Antioxidants N-Acetylcysteine and Vitamin C Improve T Cell Commitment to Memory and Long-Term Maintenance of Immunological Memory in Old Mice
Abstract
1. Introduction
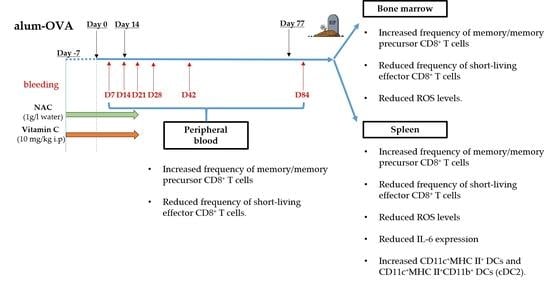
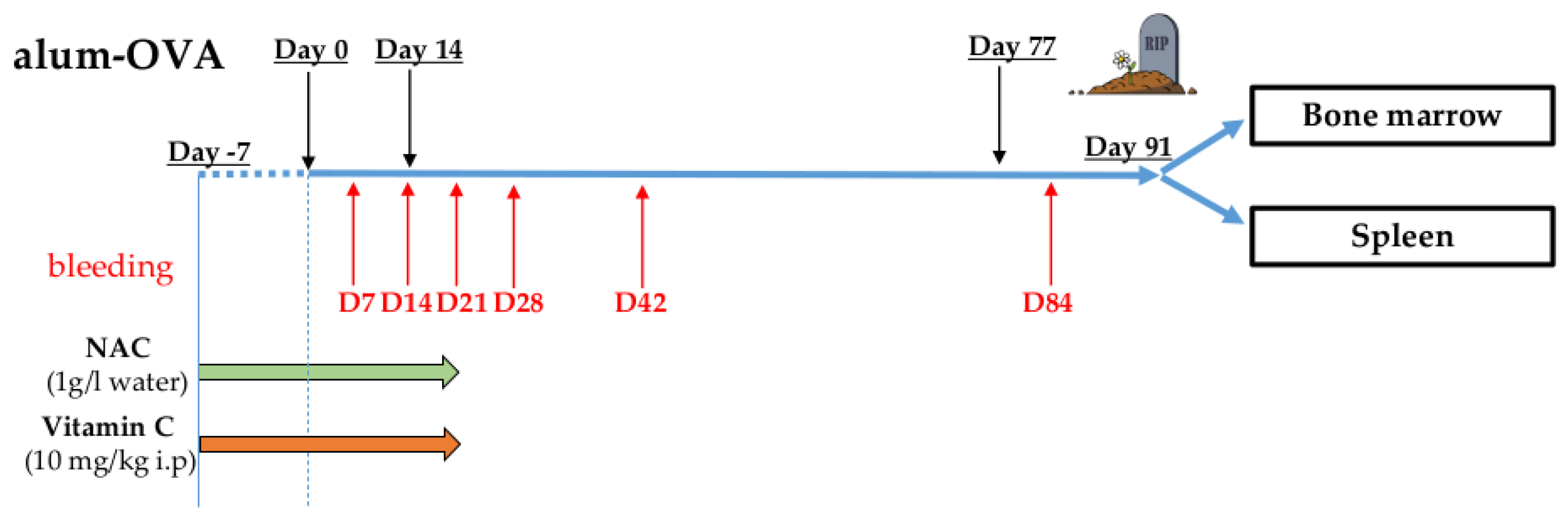
2. Materials and Methods
2.1. Mice
2.2. Material Processing and Cell Culture
2.3. Flow Cytometry
2.4. ROS Measurement
2.5. Statistical Analysis
2.6. Study Approval
3. Results
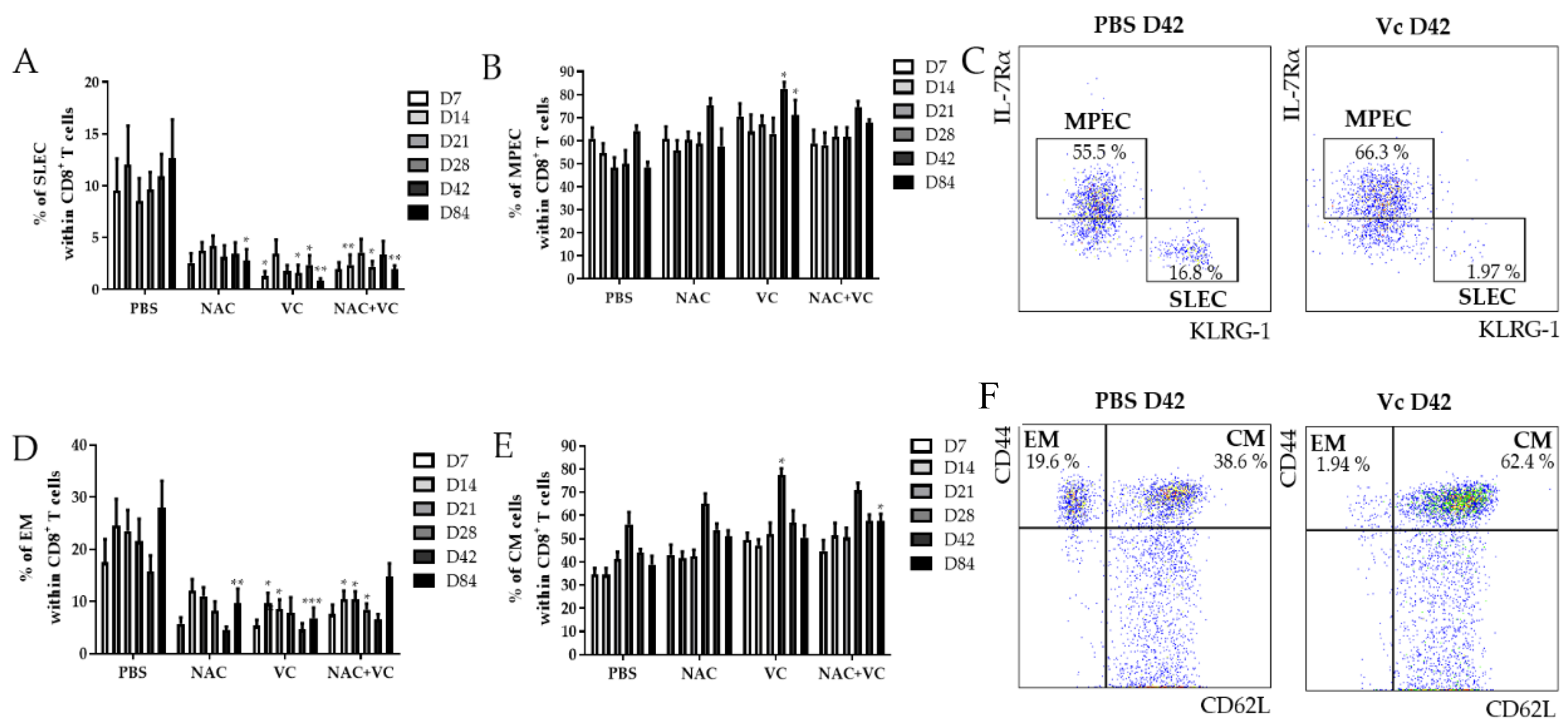
3.1. Effector/Memory T Cell Subsets Change in the Peripheral Blood (PB) after Treatment with Antioxidants
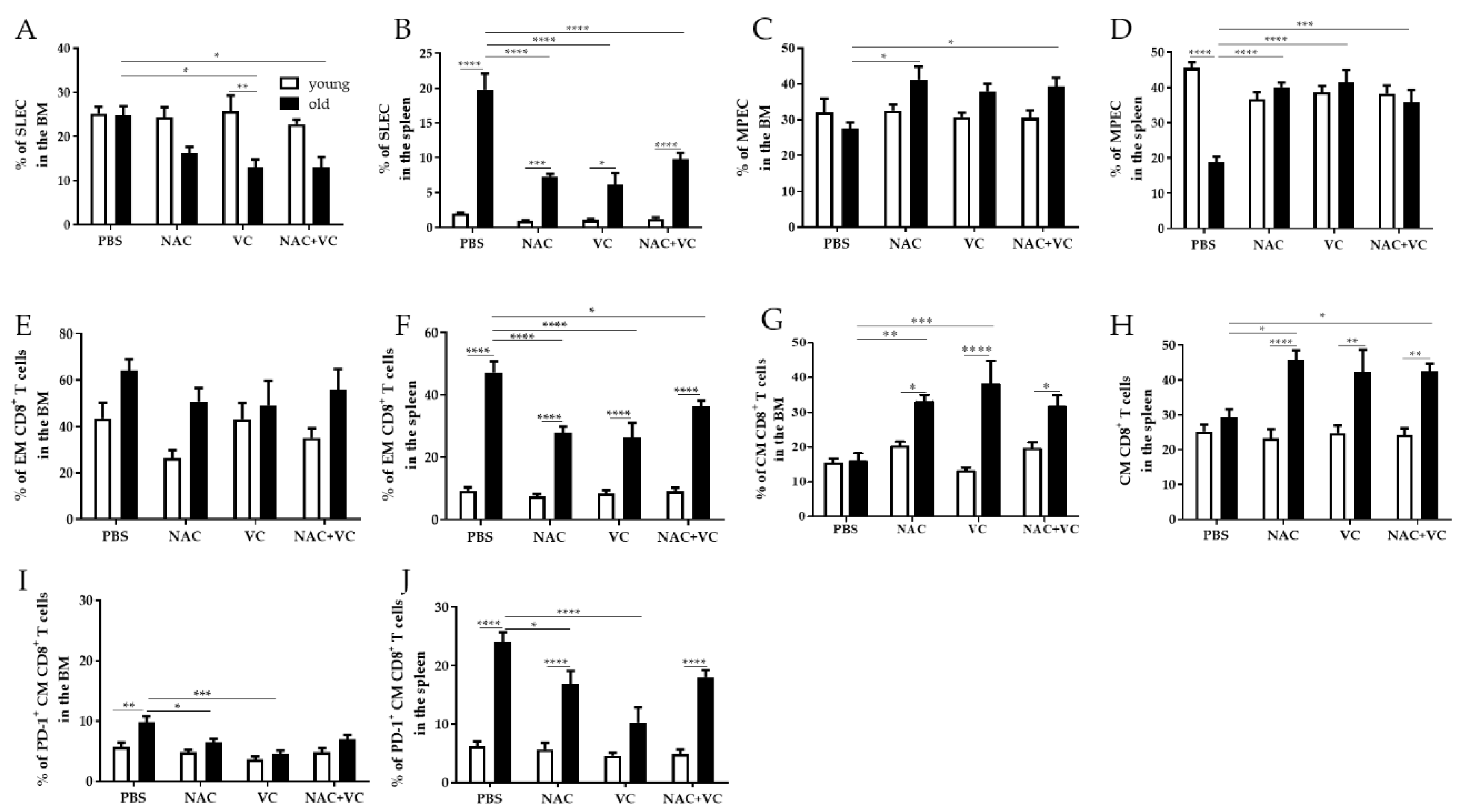
3.2. Effector/Memory T Cell Subsets Change in the BM and in the Spleen after Treatment with Antioxidants
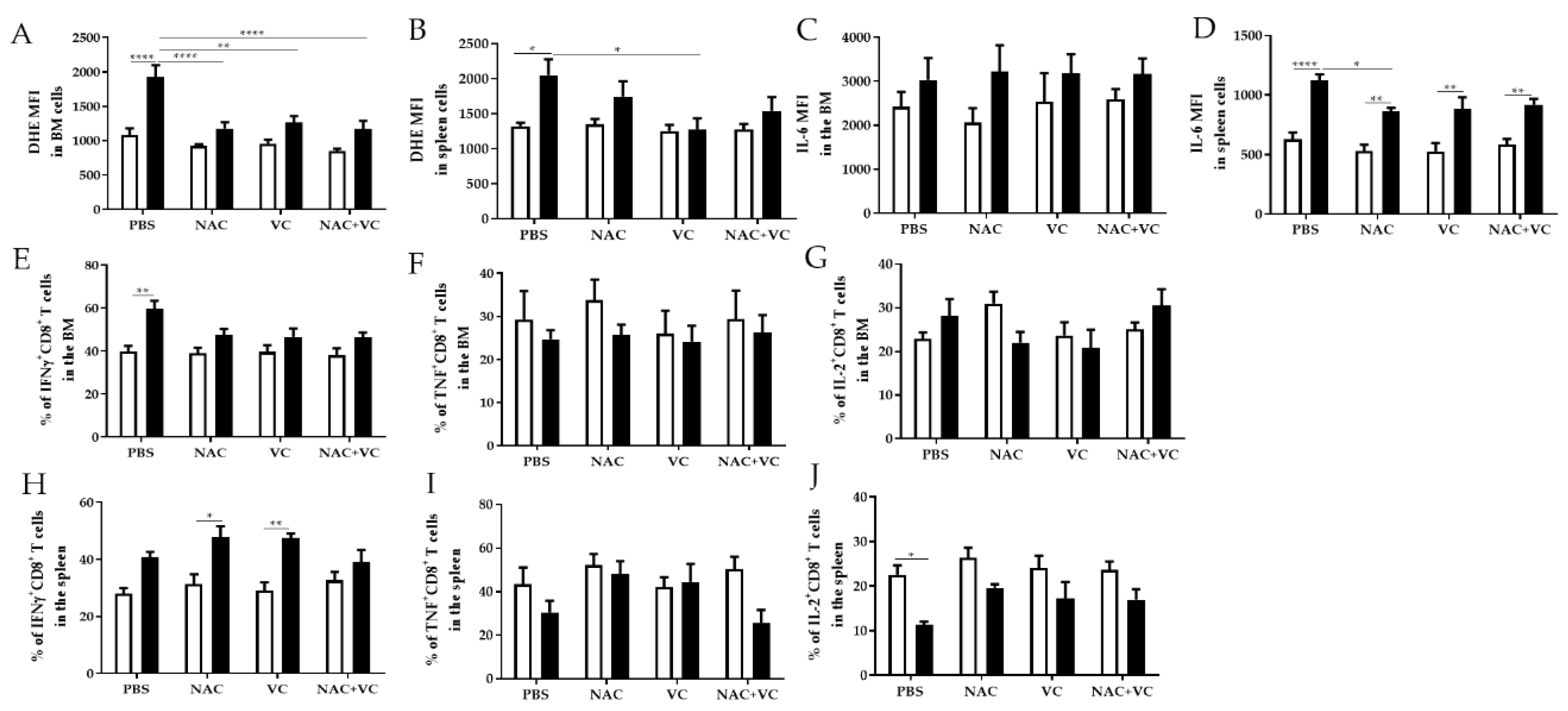
3.3. Antioxidant Treatments Affect ROS Levels and Pro-Inflammatory Molecules in the BM and Spleen
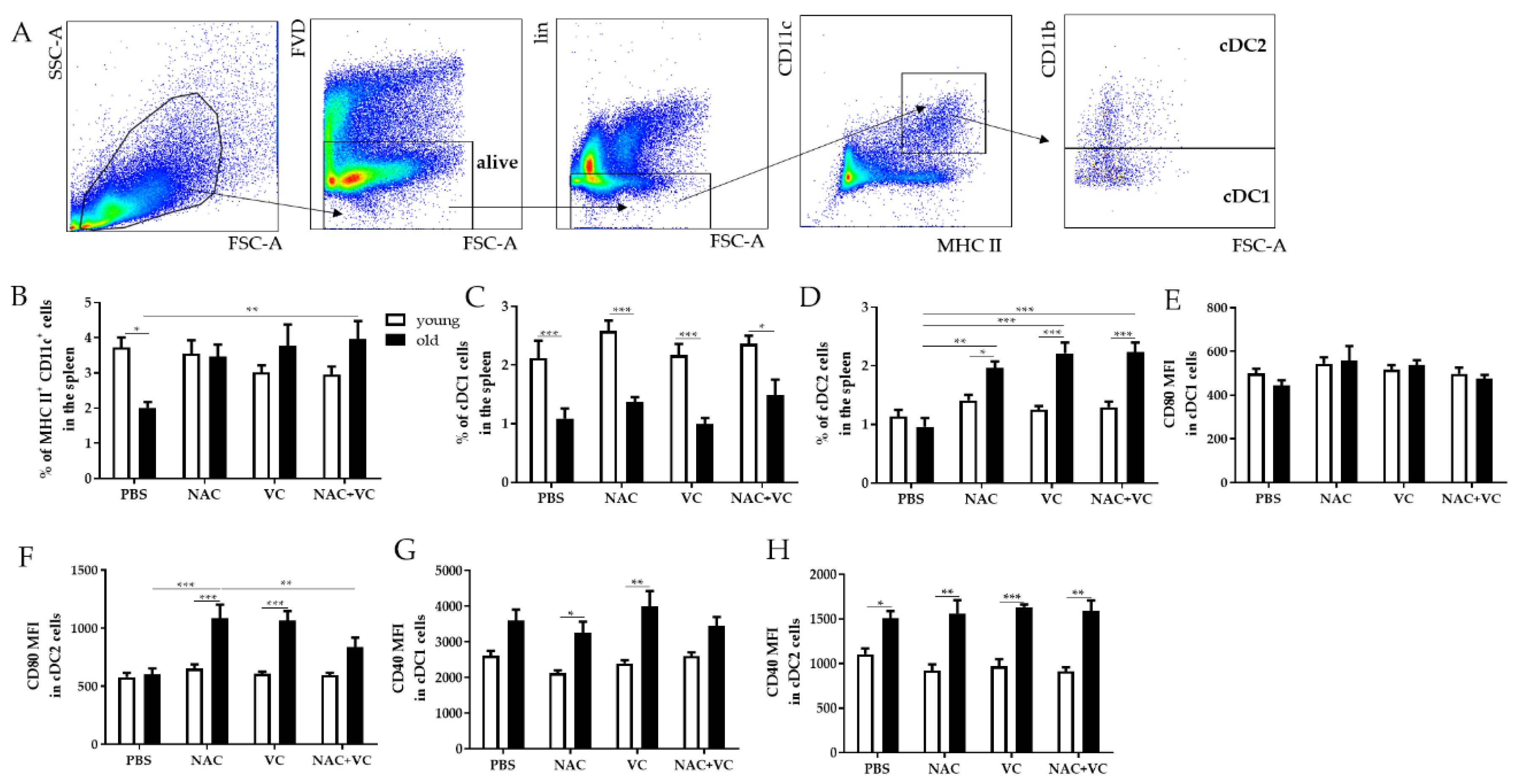
3.4. Dendritic Cell Subsets Change in the Spleen after Antioxidant Treatments
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- George, A.J.T.; Ritter, M.A. Thymic involution with ageing. Obsolescence or good housekeeping? Immunol. Today 1996, 17, 267–272. [Google Scholar] [CrossRef]
- Aspinall, R.; Andrew, D. Thymic involution in aging. J. Clin. Immunol. 2000, 20, 250–256. [Google Scholar] [CrossRef] [PubMed]
- Boyman, O.; Letourneau, S.; Krieg, C.; Sprent, J. Homeostatic proliferation and survival of naive and memory T cells. Eur. J. Immunol. 2009, 39, 2088–2094. [Google Scholar] [CrossRef] [PubMed]
- Wikby, A.; Nilsson, B.O.; Forsey, R.; Thompson, J.; Strindhall, J.; Lofgren, S.; Ernerudh, J.; Pawelec, G.; Ferguson, F.; Johansson, B. The immune risk phenotype is associated with IL-6 in the terminal decline stage: Findings from the Swedish NONA immune longitudinal study of very late life functioning. Mech. Ageing Dev. 2006, 127, 695–704. [Google Scholar] [CrossRef] [PubMed]
- Dumitriu, I.E. The life (and death) of CD4 + CD28(null) T cells in inflammatory diseases. Immunology 2015, 146, 185–193. [Google Scholar] [CrossRef] [PubMed]
- Tokoyoda, K.; Hauser, A.E.; Nakayama, T.; Radbruch, A. Organization of immunological memory by bone marrow stroma. Nat. Rev. Immunol. 2010, 10, 193–200. [Google Scholar] [CrossRef]
- Okhrimenko, A.; Grün, J.R.; Westendorf, K.; Fang, Z.; Reinke, S.; von Roth, P.; Wassilew, G.; Kuhl, A.A.; Kudernatsch, R.; Demski, S.; et al. Human memory T cells from the bone marrow are resting and maintain long-lasting systemic memory. Proc. Natl. Acad. Sci. USA 2014, 111, 9229–9234. [Google Scholar] [CrossRef]
- Herndler-Brandstetter, D.; Landgraf, K.; Tzankov, A.; Jenewein, B.; Brunauer, R.; Laschober, G.T.; Parson, W.; Kloss, F.; Gassner, R.; Lepperdinger, G.; et al. The impact of aging on memory T cell phenotype and function in the human bone marrow. J. Leukoc Biol. 2012, 91, 197–205. [Google Scholar] [CrossRef]
- Li, J.; Valentin, A.; Ng, S.; Beach, R.K.; Alicea, C.; Bergamaschi, C.; Felber, B.K.; Pavlakis, G.N. Differential effects of IL-15 on the generation, maintenance and cytotoxic potential of adaptive cellular responses induced by DNA vaccination. Vaccine 2015, 33, 1188–1196. [Google Scholar] [CrossRef][Green Version]
- Osborne, L.C.; Dhanji, S.; Snow, J.W.; Priatel, J.J.; Ma, M.C.; Miners, M.J.; Teh, H.; Goldsmith, M.A.; Abraham, N. Impaired CD8 T cell memory and CD4 T cell primary responses in IL-7R alpha mutant mice. J. Exp. Med. 2007, 204, 619–631. [Google Scholar] [CrossRef]
- Pangrazzi, L.; Meryk, A.; Naismith, E.; Koziel, R.; Lair, J.; Krismer, M.; Trieb, K.; Grubeck-Loebenstein, B. “Inflamm-aging” influences immune cell survival factors in human bone marrow. Eur. J. Immunol. 2017, 47, 481–492. [Google Scholar] [CrossRef] [PubMed]
- Pangrazzi, L.; Naismith, E.; Meryk, A.; Keller, M.; Jenewein, B.; Trieb, K.; Grubeck-Loebenstein, B. Increased IL-15 Production and Accumulation of Highly Differentiated CD8+ Effector/Memory T Cells in the Bone Marrow of Persons with Cytomegalovirus. Front. Immunol. 2017, 8, 715. [Google Scholar] [CrossRef] [PubMed]
- Naismith, E.; Pangrazzi, L. The impact of oxidative stress, inflammation, and senescence on the maintenance of immunological memory in the bone marrow in old age. Biosci. Rep. 2019, 39, BSR20190371. [Google Scholar] [CrossRef] [PubMed]
- McKee, A.S.; Munks, M.W.; MacLeod, M.K.; Fleenor, C.J.; Van Rooijen, N.; Kappler, J.W.; Marrack, P. Alum induces innate immune responses through macrophage and mast cell sensors, but these sensors are not required for alum to act as an adjuvant for specific immunity. J. Immunol. 2009, 183, 4403–4414. [Google Scholar] [CrossRef]
- Lehmann, D.; Karussis, D.; Misrachi-Koll, R.; Shezen, E.; Ovadia, H.; Abramsky, O. Oral administration of the oxidant-scavenger N-acetyl--cysteine inhibits acute experimental autoimmune encephalomyelitis. J. Neuroimmunol. 1994, 50, 35–42. [Google Scholar] [CrossRef]
- Joshi, N.S.; Cui, W.; Chandele, A.; Lee, H.K.; Urso, D.R.; Hagman, J.; Gapin, L.; Kaech, S.M. Inflammation directs memory precursor and short-lived effector CD8(+) T cell fates via the graded expression of T-bet transcription factor. Immunity 2007, 27, 281–295. [Google Scholar] [CrossRef]
- Remmerswaal, E.; Hombrink, P.; Nota, B.; Pircher, H.; Ten Berge, I.; van Lier, R.; van Aalderen, M.C. Expression of IL-7Rα and KLRG1 defines functionally distinct CD8+ T-cell populations in humans. Eur. J. Immunol. 2019, 49, 694–708. [Google Scholar] [CrossRef]
- Guilliams, M. Dendritic cells, monocytes and macrophages: A unified nomenclature based on ontogeny. Nat. Rev. Immunol. 2014, 14, 571–578. [Google Scholar] [CrossRef]
- Van der Merwe, P.A.; Bodian, D.L.; Daenke, S.; Linsley, P.; Davis, S.J. CD80 (B7–1) binds both CD28 and CTLA-4 with a low affinity and very fast kinetics. J. Exp. 1997, 185, 393–403. [Google Scholar] [CrossRef]
- Ma, D.Y.; Clark, E.A. The role of CD40 and CD154/CD40L in dendritic cells. Semin. Immunol. 2009, 21, 265–272. [Google Scholar] [CrossRef]
- Boraschi, D.; Aguado, M.T.; Dutel, C.; Goronzy, J.; Louis, J.; Grubeck-Loebenstein, B.; Rappuoli, R.; del Giudice, G. The gracefully aging immune system. Sci. Transl. Med. 2013, 5, 185. [Google Scholar] [CrossRef] [PubMed]
- Pangrazzi, L.; Reidla, J.; Carmona Arana, J.A.; Naismith, E.; Miggitsch, C.; Meryk, A.; Keller, M.; Krause, A.A.N.; Melzer, F.L.; Trieb, K. CD28 and CD57 define four populations with distinct phenotypic properties within human CD8 + T cells. Eur. J. Immunol. 2020, 50, 363–379. [Google Scholar] [CrossRef] [PubMed]
- Northfield, J.; Lucas, M.; Jones, H.; Young, N.T.; Klenerman, P. Does memory improve with age? CD85j (ILT-2/LIR-1) expression on CD8 T cells correlates with ‘memory inflation’ in human cytomegalovirus infection. Immunol. Cell Biol. 2005, 83, 182–188. [Google Scholar] [CrossRef]
- Naismith, E.; Pangrazzi, L.; Grasse, M.; Keller, M.; Miggitsch, C.; Weinberger, B.; Trieb, K.; Grubeck-Loebenstein, B. Peripheral antibody concentrations are associated with highly differentiated T cells and inflammatory processes in the human bone marrow. Immun. Ageing 2019, 16, 21. [Google Scholar] [CrossRef] [PubMed]
- Rushworth, G.F.; Megson, I.L. Existing and potential therapeutic uses for N-acetylcysteine: The need for conversion to intracellular glutathione for antioxidant benefits. Pharmacol. Ther. 2014, 141, 150–159. [Google Scholar] [CrossRef] [PubMed]
- Elbini Dhouib, I.; Jallouli, M.; Annabi, A.; Gharbi, N.; Elfazaa, S.; Lasram, M.M. A minireview on N-acetylcysteine: An old drug with new approaches. Life Sci. 2016, 15, 359–363. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.Y. N-acetylcysteine, reactive oxygen species and beyond. Cancer Biol. Ther. 2010, 9, 109–110. [Google Scholar] [CrossRef]
- Jacob, R.A.; Sotoudeh, G. Vitamin C function and status in chronic disease. Nutr. Clin. Care 2002, 5, 66–74. [Google Scholar] [CrossRef]
- Łukawski, M.; Dałek, P.; Borowik, T.; Foryś, A.; Langner, M.; Witkiewicz, W.; Przybylo, M. New oral liposomal vitamin C formulation: Properties and bioavailability. J. Liposome Res. 2020, 30, 227–234. [Google Scholar] [CrossRef]
- De Flora, S.; Grassi, C.; Carati, L. Attenuation of influenza-like symptomatology and improvement of cell-mediated immunity with long-term N-acetylcysteine treatment. Eur. Respir. J. 1997, 10, 1535–1541. [Google Scholar] [CrossRef]
- Arranz, L.; Fernández, C.; Rodríguez, A.; Ribera, J.M.; De la Fuente, M. The glutathione precursor N-acetylcysteine improves immune function in postmenopausal women. Free Radic. Biol. Med. 2008, 45, 1252–1262. [Google Scholar] [CrossRef] [PubMed]
- Hemilä, H. Vitamin C and Infections. Nutrients 2017, 9, 339. [Google Scholar] [CrossRef] [PubMed]
- Tokoyoda, K.; Zehentmeier, S.; Hegazy, A.N.; Albrecht, I.; Grun, J.R.; Lohning, M.; Radbruch, A. Professional memory CD4+ T lymphocytes preferentially reside and rest in the bone marrow. Immunity 2009, 30, 721–730. [Google Scholar] [CrossRef] [PubMed]
- Crauste, F.; Mafille, J.; Boucinha, L.; Djebali, S.; Gandrillon, O.; Marvel, J.; Arpin, C. Identification of Nascent Memory CD8 T Cells and Modeling of Their Ontogeny. Cell Syst. 2017, 4, 306–317. [Google Scholar] [CrossRef] [PubMed]
- Sallusto, F.; Lenig, D.; Förster, R.; Lipp, M.; Lanzavecchia, A. Two subsets of memory T lymphocytes with distinct homing potentials and effector functions. Nature 1999, 401, 708–712. [Google Scholar] [CrossRef]
- Francisco, L.M.; Sage, P.T.; Sharpe, A.H. The PD-1 pathway in tolerance and autoimmunity. Immunol. Rev. 2010, 236, 219–242. [Google Scholar] [CrossRef]
- Franceschi, C.; Bonafe, M.; Valensin, S.; Olivieri, F.; De Luca, M.; Ottaviani, E.; De Benedictis, G. Inflamm-aging. An evolutionary perspective on immunosenescence. Ann. N. Y. Acad. Sci. 2000, 908, 244–254. [Google Scholar] [CrossRef]
- Krishnaswamy, J.K.; Gowthaman, U.; Zhang, B.; Mattsson, J.; Szeponik, L.; Liu, D.; Wu, R.; White, T.; Calabro, S.; Xu, L.; et al. Migratory CD11b+ conventional dendritic cells induce T follicular helper cell-dependent antibody responses. Sci. Immunol. 2017, 2. [Google Scholar] [CrossRef]
- Yang, Y.; Bazhin, A.V.; Werner, J.; Karakhanova, S. Reactive oxygen species in the immune system. Int. Rev. Immunol. 2013, 32, 249–270. [Google Scholar] [CrossRef]
- Seifirad, S.; Ghaffari, A.; Amoli, M.M. The antioxidants dilemma: Are they potentially immunosuppressants and carcinogens? Front. Physiol. 2014, 5, 245. [Google Scholar] [CrossRef][Green Version]
- Smetana, J.; Chlibek, R.; Shaw, J.; Splino, M.; Prymula, R. Influenza vaccination in the elderly. Hum. Vaccin. Immunother. 2018, 4, 540–549. [Google Scholar] [CrossRef] [PubMed]
| Antigen | Fluorocrome | Company | Clone |
|---|---|---|---|
| KLRG-1 | BV421 | Biolegend | 2F1/KLRG1 |
| CD44 | FITC | Biolegend | IM7 |
| IL-7Rα | PE | Biolegend | A7R34 |
| CD8 | PeCy7 | Biolegend | 53-6.7 |
| CD8 | PerCp | Biolegend | 53-6.7 |
| CD62L | APC | Biolegend | MEL-14 |
| CD4 | BV510 | Biolegend | GK1.5 |
| CD4 | Pecy7 | Biolegend | GK1.5 |
| CD3 | APC-Vio770 | Miltenyi | REA641 |
| PD-1 | PE | Biolegend | 29F.1A12 |
| IL-6 | APC | Biolegend | MP5-20F3 |
| IFNg | PE | Biolegend | XMG1.2 |
| TNF | FITC | Biolegend | MP6-XT22 |
| IL-2 | APC | Biolegend | JES6-5H4 |
| CD3 | BV510 | Biolegend | 17A2 |
| CD11c | BV510 | Biolegend | N418 |
| MHC II | FITC | Biolegend | M5/114.15.2 |
| CD40 | PE | Biolegend | 3/23 |
| NKp46 | PerCp | Biolegend | 29A1.4 |
| CD19 | PerCp | Biolegend | 6D5 |
| CD3 | PerCp | Biolegend | 145-2C11 |
| CD80 | APC | Biolegend | 16-10A1 |
| CD11b | APC-Cy7 | Miltenyi | M1/70.15.11.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meryk, A.; Grasse, M.; Balasco, L.; Kapferer, W.; Grubeck-Loebenstein, B.; Pangrazzi, L. Antioxidants N-Acetylcysteine and Vitamin C Improve T Cell Commitment to Memory and Long-Term Maintenance of Immunological Memory in Old Mice. Antioxidants 2020, 9, 1152. https://doi.org/10.3390/antiox9111152
Meryk A, Grasse M, Balasco L, Kapferer W, Grubeck-Loebenstein B, Pangrazzi L. Antioxidants N-Acetylcysteine and Vitamin C Improve T Cell Commitment to Memory and Long-Term Maintenance of Immunological Memory in Old Mice. Antioxidants. 2020; 9(11):1152. https://doi.org/10.3390/antiox9111152
Chicago/Turabian StyleMeryk, Andreas, Marco Grasse, Luigi Balasco, Werner Kapferer, Beatrix Grubeck-Loebenstein, and Luca Pangrazzi. 2020. "Antioxidants N-Acetylcysteine and Vitamin C Improve T Cell Commitment to Memory and Long-Term Maintenance of Immunological Memory in Old Mice" Antioxidants 9, no. 11: 1152. https://doi.org/10.3390/antiox9111152
APA StyleMeryk, A., Grasse, M., Balasco, L., Kapferer, W., Grubeck-Loebenstein, B., & Pangrazzi, L. (2020). Antioxidants N-Acetylcysteine and Vitamin C Improve T Cell Commitment to Memory and Long-Term Maintenance of Immunological Memory in Old Mice. Antioxidants, 9(11), 1152. https://doi.org/10.3390/antiox9111152