Safety Evaluation of α-Lipoic Acid Supplementation: A Systematic Review and Meta-Analysis of Randomized Placebo-Controlled Clinical Studies
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Study Selection Criteria
2.3. Data Extraction
2.4. Quality Assessment
2.5. Data Synthesis
2.6. Additional Analysis
2.7. Publication Biases
3. Results
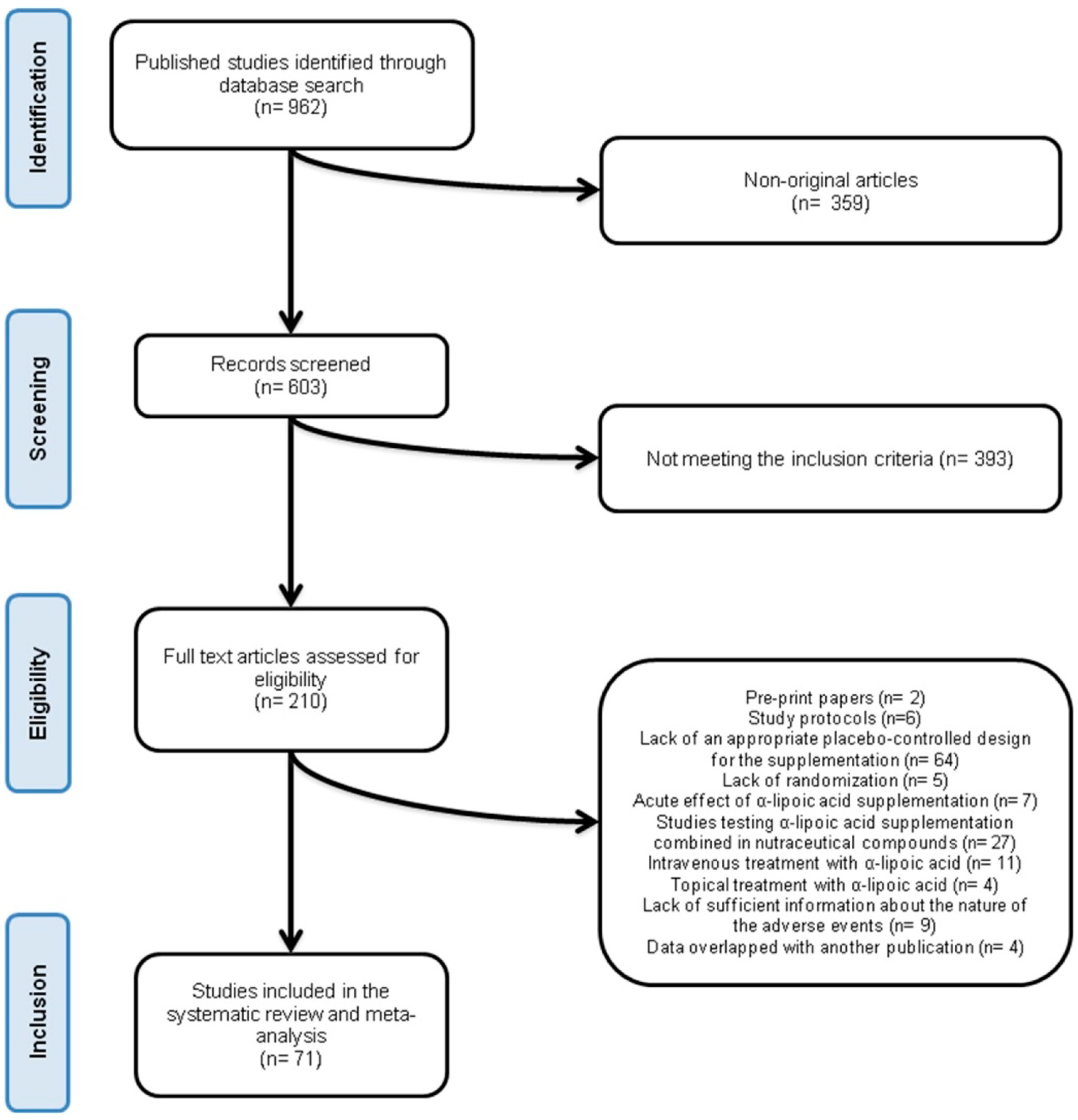
3.1. Flow and Characteristics of the Included Studies
3.2. Risk of Bias Assessment
3.3. Primary Outcomes
3.3.1. Hypoglycaemic Episodes
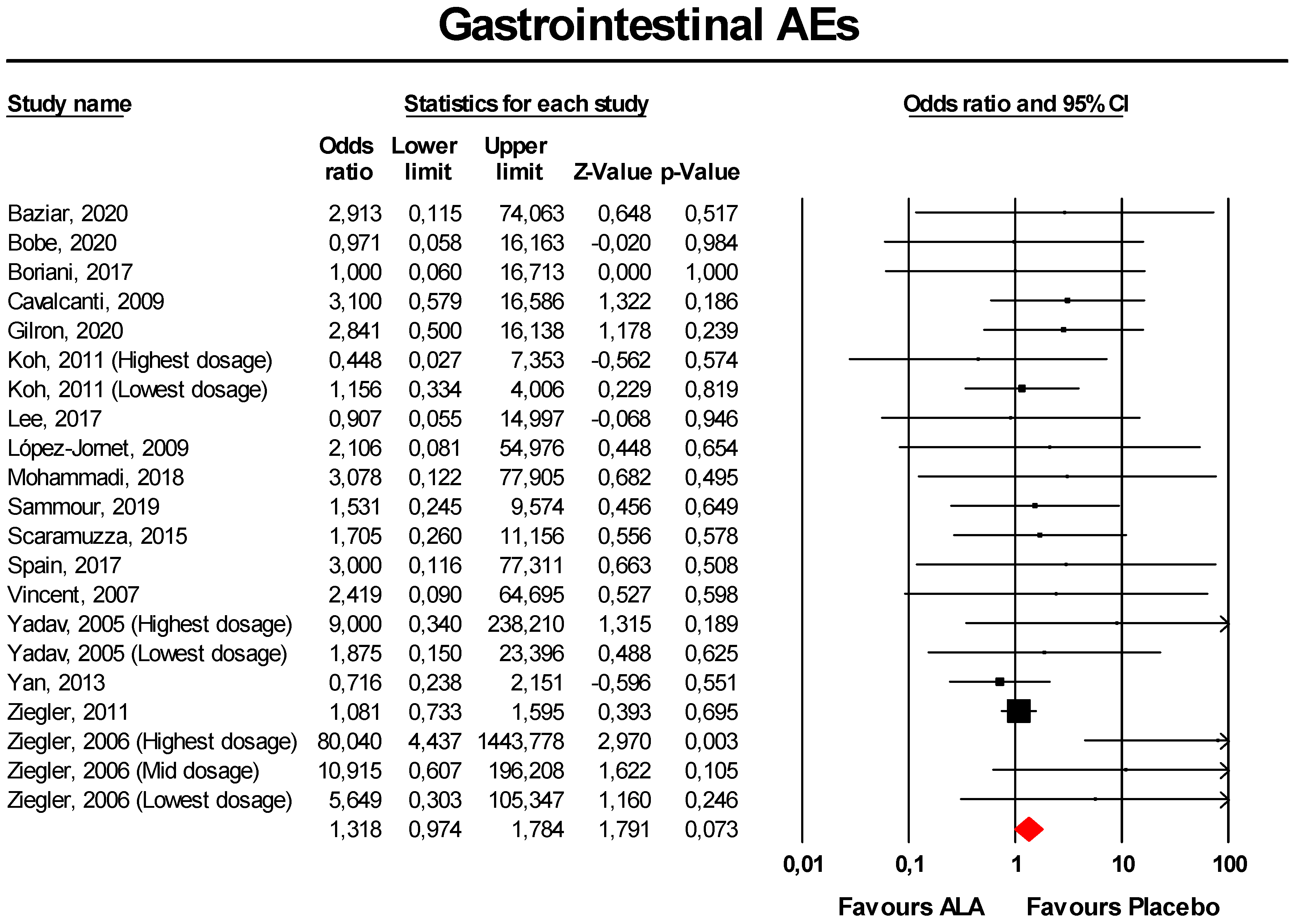
3.3.2. Gastrointestinal AEs
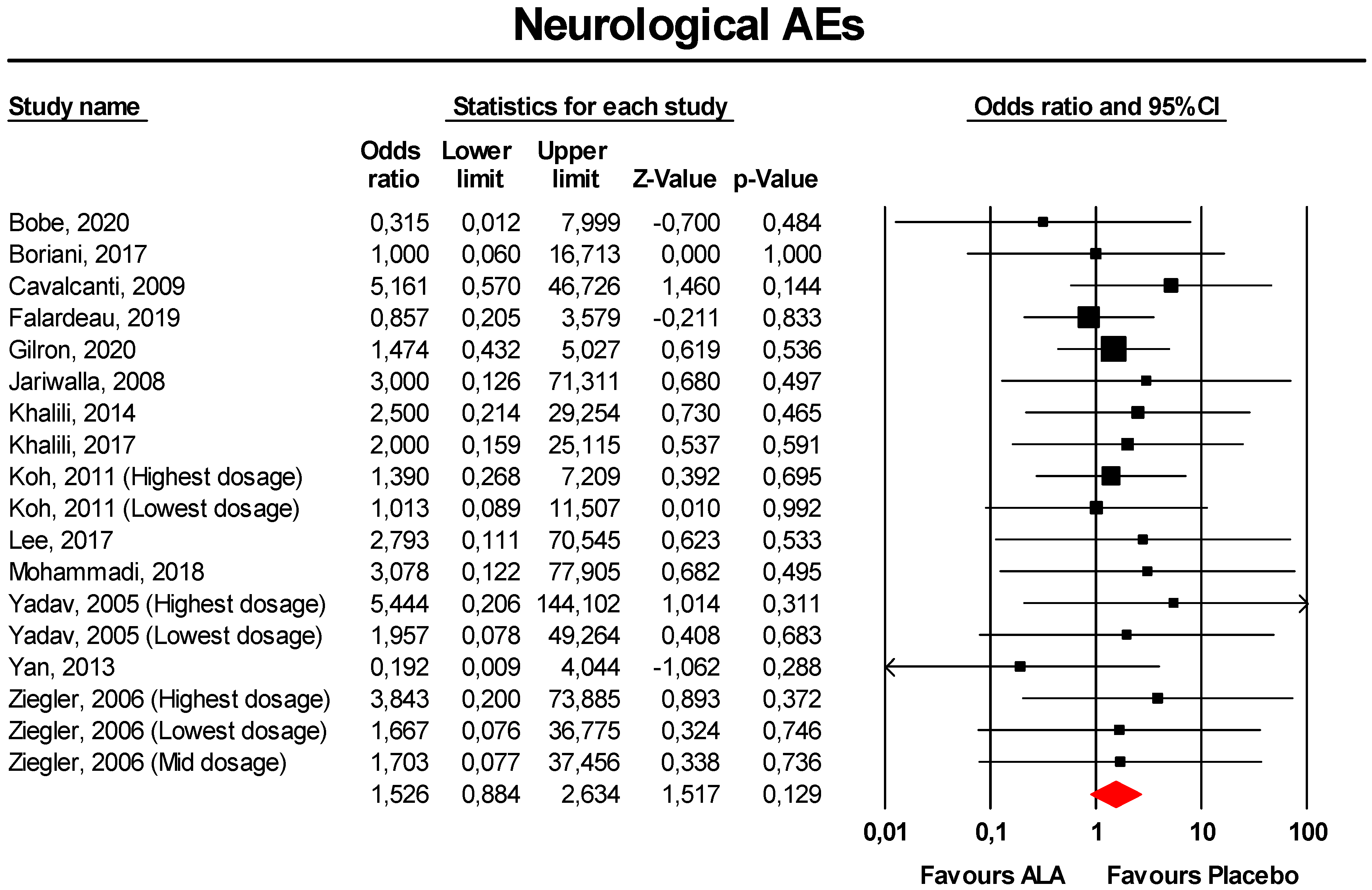
3.3.3. Neurological AEs
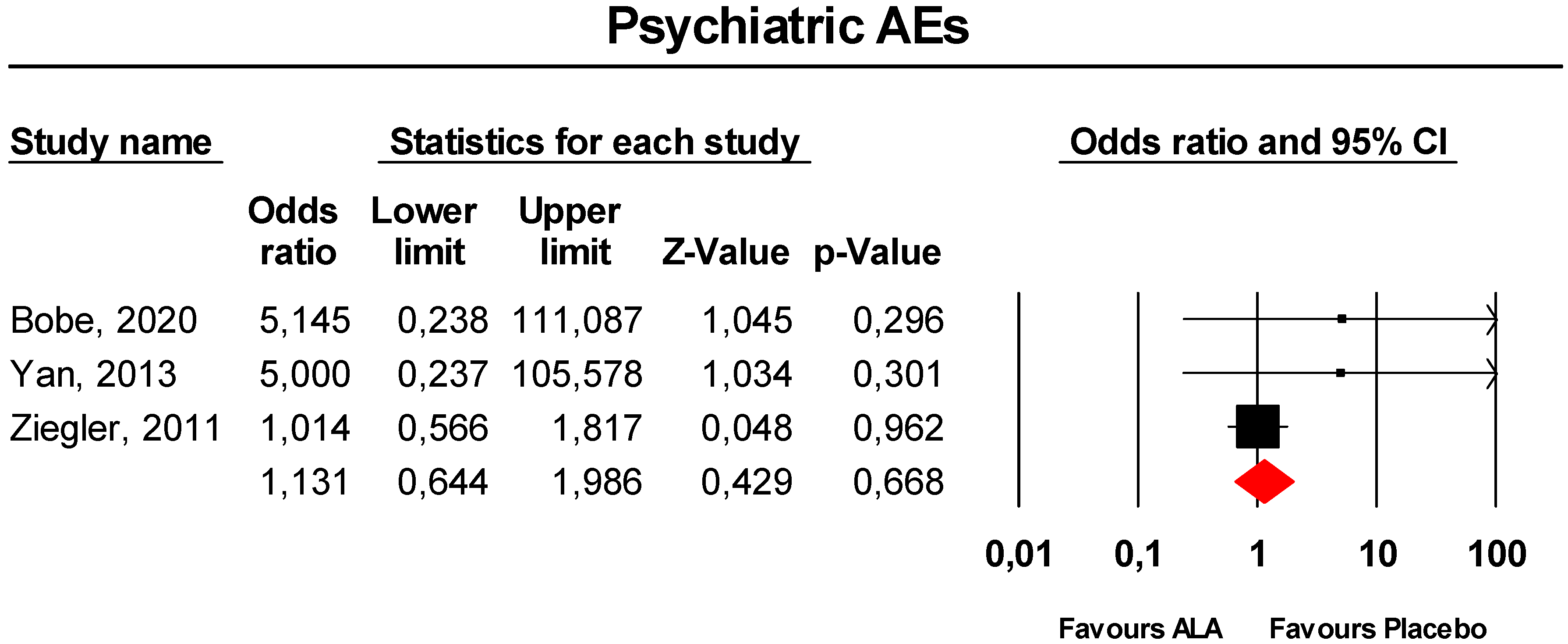
3.3.4. Psychiatric Disorders
3.3.5. Musculoskeletal AEs
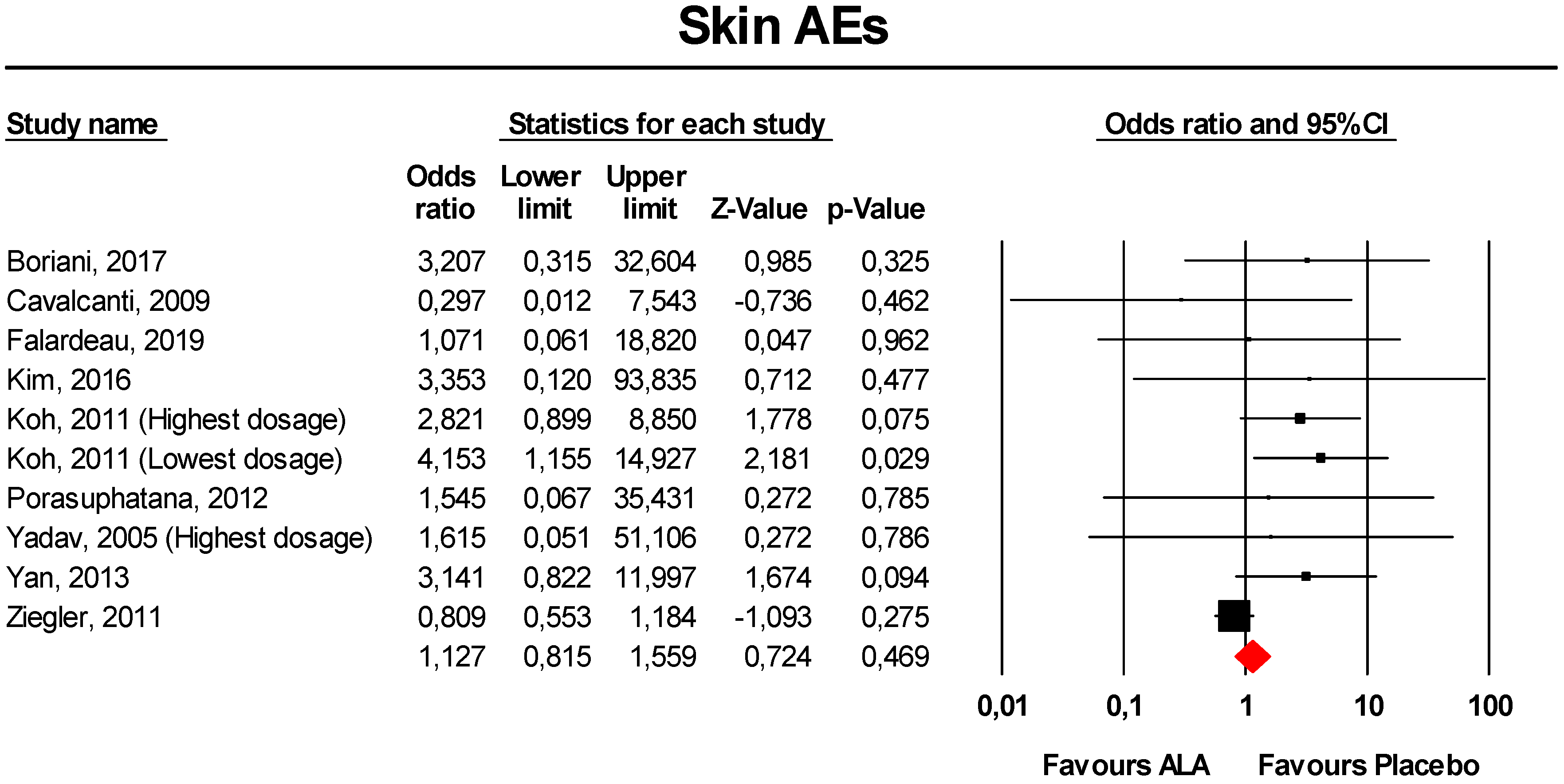
3.3.6. Skin AEs
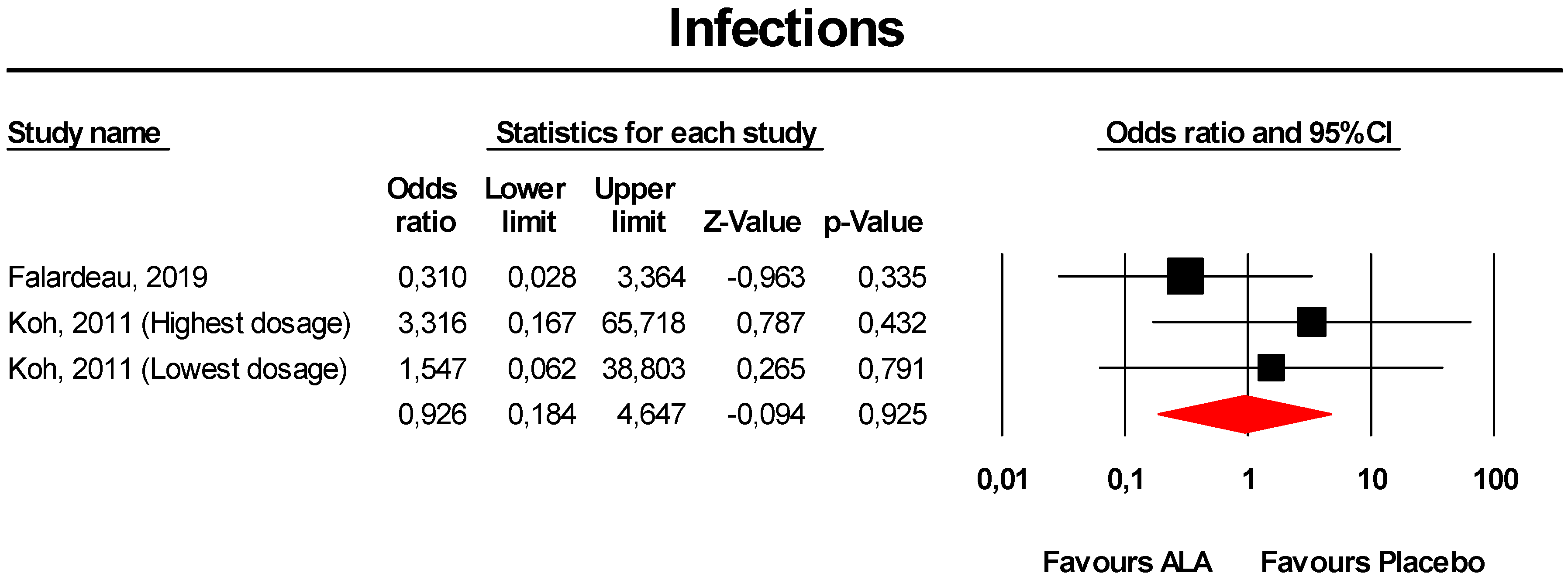
3.3.7. Infections
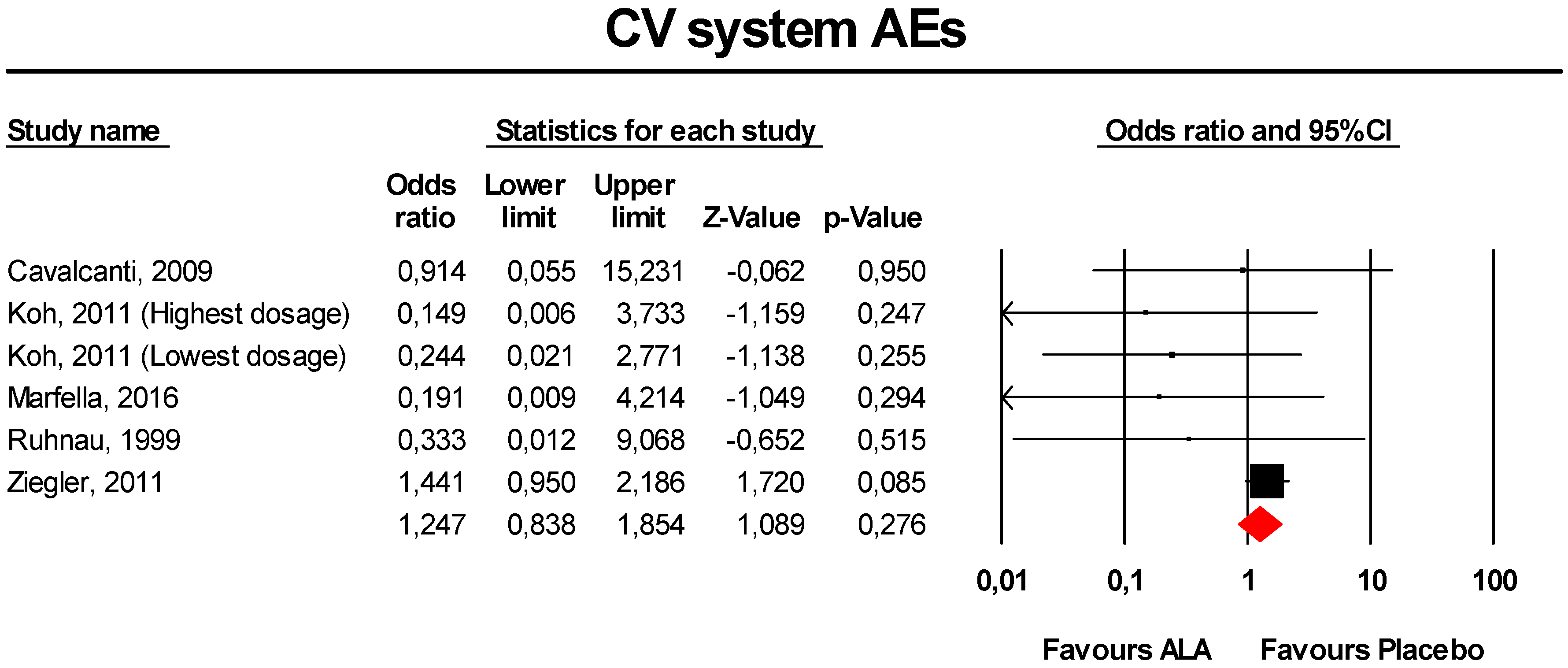
3.3.8. CV System AEs
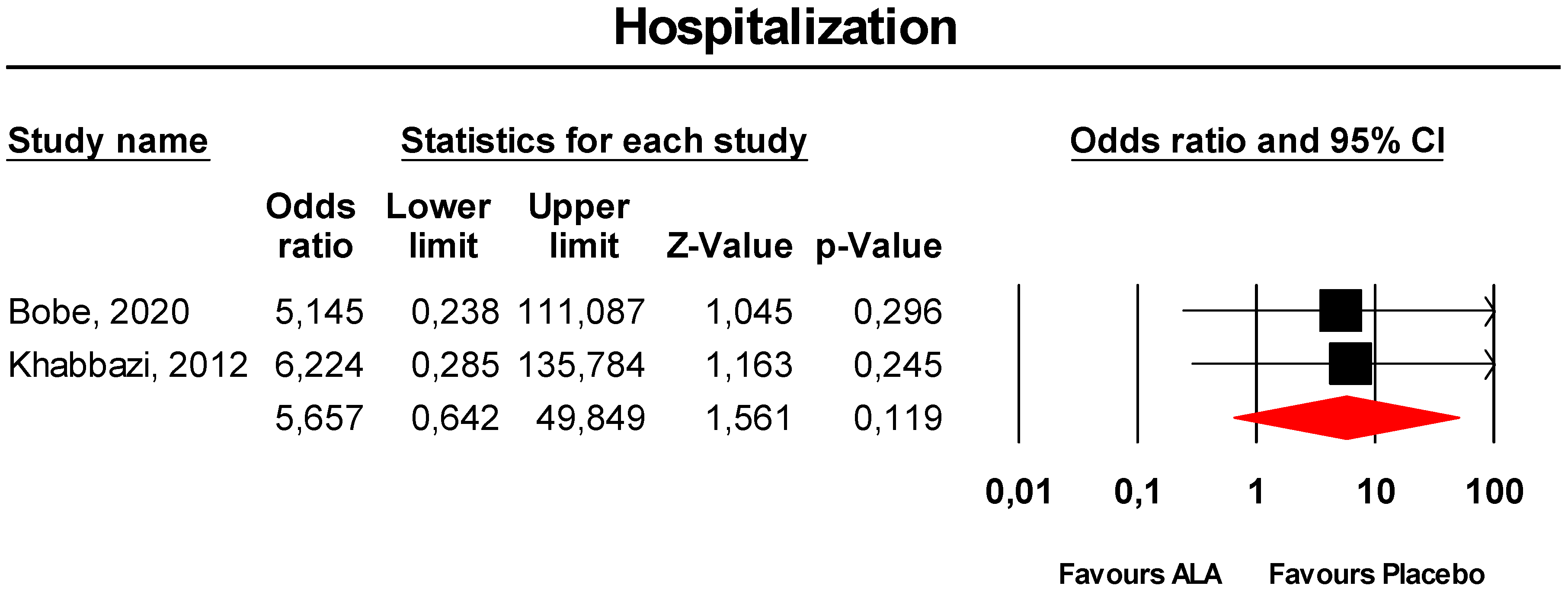
3.3.9. Hospitalisation
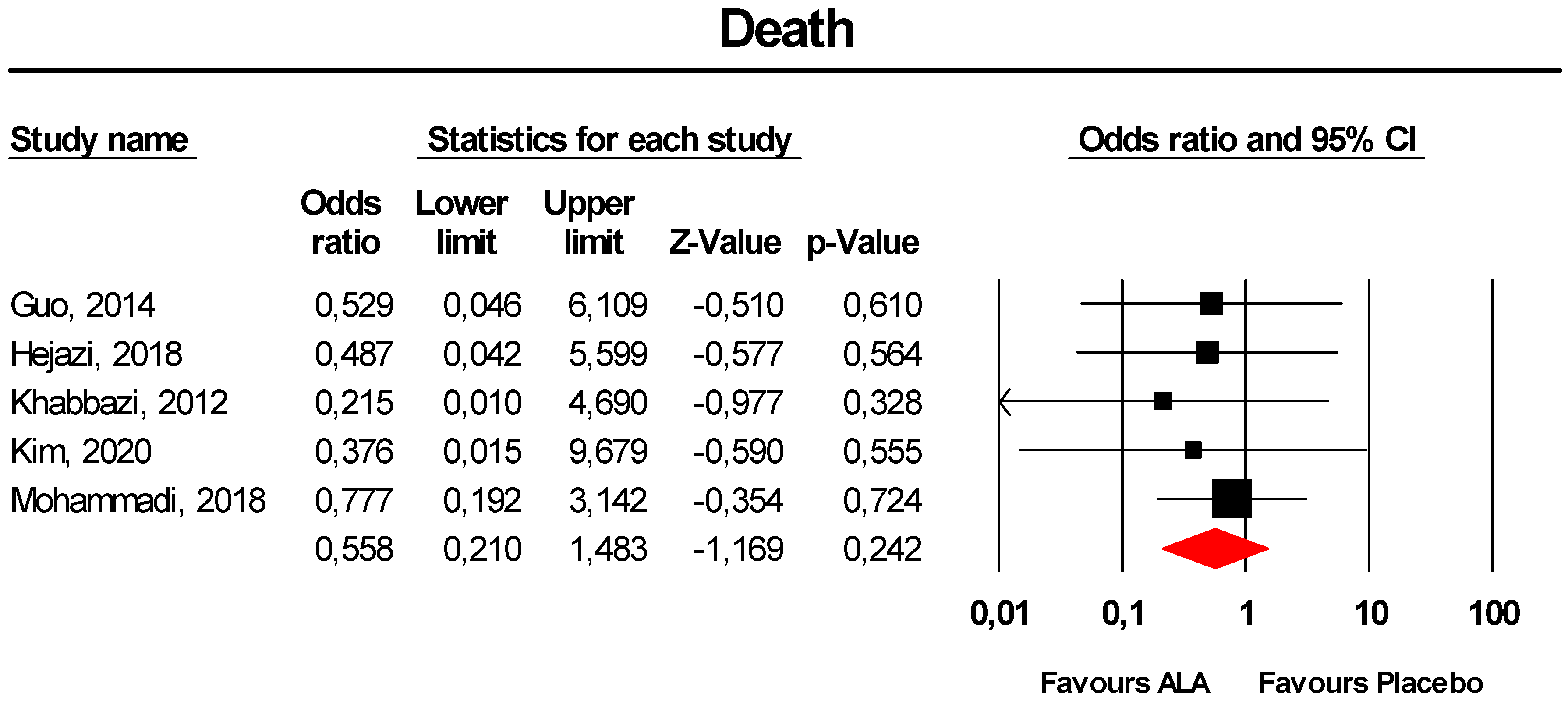
3.3.10. Death
3.4. Additional Analyses
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Tibullo, D.; Li Volti, G.; Giallongo, C.; Grasso, S.; Tomassoni, D.; Anfuso, C.D.; Lupo, G.; Amenta, F.; Avola, R.; Bramanti, V. Biochemical and clinical relevance of alpha lipoic acid: Antioxidant and anti-inflammatory activity, molecular pathways and therapeutic potential. Inflamm. Res. 2017, 66, 947–959. [Google Scholar] [CrossRef] [PubMed]
- Biewenga, G.P.; Haenen, G.R.; Bast, A. The pharmacology of the antioxidant lipoic acid. Gen. Pharmacol. 1997, 29, 315–331. [Google Scholar] [CrossRef]
- Pershadsingh, H.A. Alpha-lipoic acid: Physiologic mechanisms and indications for the treatment of metabolic syndrome. Expert Opin. Investig. Drugs 2007, 16, 291–302. [Google Scholar] [CrossRef] [PubMed]
- Rahmanabadi, A.; Mahboob, S.; Amirkhizi, F.; Hosseinpour-Arjmand, S.; Ebrahimi-Mameghani, M. Oral α-lipoic acid supplementation in patients with non-alcoholic fatty liver disease: Effects on adipokines and liver histology features. Food Funct. 2019, 10, 4941–4952. [Google Scholar] [CrossRef] [PubMed]
- Hosseinpour-Arjmand, S.; Amirkhizi, F.; Ebrahimi-Mameghani, M. The effect of alpha-lipoic acid on inflammatory markers and body composition in obese patients with non-alcoholic fatty liver disease: A randomized, double-blind, placebo-controlled trial. J. Clin. Pharm. Ther. 2019, 44, 258–267. [Google Scholar] [CrossRef] [PubMed]
- Yukina, M.; Nuralieva, N.; Solovyev, M.; Troshina, E.; Vasilyev, E. Insulin autoimmune syndrome. Endocrinol. Diabetes Metab. Case Rep. 2020, 2020, 19–0159. [Google Scholar] [CrossRef]
- Izzo, V.; Greco, C.; Corradini, D.; Infante, M.; Staltari, M.T.; Romano, M.; Bellia, A.; Lauro, D.; Uccioli, L. Insulin autoimmune syndrome in an Argentine woman taking α-lipoic acid: A case report and review of the literature. SAGE Open Med. Case Rep. 2018, 6, 2050313X18819601. [Google Scholar] [CrossRef]
- Bresciani, E.; Bussi, A.; Bazzigaluppi, E.; Balestrieri, G. Insulin autoimmune syndrome induced by α-lipoic acid in a Caucasian woman: Case report. Diabetes Care. 2011, 34, e146. [Google Scholar] [CrossRef]
- Gatti, M.; Ippoliti, I.; Poluzzi, E.; Antonazzo, I.C.; Moro, P.A.; Moretti, U.; Menniti-Ippolito, F.; Mazzanti, G.; De Ponti, F.; Raschi, E. Assessment of adverse reactions to α-lipoic acid containing dietary supplements through spontaneous reporting systems. Clin. Nutr. 2020. E-pub ahead of print. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. BMJ 2009, 339, b2535. [Google Scholar] [CrossRef]
- Higgins, J.; Green, S. Cochrane Handbook for Systematic Reviews of Interventions; Version 5.0. 2. 2009; John Wiley and Sons Ltd: Chichester, UK, 2010. [Google Scholar]
- Fogacci, F.; Ferri, N.; Toth, P.P.; Ruscica, M.; Corsini, A.; Cicero, A.F.G. Efficacy and Safety of Mipomersen: A Systematic Review and Meta-Analysis of Randomized Clinical Trials. Drugs 2019, 79, 751–766. [Google Scholar] [CrossRef] [PubMed]
- Guyatt, G.H.; Oxman, A.D.; Vist, G.E.; Kunz, R.; Falck-Ytter, Y.; Alonso-Coello, P.; Schünemann, H.J.; GRADE Working Group. GRADE: An emerging consensus on rating quality of evidence and strength of recommendations. BMJ 2008, 336, 924–926. [Google Scholar] [CrossRef]
- Borenstein, M.; Hedges, L.; Higgins, J.; Rothstein, H. Comprehensive Meta-Analysis Version 3; Biostat: Englewood, NJ, USA, 2005; Volume 104. [Google Scholar]
- Higgins, J.P.T.; Thomas, J.; Chandler, J.; Cumpston, M.; Li, T.; Page, M.J.; Welch, V.A. Cochrane Handbook for Systematic Reviews of Interventions; Version 6.0 (updated July 2019); Cochrane: London, UK, 2019; Available online: www.training.cochrane.org/handbook (accessed on 15 September 2020).
- Melsen, W.G.; Bootsma, M.C.; Rovers, M.M.; Bonten, M.J. The effects of clinical and statistical heterogeneity on the predictive values of results from meta-analyses. Clin. Microbiol. Infect. 2004, 20, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Haenszel, W.; Hon, N.B. Statistical approaches to the study of cancer with particular reference to case registers. J. Chronic. Dis. 1956, 4, 589–599. [Google Scholar] [CrossRef]
- Fogacci, S.; Fogacci, F.; Banach, M.; Michos, E.D.; Hernandez, A.V.; Lip, G.Y.H.; Blaha, M.J.; Toth, P.P.; Borghi, C.; Cicero, A.F.G. Lipid and Blood Pressure Meta-analysis Collaboration (LBPMC) Group. Vitamin D supplementation and incident preeclampsia: A systematic review and meta-analysis of randomized clinical trials. Clin. Nutr. 2020, 39, 1742–1752. [Google Scholar] [CrossRef]
- Fogacci, F.; Banach, M.; Mikhailidis, D.P.; Bruckert, E.; Toth, P.P.; Watts, G.F.; Reiner, Ž.; Mancini, J.; Rizzo, M.; Mitchenko, O.; et al. Safety of red yeast rice supplementation: A systematic review and meta-analysis of randomized controlled trials. Pharmacol. Res. 2019, 143, 1–16. [Google Scholar] [CrossRef]
- Ahmadi, A.; Mazooji, N.; Roozbeh, J.; Mazloom, Z.; Hasanzade, J. Effect of alpha-lipoic acid and vitamin E supplementation on oxidative stress, inflammation, and malnutrition in hemodialysis patients. Iran J. Kidney Dis. 2013, 7, 461–467. [Google Scholar] [PubMed]
- Ansar, H.; Mazloom, Z.; Kazemi, F.; Hejazi, N. Effect of alpha-lipoic acid on blood glucose, insulin resistance and glutathione peroxidase of type 2 diabetic patients. Saudi Med. J. 2011, 32, 584–588. [Google Scholar]
- Aslfalah, H.; Jamilian, M.; Rafiei, F.; Khosrowbeygi, A. Reduction in maternal serum values of glucose and gamma-glutamyltransferase after supplementation with alpha-lipoic acid in women with gestational diabetes mellitus. J. Obstet. Gynaecol. Res. 2019, 45, 313–317. [Google Scholar] [CrossRef]
- Aslfalah, H.; Jamilian, M.; Khosrowbeygi, A. Elevation of the adiponectin/leptin ratio in women with gestational diabetes mellitus after supplementation with alpha-lipoic acid. Gynecol. Endocrinol. 2019, 35, 271–275. [Google Scholar] [CrossRef]
- Baumgartner, S.; Mensink, R.P.; Haenen, G.R.; Bast, A.; Binder, C.J.; Bekers, O.; Husche, C.; Lütjohann, D.; Plat, J. The effects of vitamin E or lipoic acid supplementation on oxyphytosterols in subjects with elevated oxidative stress: A randomized trial. Sci. Rep. 2017, 7, 15288. [Google Scholar] [CrossRef] [PubMed]
- Baziar, N.; Nasli-Esfahani, E.; Djafarian, K.; Qorbani, M.; Hedayati, M.; Mishani, M.A.; Faghfoori, Z.; Ahmaripour, N.; Hosseini, S. The Beneficial Effects of Alpha Lipoic Acid Supplementation on Lp-PLA2 Mass and Its Distribution between HDL and apoB-Containing Lipoproteins in Type 2 Diabetic Patients: A Randomized, Double-Blind, Placebo-Controlled Trial. Oxid. Med. Cell Longev. 2020, 2020, 5850865. [Google Scholar] [CrossRef] [PubMed]
- Bobe, G.; Michels, A.J.; Zhang, W.J.; Purnell, J.Q.; Woffendin, C.; Pereira, C.; Vita, J.A.; Thomas, N.O.; Traber, M.G.; Frei, B.; et al. A Randomized Controlled Trial of Long-Term (R)-α-Lipoic Acid Supplementation Promotes Weight Loss in Overweight or Obese Adults without Altering Baseline Elevated Plasma Triglyceride Concentrations. J. Nutr. 2020, 150, 2336–2345. [Google Scholar] [CrossRef] [PubMed]
- Boriani, F.; Granchi, D.; Roatti, G.; Merlini, L.; Sabattini, T.; Baldini, N. Alpha-lipoic Acid After Median Nerve Decompression at the Carpal Tunnel: A Randomized Controlled Trial. J. Hand Surg. Am. 2017, 42, 236–242. [Google Scholar] [CrossRef] [PubMed]
- Carbone, M.; Pentenero, M.; Carrozzo, M.; Ippolito, A.; Gandolfo, S. Lack of efficacy of alpha-lipoic acid in burning mouth syndrome: A double-blind, randomized, placebo-controlled study. Eur. J. Pain 2009, 13, 492–496. [Google Scholar] [CrossRef]
- Cavalcanti, D.R.; da Silveira, F.R. Alpha lipoic acid in burning mouth syndrome—A randomized double-blind placebo-controlled trial. J. Oral Pathol. Med. 2009, 38, 254–261. [Google Scholar] [CrossRef]
- Durastanti, V.; Tinelli, E.; Di Rezze, S.; Berardelli, A.; Millefiorini, E. Alpha lipoic acid as add-on therapy to subcutaneous interferon β-1a for relapsing-remitting multiple sclerosis: A pilot study. IJABPT 2016, 7, 336–341. [Google Scholar]
- El Amrousy, D.; El-Afify, D. Effects of alpha lipoic acid as a supplement in obese children and adolescents. Cytokine 2020, 130, 155084. [Google Scholar] [CrossRef]
- Falardeau, J.; Fryman, A.; Wanchu, R.; Marracci, G.H.; Mass, M.; Wooliscroft, L.; Bourdette, D.N.; Murchison, C.F.; Hills, W.L.; Yadav, V. Oral lipoic acid as a treatment for acute optic neuritis: A blinded, placebo controlled randomized trial. Mult. Scler. J. Exp. Transl. Clin. 2019, 5, 2055217319850193. [Google Scholar] [CrossRef]
- Femiano, F.; Scully, C. Burning mouth syndrome (BMS): Double blind controlled study of alpha-lipoic acid (thioctic acid) therapy. J. Oral Pathol. Med. 2002, 31, 267–269. [Google Scholar] [CrossRef] [PubMed]
- Georgakouli, K.; Fatouros, I.G.; Fragkos, A.; Tzatzakis, T.; Deli, C.K.; Papanikolaou, K.; Koutedakis, Y.; Jamurtas, A.Z. Exercise and Redox Status Responses Following Alpha-Lipoic Acid Supplementation in G6PD Deficient Individuals. Antioxidants 2018, 7, 162. [Google Scholar] [CrossRef]
- Gianturco, V.; Bellomo, A.; D’Ottavio, E.; Formosa, V.; Iori, A.; Mancinella, M.; Troisi, G.; Marigliano, V. Impact of therapy with alpha-lipoic acid (ALA) on the oxidative stress in the controlled NIDDM: A possible preventive way against the organ dysfunction? Arch. Gerontol. Geriatr. 2009, 49, 129–133. [Google Scholar] [CrossRef] [PubMed]
- Gilron, I.; Robb, S.; Tu, D.; Holden, R.; Towheed, T.; Ziegler, D.; Wang, L.; Milev, R.; Gray, C. Double-blind, randomized, placebo-controlled crossover trial of alpha-lipoic acid for the treatment of fibromyalgia pain: The IMPALA trial. Pain 2020. [Google Scholar] [CrossRef] [PubMed]
- Gosselin, L.E.; Chrapowitzky, L.; Rideout, T.C. Metabolic effects of α-lipoic acid supplementation in pre-diabetics: A randomized, placebo-controlled pilot study. Food Funct. 2019, 10, 5732–5738. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Jones, D.; Palmer, J.L.; Forman, A.; Dakhil, S.R.; Velasco, M.R.; Weiss, M.; Gilman, P.; Mills, G.M.; Noga, S.J.; et al. Oral alpha-lipoic acid to prevent chemotherapy-induced peripheral neuropathy: A randomized, double-blind, placebo-controlled trial. Support. Care Cancer 2014, 22, 1223–1231. [Google Scholar] [CrossRef] [PubMed]
- Haghighian, H.K.; Haidari, F.; Mohammadi-Asl, J.; Dadfar, M. Randomized, triple-blind, placebo-controlled clinical trial examining the effects of alpha-lipoic acid supplement on the spermatogram and seminal oxidative stress in infertile men. Fertil. Steril. 2015, 104, 318–324. [Google Scholar] [CrossRef]
- Hejazi, N.; Mazloom, Z.; Zand, F.; Rezaianzadeh, A.; Nikandish, R. The Beneficial Effects of α-Lipoic Acid in Critically Ill Patients: A Prospective, Randomized, Double-Blind, Placebo-Controlled Trial. Asian J. Anesthesiol. 2018, 56, 45–55. [Google Scholar]
- Huang, E.A.; Gitelman, S.E. The effect of oral alpha-lipoic acid on oxidative stress in adolescents with type 1 diabetes mellitus. Pediatr. Diabetes 2008, 9, 69–73. [Google Scholar] [CrossRef]
- Huerta, A.E.; Prieto-Hontoria, P.L.; Sáinz, N.; Martínez, J.A.; Moreno-Aliaga, M.J. Supplementation with α-Lipoic Acid Alone or in Combination with Eicosapentaenoic Acid Modulates the Inflammatory Status of Healthy Overweight or Obese Women Consuming an Energy-Restricted Diet. J. Nutr. 2016, 146. [Google Scholar] [CrossRef]
- Huerta, A.E.; Navas-Carretero, S.; Prieto-Hontoria, P.L.; Martínez, J.A.; Moreno-Aliaga, M.J. Effects of α-lipoic acid and eicosapentaenoic acid in overweight and obese women during weight loss. Obesity 2015, 23, 313–321. [Google Scholar] [CrossRef]
- Jacob, S.; Ruus, P.; Hermann, R.; Tritschler, H.J.; Maerker, E.; Renn, W.; Augustin, H.J.; Dietze, G.J.; Rett, K. Oral administration of RAC-alpha-lipoic acid modulates insulin sensitivity in patients with type-2 diabetes mellitus: A placebo-controlled pilot trial. Free Radic. Biol. Med. 1999, 27, 309–314. [Google Scholar] [CrossRef]
- Jamshidi, K.; Abdollahzad, H.; Nachvak, M.; Rezaei, M.; Golpayegani, M.R.; Sharifi Zahabi, E. Effects of Alpha-Lipoic Acid Supplementation on Cardiovascular Disease Risk Factors in β-Thalassemia Major Patients: A Clinical Trial Crossover Study. J. Blood Med. 2020, 11, 131–139. [Google Scholar] [CrossRef]
- Jariwalla, R.J.; Lalezari, J.; Cenko, D.; Mansour, S.E.; Kumar, A.; Gangapurkar, B.; Nakamura, D. Restoration of blood total glutathione status and lymphocyte function following alpha-lipoic acid supplementation in patients with HIV infection. J. Altern. Complement Med. 2008, 14, 139–146. [Google Scholar] [CrossRef]
- Khabbazi, T.; Mahdavi, R.; Safa, J.; Pour-Abdollahi, P. Effects of alpha-lipoic acid supplementation on inflammation, oxidative stress, and serum lipid profile levels in patients with end-stage renal disease on hemodialysis. J. Ren. Nutr. 2012, 22, 244–250. [Google Scholar] [CrossRef] [PubMed]
- Khalili, M.; Soltani, M.; Moghadam, S.A.; Dehghan, P.; Azimi, A.; Abbaszadeh, O. Effect of alpha-lipoic acid on asymmetric dimethylarginine and disability in multiple sclerosis patients: A randomized clinical trial. Electron. Physician 2017, 9, 4899–4905. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Khalili, M.; Azimi, A.; Izadi, V.; Eghtesadi, S.; Mirshafiey, A.; Sahraian, M.A.; Motevalian, A.; Norouzi, A.; Sanoobar, M.; Eskandari, G.; et al. Does lipoic acid consumption affect the cytokine profile in multiple sclerosis patients: A double-blind, placebo-controlled, randomized clinical trial. Neuroimmunomodulation 2014, 21, 291–296. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.J.; Hunter, A.; Brucker, A.J.; Hahn, P.; Gehrs, K.; Patel, A.; Edwards, A.O.; Li, Y.; Khurana, R.N.; Nissim, I.; et al. Orally Administered Alpha Lipoic Acid as a Treatment for Geographic Atrophy: A Randomized Clinical Trial. Ophthalmol. Retin. 2020, 4, 889–898. [Google Scholar] [CrossRef] [PubMed]
- Kim, N.W.; Song, Y.M.; Kim, E.; Cho, H.S.; Cheon, K.A.; Kim, S.J.; Park, J.Y. Adjunctive α-lipoic acid reduces weight gain compared with placebo at 12 weeks in schizophrenic patients treated with atypical antipsychotics: A double-blind randomized placebo-controlled study. Int. Clin. Psychopharmacol. 2016, 31, 265–274. [Google Scholar] [CrossRef] [PubMed]
- Koh, E.H.; Lee, W.J.; Lee, S.A.; Kim, E.H.; Cho, E.H.; Jeong, E.; Kim, D.W.; Kim, M.S.; Park, J.Y.; Park, K.G.; et al. Effects of alpha-lipoic Acid on body weight in obese subjects. Am. J. Med. 2011, 124, 85.e1–85.e8. [Google Scholar] [CrossRef]
- Lampitella, A.; Rossi, E.; Carrino, F.; Griffo, P.; Carrino, R. Effetto dell’acido lipoico e della vitamina E sulla polineuropatia diabetica: Analisi statistica. Prog. Nutr. 2005, 7, 116–134. (In Italian) [Google Scholar]
- Lee, S.J.; Jeong, S.J.; Lee, Y.C.; Lee, Y.H.; Lee, J.E.; Kim, C.H.; Min, K.W.; Cha, B.Y. Effects of High-Dose α-Lipoic Acid on Heart Rate Variability of Type 2 Diabetes Mellitus Patients with Cardiac Autonomic Neuropathy in Korea. Diabetes Metab. J. 2017, 41, 275–283. [Google Scholar] [CrossRef] [PubMed]
- Loy, B.D.; Fling, B.W.; Horak, F.B.; Bourdette, D.N.; Spain, R.I. Effects of lipoic acid on walking performance, gait, and balance in secondary progressive multiple sclerosis. Complement Ther. Med. 2018, 41, 169–174. [Google Scholar] [CrossRef] [PubMed]
- López-D’alessandro, E.; Escovich, L. Combination of alpha lipoic acid and gabapentin, its efficacy in the treatment of Burning Mouth Syndrome: A randomized, double-blind, placebo controlled trial. Med. Oral Patol. Oral Cir. Bucal. 2011, 16, e635–e640. [Google Scholar] [CrossRef] [PubMed]
- López-Jornet, P.; Camacho-Alonso, F.; Leon-Espinosa, S. Efficacy of alpha lipoic acid in burning mouth syndrome: A randomized, placebo-treatment study. J. Oral Rehabil. 2009, 36, 52–57. [Google Scholar] [CrossRef] [PubMed]
- Magis, D.; Ambrosini, A.; Sándor, P.; Jacquy, J.; Laloux, P.; Schoenen, J. A randomized double-blind placebo-controlled trial of thioctic acid in migraine prophylaxis. Headache 2007, 47, 52–57. [Google Scholar] [CrossRef] [PubMed]
- Manning, P.J.; Sutherland, W.H.; Williams, S.M.; Walker, R.J.; Berry, E.A.; De Jong, S.A.; Ryalls, A.R. The effect of lipoic acid and vitamin E therapies in individuals with the metabolic syndrome. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 543–549. [Google Scholar] [CrossRef]
- Marfella, R.; Barbieri, M.; Sardu, C.; Rizzo, M.R.; Siniscalchi, M.; Paolisso, P.; Ambrosino, M.; Fava, I.; Materazzi, C.; Cinquegrana, G.; et al. Effects of α-lipoic acid therapy on sympathetic heart innervation in patients with previous experience of transient takotsubo cardiomyopathy. J. Cardiol. 2016, 67, 153–161. [Google Scholar] [CrossRef]
- Marshall, A.W.; Graul, R.S.; Morgan, M.Y.; Sherlock, S. Treatment of alcohol-related liver disease with thioctic acid: A six month randomised double-blind trial. Gut 1982, 23, 1088–1093. [Google Scholar] [CrossRef]
- Martins, V.D.; Manfredini, V.; Peralba, M.C.; Benfato, M.S. Alpha-lipoic acid modifies oxidative stress parameters in sickle cell trait subjects and sickle cell patients. Clin. Nutr. 2009, 28, 192–197. [Google Scholar] [CrossRef]
- Mendes, P.R.; Félix Ddos, S.; Silva, P.C.; Pereira, G.H.; Simões, M.O. Effect of alpha lipoic acid on the blood cell count and iron kinetics in hypertensive patients. Nutr. Hosp. 2014, 31, 883–889. [Google Scholar]
- Mendoza-Núñez, V.M.; García-Martínez, B.I.; Rosado-Pérez, J.; Santiago-Osorio, E.; Pedraza-Chaverri, J.; Hernández-Abad, V.J. The Effect of 600 mg Alpha-lipoic Acid Supplementation on Oxidative Stress, Inflammation, and RAGE in Older Adults with Type 2 Diabetes Mellitus. Oxid. Med. Cell Longev. 2019, 2019, 3276958. [Google Scholar] [CrossRef] [PubMed]
- Mirtaheri, E.; Pourghassem Gargari, B.; Kolahi, S.; Asghari-Jafarabadi, M.; Hajaliloo, M. Effect of alpha-lipoic acid supplementation on serum lipid profile in women with rheumatoid arthritis. NFSR 2014, 1, 11–18. [Google Scholar]
- Mohammadi, V.; Khorvash, F.; Feizi, A.; Askari, G. Does Alpha-lipoic Acid Supplementation Modulate Cardiovascular Risk Factors in Patients with Stroke? A Randomized, Double-blind Clinical Trial. Int. J. Prev. Med. 2018, 9, 34. [Google Scholar] [PubMed]
- Mohammadi, V.; Khalili, M.; Eghtesadi, S.; Dehghani, S.; Jazayeri, S.; Aghababaee, S.K.; Sabour, H.; Saberi, H.; Eghtesadi, M.; Gohari, M.R. The effect of alpha-lipoic acid (ALA) supplementation on cardiovascular risk factors in men with chronic spinal cord injury: A clinical trial. Spinal Cord. 2015, 53, 621–624. [Google Scholar] [CrossRef] [PubMed]
- Mollo, R.; Zaccardi, F.; Scalone, G.; Scavone, G.; Rizzo, P.; Navarese, E.P.; Manto, A.; Pitocco, D.; Lanza, G.A.; Ghirlanda, G.; et al. Effect of α-lipoic acid on platelet reactivity in type 1 diabetic patients. Diabetes Care 2012, 35, 196–197. [Google Scholar] [CrossRef]
- Monroy Guízar, E.A.; García Benavides, L.; Ambriz Plascencia, A.R.; Pascoe González, S.; Totsuka Sutto, S.E.; Cardona Muñoz, E.G.; Méndez-Del Villar, M. Effect of Alpha-Lipoic Acid on Clinical and Neurophysiologic Recovery of Carpal Tunnel Syndrome: A Double-Blind, Randomized Clinical Trial. J. Med. Food 2018, 21, 521–526. [Google Scholar] [CrossRef]
- Palacios-Sánchez, B.; Moreno-López, L.A.; Cerero-Lapiedra, R.; Llamas-Martínez, S.; Esparza-Gómez, G. Alpha lipoic acid efficacy in burning mouth syndrome. A controlled clinical trial. Med. Oral Patol. Oral Cir. Bucal. 2015, 20, e435–e440. [Google Scholar] [CrossRef]
- Porasuphatana, S.; Suddee, S.; Nartnampong, A.; Konsil, J.; Harnwong, B.; Santaweesuk, A. Glycemic and oxidative status of patients with type 2 diabetes mellitus following oral administration of alpha-lipoic acid: A randomized double-blinded placebo-controlled study. Asia Pac. J. Clin. Nutr. 2012, 21, 12–21. [Google Scholar]
- Pourghasem Gargari, B.; Kolahi, S.; Dehghan, P.; Khabbazi, A.; Mirtaheri, E. Effects of alpha-lipoic acid supplementation on clinical status and anthropometric indices in women with rheumatoid arthritis. Curr. Top. Nutraceutical Res. 2015, 13, 33–40. [Google Scholar]
- Ruhnau, K.J.; Meissner, H.P.; Finn, J.R.; Reljanovic, M.; Lobisch, M.; Schütte, K.; Nehrdich, D.; Tritschler, H.J.; Mehnert, H.; Ziegler, D. Effects of 3-week oral treatment with the antioxidant thioctic acid (alpha-lipoic acid) in symptomatic diabetic polyneuropathy. Diabetic Med. 1999, 16, 1040–1043. [Google Scholar] [CrossRef]
- Safa, J.; Ardalan, M.R.; Rezazadehsaatlou, M.; Mesgari, M.; Mahdavi, R.; Jadid, M.P. Effects of alpha lipoic acid supplementation on serum levels of IL-8 and TNF-α in patient with ESRD undergoing hemodialysis. Int. Urol. Nephrol. 2014, 46, 1633–1638. [Google Scholar] [CrossRef] [PubMed]
- Sammour, H.; Elkholy, A.; Rasheedy, R.; Fadel, E. The effect of alpha lipoic acid on uterine wound healing after primary cesarean section: A triple-blind placebo-controlled parallel-group randomized clinical trial. Arch. Gynecol. Obstet. 2019, 299, 665–673. [Google Scholar] [CrossRef] [PubMed]
- Sardu, C.; Santulli, G.; Santamaria, M.; Barbieri, M.; Sacra, C.; Paolisso, P.; D’Amico, F.; Testa, N.; Caporaso, I.; Paolisso, G.; et al. Effects of Alpha Lipoic Acid on Multiple Cytokines and Biomarkers and Recurrence of Atrial Fibrillation Within 1 Year of Catheter Ablation. Am. J. Cardiol. 2017, 119, 1382–1386. [Google Scholar] [CrossRef]
- Scaramuzza, A.; Giani, E.; Redaelli, F.; Ungheri, S.; Macedoni, M.; Giudici, V.; Bosetti, A.; Ferrari, M.; Zuccotti, G.V. Alpha-Lipoic Acid and Antioxidant Diet Help to Improve Endothelial Dysfunction in Adolescents with Type 1 Diabetes: A Pilot Trial. J. Diabetes Res. 2015, 2015, 474561. [Google Scholar] [CrossRef] [PubMed]
- Sola, S.; Mir, M.Q.; Cheema, F.A.; Khan-Merchant, N.; Menon, R.G.; Parthasarathy, S.; Khan, B.V. Irbesartan and lipoic acid improve endothelial function and reduce markers of inflammation in the metabolic syndrome: Results of the Irbesartan and Lipoic Acid in Endothelial Dysfunction (ISLAND) study. Circulation 2005, 111, 343–348. [Google Scholar] [CrossRef] [PubMed]
- Spain, R.; Powers, K.; Murchison, C.; Heriza, E.; Winges, K.; Yadav, V.; Cameron, M.; Kim, E.; Horak, F.; Simon, J.; et al. Lipoic acid in secondary progressive MS: A randomized controlled pilot trial. Neurol. Neuroimmunol. Neuroinflamm. 2017, 4, e374. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.D.; Dong, Y.D.; Fan, R.; Zhai, L.L.; Bai, Y.L.; Jia, L.H. Effect of (R)-α-lipoic acid supplementation on serum lipids and antioxidative ability in patients with age-related macular degeneration. Ann. Nutr. Metab. 2012, 60, 293–297. [Google Scholar] [CrossRef]
- Tromba, L.; Perla, F.M.; Carbotta, G.; Chiesa, C.; Pacifico, L. Effect of Alpha-Lipoic Acid Supplementation on Endothelial Function and Cardiovascular Risk Factors in Overweight/Obese Youths: A Double-Blind, Placebo-Controlled Randomized Trial. Nutrients 2019, 11, 375. [Google Scholar] [CrossRef] [PubMed]
- Udupa, A.; Nahar, P.; Shah, S.; Kshirsagar, M.; Ghongane, B. A comparative study of effects of omega-3 Fatty acids, alpha lipoic Acid and vitamin e in type 2 diabetes mellitus. Ann. Med. Health Sci. Res. 2013, 3, 442–446. [Google Scholar] [PubMed]
- Vincent, H.K.; Bourguignon, C.M.; Vincent, K.R.; Taylor, A.G. Effects of alpha-lipoic acid supplementation in peripheral arterial disease: A pilot study. J. Altern. Complement Med. 2007, 13, 577–584. [Google Scholar] [CrossRef] [PubMed]
- Yadav, V.; Marracci, G.; Lovera, J.; Woodward, W.; Bogardus, K.; Marquardt, W.; Shinto, L.; Morris, C.; Bourdette, D. Lipoic acid in multiple sclerosis: A pilot study. Mult. Scler. 2005, 11, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Yan, W.; Li, N.; Hu, X.; Huang, Y.; Zhang, W.; Wang, Q.; Wang, F.; Wang, C.; Zhai, X.; Xu, R.; et al. Effect of oral ALA supplementation on oxidative stress and insulin sensitivity among overweight/obese adults: A double-blinded, randomized, controlled, cross-over intervention trial. Int. J. Cardiol. 2013, 167, 602–603. [Google Scholar] [CrossRef] [PubMed]
- Zembron-Lacny, A.; Gajewski, M.; Naczk, M.; Dziewiecka, H.; Siatkowski, I. Physical activity and alpha-lipoic acid modulate inflammatory response through changes in thiol redox status. J. Physiol. Biochem. 2013, 69, 397–404. [Google Scholar] [CrossRef] [PubMed]
- Zembron-Lacny, A.; Slowinska-Lisowska, M.; Szygula, Z.; Witkowski, K.; Szyszka, K. The comparison of antioxidant and hematological properties of N-acetylcysteine and alpha-lipoic acid in physically active males. Physiol. Res. 2009, 58, 855–861. [Google Scholar]
- Ziegler, D.; Low, P.A.; Litchy, W.J.; Boulton, A.J.; Vinik, A.I.; Freeman, R.; Samigullin, R.; Tritschler, H.; Munzel, U.; Maus, J.; et al. Efficacy and safety of antioxidant treatment with α-lipoic acid over 4 years in diabetic polyneuropathy: The NATHAN 1 trial. Diabetes Care 2011, 34, 2054–2060. [Google Scholar] [CrossRef]
- Ziegler, D.; Ametov, A.; Barinov, A.; Dyck, P.J.; Gurieva, I.; Low, P.A.; Munzel, U.; Yakhno, N.; Raz, I.; Novosadova, M.; et al. Oral treatment with alpha-lipoic acid improves symptomatic diabetic polyneuropathy: The SYDNEY 2 trial. Diabetes Care 2006, 29, 2365–2370. [Google Scholar] [CrossRef]
- Mahady, G.B. Global harmonization of herbal health claims. J. Nutr. 2001, 131, 1120S–1123S. [Google Scholar] [CrossRef]
- Thakkar, S.; Anklam, E.; Xu, A.; Ulberth, F.; Li, J.; Li, B.; Hugas, M.; Sarma, N.; Crerar, S.; Swift, S.; et al. Regulatory landscape of dietary supplements and herbal medicines from a global perspective. Regul. Toxicol. Pharmacol. 2020, 114, 104647. [Google Scholar] [CrossRef]
- Han, T.; Bai, J.; Liu, W.; Hu, Y. A systematic review and meta-analysis of α-lipoic acid in the treatment of diabetic peripheral neuropathy. Eur. J. Endocrinol. 2012, 167, 465–471. [Google Scholar] [CrossRef]
- Parente, E.; Colannino, G.; Picconi, O.; Monastra, G. Safety of oral alpha-lipoic acid treatment in pregnant women: A retrospective observational study. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 4219–4227. [Google Scholar]
- Rahimlou, M.; Asadi, M.; Banaei Jahromi, N.; Mansoori, A. Alpha-lipoic acid (ALA) supplementation effect on glycemic and inflammatory biomarkers: A Systematic Review and meta- analysis. Clin. Nutr. ESPEN 2019, 32, 16–28. [Google Scholar] [CrossRef] [PubMed]
- Tabrizi, R.; Borhani-Haghighi, A.; Mirhosseini, N.; Lankarani, K.B.; Naghibzadeh-Tahami, A.; Akbari, M.; Heydari, S.T.; Sangari, M.; Kolahdooz, F.; Raygan, F.; et al. The effects of alpha-lipoic acid supplementation on fasting glucose and lipid profiles among patients with stroke: A systematic review and meta-analysis of randomized controlled trials. J. Diabetes Metab. Disord. 2019, 18, 585–595. [Google Scholar] [CrossRef]
- Akbari, M.; Ostadmohammadi, V.; Lankarani, K.B.; Tabrizi, R.; Kolahdooz, F.; Khatibi, S.R.; Asemi, Z. The effects of alpha-lipoic acid supplementation on glucose control and lipid profiles among patients with metabolic diseases: A systematic review and meta-analysis of randomized controlled trials. Metabolism 2018, 87, 56–69. [Google Scholar] [CrossRef] [PubMed]
- Eisenberg, D.M.; Davis, R.B.; Ettner, S.L.; Appel, S.; Wilkey, S.; Van Rompay, M.; Kessler, R.C. Trends in alternative medicine use in the United States, 1990–1997: Results of a follow-up national survey. JAMA 1998, 280, 1569–1575. [Google Scholar] [CrossRef] [PubMed]
- Mijnhout, G.S.; Kollen, B.J.; Alkhalaf, A.; Kleefstra, N.; Bilo, H.J. Alpha lipoic Acid for symptomatic peripheral neuropathy in patients with diabetes: A meta-analysis of randomized controlled trials. Int. J. Endocrinol. 2012, 2012, 456279. [Google Scholar] [CrossRef] [PubMed]
- Ziegler, D.; Nowak, H.; Kempler, P.; Vargha, P.; Low, P.A. Treatment of symptomatic diabetic polyneuropathy with the antioxidant alpha-lipoic acid: A meta-analysis. Diabet Med. 2004, 21, 114–121. [Google Scholar] [CrossRef]
- Vajdi, M.; Abbasalizad Farhangi, M. Alpha-lipoic acid supplementation significantly reduces the risk of obesity in an updated systematic review and dose response meta-analysis of randomised placebo-controlled clinical trials. Int. J. Clin. Pract. 2020, 74, e13493. [Google Scholar] [CrossRef]
- Ebada, M.A.; Fayed, N.; Fayed, L.; Alkanj, S.; Abdelkarim, A.; Farwati, H.; Hanafy, A.; Negida, A.; Ebada, M.; Noser, Y. Efficacy of Alpha-lipoic Acid in The Management of Diabetes Mellitus: A Systematic Review and Meta-analysis. Iran J. Pharm. Res. 2019, 18, 2144–2156. [Google Scholar]
- Sandercock, P. The authors say: ‘The data are not so robust because of heterogeneity’—So, how should I deal with this systematic review? Meta-analysis and the clinician. Cerebrovasc. Dis. 2011, 31, 615–620. [Google Scholar] [CrossRef]

| Author, Year | Location | Study Design | Treatment Duration | Main Inclusion Criteria and Underlying Disease | Study Group | Enrolled Subjects (n) | Age (years; mean ± SD) | Male [n (%)] |
|---|---|---|---|---|---|---|---|---|
| Ahmadi, 2013 [20] | Iran | Randomized, single-blind, placebo-controlled, parallel-group, clinical study | 2 months | End-stage renal disease on haemodialysis (≥2 times/week for ≥1 year) | 600 mg/day α-lipoic acid | 20 | 48.8 ± 11.2 | 14 (70) |
| Placebo | 24 | 48.9 ± 12.5 | 9 (38) | |||||
| Ansar, 2011 [21] | Iran | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 8 weeks | Type 2 diabetes mellitus FPG > 126 mg/dL | 300 mg/day α-lipoic acid | 29 | 49 ± 9.1 | 6 (21) |
| Placebo | 28 | 51.8 ± 8.3 | 8 (29) | |||||
| Aslfalah, 2019a [22] | Iran | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 8 weeks | Gestational diabetes mellitus | 100 mg/day α-lipoic acid | 30 | 30.96 ± 0.93 | 0 (0) |
| Placebo | 30 | 31.1 ± 0.92 | 0 (0) | |||||
| Aslfalah, 2019b [23] | Iran | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 8 weeks | Gestational diabetes mellitus | 100 mg/day α-lipoic acid | 30 | 30.96 ± 0.93 | 0 (0) |
| Placebo | 30 | 31.1 ± 0.92 | 0 (0) | |||||
| Baumgartner, 2017 [24] | The Netherlands | Randomized, double-blind, placebo-controlled, crossover, clinical study | 4 weeks | Impaired glucose tolerance or non-insulin-dependent type 2 diabetes BMI ≥ 20 kg/m2 and ≤35 kg/m2 | 600 mg/day α-lipoic acid | 20 | 63.1 ± 5.8 | 16 (80) |
| Placebo | ||||||||
| Baziar, 2020 [25] | Iran | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 8 weeks | Non-insulin-dependent diabetes mellitus HbA1c < 7% BMI ≥ 18.5 kg/m2 and ≤29.9 kg/m2 | 1200 mg/day α-lipoic acid | 35 | 52.66 ± 4.81 | 15 (43) |
| Placebo | 35 | 53.34 ± 4.45 | 16 (46) | |||||
| Bobe, 2020 [26] | United States of America | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 24 weeks | Sedentary lifestyle BMI ≥ 27 kg/m2 TG ≥ 150 mg/dL FPG < 125 mg/dL | 600 mg/day α-lipoic acid | 40 | 38 ± 10 * | 12 (39) * |
| Placebo | 41 | 40 ± 8 | 16 (48) * | |||||
| Boriani, 2017 [27] | Italy | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 40 days | Primary tunnel carpal syndrome at least one of the following findings: anaesthesia or paraesthesia in the median nerve territory, positive Tinel sign, Phalen or reverse Phalen manoeuvres, and positive nerve conduction studies irrespective of severity | 800 mg/day α-lipoic acid | 32 | 57.3 ± 12 | 13 (41) |
| Placebo | 32 | 58.5 ± 11 | 9 (28) | |||||
| Carbone, 2009 [28] | Italy | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 8 weeks | Burning mouth syndrome | 800 mg/day α-lipoic acid | 22 | NA | NA |
| Placebo | 22 | NA | NA | |||||
| Cavalcanti, 2009 [29] | Brazil | Randomized, double-blind, placebo-controlled, crossover, clinical study | 30 days | Burning mouth syndrome | 600 mg/day α-lipoic acid | 38 | 63.1 (36–78) § | 4 (11) |
| Placebo | ||||||||
| Durastanti, 2016 [30] | Italy | Randomized, double-blind, placebo-controlled, parallel-group, pilot clinical study | 2 years | Relapsing-remitting multiple sclerosis EDSS score ≤ 3.5 | 800 mg/day α-lipoic acid during the first year and 400 mg/day α-lipoic acid during the second year | 7 | 33 (26–43) ° | 2 (29) |
| Placebo | 6 | 28.5 (22.5–44.3) ° | 1 (17) | |||||
| El Amrousy, 2020 [31] | Egypt | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 3 months | Obese healthy children and adolescents BMI > 95th percentile for age and sex | 600 mg/day α-lipoic acid | 40 | 12.3 ± 1.5 | 16 (40) |
| Placebo | 40 | 12.4 ± 1.4 | 18 (45) | |||||
| Falardeau, 2019 [32] | United States of America | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 6 weeks | Unilateral acute optic neuritis | 1200 mg/day α-lipoic acid | 15 | 41.2 ± 10.51 | 7 (47) |
| Placebo | 16 | 36.1 ± 9.84 | 4 (25) | |||||
| Femiano, 2002 [33] | Spain | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 2 months | Burning mouth syndrome | 600 mg/day α-lipoic acid | 30 | 45 (22–68) § | 18 (30) |
| Placebo | 30 | |||||||
| Georgakouli, 2018 [34] | Greece | Randomized, double-blind, placebo-controlled, crossover, clinical study | 4 weeks | Healthy status | 600 mg/day α-lipoic acid | 8 | 38.4 ± 5.6 | 8 (100) |
| Placebo | ||||||||
| Gianturco, 2009 [35] | Italy | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 4 weeks | Diabetes mellitus HbA1c < 7% | 400 mg/day α-lipoic acid | 7 | 61 ± 7 | 4 (57) |
| Placebo | 7 | 58 ± 16 | 4 (57) | |||||
| Gilron, 2020 [36] | Canada | Randomized, double-blind, placebo-controlled, crossover, clinical study | 5 weeks | Fibromyalgia daily moderate pain (≥4/10 on a NRS) for ≥3 months | 600 mg/day α-lipoic acid during the first week; 1200 mg/day α-lipoic acid during the second week; 1800 mg/day α-lipoic acid during the third and the fourth weeks | 27 | 57 (25–74) § | 5 (19) |
| Placebo | ||||||||
| Gosselin, 2019 [37] | United States of America | Randomized, double-blind, placebo-controlled, crossover, clinical study | 1 month | Sedentary lifestyle FPG ≥ 100 mg/dL and ≤125 mg/dL BMI ≥ 25 kg/m2 and ≤40 kg/m2 | 600 mg/day α-lipoic acid | 12 | 47.1 ± 2.9 | 4 (33) |
| Placebo | ||||||||
| Guo, 2014 [38] | United States of America | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 24 weeks | Cancer patients receiving chemotherapy with cisplatin or oxaliplatin | 1800 mg/day α-lipoic acid | 122 | 55 ± 11 | 66 (54) |
| Placebo | 121 | 57 ± 12 | 63 (52) | |||||
| Haghighian, 2015 [39] | Iran | Randomized, triple-blind, placebo-controlled, parallel-group, clinical study | 12 weeks | Idiopathic asthenozoospermia BMI < 30 kg/m2 | 600 mg/day α-lipoic acid | 24 | 32.98 ± 5.35 * | 24 (100) |
| Placebo | 24 | 34.12 ± 4.79 * | 24 (100) | |||||
| Hejazi, 2018 [40] | Iran | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 10 days | Candidates for enteral feeding and expected to stay in the intensive care unit for ≥7 days | 2700 mg/day α-lipoic acid | 40 | 51.2 ± 17 | 17 (43) |
| Placebo | 40 | 57.4 ± 19 | 25 (63) | |||||
| Huang, 2008 [41] | United States of America | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 3 months | Pubertal or postpubertal adolescents with type 1 diabetes | 600–1200 mg/day (14–21 mg/kg/day) α-lipoic acid | 30 | 14 ± 2.4 | 13 (43) |
| Placebo | 10 | 15 ± 1.9 | 7 (70) | |||||
| Huerta, 2016 [42] | Spain | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 10 weeks | Sedentary lifestyle BMI ≥ 27.5 kg/m2 and ≤40 kg/m2 | 300 mg/day α-lipoic acid | 6 | 35.5 ± 8.4 | 0 (0) |
| Placebo | 6 | 41.8 ± 6.6 | 0 (0) | |||||
| Huerta, 2015 [43] | Spain | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 10 weeks | Healthy status regular menstrual cycles BMI ≥ 27.5 kg/m2 and ≤40 kg/m2 | 300 mg/day α-lipoic acid | 26 | 39 ± 8 * | 0 (0) |
| Placebo | 31 | 38 ± 7 * | 0 (0) | |||||
| Jacob, 1999 [44] | Germany | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 4 weeks | Well-controlled type 2 diabetes mellitus | 1800 mg/day α-lipoic acid | 18 | 62.1 ± 3 | 10 (56) |
| 1200 mg/day α-lipoic acid | 18 | 60.9 ± 2.2 | 11 (61) | |||||
| 600 mg/day α-lipoic acid | 19 | 58.1 ± 2.8 | 10 (53) | |||||
| Placebo | 19 | 60.4 ± 2.4 | 12 (63) | |||||
| Jamshidi, 2020 [45] | Iran | Randomized, double-blind, placebo-controlled, crossover, clinical study | 8 weeks | β-thalassemia major | 600 mg/day α-lipoic acid | 20 | 23.5 ± 5.47 | 13 (65) |
| Placebo | ||||||||
| Jariwalla, 2008 [46] | United States of America | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 6 months | HIV infection HIV-RNA viral load > 10.000 copies/cm3 despite HAART CD4+ cell count ≥ 50 cells/mm3 | 900 mg/day α-lipoic acid | 18 | 47.2 ± 6.8 | 29 (88) |
| Placebo | 15 | 43.7 ± 7.6 | ||||||
| Khabbazi, 2012 [47] | Iran | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 8 weeks | Patients with end-stage renal disease on haemodialysis | 600 mg/day α-lipoic acid | 31 | 53.83 ± 13.29 | 16 (52) |
| Placebo | 32 | 54.04 ± 13.96 | 18 (56) | |||||
| Khalili, 2017 [48] | Iran | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 12 weeks | Relapsing-remitting multiple sclerosis | 1200 mg/day α-lipoic acid | 15 | 32.3 ± 6.2 * | 5 (42) * |
| Placebo | 16 | 32.2 ± 10.5 * | 1 (8) * | |||||
| Khalili, 2014 [49] | Iran | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 12 weeks | Relapsing-remitting multiple sclerosis | 1200 mg/day α-lipoic acid | 26 | 31.4 ± 6.2 * | 7 (27) |
| Placebo | 34 | 28.7 ± 9 * | 9 (26) | |||||
| Kim, 2020 [50] | South Korea | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 18 months | Geographic atrophy | 1200 mg/day α-lipoic acid | 26 | 80.6 ± 6.5 | 8 (31) |
| Placebo | 27 | 79 ± 7 | 11 (41) | |||||
| Kim, 2016 [51] | South Korea | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 12 weeks | Chronic schizophrenia in rehabilitation significant weight gain after starting treatment with atypical antipsychotics | 600–1800 mg/day α-lipoic acid | 10 | 40.5 ± 6.65 | 4 (40) |
| Placebo | 12 | 40.08 ± 9.14 | 7 (58) | |||||
| Koh, 2011 [52] | Republic of Korea | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 20 weeks | BMI ≥ 30 kg/m2 or BMI ≥ 27.5 kg/m2 and ≤40 kg/m2 if hypertension, diabetes mellitus and/or hypercholesterolemia coexisted | 1800 mg/day α-lipoic acid | 120 | 41.4 ± 1 | 82 (68) |
| 1200 mg/day α-lipoic acid | 120 | 41.6 ± 1.1 | 79 (66) | |||||
| Placebo | 120 | 40.7 ± 1.1 | 74 (62) | |||||
| Lampitella, 2005 [53] | Italy | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 6 months | Type 2 diabetes mellitus | 600 mg/day α-lipoic acid | 20 | NA | NA |
| Placebo | 20 | NA | NA | |||||
| Lee, 2017 [54] | Republic of Korea | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 24 weeks | Diabetic cardiac autonomic neuropathy | 600-1200 mg/day α-lipoic acid | 46 | 64.37 ± 7.8 | 27 (59) |
| Placebo | 45 | 62.4 ± 9.1 | 20 (44) | |||||
| Loy, 2018 [55] | United States of America | Randomized, double-blind, placebo-controlled, parallel-group, pilot clinical study | 2 years | Multiple sclerosis disability progression in absence of clinical relapse for 5 years EDSS ≤ 6.0 ability to walk ≥ 25 feet without aid | 1200 mg/day α-lipoic acid | 11 | 55.8 ± 5.7 | 5 (45) |
| Placebo | 10 | 55.7 ± 4.1 | 5 (50) | |||||
| López-D’alessandro, 2011 [56] | Argentina | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 2 months | Burning mouth syndrome | 600 g/day α-lipoic acid | 20 | NA | NA |
| Placebo | 60 | NA | NA | |||||
| López-Jornet, 2009 [57] | Spain | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 8 weeks | Burning mouth syndrome | 800 mg/day α-lipoic acid | 30 | 64.37 ± 11.61 | 6 (10) |
| Placebo | 30 | |||||||
| Magis, 2007 [58] | Belgium | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 3 months | Migraine with or without aura | 600 mg/day α-lipoic acid | 26 | 37.46 ± 13.43 | 4 (15) |
| Placebo | 18 | 38.94 ± 8.05 | 2 (11) | |||||
| Manning, 2013 [59] | New Zeland | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 1 year | Metabolic syndrome | 600 mg/day α-lipoic acid | 34 | 55 ± 10 | 14 (41) |
| Placebo | 40 | 57 ± 9 | 15 (38) | |||||
| Marfella, 2016 [60] | Italy | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 12 months | Takotsubo cadiomyopathy | 600 mg/day α-lipoic acid | 24 | 63.7 ± 6.5 | 0 (0) |
| Placebo | 24 | 63.9 ± 5.2 | 0 (0) | |||||
| Marshall, 1982 [61] | United Kingdom | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 24 weeks | Alcohol related liver disease | 300 mg/day α-lipoic acid | 20 | 50.7 ± 1.9 | 17 (85) |
| Placebo | 20 | 46.4 ± 2.7 | 15 (75) | |||||
| Martins, 2009 [62] | Brazil | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 3 months | Sickle cell disease | 200 mg/day α-lipoic acid | 10 | 17.7 ± 9.6 | 6 (60) |
| Placebo | 10 | 17 ± 11 | 5 (50) | |||||
| Sickle cell trait | 200 mg/day α-lipoic acid | 10 | 31.3 ± 15.4 | 2 (20) | ||||
| Placebo | 10 | 29.7 ± 10.8 | 2 (20) | |||||
| Healthy status | 200 mg/day α-lipoic acid | 10 | 23.5 ± 11 | 4 (40) | ||||
| Placebo | 10 | 23.3 ± 11 | 3 (30) | |||||
| Mendes, 2014 [63] | Brazil | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 12 weeks | Arterial hypertension | 600 mg/day α-lipoic acid | 32 | NA | NA |
| Placebo | 28 | NA | NA | |||||
| Mendoza-Núñez, 2019 [64] | Mexico | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 6 months | Type 2 diabetes mellitus without complications or comorbidity, treated with two tablets of glibenclamide/metformin (5/500 mg) per day BMI < 35 kg/m2 sedentary lifestyle | 600 mg/day α-lipoic acid | 50 | 63 ± 1 * | NA |
| Placebo | 50 | 64 ± 1 * | NA | |||||
| Mirtaheri, 2014 [65] | Iran | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 8 weeks | Rheumatoid arthritis | 1200 mg/day α-lipoic acid | 35 | 36.09 ± 8.77 * | 0 (0) |
| Placebo | 35 | 38.28 ± 8.63 * | 0 (0) | |||||
| Mohammadi, 2018 [66] | Iran | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 12 weeks | Previous thrombotic or embolic stroke BMI ≥ 18.5 kg/m2 and ≤35 kg/m2 | 600 mg/day α-lipoic acid | 40 | 62.33 ± 6.19 | NA |
| Placebo | 40 | 64.23 ± 8.01 | NA | |||||
| Mohammadi, 2015 [67] | Iran | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 12 weeks | Spinal cord injury since ≥ 1 year BMI ≥ 18.5 kg/m2 | 600 mg/day α-lipoic acid | 28 | 39 ± 6.44 | 28 (100) |
| Placebo | 30 | 36.8 ± 7.48 | 30 (100) | |||||
| Mollo, 2012 [68] | Italy | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 5 weeks | Type 1 diabetes | 600 mg/day α-lipoic acid | 26 | 43 ± 9 | 15 (58) |
| Placebo | 25 | 46 ± 11 | 12 (48) | |||||
| Monroy Guízar, 2018 [69] | Mexico | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 3 months | Idiopathic carpal tunnel syndrome | 600 mg/day α-lipoic acid | 10 | 45.3 † | 1 (10) |
| Placebo | 10 | 48.4 † | 1 (10) | |||||
| Palacios-Sánchez, 2015 [70] | Spain | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 2 months | Burning mouth syndrome | 600 mg/day α-lipoic acid | 30 | 62.13 (36–86) § | 5 (8) |
| Placebo | 30 | |||||||
| Porasuphatana, 2012 [71] | Thailand | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 6 months | Type 2 diabetes mellitus with microalbuminuria | 1200 mg/day α-lipoic acid | 7 | 47.07 ± 2.18 | 1 (14) |
| 900 mg/day α-lipoic acid | 7 | 44 ± 2 | 1 (14) | |||||
| 600 mg/day α-lipoic acid | 8 | 45.7 ± 1.68 | 3 (38) | |||||
| 300 mg/day α-lipoic acid | 8 | 42.5 ± 1.12 | 4 (50) | |||||
| Placebo | 8 | 42.9 ± 2.52 | 1 (13) | |||||
| Pourghasem Gargari, 2014 [72] | Iran | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 8 weeks | Rheumatoid arthritis DAS28 < 5.1 BMI < 40 kg/m2 | 1200 mg/day α-lipoic acid | 35 | 36.1 ± 8.8 | 0 (0) |
| Placebo | 35 | 38.3 ± 8.6 | 0 (0) | |||||
| Rahmanabadi, 2019 [4] | Iran | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 12 weeks | Non-alcoholic fatty liver disease BMI ≥ 30 kg/m2 and ≤40 kg/m2 | 1200 mg/day α-lipoic acid | 25 | 40.28 ± 5.5 | 13 (52) |
| Placebo | 25 | 37.52 ± 9.67 | 14 (56) | |||||
| Ruhnau, 1999 [73] | Germany | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 3 weeks | Type 2 diabetes mellitus with distal symmetrical polyneuropathy | 1800 mg/day α-lipoic acid | 12 | 60.5 ± 6.9 | 6 (50) |
| Placebo | 12 | 62.1 ± 4.5 | 6 (50) | |||||
| Safa, 2014 [74] | Iran | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 12 months | End-stage renal disease on haemodialysis ≥ 6 months | 600 mg/day α-lipoic acid | 30 | 59.3 ± 10.47 | 21 (70) |
| Placebo | 31 | 55.2 ± 13.43 | 21 (68) | |||||
| Sammour, 2019 [75] | Egypt | Randomized, triple-blind, placebo-controlled, parallel-group, clinical study | 6 weeks | Primary caesarean section in singleton term pregnancy | 1200 mg/day α-lipoic acid | 51 | 25.3 ± 5.1 | 0 (0) |
| Placebo | 51 | 25.1 ± 5.4 | 0 (0) | |||||
| Sardu, 2017 [76] | Italy | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 12 months | Paroxysmal, symptomatic atrial fibrillation ≥ 6 months refractory to ≥1 class 1–3 antiarrhythmic drugs and treated with catheter ablation | 600 mg/day α-lipoic acid | 33 | 58.8 ± 6.7 | 15 (45) |
| Placebo | 40 | 61.5 ± 8.1 | 23 (58) | |||||
| Scaramuzza, 2015 [77] | Italy | Randomized, double-blind, placebo-controlled, parallel-group, pilot clinical study | 6 months | Type 1 diabetes endothelial dysfunction | 800 mg/day α-lipoic acid | 25 | 16.1 ± 3.1 | 15 (60) |
| Placebo | 27 | 16 ± 3.4 | 16 (59) | |||||
| Sola, 2005 [78] | United Stated of America | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 4 weeks | Metabolic syndrome | 300 mg/day α-lipoic acid | 15 | 46 ± 15 | 5 (33) |
| Placebo | 14 | 44 ± 13 | 6 (43) | |||||
| Spain, 2017 [79] | United Stated of America | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 2 years | Multiple sclerosis disability progression in absence of clinical relapse for 5 years | 1200 mg/day α-lipoic acid | 27 | 57.9 ± 6.7 | 11 (41) |
| Placebo | 24 | 59.7 ± 6 | 9 (38) | |||||
| Sun, 2012 [80] | China | Randomized, blind, placebo-controlled, parallel-group, clinical study | 3 months | Dry form of age-related macular degeneration | 600 mg/day α-lipoic acid | 32 | 65.8 ± 7.9 | 11 (35) |
| Placebo | 30 | 64.5 ± 8.1 | 10 (33) | |||||
| Tromba, 2019 [81] | Italy | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 12 weeks | BMI ≥ 85th percentile for age and sex | 800 mg/day α-lipoic acid | 34 | 11.5 ± 1.9 * | 16 (50) * |
| Placebo | 33 | 11.1 ± 2.1 * | 20 (63) * | |||||
| Udupa, 2013 [82] | India | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 90 days | Type 2 diabetes mellitus FGP ≥ 110 mg/dL and ≤250 mg/dL | 300 mg/day α-lipoic acid | 25 | 53.5 ± 1.4 | 12 (48) |
| Placebo | 25 | 53.8 ± 2.1 | 15 (60) | |||||
| Vincent, 2007 [83] | United States of America | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 3 months | ABI ≥ 0.3 and ≤0.9 claudication pain with walking | 600 mg/day α-lipoic acid | 16 | 75.1 ± 8.2 | 9 (56) |
| Placebo | 12 | 70.7 ± 18.9 | 6 (50) | |||||
| Yadav, 2005 [84] | United States of America | Randomized, double-blind, placebo-controlled, parallel-group, pilot clinical study | 14 days | Multiple sclerosis EDSS score ≤ 7.5 | 2400 mg/day α-lipoic acid | 8 | 44.5 (34–56) § | 0 (0) |
| 1200 mg/day α-lipoic acid | 16 | NA | 2 (13) | |||||
| Placebo | 9 | 50 (36–66) § | 2 (22) | |||||
| Yan, 2013 [85] | China | Randomized, double-blind, placebo-controlled, crossover, clinical study | 8 weeks | BMI ≥ 25 kg/m2 ≥1 of borderline hypertension, dyslipidemia, or impaired FPG | 1200 mg/day α-lipoic acid | 103 | NA | NA |
| Placebo | ||||||||
| Zembron-Lacny, 2013 [86] | Poland | Randomized, double-blind, placebo-controlled, crossover, clinical study | 10 days | Healthy status | 1200 mg/day α-lipoic acid | 16 | 20.7 ± 0.9 | 16 (100) |
| Placebo | ||||||||
| Zembron-Lacny, 2009 [87] | Poland | Randomized, double-blind, placebo-controlled, crossover, clinical study | 8 days | Physical education students healthy status forced training experience ≥3 years | 1200 mg/day α-lipoic acid | 13 | 25.5 ± 6 | 13 (100) |
| Placebo | ||||||||
| Ziegler, 2011 [88] | Canada, Croatia, Denmark, France, Italy, Spain, The Netherlands, United Kingdom, United States of America | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 4 years | Type 1 or 2 diabetes (duration ≥1 year) stage 1 or 2a distal symmetric sensorimotor polyneuropathy due to diabetes stable insulin regimen NIS[LL]+7 ≥ 2 one of the following abnormalities: abnormal nerve conduction attributes in two separate nerves ≥ 99th percentile for distal latency or ≤1st percentile for nerve conduction velocity or amplitude OR HRBD ≥ 1st percentile or TSS in the feet< 5 | 600 mg/day α-lipoic acid | 231 | 53.3 ± 8.3 | 152 (66) |
| Placebo | 225 | 53.9 ± 7.6 | 154 (67) | |||||
| Ziegler, 2006 [89] | Israel and Russia | Randomized, double-blind, placebo-controlled, parallel-group, clinical study | 5 weeks | Type 1 or 2 diabetes HbA1c < 10% symptomatic distal symmetric polyneuropathy due to diabetes TSS > 7.5 NIS[LL] ≥ 2 absent or decreased pain sensation according to pin-prick test | 1800 mg/day α-lipoic acid | 46 | 59 ± 9 | 19 (41) |
| 1200 mg/day α-lipoic acid | 47 | 59 ± 12 | 19 (40) | |||||
| 600 mg/day α-lipoic acid | 45 | 56 ± 12 | 20 (44) | |||||
| Placebo | 43 | 57 ± 11 | 15 (35) |
| Author, Year | Sequence Generation | Allocation Concealment | Blinding to Participants, Personnel and Outcome Assessment | Incomplete Outcome Data | Selective Outcome Reporting | Other Potential Threats to Validity |
|---|---|---|---|---|---|---|
| Ahmadi, 2013 [20] | L | L | H | L | L | U |
| Ansar, 2011 [21] | L | L | L | L | U | L |
| Aslfalah, 2019a [22] | L | L | L | L | L | L |
| Aslfalah, 2019b [23] | L | L | L | L | L | L |
| Baumgartner, 2017 [24] | L | L | L | L | L | L |
| Baziar, 2020 [25] | L | L | L | L | L | L |
| Bobe, 2020 [26] | L | L | L | L | L | L |
| Boriani, 2017 [27] | L | L | L | L | L | L |
| Carbone, 2009 [28] | L | L | L | L | L | L |
| Cavalcanti, 2009 [29] | L | L | L | L | L | L |
| Durastanti, 2016 [30] | L | L | L | U | U | U |
| El Amrousy, 2020 [31] | L | L | L | L | L | L |
| Falardeau, 2019 [32] | L | L | L | L | L | L |
| Femiano, 2002 [33] | U | L | L | L | U | U |
| Georgakouli, 2018 [34] | L | L | L | L | L | L |
| Gianturco, 2009 [35] | L | L | L | L | U | L |
| Gilron, 2020 [36] | L | L | L | L | L | L |
| Gosselin, 2019 [37] | L | L | L | L | L | L |
| Guo, 2014 [38] | L | L | L | L | L | L |
| Haghighian, 2015 [39] | L | L | L | L | L | L |
| Hejazi, 2018 [40] | L | L | L | L | L | L |
| Huang, 2008 [41] | L | L | L | L | L | L |
| Huerta, 2016 [42] | L | L | L | L | L | L |
| Huerta, 2015 [43] | L | L | L | L | L | L |
| Jacob, 1999 [44] | L | L | L | L | U | H |
| Jamshidi, 2020 [45] | L | L | L | L | L | L |
| Jariwalla, 2008 [46] | L | L | L | L | U | H |
| Khabbazi, 2012 [47] | L | L | L | L | L | L |
| Khalili, 2017 [48] | L | L | L | L | L | L |
| Khalili, 2014 [49] | L | L | L | L | L | L |
| Kim, 2020 [50] | L | L | L | L | L | L |
| Kim, 2016 [51] | L | L | L | L | L | L |
| Koh, 2011 [52] | L | L | L | L | L | L |
| Lampitella, 2005 [53] | L | U | U | L | L | U |
| Lee, 2017 [54] | L | L | L | L | L | L |
| Loy, 2018 [55] | L | L | L | L | L | L |
| López- D’Alessandro, 2011 [56] | L | L | L | H | H | U |
| López-Jornet, 2009 [57] | L | L | L | L | L | L |
| Magis, 2007 [58] | L | L | L | L | L | L |
| Manning, 2013 [59] | L | L | L | L | L | L |
| Marfella, 2016 [60] | L | L | U | L | L | U |
| Marshall, 1982 [61] | L | L | L | L | L | L |
| Martins, 2009 [62] | L | L | U | L | L | U |
| Mendes, 2014 [63] | L | L | L | L | H | U |
| Mendoza- Núñez, 2019 [64] | L | L | L | L | L | L |
| Mirtaheri, 2014 [65] | L | L | L | L | L | L |
| Mohammadi, 2018 [66] | L | L | L | L | L | L |
| Mohammadi, 2015 [67] | L | L | L | L | L | L |
| Mollo, 2012 [68] | L | L | L | L | L | L |
| Monroy Guízar, 2018 [69] | L | L | L | L | L | L |
| Palacios- Sánchez, 2015 [70] | L | L | L | L | L | L |
| Porasuphatana, 2012 [71] | L | L | L | L | L | H |
| Pourghasem Gargari, 2014 [72] | L | L | L | L | L | L |
| Rahmanabadi, 2019 [4] | L | L | L | L | L | L |
| Ruhnau, 1999 [73] | L | L | L | L | L | L |
| Safa, 2014 [74] | L | L | L | L | L | L |
| Sammour, 2019 [75] | L | L | L | L | L | L |
| Sardu, 2017 [76] | L | L | L | L | L | L |
| Scaramuzza, 2015 [77] | L | L | L | L | L | L |
| Sola, 2005 [78] | L | L | L | L | L | L |
| Spain, 2017 [79] | L | L | L | L | L | L |
| Sun, 2012 [80] | L | U | U | L | L | U |
| Tromba, 2019 [81] | L | L | L | L | L | L |
| Udupa, 2013 [82] | L | L | L | L | L | L |
| Vincent, 2007 [83] | L | L | L | L | L | L |
| Yadav, 2005 [84] | L | L | L | L | L | L |
| Yan, 2013 [85] | L | L | L | L | L | L |
| Zembron- Lacny, 2013 [86] | L | L | L | L | L | L |
| Zembron- Lacny, 2009 [87] | L | L | L | L | L | L |
| Ziegler, 2011 [88] | L | L | L | L | L | L |
| Ziegler, 2006 [89] | L | L | L | L | L | L |
| AEs | Smoking Habit | Cardiovascular Disease | Diabetes | Pregnancy | Neurological Disorders | Rheumatic Affections | Children and/or Adolescents | Severe Renal Impairment | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Yes | No | Yes | No | Yes | No | Yes | No | Yes | No | Yes | No | Yes | No | Yes | No | ||
| Gastrointestinal AEs | Number of reported AEs (active arm/placebo arm) | -/- | 4/2 | 2/0 | 97/88 | 137/77 | 17/14 | 3/2 | 180/97 | 144/76 | -/- | 5/2 | 4/3 | 3/2 | 180/97 | -/- | 94/81 |
| Odd ratio | - | 1.192 | 2.734 | 1.103 | 1.267 | 1.155 | 1.531 | 1.313 | 1.295 | - | 2.841 | 1.433 | 1.705 | 1.309 | - | 1.158 | |
| 95% CI (lower limit; upper limit) | - | 0.265; 5.361 | 0.273; 27.383 | 0.781; 1.558 | 0.879; 1.827 | 0.540; 2.468 | 0.245; 9.574 | 0.966; 1.784 | 0.897; 1.869 | - | 0.500; 16.138 | 0.300; 6.833 | 0.260; 11.156 | 0.964; 1.779 | - | 0.811; 1.653 | |
| Z-value | - | 0.229 | 0.856 | 0.556 | 1.268 | 0.371 | 0.456 | 1.740 | 1.382 | - | 1.178 | 0.451 | 0.556 | 1.724 | - | 0.809 | |
| I2 (%) | - | 0 | 0 | 0 | 50 | 0 | 0 | 0 | 48 | - | 0 | 0 | 0 | 0 | - | 0 | |
| P-value | - | 0.819 | 0.392 | 0.578 | 0.205 | 0.711 | 0.649 | 0.082 | 0.167 | - | 0.239 | 0.652 | 0.578 | 0.085 | - | 0.418 | |
| Neurological AEs | Number of reported AEs (active arm/placebo arm) | -/- | 6/2 | 1/0 | 19/18 | 10/0 | 18/14 | -/- | 50/23 | 25/9 | -/- | 8/6 | 0/1 | -/- | 50/23 | -/- | 22/16 |
| Odd ratio | - | 1.024 | 3.078 | 1.153 | 2.368 | 1.268 | - | 1.526 | 1.718 | - | 1.474 | 0.315 | - | 1.526 | - | 3.078 | |
| 95% CI (lower limit; upper limit) | - | 0.236; 4.442 | 0.122; 77.905 | 0.544; 2.442 | 0.884; 2.634 | 0.552; 2.914 | - | 0.884; 2.634 | 0.742; 3.977 | - | 0.432; 5.027 | 0.012; 7.999 | - | 0.884; 2.634 | - | 0.122; 77.905 | |
| Z-value | - | 0.032 | 0.682 | 0.371 | 1.517 | 0.560 | - | 1.517 | 1.264 | - | 0.619 | −0.700 | - | 1.517 | - | 0.682 | |
| I2 (%) | - | 0 | 0 | 0 | 0 | 0 | - | 0 | 0 | - | 0 | 0 | - | 0 | - | 0 | |
| P-value | - | 0.974 | 0.495 | 0.711 | 0.129 | 0.575 | - | 0.129 | 0.206 | - | 0.536 | 0.484 | - | 0.129 | - | 0.495 | |
| Psychiatric AEs | Number of reported AEs (active arm/placebo arm) | -/- | 2/0 | -/- | 30/25 | 26/25 | 4/0 | -/- | 30/25 | 26/25 | -/- | -/- | 2/0 | -/- | 30/25 | -/- | 28/25 |
| Odd ratio | - | 5.145 | - | 1.131 | 1.014 | 5.071 | - | 1.131 | 1.014 | - | - | 5.145 | - | 1.131 | - | 1.073 | |
| 95% CI (lower limit; upper limit) | - | 0.238; 111.087 | - | 0.644; 1.986 | 0.566; 1.817 | 0.582; 44.174 | - | 0.644; 1.986 | 0.566; 1.817 | - | - | 0.238; 111.087 | - | 0.644; 1.986 | - | 0.605; 1.903 | |
| Z-value | - | 1.045 | - | 0.429 | 0.048 | 1.470 | - | 0.429 | 0.048 | - | - | 1.045 | - | 0.429 | - | 0.242 | |
| I2 (%) | - | 0 | - | 0 | 0 | 0 | - | 0 | 0 | - | - | 0 | - | 0 | - | 0 | |
| P-value | - | 0.296 | - | 0.668 | 0.962 | 0.142 | - | 0.668 | 0.962 | - | - | 0.296 | - | 0.668 | - | 0.809 | |
| Musculoskeletal AEs | Number of reported AEs (active arm/placebo arm) | -/- | 1/0 | -/- | 3/5 | -/- | 3/4 | -/- | 5/5 | 4/4 | -/- | 0/1 | 1/0 | -/- | 5/5 | -/- | 3/5 |
| Odd ratio | - | 3.000 | - | 0.625 | - | 0.738 | - | 0.761 | 0.683 | - | 0.321 | 3.000 | - | 0.761 | - | 0.625 | |
| 95% CI (lower limit; upper limit) | - | 0.118; 76.161 | - | 0.147; 2.661 | - | 0.146; 3.723 | - | 0.220; 2.635 | 0.156; 2.997 | - | 0.013; 8.241 | 0.118; 76.161 | - | 0.220; 2.635 | - | 0.147; 2.661 | |
| Z-value | - | 0.666 | - | −0.636 | - | −0.368 | - | −0.431 | −0.505 | - | −0.686 | 0.666 | - | −0.431 | - | −0.636 | |
| I2 (%) | - | 0 | - | 0 | - | 0 | - | 0 | 0 | - | 0 | 0 | - | 0 | - | 0 | |
| P-value | - | 0.506 | - | 0.525 | - | 0.713 | - | 0.666 | 0.614 | - | 0.493 | 0.506 | - | 0.666 | - | 0.525 | |
| Skin AEs | Number of reported AEs (active arm/placebo arm) | -/- | 21/4 | -/- | 92/94 | 83/90 | 14/6 | -/- | 139/103 | 83/91 | 1/0 | -/- | -/- | -/- | 139/103 | 2/0 | 104/95 |
| Odd ratio | - | 2.821 | - | 0.912 | 0.816 | 2.258 | - | 1.127 | 0.819 | 3.353 | - | - | - | 1.127 | 1.545 | 0.932 | |
| 95% CI (lower limit; upper limit) | - | 0.899; 8.850 | - | 0.635; 1.308 | 0.559; 1.191 | 0.851; 5.992 | - | 0.815; 1.559 | 0.563; 1.192 | 0.120; 93.835 | - | - | - | 0.815; 1.559 | 0.067; 35.431 | 0.653; 1.331 | |
| Z-value | - | 1.778 | - | −0.502 | −1.052 | 1.636 | - | 0.724 | −1.041 | 0.712 | - | - | - | 0.724 | 0.272 | −0.387 | |
| I2 (%) | - | 0 | - | 29 | 0 | 0 | - | 34 | 0 | 0 | - | - | - | 34 | 0 | 36 | |
| P-value | - | 0.075 | - | 0.616 | 0.293 | 0.102 | - | 0.469 | 0.298 | 0.477 | - | - | - | 0.469 | 0.785 | 0.699 | |
| Infections | Number of reported AEs (active arm/placebo arm) | -/- | 3/0 | -/- | 1/3 | -/- | 1/3 | -/- | 5/3 | 1/3 | -/- | -/- | -/- | -/- | 5/3 | -/- | 4/3 |
| Odd ratio | - | 3.316 | - | 0.310 | - | 0.310 | - | 0.926 | 0.310 | - | - | - | - | 0.926 | - | 0.780 | |
| 95% CI (lower limit; upper limit) | - | 0.167; 65.718 | - | 0.028; 3.364 | - | 0.028; 3.364 | - | 0.184; 4.647 | 0.028; 3.364 | - | - | - | - | 0.184; 4.647 | - | 0.121; 5.028 | |
| Z-value | - | 0.787 | - | −0.963 | - | −0.963 | - | −0.094 | −0.963 | - | - | - | - | −0.094 | - | −0.262 | |
| I2 (%) | - | 0 | - | 0 | - | 0 | - | 0 | 0 | - | - | - | - | 0 | - | 32 | |
| P-value | - | 0.432 | - | 0.335 | - | 0.335 | - | 0.925 | 0.335 | - | - | - | - | 0.925 | - | 0.793 | |
| CV system AEs | Number of reported AEs (active arm/placebo arm) | -/- | 0/1 | 0/2 | 71/53 | 71/54 | 1/3 | -/- | 73/60 | 71/54 | -/- | -/- | 0/1 | -/- | 73/60 | -/- | 71/57 |
| Odd ratio | - | 0.149 | 0.191 | 1.441 | 1.409 | 0.450 | - | 1.247 | 1.409 | - | - | 0.333 | - | 1.247 | - | 1.313 | |
| 95% CI (lower limit; upper limit) | - | 0.006; 3.733 | 0.009; 4.214 | 0.950; 2.186 | 0.932; 2.130 | 0.056; 3.608 | - | 0.838; 1.854 | 0.932; 2.130 | - | - | 0.012; 9.068 | - | 0.838; 1.854 | - | 0.875; 1.972 | |
| Z-value | - | −1.159 | −1.049 | 1.720 | 1.625 | −0.752 | - | 1.089 | 1.625 | - | - | −0.652 | - | 1.089 | - | 1.314 | |
| I2 (%) | - | 0 | 0 | 0 | 0 | 0 | - | 16 | 0 | - | - | 0 | - | 16 | - | 27 | |
| P-value | - | 0.247 | 0.294 | 0.085 | 0.104 | 0.452 | - | 0.276 | 0.104 | - | - | 0.515 | - | 0.276 | - | 0.189 | |
| Hospitalisation | Number of reported AEs (active arm/placebo arm) | -/- | 4/0 | -/- | 2/0 | -/- | 2/0 | -/- | 4/0 | -/- | -/- | -/- | 2/0 | -/- | 4/0 | 2/0 | 2/0 |
| Odd ratio | - | 5.657 | - | 5.145 | - | 5.145 | - | 5.657 | - | - | - | 5.145 | - | 5.657 | 6.224 | 5.145 | |
| 95% CI (lower limit; upper limit) | - | 0.642; 49.849 | - | 0.238; 111.087 | - | 0.238; 111.087 | - | 0.642; 49.849 | - | - | - | 0.238; 111.087 | - | 0.642; 49.849 | 0.285; 135.784 | 0.238; 111.087 | |
| Z-value | - | 1.561 | - | 1.045 | - | 1.045 | - | 1.561 | - | - | - | 1.045 | - | 1.561 | 1.163 | 1.045 | |
| I2 (%) | - | 0 | - | 0 | - | 0 | - | 0 | - | - | - | 0 | - | 0 | 0 | 0 | |
| P-value | - | 0.119 | - | 0.296 | - | 0.296 | - | 0.119 | - | - | - | 0.296 | - | 0.119 | 0.245 | 0.296 | |
| Death | Number of reported AEs (active arm/placebo arm) | -/- | 0/2 | 4/5 | -/- | -/- | 1/2 | -/- | 6/12 | 1/3 | -/- | -/- | -/- | -/- | 6/12 | 0/2 | 6/9 |
| Odd ratio | - | 0.215 | 0.777 | - | - | 0.529 | - | 0.558 | 0.468 | - | - | - | - | 0.558 | 0.215 | 0.657 | |
| 95% CI (lower limit; upper limit) | - | 0.010; 4.690 | 0.192; 3.142 | - | - | 0.046; 6.109 | - | 0.210; 1.483 | 0.066; 3.300 | - | - | - | - | 0.210; 1.483 | 0.010; 4.690 | 0.222; 1.947 | |
| Z-value | - | −0.977 | −0.354 | - | - | −0.510 | - | −1.169 | −0.762 | - | - | - | - | −1.169 | −0.977 | −0.758 | |
| I2 (%) | - | 0 | 0 | - | - | 0 | - | 0 | 0 | - | - | - | - | 0 | 0 | 0 | |
| P-value | - | 0.328 | 0.724 | - | - | 0.610 | - | 0.242 | 0.446 | - | - | - | - | 0.242 | 0.328 | 0.448 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fogacci, F.; Rizzo, M.; Krogager, C.; Kennedy, C.; Georges, C.M.G.; Knežević, T.; Liberopoulos, E.; Vallée, A.; Pérez-Martínez, P.; Wenstedt, E.F.E.; et al. Safety Evaluation of α-Lipoic Acid Supplementation: A Systematic Review and Meta-Analysis of Randomized Placebo-Controlled Clinical Studies. Antioxidants 2020, 9, 1011. https://doi.org/10.3390/antiox9101011
Fogacci F, Rizzo M, Krogager C, Kennedy C, Georges CMG, Knežević T, Liberopoulos E, Vallée A, Pérez-Martínez P, Wenstedt EFE, et al. Safety Evaluation of α-Lipoic Acid Supplementation: A Systematic Review and Meta-Analysis of Randomized Placebo-Controlled Clinical Studies. Antioxidants. 2020; 9(10):1011. https://doi.org/10.3390/antiox9101011
Chicago/Turabian StyleFogacci, Federica, Manfredi Rizzo, Christoffer Krogager, Cormac Kennedy, Coralie M.G. Georges, Tamara Knežević, Evangelos Liberopoulos, Alexandre Vallée, Pablo Pérez-Martínez, Eliane F.E. Wenstedt, and et al. 2020. "Safety Evaluation of α-Lipoic Acid Supplementation: A Systematic Review and Meta-Analysis of Randomized Placebo-Controlled Clinical Studies" Antioxidants 9, no. 10: 1011. https://doi.org/10.3390/antiox9101011
APA StyleFogacci, F., Rizzo, M., Krogager, C., Kennedy, C., Georges, C. M. G., Knežević, T., Liberopoulos, E., Vallée, A., Pérez-Martínez, P., Wenstedt, E. F. E., Šatrauskienė, A., Vrablík, M., & Cicero, A. F. G. (2020). Safety Evaluation of α-Lipoic Acid Supplementation: A Systematic Review and Meta-Analysis of Randomized Placebo-Controlled Clinical Studies. Antioxidants, 9(10), 1011. https://doi.org/10.3390/antiox9101011









