Aerobic Exercise and Healthy Nutrition as Neuroprotective Agents for Brain Health in Patients with Parkinson’s Disease: A Critical Review of the Literature
Abstract
1. Parkinson’s Disease (PD): Epidemiology and Clinical Features
2. PD and Aerobic Exercise
3. PD and Nutrition
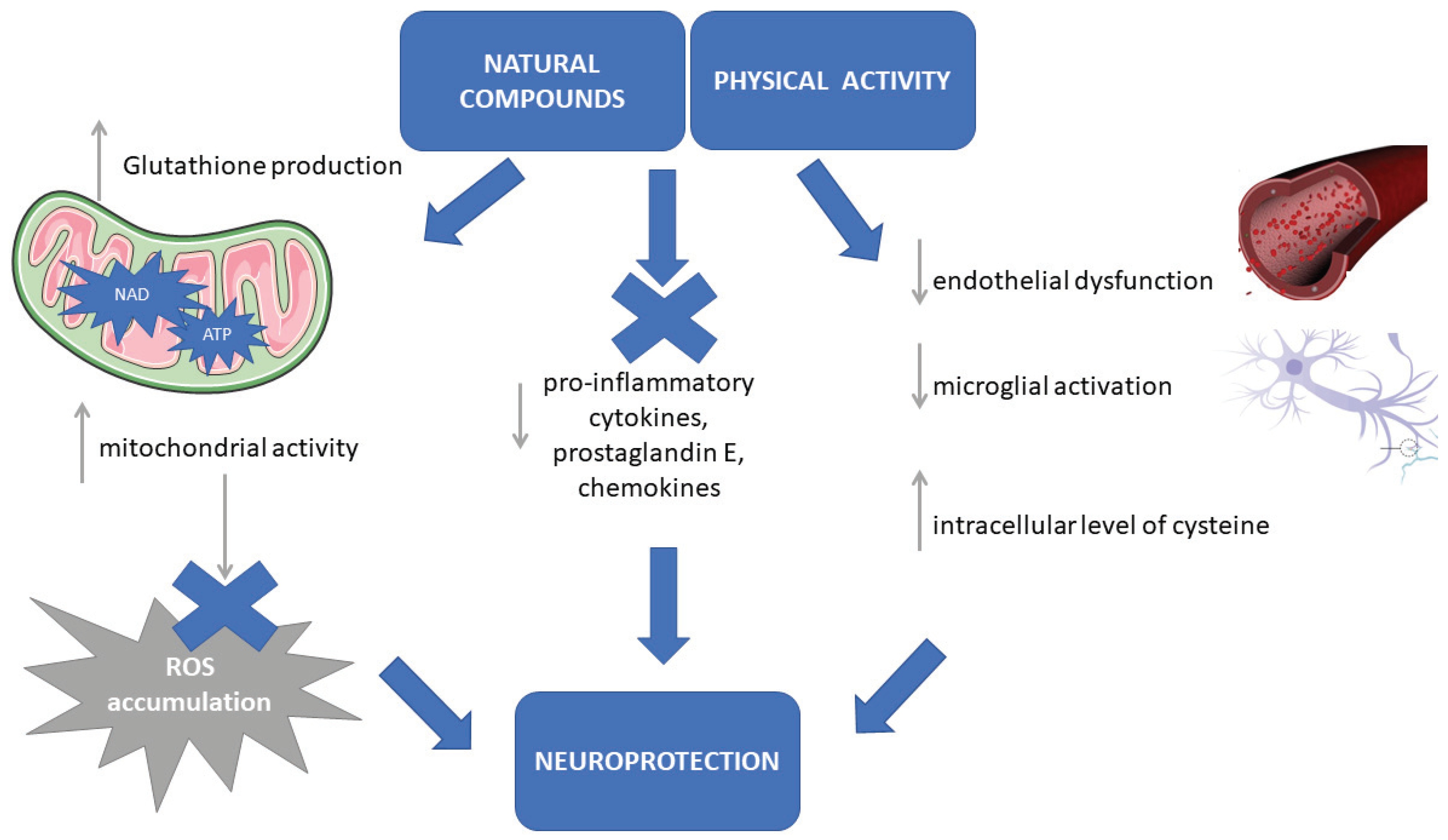
4. Neuroprotective Effects of Aerobic Exercise and Balanced Diet in PD
5. Implications for PD Therapeutic Plan and Rehabilitation
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Poewe, W.; Seppi, K.; Tanner, C.M.; Halliday, G.M.; Brundin, P.; Volkmann, J.; Schrag, A.-E.; Land, A.E. Parkinson’s disease. Nat. Rev. Dis. Primers 2017, 3, 17013. [Google Scholar] [CrossRef] [PubMed]
- Kalia, L.V.; Lang, A.E. Parkinson disease in 2015: Evolving basic, pathological and clinical concepts in PD. Nat. Rev. Neurol. 2016, 12, 65–66. [Google Scholar] [CrossRef] [PubMed]
- Meade, R.M.; Fairlie, D.P.; Mason, J.M. Alpha-synuclein structure and Parkinson’s disease—Lesson and emerging principles. Mol. Neurodegener. 2019, 14, 29. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, M.J.; Okun, M.S. Choosing a Parkinson disease treatment. JAMA. 2020, 323, 1420. [Google Scholar] [CrossRef] [PubMed]
- Monteiro, E.P.; Franzoni, L.T.; Cubillos, D.M.; de Oliveira Fagundes, A.; Carcalho, A.R.; Oliveira, H.B.; Pantoja, P.D.; Schuch, F.B.; Rieder, C.R.; Martinez, F.G.; et al. Effects of Nordic walking training on functional parameters in Parkinson’s disease: A randomized controlled trial. Scand. J. Med. Sci. Sports 2017, 27, 351–358. [Google Scholar] [CrossRef]
- Muthuraju, S.; Zakaria, R.; Karruppan, M.K.M.; Al-Rahbui, B. The Role of Neuroinflammation in Cellular Damage in Neurodegenerative Diseases. Biomed Res. Int. 2020, 2020. [Google Scholar] [CrossRef]
- Koutzoumis, D.N.; Vergara, M.; Pino, J.; Buddendorff, J.; Khoshbouei, H.; Mandel, R.J.; Torresd, G.E. Alterations of the gut microbiota with antibiotics protects dopamine neuron loss and improve motor deficits in a pharmacological rodent model of Parkinson’s disease. Exp. Neurol. 2020, 325, 113159. [Google Scholar] [CrossRef]
- Marinus, J.; Zhu, K.; Marras, C.; Aarsland, D.; van Hilten, J.J. Risk factors for non-motor symptoms in Parkinson’s disease. Lancet Neurol. 2018, 17, 559–568. [Google Scholar] [CrossRef]
- Wojtala, J.; Heber, I.A.; Neuser, P.; Heller, J.; Kalbe, E.; Rehberg, S.P.; Storch, A.; Linse, K.; Schneider, C.; Gräber, S. Cognitive decline in Parkinson’s disease: The impact of the motor phenotype on cognition. J. Neurol. Neurosurg. Psychiatry 2019, 90, 171–179. [Google Scholar] [CrossRef]
- Vasconcellos, L.F.R.; Pereira, J.S.; Charchat-Fichman, H.; Greca, D.; Cruz, M.; Blum, A.L.; Spitz, M. Mild cognitive impairment in Parkinson’s disease: Characterization and impact on quality of life according to subtype. Geriatr. Gerontol. Int. 2019, 19, 497–502. [Google Scholar] [CrossRef]
- Hoogland, J.; van Wanrooij, L.L.; Boel, J.A.; Goldman, J.G.; Stebbins, G.T.; Dalrymple-Alford, J.C.; Marras, C.; Adler, C.H.; Junque, C.; Pedersen, K.F.; et al. Detecting Mild Cognitive Deficits in Parkinson’s Disease: Comparison of Neuropsychological Tests. Mov. Disord. 2018, 33, 1750–1759. [Google Scholar] [CrossRef] [PubMed]
- Cammisuli, D.M.; Crowe, S. Spatial disorientation and executive dysfunction in elderly nondemented patients with Parkinson’s disease. Neuropsychiatr. Dis. Treat. 2018, 14, 2531–2539. [Google Scholar] [CrossRef] [PubMed]
- Lezak, M.D.; Howieson, D.B.; Loring, H.J.; Fisher, J.S. Neuropsychological Assessment; Oxford University Press: Oxford, UK, 2004. [Google Scholar]
- Gazzaniga, M.S.; Ivry, R.B.; Mangun, G.R.; Steven, M.S. Cognitive Neuroscience: The Biology of Mind; Norton: New York, NY, USA, 2006; pp. 87–90. [Google Scholar]
- Silveira, C.R.A.; Roy, E.A.; Almeida, Q.J. Acute effects of aerobic exercise on cognitive function in individuals with Parkinson’s disease. Neurosci. Lett. 2018, 671, 60–65. [Google Scholar] [CrossRef] [PubMed]
- Perez-Pardo, P.; Kliest, T.; Dodiya, H.B.; Broersen, L.M.; Garssen, J.; Keshavarzian, A. The gut-brain axis in Parkinson’s disease: Possibilities for food-based therapy. Eur. J. Pharmacol. 2017, 817, 85–95. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, H.; Piazza, B.R.; Forsyth, C.B.; Keshavarzian, A. Nutrition and gastrointestinal health as modulators of Parkinson’s disease. In Pharma-Nutrition. AAPS Advances in the Pharmaceutical Sciences Series; Folkerts, G., Garssen, J., Eds.; Springer International Publishing: New York, NY, USA, 2014; pp. 213–242. [Google Scholar]
- Martin, B.; Mattson, M.P.; Maudsley, S. Caloric restriction and intermittent fasting: Two potential diets for successful brain aging. Ageing Res. Rev. 2006, 5, 332–353. [Google Scholar] [CrossRef] [PubMed]
- Sutoo, D.; Akiyama, K. Regulation of brain function by exercise. Neurobiol. Dis. 2003, 13, 1–14. [Google Scholar] [CrossRef]
- Xu, Q.; Park, Y.; Huang, X.; Hollenbeck, A.; Blair, A.; Schatzkin, A.; Chen, H. Physical activities and future risk of Parkinson disease. Neurology 2010, 75, 341–348. [Google Scholar] [CrossRef]
- Alonso-Frech, F.; Sanahuja, J.J.; Rodriguez, A.M. Exercise and physical therapy in early management of parkinson disease. Neurologist 2011, 17, S47–S53. [Google Scholar] [CrossRef]
- Macpherson, H.; Teo, W.-P.; Schneider, L.A.; Smith, A.E. A Life-Long Approach to Physical Activity for Brain Health. Front. Aging Neurosci. 2017, 9, 147. [Google Scholar] [CrossRef]
- Logroscino, G.; Sesso, H.D.; Paffenbarger, R.S., Jr.; Lee, I.-M. Physical Activity and Risk of Parkinson’s Disease: A Prospective Cohort Study. J. Neurol. Neurosurg. Psychiatry 2006, 77, 1318–1322. [Google Scholar] [CrossRef]
- Wu, P.-L.; Lee, M.; Huang, T.-H. Effectiveness of physical activity on patients with depression and Parkinson’s disease: A systematic review. PLoS ONE 2017, 12, 2017. [Google Scholar] [CrossRef]
- Silva, F.C.; Iop, R.D.R.; de Oliveira, L.C.; Boll, A.M.; de Alvarenga, J.G.S.; Gutierres Filho, P.J.B.; de Melo, L.M.A.B.; Xavier, A.J.; da Silva, R. Effects of physical exercise programs on cognitive function in Parkinson’s disease patients: A systematic review of randomized controlled trials of the last 10 years. PLoS ONE 2018, 13, e0193113. [Google Scholar] [CrossRef] [PubMed]
- Johansson, H.; Hagströmer, M.; Grooten, W.J.A.; Franzén, E. Exercise-Induced Neuroplasticity in Parkinson’s Disease: A Metasynthesis of the Literature. Neural Plast. 2020, 2020, 8961493. [Google Scholar] [CrossRef] [PubMed]
- Wahid, A.; Manek, N.; Nichols, M.; Kelly, P.; Foster, C.; Webster, P.; Kaur, A.; Smith, C.F.; Wilkins, E.; Rayner, M. Quantifying the association between physical activity and cardiovascular disease and diabetes: A Systematic Review and Meta-Analysis. J. Am. Hear. Assoc. 2016, 5, e0002495. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.; Alkhawam, H.; Madenieh, R.; Shah, N.; Kosmas, C.E.; Vittorio, T.J. Aerobic vs anaerobic exercise training effects on the cardiovascular system. World J. Cardiol. 2017, 9, 134–138. [Google Scholar] [CrossRef] [PubMed]
- Cotman, C.W.; Bertchtols, N.C.; Christie, L.A. Exercise builds brain health: Key roles of growth factor cascades and inflammation. Trends Neurosci. 2007, 30, 464–472. [Google Scholar] [CrossRef]
- Chen, H.; Zhang, S.M.; Schwarzschild, M.A.; Hernan, M.A.; Ascherio, A. Physical activity and the risk of Parkinson disease. Neurology 2005, 64, 664–669. [Google Scholar] [CrossRef]
- Ahlskog, J.E. Aerobic exercise: Evidence for a direct brain effect to slow Parkinson disease progression. Mayo Clin. Proc. 2018, 93, 360–372. [Google Scholar] [CrossRef]
- Agnati, L.F.; Benfenati, F.; Solfrini, V.; Biagini, G.; Fuxe, K.; Guidolin, D.; Carani, C.; Zini, I. Brain aging and neuronal plasticity. Ann. N Y Acad. Sci. 1992, 673, 180–186. [Google Scholar] [CrossRef]
- Hirsh, M.A.; Farley, B.G. Exercise and neuroplasticity in persons living with Parkinson’s disease. Eur. H. Phy. Rehabil. Med. 2009, 45, 215–229. [Google Scholar]
- Wang, Z.; Myers, K.G.; Guo, Y.; Ocampo, M.A.; Pang, R.D.; Jakowec, M.W.; Holscheider, D.P. Functional reorganization of motor and limbic circuits after exercise training in a rat model of bilateral parkinsonism. PLoS ONE 2013, 8, e80058. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Guo, Y.; Myers, K.G.; Heints, R.; Peng, Y.-H.; Maarek, J.-M.I.; Holschneider, D.P. Exercise alters resting-state functional connectivity of motor circuits in parkinsonian rats. Neurobiol. Aging 2014, 36, 536–544. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Al-Jarrarh, M.; Jamous, M.; Al Zailaey, K.; Bweir, S.O. Endurance exercise training promotes angiogenesis in the brain of chronic/progressive mouse model of Parkinson’s disease. NeuroRehabilitation 2010, 26, 369–373. [Google Scholar] [CrossRef]
- Nadeau, A.; Pourcher, E.; Corbeil, P. Effects of 24wk of treadmill training on gait performance in Parkinson’s disease. Med. Sci. Sport. Exerc. 2014, 46, 645–655. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, K.; Quadros, A.C., Jr.; Santos, R.F.; Stella, F.; Gobbi, L.T.; Gobbi, S. Benefits of physical exercise on executive functions in older people with Parkinson’s disease. Brain Cogn. 2009, 69, 435–441. [Google Scholar] [CrossRef] [PubMed]
- Cruise, K.E.; Buck, R.S.; Loftus, A.M.; Newton, R.U.; Pegoraro, R.; Thomas, M.G. Exercise and Parkinson’s: Benefits for cognition and quality of life. Acta Neurol. Scand. 2020, 123, 13–19. [Google Scholar] [CrossRef]
- Reuter, I.; Mehenert, S.; Leone, P.; Kaps, M.; Oechsner, M.; Engelhardt, M. Effects of a flexibility and relaxation programme, walking and Nordic walking on Parkinson’s disease. J. Agin. Res. 2011, 232473, 1–18. [Google Scholar] [CrossRef]
- Ebersbach, G.; Eberschbach, A.; Gandor, F.; Wegner, B.; Wissel, J.; Kupsch, A. Impact of physical exercise on reaction time in patients with Parkinson’s disease—data from the Berlin BIG study. Arch. Phys. Med. Rehabil. 2014, 95, 996–999. [Google Scholar] [CrossRef]
- Cagusi, L.; Solla, P.; Serpe, R.; Carzedda, T.; Piras, L.; Onaggianu, M.; Silviaa, G.; Andread, D.B.; Marcoe, B.; Antoninoc, C.; et al. Effect of a nordic walking program on motor and non-motor symptoms, functional performance and body composition in patients with Parkinson’s disease. NeuroRehabilitation 2015, 37, 245–254. [Google Scholar] [CrossRef]
- Duchesne, C.; Longu, O.; Nadeau, A.; Robillard, M.E.; Boré, A.; Bobeuf, F.; Lafontainef, A.L.; Gheyseng, F.; Bhererah, L.; Doyonabc, J. Enhancing both motor and cognitive functioning in Parkinson’s disease: Aerobic exercise as a rehabilitative intervention. Brain Cogn. 2015, 99, 68–77. [Google Scholar] [CrossRef]
- Picelli, A.; Varalta, V.; Melotti, C.; Zatenalo, V.; Fonte, C.; Amato, S.; Saltuari, L.; Santamato, A.; Fiore, P.; Smania, N. Effects of treadmill training on cognitive and motor features of patients with mild to moderate Parkinson’s disease: A pilot single-blind, randomized controlled trial. Funct. Neurol. 2016, 31, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Koop, M.K.; Rosenfeldt, A.B.; Alberts, J.L. Mobility improves after high intensity aerobic exercise in individuals with Parkinson’s disease. J. Neurol. Sci. 2019, 15, 187–193. [Google Scholar] [CrossRef] [PubMed]
- Bello, O.; Sanchez, J.A.; Lopez-Alonso, V.; Márquez, G.; Morenilla, L.; Castro, X.; Giraldezb, M.; Santos-Garcíac, D.; Fernandez-del-Olmob, M. The effects of treadmill or overground walking training program on gait in Parkinson’s disease. Gait Posture 2013, 38, 590–595. [Google Scholar] [CrossRef] [PubMed]
- Palasz, E.; Niewiadomski, W.; Gasiorowska, A.; Wysocka, A.; Stepniewska, A.; Niewiadomska, G. Exercise-Induced Neuroprotection and Recovery of Motor Function in Animal Models of Parkinson’s disease. Front. Neurol. 2019, 10, 1143. [Google Scholar] [CrossRef]
- Speelma, A.D.; van de Warrenbur, B.P.; van Nimwegen, M.; Petzinger, G.M.; Munneke, M.; Bloem, B.R. How might physical activity benefit patients with Parkinson disease? Nat. Rev. Neurol. 2011, 7, 528–534. [Google Scholar] [CrossRef]
- Dutra, M.F.; Jaege, M.; Ilha, J.; Kalil-Gaspa, P.I.; Marcuzz, S.; Achava, M. Exercise improves motor deficits and alters striatal GFAP expression in a 6-OHDA-induced rat model of Parkinson’s disease. Neurol. Sci. 2012, 33, 1137–1144. [Google Scholar] [CrossRef]
- Larsen, J.O.; Skalicky, M.; Viidik, A. Does long-term physical exercise counteract age-related Purkinje cell loss? A stereological study of rat cerebellum. J. Comp. Neurol. 2000, 428, 213–222. [Google Scholar] [CrossRef]
- Olaseinde, T.; Oyeleye, S.I.; Ognunsuyi, O.B.; Ogunruku, O. Functional foods in the management of neurodegenerative disease. In Functional Foods: Unloking the Medicine in Food; Oboh, G., Ed.; Graceland Prints: Memphis, TN, USA, 2017; pp. 72–81. [Google Scholar]
- De Lau, L.M.L.; Bornebroek, M.; Witteman, J.M.C.; Hofman, A.; Koudstaal, P.J.; Breteler, M.M.B. Dietary fatty acids and the risk of Parkinson disease: The Rotterdam study. Neurology 2005, 64, 2040–2045. [Google Scholar] [CrossRef]
- De Lau, L.M.L.; Koudstaad, P.J.; Witteman, J.C.M.; Hofman, A.; Breteler, M.M.B. Dietary folate, vitamin B12, and vitamin B6 and the risk of Parkinson disease. Neurology 2006, 67, 315–318. [Google Scholar] [CrossRef]
- Logroscino, G.; Gao, X.; Chen, H.; Wing, A.L.; Ascherio, A. Dietary iron intake and risk of Parkinson’s disease. Am. J. Epidemiol. 2008, 168, 1381–1388. [Google Scholar] [CrossRef]
- Ross, G.W.; Abbott, R.D.; Petrovitch, H.; Morens, D.M.; Grandinetti, A.; Tung, K.-H.; Tanner, C.M.; Masaki, K.H.; Blanchette, P.L.; Curb, J.D.; et al. Association of coffee and caffeine intake with the risk of Parkinson disease. JAMA 2000, 283, 2674–2679. [Google Scholar] [CrossRef] [PubMed]
- Park, M.; Ross, G.W.; Petrovich, H.; White, L.R.; Masaki, K.H.; Nelson, J.S.; Tanner, C.M.; Curb, J.D.; Blanchette, P.L.; Abbott, R.D. Consumption of milk and calcium in midlife and future risk of Parkinson disease. Neurology 2005, 64, 1047–1051. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Chen, H.; Fung, T.T.; Logroscino, G.; Schwarzshild, M.A.; Hu, F.B.; Ascherio, A. Prospective study of dietary pattern and risk of Parkinson disease. Am. J. Clin. Nutr. 2007, 86, 1486–1492. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Cassidy, A.; Schwarzshild, M.A.; Rimm, E.B.; Ascherio, A. Habitual intake of dietary flavonoids and risk of Parkinson disease. Neurology 2012, 78, 1138–1145. [Google Scholar] [CrossRef]
- Ciulla, M.; Marinelli, L.; Cacciatore, I.; Di Stefano, A. Role of dietary Supplements in the Management of Parkinson’s disease. Biomolecules 2019, 9, 271. [Google Scholar] [CrossRef]
- Martucci, M.; Ostan, R.; Biondi, F.; Bellavista, E.; Fabbri, C.; Bertarelli, C.; Salvioli, S.; Capri, M.; Franceschi, C.; Santoro, A. Mediterranean Diet and Inflammaging Within the Hormesis Paradigm. Nutr. Rev. 2017, 75, 442–455. [Google Scholar] [CrossRef]
- Makki, K.; Deehan, E.C.; Walter, J.; Bäckhed, F. The impact of dietary fiber on gut microbiota in host health and disease. Cell Host Microbe 2018, 23, 705–715. [Google Scholar] [CrossRef]
- Alcalay, R.N.; Gu, Y.; Mejia-Santana, H.; Cote, L.; Marder, K.S.; Scarmeas, N. The Association between Mediterranean Diet Adherence and Parkinson’s disease. Mov. Disord. 2012, 27, 771–774. [Google Scholar] [CrossRef]
- Lau, Y.S.; Patki, G.; Das-Panja, K.; Le, W.D.; Ahmad, S.O. Neuroprotective effects and mechanisms of exercise in a chronic mouse model od Parkinson’s disease with moderate neurodegeneration. Eur. J. Neurosci. 2011, 33, 1264–1274. [Google Scholar] [CrossRef]
- Colcombe, S.J.; Erickson, K.I.; Scalf, P.E.; Kim, J.S.; Prakash, R.; McAuley, E. Aerobic Exercise Training Increases Brain Volume in Aging Humans. J. Gerontol. A Biol. Sci. Med. Sci. 2006, 61, 1166–1170. [Google Scholar] [CrossRef]
- Kramer, A.; Hahn, S.; Cohen, N.J.; Banich, M.T.; McAuley, E.; Harrison, C.R.; Chason, J.; Vakil, E.; Bardell, L.; Boileau, R.A.; et al. Ageing, fitness and neurocognitive function. Nature 1999, 400, 418–419. [Google Scholar] [CrossRef] [PubMed]
- Paillard, T.; Rolland, Y.; de Souto Barreto, P. Protective effects of physical exercise in Alzheimer’s disease and Parkinson’s disease: A narrative review. J. Clin. Neurol. 2015, 11, 212–219. [Google Scholar] [CrossRef] [PubMed]
- Duchesne, C.; Gheysen, F.; Boré, A.; Albouy, G.; Nadeau, A.; Robillard, M.E.; Bobeuf, F.; Lafontaine, A.L.; Lungu, O.; Bherer, L.; et al. Influence of aerobic exercise training on the neural correlates of motor learning in Parkinson’s disease. NeuroImage: Clinical 2016, 12, 559–569. [Google Scholar] [CrossRef] [PubMed]
- Agim, Z.S.; Cannon, J.R. Dietary factors in the etiology of Parkinson’s disease. Biomed Res. Int. 2015, 2015, 672838. [Google Scholar] [CrossRef]
- Jackson, A.; Forsyth, B.; Shaikh, M.; Voigt, R.M.; Engen, P.A.; Ramirez, V.; Keshavarzian, A. Diet in Parkinson’s Disease: Critical Role for the Microbiome. Front. Neurol. 2019, 10, 1245. [Google Scholar] [CrossRef] [PubMed]
- Maraki, M.I.; Yannakoulia, M.; Stamelou, M.; Stefanis, L.; Xiromerisiou, G.; Kosmidis, M.H.; Dardiotis, E.; Hadjigeorgiou, G.M.; Sakka, P.; Anastasiou, C.A.; et al. Mediterranean diet adherence is related to reduced probability of prodromal Parkinson’s disease. Mov. Disord. 2019, 34, 48–57. [Google Scholar] [CrossRef]
- Leri, M.; Scuto, M.; Ontario, M.L.; Calabrese, V.; Calabrese, E.J.; Bucciantini, M.; Stefani, M. Healthy Effects of Plant Polyphenols: Molecular Mechanisms. Int. J. Mol. Sci. 2020, 21, 1250. [Google Scholar] [CrossRef]
- Schapira, H.V.; Chaudhuri, R.; Jenner, P. Non-motor features of Parkinson’s disease. Nat. Rev. Neurosci. 2017, 18, 435–450. [Google Scholar] [CrossRef]
- Cammisuli, D.M.; Ceravolo, R.; Bonuccelli, U. Non-pharmacological interventions for Parkinson’s disease-mild cognitive impairment: Future directions for research. Neural Regen. Res. 2020, 9, 1650–1651. [Google Scholar] [CrossRef]
- Schrag, A.; Selai, C.; Jahanshahi, M.; Quinn, N.P. The EQ-5D--a generic quality of life measure-is a useful instrument to measure quality of life in patients with Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry 2000, 69, 67–73. [Google Scholar] [CrossRef]
- Isaacson, S.H. Effective Treatment Strategies for Motor and Nonmotor Symptoms of Parkinson’s disease. J. Clin. Psychiatry 2020, 81. [Google Scholar] [CrossRef] [PubMed]
- Nocera, J.R.; Altmann, L.J.P.; Sapienza, C.; Okun, M.S.; Hass, C.J. Can exercise improve language and cognition in Parkinson’s disease? A case report. Neurocase 2010, 16, 301–306. [Google Scholar] [CrossRef] [PubMed]
- Seid, S.E.; Santiago, J.A.; Bilyk, H.; Potashkin, J.A. The emerging role of nutrition in Parkinson’s disease. Front. Aging Neurosci. 2014, 6, 36. [Google Scholar]
- Daniele, S.; Giacomelli, C.; Martini, C. Brain ageing and neurodegenerative disease: The role of cellular waste management. Biochem. Pharmacol. 2018, 158, 207–216. [Google Scholar] [CrossRef]
- Uc, E.Y.; Doerschug, K.C.; Magnotta, V.; Dawson, J.D.; Thomsen, T.R.; Kline, J.N.; Rizzo, M.; Newman, S.R.; Mehta, S.; Grabowski, T.J.; et al. Phase I/II randomized trial of aerobic exercise in Parkinson disease in a community setting. Neurology 2014, 83, 413–425. [Google Scholar] [CrossRef]
- Van der Kolk, N.; de vries, N.M.; Kessels, R.P.C.; Joosten, H.; Aeilko, H.Z.; Post, B.; Bloem, B.R. Effectiveness of home-based and remotely supervised aerobic exercise in Parkinson’s disease: A double-blind, randomized trial. The Lancet Neurology 2019, 11, 998–1008. [Google Scholar] [CrossRef]
- Van Nimwegen, M.; Speelman, A.D.; Hofman-van Rossum, E.J.M.; Overeem, S.; Deeg, D.J.H.; Borm, G.F.; van der Horst, M.H.L.; Bloem, B.R.; Munneke, M. Physical inactivity in Parkinson’s disease. J. Neuology 2011, 258, 2214–2221. [Google Scholar] [CrossRef]
- Cassani, E.; Barichella, M.; Ferri, V.; Pinelli, G.; Ioro, L.; Bolliri, C.; Caronnia, S.; Faiermana, S.A.; Mottolesea, A.; Pusania, C.; et al. Dietary habits in Parkinson’s disease: Adherence to Mediterranean diet. Parkinsonism Relat. Disord. 2017, 42, 40–46. [Google Scholar] [CrossRef]
- Pluck, G.C.; Brown, R.G. Apathy in Parkinson’s Disease. J. Neurol. Neurosurg. Psychiatry 2002, 73, 636–642. [Google Scholar] [CrossRef]
- Gundogdu, I.; Ozturk, E.A.; Kocer, B.; Nazli, F.; Aypac, C.; Comoglu, S.; Cakci, A. Musculoskeletal pain and deformities in Parkinson’s disease. Int. J. Clin. Exp. Med. 2016, 9, 14663–14669. [Google Scholar]
- Marek, K.; Chowdhury, S.; Siderowf, A.; Lasch, S.; Coffey, C.S.; Caspell-Garcia, C.; Simuni, T.; Jennings, D.; Tanner, C.M.; Trojanowski, J.Q.; et al. The Parkinson’s progression markers initiative (PPMI)—establishing a PD biomarker cohort. Ann. Clin. Transl. Neurol. 2018, 5, 1460–1477. [Google Scholar] [CrossRef] [PubMed]
- Polinski, N.K.; Volpicelli-Daley, L.A.; Sortwell, C.E.; Luk, K.C.; Cremades, N.; Gottler, L.M.; Froula, J.; Duffy, M.F.; Lee, V.M.Y.; Martinez, T.N.; et al. Best Practices for Generating and Using Alpha-Synuclein Pre-Formed Fibrils to Model Parkinson’s Disease in Rodents. J. Parkinsons Dis. 2018, 8, 303–322. [Google Scholar] [CrossRef] [PubMed]

| Neuroprotective Agents | Specific Effects | Nonspecific Effects |
|---|---|---|
| AE | Psychomotor learning abilities Protection of dopaminergic neurons | Ameliorating executive functions/attentional control |
| Balanced diet | Antioxidant Anti-inflammatory Neuromodulation Neuroprotection | Global enhancement of cognitive domains efficiency |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cammisuli, D.M.; Bonuccelli, U.; Daniele, S.; Martini, C.; Fusi, J.; Franzoni, F. Aerobic Exercise and Healthy Nutrition as Neuroprotective Agents for Brain Health in Patients with Parkinson’s Disease: A Critical Review of the Literature. Antioxidants 2020, 9, 380. https://doi.org/10.3390/antiox9050380
Cammisuli DM, Bonuccelli U, Daniele S, Martini C, Fusi J, Franzoni F. Aerobic Exercise and Healthy Nutrition as Neuroprotective Agents for Brain Health in Patients with Parkinson’s Disease: A Critical Review of the Literature. Antioxidants. 2020; 9(5):380. https://doi.org/10.3390/antiox9050380
Chicago/Turabian StyleCammisuli, Davide Maria, Ubaldo Bonuccelli, Simona Daniele, Claudia Martini, Jonathan Fusi, and Ferdinando Franzoni. 2020. "Aerobic Exercise and Healthy Nutrition as Neuroprotective Agents for Brain Health in Patients with Parkinson’s Disease: A Critical Review of the Literature" Antioxidants 9, no. 5: 380. https://doi.org/10.3390/antiox9050380
APA StyleCammisuli, D. M., Bonuccelli, U., Daniele, S., Martini, C., Fusi, J., & Franzoni, F. (2020). Aerobic Exercise and Healthy Nutrition as Neuroprotective Agents for Brain Health in Patients with Parkinson’s Disease: A Critical Review of the Literature. Antioxidants, 9(5), 380. https://doi.org/10.3390/antiox9050380









