Bioaccessible Raspberry Extracts Enriched in Ellagitannins and Ellagic Acid Derivatives Have Anti-Neuroinflammatory Properties
Abstract
1. Introduction
2. Materials and Methods
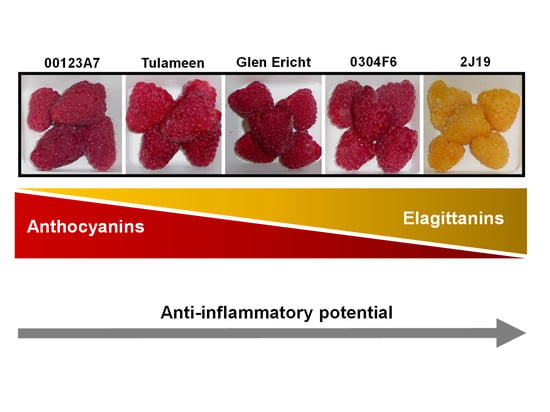
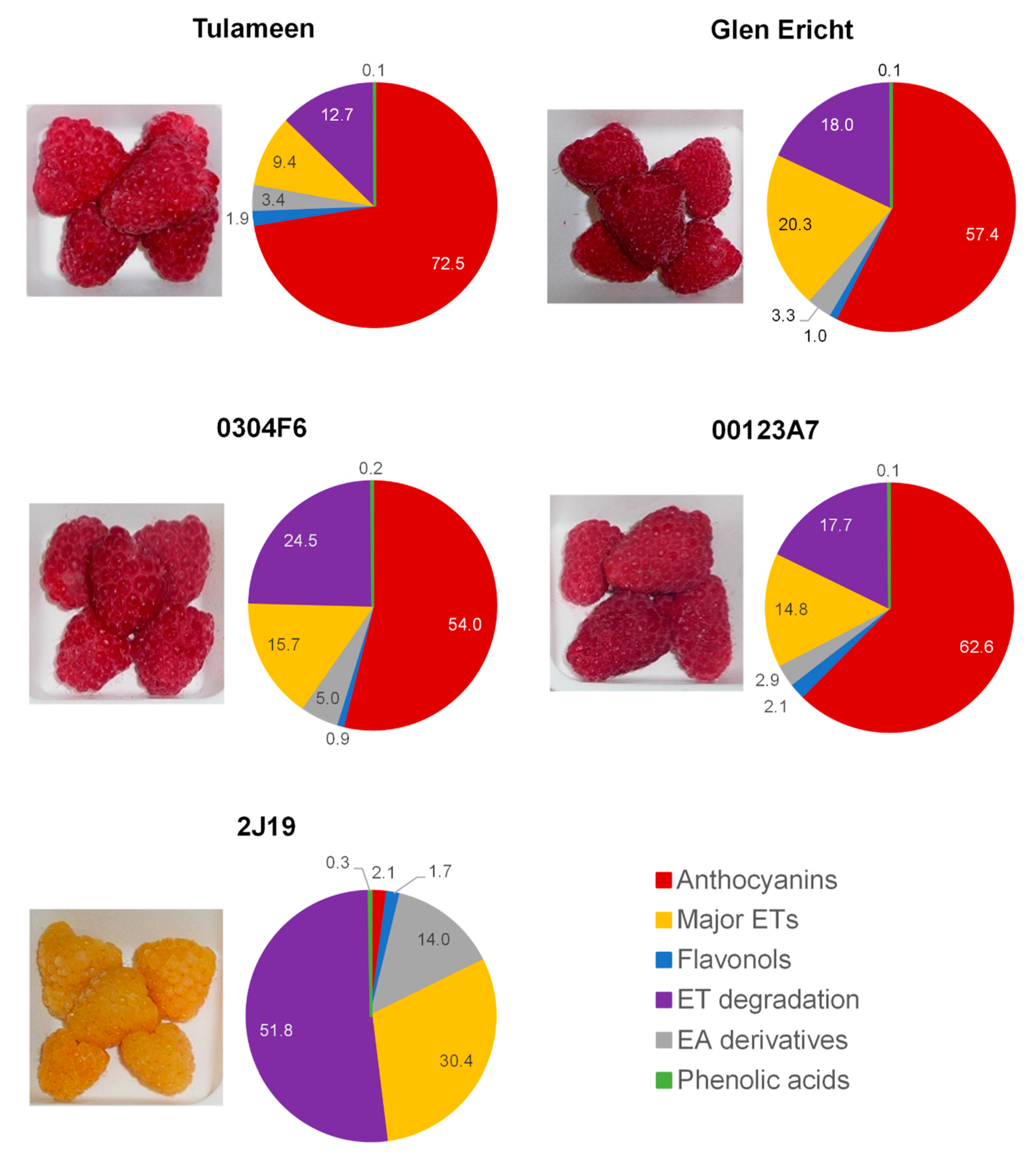
2.1. Plant Material and Characterization of Phenolic Diversity
2.2. In Vitro Digestion of Berry Powders
2.3. Liquid Chromatography-Photodiode Array-Mass Spectrometry (LC-PDA-MS)
2.4. Cell Culture Maintenance, Incubation with GIB Fractions and LPS Stimulation
2.5. Cytotoxicity Determination
2.6. Griess Reaction and ELISA
2.7. Flow Cytometry
2.8. Protein Extraction, Quantification and Western Blotting
2.9. Immunocytochemistry
2.10. Statistics
3. Results
3.1. (Poly)phenol Profiles and Effects of In Vitro Digestion
3.2. GIB Fractions Were Not Cytotoxic at Physiological Concentrations
3.3. GIB Fractions Richer in ET and Derivatives Inhibited Microglial Pro-Inflammatory Activation
3.4. J19 GIB Fraction Induced the Release of IL-10, an Important Microglial Anti-Inflammatory Marker
3.5. J19 GIB Fraction Inhibited MAPK Signaling by Repressing p38 Phosphorylation at Thr180/Tyr182
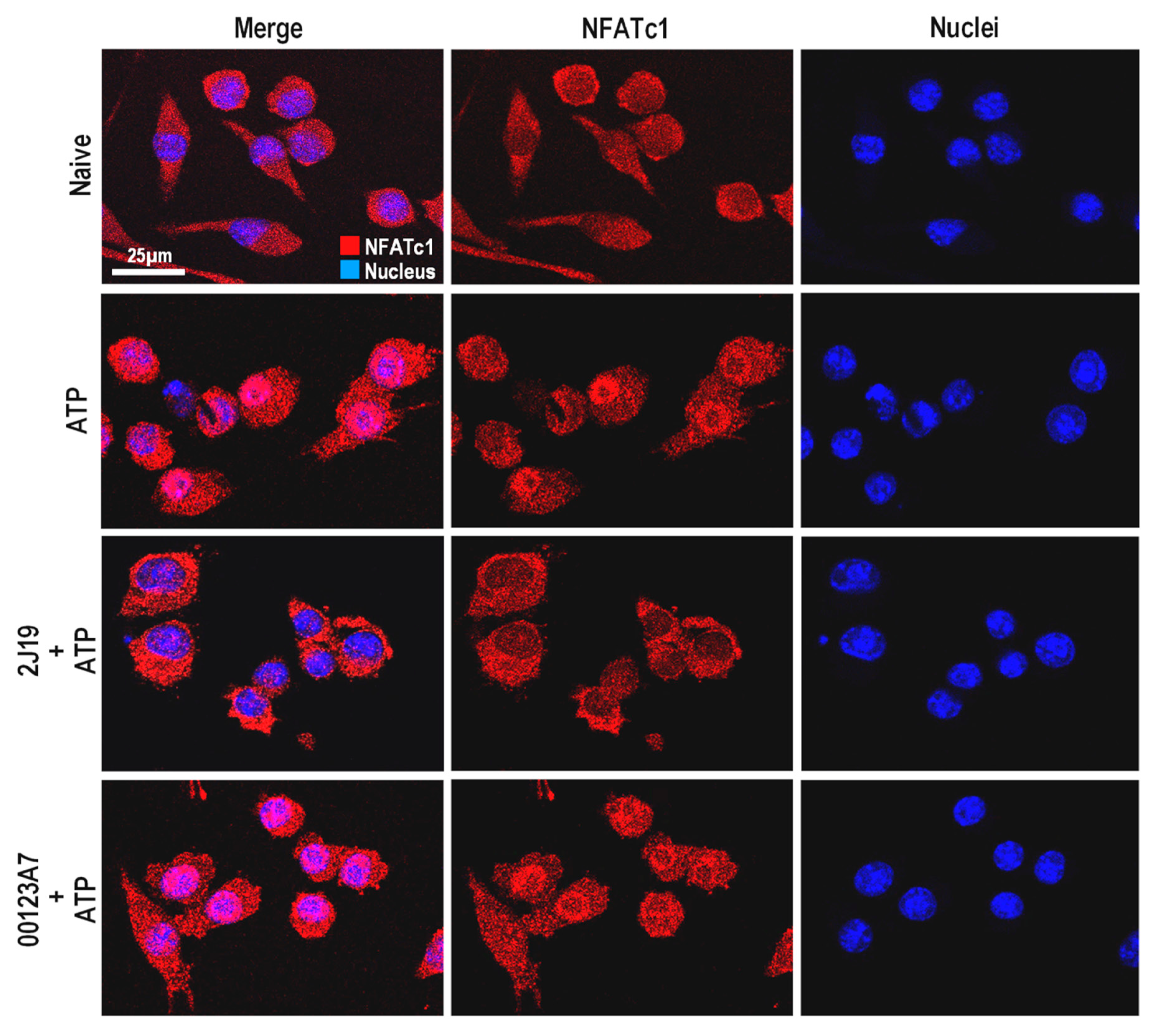
3.6. NFATc1 Nuclear Translocation Is Inhibited by 2J19
3.7. J19 GIB Fraction Represses LPS-Mediated NF-κB Activation by Reducing p65 Nuclear Translocation and Phosphorylation at Ser536
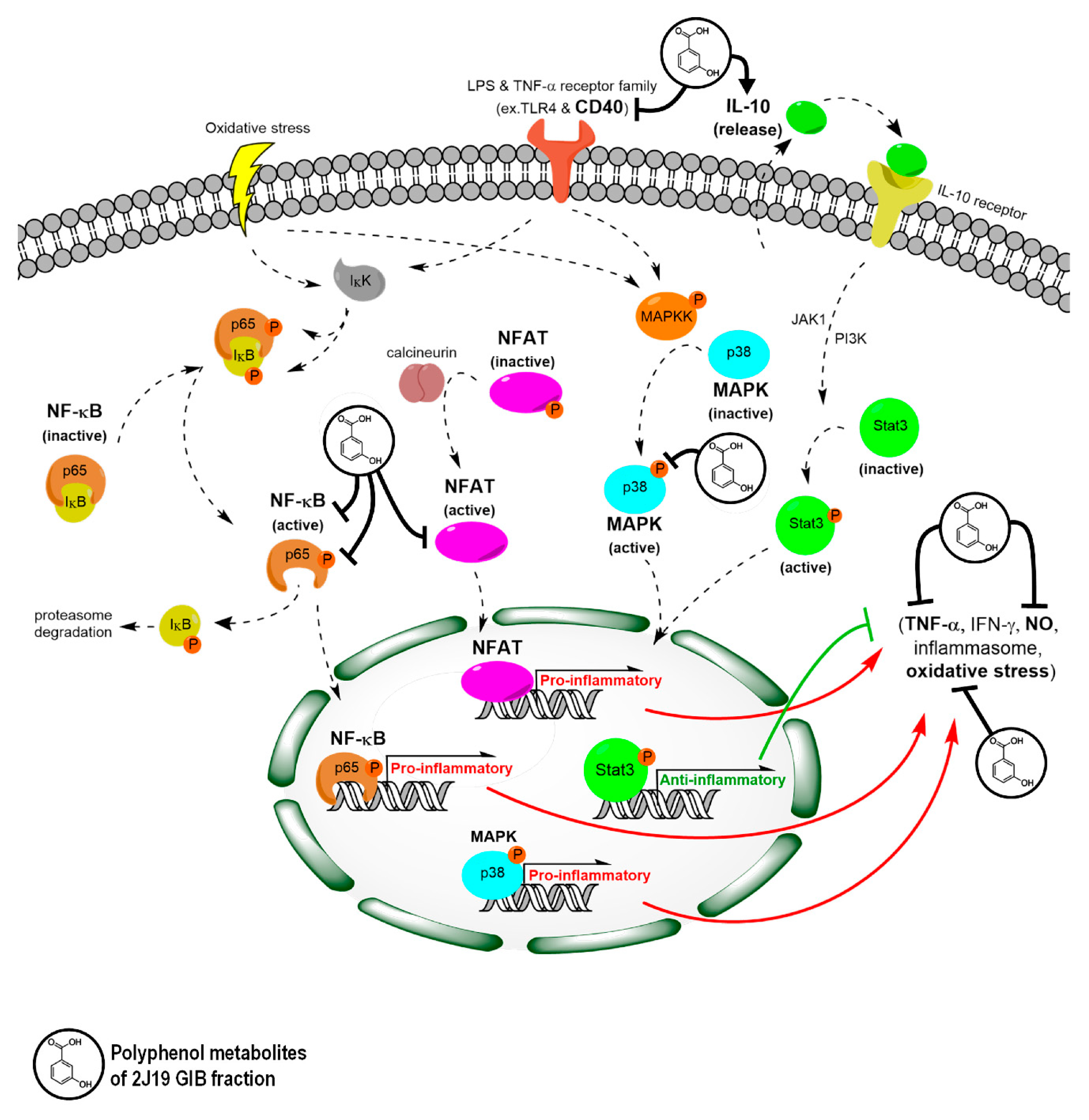
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| CNS | Central Nervous System |
| DHE | Dihydroethidium |
| EA | Ellagic acid |
| ELISA | Enzyme linked immuno sorbent assay |
| ET | Ellagitannins |
| FcγR | Fragment crystallizable-gamma receptor |
| FDW | Fruit dry weight |
| GAE | Gallic Acid Equivalent |
| GIB | Gastro-intestinal bioaccessible |
| MAPK | Mitogen-Activated Protein Kinase |
| NFATc1 | Nuclear Factor of Activated T cells, cytoplasmic 1 |
| NF-κB | Nuclear Factor kappa of activated B cells |
References
- Fernandes, A.; Ribeiro, A.R.; Monteiro, M.; Garcia, G.; Vaz, A.R.; Brites, D.; Rita, A.; Brites, D.; Vaz, A.R.; Brites, D. Secretome from SH-SY5Y APPSwe cells trigger time-dependent CHME3 microglia activation phenotypes, ultimately leading to miR-21 exosome shuttling. Biochimie 2018, 155, 67–82. [Google Scholar] [CrossRef] [PubMed]
- Lehnardt, S. Innate immunity and neuroinflammation in the CNS: The role of microglia in toll-like receptor-mediated neuronal injury. Glia 2010, 58, 253–263. [Google Scholar] [CrossRef] [PubMed]
- Cherry, J.D.; Olschowka, J.A.; O’Banion, M.K. Neuroinflammation and M2 microglia: The good, the bad, and the inflamed. J. Neuroinflamm. 2014, 11, 15. [Google Scholar] [CrossRef] [PubMed]
- Graeber, M.B.; Streit, W.J. Microglia: Biology and pathology. Acta Neuropathol. 2010, 119, 89–105. [Google Scholar] [CrossRef] [PubMed]
- Bachiller, S.; Jiménez-Ferrer, I.; Paulus, A.; Yang, Y.; Swanberg, M.; Deierborg, T.; Boza-Serrano, A. Microglia in neurological diseases: A road map to brain-disease dependent-inflammatory response. Front. Cell. Neurosci. 2018, 12. [Google Scholar] [CrossRef]
- Kettenmann, H.; Kirchhoff, F.; Verkhratsky, A. Microglia: New Roles for the Synaptic Stripper. Neuron 2013, 77, 10–18. [Google Scholar] [CrossRef]
- Guzman-Martinez, L.; Maccioni, R.B.; Andrade, V.; Navarrete, L.P.; Pastor, M.G.; Ramos-Escobar, N. Neuroinflammation as a common feature of neurodegenerative disorders. Front. Pharmacol. 2019, 10, 1008. [Google Scholar] [CrossRef]
- Wu, X.; Schauss, A.G. Mitigation of inflammation with foods. J. Agric. Food Chem. 2012, 60, 6703–6717. [Google Scholar] [CrossRef]
- Pimpao, R.C.; Dew, T.; Oliveira, P.B.; Williamson, G.; Ferreira, R.B.; Santos, C.N. Analysis of phenolic compounds in Portuguese wild and commercial berries after multienzyme hydrolysis. J. Agric. Food Chem. 2013, 61, 4053–4062. [Google Scholar] [CrossRef]
- Garcia, G.; Nanni, S.; Figueira, I.; Ivanov, I.; McDougall, G.J.; Stewart, D.; Ferreira, R.B.; Pinto, P.; Silva, R.F.M.; Brites, D.; et al. Bioaccessible (poly)phenol metabolites from raspberry protect neural cells from oxidative stress and attenuate microglia activation. Food Chem. 2017, 215, 274–283. [Google Scholar] [CrossRef]
- Tall, J.M.; Seeram, N.P.; Zhao, C.; Nair, M.G.; Meyer, R.A.; Raja, S.N. Tart cherry anthocyanins suppress inflammation-induced pain behavior in rat. Behav. Brain Resolut. 2004, 153, 181–188. [Google Scholar] [CrossRef]
- Jean-Gilles, D.; Li, L.; Ma, H.; Yuan, T.; Chichester, C.O.; Seeram, N.P.; Chichester, C.O., 3rd; Seeram, N.P. Anti-inflammatory effects of polyphenolic-enriched red raspberry extract in an antigen-induced arthritis rat model. J. Agric. Food Chem. 2012, 60, 5755–5762. [Google Scholar] [CrossRef] [PubMed]
- Galli, R.L.; Carey, A.N.; Luskin, K.A.; Bielinski, D.F.; Shukitt-Hale, B. Red raspberries can improve motor function in aged rats. J. Berry Res. 2016, 6, 97–103. [Google Scholar] [CrossRef]
- Williams, R.J.; Spencer, J.P. Flavonoids, cognition, and dementia: Actions, mechanisms, and potential therapeutic utility for Alzheimer disease. Free Radic. Biol. Med. 2012, 52, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.; Johnson, S.L.; Liu, W.; Dasilva, N.A.; Meschwitz, S.; Dain, J.A.; Seeram, N.P. Evaluation of polyphenol anthocyanin-enriched extracts of blackberry, black raspberry, blueberry, cranberry, red raspberry, and strawberry for free radical scavenging, reactive carbonyl species trapping, anti-glycation, anti-β-amyloid aggregation, and mic. Int. J. Mol. Sci. 2018, 19, 461. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Yao, G.D.; Lu, L.W.; Song, X.Y.; Lin, B.; Wang, X.B.; Huang, X.X.; Song, S.J. Neolignans from Red Raspberry (Rubus idaeus L.) Exhibit Enantioselective Neuroprotective Effects against H2O2-Induced Oxidative Injury in SH-SY5Y Cells. J. Agric. Food Chem. 2018, 66, 11390–11397. [Google Scholar] [CrossRef]
- Williamson, G.; Kay, C.D.; Crozier, A. The Bioavailability, Transport, and Bioactivity of Dietary Flavonoids: A Review from a Historical Perspective. Compr. Rev. Food Sci. Food Saf. 2018, 17, 1054–1112. [Google Scholar] [CrossRef]
- Wang, H.; Nair, M.G.; Strasburg, G.M.; Chang, Y.C.; Booren, A.M.; Gray, J.I.; DeWitt, D.L. Antioxidant and antiinflammatory activities of anthocyanins and their aglycon, cyanidin, from tart cherries. J. Nat. Prod. 1999, 62, 294–296. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, D.; Wu, Y.; Wang, D.; Wei, Y.; Wu, J.; Ji, B. Stability and absorption of anthocyanins from blueberries subjected to a simulated digestion process. Int. J. Food Sci. Nutr. 2014, 65, 440–448. [Google Scholar] [CrossRef]
- Murota, K.; Nakamura, Y.; Uehara, M. Flavonoid metabolism: The interaction of metabolites and gut microbiota. Biosci. Biotechnol. Biochem. 2018, 82, 600–610. [Google Scholar] [CrossRef]
- McDougall, G.J.; Allwood, J.W.; Pereira-Caro, G.; Brown, E.M.; Latimer, C.; Dobson, G.; Stewart, D.; Ternan, N.G.; Lawther, R.; O’Connor, G.; et al. The composition of potentially bioactive triterpenoid glycosides in red raspberry is influenced by tissue, extraction procedure and genotype. Food Funct. 2017, 8, 3469–3479. [Google Scholar] [CrossRef] [PubMed]
- Tavares, L.; Figueira, I.; MacEdo, D.; McDougall, G.J.; Leitão, M.C.; Vieira, H.L.A.; Stewart, D.; Alves, P.M.; Ferreira, R.B.; Santos, C.N. Neuroprotective effect of blackberry (Rubus sp.) polyphenols is potentiated after simulated gastrointestinal digestion. Food Chem. 2012, 131, 1443–1452. [Google Scholar] [CrossRef]
- McDougall, G.J.; Dobson, P.; Smith, P.; Blake, A.; Stewart, D. Assessing potential bioavailability of raspberry anthocyanins using an in vitro digestion system. J. Agric. Food Chem. 2005, 53, 5896–5904. [Google Scholar] [CrossRef] [PubMed]
- Coates, E.M.; Popa, G.; Gill, C.I.; McCann, M.J.; McDougall, G.J.; Stewart, D.; Rowland, I. Colon-available raspberry polyphenols exhibit anti-cancer effects on in vitro models of colon cancer. J. Carcinog. 2007, 6, 4. [Google Scholar] [CrossRef] [PubMed]
- Figueira, I.; Garcia, G.; Pimpão, R.C.; Terrasso, A.P.; Costa, I.; Almeida, A.F.; Tavares, L.; Pais, T.F.; Pinto, P.; Ventura, M.R.; et al. Polyphenols journey through blood-brain barrier towards neuronal protection. Sci. Rep. 2017, 7, 11456. [Google Scholar] [CrossRef] [PubMed]
- Remberg, S.F.; Soønsteby, A.; Aaby, K.; Heide, O.M. Influence of postflowering temperature on fruit size and chemical composition of glen ample raspberry (Rubus Idaeus L.). J. Agric. Food Chem. 2010, 58, 9120–9128. [Google Scholar] [CrossRef] [PubMed]
- McDougall, G.J.; Stewart, D. The inhibitory effects of berry polyphenols on digestive enzymes. BioFactors 2005, 23, 189–195. [Google Scholar] [CrossRef]
- Daniel, E.M.; Ratnayake, S.; Kinstle, T.; Stoner, G.D. The effects of pH and rat intestinal contents on the liberation of ellagic acid from purified and crude ellagitannins. J. Nat. Prod. 1991, 54, 946–952. [Google Scholar] [CrossRef]
- Manach, C.; Williamson, G.; Morand, C.; Scalbert, A.; Remesy, C. Bioavailability and bioefficacy of polyphenols in humans. I. Review of 97 bioavailability studies. Am J Clin Nutr 2005, 81, 230S–242S. [Google Scholar] [CrossRef]
- Pimpão, R.C.; Ventura, M.R.; Ferreira, R.B.; Williamson, G.; Santos, C.N. Phenolic sulfates as new and highly abundant metabolites in human plasma after ingestion of a mixed berry fruit purée. Br. J. Nutr. 2015, 113, 454–463. [Google Scholar] [CrossRef]
- Sun, Y.; Ma, J.; Li, D.; Li, P.; Zhou, X.; Li, Y.; He, Z.; Qin, L.; Liang, L.; Luo, X. Interleukin-10 inhibits interleukin-1β production and inflammasome activation of microglia in epileptic seizures. J. Neuroinflamm. 2019, 16, 13. [Google Scholar] [CrossRef] [PubMed]
- Magrone, T.; Spagnoletta, A.; Bizzoca, A.; Russo, M.A.; Jirillo, E.; Gennarini, G. Polyphenol effects on splenic cytokine response in post-weaning contactin 1-overexpressing transgenic mice. Molecules 2019, 24, 2205. [Google Scholar] [CrossRef] [PubMed]
- Molina, N.; Bolin, A.P.; Otton, R. Green tea polyphenols change the profile of inflammatory cytokine release from lymphocytes of obese and lean rats and protect against oxidative damage. Int. Immunopharmacol. 2015, 28, 985–996. [Google Scholar] [CrossRef] [PubMed]
- Cuenda, A.; Rousseau, S. p38 MAP-Kinases pathway regulation, function and role in human diseases. Biochim. Biophys. Acta Mol. Cell Res. 2007, 1773, 1358–1375. [Google Scholar] [CrossRef] [PubMed]
- Nagamoto-Combs, K.; Combs, C.K. Microglial Phenotype Is Regulated by Activity of the Transcription Factor, NFAT (Nuclear Factor of Activated T Cells). J. Neurosci. 2010, 30, 9641–9646. [Google Scholar] [CrossRef]
- Ferrari, D.; Stroh, C.; Schulze-Osthoff, K. P2 × 7/P2Z purinoreceptor-mediated activation of transcription factor NFAT in microglial cells. J. Biol. Chem. 1999, 274, 13205–13210. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.C. NF-κB signaling in inflammation. Signal Transduct. Target. Ther. 2017, 2, 1–9. [Google Scholar] [CrossRef]
- Sasaki, C.Y.; Barberi, T.J.; Ghosh, P.; Longo, D.L. Phosphorylation of RelA/p65 on serine 536 defines an I{kappa}B{alpha}-independent NF-{kappa}B pathway. J. Biol. Chem. 2005, 280, 34538–34547. [Google Scholar] [CrossRef]
- Ruiz, P.A.; Haller, D. Functional diversity of flavonoids in the inhibition of the proinflammatory NF-κB, IRF, and Akt signaling pathways in murine intestinal epithelial cells. J. Nutr. 2006, 136, 664–671. [Google Scholar] [CrossRef]
- Wheeler, D.S.; Catravas, J.D.; Odoms, K.; Denenberg, A.; Malhotra, V.; Wong, H.R. Epigallocatechin-3-gallate, a Green Tea-Derived Polyphenol, Inhibits IL-1β-Dependent Proinflammatory Signal Transduction in Cultured Respiratory Epithelial Cells. J. Nutr. 2004, 134, 1039–1044. [Google Scholar] [CrossRef]
- Farbood, Y.; Sarkaki, A.; Dolatshahi, M.; Taqhi Mansouri, S.M.; Khodadadi, A. Ellagic Acid Protects the Brain Against 6-Hydroxydopamine Induced Neuroinflammation in a Rat Model of Parkinson’s Disease. Basic Clin. Neurosci. 2015, 6, 83–89. [Google Scholar] [PubMed]
- Ávila-Gálvez, M.A.; Giménez-Bastida, J.A.; González-Sarrías, A.; Espín, J.C. Tissue deconjugation of urolithin A glucuronide to free urolithin A in systemic inflammation. Food Funct. 2019, 10, 3135–3141. [Google Scholar] [CrossRef] [PubMed]
- Cassidy, A.; Rogers, G.; Peterson, J.J.; Dwyer, J.T.; Lin, H.; Jacques, P.F. Higher dietary anthocyanin and flavonol intakes are associated with anti-inflammatory effects in a population of US adults. Am. J. Clin. Nutr. 2015, 102, 172–181. [Google Scholar] [CrossRef] [PubMed]
- Xie, Z.; Wei, M.; Morgan, T.E.; Fabrizio, P.; Han, D.; Finch, C.E.; Longo, V.D. Peroxynitrite Mediates Neurotoxicity of Amyloid β-Peptide 1-42- and Lipopolysaccharide-Activated Microglia. J. Neurosci. 2002, 22, 3484–3492. [Google Scholar] [CrossRef] [PubMed]
- Kipanyula, M.J.; Kimaro, W.H.; Etet, P.F.S. The Emerging Roles of the Calcineurin-Nuclear Factor of Activated T-Lymphocytes Pathway in Nervous System Functions and Diseases. J. Aging Res. 2016, 2016, 5081021. [Google Scholar] [CrossRef]
- Saijo, K.; Crotti, A.; Glass, C.K. Regulation of microglia activation and deactivation by nuclear receptors. Glia 2013, 61, 104–111. [Google Scholar] [CrossRef]
- Rojanathammanee, L.; Floden, A.M.; Manocha, G.D.; Combs, C.K. Attenuation of microglial activation in a mouse model of Alzheimer’s disease via NFAT inhibition. J. Neuroinflamm. 2015, 12, 42. [Google Scholar] [CrossRef]
- Zusso, M.; Lunardi, V.; Franceschini, D.; Pagetta, A.; Lo, R.; Stifani, S.; Frigo, A.C.; Giusti, P.; Moro, S. Ciprofloxacin and levofloxacin attenuate microglia inflammatory response via TLR4/NF-kB pathway. J. Neuroinflamm. 2019, 16, 148. [Google Scholar] [CrossRef]
- Chang, Y.; Chen, W.F.; Lin, K.H.; Hsieh, C.Y.; Chou, D.S.; Lin, L.J.; Sheu, J.R.; Chang, C.C. Novel bioactivity of ellagic acid in inhibiting human platelet activation. Evid.-Based Complement. Altern. Med. 2013, 2013, 595128. [Google Scholar] [CrossRef]
- Zhong, L.; Liu, H.; Zhang, W.; Liu, X.; Jiang, B.; Fei, H.; Sun, Z. Ellagic acid ameliorates learning and memory impairment in APP/PS1 transgenic mice via inhibition of β-amyloid production and tau hyperphosphorylation. Exp. Ther. Med. 2018, 16, 4951–4958. [Google Scholar] [CrossRef]
- Gupta, P.; Mohammad, T.; Khan, P.; Alajmi, M.F.; Hussain, A.; Rehman, M.T.; Hassan, M.I. Evaluation of ellagic acid as an inhibitor of sphingosine kinase 1: A targeted approach towards anticancer therapy. Biomed. Pharmacother. 2019, 118, 109245. [Google Scholar] [CrossRef] [PubMed]

 ; cells treated with 2J19 prior to LPS stimulation are denoted as
; cells treated with 2J19 prior to LPS stimulation are denoted as  and cells treated with 00123A7 prior to LPS stimulation are denoted as
and cells treated with 00123A7 prior to LPS stimulation are denoted as  . (B) Comparison of the p38 phosphorylation (Thr180/Tyr182) ratios between each type of pre-treatment at 0 and 30 min of LPS stimulation (mean ± SEM; n = 3). Statistical differences between 2J19 pre-treated cells and the LPS-stimulated control cells are denoted as ** p < 0.01.
. (B) Comparison of the p38 phosphorylation (Thr180/Tyr182) ratios between each type of pre-treatment at 0 and 30 min of LPS stimulation (mean ± SEM; n = 3). Statistical differences between 2J19 pre-treated cells and the LPS-stimulated control cells are denoted as ** p < 0.01.
 ; cells treated with 2J19 prior to LPS stimulation are denoted as
; cells treated with 2J19 prior to LPS stimulation are denoted as  and cells treated with 00123A7 prior to LPS stimulation are denoted as
and cells treated with 00123A7 prior to LPS stimulation are denoted as  . (B) Comparison of the p38 phosphorylation (Thr180/Tyr182) ratios between each type of pre-treatment at 0 and 30 min of LPS stimulation (mean ± SEM; n = 3). Statistical differences between 2J19 pre-treated cells and the LPS-stimulated control cells are denoted as ** p < 0.01.
. (B) Comparison of the p38 phosphorylation (Thr180/Tyr182) ratios between each type of pre-treatment at 0 and 30 min of LPS stimulation (mean ± SEM; n = 3). Statistical differences between 2J19 pre-treated cells and the LPS-stimulated control cells are denoted as ** p < 0.01.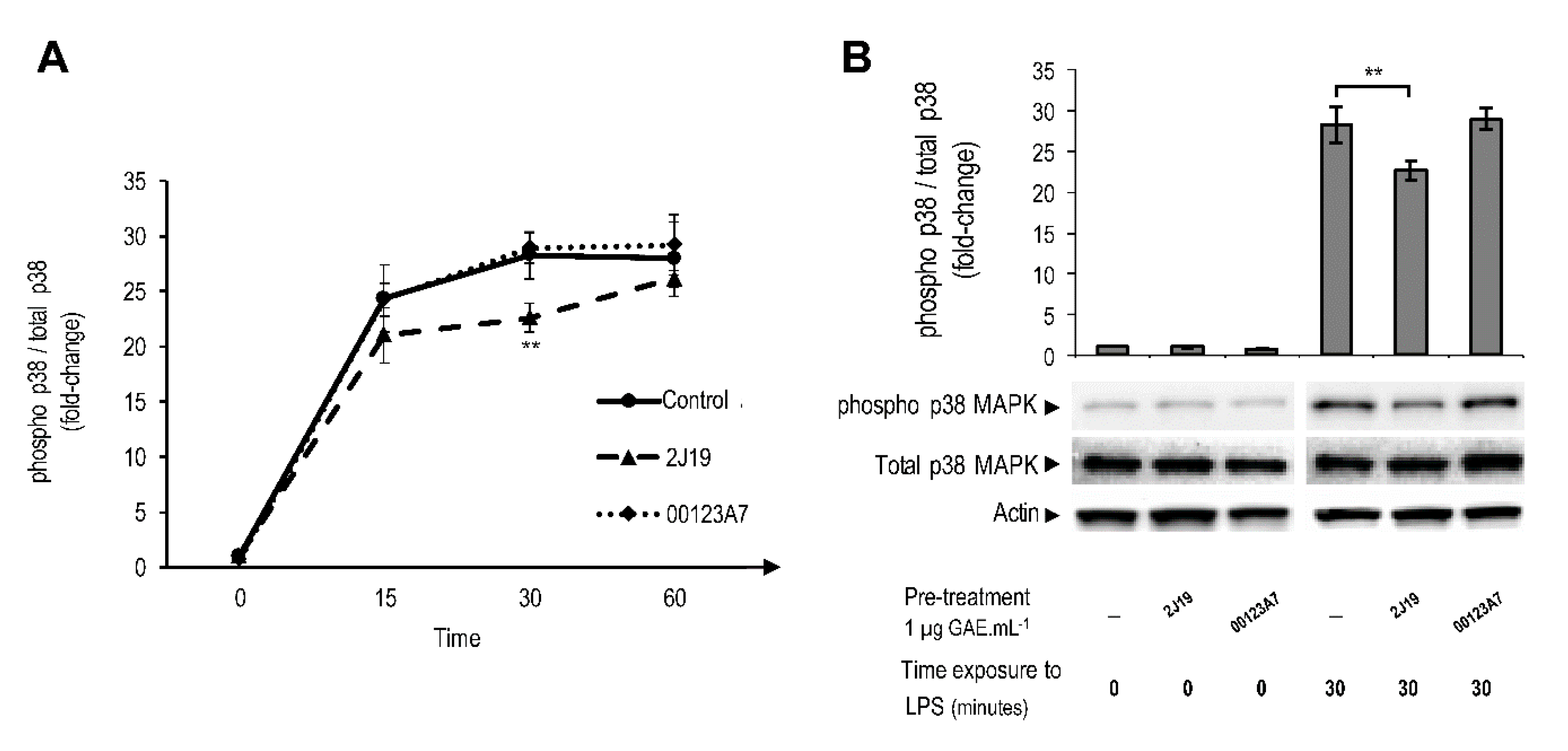
 ; cells treated with 2J19 prior to LPS stimulation are denoted by
; cells treated with 2J19 prior to LPS stimulation are denoted by  , and cells treated with 00123A7 prior to LPS stimulation are denoted by
, and cells treated with 00123A7 prior to LPS stimulation are denoted by  . (C) IκB-α protein levels and (E) p65 phosphorylation (Ser536) ratios for each GIB fraction at 0 and 30 min of LPS stimulation (mean ± SEM; n = 3). Statistical differences vs respective timepoint control are denoted as ** p < 0.01; “n.s.” means not statistically different.
. (C) IκB-α protein levels and (E) p65 phosphorylation (Ser536) ratios for each GIB fraction at 0 and 30 min of LPS stimulation (mean ± SEM; n = 3). Statistical differences vs respective timepoint control are denoted as ** p < 0.01; “n.s.” means not statistically different.
 ; cells treated with 2J19 prior to LPS stimulation are denoted by
; cells treated with 2J19 prior to LPS stimulation are denoted by  , and cells treated with 00123A7 prior to LPS stimulation are denoted by
, and cells treated with 00123A7 prior to LPS stimulation are denoted by  . (C) IκB-α protein levels and (E) p65 phosphorylation (Ser536) ratios for each GIB fraction at 0 and 30 min of LPS stimulation (mean ± SEM; n = 3). Statistical differences vs respective timepoint control are denoted as ** p < 0.01; “n.s.” means not statistically different.
. (C) IκB-α protein levels and (E) p65 phosphorylation (Ser536) ratios for each GIB fraction at 0 and 30 min of LPS stimulation (mean ± SEM; n = 3). Statistical differences vs respective timepoint control are denoted as ** p < 0.01; “n.s.” means not statistically different.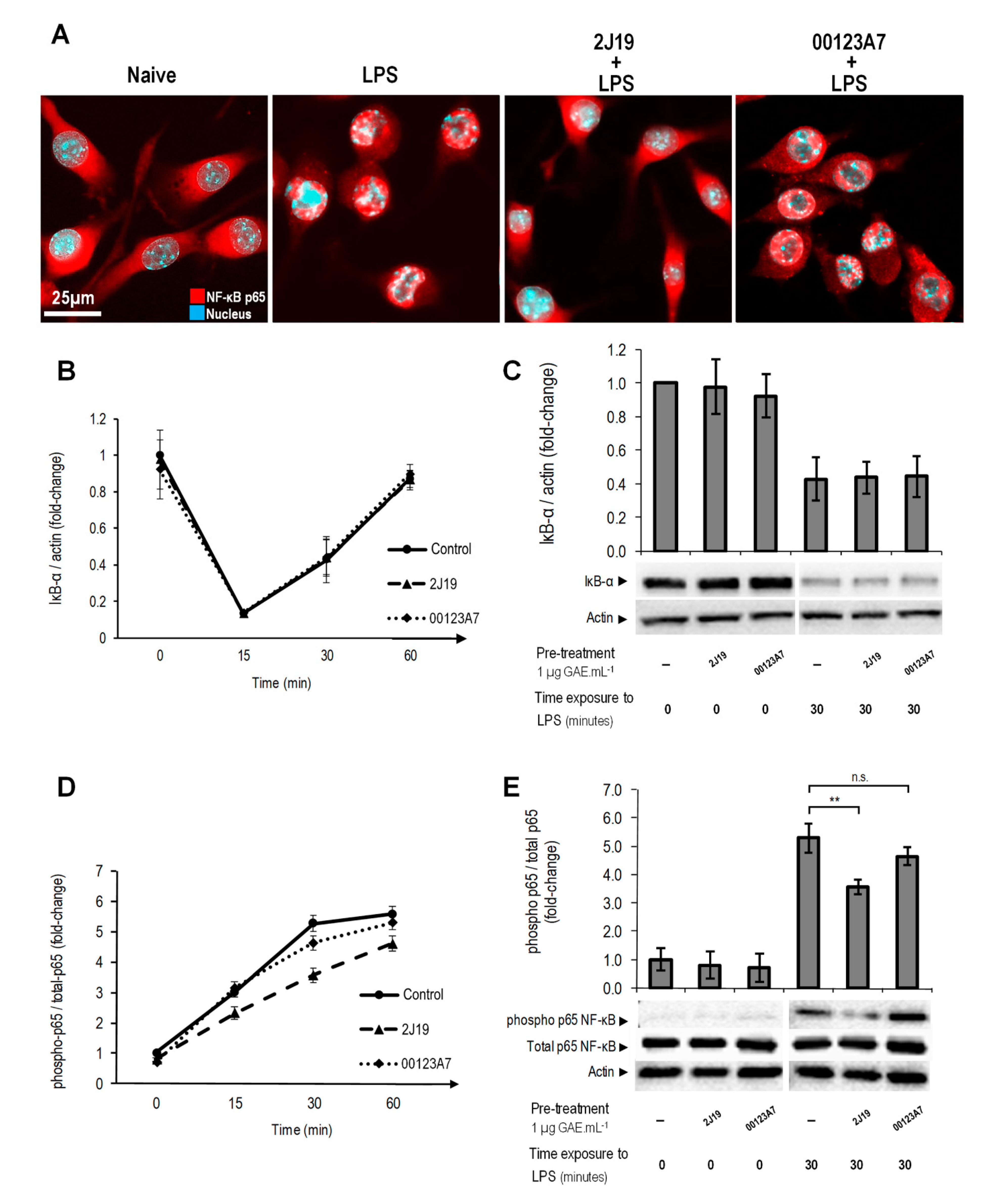
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Garcia, G.; Pais, T.F.; Pinto, P.; Dobson, G.; McDougall, G.J.; Stewart, D.; Santos, C.N. Bioaccessible Raspberry Extracts Enriched in Ellagitannins and Ellagic Acid Derivatives Have Anti-Neuroinflammatory Properties. Antioxidants 2020, 9, 970. https://doi.org/10.3390/antiox9100970
Garcia G, Pais TF, Pinto P, Dobson G, McDougall GJ, Stewart D, Santos CN. Bioaccessible Raspberry Extracts Enriched in Ellagitannins and Ellagic Acid Derivatives Have Anti-Neuroinflammatory Properties. Antioxidants. 2020; 9(10):970. https://doi.org/10.3390/antiox9100970
Chicago/Turabian StyleGarcia, Gonçalo, Teresa Faria Pais, Paula Pinto, Gary Dobson, Gordon J. McDougall, Derek Stewart, and Cláudia Nunes Santos. 2020. "Bioaccessible Raspberry Extracts Enriched in Ellagitannins and Ellagic Acid Derivatives Have Anti-Neuroinflammatory Properties" Antioxidants 9, no. 10: 970. https://doi.org/10.3390/antiox9100970
APA StyleGarcia, G., Pais, T. F., Pinto, P., Dobson, G., McDougall, G. J., Stewart, D., & Santos, C. N. (2020). Bioaccessible Raspberry Extracts Enriched in Ellagitannins and Ellagic Acid Derivatives Have Anti-Neuroinflammatory Properties. Antioxidants, 9(10), 970. https://doi.org/10.3390/antiox9100970