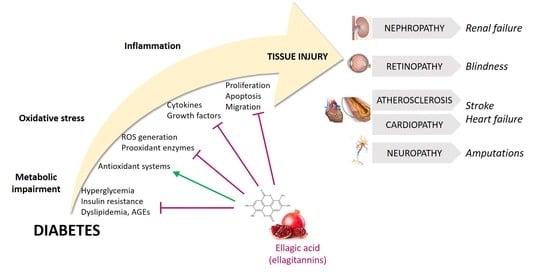
Ellagic Acid as a Tool to Limit the Diabetes Burden: Updated Evidence
Abstract
1. Introduction
2. Ellagic Acid and Glucose Control
2.1. In Vitro Studies
2.2. Animal Studies
2.2.1. Non-Diabetic Murine Models
2.2.2. Streptozotocin-Induced Diabetic Murine Models
2.2.3. Alloxan-Induced Diabetic Murine Models
2.2.4. Other Diabetic Murine Models
2.3. Human Studies
3. Anti-Inflammatory, Antioxidant, and Anti-Glycation Properties of Ellagic Acid
3.1. Ellagic Acid and Inflammation
3.1.1. In Vitro Studies
3.1.2. Animal Studies
3.1.3. Human Studies
3.2. Ellagic Acid and Oxidative Stress
3.2.1. In Vitro Studies
3.2.2. Animal Studies
3.2.3. Human Studies
3.3. Ellagic Acid and Glycation
3.3.1. In Vitro Studies
3.3.2. Animal Studies
3.3.3. Human Studies
4. Ellagic Acid and Complications of Diabetes
4.1. Microvascular Complications
4.1.1. Diabetic Kidney Disease
4.1.2. Retinopathy
4.2. Macrovascular Complications
4.2.1. Atherosclerosis
4.2.2. Cardiopathy
4.3. Neurologic-Related Complications of Diabetes
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Saeedi, P.; Petersohn, I.; Salpea, P.; Malanda, B.; Karuranga, S.; Unwin, N.; Colagiuri, S.; Guariguata, L.; Motala, A.A.; Ogurtsova, K.; et al. Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045, Results from the International Diabetes Federation Diabetes Atlas, 9th edition. Diabetes Res. Clin. Pract. 2019, 157. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, S.; Khunti, K.; Davies, M.J. Type 2 diabetes. Lancet 2017, 389, 2239–2251. [Google Scholar] [CrossRef]
- Buse, J.B.; Wexler, D.J.; Tsapas, A.; Rossing, P.; Mingrone, G.; Mathieu, C.; D’Alessio, D.A.; Davies, M.J. 2019 update to: Management of hyperglycaemia in type 2 diabetes, 2018. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetologia 2020, 63, 221–228. [Google Scholar] [CrossRef] [PubMed]
- Evert, A.B.; Dennison, M.; Gardner, C.D.; Garvey, W.T.; Lau, K.H.; MacLeod, J.; Mitri, J.; Pereira, R.F.; Rawlings, K.; Robinson, S.; et al. Nutrition Therapy for Adults With Diabetes or Prediabetes: A Consensus Report. Diabetes Care 2019, 42, 731–754. [Google Scholar] [CrossRef]
- Maritim, A.C.; Sanders, R.A.; Watkins, J.B. Diabetes, oxidative stress, and antioxidants: A review. J. Biochem. Mol. Toxicol. 2003, 17, 24–38. [Google Scholar] [CrossRef]
- Giacco, F.; Brownlee, M. Oxidative stress and diabetic complications. Circ. Res. 2010, 107, 1058–1070. [Google Scholar] [CrossRef]
- Yuan, T.; Yang, T.; Chen, H.; Fu, D.; Hu, Y.; Wang, J.; Yuan, Q.; Yu, H.; Xu, W.; Xie, X. New insights into oxidative stress and inflammation during diabetes mellitus-accelerated atherosclerosis. Redox Biol. 2019, 20, 247–260. [Google Scholar] [CrossRef]
- Del Bo, C.; Bernardi, S.; Marino, M.; Porrini, M.; Tucci, M.; Guglielmetti, S.; Cherubini, A.; Carrieri, B.; Kirkup, B.; Kroon, P.; et al. Systematic Review on Polyphenol Intake and Health Outcomes: Is there Sufficient Evidence to Define a Health-Promoting Polyphenol-Rich Dietary Pattern? Nutrients 2019, 11, 1355. [Google Scholar] [CrossRef]
- Laddha, A.P.; Kulkarni, Y.A. Tannins and vascular complications of Diabetes: An update. Phytomedicine 2019, 56, 229–245. [Google Scholar] [CrossRef]
- Sánchez-González, C.; Ciudad, C.J.; Noé, V.; Izquierdo-Pulido, M. Health benefits of walnut polyphenols: An exploration beyond their lipid profile. Crit. Rev. Food Sci. Nutr. 2017, 57, 3373–3383. [Google Scholar] [CrossRef]
- García-Conesa, M.T.; Chambers, K.; Combet, E.; Pinto, P.; Garcia-Aloy, M.; Andrés-Lacueva, C.; Pascual-Teresa, D.; Mena, P.; Konic Ristic, A.; Hollands, W.J.; et al. Meta-Analysis of the Effects of Foods and Derived Products Containing Ellagitannins and Anthocyanins on Cardiometabolic Biomarkers: Analysis of Factors Influencing Variability of the Individual Responses. Int. J. Mol. Sci. 2018, 19, 694. [Google Scholar] [CrossRef] [PubMed]
- Ríos, J.-L.; Giner, R.; Marín, M.; Recio, M. A Pharmacological Update of Ellagic Acid. Planta Med. 2018, 84, 1068–1093. [Google Scholar] [CrossRef] [PubMed]
- Landete, J.M. Ellagitannins, ellagic acid and their derived metabolites: A review about source, metabolism, functions and health. Food Res. Int. 2011, 44, 1150–1160. [Google Scholar] [CrossRef]
- García-Niño, W.R.; Zazueta, C. Ellagic acid: Pharmacological activities and molecular mechanisms involved in liver protection. Pharm. Res. 2015, 97, 84–103. [Google Scholar] [CrossRef]
- Tomás-Barberán, F.A.; González-Sarrías, A.; García-Villalba, R.; Núñez-Sánchez, M.A.; Selma, M.V.; García-Conesa, M.T.; Espín, J.C. Urolithins, the rescue of “old” metabolites to understand a “new” concept: Metabotypes as a nexus among phenolic metabolism, microbiota dysbiosis, and host health status. Mol. Nutr. Food Res. 2017, 61, 1500901. [Google Scholar] [CrossRef]
- Selma, M.V.; González-Sarrías, A.; Salas-Salvadó, J.; Andrés-Lacueva, C.; Alasalvar, C.; Örem, A.; Tomás-Barberán, F.A.; Espín, J.C. The gut microbiota metabolism of pomegranate or walnut ellagitannins yields two urolithin-metabotypes that correlate with cardiometabolic risk biomarkers: Comparison between normoweight, overweight-obesity and metabolic syndrome. Clin. Nutr. 2018, 37, 897–905. [Google Scholar] [CrossRef]
- Romo-Vaquero, M.; Cortés-Martín, A.; Loria-Kohen, V.; Ramírez-de-Molina, A.; García-Mantrana, I.; Collado, M.C.; Espín, J.C.; Selma, M.V. Deciphering the Human Gut Microbiome of Urolithin Metabotypes: Association with Enterotypes and Potential Cardiometabolic Health Implications. Mol. Nutr. Food Res. 2019, 63, 1800958. [Google Scholar] [CrossRef]
- Nakanishi, M.; Chen, Y.; Qendro, V.; Miyamoto, S.; Weinstock, E.; Weinstock, G.M.; Rosenberg, D.W. Effects of Walnut Consumption on Colon Carcinogenesis and Microbial Community Structure. Cancer Prev. Res. (Phila) 2016, 9, 692–703. [Google Scholar] [CrossRef]
- Zhao, W.; Shi, F.; Guo, Z.; Zhao, J.; Song, X.; Yang, H. Metabolite of ellagitannins, urolithin A induces autophagy and inhibits metastasis in human sw620 colorectal cancer cells. Mol. Carcinog. 2018, 57, 193–200. [Google Scholar] [CrossRef]
- Stanisławska, I.J.; Piwowarski, J.P.; Granica, S.; Kiss, A.K. The effects of urolithins on the response of prostate cancer cells to non-steroidal antiandrogen bicalutamide. Phytomedicine 2018, 46, 176–183. [Google Scholar] [CrossRef]
- Ding, X.; Jian, T.; Wu, Y.; Zuo, Y.; Li, J.; Lv, H.; Ma, L.; Ren, B.; Zhao, L.; Li, W.; et al. Ellagic acid ameliorates oxidative stress and insulin resistance in high glucose-treated HepG2 cells via miR-223/keap1-Nrf2 pathway. Biomed. Pharm. 2019, 110, 85–94. [Google Scholar] [CrossRef] [PubMed]
- Fatima, N.; Hafizur, R.M.; Hameed, A.; Ahmed, S.; Nisar, M.; Kabir, N. Ellagic acid in Emblica officinalis exerts anti-diabetic activity through the action on β-cells of pancreas. Eur. J. Nutr. 2017, 56, 591–601. [Google Scholar] [CrossRef] [PubMed]
- Rahimifard, M.; Moini-Nodeh, S.; Niaz, K.; Baeeri, M.; Jamalifar, H.; Abdollahi, M. Improvement of the functionality of pancreatic Langerhans islets via reduction of bacterial contamination and apoptosis using phenolic compounds. Iran. J. Basic Med. Sci. 2018, 21, 920–927. [Google Scholar] [CrossRef] [PubMed]
- Les, F.; Arbonés-Mainar, J.M.; Valero, M.S.; López, V. Pomegranate polyphenols and urolithin A inhibit α-glucosidase, dipeptidyl peptidase-4, lipase, triglyceride accumulation and adipogenesis related genes in 3T3-L1 adipocyte-like cells. J. Ethnopharmacol. 2018, 220, 67–74. [Google Scholar] [CrossRef]
- Li, Y.; Wen, S.; Kota, B.P.; Peng, G.; Li, G.Q.; Yamahara, J.; Roufogalis, B.D. Punica granatum flower extract, a potent alpha-glucosidase inhibitor, improves postprandial hyperglycemia in Zucker diabetic fatty rats. J. Ethnopharmacol. 2005, 99, 239–244. [Google Scholar] [CrossRef]
- Kam, A.; Li, K.M.; Razmovski-Naumovski, V.; Nammi, S.; Shi, J.; Chan, K.; Li, G.Q. A Comparative Study on the Inhibitory Effects of Different Parts and Chemical Constituents of Pomegranate on α-Amylase and α-Glucosidase. Phyther. Res. 2013, 27, 1614–1620. [Google Scholar] [CrossRef]
- Wu, S.; Tian, L. A new flavone glucoside together with known ellagitannins and flavones with anti-diabetic and anti-obesity activities from the flowers of pomegranate (Punica granatum). Nat. Prod. Res. 2019, 33, 252–257. [Google Scholar] [CrossRef]
- Bellesia, A.; Verzelloni, E.; Tagliazucchi, D. Pomegranate ellagitannins inhibit α-glucosidase activity in vitro and reduce starch digestibility under simulated gastro-intestinal conditions. Int. J. Food Sci. Nutr. 2015, 66, 85–92. [Google Scholar] [CrossRef]
- Yin, P.; Yang, L.; Xue, Q.; Yu, M.; Yao, F.; Sun, L.; Liu, Y. Identification and inhibitory activities of ellagic acid- and kaempferol-derivatives from Mongolian oak cups against α-glucosidase, α-amylase and protein glycation linked to type II diabetes and its complications and their influence on HepG2 cells’ viability. Arab. J. Chem. 2018, 11, 1247–1259. [Google Scholar] [CrossRef]
- Mohanty, I.R.; Borde, M.; Kumar, C.S.; Maheshwari, U. Dipeptidyl peptidase IV Inhibitory activity of Terminalia arjuna attributes to its cardioprotective effects in experimental diabetes: In silico, in vitro and in vivo analyses. Phytomedicine 2019, 57, 158–165. [Google Scholar] [CrossRef]
- Makino-Wakagi, Y.; Yoshimura, Y.; Uzawa, Y.; Zaima, N.; Moriyama, T.; Kawamura, Y. Ellagic acid in pomegranate suppresses resistin secretion by a novel regulatory mechanism involving the degradation of intracellular resistin protein in adipocytes. Biochem. Biophys. Res. Commun. 2012, 417, 880–885. [Google Scholar] [CrossRef] [PubMed]
- Toney, A.M.; Fan, R.; Xian, Y.; Chaidez, V.; Ramer-Tait, A.E.; Chung, S. Urolithin A, a Gut Metabolite, Improves Insulin Sensitivity Through Augmentation of Mitochondrial Function and Biogenesis. Obesity 2019, 27, 612–620. [Google Scholar] [CrossRef] [PubMed]
- Parmar, H.S.; Kar, A. Medicinal Values of Fruit Peels from Citrus sinensis, Punica granatum, and Musa paradisiaca with Respect to Alterations in Tissue Lipid Peroxidation and Serum Concentration of Glucose, Insulin, and Thyroid Hormones. J. Med. Food 2008, 11, 376–381. [Google Scholar] [CrossRef] [PubMed]
- Harzallah, A.; Hammami, M.; Kępczyńska, M.A.; Hislop, D.C.; Arch, J.R.; Cawthorne, M.A.; Zaibi, M.S. Comparison of potential preventive effects of pomegranate flower, peel and seed oil on insulin resistance and inflammation in high-fat and high-sucrose diet-induced obesity mice model. Arch. Physiol. Biochem. 2016, 122, 75–87. [Google Scholar] [CrossRef]
- Neyrinck, A.M.; Van Hée, V.F.; Bindels, L.B.; De Backer, F.; Cani, P.D.; Delzenne, N.M. Polyphenol-rich extract of pomegranate peel alleviates tissue inflammation and hypercholesterolaemia in high-fat diet-induced obese mice: Potential implication of the gut microbiota. Br. J. Nutr. 2013, 109, 802–809. [Google Scholar] [CrossRef]
- Zilani, M.N.; Sultana, T.; Rahman, S.A.; Anisuzzman, M.; Islam, M.A.; Shilpi, J.A.; Hossain, M.G. Chemical composition and pharmacological activities of Pisum sativum. BMC Complement. Altern. Med. 2017, 17, 171. [Google Scholar] [CrossRef]
- Fotschki, B.; Juśkiewicz, J.; Jurgoński, A.; Kosmala, M.; Milala, J.; Zduńczyk, Z.; Markowski, J. Grinding levels of raspberry pomace affect intestinal microbial activity, lipid and glucose metabolism in Wistar rats. Food Res. Int. 2019, 120, 399–406. [Google Scholar] [CrossRef]
- Nankar, R.P.; Doble, M. Hybrid drug combination: Anti-diabetic treatment of type 2 diabetic Wistar rats with combination of ellagic acid and pioglitazone. Phytomedicine 2017, 37, 4–9. [Google Scholar] [CrossRef]
- Farbood, Y.; Rashno, M.; Ghaderi, S.; Khoshnam, S.E.; Sarkaki, A.; Rashidi, K.; Rashno, M.; Badavi, M. Ellagic acid protects against diabetes-associated behavioral deficits in rats: Possible involved mechanisms. Life Sci. 2019, 225, 8–19. [Google Scholar] [CrossRef]
- Das, A.K.; Mandal, S.C.; Banerjee, S.K.; Sinha, S.; Saha, B.P.; Pal, M. Studies on the hypoglycaemic activity of Punica granatum seed in streptozotocin induced diabetic rats. Phyther. Res. 2001, 15, 628–629. [Google Scholar] [CrossRef]
- Manna, K.; Mishra, S.; Saha, M.; Mahapatra, S.; Saha, C.; Yenge, G.; Gaikwad, N.; Pal, R.; Oulkar, D.; Banerjee, K.; et al. Amelioration of diabetic nephropathy using pomegranate peel extract-stabilized gold nanoparticles: Assessment of NF-κB and Nrf2 signaling system. Int. J. Nanomed. 2019, 14, 1753–1777. [Google Scholar] [CrossRef] [PubMed]
- Salwe, K.J.; Sachdev, D.O.; Bahurupi, Y.; Kumarappan, M. Evaluation of antidiabetic, hypolipedimic and antioxidant activity of hydroalcoholic extract of leaves and fruit peel of Punica granatum in male Wistar albino rats. J. Nat. Sci. Biol. Med. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Bagri, P.; Ali, M.; Aeri, V.; Bhowmik, M.; Sultana, S. Antidiabetic effect of Punica granatum flowers: Effect on hyperlipidemia, pancreatic cells lipid peroxidation and antioxidant enzymes in experimental diabetes. Food Chem. Toxicol. 2009, 47, 50–54. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.Y.; Zhu, C.; Qian, T.W.; Guo, H.; Wang, D.D.; Zhang, F.; Yin, X. Extracts of black bean peel and pomegranate peel ameliorate oxidative stress-induced hyperglycemia in mice. Exp. Ther. Med. 2015, 9, 43–48. [Google Scholar] [CrossRef]
- Yeh, Y.-T.; Chiang, A.-N.; Hsieh, S.-C. Chinese Olive (Canarium album L.) Fruit Extract Attenuates Metabolic Dysfunction in Diabetic Rats. Nutrients 2017, 9, 1123. [Google Scholar] [CrossRef]
- Taheri Rouhi, S.Z.; Sarker, M.M.R.; Rahmat, A.; Alkahtani, S.A.; Othman, F. The effect of pomegranate fresh juice versus pomegranate seed powder on metabolic indices, lipid profile, inflammatory biomarkers, and the histopathology of pancreatic islets of Langerhans in streptozotocin-nicotinamide induced type 2 diabetic Sprague-Dawley rats. BMC Complement. Altern. Med. 2017, 17, 156. [Google Scholar] [CrossRef]
- Aboonabi, A.; Rahmat, A.; Othman, F. Antioxidant effect of pomegranate against streptozotocin-nicotinamide generated oxidative stress induced diabetic rats. Toxicol. Rep. 2014, 1, 915–922. [Google Scholar] [CrossRef]
- El-Beih, N.M.; Ramadan, G.; El-Husseiny, E.A.; Hussein, A.M. Effects of pomegranate aril juice and its punicalagin on some key regulators of insulin resistance and oxidative liver injury in streptozotocin-nicotinamide type 2 diabetic rats. Mol. Biol. Rep. 2019, 46, 3701–3711. [Google Scholar] [CrossRef]
- Parmar, H.S.; Kar, A. Antidiabetic potential of Citrus sinensis and Punica granatum peel extracts in alloxan treated male mice. Biofactors 2007, 31, 17–24. [Google Scholar] [CrossRef]
- Middha, S.K.; Usha, T.; Pande, V. Pomegranate peel attenuates hyperglycemic effects of alloxan-induced diabetic rats. Excli J. 2014, 13, 223–224. [Google Scholar]
- El-Hadary, A.E.; Ramadan, M.F. Phenolic profiles, antihyperglycemic, antihyperlipidemic, and antioxidant properties of pomegranate (Punica granatum) peel extract. J. Food Biochem. 2019, 43, e12803. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Barman, S. Antidiabetic and antihyperlipidemic effects of ethanolic extract of leaves of Punica granatum in alloxan-induced non-insulin-dependent diabetes mellitus albino rats. Indian J. Pharm. 2012, 44, 219–224. [Google Scholar] [CrossRef]
- Gharib, E.; Montasser Kouhsari, S. Study of the Antidiabetic Activity of Punica granatum L. Fruits Aqueous Extract on the Alloxan-Diabetic Wistar Rats. Iran J. Pharm. Res. IJPR 2019, 18, 358–368. [Google Scholar] [PubMed]
- Raafat, K.; Samy, W. Amelioration of diabetes and painful diabetic neuropathy by Punica granatum L. Extract and its spray dried biopolymeric dispersions. Evid. -Based Complement. Altern. Med. 2014, 2014. [Google Scholar] [CrossRef]
- Abu-Gharbieh, E.; Shehab, N.G. Therapeutic potentials of Crataegus azarolus var. eu-azarolus Maire leaves and its isolated compounds. BMC Complement. Altern. Med. 2017, 17, 218. [Google Scholar] [CrossRef] [PubMed]
- Yin, P.; Wang, Y.; Yang, L.; Sui, J.; Liu, Y. Hypoglycemic Effects in Alloxan-Induced Diabetic Rats of the Phenolic Extract from Mongolian Oak Cups Enriched in Ellagic Acid, Kaempferol and Their Derivatives. Molecules 2018, 23, 1046. [Google Scholar] [CrossRef] [PubMed]
- Jelodar, G.; Mohsen, M.; Shahram, S. Effect of walnut leaf, coriander and pomegranate on blood glucose and histopathology of pancreas of alloxan induced diabetic rats. Afr. J. Tradit. Complement. Altern. Med. Ajtcam. 2007, 4, 299–305. [Google Scholar] [CrossRef]
- Amin, M.M.; Arbid, M.S. Estimation of ellagic acid and/or repaglinide effects on insulin signaling, oxidative stress, and inflammatory mediators of liver, pancreas, adipose tissue, and brain in insulin resistant/type 2 diabetic rats. Appl. Physiol. Nutr. Metab. 2017, 42, 181–192. [Google Scholar] [CrossRef]
- Yoshimura, Y.; Nishii, S.; Zaima, N.; Moriyama, T.; Kawamura, Y. Ellagic acid improves hepatic steatosis and serum lipid composition through reduction of serum resistin levels and transcriptional activation of hepatic ppara in obese, diabetic KK-A(y) mice. Biochem. Biophys. Res. Commun. 2013, 434, 486–491. [Google Scholar] [CrossRef]
- Polce, S.A.; Burke, C.; França, L.M.; Kramer, B.; de Andrade Paes, A.M.; Carrillo-Sepulveda, M.A. Ellagic Acid Alleviates Hepatic Oxidative Stress and Insulin Resistance in Diabetic Female Rats. Nutrients 2018, 10, 531. [Google Scholar] [CrossRef]
- Banihani, S.A.; Makahleh, S.M.; El-Akawi, Z.; Al-Fashtaki, R.A.; Khabour, O.F.; Gharibeh, M.Y.; Saadah, N.A.; Al-Hashimi, F.H.; Al-Khasieb, N.J. Fresh pomegranate juice ameliorates insulin resistance, enhances β-cell function, and decreases fasting serum glucose in type 2 diabetic patients. Nutr. Res. 2014, 34, 862–867. [Google Scholar] [CrossRef] [PubMed]
- Kerimi, A.; Nyambe-Silavwe, H.; Gauer, J.S.; Tomás-Barberán, F.A.; Williamson, G. Pomegranate juice, but not an extract, confers a lower glycemic response on a high–glycemic index food: Randomized, crossover, controlled trials in healthy subjects. Am. J. Clin. Nutr. 2017, 106, 1384–1393. [Google Scholar] [CrossRef] [PubMed]
- Kelishadi, R.; Gidding, S.S.; Hashemi, M.; Hashemipour, M.; Zakerameli, A.; Poursafa, P. Acute and long term effects of grape and pomegranate juice consumption on endothelial dysfunction in pediatric metabolic syndrome. J. Res. Med. Sci. 2011, 16, 245–253. [Google Scholar] [PubMed]
- Parsaeyan, N.; Mozaffari-Khosravi, H.; Mozayan, M.R. Effect of pomegranate juice on paraoxonase enzyme activity in patients with type 2 diabetes. J. Diabetes Metab. Disord. 2012, 11, 11. [Google Scholar] [CrossRef]
- Shishehbor, F.; Mohammad Shahi, M.; Zarei, M.; Saki, A.; Zakerkish, M.; Shirani, F. Effects of Concentrated Pomegranate Juice on Subclinical Inflammation and Cardiometabolic Risk Factors for Type 2 Diabetes: A Quasi-Experimental Study. Int. J. Endocrinol. Metab. 2016, 14, e33835. [Google Scholar] [CrossRef]
- Sumner, M.D.; Elliott-Eller, M.; Weidner, G.; Daubenmier, J.J.; Chew, M.H.; Marlin, R.; Raisin, C.J.; Ornish, D. Effects of Pomegranate Juice Consumption on Myocardial Perfusion in Patients With Coronary Heart Disease. Am. J. Cardiol. 2005, 96, 810–814. [Google Scholar] [CrossRef]
- Cerdá, B.; Soto, C.; Albaladejo, M.D.; Martinez, P.; Sanchez-Gascon, F.; Tomás-Barberán, F.; Espin, J.C. Pomegranate juice supplementation in chronic obstructive pulmonary disease: A 5-week randomized, double-blind, placebo-controlled trial. Eur. J. Clin. Nutr. 2006, 60, 245–253. [Google Scholar] [CrossRef]
- González-Ortiz, M.; Martínez-Abundis, E.; Espinel-Bermúdez, M.C.; Pérez-Rubio, K.G. Effect of pomegranate juice on insulin secretion and sensitivity in patients with obesity. Ann. Nutr. Metab. 2011, 58, 220–223. [Google Scholar] [CrossRef]
- Tsang, C.; Smail, N.F.; Almoosawi, S.; Davidson, I.; Al-Dujaili, E.A.S. Intake of polyphenol-rich pomegranate pure juice influences urinary glucocorticoids, blood pressure and homeostasis model assessment of insulin resistance in human volunteers. J. Nutr. Sci. 2012, 1, e9. [Google Scholar] [CrossRef]
- Sohrab, G.; Nasrollahzadeh, J.; Zand, H.; Amiri, Z.; Tohidi, M.; Kimiagar, M. Effects of pomegranate juice consumption on inflammatory markers in patients with type 2 diabetes: A randomized, placebo-controlled trial. J. Res. Med. Sci. 2014, 19, 215–220. [Google Scholar]
- Asgary, S.; Sahebkar, A.; Afshani, M.R.; Keshvari, M.; Haghjooyjavanmard, S.; Rafieian-Kopaei, M. Clinical Evaluation of Blood Pressure Lowering, Endothelial Function Improving, Hypolipidemic and Anti-Inflammatory Effects of Pomegranate Juice in Hypertensive Subjects. Phyther. Res. 2014, 28, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Park, J.E.; Kim, J.Y.; Kim, J.; Kim, Y.J.; Kim, M.J.; Kwon, S.W.; Kwon, O. Pomegranate vinegar beverage reduces visceral fat accumulation in association with AMPK activation in overweight women: A double-blind, randomized, and placebo-controlled trial. J. Funct. Foods 2014, 8, 274–281. [Google Scholar] [CrossRef]
- Fuster-Muñoz, E.; Roche, E.; Funes, L.; Martínez-Peinado, P.; Sempere, J.M.; Vicente-Salar, N. Effects of pomegranate juice in circulating parameters, cytokines, and oxidative stress markers in endurance-based athletes: A randomized controlled trial. Nutrition 2016, 32, 539–545. [Google Scholar] [CrossRef] [PubMed]
- Manthou, E.; Georgakouli, K.; Deli, C.K.; Sotiropoulos, A.; Fatouros, I.G.; Kouretas, D.; Haroutounian, S.; Matthaiou, C.; Koutedakis, Y.; Jamurtas, A.Z. Effect of pomegranate juice consumption on biochemical parameters and complete blood count. Exp. Ther. Med. 2017, 14, 1756–1762. [Google Scholar] [CrossRef] [PubMed]
- Sohrab, G.; Ebrahimof, S.; Sotoudeh, G.; Neyestani, T.R.; Angoorani, P.; Hedayati, M.; Siasi, F. Effects of pomegranate juice consumption on oxidative stress in patients with type 2 diabetes: A single-blind, randomized clinical trial. Int. J. Food Sci. Nutr. 2017, 68, 249–255. [Google Scholar] [CrossRef] [PubMed]
- Moazzen, H.; Alizadeh, M. Effects of Pomegranate Juice on Cardiovascular Risk Factors in Patients with Metabolic Syndrome: A Double-Blinded, Randomized Crossover Controlled Trial. Plant Foods Hum. Nutr. 2017, 72, 126–133. [Google Scholar] [CrossRef] [PubMed]
- Heber, D.; Seeram, N.P.; Wyatt, H.; Henning, S.M.; Zhang, Y.; Ogden, L.G.; Dreher, M.; Hill, J.O. Safety and Antioxidant Activity of a Pomegranate Ellagitannin-Enriched Polyphenol Dietary Supplement in Overweight Individuals with Increased Waist Size. J. Agric. Food Chem. 2007, 55, 10050–10054. [Google Scholar] [CrossRef]
- Hosseini, B.; Saedisomeolia, A.; Wood, L.G.; Yaseri, M.; Tavasoli, S. Effects of pomegranate extract supplementation on inflammation in overweight and obese individuals: A randomized controlled clinical trial. Complement Clin. Pr. 2016, 22, 44–50. [Google Scholar] [CrossRef]
- Pérez-Ramírez, I.F.; de Diego, E.H.; Riomoros-Arranz, M.; Reynoso-Camacho, R.; Saura-Calixto, F.; Pérez-Jiménez, J. Effects of acute intake of grape/pomegranate pomace dietary supplement on glucose metabolism and oxidative stress in adults with abdominal obesity. Int. J. Food Sci. Nutr. 2019, 1–12. [Google Scholar] [CrossRef]
- Tsalamandris, S.; Antonopoulos, A.S.; Oikonomou, E.; Papamikroulis, G.A.; Vogiatzi, G.; Papaioannou, S.; Deftereos, S.; Tousoulis, D. The role of inflammation in diabetes: Current concepts and future perspectives. Eur. Cardiol. Rev. 2019, 14, 50–59. [Google Scholar] [CrossRef]
- Oguntibeju, O.O. Type 2 diabetes mellitus, oxidative stress and inflammation: Examining the links. Int. J. Physiol. Pathophysiol. Pharmacol. 2019, 11, 45–63. [Google Scholar] [PubMed]
- Rozentsvit, A.; Vinokur, K.; Samuel, S.; Li, Y.; Gerdes, A.M.; Carrillo-Sepulveda, M.A. Ellagic Acid Reduces High Glucose-Induced Vascular Oxidative Stress Through ERK1/2/NOX4 Signaling Pathway. Cell Physiol. Biochem. 2017, 44, 1174–1187. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Zhao, Y.; Aisa, H.A. Anti-inflammatory effect of pomegranate flower in lipopolysaccharide (LPS)-stimulated RAW264.7 macrophages. Pharm. Biol. 2017, 55, 2095–2101. [Google Scholar] [CrossRef] [PubMed]
- Basu, A.; Newman, E.D.; Bryant, A.L.; Lyons, T.J.; Betts, N.M. Pomegranate Polyphenols Lower Lipid Peroxidation in Adults with Type 2 Diabetes but Have No Effects in Healthy Volunteers: A Pilot Study. J. Nutr. Metab. 2013, 2013, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Sohrab, G.; Nasrollahzadeh, J.; Tohidi, M.; Zand, H.; Nikpayam, O. Pomegranate Juice Increases Sirtuin1 Protein in Peripheral Blood Mononuclear Cell from Patients with Type 2 Diabetes: A Randomized Placebo Controlled Clinical Trial. Metab. Syndr. Relat. Disord. 2018, 16, 446–451. [Google Scholar] [CrossRef] [PubMed]
- Karasu, Ç.; Cumaoğlu, A.; Gúrpinar, A.; Kartal, M.; Kovacikova, L.; Milackova, I.; Stefek, M. Aldose reductase inhibitory activity and antioxidant capacity of pomegranate extracts. Interdiscip. Toxicol. 2012, 5, 15–20. [Google Scholar] [CrossRef]
- Rozenberg, O.; Howell, A.; Aviram, M. Pomegranate juice sugar fraction reduces macrophage oxidative state, whereas white grape juice sugar fraction increases it. Atherosclerosis 2006, 188, 68–76. [Google Scholar] [CrossRef]
- Gharib, E.; Montasser Kouhsari, S.; Izad MPunica granatum, L. Fruit Aqueous Extract Suppresses Reactive Oxygen Species-Mediated p53/p65/miR-145 Expressions followed by Elevated Levels of irs-1 in Alloxan-Diabetic Rats. Cell J. 2018, 19, 520–527. [Google Scholar] [CrossRef]
- Rock, W.; Rosenblat, M.; Miller-Lotan, R.; Levy, A.P.; Elias, M.; Aviram, M. Consumption of Wonderful Variety Pomegranate Juice and Extract by Diabetic Patients Increases Paraoxonase 1 Association with High-Density Lipoprotein and Stimulates Its Catalytic Activities. J. Agric. Food Chem. 2008, 56, 8704–8713. [Google Scholar] [CrossRef]
- Sohrab, G.; Angoorani, P.; Tohidi, M.; Tabibi, H.; Kimiagar, M.; Nasrollahzadeh, J. Pomegranate (Punicagranatum) juice decreases lipid peroxidation, but has no effect on plasma advanced glycated end-products in adults with type 2 diabetes: A randomized double-blind clinical trial. Food Nutr. Res. 2015, 59, 28551. [Google Scholar] [CrossRef]
- Rosenblat, M.; Hayek, T.; Aviram, M. Anti-oxidative effects of pomegranate juice (PJ) consumption by diabetic patients on serum and on macrophages. Atherosclerosis 2006, 187, 363–371. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Rubio, M.E.; Pérez-Jiménez, J.; Martínez-Bartolomé, M.Á.; Álvarez, I.; Saura-Calixto, F. Regular consumption of an antioxidant-rich juice improves oxidative status and causes metabolome changes in healthy adults. Plant Foods Hum. Nutr. 2015, 70, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Nowotny, K.; Jung, T.; Höhn, A.; Weber, D.; Grune, T. Advanced glycation end products and oxidative stress in type 2 diabetes mellitus. Biomolecules 2015, 5, 194–222. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Ma, H.; Frost, L.; Yuan, T.; Dain, J.A.; Seeram, N.P. Pomegranate phenolics inhibit formation of advanced glycation endproducts by scavenging reactive carbonyl species. Food Funct. 2014, 5, 2996–3004. [Google Scholar] [CrossRef] [PubMed]
- Kumagai, Y.; Nakatani, S.; Onodera, H.; Nagatomo, A.; Nishida, N.; Matsuura, Y.; Kobata, K.; Wada, M. Anti-Glycation Effects of Pomegranate (Punica granatum L.) Fruit Extract and Its Components in Vivo and in Vitro. J. Agric. Food Chem. 2015, 63, 7760–7764. [Google Scholar] [CrossRef] [PubMed]
- Dorsey, P.G.; Greenspan, P. Inhibition of nonenzymatic protein glycation by pomegranate and other fruit juices. J. Med. Food 2014, 17, 447–454. [Google Scholar] [CrossRef] [PubMed]
- Raghu, G.; Jakhotia, S.; Yadagiri Reddy, P.; Kumar, P.A.; Bhanuprakash Reddy, G. Ellagic acid inhibits non-enzymatic glycation and prevents proteinuria in diabetic rats. Food Funct. 2016, 7, 1574–1583. [Google Scholar] [CrossRef]
- Alicic, R.Z.; Rooney, M.T.; Tuttle, K.R. Diabetic kidney disease: Challenges, progress, and possibilities. Clin. J. Am. Soc. Nephrol. 2017, 12, 2032–2045. [Google Scholar] [CrossRef]
- Neuen, B.L.; Young, T.; Heerspink, H.J.; Neal, B.; Perkovic, V.; Billot, L.; Mahaffey, K.W.; Charytan, D.M.; Wheeler, D.C.; Arnott, C.; et al. SGLT2 inhibitors for the prevention of kidney failure in patients with type 2 diabetes: A systematic review and meta-analysis. Lancet Diabetes Endocrinol. 2019, 7, 845–854. [Google Scholar] [CrossRef]
- Bolignano, D.; Cernaro, V.; Gembillo, G.; Baggetta, R.; Buemi, M.; D’Arrigo, G. Antioxidant agents for delaying diabetic kidney disease progression: A systematic review and meta-analysis. PLoS ONE 2017, 12, e0178699. [Google Scholar] [CrossRef]
- Chao, C.; Mong, M.; Chan, K.; Yin, M. Anti-glycative and anti-inflammatory effects of caffeic acid and ellagic acid in kidney of diabetic mice. Mol. Nutr. Food Res. 2010, 54, 388–395. [Google Scholar] [CrossRef] [PubMed]
- Mohan, M.; Waghulde, H.; Kasture, S. Effect of pomegranate juice on Angiotensin II-induced hypertension in diabetic wistar rats. Phyther. Res. 2010, 24, S196–S203. [Google Scholar] [CrossRef] [PubMed]
- Ankita, P.; Deepti, B.; Nilam, M. Flavonoid rich fraction of Punica granatum improves early diabetic nephropathy by ameliorating proteinuria and disturbed glucose homeostasis in experimental animals. Pharm. Biol. 2015, 53, 61–71. [Google Scholar] [CrossRef] [PubMed]
- Zhou, B.; Li, Q.; Wang, J.; Chen, P.; Jiang, S. Ellagic acid attenuates streptozocin induced diabetic nephropathy via the regulation of oxidative stress and inflammatory signaling. Food Chem. Toxicol. 2019, 123, 16–27. [Google Scholar] [CrossRef] [PubMed]
- Ahad, A.; Ganai, A.A.; Mujeeb, M.; Siddiqui, W.A. Ellagic acid, an NF-κB inhibitor, ameliorates renal function in experimental diabetic nephropathy. Chem. Biol. Interact. 2014, 219, 64–75. [Google Scholar] [CrossRef] [PubMed]
- Solomon, S.D.; Chew, E.; Duh, E.J.; Sobrin, L.; Sun, J.K.; VanderBeek, B.L.; Wykoff, C.C.; Gardner, T.W. Diabetic retinopathy: A position statement by the American Diabetes Association. Diabetes Care 2017, 40, 412–418. [Google Scholar] [CrossRef]
- Díaz-López, A.; Babio, N.; Martínez-González, M.A.; Corella, D.; Amor, A.J.; Fitó, M.; Estruch, R.; Arós, F.; Gómez-Gracia, E.; Fiol, M.; et al. Mediterranean diet, retinopathy, nephropathy, and microvascular diabetes Complications: A post Hoc analysis of a randomized trial. Diabetes Care 2015, 38, 2134–2141. [Google Scholar] [CrossRef]
- Sala-Vila, A.; Díaz-López, A.; Valls-Pedret, C.; Cofán, M.; García-Layana, A.; Lamuela-Raventós, R.-M.; Castañer, O.; Zanon-Moreno, V.; Martinez-Gonzalez, M.A.; Toledo, E.; et al. Dietary marine ω-3 fatty acids and incident sight-threatening retinopathy in middle-aged and older individuals with type 2 diabetes: Prospective investigation from the PREDIMED trial. JAMA Ophthalmol. 2016, 134, 1142–1149. [Google Scholar] [CrossRef]
- Raghu, G.; Akileshwari, C.; Reddy, V.S.; Reddy, G.B. Attenuation of diabetic retinopathy in rats by ellagic acid through inhibition of AGE formation. J. Food Sci. Technol. 2017, 54, 2411–2421. [Google Scholar] [CrossRef]
- Tugcu, B.; Nacaroglu, S.A.; Gedikbasi, A.; Uhri, M.; Acar, N.; Ozdemir, H. Protective effect of pomegranate juice on retinal oxidative stress in streptozotocin-induced diabetic rats. Int. J. Ophthalmol. 2017, 10, 1662–1668. [Google Scholar] [CrossRef]
- Rawshani, A.; Rawshani, A.; Franzén, S.; Sattar, N.; Eliasson, B.; Svensson, A.M.; Zethelius, B.; Miftaraj, M.; McGuire, D.K.; Rosengren, A.; et al. Risk factors, mortality, and cardiovascular outcomes in patients with type 2 diabetes. N. Engl. J. Med. 2018, 379, 633–644. [Google Scholar] [CrossRef] [PubMed]
- Catalan, M.; Herreras, Z.; Pinyol, M.; Sala-Vila, A.; Amor, A.J.; de Groot, E.; Gilabert, R.; Ros, E.; Ortega, E. Prevalence by sex of preclinical carotid atherosclerosis in newly diagnosed type 2 diabetes. Nutr. Metab. Cardiovasc. Dis. Nmcd. 2015, 25, 742–748. [Google Scholar] [CrossRef] [PubMed]
- Amor, A.J.; Catalan, M.; Pérez, A.; Herreras, Z.; Pinyol, M.; Sala-Vila, A.; Cofán, M.; Gilabert, R.; Ros, E.; Ortega, E. Nuclear magnetic resonance lipoprotein abnormalities in newly-diagnosed type 2 diabetes and their association with preclinical carotid atherosclerosis. Atherosclerosis 2016, 247. [Google Scholar] [CrossRef] [PubMed]
- McHugh, K.; DeVore, A.D.; Wu, J.; Matsouaka, R.A.; Fonarow, G.C.; Heidenreich, P.A.; Yancy, C.W.; Green, J.B.; Altman, N.; Hernandez, A.F. Heart Failure With Preserved Ejection Fraction and Diabetes: JACC State-of-the-Art Review. J. Am. Coll. Cardiol. 2019, 73, 602–611. [Google Scholar] [CrossRef]
- Sattar, N.; Rawshani, A.; Franzén, S.; Rawshani, A.; Svensson, A.M.; Rosengren, A.; McGuire, D.K.; Eliasson, B.; Gudbjörnsdottir, S. Age at Diagnosis of Type 2 Diabetes Mellitus and Associations With Cardiovascular and Mortality Risks: Findings From the Swedish National Diabetes Registry. Circulation 2019, 139, 2228–2237. [Google Scholar] [CrossRef]
- Gæde, P.; Pedersen, O. Intensive integrated therapy of type 2 diabetes: Implications for long-term prognosis. Diabetes 2004, 53. [Google Scholar] [CrossRef]
- Kozakova, M.; Morizzo, C.; Goncalves, I.; Natali, A.; Nilsson, J.; Palombo, C. Cardiovascular organ damage in type 2 diabetes mellitus: The role of lipids and inflammation. Cardiovasc. Diabetol. 2019, 18, 61. [Google Scholar] [CrossRef]
- Aviram, M.; Volkova, N.; Coleman, R.; Dreher, M.; Reddy, M.K.; Ferreira, D.; Rosenblat, M. Pomegranate Phenolics from the Peels, Arils, and Flowers Are Antiatherogenic: Studies in Vivo in Atherosclerotic Apolipoprotein E-Deficient (E 0 ) Mice and in Vitro in Cultured Macrophages and Lipoproteins. J. Agric. Food Chem. 2008, 56, 1148–1157. [Google Scholar] [CrossRef]
- De Nigris, F.; Balestrieri, M.L.; Williams-Ignarro, S.; D’Armiento, F.P.; Fiorito, C.; Ignarro, L.J.; Napoli, C. The influence of pomegranate fruit extract in comparison to regular pomegranate juice and seed oil on nitric oxide and arterial function in obese Zucker rats. Nitric Oxide 2007, 17, 50–54. [Google Scholar] [CrossRef]
- Panchal, S.K.; Ward, L.; Brown, L. Ellagic acid attenuates high-carbohydrate, high-fat diet-induced metabolic syndrome in rats. Eur. J. Nutr. 2013, 52, 559–568. [Google Scholar] [CrossRef]
- Kesavan, R.; Ganugula, R.; Avaneesh, T.; Kumar, U.; Reddy, G.B.; Dixit, M. Ellagic acid inhibits PDGF-BB-induced vascular smooth muscle cell proliferation and prevents atheroma formation in streptozotocin-induced diabetic rats. J. Nutr. Biochem. 2013, 24, 1830–1839. [Google Scholar] [CrossRef]
- Chao, P.; Hsu, C.; Yin, M. Anti-inflammatory and anti-coagulatory activities of caffeic acid and ellagic acid in cardiac tissue of diabetic mice. Nutr. Metab. 2009, 6, 33. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.H.; Peng, G.; Kota, B.P.; Li, G.Q.; Yamahara, J.; Roufogalis, B.D.; Li, Y. Pomegranate flower improves cardiac lipid metabolism in a diabetic rat model: Role of lowering circulating lipids. Br. J. Pharm. 2005, 145, 767–774. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.H.; Yang, Q.; Harada, M.; Li, G.Q.; Yamahara, J.; Roufogalis, B.D.; Li, Y. Pomegranate flower extract diminishes cardiac fibrosis in Zucker diabetic fatty rats: Modulation of cardiac endothelin-1 and nuclear factor-kappaB pathways. J. Cardiovasc. Pharm. 2005, 46, 856–862. [Google Scholar] [CrossRef]
- Savi, M.; Bocchi, L.; Mena, P.; Dall’Asta, M.; Crozier, A.; Brighenti, F.; Stilli, D.; Del Rio, D. In vivo administration of urolithin A and B prevents the occurrence of cardiac dysfunction in streptozotocin-induced diabetic rats. Cardiovasc. Diabetol. 2017, 16, 80. [Google Scholar] [CrossRef]
- Namekata, I.; Hamaguchi, S.; Wakasugi, Y.; Ohhara, M.; Hirota, Y.; Tanaka, H. Ellagic acid and gingerol, activators of the sarco-endoplasmic reticulum Ca2+-ATPase, ameliorate diabetes mellitus-induced diastolic dysfunction in isolated murine ventricular myocardia. Eur. J. Pharm. 2013, 706, 48–55. [Google Scholar] [CrossRef]
- Mauricio, D.; Alonso, N.; Gratacòs, M. Chronic Diabetes Complications: The Need to Move beyond Classical Concepts. Trends Endocrinol. Metab. 2020. [Google Scholar] [CrossRef]
- Vadini, F.; Simeone, P.G.; Boccatonda, A.; Guagnano, M.T.; Liani, R.; Tripaldi, R.; Di Castelnuovo, A.; Cipollone, F.; Consoli, A.; Santilli, F. Liraglutide improves memory in obese patients with prediabetes or early type 2 diabetes: A randomized, controlled study. Int. J. Obes. 2020. [Google Scholar] [CrossRef]
- Uzar, E.; Alp, H.; Cevik, M.U.; Fırat, U.; Evliyaoglu, O.; Tufek, A.; Altun, Y. Ellagic acid attenuates oxidative stress on brain and sciatic nerve and improves histopathology of brain in streptozotocin-induced diabetic rats. Rats. Neurol. Sci 2012, 33, 567–574. [Google Scholar] [CrossRef]
- Cambay, Z.; Baydas, G.; Tuzcu, M.; Bal, R. Pomegranate (Punica granatum L.) flower improves learning and memory performances impaired by diabetes mellitus in rats. Acta Physiol. Hung. 2011, 98, 409–420. [Google Scholar] [CrossRef]
- Huang, H.; Liao, D.; Chen, G.; Chen, H.; Zhu, Y. Lack of efficacy of pomegranate supplementation for glucose management, insulin levels and sensitivity: Evidence from a systematic review and meta-analysis. Nutr. J. 2017, 16, 67. [Google Scholar] [CrossRef] [PubMed]
- Kataja-Tuomola, M.; Sundell, J.R.; Männistö, S.; Virtanen, M.J.; Kontto, J.; Albanes, D.; Virtamo, J. Effect of alpha-tocopherol and beta-carotene supplementation on the incidence of type 2 diabetes. Diabetologia 2008, 51, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, D.J.A.; Spence, J.D.; Giovannucci, E.L.; Kim, Y.I.; Josse, R.; Vieth, R.; Blanco Mejia, S.; Viguiliouk, E.; Nishi, S.; Sahye-Pudaruth, S.; et al. Supplemental Vitamins and Minerals for CVD Prevention and Treatment. J. Am. Coll. Cardiol. 2018, 71, 2570–2584. [Google Scholar] [CrossRef] [PubMed]
- De Lorgeril, M.; Salen, P.; Martin, J.L.; Monjaud, I.; Delaye, J.; Mamelle, N. Mediterranean diet, traditional risk factors, and the rate of cardiovascular complications after myocardial infarction: Final report of the Lyon Diet Heart Study. Circulation 1999, 99, 779–785. [Google Scholar] [CrossRef]
- Estruch, R.; Ros, E.; Salas-Salvadó, J.; Covas, M.I.; Corella, D.; Arós, F.; Gómez-Gracia, E.; Ruiz-Gutiérrez, V.; Fiol, M.; Lapetra, J.; et al. Primary Prevention of Cardiovascular Disease with a Mediterranean Diet Supplemented with Extra-Virgin Olive Oil or Nuts. N. Engl. J. Med. 2018, 378, e34. [Google Scholar] [CrossRef]
- Balfegó, M.; Canivell, S.; Hanzu, F.A.; Sala-Vila, A.; Martínez-Medina, M.; Murillo, S.; Mur, T.; Ruano, E.G.; Linares, F.; Porras, N.; et al. Effects of sardine-enriched diet on metabolic control, inflammation and gut microbiota in drug-naïve patients with type 2 diabetes: A pilot randomized trial. Lipids Health Dis. 2016, 15, 78. [Google Scholar] [CrossRef]
- Sala-Vila, A.; Valls-Pedret, C.; Rajaram, S.; Coll-Padrós, N.; Cofán, M.; Serra-Mir, M.; Pérez-Heras, A.M.; Roth, I.; Freitas-Simoes, T.M.; Doménech, M.; et al. Effect of a 2-year diet intervention with walnuts on cognitive decline. The Walnuts and Healthy Aging (WAHA) study: A randomized controlled trial. Am. J. Clin. Nutr. 2020, 111, 590–600. [Google Scholar] [CrossRef]
| Reference | Sample Size | Study Design | Participants’ Characteristics | T2D (%) | Age (Years) | Sex (M/F) | BMI (kg/m2) | Interventions | Duration | Main Results |
|---|---|---|---|---|---|---|---|---|---|---|
| Pomegranate juice | ||||||||||
| [61] | 85 | NR, SA | T2D (85) | 100 | NA | 40/45 | NA | 1.5 mL/kg bw | 1 single dose | • ↓FPG and HOMA-IR |
| • ↑HOMA %B after 3 h of administration | ||||||||||
| [62] | 16 | R, SB, C | Healthy | 0 | 31 ± 5 | NA | 23 ± 3 | PJ 200 mL vs. Placebo (sugar-balanced water), before bread consumption | 1 single dose | • ↓incremental AUC and peak glucose after PJ |
| [63] | 30 | R, P | MetS, adolescents | 0 | 13.4 ± 1.1 | 16/14 | 27.1 ± 1.1 | PJ (240 mL/d) vs. grape juice 18 mL/kg/d | 1 single dose | • ≈FPG (both arms) after 4 h of consumption |
| 4 wks | • ≈FPG in both arms | |||||||||
| [64] | 50 | NR, SA | T2D | 100 | 45 ± 8 | NA | 30 ± 3 | PJ 200 mL/d | 6 wks | • ↓FPG |
| [65] | 31 | NR, SA | T2D | 100 | 46.0 ± 8.3 | 15/16 | 29.5 ± 0.7 | concentrated PJ 50 g/d | 4 wks | • ≈FPG |
| [66] | 45 | R, DB, P | stable CHD | 24 | 69.0 ± 10.0 | 40/5 | 28.5 ± 5.6 | PJ 240 mL/d vs. Placebo (modified sports beverage) | 12 wks | • ≈FPG and HbA1c |
| [67] | 60 | R, SB | T2D | 100 | 54.7 ± 8.4 | 30/30 | 27.3 ± 3.7 | PJ 200 mL/d vs. Control (untreated) | 6 wks | • ≈FPG |
| [68] | 30 | R, DB, C | MetS | 0 | 51.6 ± 10.0 | 13/17 | NA | PJ 500 mL/d vs. Placebo (equivalent in sugar and acid content, without polyphenols) | 1 wk | • ≈FPG |
| • ≈insulin | ||||||||||
| • ≈HOMA-IR | ||||||||||
| [69] | 30 | R, DB, P | stable COPD | 0 | 61.7 ± 10.0 | NA | 31.0 ± 5.3 | PJ 400 mL/d vs. Placebo (synthetic flavored drink) | 5 wks | • ≈FPG |
| [70] | 20 | R, DB, P | Obesity | 0 | 37.3 ± 9.4 | NA | 34.5 ± 3.6 | PJ 120 mL/d vs. Placebo | 4 wks | • ≈FPG, AUC glucose |
| • insulin, AUC insulin | ||||||||||
| • insulinogenic index, Matsuda index | ||||||||||
| [71] | 28 | R, DB, C | Healthy | 0 | 50.4 ± 6.1 | 12/16 | 26.8 ± 3.4 | PJ 500 mL/d vs. Placebo (water plus equivalent carbohydrates) | 4 wks | • ↓FPG, |
| • ↓insulin and HOMA-IR | ||||||||||
| [72] | 44 | R, DB, P | T2D | 100 | 55.9 ± 6.7 | 23/21 | 29.0 ± 4.0 | PJ 250 mL/d vs. Placebo (equivalent in sugar content) | 12 wks | • ≈FPG |
| • ≈insulin or HOMA-IR | ||||||||||
| [73] | 21 | R, SB, P | Hypertension | 0 | 52.9 ± 8.7 | 6/15 | 27.4 ± 3.8 | PJ 150 mL/d vs. Placebo (water) | 2 wks | • ≈FPG |
| [74] | 77 | R, DB, P | Overweight, women | 0 | 41.5 ± 12.5 | 0/77 | 28.4 ± 2.2 | PV-based beverage 200 mL/d vs. Placebo (equivalent in sugar and acid content) | 8 wks | • ≈FPG, |
| • ≈FPI, HOMA-IR | ||||||||||
| [75] | 20 | R, DB, P | Endurance-based athletes | 0 | 35.3 ± 9.9 | 20/0 | NA | PJ 200 mL/d vs. PJ diluted 1:1 with water (200 mL/d) vs. seasonal fruit (equivalent energy) | 3 wks | • ≈FPG |
| [76] | 10 | R, SB | Healthy | 0 | 31.8 ± 6.6 | 5/5 | NA | PJ 500 mL/d vs. Placebo | 2 wks | • ≈FPG |
| Pomegranate extracts | ||||||||||
| [62] | 16 | R, DB, C | Healthy | 0 | 26 ± 6 | NA | 23 ± 2 | 400 mg PE vs. 200 mg placebo+200 mg PE vs. 400 mg placebo (before bread consumption) | 1 single dose | • ≈Incremental AUC |
| • ≈peak glucose | ||||||||||
| [77] | 64 | R, DB, P | Overweight and increased waist size | 0 | 35–65 | NA | NA | PFE 710 mg or 1420 mg vs. Placebo | 4 wks | • ≈FPG |
| [78] | 42 | R, DB, P | Overweight/obese | NA | 30–60 | NA | 31.8 ± 4.5 | PFE 1000 mg/d vs. Placebo (cellulose capsules) | 30 d | • ↓FPG, |
| • ↓FPI, HOMA-IR | ||||||||||
| Pomegranate-based dietary supplements | ||||||||||
| [79] | 20 | NR, SB | Abdominal obesity | 0 | 48 ± 4 | 10/10 | 29.7 ± 2.7 | Dietary supplement prepared from pomegranate and grape pomaces (50:50) 10 g vs. Control (untreated) | 1 single dose | • ≈Glucose after OGTT (either administered simultaneously or 10 h before of the OGTT) |
| • ≈HOMA-IR | ||||||||||
| Reference | Study Model | Interventions | Duration | Main Results |
|---|---|---|---|---|
| Diabetic Kidney Disease | ||||
| [41] | STZ-induced diabetic mice (BALB/c), males | Control vs. PPE-AuNP 5 (15 or 25 mg/kg every 2nd day) | 10 d | • ↓renal histopathology alterations |
| • ↓renal fibrosis markers (TGF-β and Col IV) | ||||
| • ↓renal oxidative stress (↓ROS, ↓LPO, ↓nitrite; ↓MAPK pathway; ↓NOX4 and p47phox expression; ↑SOD, ↑GSH, ↑Nrf2 activation) | ||||
| • ↓renal inflammation (↓NF-κB and STAT3 pathways: IL-1β, IFN-γ, IL-6, IL-10, TNF-α, COX2) | ||||
| • ↓renal AGEs and RAGE | ||||
| • ↓urinary urea and creatinine | ||||
| [97] | STZ-induced diabetic rats (Wistar), males | Control vs. EA (0.2 or 2%) | 12 wks | • ↓protein glycation in glomeruli (IgG cross-linking, CML accumulation) |
| • ↓ renal RAGE expression | ||||
| • ↓ renal TGF-β expression | ||||
| • ↓accumulation of ECM | ||||
| • ↑podocyte specific markers (podocin, nephrin) | ||||
| • ↓UACR and urinary urea | ||||
| [101] | STZ-induced diabetic mice (Balb/cA), males | Control (CA, 2.5 or 5%) vs. EA (2.5 or 5%) | 12 wks | • ↓renal AGEs (CML, pentosidine, sorbitol, fructose) |
| • ↓renal polyol pathway (↓AR and SDH activity) | ||||
| • ↓renal inflammation markers (IL-1β, IL-6, TNF-α, MCP-1) | ||||
| • ↓serum BUN | ||||
| • ↑creatinine clearance | ||||
| [102] | STZ-induced diabetic rats (Wistar), +/−Ang II), males | Control vs. PJ (100 or 300 mg/kg/d) | 4 wks | • ↓renal histopathology tubular alterations (no mesangial protection) |
| • ↓renal oxidative stress/lipid peroxidation (↓MDA, ↑GSH, ↑SOD, ↑CAT) | ||||
| [103] | STZ-induced diabetic rats (Wistar), either sex | Control vs. PLE (50, 100 or 200 mg/kg/d) | 4 wks | • ↓renal histopathology alterations and inflammatory cells infiltration |
| • ↓renal oxidative stress/peroxidation (↓MDA, ↑GSH, ↑SOD, ↑CAT) | ||||
| • ↓albuminuria | ||||
| • ↓Serum BUN and creatinine | ||||
| • ↑creatinine clearance | ||||
| [104] | STZ-induced diabetic mice (ICR), males | Control vs. EA (50, 100 or 150 mg/kg/d) vs. IRB (180 mg/kg/d) | 4 wks | • ↓renal histopathology alterations and inflammatory cells infiltration (≈IRB) |
| • ↓renal inflammation (↓TLR4/NF-κB) | ||||
| • ↓albuminuria (≈IRB) | ||||
| • ↓serum creatinine (≈IRB) | ||||
| • ↓serum oxidative stress (↓MDA and ↑SOD levels) (≈IRB) | ||||
| [105] | HFD/low-dose STZ-induced type 2 diabetic rats (Wistar), males | Control vs. EA (20 or 40 mg/kg/d) | 16 wks | • ↓renal histopathology tubular alterations (no mesangial protection) |
| • ↓renal PAS accumulation | ||||
| • ↓renal expression of NF-κBp65, TGF-β and fibronectin | ||||
| • ↓serum inflammatory cytokines (IL-1β, IL-6, TNF-α) | ||||
| • ↓renal oxidative stress/peroxidation (↓MDA, ↑GSH, ↑antioxidant enzymes activity) | ||||
| • ↓serum creatinine, BUN, proteinuria | ||||
| • ↑creatinine clearance | ||||
| Retinopathy | ||||
| [109] | STZ-induced diabetic rats (Wistar-NIN), males | Control vs. EA (0.2 or 2%) | 12 wks | • ↑retinal thickness |
| • ↓retinal AGE (CML) and RAGE | ||||
| • ↓retinal pro-apoptotic markers (Bax) | ||||
| • ↓retinal neovascularization markers (HIF-1α and VEGF) | ||||
| • ↓retinal gliosis markers (GFAP) | ||||
| • Improvement electroretinogram abnormalities | ||||
| [110] | STZ-induced diabetic rats (Sprague-Dawley), either sex | Control vs. PJ (100 µL/d) | 10 wks | • ↓renal histopathology tubular alterations (no mesangial protection) |
| • ↓retinal oxidative stress/lipid peroxidation (↓8OHdG, ↓MDA, ↑GSH, ↑GSH-Px) | ||||
| • ↓retinal eNOS and P65 staining | ||||
| Reference | Study Model | Interventions | Duration | Main Results |
|---|---|---|---|---|
| Atherosclerosis, animal studies | ||||
| [118] | ApoE-deficient mice, males | Control vs. PPE or PAE or PFE or PSE (200 µg GAE/d) | 13 wks | • ↓atherosclerotic lesion size (no effect in PAE or PSE) |
| • ↓serum lipoperoxides (PFE and PAE) | ||||
| • ↑serum PON1 activity (only PAE) | ||||
| • ↓macrophage oxidized-LDL uptake (only PPE) | ||||
| • ↑HDL-cholesterol efflux (only PAE) | ||||
| Control vs. PJ (200 µg GAE/d) | 13 wks | • ↓atherosclerotic lesion size | ||
| • ↓serum lipoperoxides | ||||
| • ↑serum PON1 activity | ||||
| • ↓macrophage LDL and oxidized-LDL uptake | ||||
| • ↑HDL-cholesterol efflux | ||||
| [119] | Obese Zucker rats, females | Control (atherogenic diet) vs. PFE (30 µL/d)- supplemented atherogenic diet | 5 wks | • ↑vasodilatation in response to acetylcholine |
| Control (atherogenic diet) vs. PJ (30 µL/d)- supplemented atherogenic diet | 5 wks | • ↑vasodilatation in response to acetylcholine | ||
| [120] | High-carbohydrate, HFD-induced MetS rats (Wistar), males | Control vs. EA (0.8 g/kg food) | 8 wks | • ↑vasodilatation in response to acetylcholine |
| • ↑aortic contractile in response to noradrenalin | ||||
| [121] | STZ-induced diabetic rats (NIN-Wistar), males | Control vs. EA (2%) | 12 wks | • ↓medial layer thickness |
| • ↓lipid accumulation | ||||
| • ↓deposition of collagen | ||||
| • ↓cyclin D1 expression in media (SMC proliferation marker) | ||||
| Atherosclerosis, human studies | ||||
| [63] | MetS, adolescents (n = 30) | PJ (240 mL/d) vs. grape juice (18 mL/kg/d) | 4 wks | • ↑FMD, both interventions |
| [73] | Hypertension (n = 20) | Placebo (water) vs. PJ (150 mL/d) | 2 wks | • no improvements in FMD |
| Cardiopathy, animal studies | ||||
| [30] | Myocardial infarction in STZ-induced diabetic rats (Wistar), males | Control vs. Terminalia arjuna extract (500 mg/kg/d) or vildagliptin (10 mg/kg/d) | 4 wks | • ↓focal myofiber loss |
| • ↓inflammation | ||||
| • ↓necrosis | ||||
| • ↓edema | ||||
| • ↓cardiac parameters (CPK-MB, but less than vildagliptin) | ||||
| [120] | High-carbohydrate, HFD-induced MetS rats (Wistar), males | Control vs. EA (0.8 g/kg food) | 8 wks | • ↓cardiac infiltration of inflammatory cells |
| • ↓cardiac collagen deposition | ||||
| • ↑cardiac hemodynamic performance (↑EF; ↓LVIDs; ↓systolic volume, ↓fractional shortening; ↓estimated LV mass; ↓LV diastolic stiffness) | ||||
| • ↓cardiac NF-κB expression | ||||
| • ↑cardiac CPT-1 expression | ||||
| [122] | STZ-induced diabetic mice (Balb/c), males | Control vs. EA (2%) | 12 wks | • ↓cardiac triglyceride content (but not cholesterol) |
| • ↓cardiac inflammation (IL-1β; IL-6; TNF-α; MCP-1) | ||||
| • ↓cardiac oxidative stress (↓MDA, ↓ROS; ↑GSH, ↑CAT, ↑SOD, ↑GSH-Px) | ||||
| [123] | Zucker diabetic fatty rats, males | Control vs. PFE (500 mg/kg/d) | 6 wks | • ↓cardiac triglyceride content (but not cholesterol) |
| • ↓abnormal cardiac upregulation lipogenic genes (FATP, PPARα, CPT-1, ACO, AMPKα2) | ||||
| [124] | Zucker diabetic fatty rats, males | Control vs. PFE (500 mg/kg/d) | 6 wks | • ↓interstitial and perivascular collagen deposit |
| • ↓cardiac fibrosis markers (fibronectin, collagen I and III, ET-1 and ETA) | ||||
| • ↓NF-κB pathway activation | ||||
| [125] | STZ-induced diabetic rats (Wistar), males | Control vs. Urolithin A or Urolithin B (2.5 mg/kg/d) | 3 wks | • ↑cardiac hemodynamic performance, specially urolithin B (↑LVSP; ↑+dP/dtmax, ↓IVCT, ↓Tcycle) |
| • Improvement of cardiomyocyte mechanics and calcium transients | ||||
| • ↓fractalkine (cardiac pro-inflammatory cytokine) | ||||
| Cardiopathy, human studies | ||||
| [66] | CHD and myocardial ischemia (n = 45) | Placebo vs. PJ (240 mL/d) | 13 wks | • ↓stress-induced ischemia (in myocardial perfusion single-photon emission computed tomographic technetium-99m tetrofosmin scintigraphy) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Amor, A.J.; Gómez-Guerrero, C.; Ortega, E.; Sala-Vila, A.; Lázaro, I. Ellagic Acid as a Tool to Limit the Diabetes Burden: Updated Evidence. Antioxidants 2020, 9, 1226. https://doi.org/10.3390/antiox9121226
Amor AJ, Gómez-Guerrero C, Ortega E, Sala-Vila A, Lázaro I. Ellagic Acid as a Tool to Limit the Diabetes Burden: Updated Evidence. Antioxidants. 2020; 9(12):1226. https://doi.org/10.3390/antiox9121226
Chicago/Turabian StyleAmor, Antonio J., Carmen Gómez-Guerrero, Emilio Ortega, Aleix Sala-Vila, and Iolanda Lázaro. 2020. "Ellagic Acid as a Tool to Limit the Diabetes Burden: Updated Evidence" Antioxidants 9, no. 12: 1226. https://doi.org/10.3390/antiox9121226
APA StyleAmor, A. J., Gómez-Guerrero, C., Ortega, E., Sala-Vila, A., & Lázaro, I. (2020). Ellagic Acid as a Tool to Limit the Diabetes Burden: Updated Evidence. Antioxidants, 9(12), 1226. https://doi.org/10.3390/antiox9121226