Anthocyanins in Blueberries Grown in Hot Climate Exert Strong Antioxidant Activity and May Be Effective against Urinary Tract Bacteria
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Samples
2.3. Extraction of Anthocyanin Fraction
2.4. Total Anthocyanin Content (TAC)
2.5. Oxygen Radical Absorbance Capacity (ORAC)
2.6. Identification of Anthocyanins: UHPLC-MS/MS Orbitrap
2.7. Bacterial Strains
2.7.1. Standard Strains
2.7.2. Uropathogenic Bacteria Isolated from UTI Patients
2.7.3. Preparation of Bacterial Strains
2.8. Determination of the Minimum Inhibitory Concentration (MIC)
2.9. Determination of the Minimum Bactericidal Concentration (MBC)
2.10. Statistical Analysis
3. Results and Discussion
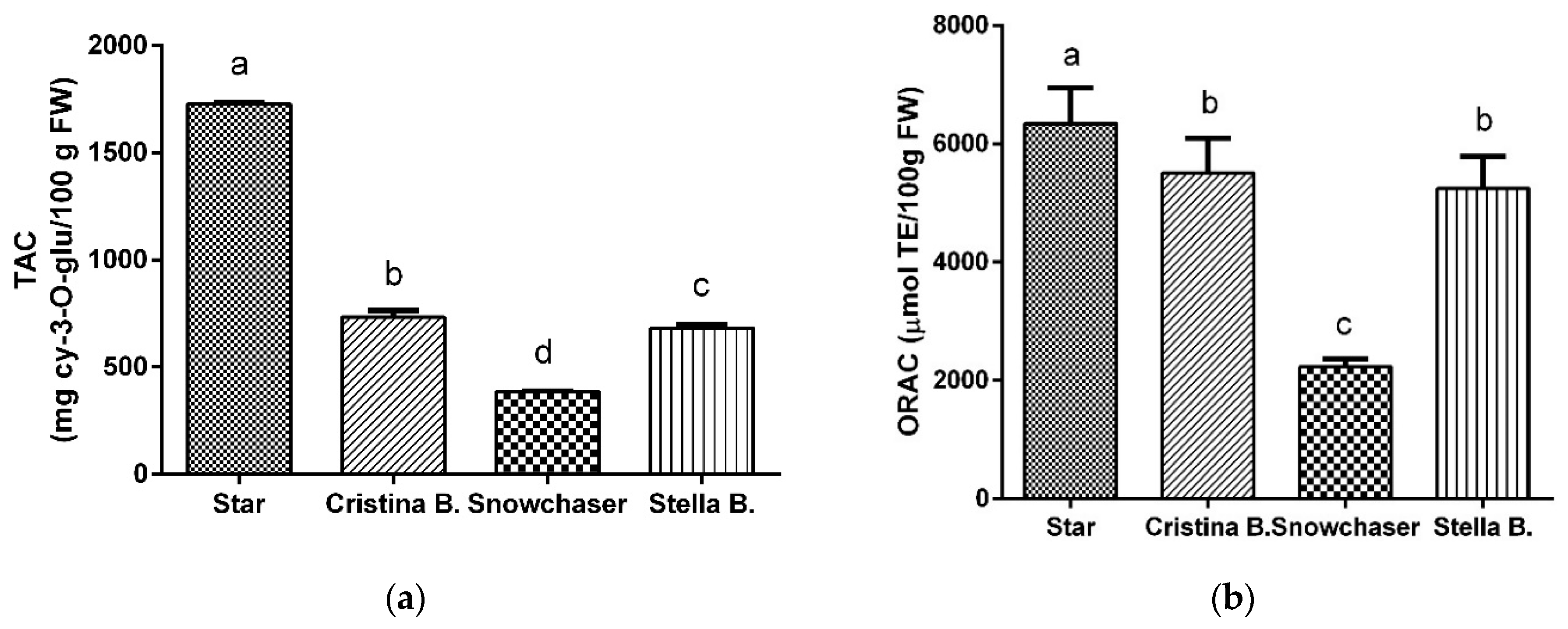
3.1. Total Anthocyanins Content (TAC)
3.2. Antioxidant Capacity: ORAC Assay
3.3. Anthocyanin Profile
3.4. Antibacterial Activity
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tsuda, T. Dietary anthocyanin-rich plants: Biochemical basis and recent progress in health benefits studies. Mol. Nutr. Food Res. 2012, 56, 159–170. [Google Scholar] [CrossRef]
- Jara-Palacios, M.J.; Santisteban, A.; Gordillo, B.; Hernanz, D.; Heredia, F.J.; Escudero-Gilete, M.L. Comparative study of red berry pomaces (blueberry, red raspberry, red currant and blackberry) as source of antioxidants and pigments. Eur. Food Res. Technol. 2019, 245, 1–9. [Google Scholar] [CrossRef]
- Fibigr, J.; Satinsky, D.; Solich, P. A UHPLC method for the rapid separation and quantification of anthocyanins in acai berry and dry blueberry extracts. J. Pharm. Biomed. Anal. 2017, 143, 204–213. [Google Scholar] [CrossRef]
- Kim, J.G.; Kim, H.L.; Kim, S.J.; Park, K.-S. Fruit quality, anthocyanin and total phenolic contents, and antioxidant activities of 45 blueberry cultivars grown in Suwon, Korea. J. Zhejiang Univ. Sci. B 2013, 14, 793–799. [Google Scholar] [CrossRef]
- Reque, P.M.; Steffens, R.S.; Silva, A.M.D.; Jablonski, A.; Flôres, S.H.; Rios, A.D.O.; Jong, E.V.D. Characterization of blueberry fruits (Vaccinium spp.) and derived products. Food Sci. Technol. (Camp.) 2014, 34, 773–779. [Google Scholar] [CrossRef]
- Ongkowijoyo, P.; Luna-Vital, D.A.; Gonzalez De Mejia, E. Extraction techniques and analysis of anthocyanins from food sources by mass spectrometry: An update. Food Chem. 2018, 250, 113–126. [Google Scholar] [CrossRef]
- Bendokas, V.; Skemiene, K.; Trumbeckaite, S.; Stanys, V.; Passamonti, S.; Borutaite, V.; Liobikas, J. Anthocyanins: From plant pigments to health benefits at mitochondrial level. Crit. Rev. Food Sci. Nutr. 2019. [Google Scholar] [CrossRef]
- Fernandes, I.; Faria, A.; Calhau, C.; De Freitas, V.; Mateus, N. Bioavailability of anthocyanins and derivatives. J. Funct. Foods 2014, 7, 54–66. [Google Scholar] [CrossRef]
- Cassidy, A.; Bertoia, M.; Chiuve, S.; Flint, A.; Forman, J.; Rimm, E.B. Habitual intake of anthocyanins and flavanones and risk of cardiovascular disease in men. Am. J. Clin. Nutr. 2016, 104, 587–594. [Google Scholar] [CrossRef]
- Cassidy, A.; Mukamal, K.J.; Liu, L.; Franz, M.; Eliassen, A.H.; Rimm, E.B. High anthocyanin intake is associated with a reduced risk of myocardial infarction in young and middle-aged women. Circulation 2013, 127, 188–196. [Google Scholar] [CrossRef]
- McCullough, M.L.; Peterson, J.J.; Patel, R.; Jacques, P.F.; Shah, R.; Dwyer, J.T. Flavonoid intake and cardiovascular disease mortality in a prospective cohort of US adults. Am. J. Clin. Nutr. 2012, 95, 454–464. [Google Scholar] [CrossRef]
- Zamora-Ros, R.; Knaze, V.; Luján-Barroso, L.; Slimani, N.; Romieu, I.; Touillaud, M.; Kaaks, R.; Teucher, B.; Mattiello, A.; Grioni, S.; et al. Estimation of the intake of anthocyanidins and their food sources in the European Prospective Investigation into Cancer and Nutrition (EPIC) study. Br. J. Nutr. 2011, 106, 1090–1099. [Google Scholar] [CrossRef]
- De Pascual-Teresa, S.; Moreno, D.A.; García-Viguera, C. Flavanols and anthocyanins in cardiovascular health: A review of current evidence. Int. J. Mol. Sci. 2010, 11, 1679–1703. [Google Scholar] [CrossRef]
- Howell, A.B. Bioactive compounds in cranberries and their role in prevention of urinary tract infections. Mol. Nutr. Food Res. 2007, 51, 732–737. [Google Scholar] [CrossRef]
- Jensen, H.D.; Struve, C.; Christensen, S.B.; Krogfelt, K.A. Cranberry juice and combinations of its organic acids are effective against experimental urinary tract infection. Front. Microbiol. 2017, 8, 542. [Google Scholar] [CrossRef]
- Jepson, R.G.; Williams, G.; Craig, J.C. Cranberries for preventing urinary tract infections. Cochrane Database Syst. Rev. 2012, 10, CD001321. [Google Scholar] [CrossRef]
- Milbury, P.E.; Vita, J.A.; Blumberg, J.B. Anthocyanins are bioavailable in humans following an acute dose of cranberry juice. J. Nutr. 2010, 140, 1099–1104. [Google Scholar] [CrossRef]
- Jepson, R.G.; Craig, J.C. A systematic review of the evidence for cranberries and blueberries in UTI prevention. Mol. Nutr. Food Res. 2007, 51, 738–745. [Google Scholar] [CrossRef]
- Cassidy, A. Berry anthocyanin intake and cardiovascular health. Mol. Aspects Med. 2018, 61, 76–82. [Google Scholar] [CrossRef] [PubMed]
- Timmers, M.A.; Grace, M.H.; Yousef, G.G.; Lila, M.A. Inter- and intra-seasonal changes in anthocyanin accumulation and global metabolite profiling of six blueberry genotypes. J. Food Compost. Anal. 2017, 59, 105–110. [Google Scholar] [CrossRef]
- Brito, A.; Areche, C.; Sepúlveda, B.; Kennelly, E.; Simirgiotis, M. Anthocyanin characterization, total phenolic quantification and antioxidant features of some Chilean edible berry extracts. Molecules 2014, 19, 10936–10955. [Google Scholar] [CrossRef]
- Wang, S.Y.; Chen, H.; Camp, M.J.; Ehlenfeldt, M.K. Genotype and growing season influence blueberry antioxidant capacity and other quality attributes. Int. J. Food Sci. Technol. 2012, 47, 1540–1549. [Google Scholar] [CrossRef]
- Cerezo, A.B.; Cuevas, E.; Winterhalter, P.; Garcia-Parrilla, M.C.; Troncoso, A.M. Isolation, identification, and antioxidant activity of anthocyanin compounds in Camarosa strawberry. Food Chem. 2010, 123, 574–582. [Google Scholar] [CrossRef]
- Wu, Y.; Zhou, Q.; Chen, X.-Y.; Li, X.; Wang, Y.; Zhang, J.-L. Comparison and screening of bioactive phenolic compounds in different blueberry cultivars: Evaluation of anti-oxidation and α-glucosidase inhibition effect. Food Res. Int. 2017, 100, 312–324. [Google Scholar] [CrossRef]
- Prior, R.L.; Cao, G.; Martin, A.; Sofic, E.; McEwen, J.; O’Brien, C.; Lischner, N.; Ehlenfeldt, M.; Kalt, W.; Krewer, G.; et al. Antioxidant capacity as influenced by total phenolic and anthocyanin content, maturity, and variety of Vaccinium species. J. Agric. Food Chem. 1998, 46, 2686–2693. [Google Scholar] [CrossRef]
- US Department of Agriculture. USDA Database for the Flavonoid Content of Selected Foods: Release 3.1. US Department of Agriculture, Washington (DC). 2014. Available online: https://data.nal.usda.gov/dataset/usda-database-flavonoid-content-selected-foods-release-31-may-2014 (accessed on 30 April 2020).
- Phenol Explorer. Available online: http://phenol-explorer.eu/ (accessed on 28 April 2020).
- Wu, X.; Beecher, G.R.; Holden, J.M.; Haytowitz, D.B.; Gebhardt, S.E.; Prior, R.L. Concentrations of anthocyanins in common foods in the United States and estimation of normal consumption. J. Agric. Food Chem. 2006, 54, 4069–4075. [Google Scholar] [CrossRef]
- Kalt, W.; Cassidy, A.; Howard, L.R.; Krikorian, R.; Stull, A.J.; Tremblay, F.; Zamora-Ros, R. Recent research on the health benefits of blueberries and their anthocyanins. Adv. Nutr. 2019. [Google Scholar] [CrossRef]
- Spinardi, A.; Cola, G.; Gardana, C.S.; Mignani, I. Variation of anthocyanin content and profile throughout fruit development and ripening of highbush blueberry cultivars grown at two different altitudes. Front. Plant Sci. 2019, 10, 1319. [Google Scholar] [CrossRef]
- Bunea, A.; Rugina, D.; Sconta, Z.; Pop, R.M.; Pintea, A.; Socaciu, C.; Tabaran, F.; Grootaert, C.; Struijs, K.; VanCamp, J. Anthocyanin determination in blueberry extracts from various cultivars and their antiproliferative and apoptotic properties in B16-F10 metastatic murine melanoma cells. Phytochemistry 2013, 95, 436–444. [Google Scholar] [CrossRef]
- Pertuzatti, P.B.; Barcia, M.T.; Rebello, L.P.G.; Gómez-Alonso, S.; Duarte, R.M.T.; Duarte, M.C.T.; Godoy, H.T.; Hermosín-Gutiérrez, I. Antimicrobial activity and differentiation of anthocyanin profiles of rabbiteye and highbush blueberries using HPLC–DAD–ESI-MS n and multivariate analysis. J. Funct. Foods 2016, 26, 506–516. [Google Scholar] [CrossRef]
- Prior, R.L.; Lazarus, S.A.; Cao, G.H.; Muccitelli, H.; Hammerstone, J.F. Identification of procyanidins and anthocyanins in blueberries and cranberries (Vaccinium spp.) using high-performance liquid chromatography/mass spectrometry. J. Agric. Food Chem. 2001, 49, 1270–1276. [Google Scholar] [CrossRef] [PubMed]
- Silva, S.; Costa, E.M.; Calhau, C.; Morais, R.M.; Pintado, M.M.E. Production of a food grade blueberry extract rich in anthocyanins: Selection of solvents, extraction conditions and purification method. J. Food Meas. Charact. 2017, 11, 1248–1253. [Google Scholar] [CrossRef]
- Andreotti, C.; Castagnoli, M.; Maltoni, M.L.; Magnani, S.; Fontanari, M.; Carlini, A.; Marchesini, A.; Pititto, A.; Faedi, W.; Baudino, M. Quality and Phenolic Composition of Blueberries Cultivated in Italy; International Society for Horticultural Science (ISHS): Leuven, Belgium, 2014. [Google Scholar]
- Ribera, A.E.; Reyes-Diaz, M.; Alberdi, M.; Zuñiga, G.E.; Mora, M.L. antioxidant compounds in skin and pulp of fruits change among genotypes and maturity stages in highbush blueberry (Vaccinium corymbosum l.) grown in Southern Chile. J. Soil Sci. Plant Nutr. 2010, 10, 509–536. [Google Scholar] [CrossRef]
- Castrejón, A.D.R.; Eichholz, I.; Rohn, S.; Kroh, L.W.; Huyskens-Keil, S. Phenolic profile and antioxidant activity of highbush blueberry (Vaccinium corymbosum L.) during fruit maturation and ripening. Food Chem. 2008, 109, 564–572. [Google Scholar] [CrossRef]
- Yousef, G.G.; Brown, A.F.; Funakoshi, Y.; Mbeunkui, F.; Grace, M.H.; Ballington, J.R.; Loraine, A.; Lila, M.A. Efficient quantification of the health-relevant anthocyanin and phenolic acid profiles in commercial cultivars and breeding selections of blueberries (vaccinium spp.). J. Agric. Food Chem. 2013, 61, 4806–4815. [Google Scholar] [CrossRef]
- Tan, K.; Lee, W.S.; Gan, H.; Wang, S. Recognising blueberry fruit of different maturity using histogram oriented gradients and colour features in outdoor scenes. Biosys. Eng. 2018, 176, 59–72. [Google Scholar] [CrossRef]
- Junta de Andalucía, Consejería de Agricultura, Ganadería, Pesca y Desarrollo Sostenible.Observatorio de Precios y Mercados. Available online: http://www.juntadeandalucia.es/agriculturaypesca/observatorio/servlet/FrontController?action=RecordContent&table=11113&element=2962762&subsector=19& (accessed on 29 April 2020).
- Cardeñosa, V.; Girones-Vilaplana, A.; Muriel, J.L.; Moreno, D.A.; Moreno-Rojas, J.M. Influence of genotype, cultivation system and irrigation regime on antioxidant capacity and selected phenolics of blueberries (Vaccinium corymbosum L.). Food Chem. 2016, 202, 276–283. [Google Scholar] [CrossRef]
- Jimenes, I.M.; da Silva, S.R.; Tezotto-Uliana, J.V.; Cantuarias-Avilés, T. Atributos de qualidade em frutos de mirtileiro ‘Snowchaser’ de baixa exigência em frio cultivados no Brasil. Rev. Bras. Frutic. 2018, 40. [Google Scholar] [CrossRef]
- Ogden, A.B.; Van Iersel, M.W. Southern highbush blueberry production in high tunnels: Temperatures, development, yield, and fruit quality during the establishment years. Hortscience 2009, 44, 1850–1856. [Google Scholar] [CrossRef]
- Das, Q.; Islam, M.R.; Marcone, M.F.; Warriner, K.; Diarra, M.S. Potential of berry extracts to control foodborne pathogens. Food Control 2016. [Google Scholar] [CrossRef]
- Khalifa, H.O.; Kamimoto, M.; Shimamoto, T.; Shimamoto, T. Antimicrobial effects of blueberry, raspberry, and strawberry aqueous extracts and their effects on virulence gene expression in Vibrio cholerae. Phytother. Res. 2015, 29, 1791–1797. [Google Scholar] [CrossRef] [PubMed]
- Lacombe, A.; Tadepalli, S.; Hwang, C.A.; Wu, V.C. Phytochemicals in lowbush wild blueberry inactivate Escherichia coli O157:H7 by damaging its cell membrane. Foodborne Pathog. Dis. 2013, 10, 944–950. [Google Scholar] [CrossRef]
- Lacombe, A.; Wu, V.C.H.; White, J.; Tadepalli, S.; Andre, E.E. The antimicrobial properties of the lowbush blueberry (Vaccinium angustifolium) fractional components against foodborne pathogens and the conservation of probiotic Lactobacillus rhamnosus. Food Microbiol. 2012, 30, 124–131. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.J.; Biswas, R.; Phillips, R.D.; Chen, J. Antibacterial activities of blueberry and muscadine phenolic extracts. J. Food Sci. 2011, 76, M101–M105. [Google Scholar] [CrossRef] [PubMed]
- Shen, X.; Sun, X.; Xie, Q.; Liu, H.; Zhao, Y.; Pan, Y.; Hwang, C.-A.; Wu, V.C.H. Antimicrobial effect of blueberry (Vaccinium corymbosum L.) extracts against the growth of Listeria monocytogenes and Salmonella Enteritidis. Food Control 2014, 35, 159–165. [Google Scholar] [CrossRef]
- Silva, S.; Costa, E.M.; Mendes, M.; Morais, R.M.; Calhau, C.; Pintado, M.M. Antimicrobial, antiadhesive and antibiofilm activity of an ethanolic, anthocyanin-rich blueberry extract purified by solid phase extraction. J. Appl. Microbiol. 2016, 121, 693–703. [Google Scholar] [CrossRef]
- Sun, X.H.; Hao, L.R.; Xie, Q.C.; Lan, W.Q.; Zhao, Y.; Pan, Y.J.; Wu, V.C. Antimicrobial effects and membrane damage mechanism of blueberry (Vaccinium corymbosum L.) extract against Vibrio parahaemolyticus. Food Control 2020, 111. [Google Scholar] [CrossRef]
- Sun, X.H.; Zhou, T.T.; Wei, C.H.; Lan, W.Q.; Zhao, Y.; Pan, Y.J.; Wu, V.C. Antibacterial effect and mechanism of anthocyanin rich Chinese wild blueberry extract on various foodborne pathogens. Food Control 2018, 94, 155–161. [Google Scholar] [CrossRef]
- Zhou, T.-T.; Wei, C.-H.; Lan, W.-Q.; Zhao, Y.; Pan, Y.-J.; Sun, X.-H.; Wu, V.C.H. The effect of Chinese wild blueberry fractions on the growth and membrane integrity of various foodborne pathogens. J. Food Sci. 2020. [Google Scholar] [CrossRef]
- Zimmer, K.R.; Blum-Silva, C.H.; Souza, A.L.K.; WulffSchuch, M.; Reginatto, F.H.; Pereira, C.M.P.; Macedo, A.J.; Lencina, C.L. The antibiofilm effect of blueberry fruit cultivars against Staphylococcus epidermidis and Pseudomonas aeruginosa. J. Med. Food 2014, 17, 324–331. [Google Scholar] [CrossRef]
- Denev, P.; Ciz, M.; Kratchanova, M.; Blazheva, D. Black chokeberry (Aronia melanocarpa) polyphenols reveal different antioxidant, antimicrobial and neutrophil-modulating activities. Food Chem. 2019, 284, 108–117. [Google Scholar] [CrossRef] [PubMed]
- Jurikova, T.; Skrovankova, S.; Mlcek, J.; Balla, S.; Snopek, L. Bioactive compounds, antioxidant activity, and biological effects of European ranberry (Vaccinium oxycoccos). Molecules 2019, 24, 24. [Google Scholar] [CrossRef]
- Ofek, I.; Goldhar, J.; Zafriri, D.; Lis, H.; Adar, R.; Sharon, N. Anti-Escherichia-coli adhesin activity of cranberry and blueberry juices. N. Engl. J. Med. 1991, 324, 1599. [Google Scholar] [PubMed]
- Schmidt, B.M.; Howell, A.B.; McEniry, B.; Knight, C.T.; Seigler, D.; Erdman, J.W., Jr.; Lila, M.A. Effective separation of potent antiproliferation and antiadhesion components from wild blueberry (Vaccinium angustifolium Ait.) fruits. J. Agric. Food Chem. 2004, 52, 6433–6442. [Google Scholar] [CrossRef] [PubMed]
- Dorneanu, R.; CIOANCĂ, O.; Chifiriuc, O.; Albu, E.; TUCHILUŞ, C.; Mircea, C.; Salamon, I.; HĂNCIANU, M. Synergic benefits of Aronia melanocarpa anthocyanin–rich extracts and antibiotics used for urinary tract infections. FARMACIA 2017, 65, 778–783. [Google Scholar]
- Salaheen, S.; Peng, M.; Joo, J.; Teramoto, H.; Biswas, D. Eradication and sensitization of methicillin resistant Staphylococcus aureus to methicillin with bioactive extracts of berry pomace. Front. Microbiol. 2017, 8, 253. [Google Scholar] [CrossRef]
- Nohynek, L.J.; Alakomi, H.L.; Kahkonen, M.P.; Heinonen, M.; Helander, I.M.; Oksman-Caldentey, K.M.; Puupponen-Pimia, R.H. Berry phenolics: Antimicrobial properties and mechanisms of action against severe human pathogens. Nutr. Cancer 2006, 54, 18–32. [Google Scholar] [CrossRef]
- Marhuenda, J.; Alemán, M.D.; Gironés-Vilaplana, A.; Pérez, A.; Caravaca, G.; Figueroa, F.; Mulero, J.; Zafrilla, P. Phenolic composition, antioxidant activity, and in vitro availability of four different berries. J. Chem. 2016, 2016. [Google Scholar] [CrossRef]
- Hornedo-Ortega, R.; Álvarez-Fernández, M.A.; Cerezo, A.B.; Garcia-Garcia, I.; Troncoso, A.M.; Garcia-Parrilla, M.C. Influence of fermentation process on the anthocyanin composition of wine and vinegar elaborated from strawberry. J. Food Sci. 2017, 82, 364–372. [Google Scholar] [CrossRef]
- Giusti, M.M.; Wrolstad, R.E. Characterization and measurement of anthocyanins by UV-visible spectroscopy. Curr. Protoc. Food Anal. Chem. 2001, 1, F1.2.1–F1.2.13. [Google Scholar] [CrossRef]
- Ou, B.; Hampsch-Woodill, M.; Prior, R.L. Development and validation of an improved oxygen radical absorbance capacity assay using fluorescein as the fluorescent probe. J. Agric. Food Chem. 2001, 49, 4619–4626. [Google Scholar] [CrossRef] [PubMed]
- Holm, A.; Aabenhus, R. Urine sampling techniques in symptomatic primary-care patients: A diagnostic accuracy review. BMC Fam. Pract. 2016, 17, 72. [Google Scholar] [CrossRef] [PubMed]
- Abrar, S.; Ain, N.U.; Liaqat, H.; Hussain, S.; Rasheed, F.; Riaz, S. Distribution of blaCTX-M, blaTEM, blaSHV and blaOXA genes in extended-spectrum-beta-lactamase-producing clinical isolates: A three-year multi-center study from Lahore, Pakistan. Antimicrob Resist. Infect Control. 2019, 8, 80. [Google Scholar] [CrossRef] [PubMed]
- Facklam, R.; Elliott, J.A. Identification, classification, and clinical relevance of catalase-negative, Gram-positive Cocci, excluding the Streptococci and Enterococci. Clin. Microbiol. Rev. 1995, 8, 479–495. [Google Scholar] [CrossRef] [PubMed]
- Preuss, H.G.; Echard, B.; Enig, M.; Brook, I.; Elliott, T.B. Minimum inhibitory concentrations of herbal essential oils and monolaurin for Gram-positive and Gram-negative bacteria. Mol. Cell. Biochem. 2005, 272, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Semeniuc, C.A.; Pop, C.R.; Rotar, A.M. Antibacterial activity and interactions of plant essential oil combinations against Gram-positive and Gram-negative bacteria. J. Food Drug Anal. 2017, 25, 403–408. [Google Scholar] [CrossRef] [PubMed]
- Addinsoft. XLSTAT Statistical and Data Analysis Solution; Addinsoft: Long Island, NY, USA, 2020. [Google Scholar]
- Rodriguez-Mateos, A.; Cifuentes-Gomez, T.; Tabatabaee, S.; Lecras, C.; Spencer, J.P. Procyanidin, anthocyanin, and chlorogenic acid contents of highbush and lowbush blueberries. J. Agric. Food Chem. 2012, 60, 5772–5778. [Google Scholar] [CrossRef]
- Wang, H.; Guo, X.; Hu, X.; Li, T.; Fu, X.; Liu, R.H. Comparison of phytochemical profiles, antioxidant and cellular antioxidant activities of different varieties of blueberry (Vaccinium spp.). Food Chem. 2017, 217, 773–781. [Google Scholar] [CrossRef]
- Kalt, W.; Ryan, D.A.; Duy, J.C.; Prior, R.L.; Ehlenfeldt, M.K.; Vander Kloet, S.P. Interspecific variation in anthocyanins, phenolics, and antioxidant capacity among genotypes of highbush and lowbush blueberries (Vaccinium section cyanococcus spp.). J. Agric. Food Chem. 2001, 49, 4761–4767. [Google Scholar] [CrossRef]
- Wang, Y.; Zhu, J.; Meng, X.; Liu, S.; Mu, J.; Ning, C. Comparison of polyphenol, anthocyanin and antioxidant capacity in four varieties of Lonicera caerulea berry extracts. Food Chem. 2016, 197, 522–529. [Google Scholar] [CrossRef]
- Cho, M.J.; Howard, L.R.; Prior, R.L.; Clark, J.R. Flavonoid glycosides and antioxidant capacity of various blackberry, blueberry and red grape genotypes determined by high-performance liquid chromatography/mass spectrometry. J. Sci. Food Agric. 2004, 84, 1771–1782. [Google Scholar] [CrossRef]
- Huang, H.; Sun, Y.; Lou, S.; Li, H.; Ye, X. In vitro digestion combined with cellular assay to determine the antioxidant activity in Chinese bayberry (Myrica rubra Sieb. et Zucc.) fruits: A comparison with traditional methods. Food Chem. 2014, 146, 363–370. [Google Scholar] [CrossRef] [PubMed]
- Borges, G.; Degeneve, A.; Mullen, W.; Crozier, A. Identification of flavonoid and phenolic antioxidants in black currants, blueberries, raspberries, red currants, and cranberries. J. Agric. Food Chem. 2010, 58, 3901–3909. [Google Scholar] [CrossRef] [PubMed]
- Gavrilova, V.; Kajdzanoska, M.; Gjamovski, V.; Stefova, M. Separation, characterization and quantification of phenolic compounds in blueberries and red and black currants by HPLC-DAD-ESI-MSn. J. Agric. Food Chem. 2011, 59, 4009–4018. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.Y.; Chen, C.-T.; Sciarappa, W.; Wang, C.Y.; Camp, M.J. Fruit quality, antioxidant capacity, and flavonoid content of organically and conventionally grown blueberries. J. Agric. Food Chem. 2008, 56, 5788–5794. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Prior, R.L. Systematic identification and characterization of anthocyanins by HPLC-ESI-MS/MS in common foods in the United States: Fruits and berries. J. Agric. Food Chem. 2005, 53, 2589–2599. [Google Scholar] [CrossRef]
- Veberic, R.; Slatnar, A.; Bizjak, J.; Stampar, F.; Mikulic-Petkovsek, M. Anthocyanin composition of different wild and cultivated berry species. LWT Food Sci. Technol. 2015, 60, 509–517. [Google Scholar] [CrossRef]
- Moze, S.; Polak, T.; Gasperlin, L.; Koron, D.; Vanzo, A.; Poklar Ulrih, N.; Abram, V. Phenolics in Slovenian bilberries ( Vaccinium myrtillus L.) and blueberries (Vaccinium corymbosum L.). J. Agric. Food Chem. 2011, 59, 6998–7004. [Google Scholar] [CrossRef]
- Kim, S.A.; Kim, H.W.; Rhee, M.S. Cranberry extract with enhanced bactericidal activities against uropathogenic Escherichia coli within one minute of treatment. LWT 2019, 113. [Google Scholar] [CrossRef]
- Hayouni, E.A.; Miled, K.; Boubaker, S.; Bellasfar, Z.; Abedrabba, M.; Iwaski, H.; Oku, H.; Matsui, T.; Limam, F.; Hamdi, M. Hydroalcoholic extract based-ointment from Punica granatum L. peels with enhanced in vivo healing potential on dermal wounds. Phytomedicine 2011, 18, 976–984. [Google Scholar] [CrossRef]
- Lacombe, A.; Wu, V.C.H. The potential of berries to serve as selective inhibitors of pathogens and promoters of beneficial microorganisms. Food Qual. Saf. 2017, 1, 3–12. [Google Scholar] [CrossRef]
- Salaheen, S.; Jaiswal, E.; Joo, J.; Peng, M.; Ho, R.; Oconnor, D.; Adlerz, K.; Aranda-Espinoza, J.H.; Biswas, D. Bioactive extracts from berry byproducts on the pathogenicity of Salmonella Typhimurium. Int. J. Food Microbiol. 2016, 237, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Marcon, J.; Schubert, S.; Stief, C.G.; Magistro, G. In vitro efficacy of phytotherapeutics suggested for prevention and therapy of urinary tract infections. Infection 2019, 47, 937–944. [Google Scholar] [CrossRef] [PubMed]
- Silva, S.; Costa, E.M.; Pereira, M.F.; Costa, M.R.; Pintado, M.E. Evaluation of the antimicrobial activity of aqueous extracts from dry Vaccinium corymbosum extracts upon food microorganism. Food Control 2013, 34, 645–650. [Google Scholar] [CrossRef]
- Santos, T.R.J.; de Aquino Santana, L.C.L. Antimicrobial potential of exotic fruits residues. S. Afr. J. Bot. 2019, 124, 338–344. [Google Scholar] [CrossRef]
- Cătunescu, G.M.; Rotar, A.M.; Pop, C.R.; Diaconeasa, Z.; Bunghez, F.; Socaciu, M.-I.; Semeniuc, C.A. Influence of extraction pre-treatments on some phytochemicals and biological activity of Transylvanian cranberries (Vaccinium vitis-idea L.). LWT 2019, 102, 385–392. [Google Scholar] [CrossRef]
- Diaconeasa, Z.; Iuhas, C.I.; Ayvaz, H.; Rugină, D.; Stanilă, A.; Dulf, F.; Bunea, A.; Socaci, S.A.; Socaciu, C.; Pintea, A. Phytochemical characterization of commercial processed blueberry, blackberry, blackcurrant, cranberry, and raspberry and their antioxidant activity. Antioxidants 2019, 8, 540. [Google Scholar] [CrossRef]
- Ibrahim, O.M.S. In vivo and in vitro antibacterial activities of cranberry extract against E. coli O157:H7 in urinary tract infected rats. Adv. Anim. Vet. Sci. 2015, 3, 233–244. [Google Scholar] [CrossRef]
- Prieto, L.; Esteban, M.; Salinas, J.; Adot, J.M.; Arlandis, S.; Peri, L.; Cozar, J.M. Consensus document of the Spanish Urological Association on the management of uncomplicated recurrent urinary tract infections. Actas Urológicas Españolas Engl. Ed. 2015, 39, 339–348. [Google Scholar] [CrossRef]
| Variety | Ratio (mg Anthocyanin Extract/100 g FW) |
|---|---|
| Star | 704.9 |
| Snowchaser | 253.8 |
| Cristina Blue | 431.7 |
| Stella Blue | 439.1 |
| Variety | ORAC (µmol TE/100g FW) | Cropfile | References |
|---|---|---|---|
| Blomidom | 5538 ± 388 | USA | [73] |
| Northland | 6747 ± 121 | USA | [73] |
| Northblue | 4976 ± 722 | USA | [73] |
| North Country | 6265 ± 699 | USA | [73] |
| Chipava | 5856 ±165 | USA | [73] |
| Elliott | 4098 ± 436 | USA | [73] |
| Darrow | 3994 ± 522 | USA | [73] |
| Bluecrop | 4491 ± 190 | USA | [73] |
| Primary Operation Blue | 3649 ± 473 | USA | [73] |
| Send a Blow | 5070 ± 179 | USA | [73] |
| Berkley | 2627 ± 364 | USA | [73] |
| JK-M7 | 4114 ± 344 | USA | [73] |
| Brie G Kobita | 2737 ± 394 | USA | [73] |
| Duke | 3145 ± 263 | USA | [73] |
| Bluegold | 2121 ± 326 | Romania | [31] |
| Nui | 2235 ± 677 | Romania | [31] |
| Darrow | 2543 ± 219 | Romania | [31] |
| Legacy | 2899 ± 531 | Romania | [31] |
| Nelson | 3027 ± 474 | Romania | [31] |
| Hanna’s Choice | 2036 ± 223 | Romania | [31] |
| Toro | 3458 ± 325 | Romania | [31] |
| Retention Time (min) | Compounds | Molecular Formula (M+) | Calculated Mass (m/z) | Accurate Mass (m/z) | Error (ppm) | MS/MS Fragments | Variety |
|---|---|---|---|---|---|---|---|
| 4.03 | Delphinidin-3-O-galactoside | C21H21O12 | 465.1028 | 465.1023 | −1.0441 | 303.0501 | A; B; C; D |
| 4.30 | Delphinidin-3-O-glucoside * | C21H21O12 | 465.1028 | 465.1025 | −0.4535 | 303.0502 | A; B; C; D |
| 4.44 | Cyanidin-3-galactoside * | C21H21O11 | 449.1078 | 449.1073 | −1.1349 | 287.0552 | A; B; C; D |
| 4.47 | Delphinidin-3-O-arabinoside | C20H19O11 | 435.0922 | 435.0918 | −0.8334 | 303.0501 | A; B; C; D |
| 4.60 | Cyanidin-3-O-glucoside * | C21H21O11 | 449.1078 | 449.1073 | −1.1349 | 287.0543 | A; B; C; D |
| 4.65 | Petunidin-3-O-galactoside | C22H23O12 | 479.1184 | 479.1180 | −0.9383 | 317.0658 | A; B; C; D |
| 4.68 | Cyanidin-3-O-rutinoside | C27H31O15 | 595.1658 | 595.1650 | −1.1945 | 449.10784/ 287.05501 | D |
| 4.73 | Cyanidin-3-O-arabinoside | C20H19O10 | 419.0973 | 419.0968 | −1.1173 | 287.0550 | A; B; C; D |
| 4.76 | Petunidin-3-O-glucoside | C22H23O12 | 479.1184 | 479.1179 | −1.1346 | 317.0645 | A; B; C; D |
| 7.77 | Petunidin-3-O-rutinoside | C28H33O16 | 625.1763 | 625.1766 | 0.4215 | 479.1180 | D |
| 4.89 | Peonidin-3-O-galactoside | C22H23O11 | 463.1235 | 463.1230 | −1.1500 | 301.0708 | A; B; C; D |
| 4.89 | Petunidin-3-O-arabinoside | C21H21O11 | 449.1078 | 449.1073 | −1.1300 | 317.0656 | A; B; C; D |
| 4.99 | Malvidin-3-O-galactoside | C23H25O12 | 493.1341 | 493.1335 | −1.0913 | 331.0812 | A; B; C; D |
| 4.99 | Peonidin-3-O-glucoside * | C22H23O11 | 463.1235 | 463.1231 | −0.7546 | 301.0694 | A; B; C; D |
| 5.04 | Peonidin-3-O-rutinoside | C28H33O15 | 609.1814 | 609.1804 | −1.7904 | 301.0691 | A; B; C; D |
| 5.10 | Malvidin-3-O-glucoside * | C23H25O12 | 493.1341 | 493.1336 | −0.8437 | 331.0802 | A; B; C; D |
| 5.19 | Cyanidin-3-(6″-acetyl) galactoside | C23H23O12 | 491.1184 | 491.1181 | −0.6047 | 287.0551 | A; C |
| 5.22 | Malvidin-3-O-arabinoside | C22H23O11 | 463.1235 | 463.1231 | −0.9568 | 331.0813 | A; B; C; D |
| 5.33 | Delphinidin-3-(6″-acetyl) glucoside | C23H23O13 | 507.1133 | 507.1133 | −0.7986 | 303.0497 | A |
| 5.33 | Petunidin-3-(6″-acetyl) galactoside | C24H25O13 | 521.1290 | 521.1236 | −1.1710 | 317.0657 | A; B; D |
| 5.39 | Cyanidin-3-(6-acetyl) glucoside | C23H23O12 | 491.1184 | 491.1182 | −0.3561 | 287.0551 | A; C; D |
| 5.47 | Malvidin-3-(6″-acetyl) galactoside | C25H27O13 | 535.1145 | 535.1441 | −0.9590 | 331.0801 | D |
| 5.48 | Petunidin-3-(6″-acetyl) glucoside | C24H25O13 | 521.1290 | 521.1289 | −0.2340 | 317.0651 | A; B; C; D |
| 5.53 | Peonidin-3-(6″-acetyl) glucoside | C24H25O12 | 505.1341 | 505.1343 | 0.4501 | 301.0707 | A; B; C; D |
| 5.58 | Delphinidin-3-(6″-malonyl) glucoside | C24H23O15 | 551.1032 | 551.1034 | 0.4419 | 303.0501 | A; B; D |
| Type of Strain | UTI Strains | ATCC Standard Strains | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Gram staining of the strains | Gram-negative | Gram-positive | Gram-negative | Gram-positive | |||||
| Strains | Klebsiella pneumoniae ssp. pneumoniae | Providencia stuartii | Escherichia coli β-Haemolytic | Pseudomonas aeruginosa | Micrococcus spp. | Escherichia coli ATCC 25922 | Salmonella Enteritidis ATCC 13076 | Listeria monocytogenes ATCC 19114 | |
| Sample | |||||||||
| Cristina Blue | 9.52 ± 0 Ac | 1.78 ± 0.65 A,a | 3.74 ± 1.372 A,b | 0.85 ± 0.31 A,a | 1.41 ± 0.65 A,a | 3.74 ± 1.37 A,b | 2.16 ± 0 C,a | 4.54 ± 0 D,b | |
| Star | 9.52 ± 0 Ad | 1.78 ± 0.65 A,a,b | 3.74 ± 1.372 A,c | 0.49 ± 0 A,a | 2.57 ± 1.79 A,b,c | 3.74 ± 1.37 A,c | 1.03 ± 0 B,a,b | 2.16 ± 0 C,a,b,c | |
| Snowchaser | 9.52 ± 0 Ad | 1.78 ± 0.65 A,a,b | 3.74 ± 1.372 A,c | 0.58 ± 0.41 A,a | 1.78 ± 0.65 A,a,b | 3.74 ± 1.37 A,c | 1.78 ± 0.65 C,a,b | 2.16 ± 0 C,b | |
| Stella Blue | 9.52 ± 0 Ad | 1.78 ± 0.65 A,a,b | 3.74 ± 1.372 A,c | 0.40 ± 0.15 A,a | 0.76 ± 0.46 A,a,b | 3.74 ± 1.37 A,c | 2.16 ± 0 C,b | 1.41 ± 0.653 B,a,b | |
| Positive control (µg/mL) | 0.28 ± 0.20Aa | 1.45 ± 0.67 A,b | 0.12 ± 0.11 A,a | 3.85 ± 1.41 A,c | 0.02 ± 0 A,a | 0.05 ± 0.05 A,a | 0.69 ± 0.32 A,a,b | 0.02±0 A,b | |
| Type of Strain | UTI Strains | ATCC Standard Strains | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Gram staining of the strains | Gram-negative | Gram-positive | Gram-negative | Gram-positive | |||||
| Strains | Klebsiella pneumoniae ssp. pneumoniae | Providencia stuartii | Escherichia coli β-Haemolytic | Pseudomonas aeruginosa | Micrococcus spp. | Escherichia coli ATCC 25922 | Salmonella Enteritidis ATCC 13076 | Listeria monocytogenes ATCC 19114 | |
| Sample | |||||||||
| Cristina Blue | 9.52 | 4.54 | 4.54 | 1.03 | 2.16 | 4.54 | 2.16 | 4.54 | |
| Star | 9.52 | 4.54 | 9.52 | 1.03 | 4.54 | 4.54 | 4.54 | 9.52 | |
| Snowchaser | 9.52 | 2.16 | 4.54 | 1.03 | 2.16 | 4.54 | 4.54 | 4.54 | |
| Stella Blue | 9.52 | 2.16 | 4.54 | 1.03 | 1.03 | 9.52 | 2.16 | 2.16 | |
| Type of strain | UTI Strains | ATCC Standard Strains | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Gram staining of the strains | Gram-negative | Gram-positive | Gram-negative | Gram-positive | |||||
| Strains | Klebsiella pneumoniae ssp. pneumoniae | Providencia stuartii | Escherichia coli β-Haemolytic | Pseudomonas aeruginosa | Micrococcus spp. | Escherichia coli ATCC 25922 | Salmonella Enteritidis ATCC 13076 | Listeria monocytogenes ATCC 19114 | |
| Sample | |||||||||
| TAC | 0.69 | −0.24 | −0.81 | −0.08 | 0.86 | 0.69 | 0.96 | −0.22 | |
| ORAC | 0.13 | −0.04 | −0.25 | 0.18 | 0.39 | 0.70 | 0.56 | 0.15 | |
| Linear Regression Equations | Goodness of Fit, R2 | Statistical Significance of the Model, p |
|---|---|---|
| MIC S. ATCC = 1.74 − 1.46 × 10−3 × TAC + 2.69 × 10−4 × ORAC | 1.00 | 0.002 |
| MBC E. UTI = 3.64 + 5.83 × 10−3 × TAC − 6.00 × 10−4 × ORAC | 0.99 | 0.023 |
| MBC S. ATCC = 5.64 + 3.62 × 10−3 − 1.12 × 10−3 × ORAC | 0.99 | 0.033 |
| MBC E. UTI = 2.03 + 4.33 × 10−3 × TAC | 0.92 | 0.039 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cerezo, A.B.; Cătunescu, G.M.; González, M.M.-P.; Hornedo-Ortega, R.; Pop, C.R.; Rusu, C.C.; Chirilă, F.; Rotar, A.M.; Garcia-Parrilla, M.C.; Troncoso, A.M. Anthocyanins in Blueberries Grown in Hot Climate Exert Strong Antioxidant Activity and May Be Effective against Urinary Tract Bacteria. Antioxidants 2020, 9, 478. https://doi.org/10.3390/antiox9060478
Cerezo AB, Cătunescu GM, González MM-P, Hornedo-Ortega R, Pop CR, Rusu CC, Chirilă F, Rotar AM, Garcia-Parrilla MC, Troncoso AM. Anthocyanins in Blueberries Grown in Hot Climate Exert Strong Antioxidant Activity and May Be Effective against Urinary Tract Bacteria. Antioxidants. 2020; 9(6):478. https://doi.org/10.3390/antiox9060478
Chicago/Turabian StyleCerezo, Ana B., Giorgiana M. Cătunescu, Mercedes Martínez-Pais González, Ruth Hornedo-Ortega, Carmen R. Pop, Crina Claudia Rusu, Flore Chirilă, Ancuța M. Rotar, M. Carmen Garcia-Parrilla, and Ana M. Troncoso. 2020. "Anthocyanins in Blueberries Grown in Hot Climate Exert Strong Antioxidant Activity and May Be Effective against Urinary Tract Bacteria" Antioxidants 9, no. 6: 478. https://doi.org/10.3390/antiox9060478
APA StyleCerezo, A. B., Cătunescu, G. M., González, M. M.-P., Hornedo-Ortega, R., Pop, C. R., Rusu, C. C., Chirilă, F., Rotar, A. M., Garcia-Parrilla, M. C., & Troncoso, A. M. (2020). Anthocyanins in Blueberries Grown in Hot Climate Exert Strong Antioxidant Activity and May Be Effective against Urinary Tract Bacteria. Antioxidants, 9(6), 478. https://doi.org/10.3390/antiox9060478








