Phytochemical Profile and Antioxidant Capacity of Coffee Plant Organs Compared to Green and Roasted Coffee Beans
Abstract
1. Introduction
2. Materials and Methods
2.1. Seeds
2.2. Plant Material and Growing Conditions
2.3. Sample Preparation
2.3.1. Coffee Seeds
2.3.2. Leaves
2.3.3. Stems
- (1)
- The herbaceous stem (HS), consisting of the green parts of the main stem and the green branches.
- (2)
- The woody stem (WS), consisting of the ligneous brown part of the stem above the root collar.
2.3.4. Roots
2.4. Extraction and Analysis
2.4.1. Chlorophylls and Carotenoids Determination
2.4.2. Sample Processing for Phytochemical and Antioxidant Capacity Determination
2.4.3. Sample Extraction
2.4.4. Alkaloids and Phenolic Compounds Determination
2.4.5. Sucrose Extraction and Determination
2.4.6. Total Phenolic Content (TPC) Determination
2.4.7. Antioxidant Capacity Determination
2,2–Diphenyl–1–picryl hydrazyl (DPPH) Radical Assay
2,2–Azino bis (3–ethyl benzothiazoline–6–sulphonic acid) Radical (ABTS) Assay
Ferric Ion Reducing Antioxidant Power (FRAP) Assay
2.5. Statistical Analysis
3. Results
3.1. HPLC Phytochemical Composition
3.1.1. Coffee Beans
3.1.2. Leaves
3.1.3. Stems
3.1.4. Roots
3.2. Total Phenolic Content and Antioxidant Capacities of the Coffee Plant Organs
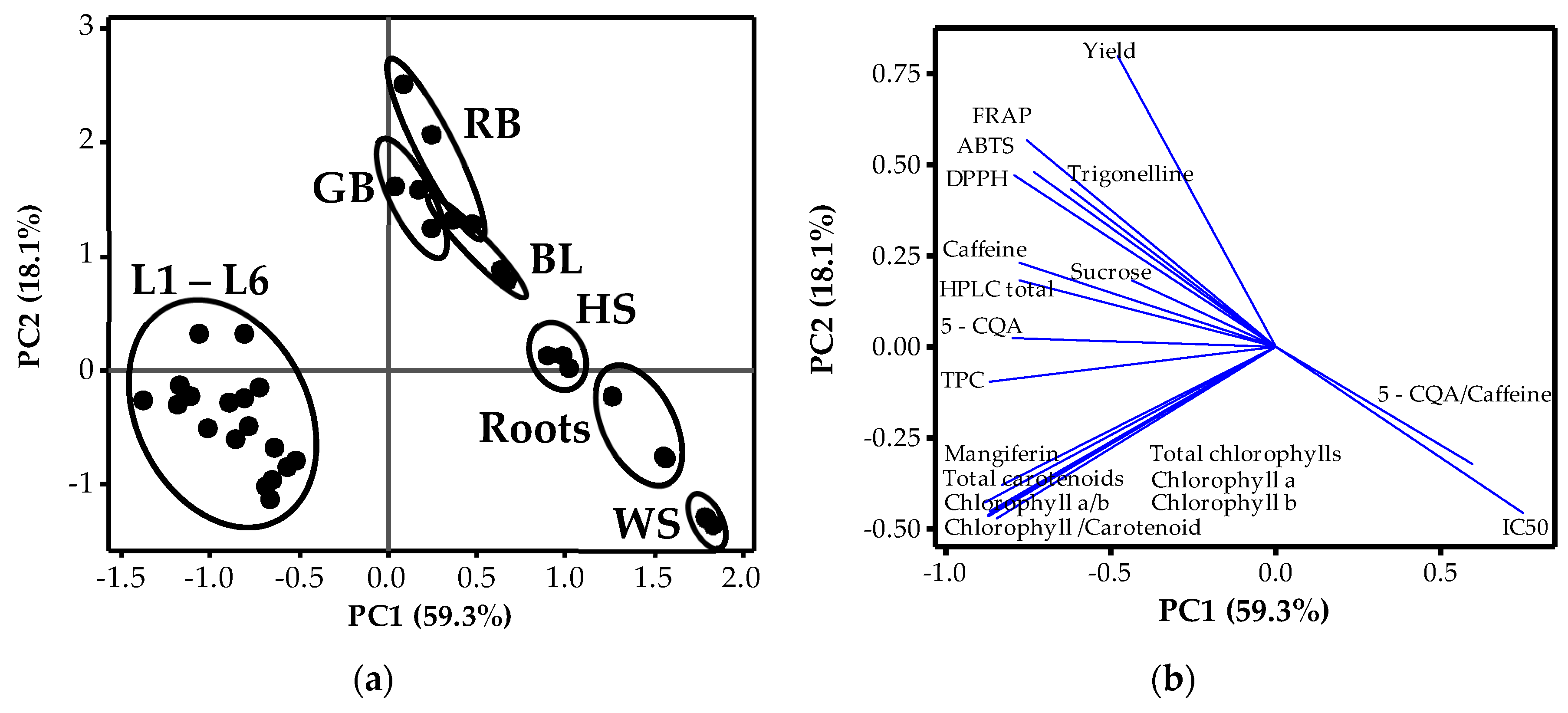
3.3. Relationships amongst Phytochemicals and Plant Organs
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- International Coffee Organization (ICO). Historical Data on the Global Coffee Trade. Available online: http://www.ico.org/new_historical.asp (accessed on 17 January 2019).
- Farah, A.; Donangelo, C.M. Phenolic compounds in coffee. Braz. J. Plant Physiol. 2006, 18, 23–36. [Google Scholar] [CrossRef]
- Koshiro, Y.; Jackson, M.C.; Katahira, R.; Wang, M.L.; Nagai, C.; Ashihara, H. Biosynthesis of chlorogenic acids in growing and ripening fruits of Coffea arabica and Coffea canephora plants. Z. Nat. Sect. C J. Biosci. 2007, 62, 731–742. [Google Scholar] [CrossRef]
- Koshiro, Y.; Zheng, X.Q.; Wang, M.L.; Nagai, C.; Ashihara, H. Changes in content and biosynthetic activity of caffeine and trigonelline during growth and ripening of Coffea arabica and Coffea canephora fruits. Plant Sci. 2006, 171, 242–250. [Google Scholar] [CrossRef]
- Farah, A.; Monteiro, M.; Donangelo, C.M.; Lafay, S. Chlorogenic acids from green coffee extract are highly bioavailable in humans. J. Nutr. 2008, 138, 2309–2315. [Google Scholar] [CrossRef] [PubMed]
- Patay, É.B.; Bencsik, T.; Papp, N. Phytochemical overview and medicinal importance of Coffea species from the past until now. Asian Pac. J. Trop. Med. 2016, 9, 1127–1135. [Google Scholar] [CrossRef] [PubMed]
- De Melo Pereira, G.V.; de Carvalho Neto, D.P.; Júnior, A.I.M.; Vásquez, Z.S.; Medeiros, A.B.; Vandenberghe, L.P.; Soccol, C.R. Exploring the impacts of postharvest processing on the aroma formation of coffee beans—A review. Food Chem. 2019, 272, 441–452. [Google Scholar] [CrossRef] [PubMed]
- Poisson, L.; Auzanneau, N.; Mestdagh, F.; Blank, I.; Davidek, T. New Insight into the role of sucrose in the generation of α-diketones upon coffee roasting. J. Agric. Food Chem. 2018, 66, 2422–2431. [Google Scholar] [CrossRef]
- Amrani-Hemaimi, M.; Cerny, C.; Fay, L.B. Mechanisms of formation of alkylpyrazines in the maillard reaction. J. Agric. Food Chem. 1995, 43, 2818–2822. [Google Scholar] [CrossRef]
- Stadler, R.H.; Varga, N.; Hau, J.; Arce Vera, F.; Welti, D.H. Alkylpyridiniums. 1. Formation in model systems via thermal degradation of trigonelline. J. Agric. Food Chem. 2002, 50, 1192–1199. [Google Scholar] [CrossRef]
- Fuller, M.; Rao, N.Z. The effect of time, roasting temperature, and grind size on caffeine and chlorogenic acid concentrations in cold brew coffee. Sci. Rep. 2017, 7, 1–9. [Google Scholar] [CrossRef]
- Ky, C.L.; Louarn, J.; Dussert, S.; Guyot, B.; Hamon, S.; Noirot, M. Caffeine, trigonelline, chlorogenic acids and sucrose diversity in wild Coffea arabica L. and C. canephora P. accessions. Food Chem. 2001, 75, 223–230. [Google Scholar] [CrossRef]
- Talamond, P.; Mondolot, L.; Gargadennec, A.; de Kochko, A.; Hamon, S.; Fruchier, A.; Campa, C. First report on mangiferin (C-glucosyl-xanthone) isolated from leaves of a wild coffee plant, Coffea pseudozanguebariae (Rubiaceae). Acta Bot. Gall. 2008, 155, 513–519. [Google Scholar] [CrossRef]
- Campa, C.; Mondolot, L.; Rakotondravao, A.; Bidel, L.P.; Gargadennec, A.; Couturon, E.; La Fisca, P.; Rakotomalala, J.J.; Jay-Allemand, C.; Davis, A.P. A survey of mangiferin and hydroxycinnamic acid ester accumulation in coffee (Coffea) leaves: Biological implications and uses. Ann. Bot. 2012, 110, 595–613. [Google Scholar] [CrossRef] [PubMed]
- Imran, M.; Arshad, M.S.; Butt, M.S.; Kwon, J.H.; Arshad, M.U.; Sultan, M.T. Mangiferin: A natural miracle bioactive compound against lifestyle related disorders. Lipids Health Dis. 2017, 16, 84. [Google Scholar] [CrossRef]
- Chen, X.M.; Ma, Z.; Kitts, D.D. Effects of processing method and age of leaves on phytochemical profiles and bioactivity of coffee leaves. Food Chem. 2018, 249, 143–153. [Google Scholar] [CrossRef]
- Stahl, W.; Sies, H. Antioxidant activity of carotenoids. Mol. Asp. Med. 2003, 24, 345–351. [Google Scholar] [CrossRef]
- Lanfer-Marquez, U.M.; Barros, R.M.C.; Sinnecker, P. Antioxidant activity of chlorophylls and their derivatives. Food Res. Int. 2005, 38, 885–891. [Google Scholar] [CrossRef]
- Wangcharoen, W.; Phimphilai, S. Chlorophyll and total phenolic contents, antioxidant activities and consumer acceptance test of processed grass drinks. J. Food Sci. Technol. 2016, 53, 4135–4140. [Google Scholar] [CrossRef]
- Alvarez-Jubete, L.; Wijngaard, H.; Arendt, E.K.; Gallagher, E. Polyphenol composition and in vitro antioxidant activity of amaranth, quinoa buckwheat and wheat as affected by sprouting and baking. Food Chem. 2010, 119, 770–778. [Google Scholar] [CrossRef]
- Lindenmeier, M.; Hofmann, T. Influence of baking conditions and precursor supplementation on the amounts of the antioxidant pronyl-l-lysine in bakery products. J. Agric. Food Chem. 2004, 52, 350–354. [Google Scholar] [CrossRef]
- Aerts, R.J.; Baumann, T.W. Distribution and utilization of chlorogenic acid in Coffea seedlings. J. Exp. Bot. 1994, 45, 497–503. [Google Scholar] [CrossRef]
- Zheng, X.Q.; Ashihara, H. Distribution, biosynthesis and function of purine and pyridine alkaloids in Coffea arabica seedlings. Plant Sci. 2004, 166, 807–813. [Google Scholar] [CrossRef]
- Gebreselassie, W.; Mohammed, A.; Netsere, A. Pre-sowing treatment of coffee (Coffea arabica L.) seeds to enhance emergence and subsequent growth of seedlings. Res. J. Seed Sci. 2010, 3, 218–226. [Google Scholar] [CrossRef]
- Hoagland, D.C.; Arnon, D.I. The water culture method for growing plant without soil. Circ. Calif. Agric. Exp. Stn. 1950, 347, 32. [Google Scholar]
- Porra, R.; Thompson, W.A.; Kriedemann, P.E. Determination of accurate extinction coefficients and simultaneous equations for assaying chlorophylls a and b extracted with four different solvents: Verification of the concentration of chlorophyll standards by atomic absorption spectroscopy. Biochim. Biophys. Acta 1989, 975, 384–394. [Google Scholar] [CrossRef]
- Lichtenthaler, H.K.; Buschmann, C. Chlorophylls and carotenoids: Measurement and characterization by UV-VIS spectroscopy. Curr. Protoc. Food Analyical Chem. 2001, 1, F4.3.1–F4.3.8. [Google Scholar] [CrossRef]
- Chen, X.M.; Tait, A.R.; Kitts, D.D. Flavonoid composition of orange peel and its association with antioxidant and anti-inflammatory activities. Food Chem. 2017, 218, 15–21. [Google Scholar] [CrossRef]
- Xu, F.; Tan, X.; Wang, Z. Effects of sucrose on germination and seedling development of Brassica napus. Int. J. Biol. 2010, 2, 150–154. [Google Scholar] [CrossRef]
- Mazzafera, P.; Robinson, S.P. Characterization of polyphenol oxidase in coffee. Phytochemistry 2000, 55, 285–296. [Google Scholar] [CrossRef]
- Montavon, P.; Duruz, E.; Rumo, G.; Pratz, G. Evolution of green coffee protein profiles with maturation and relationship to coffee cup quality. J. Agric. Food Chem. 2003, 51, 2328–2334. [Google Scholar] [CrossRef]
- De Maria, C.A.B.; Trugo, L.C.; Moreira, R.F.A.; Werneck, C.C. Composition of green coffee fractions and their contribution to the volatile profile formed during roasting. Food Chem. 1994, 50, 141–145. [Google Scholar] [CrossRef]
- Sakiyama, N.S.; Picoli, E.A.D.T.; Oliveira, A.C.B.D.; Caixeta, E.T.; Zambolim, L.; Martinez, H.E.P.; Pereira, A.A. Triple plagiotropic branch in coffee: A new promising mutant? Crop Breed. Appl. Biotechnol. 2017, 17, 408–411. [Google Scholar] [CrossRef]
- Kamble, P.N.; Giri, S.P.; Mane, R.S.; Tiwana, A. Estimation of chlorophyll content in young and adult leaves of some selected plants. Univers. J. Environ. Res. Technol. 2015, 5, 306–310. [Google Scholar] [CrossRef]
- Fiedor, J.; Burda, K. Potential role of carotenoids as antioxidants in human health and disease. Nutrients 2014, 6, 466–488. [Google Scholar] [CrossRef]
- Mittler, R. Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci. 2002, 7, 405–410. [Google Scholar] [CrossRef]
- Asada, K. The water-water cycle in chloroplasts: Scavenging of active oxygens and dissipation of excess photons. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1999, 50, 601–639. [Google Scholar] [CrossRef]
- Van Den Ende, W.; Valluru, R. Sucrose, sucrosyl oligosaccharides, and oxidative stress: Scavenging and salvaging? J. Exp. Bot. 2009, 60, 9–18. [Google Scholar] [CrossRef]
- Brewer, M.S. Natural antioxidants: Sources, compounds, mechanisms of action, and potential applications. Compr. Rev. Food Sci. Food Saf. 2011, 10, 221–247. [Google Scholar] [CrossRef]
- Das, K.; Roychoudhury, A. Reactive oxygen species (ROS) and response of antioxidants as ROS-scavengers during environmental stress in plants. Front. Environ. Sci. 2014, 2, 53. [Google Scholar] [CrossRef]
- Ramalho, J.C.; Rodrigues, A.P.; Lidon, F.C.; Marques, L.M.; Leitão, A.E.; Fortunato, A.S.; Pais, I.P.; Silva, M.J.; Scotti-Campos, P.; Lopes, A.; et al. Stress cross-response of the antioxidative system promoted by superimposed drought and cold conditions in Coffea spp. PLoS ONE 2018, 13, e0198694. [Google Scholar] [CrossRef]
- Lunn, J.E. Sucrose metabolism. In Encyclopedia of Life Sciences; Wiley: Basingstoke, UK, 2016; pp. 1–9. [Google Scholar] [CrossRef]
- Hammond, J.P.; White, P.J. Sucrose transport in the phloem: Integrating root responses to phosphorus starvation. J. Exp. Bot. 2008, 59, 93–109. [Google Scholar] [CrossRef] [PubMed]
- Franklin, G.; Dias, A.C.P. Chlorogenic acid participates in the regulation of shoot, root and root hair development in Hypericum perforatum. Plant Physiol. Biochem. 2011, 49, 835–842. [Google Scholar] [CrossRef] [PubMed]
- Sreekumar, S.; Soniya, E.V. Artificial micrornas promote high-level production of biomolecules through metabolic engineering of phenylpropanoid pathway. CRC Crit. Rev. Plant Sci. 2017, 36, 353–366. [Google Scholar] [CrossRef]
- Van Moerkercke, A.; Schauvinhold, I.; Pichersky, E.; Haring, M.A.; Schuurink, R.C. A plant thiolase involved in benzoic acid biosynthesis and volatile benzenoid production. Plant J. 2009, 60, 292–302. [Google Scholar] [CrossRef]
- Shimizu, M.M.; Mazzafera, P. A role for trigonelline during imbibition and germination of coffee seeds. Plant Biol. 2000, 2, 605–611. [Google Scholar] [CrossRef]
- Ashihara, H.; Watanabe, S. Accumulation and function of trigonelline in non-leguminous plants. Nat. Prod. Commun. 2014, 9, 795–798. [Google Scholar] [CrossRef]
- Heaton, J.W.; Marangoni, A.G. Chlorophyll degradation in processed foods and senescent plant tissues. Trends Food Sci. Technol. 1996, 7, 8–15. [Google Scholar] [CrossRef]
- Harborne, J.B.; Williams, C.A. Advances in flavonoid research since 1992. Phytochemistry 2000, 55, 481–504. [Google Scholar] [CrossRef]
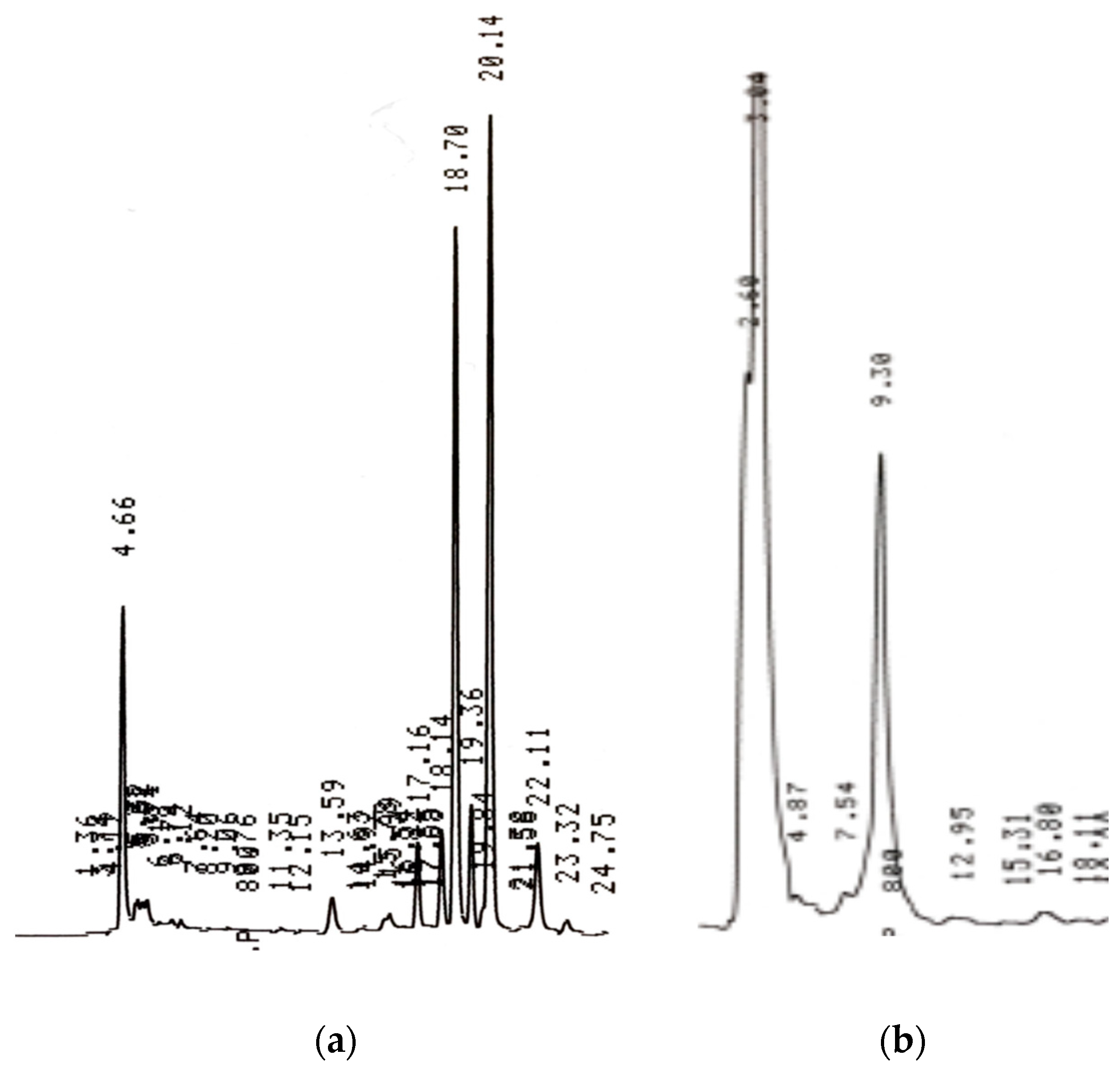
| Time (min) | Methanol (100%) | Acetic Acid (2%) |
|---|---|---|
| 0 | 15 | 85 |
| 4 | 15 | 85 |
| 10 | 35 | 65 |
| 22 | 43 | 57 |
| 24 | 60 | 40 |
| 28 | 60 | 40 |
| 30 | 15 | 85 |
| 40 | 15 | 85 |
| Sample (Plant Organ) | Caffeine | Trigonelline | 5–CQA | Mangiferin | Sucrose | 5–CQA/Caffeine | HPLC Total | |
|---|---|---|---|---|---|---|---|---|
| Seeds | GB | 0.97 ± 0.09 b,c | 0.65 ± 0.05 a,b,c | 3.13 ± 0.33 a,b,c | 0.00 ± 0.00 c | 4.95 ± 0.40 a | 3.23 ± 0.03 c | 9.70 ± 0.73 a |
| RB | 1.30 ± 0.13 a,b | 0.85 ± 0.01 a | 1.00 ± 0.02 e | 0.00 ± 0.00 c | 0.14 ± 0.05 g | 0.77 ± 0.07 c | 3.29 ± 0.20 g,h | |
| Leaves | L1 | 0.87 ± 0.03 c | 0.67 ± 0.22 a,b,c | 3.97 ± 0.61 a | 0.09 ± 0.03 a | 2.63 ± 0.55 b | 4.58 ± 0.85 c | 8.23 ± 0.18 b |
| L2 | 0.78 ± 0.13 c | 0.72 ± 0.26 a,b | 3.35 ± 0.37 a,b | 0.05 ± 0.01b | 2.30 ± 0.16 b,c | 4.46 ± 1.26 c | 7.19 ± 0.15 b,c | |
| L3 | 0.90 ± 0.37 c | 0.68 ± 0.20 a,b,c | 3.21 ± 0.24 a,b,c | 0.05 ± 0.01b | 2.35 ± 0.34 b | 4.15 ± 2.10 c | 7.19 ± 0.43 b,c | |
| L4 | 1.35 ± 0.07 a | 0.76 ± 0.12 a,b | 2.83 ± 0.23 b,c | 0.05 ± 0.00b | 1.95 ± 0.08 b,c,d | 2.10 ± 0.06 c | 6.93 ± 0.20 c,d | |
| L5 | 1.12 ± 0.00 a,b,c | 0.44 ± 0.03 b,c,d | 2.35 ± 0.11 c,d | 0.04 ± 0.01b | 1.85 ± 0.16 b,c,d | 2.09 ± 0.09 c | 5.81 ± 0.06 d,e | |
| L6 | 1.05 ± 0.01 a,b,c | 0.47 ± 0.01 b,c,d | 2.48 ± 0.30 b,c,d | 0.06 ± 0.02 a,b | 1.51 ± 0.12 c,d,e | 2.36 ± 0.28 c | 5.57 ± 0.46 e,f | |
| BL | 0.32 ± 0.00 d | 0.34 ± 0.04 c,d | 1.55 ± 0.32 d,e | 0.00 ± 0.00 c | 0.82 ± 0.12 e,f,g | 5.44 ± 0.67 c | 3.00 ± 0.42 h,i | |
| Stem | HS | 0.01 ± 0.00 d | 0.45 ± 0.04 b,c,d | 2.67 ± 0.29 b,c | 0.00 ± 0.00 c | 1.24 ± 0.35 d,e,f | 190.2 ± 58.3 b | 4.37 ± 0.60 f,g |
| WS | 0.00 ± 0.00 d | 0.20 ± 0.08 d | 0.66 ± 0.23 e | 0.00 ± 0.00 c | 0.57 ± 0.11 f,g | 369.4 ± 193.2 a | 1.42 ± 0.32 j | |
| Roots | Roots | 0.00 ± 0.00 d | 0.14 ± 0.00 d | 0.71 ± 0.44 e | 0.00 ± 0.00 c | 1.01 ± 0.30 e,f | 0.00 ± 0.00 c | 1.85 ± 0.69 i,j |
| Leaf Position | Chlorophyll a | Chlorophyll b | Total Chlorophyll | Total Carotenoids | Chlorophyll a/b | Chlorophyll/Carotenoid |
|---|---|---|---|---|---|---|
| L1 | 1.09 ± 0.28 a | 0.37 ± 0.09 b | 1.46 ± 0.36 a | 0.26 ± 0.06 a | 2.93 ± 0.20 a,b | 5.48 ± 0.27 a |
| L2 | 1.02 ± 0.06 a | 0.35 ± 0.05 b | 1.37 ± 0.09 a | 0.24 ± 0.02 a | 2.90 ± 0.26 a,b | 5.70 ± 0.02 a |
| L3 | 1.31 ± 0.12 a | 0.48 ± 0.01 a,b | 1.79 ± 0.13 a | 0.31 ± 0.04 a | 2.73 ± 0.20 a,b | 5.84 ± 0.38 a |
| L4 | 1.28 ± 0.17 a | 0.52 ± 0.03 a | 1.80 ± 0.19 a | 0.30 ± 0.04 a | 2.45 ± 0.24 b | 5.96 ± 0.22 a |
| L5 | 1.27 ± 0.05 a | 0.40 ± 0.07 a,b | 1.67 ± 0.11 a | 0.37 ± 0.09 a | 3.20 ± 0.48 a | 4.72 ± 1.23 a |
| L6 | 1.08 ± 0.02 a | 0.36 ± 0.01 b | 1.44 ± 0.02 a | 0.28 ± 0.01 a | 3.02 ± 0.09 a,b | 5.06 ± 0.14 a |
| Sample (Plant Organ) | Yield (%) | TPC Content (mg g−1 GAE) | DPPH IC50 (µg. Sample mL−1) | DPPH | ABTS | FRAP | |
|---|---|---|---|---|---|---|---|
| TEAC (µmol Trolox g−1 Sample) | |||||||
| Seeds | GB | 25.8 ± 4.40 a,b | 29.3 ± 1.40 c | 146.8 ± 12.0 c | 199.7 ± 16.1 a,b,c | 220.7 ± 22.4 a,b | 974.2 ± 83.4 a,b |
| RB | 36.4 ± 11.9 a | 35.4 ± 1.49 b,c | 87.0 ± 5.73 c | 337.0 ± 22.2 a | 325.5 ± 81.0 a | 1104.4 ± 323.3 a | |
| Leaves | L1 | 26.6 ± 0.23 a,b | 65.1 ± 14.1 a | 90.6 ± 12.1 c | 328.0 ± 43.9 a | 345.7 ± 53.3 a | 1097.4 ± 132.8 a |
| L2 | 24.2 ± 1.54 b | 66.7 ± 19.0 a | 94.0 ± 38.5 c | 302.5 ± 141.7 a | 323.8 ± 72.1 a | 1016.6 ± 271.2 a | |
| L3 | 23.1 ± 0.53 b | 63.6 ± 10.2 a | 119.0 ± 34.7 c | 268.3 ± 78.3 a,b | 306.1 ± 92.3 a | 1024.2 ± 229.2 a | |
| L4 | 22.5 ± 2.96 b | 66.4 ± 3.95 a | 115.3 ± 15.8 c | 258.2 ± 35.4 a,b | 267.0 ± 48.4 a | 969.1 ± 173.8 a,b | |
| L5 | 18.1 ± 2.89 b,c | 72.0 ± 15.6 a | 125.5 ± 10.1 c | 228.7 ± 29.1 a,b | 238.9 ± 8.12 a,b | 618.6 ± 247.5 a,b,c | |
| L6 | 18.9 ± 0.94 b | 71.5 ± 2.47 a | 132.6 ± 15.7 c | 223.4 ± 26.5 a,b | 208.8 ± 15.7 a,b | 624.1 ± 104.1 a,b,c | |
| BL | 25.6 ± 3.04 a,b | 60.4 ± 8.71 a,b | 99.8 ± 22.6 c | 304.6 ± 79.0 a | 294.5 ± 17.7 a | 755.1 ± 156.4 a,b | |
| Stem | HS | 16.9 ± 0.55 b,c | 27.8 ± 2.57 c | 263.8 ± 10.5 c | 110.9 ± 4.42 b,c,d | 124.8 ± 1.42 b,c | 469.7 ± 13.0 b,c |
| WS | 6.4 ± 0.27 c | 8.7 ± 1.17 c | 1403.1 ± 406.0 a | 21.9 ± 5.59 d | 24.9 ± 7.44 c | 110.0 ± 34.0 c | |
| Roots | Root | 14.6 ± 3.16 b,c | 15.8 ± 3.43 c | 916.1 ± 370.4 b | 36.8 ± 18.6 c,d | 41.5 ± 21.2 c | 156.7 ± 85.6 c |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Acidri, R.; Sawai, Y.; Sugimoto, Y.; Handa, T.; Sasagawa, D.; Masunaga, T.; Yamamoto, S.; Nishihara, E. Phytochemical Profile and Antioxidant Capacity of Coffee Plant Organs Compared to Green and Roasted Coffee Beans. Antioxidants 2020, 9, 93. https://doi.org/10.3390/antiox9020093
Acidri R, Sawai Y, Sugimoto Y, Handa T, Sasagawa D, Masunaga T, Yamamoto S, Nishihara E. Phytochemical Profile and Antioxidant Capacity of Coffee Plant Organs Compared to Green and Roasted Coffee Beans. Antioxidants. 2020; 9(2):93. https://doi.org/10.3390/antiox9020093
Chicago/Turabian StyleAcidri, Robert, Yumiko Sawai, Yuko Sugimoto, Takuo Handa, Daisuke Sasagawa, Tsugiyaki Masunaga, Sadahiro Yamamoto, and Eiji Nishihara. 2020. "Phytochemical Profile and Antioxidant Capacity of Coffee Plant Organs Compared to Green and Roasted Coffee Beans" Antioxidants 9, no. 2: 93. https://doi.org/10.3390/antiox9020093
APA StyleAcidri, R., Sawai, Y., Sugimoto, Y., Handa, T., Sasagawa, D., Masunaga, T., Yamamoto, S., & Nishihara, E. (2020). Phytochemical Profile and Antioxidant Capacity of Coffee Plant Organs Compared to Green and Roasted Coffee Beans. Antioxidants, 9(2), 93. https://doi.org/10.3390/antiox9020093





