [CrossRef]
- Ford, N.A.; Elsen, A.C.; Zuniga, K.; Lindshield, B.L.; Erdman, J.W., Jr. Lycopene and apo-120-lycopenal reduce cell proliferation and alter cell cycle progression in human prostate cancer cell. Nutr. Cancer 2011, 63, 256–263. [Google Scholar] [CrossRef]
- Mein, J.R.; Dolnikowski, G.G.; Ernst, H.; Russell, R.M.; Wang, X.D. Enzymatic formation of apo-carotenoids from the xanthophyll carotenoids lutein, zeaxanthin, and β-cryptoxanthin by ferret carotene-9′,10′-monooxygenase. Arch. Biochem. Biophys. 2011, 506, 109–121. [Google Scholar] [CrossRef]
- Bohn, T.; McDougall, G.J.; Alegría, A.; Alminger, M.; Arrigoni, E.; Aura, A.-M.; Brito, C.; Cilla, A.; El, S.N.; Karakaya, S.; et al. Mind the gap—Deficits in our knowledge of aspects impacting the bioavailability of phytochemicals and their metabolites—A position paper focusing on carotenoids and polyphenols. Mol. Nutr. Food Res. 2015, 59, 1307–1323. [Google Scholar] [CrossRef]
- Desmarcherlier, C.; Borel, P. Overview of carotenoid bioavailability determinants: From dietary factors to host genetic variations. Trends Food Sci. Nutr. 2017, 69, 270–280. [Google Scholar] [CrossRef]
- Winklhofer-Roob, B.M.; Rock, E.; Ribalta, J.; Shmerling, D.H.; Roob, J.M. Effects of vitamin E and carotenoid status on oxidative stress in health and disease. Evidence obtained from human intervention studies. Mol. Aspects Med. 2003, 24, 391–402. [Google Scholar] [CrossRef]
- Sommer, A.; Vyas, K.S. A global clinical view on vitamin A and carotenoids. Am. J. Clin. Nutr. 2012, 96, 1204S–1206S. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Sola, M.A.; Rodríguez-Concepción, M. Carotenoid biosynthesis in Arabidopsis: A colorful pathway. Arab. Book 2012, 10, e0158. [Google Scholar] [CrossRef] [PubMed]
- Liaaen-Jensen, S. Basic Carotenoid Chemistry. In Carotenoids in Health and Disease, 1st ed.; Krinsky, N.I., Mayne, S.T., Sies, H., Eds.; Marcel Dekker: New York, NY, USA, 2004; pp. 1–30. [Google Scholar]
- Khachik, F.; Beecher, G.R.; Goli, M.B. Separation, identification, and quantification of carotenoids in fruits, vegetables and human plasma by high performance liquid chromatography. Pure Appl. Chem. 1991, 63, 71–80. [Google Scholar] [CrossRef]
- Viera, I.; Pérez-Gálvez, A.; Roca, M. Bioaccessibility of marine carotenoids. Mar. Drugs 2018, 16, 397. [Google Scholar] [CrossRef]
- Maiani, G.; Caston, M.J.; Catasta, G.; Toti, E.; Cambrodon, I.G.; Bysted, A.; Granado-Lorencio, F.; Olmedilla-Alonso, B.; Knuthsen, P.; Valoti, M.; et al. Carotenoids: Actual knowledge on food sources, intakes, stability and bioavailability and their protective role in humans. Mol. Nutr. Food Res. 2009, 53, S194–S218. [Google Scholar] [CrossRef]
- Landrum, J.; Bone, R. Lutein, zeaxanthin, and the macular pigment. Arch. Biochem. Biophys. 2001, 385, 28–40. [Google Scholar] [CrossRef]
- Clinton, S.K.; Emenhiser, C.; Schwartz, S.J.; Bostwick, D.G.; Williams, A.W.; Moore, B.J.; Erdman, J.W., Jr. Cis-trans lycopene isomers, carotenoids, and retinol in the human prostate. Cancer Epidemiol. Biomark. Prev. 1996, 5, 823–833. [Google Scholar]
- Ríos, J.J.; Xavier, A.A.O.; Díaz-Salido, E.; Arenilla-Vélez, I.; Jarén-Galán, M.; Garrido-Fernández, J.; Aguayo-Maldonado, J.; Pérez-Gálvez, A. Xanthophyll esters are found in human colostrum. Mol. Nutr. Food Res. 2017, 61, 1700296. [Google Scholar] [CrossRef]
- Olson, J.A. Benefits and liabilities of vitamin A and carotenoids. Rev. J. Nutr. 1996, 126, 1208S–1212S. [Google Scholar] [CrossRef] [PubMed]
- Bendich, A.; Olson, J.A. Biological actions of carotenoids. FASEB J. 1989, 3, 1927–1932. [Google Scholar] [CrossRef] [PubMed]
- Weber, D.; Grune, T. The contribution of β-carotene to vitamin A supply of humans. Mol. Nutr. Food Res. 2012, 56, 251–258. [Google Scholar] [CrossRef]
- Burton, G.W.; Ingold, K.U. β-Carotene. An unusual type of lipid antioxidant. Science 1984, 224, 569–573. [Google Scholar] [CrossRef] [PubMed]
- Foote, C.S.; Denny, R.W. Chemistry of singlet oxygen. VIII. Quenching by β-carotene. J. Am. Chem. Soc. 1968, 90, 6233–6235. [Google Scholar] [CrossRef]
- Liebler, D.C. Antioxidant reactions of carotenoids. Ann. N. Y. Acad. Sci. 1993, 691, 20–31. [Google Scholar] [CrossRef]
- Sies, H.; Stahl, W.; Sevanian, A. Nutritional, dietary and postprandial oxidative stress. J. Nutr. 2005, 135, 969–972. [Google Scholar] [CrossRef]
- Stahl, W.; Sies, H. Bioactivity and protective effects of natural carotenoids. Biochim. Biophys. Acta 2005, 1740, 101–107. [Google Scholar] [CrossRef]
- Stahl, W.; Sies, H. Carotenoids and flavonoids contribute to nutritional protection against skin damage from sunlight. Mol. Biotechnol. 2007, 37, 26–30. [Google Scholar] [CrossRef]
- Jomova, K.; Kysel, O.; Madden, J.C.; Morris, H.; Enoch, S.J.; Budzak, S.; Young, A.J.; Cronin, M.T.D.; Mazur, M.; Valko, M. Electron transfer from all-trans β-carotene to the t-butyl peroxyl radical at low oxygen pressure (an EPR spectroscopy and computational study). Chem. Phys. Lett. 2009, 478, 266–270. [Google Scholar] [CrossRef]
- Mortensen, A.; Skibsted, L.H. Kinetics of photobleaching of beta-carotene in chloroform and formation of transient carotenoid species absorbing in the near infrared. Free Rad. Res. 1996, 25, 355–368. [Google Scholar] [CrossRef] [PubMed]
- Burke, M.; Edge, R.; Land, E.J.; McGarvey, D.J.; Truscott, T.G. One-electron reduction potentials of dietary carotenoid radical cations in aqueous micellar environments. FEBS Lett. 2001, 500, 132–136. [Google Scholar] [CrossRef]
- Edge, R.; Land, E.J.; McGarvey, D.; Mulroy, L.; Truscott, G. Relative one-electron reduction potentials of carotenoid radical cations and the interactions of carotenoids with the vitamin E radical cation. J. Am. Chem. Soc. 1998, 120, 4087–4090. [Google Scholar] [CrossRef]
- Mortensen, A.; Skibsted, L.H.; Willnow, A.; Everett, S.A. Re-appraisal of the tocopheroxyl radical reaction with beta-carotene: Evidence for oxidation of vitamin E by the beta-carotene radical cation. Free Radic. Res. 1998, 28, 69–80. [Google Scholar] [CrossRef]
- Britton, G. Structure and properties of carotenoids in relation to function. FASEB J. 1995, 9, 1551–1558. [Google Scholar] [CrossRef]
- Edge, R.; McGarvey, D.J.; Truscott, T.G. The carotenoids as antioxidants—A review. J. Photochem. Photobiol. B 1997, 41, 189–200. [Google Scholar] [CrossRef]
- Woodall, A.A.; Lee, S.W.; Weesie, R.j.; Jackson, M.J.; Britton, G. Oxidation of carotenoids by free radicals: Relationship between structure and reactivity. Biochim. Biophys. Acta 1997, 1336, 33–42. [Google Scholar] [CrossRef]
- Pérez-Gálvez, A.; Mínguez-Mosquera, M.I. Degradation of non-esterified and esterified xanthophylls by free radicals. Biochim. Biophys. Acta 2002, 1569, 31–34. [Google Scholar] [CrossRef]
- Terao, J. Antioxidant activity of beta-carotene-related carotenoids in solution. Lipids 1989, 24, 659–661. [Google Scholar] [CrossRef]
- Bartosz, G.; Janaszewska, A.; Ertel, D. Spectrophotomeric determination of peroxyl-radical trapping capacity. Curr. Top. Biphys. 1998, 22, 11–13. [Google Scholar]
- Jomova, K.; Valko, M. Health protective effects of carotenoids and their interactions with other biological antioxidants. Eur. J. Med. Chem. 2013, 70, 102–110. [Google Scholar] [CrossRef]
- Krinsky, N.I. Carotenoids as antioxidants. Nutrition 2001, 17, 815–817. [Google Scholar] [CrossRef]
- Niki, E. Free radical initiators as source of water- or lipid-soluble peroxyl radicals. Methods Enzymol. 1990, 186, 100–108. [Google Scholar] [PubMed]
- Bangham, A.D. The first description of liposomes. Curr. Content 1989, 13, 14. [Google Scholar]
- Tsuchihashi, H.; Kigoshi, M.; Iwatsuki, M.; Niki, E. Action of β-carotene as an antioxidant against lipid peroxidation. Arch. Biochem. Biophys. 1995, 323, 137–147. [Google Scholar] [CrossRef] [PubMed]
- Liebler, D.C.; Stratton, S.P.; Kaysen, K.L. Antioxidant actions of beta-carotene in liposomal and microsomal membranes: Role of carotenoid-membrane incorporation and alpha-tocopherol. Arch. Biochem. Biophys. 1997, 338, 244–250. [Google Scholar] [CrossRef] [PubMed]
- Woodall, A.A.; Britton, G.; Jackson, M.J. Carotenoids and protection of phospholipids in solution or in liposomes against oxidation by peroxyl-radicals: Relationship between carotenoid structure and protective ability. Biochim. Biophys. Acta 1997, 1336, 575–586. [Google Scholar] [CrossRef]
- Sujak, A.; Gabrielska, J.; Grudziński, W.; Borc, R.; Mazurek, P.; Gruszecki, W.I. Lutein and zeaxanthin as protectors of lipid membranes against oxidative damage: The structural aspects. Arch. Biochem. Biophys. 1999, 371, 301–307. [Google Scholar] [CrossRef]
- Barros, M.P.; Pinto, E.; Colepicolo, P.; Pedersén, M. Astaxanthin and peridinin inhibit oxidative damage in Fe(2+)-loaded liposomes: Scavenging oxyradicals or changing membrane permeability? Biochem. Biophys. Res. Commu. 2001, 288, 225–232. [Google Scholar] [CrossRef]
- Wrona, M.; Korytowski, W.; Różanowska, M.; Sarna, T.; Truscott, T.G. Cooperation of antioxidants in protection against photosensitized oxidation. Free Rad. Biol. Med. 2003, 35, 1319–1329. [Google Scholar] [CrossRef]
- Schroeder, M.T.; Becker, E.M.; Skibsted, L.H. Molecular mechanism of antioxidant synergism of tocotrienols and carotenoids in palm oil. J. Agric. Food Chem. 2006, 54, 3445–3453. [Google Scholar] [CrossRef] [PubMed]
- Becker, E.M.; Ntouma, G.; Skibsted, L.H. Synergism and antagonism between quercetin and other chain-breaking antioxidants in lipid systems of increasing structural organization. Food Chem. 2007, 103, 1288–1296. [Google Scholar] [CrossRef]
- McNulty, H.; Jacob, R.F.; Mason, R.P. Biologic activity of carotenoids related to distinct membrane physicochemical interactions. Am. J. Cardiol. 2008, 101, 20D–29D. [Google Scholar] [CrossRef]
- Esterbauer, H.; Striegl, G.; Puhl, H.; Rotheneder, M. Continuous monitoring of in vitro oxidation of human low density lipoprotein. Free Radic. Res. Commun. 1989, 6, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Princen, H.M.; van Poppel, G.; Vogelezang, C.; Buytenhek, R.; Kok, F.J. Supplementation with vitamin E but not β-carotene in vivo protects low density lipoprotein from lipid peroxidation in vitro. Effect of cigarette smoking. Arterioscler. Thromb. Vasc. Biol. 1992, 12, 554–562. [Google Scholar] [CrossRef]
- Hininger, I.; Chopra, M.; Thurnham, D.I.; Laporte, F.; Richard, M.-J.; Favier, A.; Roussel, A.-M. Effect of increased fruit and vegetable intake on the susceptibility of lipoprotein oxidation in smokers. Eur. J. Clin. Nutr. 1997, 51, 601–606. [Google Scholar] [CrossRef] [PubMed]
- Kiokias, S.; Gordon, M.H. Dietary supplementation with a natural carotenoid mixture decreases oxidative stress. Eur. J. Clin. Nutr. 2003, 57, 1135–1140. [Google Scholar] [CrossRef]
- Upritchard, J.E.; Schuurman, C.R.W.C.; Wiersma, A.; Tijburg, L.B.M.; Coolen, S.A.J.; Rijken, P.J.; Wiseman, S.A. Spread supplemented with moderate doses of vitamin E and carotenoids reduces lipid peroxidation in healthy, nonsmoking adults. Am. J. Clin. Nutr. 2003, 78, 985–992. [Google Scholar] [CrossRef]
- Wang, Z.; Ciabattoni, G.; Créminon, C.; Lawson, J.; Fitzgerald, G.A.; Patrono, C.; Maclouf, J. Immunological characterization of urinary 8-epi-prostaglandin F2 excretion in man. J. Pharmacol. Exp. Ther. 1995, 275, 94–100. [Google Scholar]
- Visioli, F.; Riso, P.; Grande, S.; Galli, C.; Porrini, M. Protective activity of tomato products on in vivo markers of lipid oxidation. Eur. J. Nutr. 2003, 42, 201–206. [Google Scholar] [CrossRef]
- Milde, J.; Elstner, E.F.; Grassmann, J. Synergistic effects of phenolics and carotenoids on human low-density lipoprotein oxidation. Mol. Nutr. Food Res. 2007, 51, 956–961. [Google Scholar] [CrossRef]
- Neyestani, T.R.; Shariatzadeh, N.; Gharavi, A.; Kalyi, A.; Khalaji, N. Physiological dose of lycopene suppressed oxidative stress and enhanced serum levels of immunoglobulin M in patients with Type 2 diabetes mellitus: A possible role in the prevention of long-term complications. J. Endocrinol. Investig. 2007, 30, 833–838. [Google Scholar] [CrossRef] [PubMed]
- Devaraj, S.; Mathur, S.; Basu, A.; Aung, H.H.; Vasu, V.T.; Meyers, S.; Jialal, I. A dose-response study on the effects of purified lycopene supplementation on biomarkers of oxidative stress. J. Am. Coll. Nutr. 2008, 27, 267–273. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.Y.; Paik, J.K.; Kim, O.Y.; Park, H.W.; Lee, J.H.; Jang, Y.; Lee, J.H. Effects of lycopene supplementation on oxidative stress and markers of endothelial function in healthy men. Atherosclerosis 2011, 215, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Burri, B.J. Carotenoids and gene expression. Nutrition 2000, 16, 577–578. [Google Scholar] [CrossRef]
- Palozza, P.; Serini, S.; Torsello, A.; Di Nicuolo, F.; Piccioni, E.; Ubaldi, V.; Pioli, C.; Wolf, F.I.; Calviello, G. β-carotene regulates NF-κB DNA-binding activity by a redox mechanism in human leukemia and colon adenocarcinoma cells. J. Nutr. 2003, 133, 381–388. [Google Scholar] [CrossRef]
- Liu, C.L.; Chiu, Y.T.; Hu, M.L. Fucoxanthin enhances HO-1 and NQO1 expression in murine hepatic BNL CL.2 cells through activation of the Nrf2/ARE system partially by its pro-oxidant activity. J. Agric. Food Chem. 2011, 59, 11344–11351. [Google Scholar] [CrossRef]
- Palozza, P.; Catalano, A.; Simone, R.; Cittadini, A. Lycopene as a guardian of redox signalling. Acta Biochim. Pol. 2012, 59, 21–25. [Google Scholar] [CrossRef]
- Stahl, W.; von Laar, J.; Martin, H.D.; Emmerich, T.; Sies, H. Stimulation of gap junctional communication: Comparison of acyclo-retinoic acid and lycopene. Arch. Biochem. Biophys. 2000, 373, 271–274. [Google Scholar] [CrossRef]
- Aust, O.; Ale-Agha, N.; Zhang, L.; Wollersen, H.; Sies, H.; Stahl, W. Lycopene oxidation product enhances gap junctional communication. Food Chem. Toxicol. 2003, 41, 1399–1407. [Google Scholar] [CrossRef]
- Echtay, K.S.; Esteves, T.C.; Pakay, J.L.; Jekabsons, M.B.; Lambert, A.J.; Portero-Otín, M.; Pamplona, R.; Vidal-Puig, A.J.; Wang, S.; Roebuck, S.J.; et al. A signalling role for 4-hydroxy-2-nonenal in regulation of mitochondrial uncoupling. EMBO J. 2003, 22, 4103–4110. [Google Scholar] [CrossRef] [PubMed]
- Siems, W.; Capuozzo, E.; Crifò, C.; Sommerburg, O.; Langhans, C.-D.; Schlipalius, L.; Wiswedel, I.; Kraemer, K.; Salerno, C. Carotenoid cleavage products modify respiratory burst and induce apoptosis of human neutrophils. Biochim. Biophys. Acta 2003, 1639, 27–33. [Google Scholar] [CrossRef]
- Amengual, J.; Lobo, G.P.; Golczak, M.; Li, H.N.; Klimova, T.; Hoppel, C.L.; Wyss, A.; Palczewski, K.; von Lintig, J. A mitochondrial enzyme degrades carotenoids and protects against oxidative stress. FASEB J. 2011, 25, 948–959. [Google Scholar] [CrossRef] [PubMed]
- Martin, K.R.; Failla, M.L.; Smith, J.C. Beta-carotene and lutein protect HepG2 human liver cells against oxidant-induced damage. J. Nutr. 1996, 126, 2098–2106. [Google Scholar] [CrossRef] [PubMed]
- Palozza, P.; Luberto, C.; Calviello, G.; Ricci, P.; Bartoli, G.M. Antioxidant and prooxidant role of beta-carotene in murine normal and tumor thymocytes: Effects of oxygen partial pressure. Free Radic. Biol. Med. 1997, 22, 1065–1073. [Google Scholar] [CrossRef]
- Bohn, T. Carotenoids and Markers of Oxidative Stress in Human Observational Studies and Intervention Trials: Implications for Chronic Diseases. Antioxidants 2019, 8, 179. [Google Scholar] [CrossRef]
- Lowe, G.; Booth, L.A.; Young, A.J.; Bilton, Y.R. Lycopene and b-carotene protect against oxidative damage in HT29 cells at low concentrations but rapidly lose this capacity at higher doses. Free Radic. Res. 1999, 30, 141–151. [Google Scholar] [CrossRef]
- Matos, H.R.; Di Mascio, P.; Medeiros, M.H.G. Protective effect of lycopene on lipid peroxidation and oxidative DNA damage in cell culture. Arch. Biochem. Biophys. 2000, 383, 56–59. [Google Scholar] [CrossRef]
- Yeh, S.-L.; Hu, M.-L. Antioxidant and pro-oxidant effects of lycopene in comparison with b-carotene on oxidant-induced damage in Hs68 cells. J. Nutr. Biochem. 2000, 11, 548–554. [Google Scholar] [CrossRef]
- Offord, E.A.; Gautier, J.C.; Avanti, O.; Scaletta, C.; Runge, F.; Krämer, K.; Applegate, L.A. Photoprotective potential of lycopene, beta-carotene, vitamin E, vitamin C and carnosic acid in UVA-irradiated human skin fibroblasts. Free Radic. Biol. Med. 2002, 32, 1293–1303. [Google Scholar] [CrossRef]
- Ochiai, T.; Ohno, S.; Soeda, S.; Tanaka, H.; Shoyama, Y.; Shimeno, H. Crocin prevents the death of rat pheochromyctoma (PC-12) cells by its antioxidant effects stronger than those of alpha-tocopherol. Neurosci. Lett. 2004, 362, 61–64. [Google Scholar] [CrossRef]
- Chucair, A.J.; Rotstein, N.P.; Sangiovanni, J.P.; During, A.; Chew, E.Y.; Politi, L.E. Lutein and zeaxanthin protect photoreceptors from apoptosis induced by oxidative stress: Relation with docosahexaenoic acid. Investig. Ophthalmol. Vis. Sci. 2007, 48, 5168–5177. [Google Scholar] [CrossRef]
- Ajila, C.M.; Prasada Rao, U.J. Protection against hydrogen peroxide induced oxidative damage in rat erythrocytes by Mangifera indica L. peel extract. Food Chem. Toxicol. 2008, 46, 303–309. [Google Scholar] [CrossRef]
- Camera, E.; Mastofrancesco, A.; Fabbri, C.; Daubrawa, F.; Picardo, M.; Sies, H.; Stahl, W. Astaxanthin, canthaxanthin and beta-carotene differently affect UVA-induced oxidative damage and expression of oxidative stress-responsive enzymes. Exp. Dermatol. 2009, 18, 222–231. [Google Scholar] [CrossRef]
- Wolf, A.M.; Asoh, S.; Hirnuma, H.; Ohsawa, I.; Iio, K.; Satou, A.; Ishikura, M.; Ohta, S. Astaxanthin protects mitochondrial redox state and functional integrity against oxidative stress. J. Nutr. Biochem. 2010, 21, 381–389. [Google Scholar] [CrossRef]
- Yamauchi, M.; Tsuruma, K.; Imai, S.; Nakanishi, T.; Umigai, N.; Shimazawa, M.; Hara, H. Crocetin prevents retinal degeneration induced by oxidative and endoplasmic reticulum stresses via inhibition of caspase activity. Eur. J. Pharmacol. 2011, 650, 110–119. [Google Scholar] [CrossRef]
- Fernández-García, E.; Carvajal-Lérida, I.; Pérez-Gálvez, A. Carotenoids exclusively synthesized in red pepper (capsanthin and capsorubin) protect human dermal fibroblasts against UVB induced DNA damage. Photochem. Photobiol. Sci. 2016, 15, 1204–1211. [Google Scholar] [CrossRef]
- Usuki, R.; Endo, Y.; Kaneda, T. Prooxidant activities of chlorophylls and pheophytins on the photooxidation of edible oils. Agric. Biol. Chem. 1984, 48, 991–994. [Google Scholar] [CrossRef]
- Rigane, G.; Bouaziz, M.; Sayadi, S.; Salem, R.B. Effect of storage on refined olive oil composition: Stabilization by addition of chlorophyll pigments and squalene. J. Oleo Sci. 2013, 62, 981–987. [Google Scholar] [CrossRef]
- Ferruzzi, M.G.; Bohm, V.; Courtney, P.D.; Schwartz, S.J. Antioxidant and Antimutagenic Activity of Dietary Chlorophyll Derivatives Determined by Radical Scavenging and Bacterial Reverse Mutagenesis Assays. J. Food Sci. 2002, 67, 2589–2595. [Google Scholar] [CrossRef]
- Lanfer-Marquez, U.M.; Barros, R.M.C.; Sinnecker, P. Antioxidant activity of chlorophylls and their derivatives. Food Res. Int. 2005, 38, 885–891. [Google Scholar] [CrossRef]
- Nagini, S.; Palitti, F.; Natarajan, A.T. Chemopreventive potential of chlorophyllin: A review of the mechanisms of action and molecular targets. Nutr. Cancer 2015, 67, 203–211. [Google Scholar] [CrossRef]
- Tumolo, T.; Lanfer-Marquez, U.M. Copper chlorophyllin: A food colorant with bioactive properties? Food Res. Int. 2012, 46, 451–459. [Google Scholar] [CrossRef]
- Ferruzzi, M.G.; Blakeslee, J. Digestion, absorption, and cancer preventative activity of dietary chlorophyll derivatives. Nutr. Res. 2007, 27, 1–12. [Google Scholar] [CrossRef]
- Ferruzzi, M.G.; Failla, M.L.; Schwartz, S.J. Assessment of degradation and intestinal cell uptake of carotenoids and chlorophyll derivatives from spinach puree using an in vitro digestion and Caco-2 human cell model. J. Agric. Food Chem. 2001, 49, 2082–2089. [Google Scholar] [CrossRef]
- Gallardo-Guerrero, L.; Gandul-Rojas, B.; Mínguez-Mosquera, M.I. Digestive stability, micellarization, and uptake by Caco-2 human intestinal cell of chlorophyll derivatives from different preparations of pea (Pisum sativum L.). J. Agric. Food Chem. 2008, 56, 8379–8386. [Google Scholar] [CrossRef]
- Gandul-Rojas, B.; Gallardo-Guerrero, L.; Mínguez-Mosquera, M.I. Influence of the chlorophyll pigment structure on its transfer from an oily food matrix to intestinal epithelium cells. J. Agric. Food Chem. 2009, 57, 5306–5314. [Google Scholar] [CrossRef]
- Chen, K.; Roca, M. In vitro digestion of chlorophyll pigments from edible seaweeds. J. Funct. Foods 2018, 40, 400–407. [Google Scholar] [CrossRef]
- Chen, K.; Roca, M. Cooking Effects on Bioaccessibility of Chlorophyll Pigments of the Main Edible Seaweeds. Food Chem. 2019, 295, 101–109. [Google Scholar] [CrossRef]
- Chen, K.; Roca, M. In vitro bioavailability of chlorophyll pigments from edible seaweeds. J. Func. Foods 2018, 41, 25–33. [Google Scholar] [CrossRef]
- Viera, I.; Chen, K.; Ríos, J.J.; Benito, I.; Pérez-Gálvez, A.; Roca, M. First-pass metabolism of chlorophylls in mice. Mol. Nutr. Food Res. 2018, 62, 1800562. [Google Scholar] [CrossRef] [PubMed]
- Egner, P.A.; Stansbury, K.H.; Snyder, E.P.; Rogers, M.E.; Hintz, P.A.; Kensler, T.W. Identification and characterization of chlorin e4 ethyl ester in sera of individuals participating in the chlorophyllin chemoprevention trial. Chem. Res. Toxicol. 2000, 13, 900–906. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, A.S.; Nogara, G.P.; Menezes, C.R.; Cichoski, A.J.; Mercadante, A.Z.; Jacob-Lopes, E.; Zepka, L.Q. Identification of chlorophyll molecules with peroxyl radical scavenger capacity in microalgae Phormidium autumnale using ultrasound-assisted extraction. Food Res. Int. 2017, 99, 1036–1041. [Google Scholar] [CrossRef]
- Hsu, C.Y.; Yang, C.M.; Chen, C.M.; Chao, P.Y.; Hu, S.P. Effects of chlorophyll-related compounds on hydrogen peroxide induced DNA damage within human lymphocytes. J. Agric. Food Chem. 2005, 6, 2746–2750. [Google Scholar] [CrossRef]
- Hoshina, C.; Tomita, K.; Shioi, Y. Antioxidant activity of chlorophylls: Its structure-activity relationship. In Photosynthesis: Mechanisms Effects; Garab, G., Ed.; Springer: Dordrecht, The Netherlands, 1998; Volume 4, pp. 3281–3284. [Google Scholar]
- Suzuki, N.; Hatate, H.; Kanamori, N.; Nomoto, T.; Namiki, M.; Yoda, B. Antioxidative Activity of Chlorophyll Derivatives, Oxygen Stress Relievers, to Superoxide Measured by Using Cypridina Luciferin Analogues. Fish. Sci. 1995, 61, 65–67. [Google Scholar] [CrossRef][Green Version]
- Kang, Y.-R.; Park, J.; Jung, S.K.; Chang, Y.H. Synthesis, characterization, and functional properties of chlorophylls, pheophytins, and Zn-pheophytins. Food Chem. 2018, 245, 943–950. [Google Scholar] [CrossRef]
- Endo, Y.; Usuki, R.; Kaneda, T. Antioxidant effects of chlorophyll and pheophytin on the autoxidation of oils in the dark. II. The mechanism of antioxidative action of chlorophyll. J. Am. Oil Chem. Soc. 1985, 62, 1387–1390. [Google Scholar] [CrossRef]
- Viera, I.; Pérez-Gálvez, A.; Roca, M. Green Natural Colorants. Molecules 2019, 24, 154. [Google Scholar] [CrossRef]
- Sato, M.; Imai, K.; Kimura, R.; Murata, T. Effect of sodium copper chlorophyllin on lipid peroxidation. VI. Effect of its administration on mitochondrial and microsomal lipid peroxidation in rat liver. Chem. Pharm. Bull. 1984, 32, 716–722. [Google Scholar] [CrossRef]
- Gerić, M.; Gajski, G.; Mihaljević, B.; Miljanić, S.; Domijan, A.M.; Garaj-Vrhovac, V. Radioprotective properties of food colorant sodium copper chlorophyllin on human peripheral blood cells in vitro. Mutat. Res. 2019, 845, 403027. [Google Scholar] [CrossRef]
- Zhan, R.; Wu, J.; Ouyang, J. In vitro Antioxidant Activities of Sodium Zinc and Sodium Iron Chlorophyllins from Pine Needles. Food Technol. Biotechnol. 2014, 52, 505–510. [Google Scholar] [CrossRef]
- Hsu, C.-Y.; Chao, P.-Y.; Hu, S.-P.; Yang, C.-M. The Antioxidant and Free Radical Scavenging Activities of Chlorophylls and Pheophytins. Food Nutr. Sci. 2013, 4, 35234. [Google Scholar] [CrossRef]
- Jeon, Y.-M.; Lee, H.-S.; Jeong, D.; Oh, H.-K.; Ra, K.-H.; Lee, M.-Y. Antimicrobial photodynamic therapy using chlorin e6 with halogen light for acne bacteria-induced inflammation. Life Sci. 2015, 124, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Serpeloni, J.M.; Grotto, D.; Aissa, A.F.; Mercadante, A.Z.; Bianchi, M.d.L.P.; Antunes, L.M.G. An evaluation, using the comet assay and the micronucleus test, of the antigenotoxic effects of chlorophyll b in mice. Mutat. Res. Genet. Toxicol. Environ. 2011, 725, 50–56. [Google Scholar] [CrossRef]
- Le Tutour, B.; Benslimane, F.; Gouleau, M.; Gouygou, J.P.; Saadan, B.; Quemeneur, F. Antioxidant and pro-oxidant activities of the brown algae, Laminaria digitata, Himanthalia elongata, Fucus vesiculosus, Fucus serratus and Ascophyllum nodosum. J. Appl. Phycol. 1998, 10, 121–129. [Google Scholar] [CrossRef]
- Osuna-Ruiz, I.; López-Saiz, C.-M.; Burgos-Hernández, A.; Velázquez, C.; Nieves-Soto, M.; Hurtado-Oliva, M.A. Antioxidant, antimutagenic and antiproliferative activities in selected seaweed species from Sinaloa, Mexico. Pharm. Biol. 2016, 54, 2196–2210. [Google Scholar] [CrossRef] [PubMed]
- Cho, M.; Lee, H.-S.; Kang, I.-J.; Won, M.-H.; You, S. Antioxidant properties of extract and fractions from Enteromorpha prolifera, a type of green seaweed. Food Chem. 2011, 127, 999–1006. [Google Scholar] [CrossRef] [PubMed]
- Batista, A.P.; Niccolai, A.; Bursic, I.; Sousa, I.; Raymundo, A.; Rodolfi, L.; Biondi, N.; Tredici, M.R. Microalgae as Functional Ingredients in Savory Food Products: Application to Wheat Crackers. Foods 2019, 23, 611. [Google Scholar] [CrossRef]
- Zen, C.K.; Tiepo, C.B.V.; da Silva, R.V.; Reinehr, C.O.; Gutkoski, L.C.; Oro, T.; Colla, L.M. Development of functional pasta with microencapsulated Spirulina: Technological and sensorial effects. J. Sci. Food Agric. 2020, 30, 2018–2026. [Google Scholar] [CrossRef]
- Sarker, U.; Oba, S. Nutrients, minerals, pigments, phytochemicals, and radical scavenging activity in Amaranthus blitum leafy vegetables. Sci. Rep. 2020, 10, 3868. [Google Scholar] [CrossRef]
- Durga devi, M.; Banu, N. Study of Antioxidant Activity of Chlorophyll from Some Medicinal Plants Paripex. Indian J. Res. 2015, 4, 6–8. [Google Scholar]
- Alvarez-Parrilla, E.; de la Rosa, L.A.; Amarowicz, R.; Shahidi, F. Antioxidant Activity of Fresh and Processed Jalapeño and Serrano Peppers. J. Agric. Food Chem. 2011, 59, 163–173. [Google Scholar] [CrossRef]
- Suparmi, S.; Fasitasari, M.; Martosupono, M.; Mangimbulude, J.C. Comparisons of Curative Effects of Chlorophyll from Sauropus androgynus (L) Merr Leaf Extract and Cu-Chlorophyllin on Sodium Nitrate-Induced Oxidative Stress in Rats. J. Toxicol. 2016, 2016, 8515089. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.H.; Peng, H.; Woo, M.W.; Zeng, X.A.; Brennan, M.; Brennan, C.S. Preparation and characterization of whey protein isolate-chlorophyll microcapsules by spray drying: Effect of WPI ratios on the physicochemical and antioxidant properties. J. Food Eng. 2020, 267, 109729. [Google Scholar] [CrossRef]
- Kamat, J.P.; Boloor, K.K.; Devasagayam, T.P.A. Chlorophyllin as an effective antioxidant against membrane damage in vitro and ex vivo. BBA Mol. Cell Biol. Lip. 2000, 1487, 113–127. [Google Scholar] [CrossRef]
- Kumar, S.S.; Shankar, B.; Sainis, K.B. Effect of chlorophyllin against oxidative stress in splenic lymphocytes in vitro and in vivo. BBA Gen. Subj. 2004, 1672, 100–111. [Google Scholar] [CrossRef]
- Choi, K.; Ortega, M.T.; Jeffery, B.; Riviere, J.E.; Monteiro-Riviere, N.A. Oxidative stress response in canine in vitro liver, kidney and intestinal models with seven potential dietary ingredients. Toxicol. Lett. 2016, 22, 49–59. [Google Scholar] [CrossRef]
- Vaňková, K.; Marková, I.; Jašprová, J.; Dvořák, A.; Subhanová, I.; Zelenka, J.; Novosádová, I.; Rasl, J.; Vomastek, T.; Sobotka, R.; et al. Chlorophyll-Mediated Changes in the Redox Status of Pancreatic Cancer Cells Are Associated with Its Anticancer Effects. Oxid. Med. Cell Longev. 2018, 2, 4069167. [Google Scholar] [CrossRef]
- Sato, M.; Fujimoto, I.; Sakai, T.; Aimoto, T.; Kimura, R.; Murata, T. Effect of sodium copper chlorophyllin on lipid peroxidation. IX On the antioxidative components in commercial preparations of sodium copper chlorophyllin. Chem. Pharm. Bull. 1986, 34, 2428–2434. [Google Scholar] [CrossRef]
- Patar, A.K.; Bhan, S.; Syiem, D.; Sharma, A. Ameliorative Effect of Chlorophyllin on Oxidative Stress in Experimental Model of Diabetes. Int. J. Phytomed. 2016, 8, 506–513. [Google Scholar] [CrossRef][Green Version]
- Wunderlich, A.L.M.; Azevedo, S.C.S.F.; Yamada, L.A.; Bataglini, C.; Previate, C.; Campanholi, K.S.S.; Pereira, P.C.S.; Caetano, W.; Kaplum, V.; Nakamura, C.V.; et al. Chlorophyll treatment combined with photostimulation increases glycolysis and decreases oxidative stress in the liver of type 1 diabetic rats. Braz. J. Med. Biol. Res. 2020, 53, e8389. [Google Scholar] [CrossRef]
- Gomes, B.B.; Barros, S.B.M.; Andrade-Wartha, E.R.S.; Silva, A.M.O.; Silva, V.V.; Lanfer-Marquez, U.M. Bioavailability of dietary sodium copper chlorophyllin and its effect on antioxidant defence parameters of Wistar rats. J. Sci. Food Agric. 2009, 89, 2003–2010. [Google Scholar] [CrossRef]
- Patar, A.K.; Sharma, A.; Syiem, D.; Bhan, S. Chlorophyllin supplementation modulates hyperglycemia-induced oxidative stress and apoptosis in liver of streptozotocin-administered mice: Chlorophyllin modulates hyperglycemia-induced oxidative stress and apoptosis. BioFactors 2018, 44, 418–430. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, P.; Fielden, E.M. Primary Free Radical Processes in DNA. In Advances in Radiation Biology; Lett, J.T., Sinclair, W.K., Eds.; Academic Press: New York, NY, USA, 1993; pp. 53–120. [Google Scholar]
- Kumar, S.S.; Chaubey, R.C.; Devasagayam, T.P.; Priyadarsini, K.I.; Chauhan, P.S. Inhibition of radiation-induced DNA damage in plasmid pBR322 by chlorophyllin and possible mechanism(s) of action. Mutat. Res. 1999, 10, 71–79. [Google Scholar] [CrossRef]
- Ibrahim, M.A.; Elbehairy, A.M.; Ghoneim, M.A.; Amer, H.A. Protective effect of curcumin and chlorophyllin against DNA mutation induced by cyclophosphamide or benzo[a]pyrene. Z. Naturforsch. 2007, 62, 215–222. [Google Scholar] [CrossRef]
- Kavitha, K.; Thiyagarajan, P.; Rathna Nandhini, J.; Mishra, R.; Nagini, S. Chemopreventive effects of diverse dietary phytochemicals against DMBA-induced hamster buccal pouch carcinogenesis via the induction of Nrf2-mediated cytoprotective antioxidant, detoxification, and DNA repair enzymes. Biochimie 2013, 95, 1629–1639. [Google Scholar] [CrossRef]
- Dalle-Donne, I.; Rossi, R.; Giustarini, D.; Milzani, A.; Colombo, R. Protein carbonyl groups as biomarkers of oxidative stress. Clinic. Chim. Acta 2003, 329, 23–38. [Google Scholar] [CrossRef]
- Zhang, D.; Robinson, K.; Mihai, D.M.; Washington, I. Sequestration of ubiquitous dietary derived pigments enables mitochondrial light sensing. Sci. Rep. 2016, 6, 34320. [Google Scholar] [CrossRef]
- Zhang, M.; An, C.; Gao, Y.; Leak, R.K.; Chen, J.; Zhang, F. Emerging roles of Nrf2 and phase II antioxidant enzymes in neuroprotection. Prog. Neurobiol. 2013, 100, 30–47. [Google Scholar] [CrossRef] [PubMed]
- Fahey, J.W.; Stephenson, K.K.; Dinkova-Kostova, A.T.; Egner, P.A.; Kensler, T.W.; Talalay, P. Chlorophyll, chlorophyllin and related tetrapyrroles are significant inducers of mammalian phase 2 cytoprotective genes. Carcinogenesis 2005, 26, 1247–1255. [Google Scholar] [CrossRef]
- Suryavanshi, S.; Sharma, D.; Checker, R.; Thoh, M.; Gota, V.; Sandur, S.K.; Sainis, K.B. Amelioration of radiation-induced hematopoietic syndrome by an antioxidant chlorophyllin through increased stem cell activity and modulation of hematopoiesis. Free Radic. Biol. Med. 2015, 85, 56–70. [Google Scholar] [CrossRef]
- Ozcan, M.; Esendagli, G.; Musdal, Y.; Canpinar, H.; Bacanlı, M.; Anlar, H.G.; Esendağlı-Yılmaz, G.; Beyramzadeh, M.; Aksoy, Y. Dual actions of the antioxidant chlorophyllin, a glutathione transferase P1-1 inhibitor, in tumorigenesis and tumor progression. J. Cell Biochem. 2019, 120, 7045–7055. [Google Scholar] [CrossRef]
- McCarty, M.F. The chlorophyll metabolite phytanic acid is a natural rexinoid—Potential for treatment and prevention of diabetes. Med. Hypotheses 2001, 56, 217–219. [Google Scholar] [CrossRef]
- Mukherjee, R.; Davies, P.J.A.; Crombie, D.L.; Bischoff, E.D.; Cesario, R.M.; Jow, L.; Hamann, L.G.; Boehm, M.F.; Mondon, C.E.; Nadzan, A.M.; et al. Sensitization of diabetic and obese mice to insulin by retinoid X receptor agonists. Nature 1997, 386, 407–410. [Google Scholar] [CrossRef]
- Wang, E.; Wink, M. Chlorophyll enhances oxidative stress tolerance in Caenorhabditis elegans and extends its lifespan. Peer J. 2016, 4, e1879. [Google Scholar] [CrossRef]
- Zhao, Y.; Yang, R.; Rui, Q.; Wang, D. Intestinal Insulin Signaling Encodes Two Different Molecular Mechanisms for the Shortened Longevity Induced by Graphene Oxide in Caenorhabditis elegans. Sci. Rep. 2016, 6, 24024. [Google Scholar] [CrossRef]
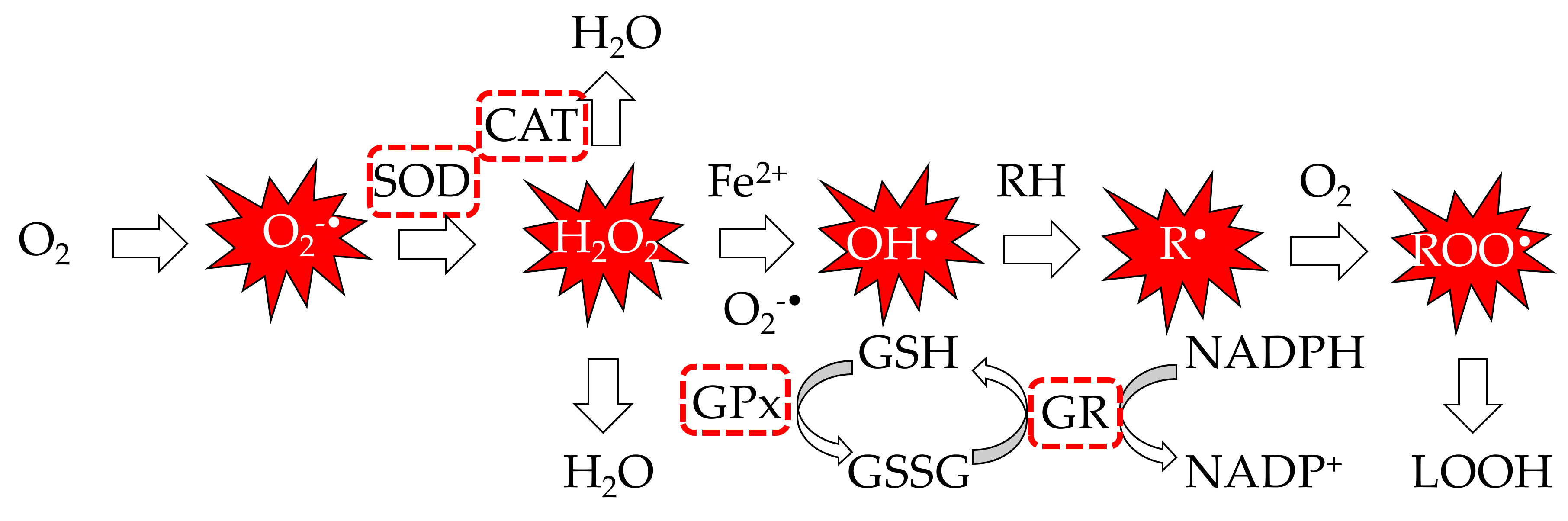
| Method | Advantages | Disadvantages | Mechanism |
|---|---|---|---|
| ORAC |
|
| HAT1: Decrease of luminescence of target compound by peroxyl radicals. |
| TRAP | HAT: Decay of fluorescent target once peroxided. | ||
| FRAP |
|
| SET: Formation of a reduced ferric compound |
| CUPRAC |
|
| SET: Formation of a reduced cupric compound |
| DPPH |
| Mixed-mode: Ability to scavenge DPPH radical | |
| ABTS |
*Short stabilization times. *Applicable to geometric carotenoid isomers [41].
|
| Mixed-mode: Formation of an oxidized radical cation. |
| Biomarker | Direct In Vivo Measurement | Alternatives | Not Allowed |
|---|---|---|---|
| Protein | A method that allows separation and identification of oxidative changes in amino acids (e.g., protein tyrosine nitration products by LC-MS) |
| |
| Lipid |
|
|
|
| DNA |
|
|
|
| Experimental Approach 1 | Composition of the Membrane Model; Induction of Oxidative Stress; Biomarker(s) | Carotenoid(s) | Observed Effect | Reference |
|---|---|---|---|---|
| Lipid peroxidation in aqueous dispersion of lipids | Dimyristoil phosphatidylcholine liposomes or soybean phosphatidylcholine liposomes enriched with β-carotene; AAPH and AMVN peroxyl-radical inducers; phospholipid hydroperoxides, carotenoid bleaching | β-carotene | Potential of the antioxidant capacity of β-carotene is limited by the carotenoid autoxidation process that continues the chain oxidation of membrane lipids | [103] |
| Lipid peroxidation of liposomal bilayers | Dilinoleoylphosphatidylcholine liposomes enriched with β-carotene; AAPH radical inducer; conjugated dienes, carotenoid bleaching | β-carotene | β-carotene is an effective chain-breaking antioxidant when it is incorporated into the membranes | [104] |
| Lipid peroxidation in artificial membranes | Egg-yolk phosphatidylcholine liposomes enriched with a single carotenoid; AAPH and AMVN peroxyl-radical inducers; phospholipid hydroperoxides, carotenoid bleaching | Astaxanthin, β-carotene, canthaxanthin, lycopene, zeaxanthin; Antioxidant activity was compared with α-tocopherol | Different reactivity toward free radicals was correlated with the structural arrangement of the carotenoid, its position, and orientation in the bilayer | [105] |
| Lipid peroxidation in artificial membranes | Egg-yolk phosphatidylcholine liposomes enriched with a single carotenoid; UV radiation and AAPH peroxyl-radical inducer; TBARS, carotenoid bleaching | Lutein, zeaxanthin | Both xanthophylls are effective membrane antioxidants towards different radical induction processes; extension of lipid peroxidation was reduced by 35%; both xanthophylls promoted different changes in the organization of the lipids in the bilayer | [106] |
| Photoperoxidation of multilayer liposomes | Egg-yolk phosphatidylcholine liposomes enriched with individual carotenoid; hydrogen peroxide, tert-butyl hydroperoxide, ascorbic acid, and Fe+2-EDTA radical inducers; TBARS, iron concentration in liposomes | Astaxanthin, lycopene, peridinin | Structure of the carotenoids induced changes in the permeability of the membranes to radical inducers; a reduction of 25% of TBARS was observed after the addition of carotenoids to liposomes | [107] |
| Photoperoxidation of multilayer liposomes | Dimyristoil phosphatidylcholine/palmitoyl-oleoyl phosphatidylcholine/cholesterol liposomes enriched with individual or combined antioxidants; aerobic photo-peroxidation; lipid hydroperoxides, carotenoid bleaching, oxygen photo-uptake | Zeaxanthin | Zeaxanthin is an efficient membrane antioxidant toward singlet oxygen with a synergistic effect with α-tocopherol; Zeaxanthin is 30 times more effective than α-tocopherol in inhibiting photosensitized lipid peroxidation | [108] |
| Lipid peroxidation in unilamellar liposomes | Soybean phosphatidylcholine liposomes enriched with combined antioxidants; AAPH peroxyl-radical inducer; conjugated dienes | β-carotene, lycopene | Synergistic effects of carotenoids and tocopherols toward the lipid peroxidation process | [109] |
| Lipid peroxidation in unilamellar liposomes | Soybean phosphatidylcholine liposomes enriched with combined antioxidants; AAPH peroxyl-radical inducer; conjugated dienes | Astaxanthin | Synergistic effects of astaxanthin with both hydrophilic and lipophilic antioxidants were not significant | [110] |
| Lipid peroxidation in artificial membranes | Multilamellar liposomes enriched with PUFA/carotenoid; autoxidation; hydroperoxides, membrane interactions | Astaxanthin, β-carotene, lutein, lycopene, zeaxanthin | Apolar carotenoids behaved as membrane pro-oxidants, increasing the peroxide formation by 90–120%, whereas astaxanthin behaved as an antioxidant, decreasing the peroxide formation by 30% | [111] |
| Experimental Approach | Participants; Supplementation Protocol; Biomarkers of Oxidative Stress; Relevant Methodologies | Carotenoids | Observed Effect | Reference |
|---|---|---|---|---|
| Supplementation/ex vivo LDL oxidation | Group of male nonsmokers and smokers; supplementation with β-carotene (2 × 20 mg daily for two weeks, and then 20 mg daily for 12 weeks); lipid peroxidation of LDL isolates [112] | β-carotene | No protective effect of LDL susceptibility to oxidation despite the observed increase in plasma β-carotene levels | [113] |
| Supplementation/ex vivo biomarkers of oxidative stress | Group of 11 smokers and 11 nonsmokers; supplementation with fruits and vegetables providing 30 mg carotenoids/day for 2 weeks; lipid peroxidation of LDL isolate supplementation, oxidative stress biomarkers of plasma [112] | α-carotene, β-carotene, lutein, lycopene, α-cryptoxanthin, β-cryptoxanthin | Inhibition of LDL susceptibility to oxidation for the smokers and nonsmokers. LDL resistance to oxidation increased 14% in smokers and 28% in the nonsmokers group after supplementation | [114] |
| Supplementation/ex vivo biomarkers of oxidative stress | Group of 32 healthy volunteers; double-blind randomized, placebo-controlled trial, supplementation with a mixture of carotenoids providing 7.6 mg carotenoids/day for 3 weeks; lipid peroxidation of LDL isolates, DNA damage, ORAC [8,112] | Lycopene, palm oil carotenes, marigold extract carotenoids, paprika carotenoids, bixin | The carotenoid supplementation reduced the LDL oxidizability (by 20.4% in the supplemented group) and DNA damage assessed by urine biomarkers; the effect was not observed with the ORAC assay | [115] |
| Supplementation/ex vivo biomarkers of oxidative stress | Group of 105 healthy volunteers; randomized, double-blind, placebo-controlled; commercial spread providing with a mixture of carotenoids at different doses for 11 weeks; lipid peroxidation of LDL isolates, plasma FRAP, MDA, serum arylesterase activity, plasma F2α-isoprostanes; [28,112] | Lycopene, lutein, α-carotene, β-carotene | Moderate amounts of carotenoids resulted in a significantly increased resistance of LDL to oxidation and lower plasma peroxidation biomarkers (17% increase of LDL resistance to oxidation, 18% increase of lag-phase, and 15% reduction in the F2-isoprostane level) | [116] |
| Supplementation/ex vivo LDL oxidation | Group of 12 healthy female volunteers; supplementation with tomato products providing 8 mg lycopene/day for 21 days; lipid peroxidation of LDL isolates, urinary 8-iso-PGF2α [112,117] | Lycopene | Decrease in LDL oxidizability (22%) and significant lower excretion of 8-iso-PGF2α (53%) regarding the values reached in the control group | [118] |
| In vitro loading of LDL/biomarkers of oxidative stress | Group of 10 volunteers donated plasma samples for LDL isolation; in vitro loading was performed with lycopene or lutein via emulgent and incubation; lipid peroxidation and oxidation of ApoB of LDL isolates [108] | Lycopene, lutein | Carotenoids were not effective antioxidants of the LDL | [119] |
| Group of 35 patients with T2DM; double-blind, placebo-controlled; supplementation with lycopene, 10 mg/day for 8 weeks; total antioxidant capacity assessed via ABTS, MDA, humoral immunity biomarkers | Lycopene | Increased ratio of total antioxidant capacity to MDA values and attenuated pro-atherogenic immune response | [120] | |
| Supplementation/ex vivo LDL oxidation | Group of 77 healthy male and female volunteers; double-blind randomized, placebo-controlled trial, lycopene supplement at different doses for 8 weeks; lipid peroxidation of LDL isolates, MDA and HNE, urinary 8-iso-PGF2α, DNA damage markers | Lycopene | Significant decrease in DNA damage (8.9%) and urinary 8-iso-PGF2α levels (23%) in the supplemented group; no significant effect was observed in biomarkers of lipid peroxidation | [121] |
| Supplementation/ex vivo biomarkers of oxidative stress | Group of 126 healthy men; randomized placebo-controlled trial, lycopene supplementation at different doses for 8 weeks; SOD activity in plasma, DNA damage, biomarkers of endothelial function | Lycopene, β-carotene | Increase in SOD activity (2.37 units/mL) and prevention of DNA damage (for the 15 mg/day suppl. group); beneficial effects in subjects with relatively impaired endothelial cell function | [122] |
| Experimental Approach | Cell Model; Induction of Oxidative Stress; Biomarker(s) | Carotenoids | Observed Effect | Reference |
|---|---|---|---|---|
| Cellular membrane oxidation | Human HepG2 cells; tert-butyl hydroperoxide; lipid peroxidation and cellular leakage of lactate dehydrogenase | Micellar β-carotene (1.1 μmol/L) or lutein (10.9 μmol/L) | Protection of cellular membrane toward oxidant-induced changes | [133] |
| Lipid peroxidation | Normal and tumor thymocytes; AAPH and xanthine/xanthine oxidase, at low or high pO2; MDA and conjugated dienes | β-carotene in THF (6.3 mg/mL) to yield 10 μM–20 μM carotenoid concentration | Oxygen tension was a significant factor of β-carotene antioxidant efficiency. Lipid peroxidation rate increased 2.2-fold and 1.8-fold at 760 mm Hg pO2 | [134] |
| DNA damage | HT29; xanthine/xanthine oxidase; oxidation of DNA and membrane integrity | Lycopene, β-carotene in THF to yield 1–10 μM concentration | Protection of oxidatively-induced DNA damage and membrane integrity Mean relative tail moment was reduced a 50% at 2.5 μM carotenoid concentration | [135] |
| Lipid peroxidation and DNA damage | CV1-P monkey cells; ferric nitrolotriacetate plus ascorbate; lipid peroxidation (TBARS) and 8-oxodGuo | Lycopene in THF to yield 3 mM concentration | Protection of mammalian cells against membrane and DNA damage. A 77% reduction in 8-oxodGuo level in lycopene-treated cells | [136] |
| Lipid peroxidation and DNA damage | Hs68 human foreskin fibroblasts; AAPH and AMVN and ferric nitrilotriacetate; lipid peroxidation, 8-OH-dG, comet assay | Lycopene, β-carotene in THF to yield 10 or 20 μM concentration | Both carotenoids performed antioxidant and pro-oxidant actions depending on the source of oxidative damage | [137] |
| UVA-photoprotection | Human skin fibroblasts; UVA radiation; metalloprotease 1 mRNA | Lycopene, β-carotene (nanoparticle formulation) | Reduction of the biomarker was only observed in the presence of vitamin E. Only a small induction of HO-1 was observed (1-2-fold) for lycopene or β-carotene treatments | [138] |
| Cellular membrane oxidation | Rat pheochromocytoma PC-12; deprivation of essential nutrients; peroxidation of membrane lipids and SOD activity | Crocin (0.1–10 μM) | Crocin was able to function as a chain-breaking antioxidant, restoring SOD activity (54% of the normal values) and maintained 60% of the neuron morphology | [139] |
| Oxidative stress of photoreceptors | Culture of rat retinal neurons; induced oxidative stress by paraquat and H2O2; apoptosis, mitochondrial membrane potential, cytochrome c translocation, and opsin expression | Lutein, zeaxanthin and β-carotene in 0.05% Tween solution | Carotenoids reduced the oxidative-stress-induced apoptosis as well as the other evaluated biomarkers. The 2.5-fold increase in photoreceptor cell death was suppressed in carotenoid-treated cells | [140] |
| Hydrogen peroxide damage | Rat erythrocytes (ex vivo); hydrogen-induced hemolysis; lipid peroxidation of membrane | Peel extracts of unripe and ripe mango fruits containing carotenoids. 5–25 μg of gallic acid equivalent in PBS | Protection against membrane protein degradation and morphological changes. 50% hemolysis inhibition was obtained at 11.5–20.9 μg GAE | [141] |
| UVA-photoprotection | Human dermal fibroblasts; UVA radiation; reactive oxygen species, apoptosis cascade enzymes, heme oxygenase expression | Astaxanthin, canthaxanthin, and β-carotene in THF to yield 0.5–10 μM concentration | Astaxanthin exerted a higher protective effect towards photo-oxidative damage. Measured ROS decreased by 30% and 50% in cells treated with astaxanthin at 5 μM | [142] |
| Functional integrity and mitochondrial redox state | Transfected HeLa human cervical cancer cells; hydrogen peroxide; redox-sensitive fluorescent protein imaging recording, mitochondrial membrane potential, superoxide levels | Astaxanthin in DMSO to yield 800 nM concentration | Reduction of basal oxidative stress, maintenance of mitochondrial membrane potential, improvement of the mitochondrial redox state | [143] |
| Mitochondrial function | Human HepG2 cells; carotenoid induction of ROS; ROS observed by fluorescence microscopy | Zeaxanthin, lutein and their 3-dehydro- derivatives | Mitochondrial carotenoid-oxygenase degraded carotenoids to protect the organelle functionality | [131] |
| Retinal degeneration | Mouse retinal ganglion cells RGC 5; tunicamycin, hydrogen peroxide; cell death, apoptosis cascade enzymes, nuclear layer thickness | Crocetin (0.1% in DMSO/PBS) to yield 0.1–3 μM concentration | Protective effects against retinal damage. Crocetin increased the protective effect against cell damage 5-fold | [144] |
| Photoprotective effect against UVB light | CCD-1064Sk human dermal fibroblasts; UVB irradiation; comet assay, UVB-induced cellular apoptosis | Capsanthin, capsorubin in THF/FBS to yield 1 μM concentration | The tested carotenoids decreased markers for UVB-induced apoptosis and interfered with cellular responses activated by UVB-mediated damage. DNA damage was decreased by 50% in capsanthin/capsorubin-treated cells after UVB irradiation | [145] |
| Method | Pigment | Concentration | Activity | Reference |
|---|---|---|---|---|
| β-carotene bleaching (% inhibition of oxidation) | Cu–chlorophyllin | 681 μM | 80% | [149] |
| Pheophorbide b | 80% | |||
| Pheophytin b | 75% | |||
| Pheophorbide a | 75% | |||
| Chlorophyll a | 40% | |||
| Pheophytin a | 70% | |||
| β-carotene bleaching | Chlorophyll | 0.05 μg/μL | 49.63% | [165] |
| Pheophytin | 13.44% | |||
| Zn–pheophytin | 66.43% | |||
| β-carotene bleaching | Zn–chlorophyllin | 5 mg/mL | 82.00% | [170] |
| Cu–chlorophyllin | 74.40% | |||
| Iron–chlorophyllin | 90.20% | |||
| DPPH (% radical scavenging) | Cu–chlorophyllin | 1 mM | 39% | [149] |
| Pheophorbide b | 1 mM | <12% | ||
| Pheophytin b | 1 mM | <12% | ||
| Pheophorbide a | 1 mM | <12% | ||
| Chlorophyll a | 1 mM | <12% | ||
| Pheophytin a | <12% | |||
| DPPH | Pheophytin a | 200 μM | 55% | [171] |
| Pheophytin b | 200 μM | 50% | ||
| Chlorophyll a | 200 μM | 40% | ||
| Chlorophyll b | 200 μM | 44% | ||
| DPPH | Chlorophyll | 0.05 μg/μL | 13.89% | [165] |
| Pheophytin | 13.44% | |||
| Zn–pheophytin | 66.43% | |||
| DPPH | Zn–chlorophyllin | 5 mg/mL | 37.90% | [170] |
| Cu–chlorophyllin | 93.50% | |||
| Fe–chlorophyllin | 26.50% | |||
| DPPH (TEAC) | Pyropheophytin a | 100 mM | 0.02 | [148] |
| Pheophytin a | 0.04 | |||
| Pheophytin b | 0.05 | |||
| Chlorophyll b | 0.06 | |||
| Zn–Pheophytin b | 0.13 | |||
| Chlorophyll a | 0.19 | |||
| Pheophorbide a | 0.21 | |||
| Chlorin e4 | 0.26 | |||
| Zn-Pyropheophytin a | 0.44 | |||
| Zn-Pheophytin a | 0.51 | |||
| Chlorin e6 | 0.6 | |||
| Cu-Chlorin | 0.81 | |||
| Cu–Pheophorbide a | 0.98 | |||
| Cu–Pheophytin a | 0.99 | |||
| Crude SCC | 1.04 | |||
| Cu–Chlorin e6 | 2.88 | |||
| DPPH (I50) | Pheophorbide a | 120 μM | [149] | |
| Pheophorbide b | 75 μM | |||
| Chlorophyllin | 360 μM | |||
| Chlorophyllide a | >800 μM | |||
| Chlorophyllide b | >800 μM | |||
| DPPH (EC50) | Chlorin e6 | 23 μg/mL | [162] | |
| ABTS (EC50) | Chlorin e6 | 52 μg/mL | [172] | |
| ORAC (TEAC) | Chlorin e6 | 12.5 μg/mL | 27 μM | [172] |
| ABTS (TEAC) | Pheophytin a | 100 mM | 0.02 | [148] |
| Pheophytin b | 0.08 | |||
| Pyropheophytin a | 0.16 | |||
| Chlorophyll b | 0.23 | |||
| Zn–pheophytin b | 0.29 | |||
| Zn–pheophytin a | 0.43 | |||
| Pheophorbide a | 0.45 | |||
| Chlorin e4 | 0.53 | |||
| Cu–pheophytin a | 0.58 | |||
| Chlorin e6 | 0.64 | |||
| Zn–pyropheophytin a | 0.67 | |||
| Chlorophyll a | 0.73 | |||
| Crude SCC | 1.25 | |||
| Cu–chlorin e4 | 1.35 | |||
| Cu–chlorin e6 | 2.25 | |||
| Cu–pheophorbide | 2.4 | |||
| TBARS Kidney | Chlorophyll b | 0.2 mg/kg b.w. | 61.16 nmol/g | [173] |
| 0.5 mg/kg b.w. | 62.06 nmol/g | |||
| TBARS | Chlorophyll b | 0.2 mg/kg b.w. | 41.29 nmol/g | [173] |
| Liver | 0.5 mg/kg b.w. | 45.90 nmol/g | ||
| Comet assay | Pheophytin a | 50 μM | 3500 | [171] |
| Pheophytin b | 50 μM | 3500 | ||
| Chlorophyll a | 50 μM | 4000 | ||
| Chlorophyll b | 50 μM | 4000 | ||
| Fe chelation | Pheophytin a | 200 μM | 65% | [171] |
| Pheophytin b | 200 μM | 65% | ||
| Chlorophyll a | 200 μM | 55% | ||
| Chlorophyll b | 200 μM | 55% | ||
| Lipid peroxidation | Pheophytin a | 100 μM | 75% | [171] |
| Pheophytin b | 100 μM | 65% | ||
| Chlorophyll a | 100 μM | 95% | ||
| Chlorophyll b | 100 μM | 75% | ||
| ROO· scavenging capacity (α-tocopherol relative) | Chlorophyll a | 308 | [171] | |
| Chlorophyll b | 386 | |||
| Comet assay (Tail moment) | Cu–chlorophyllin | 20 μM | 138 | [172] |
| Chlorophyllide a | 20 μM | 136 | ||
| Chlorophyllide b | 20 μM | 126 | ||
| Pheophorbide a | 20 μM | 100 | ||
| Pheophorbide b | 20 μM | 91 | ||
| 8-OHdG (ng/μg DNA) | Cu–chlorophyllin | 20 μM | 0.53 | [172] |
| Chlorophyllide a | 20 μM | 0.68 | ||
| Chlorophyllide b | 20 μM | 0.79 | ||
| Pheophorbide a | 20 μM | 0.55 | ||
| Pheophorbide b | 20 μM | 0.62 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pérez-Gálvez, A.; Viera, I.; Roca, M. Carotenoids and Chlorophylls as Antioxidants. Antioxidants 2020, 9, 505. https://doi.org/10.3390/antiox9060505
Pérez-Gálvez A, Viera I, Roca M. Carotenoids and Chlorophylls as Antioxidants. Antioxidants. 2020; 9(6):505. https://doi.org/10.3390/antiox9060505
Chicago/Turabian StylePérez-Gálvez, Antonio, Isabel Viera, and María Roca. 2020. "Carotenoids and Chlorophylls as Antioxidants" Antioxidants 9, no. 6: 505. https://doi.org/10.3390/antiox9060505
APA StylePérez-Gálvez, A., Viera, I., & Roca, M. (2020). Carotenoids and Chlorophylls as Antioxidants. Antioxidants, 9(6), 505. https://doi.org/10.3390/antiox9060505