Biological Activity of Phenolic Compounds in Extra Virgin Olive Oils through Their Phenolic Profile and Their Combination with Anticancer Drugs Observed in Human Cervical Carcinoma and Colon Adenocarcinoma Cells
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents and Standards
2.2. Olive Oil Samples
2.3. Extraction of Phenolic Compounds
2.4. HPLC-DAD Analysis
2.5. Total Phenols Analyses (TP)
2.6. o-Diphenols Analyses
2.7. Total Flavonoids Analyses (TF)
2.8. Scavenging Effect Assay
2.9. Cell Culture and Biological Activity
2.10. Statistical Data Analysis
3. Results
3.1. Characteristics of Phenolic Extracts
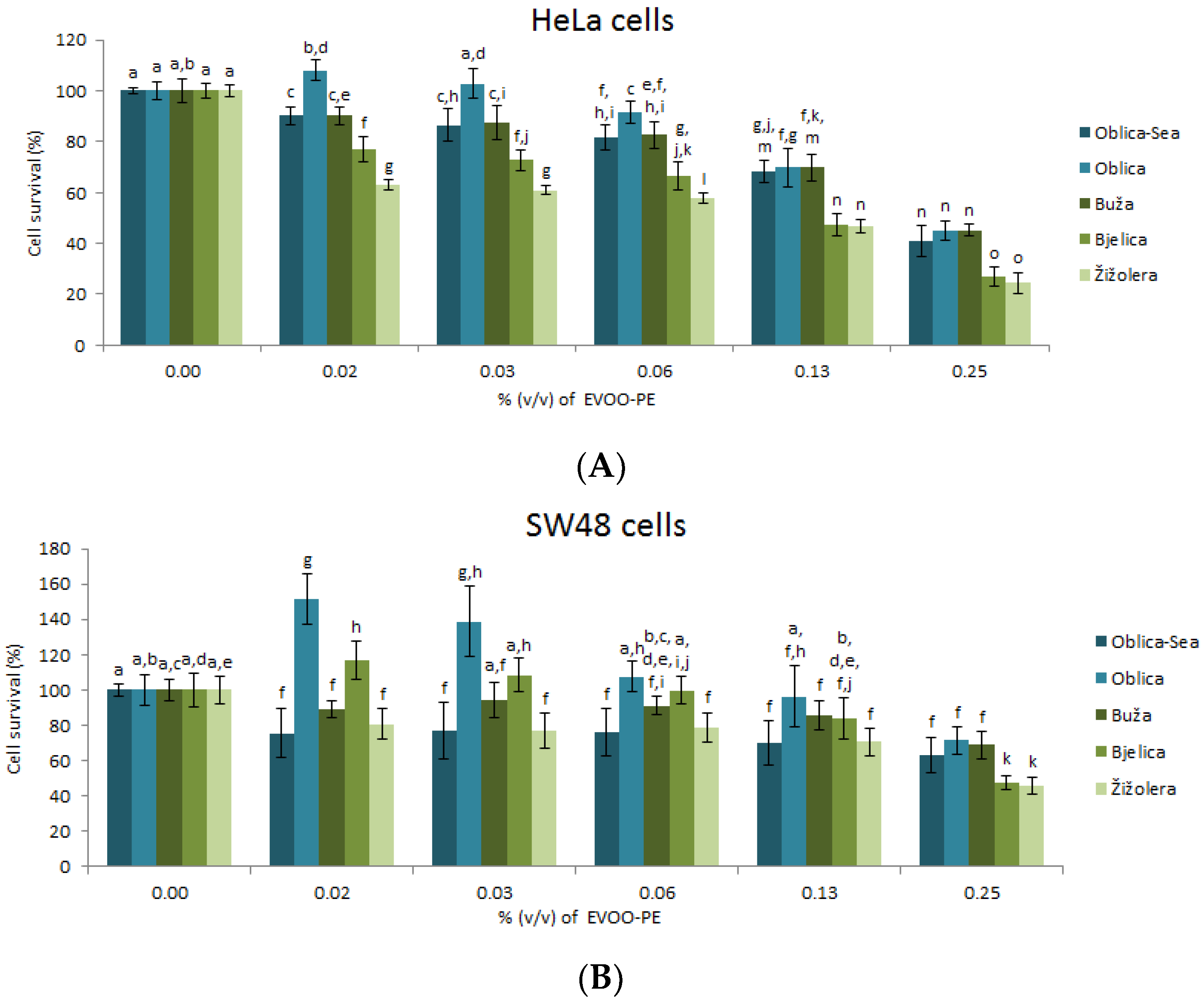
3.2. Biological Effects of EVOO-PEs on Cells Survival of HeLa and SW48
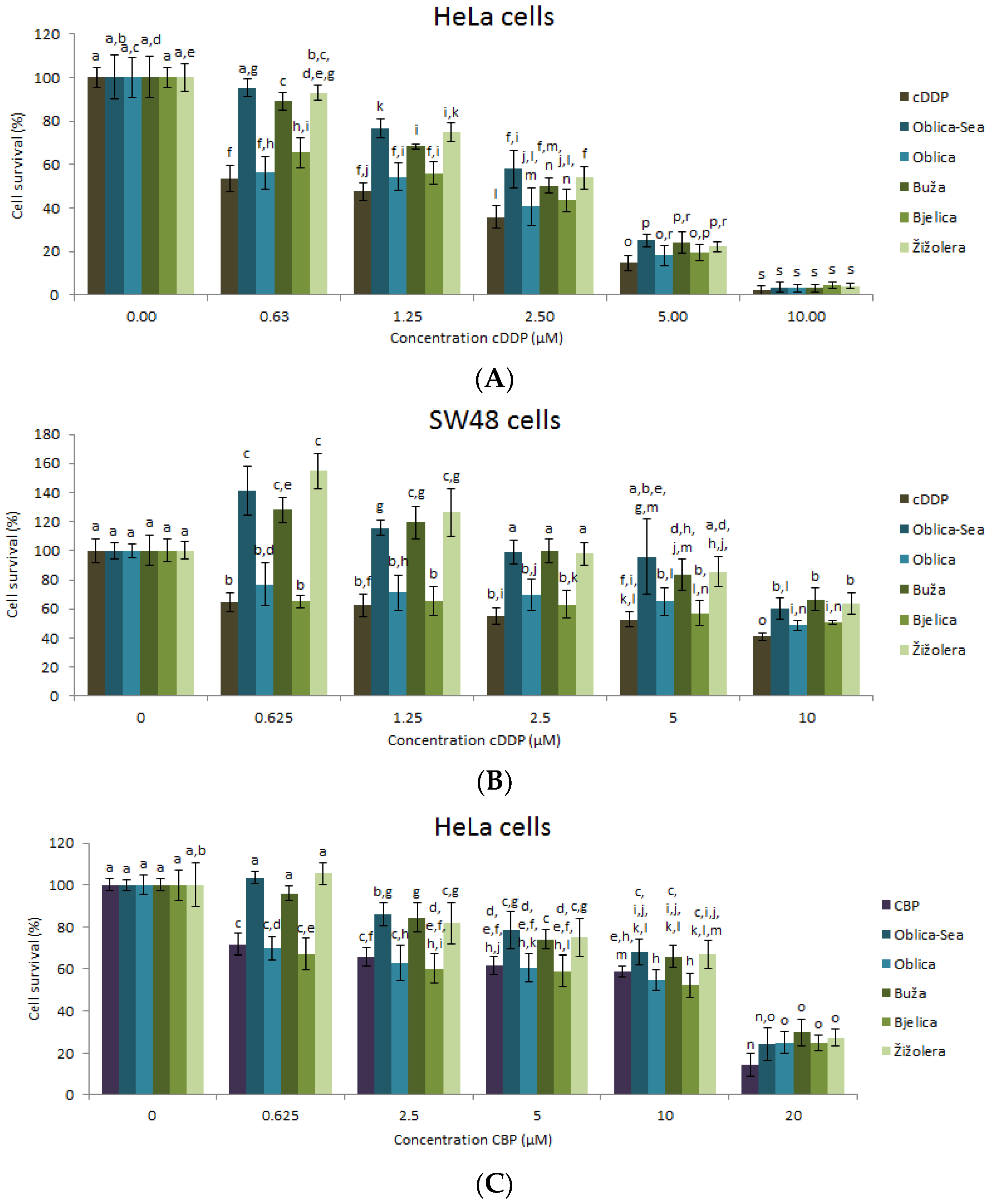
3.3. Biological Effects of EVOO-PEs in Combination with Anticancer Drugs on Cells Survival of HeLa and SW48
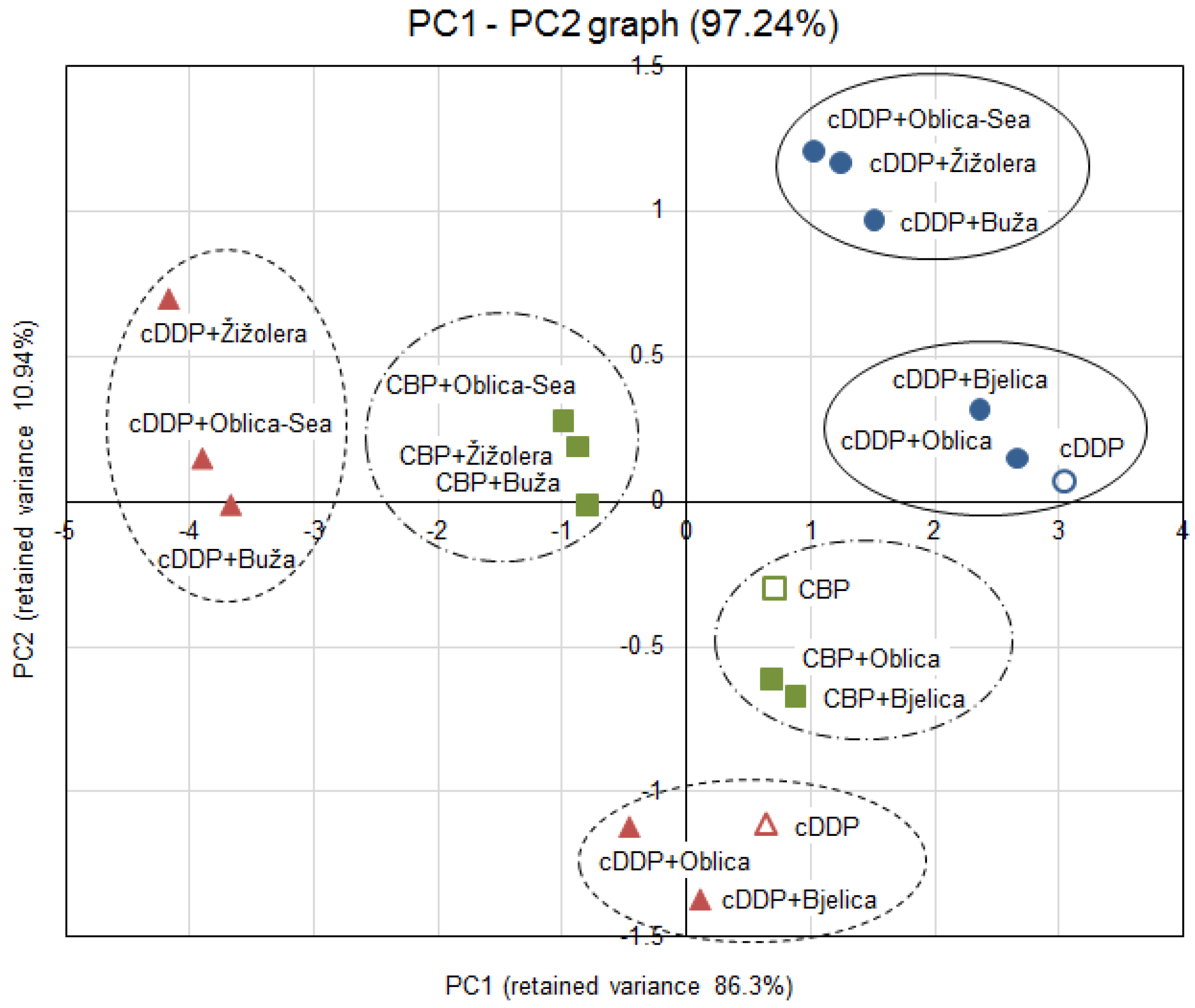
3.4. Statistical Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef]
- WHO (World Health Organisation). Available online: http://www.who.int/cancer/en/ (accessed on 11 February 2020).
- Karković Marković, A.; Torić, J.; Barbarić, M.; Jakobušić Brala, C. Hydroxytyrosol, tyrosol and derivatives and their potential effects on human health. Molecules 2019, 24, 2001. [Google Scholar] [CrossRef]
- Torić, J.; Karković Marković, A.; Jakobušić Brala, C.; Barbarić, M. Anticancer effects of olive oil polyphenols and their combinations with anticancer drugs. Acta Pharm. 2019, 69, 461–482. [Google Scholar] [CrossRef] [PubMed]
- Gorzynik-Debicka, M.; Przychodzen, P.; Cappello, F.; Kuban-Jankowska, A.; Gammazza, A.M.; Knap, N.; Wozniak, M.; Gorska-Ponikowska, M. Potential health benefits of olive oil and plant polyphenols. Int. J. Mol. Sci. 2018, 19, 686. [Google Scholar] [CrossRef] [PubMed]
- Echeverría, F.; Ortiz, M.; Valenzuela, R.; Videla, L.A. Hydroxytyrosol and cytoprotection: A projection for clinical interventions. Int. J. Mol. Sci. 2017, 18, 930. [Google Scholar] [CrossRef]
- Fabiani, R.; Rosignoli, P.; De Bartolomeo, A.; Fuccelli, R.; Servili, M.; Montedoro, G.F.; Morozzi, G. Oxidative DNA damage is prevented by extracts of olive oil, hydroxytyrosol, and other olive phenolic compounds in human blood mononuclear cells and HL60 cells. J. Nutr. 2008, 138, 1411–1416. [Google Scholar] [CrossRef] [PubMed]
- Gill, C.I.R.; Boyd, A.; McDermott, E.; McCann, M.; Servili, M.; Selvaggini, R.; Taticchi, A.; Esposto, S.; Montedoro, G.; McGlynn, H.; et al. Potential anti-cancer effects of virgin olive oil phenolson colorectal carcinogenesis modelsin vitro. Int. J. Cancer 2015, 117, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Hashim, Y.Z.H.-Y.; Worthington, J.; Allsopp, P.; Ternan, N.G.; Brown, E.M.; McCann, M.J.; Rowland, I.R.; Esposto, S.; Servili, M.; Gill, C.I.R.; et al. Virgin olive oil phenolics extract inhibit invasion of HT115 human colon cancer cells in vitro and in vivo. Food Funct. 2014, 5, 1513. [Google Scholar] [CrossRef]
- Di Francesco, A.; Falconi, A.; Di Germanio, C.; Micioni Di Bonaventura, M.V.; Costa, A.; Caramuta, S.; Del Carlo, M.; Compagnone, D.; Dainese, E.; Cifani, C.; et al. Extravirgin olive oil up-regulates CB1 tumor suppressor gene in human colon cancer cells and in rat colon via epigenetic mechanisms. J. Nutr. Biochem. 2015, 26, 250–258. [Google Scholar] [CrossRef]
- Pampaloni, B.; Mavilia, C.; Fabbri, S.; Romani, A.; Ieri, F.; Tanini, A.; Tonelli, F.; Brandi, M.L. In vitro effects of extracts of extra virgin olive oil on human colon cancer cells. Nutr. Cancer 2014, 66, 1228–1236. [Google Scholar] [CrossRef]
- Coccia, A.; Bastianelli, D.; Mosca, L.; Monticolo, R.; Panuccio, I.; Carbone, A.; Calogero, A.; Lendaro, E. Extra virgin olive oil phenols suppress migration and invasion of T24 human bladder cancer cells through modulation of matrix Metalloproteinase-2. Nutr. Cancer 2014, 66, 946–954. [Google Scholar] [CrossRef] [PubMed]
- Polini, B.; Digiacomo, M.; Carpi, S.; Bertini, S.; Gado, F.; Saccomanni, G.; Macchia, M.; Nieri, P.; Manera, C.; Fogli, S.; et al. Oleocanthal and oleacein contribute to the in vitro therapeutic potential of extra virgin oil-derived extracts in non-melanoma skin cancer. Toxicol. In Vitro 2018, 52, 243–250. [Google Scholar] [CrossRef] [PubMed]
- De Stefanis, D.; Scimè, S.; Accomazzo, S.; Catti, A.; Occhipinti, A.; Bertea, C.M.; Costelli, P. Anti-proliferative effects of an extra-virgin olive oil extract enriched in ligstroside aglycone and oleocanthal on human liver cancer cell lines. Cancers 2019, 11, 1640. [Google Scholar] [CrossRef] [PubMed]
- Oliveras-Ferraros, C.; Fernández-Arroyo, S.; Vazquez-Martin, A.; Lozano-Sánchez, J.; Cufí, S.; Joven, J.; Micol, V.; Fernández-Gutiérrez, A.; Segura-Carretero, A.; Menendez, J.A.; et al. Crude phenolic extracts from extra virgin olive oil circumvent de novo breast cancer resistance to HER1/ HER2-targeting drugs by inducing GADD45-sensed cellular stress, G2/M arrest and hyperacetylation of Histone H3. Int. J. Oncol. 2011, 38, 1533–1547. [Google Scholar] [CrossRef] [PubMed]
- Özlem, E.; Arda, N.; Erdem, G. Phenols of virgin olive oil protects nuclear DNA against oxidative damage in HeLa cells. Food Chem. Toxicol. 2012, 50, 3475–3479. [Google Scholar]
- Lewandowska, U.; Gorlach, S.; Owczarek, K.; Hrabec, E.; Szewczyk, K. Synergistic interactions between anticancer chemotherapeutics and phenolic compounds and anticancer synergy between polyphenols. Postępy Higieny i Medycyny Doświadczalnej 2014, 68, 528–540. [Google Scholar] [CrossRef]
- Wagner, H. Synergy research: Approaching a new generation of phytopharmaceuticals. Fitoterapia 2011, 82, 34–37. [Google Scholar] [CrossRef]
- Coccia, A.; Mosca, L.; Puca, R.; Mangino, G.; Rossi, A.; Lendaro, E. Extra-virgin olive oil phenols block cell cycle progression and modulate chemotherapeutic toxicity in bladder cancer cells. Oncol. Rep. 2016, 36, 3095–3104. [Google Scholar] [CrossRef]
- Torić, J.; Barbarić, M.; Jakobušić Brala, C.; Karković Marković, A.; Benčić, Đ.; Zebić Avdičević, M.; Uršić, S. Tradition and Flavour vs. Biomedical Significance: A Case of Virgin Olive Oil Obtained from Fruits Stored in Seawater; unpublished; manuscript in preparation.
- Jerman Klen, T.; Mozetič Vodopivec, B. Optimisation of olive oil phenol extraction conditions using a high-power probe ultrasonication. Food Chem. 2012, 134, 2481–2488. [Google Scholar] [CrossRef]
- Jakobušić Brala, C.; Benčić, D.; Šindrak, Z.; Barbarić, M.; Uršić, S. Labeled extra virgin olive oil as food supplement; phenolic compounds in oils from some autochthonous Croatian olives. Grasas y Aceites 2015, 66, e099. [Google Scholar] [CrossRef]
- Owen, R.W.; Giacosa, A.; Hull, W.E.; Haubner, R.; Spiegelhalder, B.; Bartsch, H. The antioxidant/anticancer potential of phenolic compounds isolated from olive oil. Eur. J. Cancer 2000, 36, 1235–1247. [Google Scholar] [CrossRef]
- Antonini, E.; Farina, A.; Leone, A.; Mazzara, E.; Urbani, S.; Selvaggini, R.; Servili, M.; Ninfali, P. Phenolic compounds and quality parameters of family farming versus protected designation of origin (PDO) extra-virgin olive oils. J. Food Compos. Anal. 2015, 43, 75–81. [Google Scholar] [CrossRef]
- Gutfinger, T. Polyphenols in olive oils. J. Am. Oil Chem. Soc. 1981, 58, 966–968. [Google Scholar] [CrossRef]
- Mateos, R.; Espartero, J.L.; Trujillo, M.; Ríos, J.J.; León-Camacho, M.; Alcudia, F.; Cert, A. Determination of phenols, flavones, and lignans in virgin olive oils by solid-phase extraction and high-performance liquid chromatography with diode array ultraviolet detection. J. Agric. Food Chem. 2001, 49, 2185–2192. [Google Scholar] [CrossRef] [PubMed]
- Kim, D. Antioxidant capacity of phenolic phytochemicals from various cultivars of plums. Food Chem. 2003, 81, 321–326. [Google Scholar] [CrossRef]
- Villaño, D.; Fernández-Pachón, M.S.; Moyá, M.L.; Troncoso, A.M.; García-Parrilla, M.C. Radical scavenging ability of polyphenolic compounds towards DPPH free radical. Talanta 2007, 71, 230–235. [Google Scholar] [CrossRef]
- Mickisch, G.; Fajta, S.; Keilhauer, G.; Schlick, E.; Tschada, R.; Alken, P. Chemosensitivity testing of primary human renal cell carcinoma by a tetrazolium based microculture assay (MTT). Urol. Res. 1990, 18, 131–136. [Google Scholar] [CrossRef]
- Sindona, G. Virtual freezing of the hemiacetal-aldehyde equilibrium of the aglycones of oleuropein and ligstroside present in olive oils from Carolea and Coratina cultivars by ionspray ionization tandem mass spectrometry. J. Mass Spectrom. 2000, 35, 461–467. [Google Scholar] [CrossRef]
- Fletcher, C.; Wilson, C.; Hutchinson, A.D.; Grunfeld, E.A. The relationship between anticipated response and subsequent experience of cancer treatment-related side effects: A meta-analysis comparing effects before and after treatment exposure. Cancer Treat. Rev. 2018, 68, 86–93. [Google Scholar] [CrossRef]
- Pistritto, G.; Trisciuoglio, D.; Ceci, C.; Garufi, A.; D’Orazi, G. Apoptosis as anticancer mechanism: Function and dysfunction of its modulators and targeted therapeutic strategies. Aging 2016, 8, 603–619. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.Z.; Kanagaratham, C.; Youssef, M.; Radzioch, D. New frontiers in cancer chemotherapy—Targeting cell death pathways. In Cell Biology—New Insights, 1st ed.; Najman, S., Ed.; InTechOpen: London, UK, 2016; pp. 93–140. ISBN 978-953-51-5420-4. [Google Scholar] [CrossRef]
- Cicerale, S.; Lucas, L.; Keast, R. Antimicrobial, antioxidant and anti-inflammatory phenolic activities in extra virgin olive oil. Curr. Opin. Biotechnol. 2012, 23, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Assaraf, Y.G.; Brozovic, A.; Gonçalves, A.C.; Jurkovicova, D.; Linē, A.; Machuqueiro, M.; Saponara, S.; Sarmento-Ribeiro, A.B.; Xavier, C.P.R.; Vasconcelos, M.H.; et al. The multi-factorial nature of clinical multidrug resistance in cancer. Drug Resist. Updates 2019, 46, 100645. [Google Scholar] [CrossRef] [PubMed]
- Brozovic, A. The relationship between platinum drug resistance and epithelial-mesenchymal transition. Arch. Toxicol. 2017, 91, 605–619. [Google Scholar] [CrossRef] [PubMed]
- Brozovic, A.; Ambriović-Ristov, A.; Osmak, M. The relationship between cisplatin-induced reactive oxygen species, glutathione, and BCL-2 and resistance to cisplatin. Crit. Rev. Toxicol. 2010, 40, 347–359. [Google Scholar] [CrossRef] [PubMed]
- Brozović, A.; Majhen, D.; Roje, V.; Mikac, N.; Jakopec, S.; Fritz, G.; Osmak, M.; Ambriović-Ristov, A. αvβ3 integrin mediated drug resistance in human laryngeal carcinoma cells is caused by glutathione dependent elimination of drug induced reactive oxidative species. Mol. Pharmacol. 2008, 74, 298–306. [Google Scholar] [CrossRef]
- Brozovic, A.; Vuković, L.; Polančac, D.S.; Arany, I.; Köberle, B.; Fritz, G.; Fiket, Z.; Majhen, D.; Ambriović-Ristov, A.; Osmak, M.; et al. Endoplasmic reticulum stress is involved in the response of human laryngeal carcinoma cells to Carboplatin but is absent in Carboplatin-resistant cells. PLoS ONE 2013, 8, e76397. [Google Scholar] [CrossRef]
- Galano, A.; Mazzone, G.; Alvarez-Diduk, R.; Marino, T.; Alvarez-Idaboy, J.R.; Russo, N. Food antioxidants: Chemical insights at the molecular level. Annu. Rev. Food Sci. Technol. 2016, 7, 335–352. [Google Scholar] [CrossRef]
| EVOO-PE (Cultivar) | |||||
|---|---|---|---|---|---|
| Oblica-Sea | Oblica | Buža | Bjelica | Žižolera | |
| TP (mg GAE/kg EVOO ± SD) | 319 ± 48 A | 682 ± 8 B | 270 ± 8 A | 423 ± 38 A | 391 ± 34 A |
| o-diphenols (mg GAE/kg EVOO ± SD) | 136 ± 4 A | 277 ± 40 B | 114 ± 3 C | 122 ± 11 A,C | 128 ± 7 A,C |
| TF (mg CE/kg EVOO ± SD) | 279 ± 53 A | 606 ± 71 B | 199 ± 3 A | 230 ± 42 A | 241 ± 6 A |
| EC50 | 158 | 191 | 190 | 208 | 198 |
| Phenolic compounds (mg/kg EVOO ± SD) | |||||
| HTyr | 12.73 ± 0.12 A | 21.23 ± 0.15 B | 4.07 ± 0.17 C | 4.85 ± 0.10 D | 4.48 ± 0.14 C,D |
| 3,4-dihydroxybenzoic acid | ND | ND | ND | ND | ND |
| Tyr | 7.69 ± 0.11 A | 9.26 ± 0.08 B | 4.57 ± 0.01 C | 5.47 ± 0.42 C,D | 5.14 ± 0.12 D |
| p-hydroxybenzoic acid | 0.11 ± 0.10 A | 0.08 ±0.02 B | 0.15 ± 0.01 A | 0.09 ± 0.02 A,B | 0.12 A ± 0.01 A |
| Homovanillyl alcohol | 0.26 ± 0.03 A | 0.43 ± 0.01 B | 0.27 ± 0.02 A | 0.17 ± 0.01 C | 0.18 ± 0.01 A,C |
| Vanillic acid | 0.95 ± 0.01 A | 0.77 ± 0.02 B | 0.92 ± 0.01 A | 0.50 ± 0.01 C | 0.69 ± 0.01 B |
| Syringic acid | ND | ND | ND | ND | ND |
| Vanillin | 0.30 ± 0.01 A | 0.54 ± 0.01 B | 0.46 ± 0.01 C | 0.29 ± 0.01 A | 0.37D ± 0.02 D |
| p-coumaric acid | 0.32 ± 0.03 A | 0.48 ± 0.01 B | 0.35 ± 0.01 C | 0.40 ± 0.02 C | 0.37 ± 0.01 C |
| Benzoic acid | 0.28 ± 0.14 A | 0.25 ± 0.14 A | 0.53 ± 0.10 A | 1.75 ± 0.19 B | 0.90 ± 0.36 A,B |
| Ferulic acid | 0.24 ± 0.04 A | 0.28 ± 0.03 A | 0.25 ± 0.01 A | 0.67 ± 0.07 B | 0.61 ± 0.04 B |
| Oleacein | 59 ± 1 A | 173 ± 6 B | 72 ± 1 C | 43 ± 1 D | 39 ± 1 E |
| o-coumaric acid | ND | ND | ND | ND | ND |
| Pinoresinol | 3.07 ± 0.04 A | 4.69 ± 0.01 B | 12.57 ± 0.06 C | 5.40 ± 0.33 B | 7.10 ± 0.16 D |
| Cinnamic acid | 0.93 ± 0.01 A | 1.06 ± 0.02 B | 0.36 ± 0.02 C | 0.41 ± 0.02 C | 0.51 ± 0.01 D |
| Apigenin | 1.36 ± 0.01 A | 0.99 ± 0.06 B | 1.01 ± 0.02 B | 0.87 ± 0.01 B | 0.85 ± 0.01 B |
| EVOO-PE (Cultivar) | |||||
|---|---|---|---|---|---|
| Oblica-Sea | Oblica | Buža | Bjelica | Žižolera | |
| HeLa IC65 (% v/v of EVOO ± SD) | 0.14 ± 0.02 A | 0.17 ± 0.02 A | 0.15 ± 0.02 A | 0.06 ± 0.01 B | 0.01 ± 0.01 C |
| SW48 IC65 (% v/v of EVOO ± SD) | 0.19 ± 0.15 A | 0.25 ± 0.03 A | 0.33 ± 0.09 A | 0.19 ± 0.01 A | 0.13 ± 0.05 A |
| HeLa IC65 | SW48 IC65 | |
|---|---|---|
| HeLa IC65 | NaN | 0.881 * |
| SW48 IC65 | 0.881 * | NaN |
| TP | 0.203 | −0.067 |
| o-diphenols | 0.498 | 0.196 |
| TF | 0.494 | 0.176 |
| EC50 | −0.50 | −0.247 |
| HTyr | 0.627 | 0.231 |
| Tyr | 0.558 | 0.12 |
| p-hydroxybenzoic acid | −0.026 | 0.332 |
| Homovanillyl alcohol | 0.799 | 0.578 |
| Vanillic acid | 0.641 | 0.593 |
| Vanillin | 0.538 | 0.56 |
| p-coumaric acid | 0.181 | 0.043 |
| Benzoic acid | −0.669 | −0.483 |
| Ferulic acid | −0.896 * | −0.782 |
| Oleacein | 0.688 | 0.474 |
| Pinoresinol | 0.037 | 0.488 |
| Cinnamic acid | 0.522 | 0.074 |
| Apigenin | 0.573 | 0.321 |
| Total phenolic alcohol | 0.617 | 0.213 |
| Total phenolic acids and derivatives | −0.562 | −0.594 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Torić, J.; Brozovic, A.; Baus Lončar, M.; Jakobušić Brala, C.; Karković Marković, A.; Benčić, Đ.; Barbarić, M. Biological Activity of Phenolic Compounds in Extra Virgin Olive Oils through Their Phenolic Profile and Their Combination with Anticancer Drugs Observed in Human Cervical Carcinoma and Colon Adenocarcinoma Cells. Antioxidants 2020, 9, 453. https://doi.org/10.3390/antiox9050453
Torić J, Brozovic A, Baus Lončar M, Jakobušić Brala C, Karković Marković A, Benčić Đ, Barbarić M. Biological Activity of Phenolic Compounds in Extra Virgin Olive Oils through Their Phenolic Profile and Their Combination with Anticancer Drugs Observed in Human Cervical Carcinoma and Colon Adenocarcinoma Cells. Antioxidants. 2020; 9(5):453. https://doi.org/10.3390/antiox9050453
Chicago/Turabian StyleTorić, Jelena, Anamaria Brozovic, Mirela Baus Lončar, Cvijeta Jakobušić Brala, Ana Karković Marković, Đani Benčić, and Monika Barbarić. 2020. "Biological Activity of Phenolic Compounds in Extra Virgin Olive Oils through Their Phenolic Profile and Their Combination with Anticancer Drugs Observed in Human Cervical Carcinoma and Colon Adenocarcinoma Cells" Antioxidants 9, no. 5: 453. https://doi.org/10.3390/antiox9050453
APA StyleTorić, J., Brozovic, A., Baus Lončar, M., Jakobušić Brala, C., Karković Marković, A., Benčić, Đ., & Barbarić, M. (2020). Biological Activity of Phenolic Compounds in Extra Virgin Olive Oils through Their Phenolic Profile and Their Combination with Anticancer Drugs Observed in Human Cervical Carcinoma and Colon Adenocarcinoma Cells. Antioxidants, 9(5), 453. https://doi.org/10.3390/antiox9050453