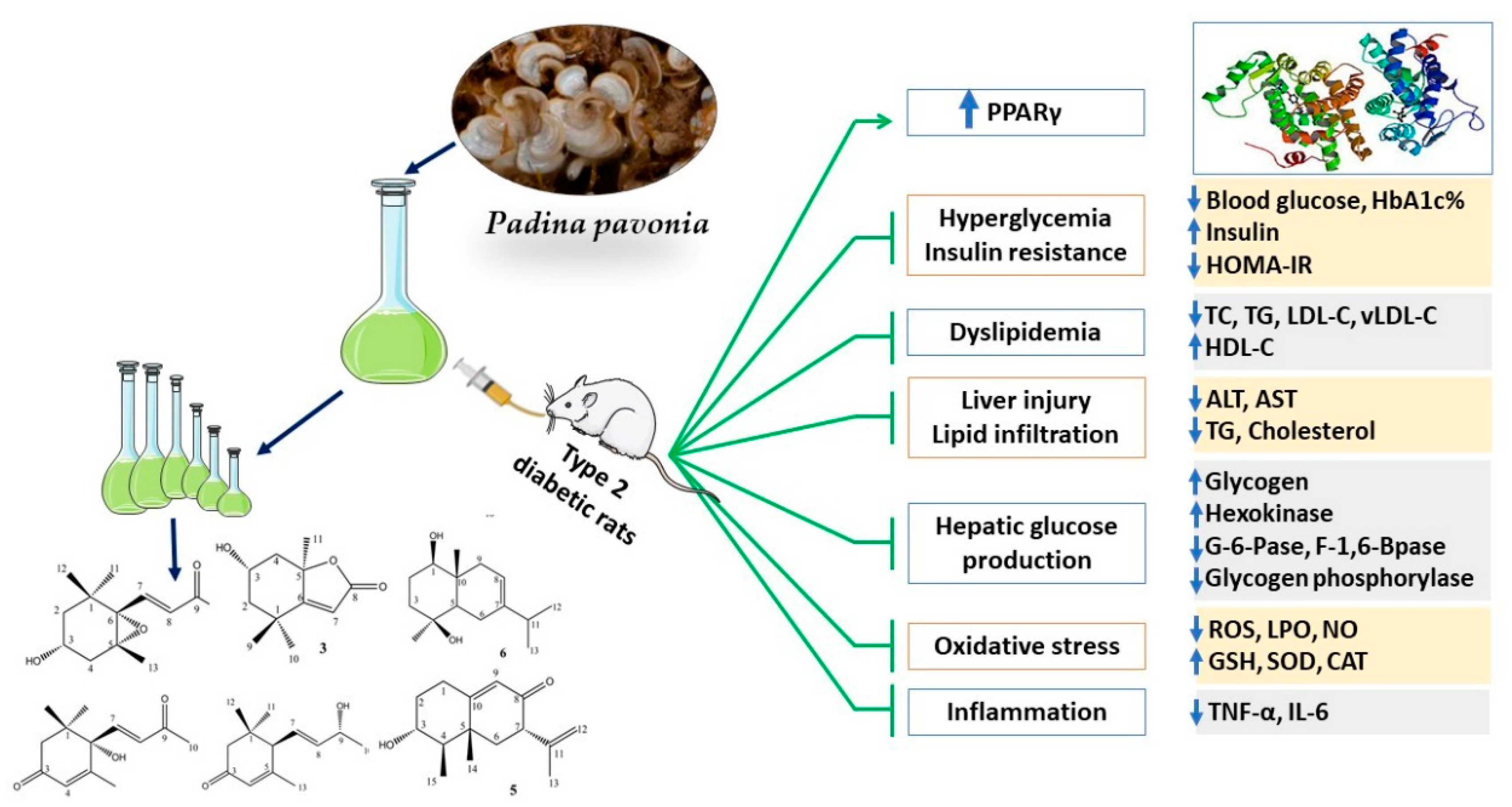
Consumption of Terpenoids-Rich Padina pavonia Extract Attenuates Hyperglycemia, Insulin Resistance and Oxidative Stress, and Upregulates PPARγ in a Rat Model of Type 2 Diabetes
Abstract
1. Introduction
2. Materials and Methods
2.1. Collection of P. pavonia, Extraction, and Isolation
2.1.1. General Experimental Procedures
2.1.2. Extraction and Isolation
3α-Hydroxy-5,6-epoxy-7-megastigmen-9-one (1)
(+)-Dehydrovomifoliol (2)
Loliolide (3)
(6R,7E,9R)-9-hydroxy-4,7-megastigmadien-3-one (4)
Petasol (5)
Oplodiol (7-Eudesmene-1b,4b-diol) (6)
2.2. Animals and Treatments
2.3. Oral Glucose Tolerance Test (OGTT)
2.4. Determination of HBA1c, Insulin, and HOMA-IR
2.5. Determination of Glycogen and Glucose-Metabolizing Enzymes
2.6. Determination of Serum and Liver Lipids
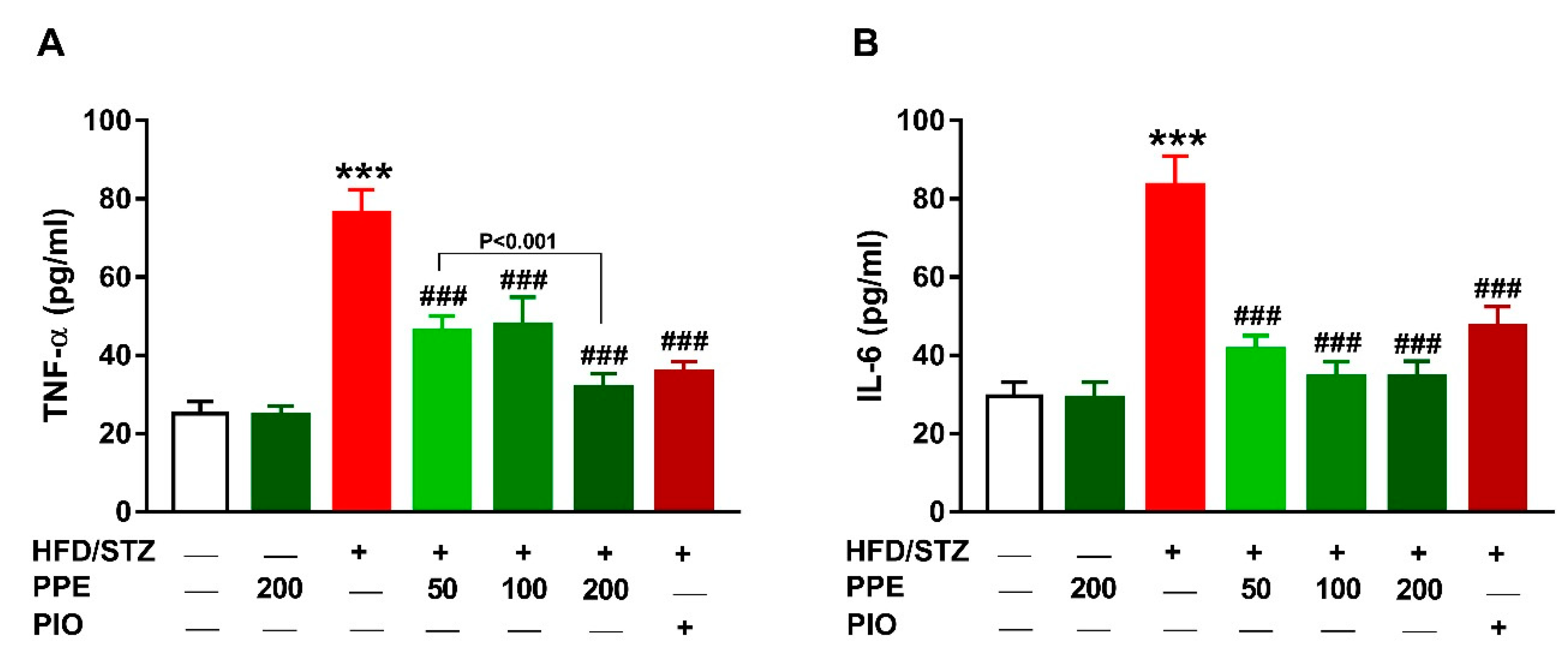
2.7. Determination of Aminotransferases and Cytokines
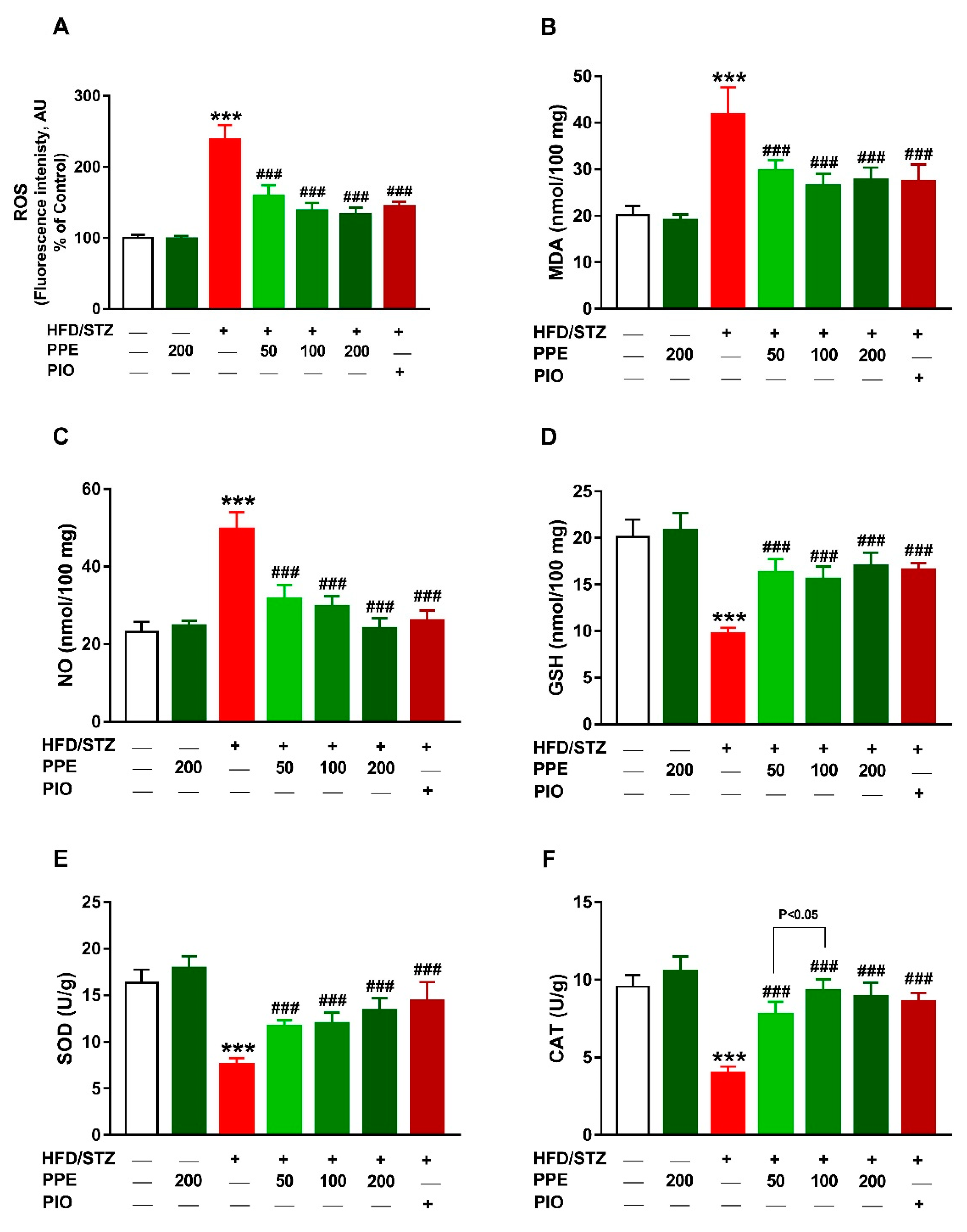
2.8. Determination of Oxidative Stress Markers and Antioxidants
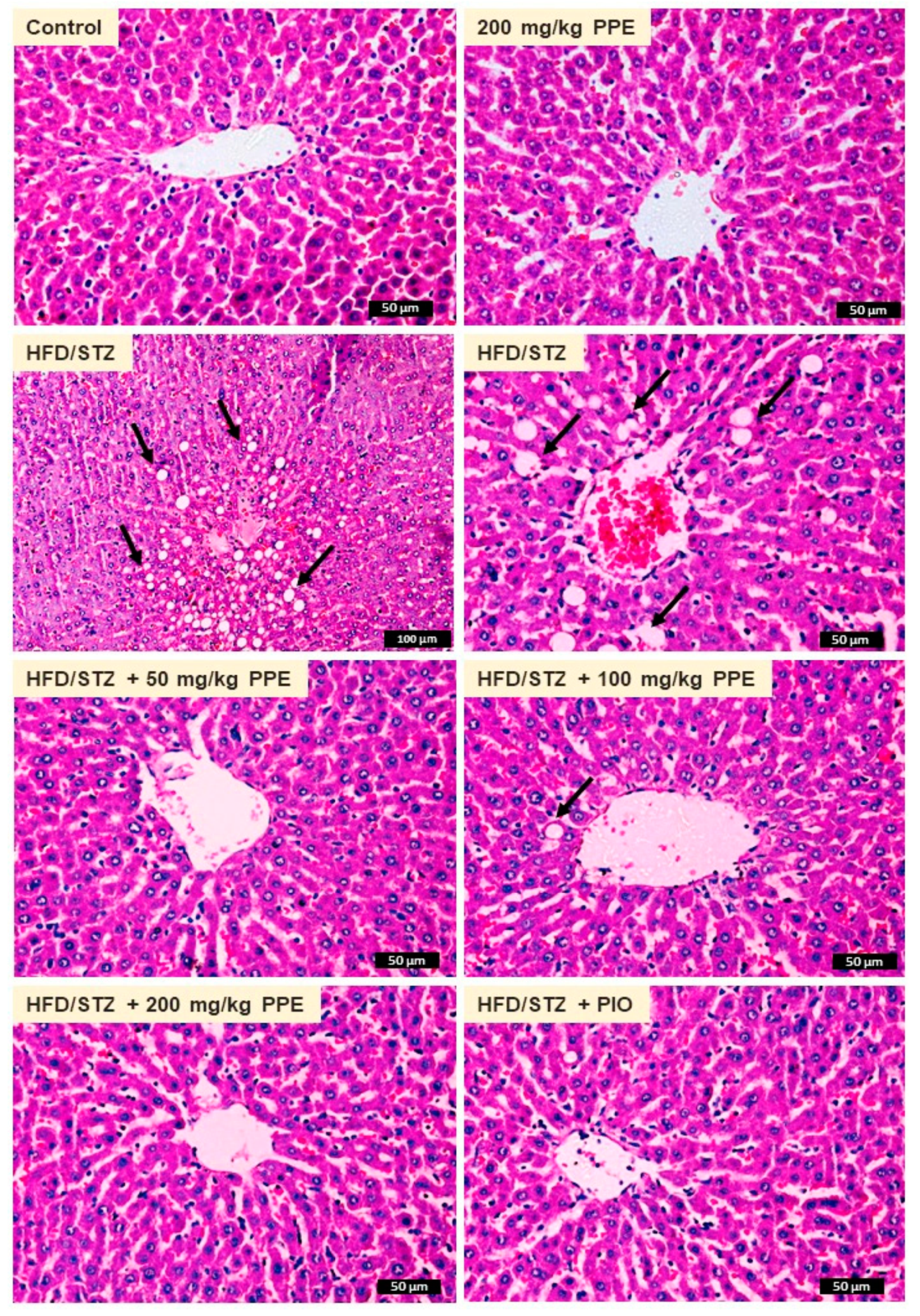
2.9. Histology
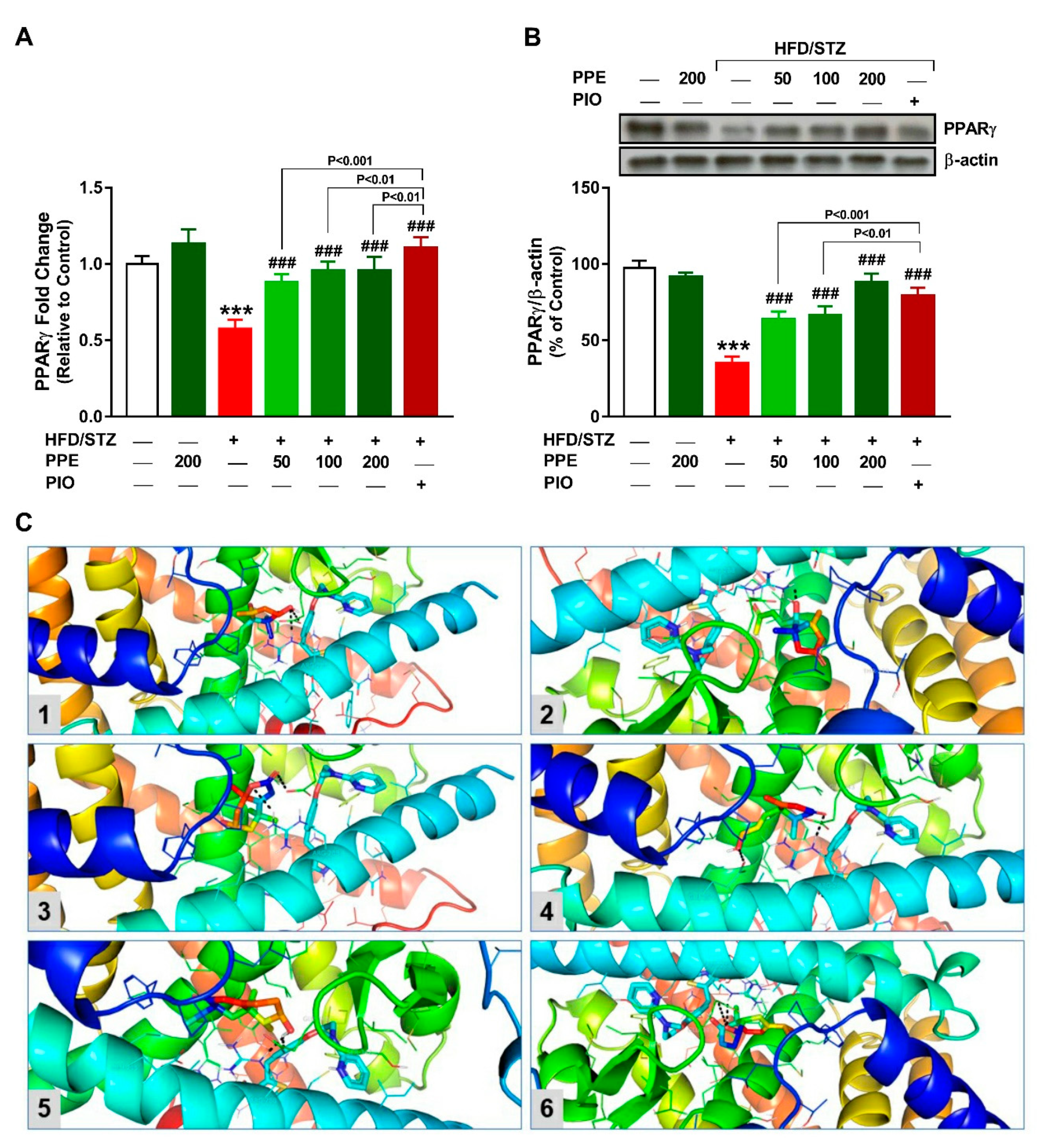
2.10. PPARγ Expression
2.11. Molecular Docking
2.12. Statistical Analysis
3. Results
3.1. Identification of the Isolated Compounds from PPE
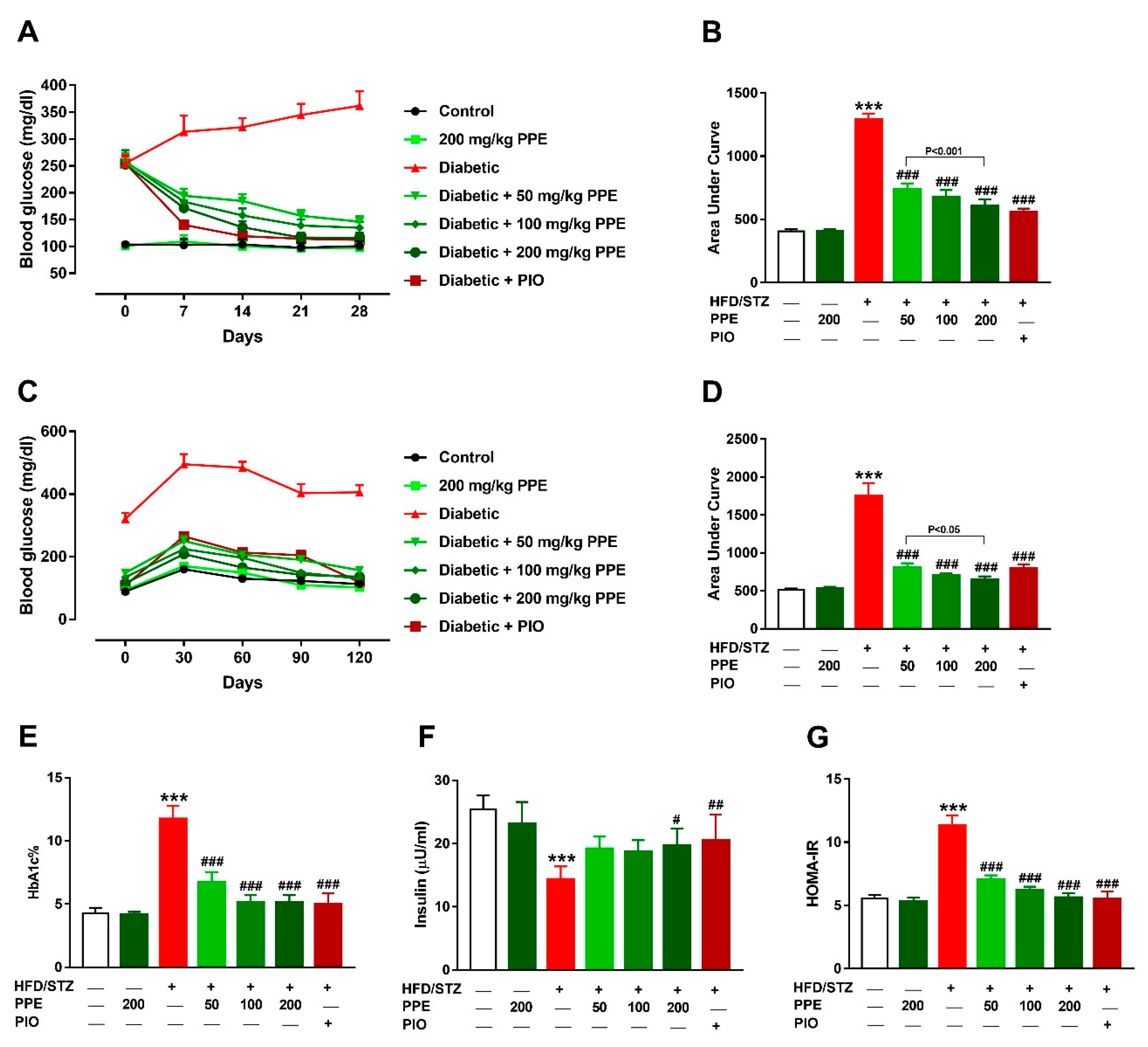
3.2. PPE Ameliorates Hyperglycemia and IR in HFD/STZ-Induced Rats
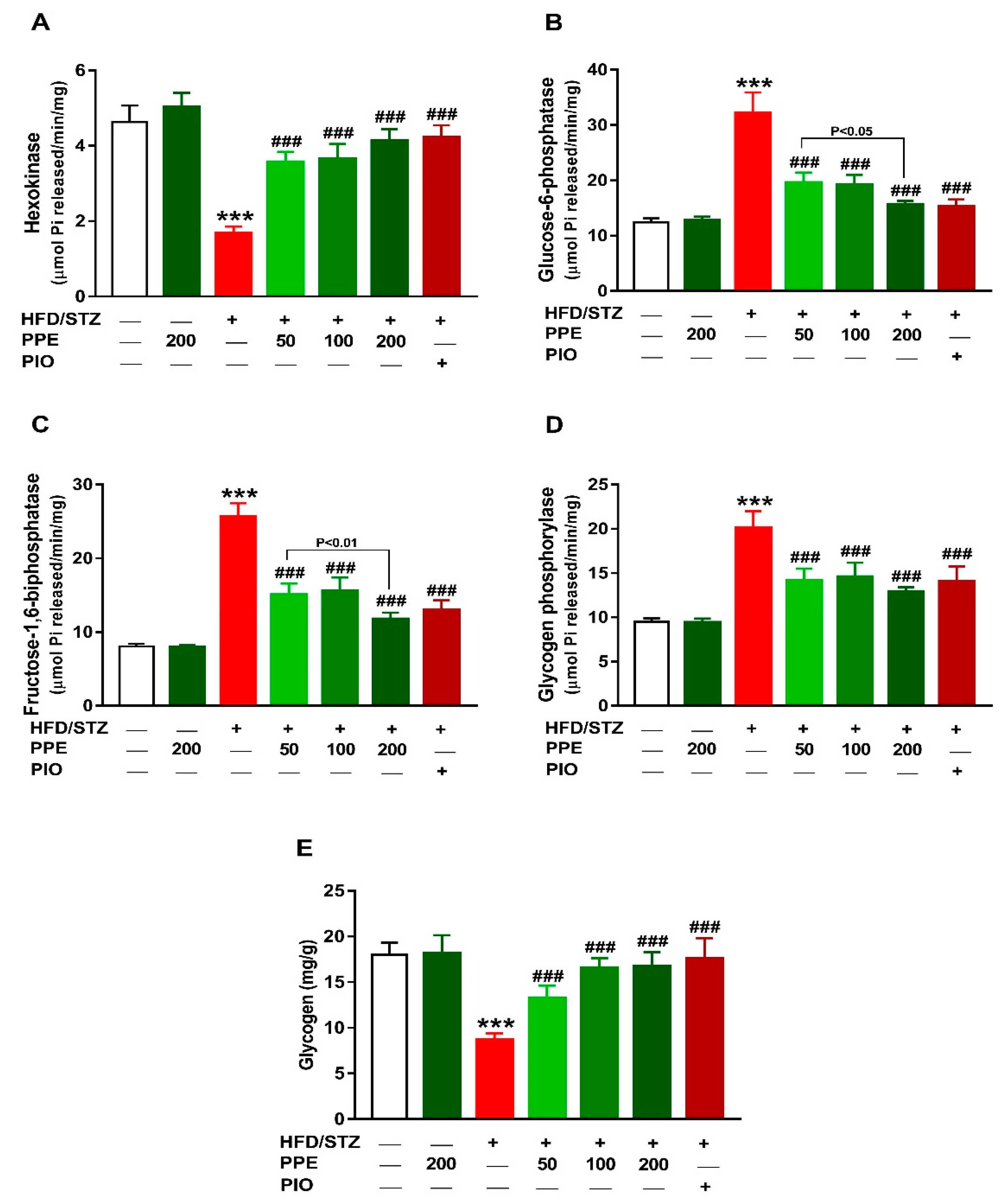
3.3. PPE Improves Liver Glucose Metabolizing Enzymes and Glycogen in HFD/STZ-Induced Rats
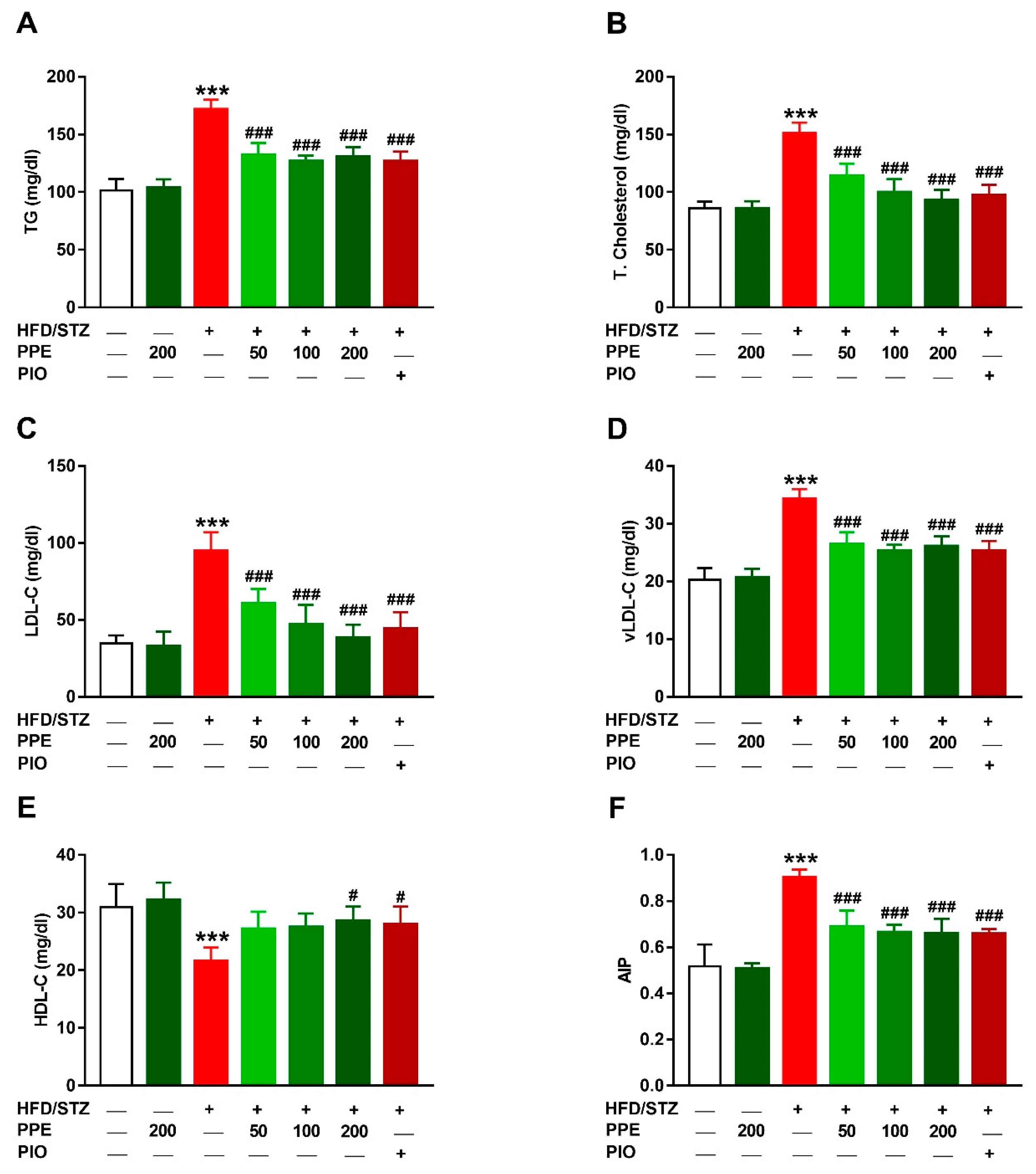
3.4. PPE Attenuates Hyperlipidemia in HFD/STZ-Induced Rats
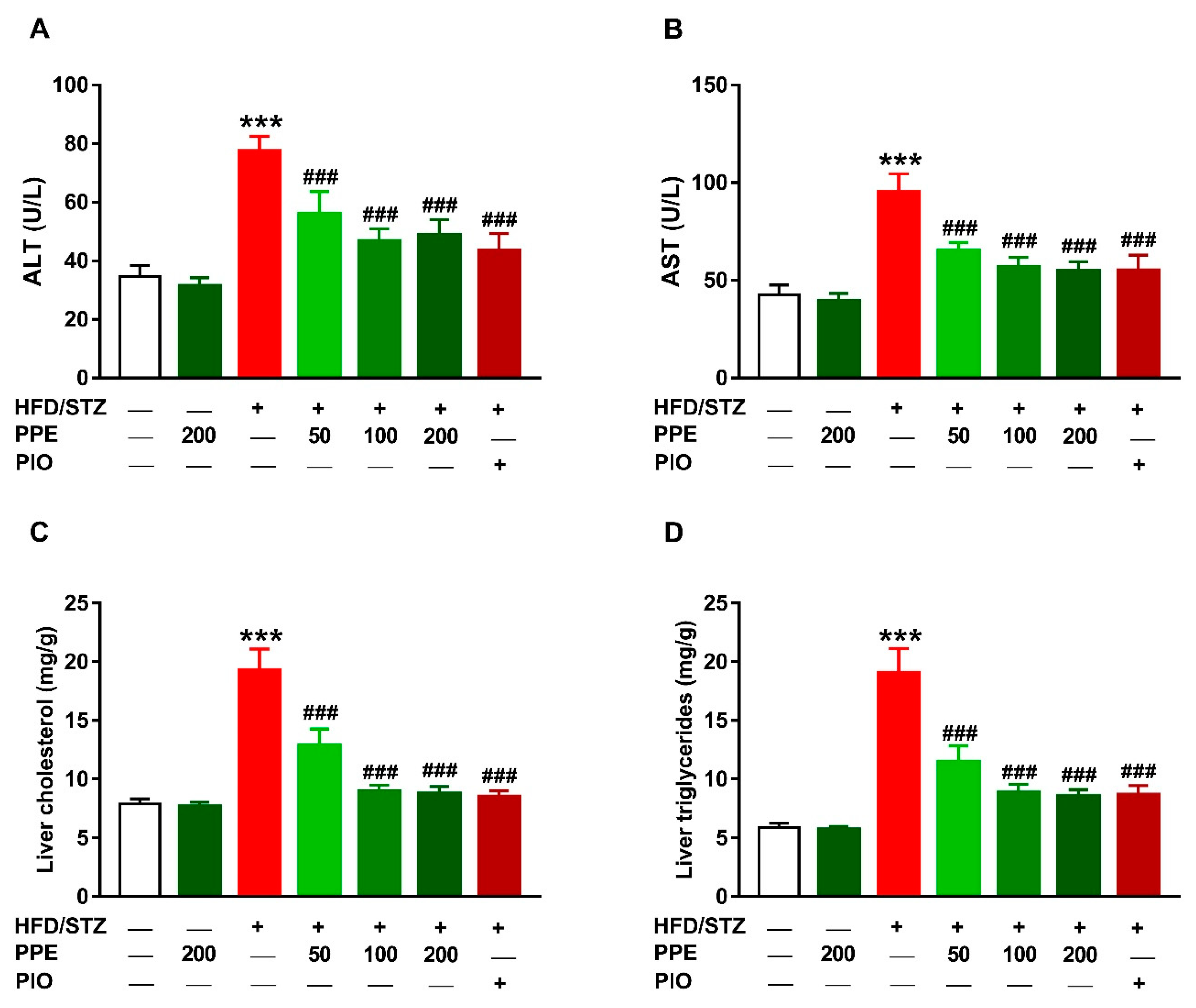
3.5. PPE Prevents Liver Injury and Lipid Accumulation in HFD/STZ-Induced Rats
3.6. PPE Attenuates Oxidative Stress and Enhances Antioxidants in HFD/STZ-Induced Rats
3.7. PPE Suppresses Inflammation in HFD/STZ-Induced Rats
3.8. PPE Upregulates Hepatic PPARγ in HFD/STZ-Induced Rats
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cho, N.H.; Shaw, J.E.; Karuranga, S.; Huang, Y.; Da Rocha Fernandes, J.D.; Ohlrogge, A.W.; Malanda, B. Idf diabetes atlas: Global estimates of diabetes prevalence for 2017 and projections for 2045. Diabetes Res. Clin. Pract. 2018, 138, 271–281. [Google Scholar] [CrossRef] [PubMed]
- Kahn, S.E.; Cooper, M.E.; Del Prato, S. Pathophysiology and treatment of type 2 diabetes: Perspectives on the past, present, and future. Lancet 2014, 383, 1068–1083. [Google Scholar] [CrossRef]
- Jellinger, P.S. Metabolic consequences of hyperglycemia and insulin resistance. Clin. Cornerstone 2007, 8, S30–S42. [Google Scholar] [CrossRef]
- Armstrong, M.J.; Adams, L.A.; Canbay, A.; Syn, W.K. Extrahepatic complications of nonalcoholic fatty liver disease. Hepatology 2014, 59, 1174–1197. [Google Scholar] [CrossRef] [PubMed]
- Petersen, M.C.; Shulman, G.I. Mechanisms of insulin action and insulin resistance. Physiol. Rev. 2018, 98, 2133–2223. [Google Scholar] [CrossRef] [PubMed]
- Halban, P.A.; Polonsky, K.S.; Bowden, D.W.; Hawkins, M.A.; Ling, C.; Mather, K.J.; Powers, A.C.; Rhodes, C.J.; Sussel, L.; Weir, G.C. Beta-cell failure in type 2 diabetes: Postulated mechanisms and prospects for prevention and treatment. Diabetes Care 2014, 37, 1751–1758. [Google Scholar] [CrossRef] [PubMed]
- Taskinen, M.R. Diabetic dyslipidaemia: From basic research to clinical practice. Diabetologia 2003, 46, 733–749. [Google Scholar] [CrossRef] [PubMed]
- Samuel, V.T.; Shulman, G.I. Mechanisms for insulin resistance: Common threads and missing links. Cell 2012, 148, 852–871. [Google Scholar] [CrossRef] [PubMed]
- Newsholme, P.; Cruzat, V.F.; Keane, K.N.; Carlessi, R.; De Bittencourt, P.I., Jr. Molecular mechanisms of ros production and oxidative stress in diabetes. Biochem. J. 2016, 473, 4527–4550. [Google Scholar] [CrossRef]
- Tiwari, B.K.; Pandey, K.B.; Abidi, A.B.; Rizvi, S.I. Markers of oxidative stress during diabetes mellitus. J. Biomark. 2013, 2013, 378790. [Google Scholar] [CrossRef]
- Bandeira Sde, M.; Guedes Gda, S.; Da Fonseca, L.J.; Pires, A.S.; Gelain, D.P.; Moreira, J.C.; Rabelo, L.A.; Vasconcelos, S.M.; Goulart, M.O. Characterization of blood oxidative stress in type 2 diabetes mellitus patients: Increase in lipid peroxidation and sod activity. Oxidative Med. Cell. Longev. 2012, 2012, 819310. [Google Scholar] [CrossRef] [PubMed]
- Rizvi, S.I.; Zaid, M.A.; Anis, R.; Mishra, N. Protective role of tea catechins against oxidation-induced damage of type 2 diabetic erythrocytes. Clin. Exp. Pharmcol. Physiol. 2005, 32, 70–75. [Google Scholar] [CrossRef] [PubMed]
- Pickup, J.C.; Chusney, G.D.; Thomas, S.M.; Burt, D. Plasma interleukin-6, tumour necrosis factor α and blood cytokine production in type 2 diabetes. Life Sci. 2000, 67, 291–300. [Google Scholar] [CrossRef]
- Sahin, N.; Orhan, C.; Erten, F.; Tuzcu, M.; Defo Deeh, P.B.; Ozercan, I.H.; Juturu, V.; Kazim, S. Effects of allyl isothiocyanate on insulin resistance, oxidative stress status, and transcription factors in high-fat diet/streptozotocin-induced type 2 diabetes mellitus in rats. J. Biochem. Mol. Toxicol. 2019, 33, e22328. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, A.M.; Ashour, M.B.; Abdel-Moneim, A.; Ahmed, O.M. Hesperidin and naringin attenuate hyperglycemia-mediated oxidative stress and proinflammatory cytokine production in high fat fed/streptozotocin-induced type 2 diabetic rats. J. Diabetes Complicat. 2012, 26, 483–490. [Google Scholar] [CrossRef] [PubMed]
- Müller, S.; Martin, S.; Koenig, W.; Hanifi-Moghaddam, P.; Rathmann, W.; Haastert, B.; Giani, G.; Illig, T.; Thorand, B.; Kolb, H. Impaired glucose tolerance is associated with increased serum concentrations of interleukin 6 and co-regulated acute-phase proteins but not tnf-alpha or its receptors. Diabetologia 2002, 45, 805–812. [Google Scholar] [CrossRef]
- Senn, J.J.; Klover, P.J.; Nowak, I.A.; Mooney, R.A. Interleukin-6 induces cellular insulin resistance in hepatocytes. Diabetes 2002, 51, 3391–3399. [Google Scholar] [CrossRef]
- Lang, C.H.; Dobrescu, C.; Bagby, G.J. Tumor necrosis factor impairs insulin action on peripheral glucose disposal and hepatic glucose output. Endocrinology 1992, 130, 43–52. [Google Scholar] [CrossRef]
- Green, A.; Dobias, S.B.; Walters, D.J.A.; Brasier, A.R. Tumor necrosis factor increases the rate of lipolysis in primary cultures of adipocytes without altering levels of hormone-sensitive lipase. Endocrinology 1994, 134, 2581–2588. [Google Scholar] [CrossRef]
- Del Aguila, L.F.; Claffey, K.P.; Kirwan, J.P. Tnf-α impairs insulin signaling and insulin stimulation of glucose uptake in C2C12 muscle cells. Am. J. Physiol. Endocrinol. Metab. 1999, 276, E849–E855. [Google Scholar] [CrossRef]
- Han, L.; Shen, W.-J.; Bittner, S.; Kraemer, F.B.; Azhar, S. PPARs: Regulators of metabolism and as therapeutic targets in cardiovascular disease. Part ii: PPAR-β/δ and PPAR-γ. Future Cardiol. 2017, 13, 279–296. [Google Scholar] [CrossRef] [PubMed]
- Tontonoz, P.; Spiegelman, B.M. Fat and beyond: The diverse biology of ppargamma. Annu. Rev. Biochem. 2008, 77, 289–312. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Liu, T.; Ng, C.Y.; Li, G. Diabetes mellitus and atrial remodeling: Mechanisms and potential upstream therapies. Cardiovasc. Ther. 2014, 32, 233–241. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, A.M.; Hozayen, W.G.; Hasan, I.H.; Shaban, E.; Bin-Jumah, M. Umbelliferone ameliorates CCl4-induced liver fibrosis in rats by upregulating ppargamma and attenuating oxidative stress, inflammation, and tgf-beta1/smad3 signaling. Inflammation 2019, 42, 1103–1116. [Google Scholar] [CrossRef]
- Mahmoud, A.M.; Mohammed, H.M.; Khadrawy, S.M.; Galaly, S.R. Hesperidin protects against chemically induced hepatocarcinogenesis via modulation of nrf2/are/ho-1, ppargamma and tgf-beta1/smad3 signaling, and amelioration of oxidative stress and inflammation. Chem. Biol. Interact. 2017, 277, 146–158. [Google Scholar] [CrossRef]
- Mahmoud, A.M.; Hussein, O.E.; Hozayen, W.G.; Abd El-Twab, S.M. Methotrexate hepatotoxicity is associated with oxidative stress, and down-regulation of ppargamma and nrf2: Protective effect of 18beta-glycyrrhetinic acid. Chem. Biol. Interact. 2017, 270, 59–72. [Google Scholar] [CrossRef]
- Kang, H.; Seo, C.; Park, Y. Marine peptides and their anti-infective activities. Mar. Drugs 2015, 13, 618–654. [Google Scholar] [CrossRef]
- Kamenarska, Z.; Gasic, M.J.; Zlatovic, M.; Rasovic, A.; Sladic, D.; Kljajic, Z.; Stefanov, K.; Seizova, K.; Najdenski, H.; Kujumgiev, A.; et al. Chemical composition of the brown alga Padina pavonia (l.) gaill. From the adriatic sea. Bot. Mar. 2002, 45, 339. [Google Scholar] [CrossRef]
- Magdel-Din Hussein, M.; Abdel-Aziz, A.; Mohamed Salem, H. Sulphated heteropolysaccharides from Padina pavonia. Phytochemistry 1980, 19, 2131–2132. [Google Scholar] [CrossRef]
- Mahmoud, A.M.; El-Derby, A.M.; Elsayed, K.N.M.; Abdella, E.M. Brown seaweeds ameliorate renal alterations in mice treated with the carcinogen azoxymethane. Int. J. Pharm Pharm. Sci. 2014, 6, 365–369. [Google Scholar]
- Mahmoud, A.M.; Abdella, E.M.; El-Derby, A.M.; Abdella, E.M. Protective effects of Turbinaria ornata and Padina pavonia against azoxymethane-induced colon carcinogenesis through modulation of ppar gamma, nf-kappab and oxidative stress. Phytother. Res. 2015, 29, 737–748. [Google Scholar] [CrossRef] [PubMed]
- Abdella, E.; Mahmoud, A.; El-Derby, A. Brown seaweeds protect against azoxymethane-induced hepatic repercussions through up-regulation of peroxisome proliferator activated receptor gamma and attenuation of oxidative stress. Pharm. Biol. 2016, 54, 2496–2504. [Google Scholar] [CrossRef] [PubMed]
- Barbalace, M.C.; Malaguti, M.; Giusti, L.; Lucacchini, A.; Hrelia, S.; Angeloni, C. Anti-inflammatory activities of marine algae in neurodegenerative diseases. Int. J. Mol. Sci. 2019, 20, 3061. [Google Scholar] [CrossRef] [PubMed]
- Abd El-Twab, S.M.; Mohamed, H.M.; Mahmoud, A.M. Taurine and pioglitazone attenuate diabetes-induced testicular damage by abrogation of oxidative stress and up-regulation of the pituitary-gonadal axis. Can. J. Physiol. Pharm. 2016, 94, 651–661. [Google Scholar] [CrossRef] [PubMed]
- Trinder, P. Determination of glucose in blood using glucose oxidase with an alternative oxygen acceptor. Ann. Clin. Biochem. 1969, 6, 24–27. [Google Scholar] [CrossRef]
- Haffner, S.M. Coronary heart disease in patients with diabetes. N. Engl. J. Med. 2000, 342, 1040–1042. [Google Scholar] [CrossRef]
- Seifter, S.; Dayton, S.; Novic, B.; Muntwyler, E. The estimation of glycogen with the anthrone reagent. Arch. Biochem. 1950, 25, 191–200. [Google Scholar]
- Brandstrup, N.; Kirk, J.E.; Bruni, C. The hexokinase and phosphoglucoisomerase activities of aortic and pulmonary artery tissue in individuals of various ages. J. Gerontol. 1957, 12, 166–171. [Google Scholar] [CrossRef]
- Koide, H.; Oda, T. Pathological occurrence of glucose-6-phosphatase in serum in liver diseases. Clin. Chim. Acta 1959, 4, 554–561. [Google Scholar]
- Freedland, R.A.; Harper, A.E. Metabolic adaptations in higher animals. V. The study of metabolic pathways by means of metabolic adaptations. J. Biol. Chem. 1959, 234, 1350–1354. [Google Scholar]
- Stalmans, W.; Hers, H.G. The stimulation of liver phosphorylase b by amp, fluoride and sulfate. A technical note on the specific determination of the a and b forms of liver glycogen phosphorylase. Eur. J. Biochem. 1975, 54, 341–350. [Google Scholar] [CrossRef]
- Fiske, C.; Subbarow, Y. The colourimetric determination of phosphorus. J. Biol. Chem. 1925, 66, 375–400. [Google Scholar]
- Ross, R. The pathogenesis of atherosclerosis. In Heart Disease: A Textbook of Cardiovascular Medicine, 4th ed.; Braunwald, E., Ed.; WB Saunders: Philadelphia, PA, USA, 1992; pp. 11061–11124. [Google Scholar]
- Folch, J.; Lees, M.; Sloane Stanley, G.H. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [PubMed]
- Hozayen, W.G.; Mahmoud, A.M.; Desouky, E.M.; El-Nahass, E.-S.; Soliman, H.A.; Farghali, A.A. Cardiac and pulmonary toxicity of mesoporous silica nanoparticles is associated with excessive ros production and redox imbalance in wistar rats. Biomed. Pharmacother. 2019, 109, 2527–2538. [Google Scholar] [CrossRef] [PubMed]
- Preuss, H.G.; Jarrell, S.T.; Scheckenbach, R.; Lieberman, S.; Anderson, R.A. Comparative effects of chromium, vanadium and Gymnema sylvestre on sugar-induced blood pressure elevations in shr. J. Am. Coll. Nutr. 1998, 17, 116–123. [Google Scholar] [CrossRef] [PubMed]
- Grisham, M.B.; Johnson, G.G.; Lancaster, J.R., Jr. Quantitation of nitrate and nitrite in extracellular fluids. Methods Enzymol. 1996, 268, 237–246. [Google Scholar] [PubMed]
- Beutler, E.; Duron, O.; Kelly, B.M. Improved method for the determination of blood glutathione. J. Lab. Clin. Med. 1963, 61, 882–888. [Google Scholar]
- Marklund, S.; Marklund, G. Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur. J. Biochem. 1974, 47, 469–474. [Google Scholar] [CrossRef]
- Cohen, G.; Dembiec, D.; Marcus, J. Measurement of catalase activity in tissue extracts. Anal. Biochem. 1970, 34, 30–38. [Google Scholar] [CrossRef]
- Mahmoud, A.M. Hesperidin protects against cyclophosphamide-induced hepatotoxicity by upregulation of pparγ and abrogation of oxidative stress and inflammation. Can. J. Physiol. Pharm. 2014, 92, 717–724. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative pcr and the 2(-delta delta c(t)) method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Pettersen, E.F.; Goddard, T.D.; Huang, C.C.; Couch, G.S.; Greenblatt, D.M.; Meng, E.C.; Ferrin, T.E. Ucsf chimera—A visualization system for exploratory research and analysis. J. Comput. Chem. 2004, 25, 1605–1612. [Google Scholar] [CrossRef] [PubMed]
- Nolte, R.T.; Wisely, G.B.; Westin, S.; Cobb, J.E.; Lambert, M.H.; Kurokawa, R.; Rosenfeld, M.G.; Willson, T.M.; Glass, C.K.; Milburn, M.V. Ligand binding and co-activator assembly of the peroxisome proliferator-activated receptor-γ. Nature 1998, 395, 137. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.U.; Yang, M.C.; Lee, K.H.; Kim, K.H.; Lee, K.R. Lignan and terpene constituents from the aerial parts of Saussurea pulchella. Arch. Pharmacal Res. 2007, 30, 1067–1074. [Google Scholar] [CrossRef] [PubMed]
- Häusler, M.; Montag, A. Isolation, identification and quantitative determination of the norisoprenoid (s)-(+)-dehydrovomifoliol in honey. Z. Lebensm. -Unters. Forsch. 1989, 189, 113–115. [Google Scholar] [CrossRef]
- He, Z.; Zhang, A.; Ding, L.; Lei, X.; Sun, J.; Zhang, L. Chemical composition of the green alga codium divaricatum holmes. Fitoterapia 2010, 81, 1125–1128. [Google Scholar] [CrossRef]
- Park, J.-H.; Lee, D.-G.; Yeon, S.-W.; Kwon, H.-S.; Ko, J.-H.; Shin, D.-J.; Park, H.-S.; Kim, Y.-S.; Bang, M.-H.; Baek, N.-I. Isolation of megastigmane sesquiterpenes from the silkworm (Bombyx mori l.) droppings and their promotion activity on ho-1 and sirt1. Arch. Pharmacal Res. 2011, 34, 533–542. [Google Scholar] [CrossRef]
- Liu, X.; Wu, Q.X.; Shi, Y.P. Terpenoids from the flower of Cacalia tangutica. J. Chin. Chem. Soc. 2005, 52, 369–374. [Google Scholar] [CrossRef]
- Kwon, H.C.; Lee, K.R. Phytochemical constituents of Artemisia japonica ssp. Littoricola. Arch. Pharmacal Res. 2001, 24, 194–197. [Google Scholar] [CrossRef]
- Laakso, M. How good a marker is insulin level for insulin resistance? Am. J. Epidemiol. 1993, 137, 959–965. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.S.; Li, P.; Huh, J.Y.; Hwang, I.J.; Lu, M.; Kim, J.I.; Ham, M.; Talukdar, S.; Chen, A.; Lu, W.J.; et al. Inflammation is necessary for long-term but not short-term high-fat diet-induced insulin resistance. Diabetes 2011, 60, 2474–2483. [Google Scholar] [CrossRef] [PubMed]
- Breyer, M.D.; Böttinger, E.; Brosius, F.C., III; Coffman, T.M.; Harris, R.C.; Heilig, C.W.; Sharma, K. Mouse models of diabetic nephropathy. J. Am. Soc. Nephrol. 2005, 16, 27–45. [Google Scholar] [CrossRef] [PubMed]
- Association, A.D. Standards of medical care in diabetes—2014. Diabetes Care 2014, 37 (Suppl. 1), S14–S80. [Google Scholar] [CrossRef] [PubMed]
- Guex, C.G.; Reginato, F.Z.; De Jesus, P.R.; Brondani, J.C.; Lopes, G.H.H.; Bauermann, L.F. Antidiabetic effects of Olea europaea l. Leaves in diabetic rats induced by high-fat diet and low-dose streptozotocin. J. Ethnopharmacol. 2019, 235, 1–7. [Google Scholar] [CrossRef]
- Liao, Z.; Zhang, J.; Liu, B.; Yan, T.; Xu, F.; Xiao, F.; Wu, B.; Bi, K.; Jia, Y. Polysaccharide from okra (Abelmoschus esculentus (l.) moench) improves antioxidant capacity via pi3k/akt pathways and nrf2 translocation in a type 2 diabetes model. Molecules 2019, 24, 1906. [Google Scholar] [CrossRef]
- Mahmoud, A.M.; Germoush, M.O.; Elgebaly, H.A.; Elsayed, K.N.M.; Hassan, S.; Mousa, N.M.; Hussein, O.E. Antidiabetic and insulin sensitizing effects of Padina pavonia and Turbenaria ornata in streptozotocin/nicotinamide diabetic rats. Asian J. Pharm Clin. Res. 2014, 7, 74–78. [Google Scholar]
- Nordlie, R.C.; Foster, J.D.; Lange, A.J. Regulation of glucose production by the liver. Annu. Rev. Nutr. 1999, 19, 379–406. [Google Scholar] [CrossRef]
- Gothandam, K.; Ganesan, V.S.; Ayyasamy, T.; Ramalingam, S. Antioxidant potential of theaflavin ameliorates the activities of key enzymes of glucose metabolism in high fat diet and streptozotocin—Induced diabetic rats. Redox Rep. 2019, 24, 41–50. [Google Scholar] [CrossRef]
- Mishra, C.; Khalid, M.A.; Fatima, N.; Singh, B.; Tripathi, D.; Waseem, M.; Mahdi, A.A. Effects of citral on oxidative stress and hepatic key enzymes of glucose metabolism in streptozotocin/high-fat-diet induced diabetic dyslipidemic rats. Iran. J. Basic Med. Sci. 2019, 22, 49–57. [Google Scholar]
- Gupta, D.; Raju, J.; Prakash, J.; Baquer, N.Z. Change in the lipid profile, lipogenic and related enzymes in the livers of experimental diabetic rats: Effect of insulin and vanadate. Diabetes Res. Clin. Pract. 1999, 46, 1–7. [Google Scholar] [CrossRef]
- Ahmed, O.M.; Moneim, A.A.; Mahmoud, A.M.; Yazid, I.A. Antihyperglycemic, antihyperlipidemic and antioxidant effects and the probable mechanisms of action of Ruta graveolens infusion and rutin in nicotinamide-streptozotocin-induced diabetic rats. Diabetol. Croat. 2010, 39, 15–35. [Google Scholar]
- Pilkis, S.J.; Claus, T.H. Hepatic gluconeogenesis/glycolysis: Regulation and structure/function relationships of substrate cycle enzymes. Annu. Rev. Nutr. 1991, 11, 465–515. [Google Scholar] [CrossRef] [PubMed]
- Roden, M.; Bernroider, E. Hepatic glucose metabolism in humans—Its role in health and disease. Best Pract. Res. Clin. Endocrinol. Metab. 2003, 17, 365–383. [Google Scholar] [CrossRef]
- Rui, L. Energy metabolism in the liver. Compr. Physiol. 2014, 4, 177–197. [Google Scholar] [PubMed]
- Golden, S.; Wals, P.A.; Okajima, F.; Katz, J. Glycogen synthesis by hepatocytes from diabetic rats. Biochem. J. 1979, 182, 727–734. [Google Scholar] [CrossRef]
- Postic, C.; Dentin, R.; Girard, J. Role of the liver in the control of carbohydrate and lipid homeostasis. Diabetes Metab. 2004, 30, 398–408. [Google Scholar] [CrossRef]
- Reaven, G.M. Compensatory hyperinsulinemia and the development of an atherogenic lipoprotein profile: The price paid to maintain glucose homeostasis in insulin-resistant individuals. Endocrinol. Metab. Clin. N. Am. 2005, 34, 49–62. [Google Scholar] [CrossRef]
- Ahmed, O.M.; Mahmoud, A.M.; Abdel-Moneim, A.; Ashour, M.B. Antidiabetic effects of hesperidin and naringin in type 2 diabetic rats. Diabetol. Croat. 2012, 41, 53–67. [Google Scholar]
- Betteridge, D.J. Effects of pioglitazone on lipid and lipoprotein metabolism. Diabetes Obes. Metab. 2007, 9, 640–647. [Google Scholar] [CrossRef]
- Kazumi, T.; Hirano, T.; Odaka, H.; Ebara, T.; Amano, N.; Hozumi, T.; Ishida, Y.; Yoshino, G. Vldl triglyceride kinetics in wistar fatty rats, an animal model of niddm: Effects of dietary fructose alone or in combination with pioglitazone. Diabetes 1996, 45, 806–811. [Google Scholar] [CrossRef] [PubMed]
- Levinthal, G.N.; Tavill, A.S. Liver disease and diabetes mellitus. Clin. Diabetes 1999, 17, 73–81. [Google Scholar]
- Nannipieri, M.; Gonzales, C.; Baldi, S.; Posadas, R.; Williams, K.; Haffner, S.M.; Stern, M.P.; Ferrannini, E. Liver enzymes, the metabolic syndrome, and incident diabetes: The mexico city diabetes study. Diabetes Care 2005, 28, 1757–1762. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, J.; Nazratun Nafizah, A.H.; Zariyantey, A.H.; Budin, S.B. Mechanisms of diabetes-induced liver damage: The role of oxidative stress and inflammation. Sultan Qaboos Univ. Med. J. 2016, 16, e132–e141. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, A.M. Exercise amaliorates metabolic disturbances and oxidative stress in diabetic cardiomyopathy: Possible underlying mechanisms. In Exercise for Cardiovascular Disease Prevention and Treatment: From Molecular to Clinical, Part 1; Xiao, J., Ed.; Springer: Singapore, 2017; pp. 207–230. [Google Scholar]
- Aladaileh, S.H.; Abukhalil, M.H.; Saghir, S.A.M.; Hanieh, H.; Alfwuaires, M.A.; Almaiman, A.A.; Bin-Jumah, M.; Mahmoud, A.M. Galangin activates nrf2 signaling and attenuates oxidative damage, inflammation, and apoptosis in a rat model of cyclophosphamide-induced hepatotoxicity. Biomolecules 2019, 9, 346. [Google Scholar] [CrossRef] [PubMed]
- Ranneh, Y.; Akim, A.M.; Hamid, H.A.; Khazaai, H.; Fadel, A.; Mahmoud, A.M. Stingless bee honey protects against lipopolysaccharide induced-chronic subclinical systemic inflammation and oxidative stress by modulating nrf2, nf-κb and p38 mapk. Nutr. Metab. 2019, 16, 15. [Google Scholar] [CrossRef]
- Elgebaly, H.A.; Mosa, N.M.; Allach, M.; El-Massry, K.F.; El-Ghorab, A.H.; Al Hroob, A.M.; Mahmoud, A.M. Olive oil and leaf extract prevent fluoxetine-induced hepatotoxicity by attenuating oxidative stress, inflammation and apoptosis. Biomed. Pharm. 2018, 98, 446–453. [Google Scholar] [CrossRef]
- Mahmoud, A.M.; Hussein, O.E.; Abd El-Twab, S.M.; Hozayen, W.G. Ferulic acid protects against methotrexate nephrotoxicity via activation of nrf2/are/ho-1 signaling and pparγ, and suppression of nf-κb/nlrp3 inflammasome axis. Food Funct. 2019, 10, 4593–4607. [Google Scholar] [CrossRef]
- Abd El-Twab, S.M.; Hussein, O.E.; Hozayen, W.G.; Bin-Jumah, M.; Mahmoud, A.M. Chicoric acid prevents methotrexate-induced kidney injury by suppressing nf-κb/nlrp3 inflammasome activation and up-regulating nrf2/are/ho-1 signaling. Inflamm. Res. 2019, 68, 511–523. [Google Scholar] [CrossRef]
- Kersten, S.; Desvergne, B.; Wahli, W. Roles of ppars in health and disease. Nature 2000, 405, 421–424. [Google Scholar] [CrossRef]
- Remels, A.H.; Langen, R.C.; Gosker, H.R.; Russell, A.P.; Spaapen, F.; Voncken, J.W.; Schrauwen, P.; Schols, A.M. Ppargamma inhibits nf-kappab-dependent transcriptional activation in skeletal muscle. Am. J. Physiol. Endocrinol. Metab. 2009, 297, E174–E183. [Google Scholar] [CrossRef] [PubMed]
- Okuno, Y.; Matsuda, M.; Miyata, Y.; Fukuhara, A.; Komuro, R.; Shimabukuro, M.; Shimomura, I. Human catalase gene is regulated by peroxisome proliferator activated receptor-gamma through a response element distinct from that of mouse. Endocr. J. 2010, 57, 303–309. [Google Scholar] [CrossRef] [PubMed]
- Hwang, J.; Kleinhenz, D.J.; Lassegue, B.; Griendling, K.K.; Dikalov, S.; Hart, C.M. Peroxisome proliferator-activated receptor-gamma ligands regulate endothelial membrane superoxide production. Am. J. Physiol. Cell Physiol. 2005, 288, C899–C905. [Google Scholar] [CrossRef] [PubMed]
- Park, S.H.; Choi, E.; Kim, S.; Kim, D.S.; Kim, J.H.; Chang, S.; Choi, J.S.; Park, K.J.; Roh, K.-B.; Lee, J.; et al. Oxidative stress-protective and anti-melanogenic effects of loliolide and ethanol extract from fresh water green algae, prasiola japonica. Int. J. Mol. Sci. 2018, 19, 2825. [Google Scholar] [CrossRef] [PubMed]

| 1 | 2 | 3 | ||||
|---|---|---|---|---|---|---|
| Position | δH (J in Hz) | δC | δH (J in Hz) | δC | δH (J in Hz) | δC |
| 1 | 37.58 | 42.51 | 35.38 | |||
| 2 | 1.66 (1H, m, H-2α) 1.33 (1H, m, H-2β) | 49.21 | 2.64 (1H, d, J = 16.7, H-2α), 2.41 (1H, d, J = 16.7, H-2β) | 48.95 | 1.94 (1H, dd, J = 13.4; 2.7, H-2α), 1.33 (1H, dt, J = 13.11, 3.4, H-2β) | 50.14 |
| 3 | 3.89 (1H, m) | 63.72 | 197.87 | 4.01 (1H, m, H-3) | 63.75 | |
| 4 | 1.71 (1H, m, H-4α), 2.43 (1H, dd, J = 13.9, 4.8, H-4β) | 41.49 | 6.12 (1H, br. s) | 129.11 | 2.87 (1H, dd, J = 13.4; 2.7, H-4α), 1.22 (1H, dt, J = 12.67, 3.6, H-4β) | 48.87 |
| 5 | 68.53 | 162.31 | 88.33 | |||
| 6 | 70.16 | 80.19 | 184.93 | |||
| 7 | δ 7.15 (1H, d, J = 14.8) | 143.57 | 6.91 (1H, d, J = 15.2) | 147.14 | 5.91 (1H, s, H-7) | 116.51 |
| 8 | 6.34 (1H, d, J = 14.8) | 133.65 | 6.52 (1H, d, J = 15.2) | 133.16 | 173.64 | |
| 9 | 196.44 | 197.15 | 1.28 (3H, s, H-9) | 25.26 | ||
| 10 | 2.32 (3H, s, H-10) | 27.76 | 2.29 (3H, s) | 30.01 | 1.17 (3H, s, H-10) | 30.29 |
| 11 | 0.97 (3H, s, H-11) | 26.25 | 1.22 (3H, s) | 22.94 | 1.62 (3H, s, H-11) | 26.62 |
| 12 | 1.19 (3H, s) | 28.92 | 1.11 (3H, s) | 25.49 | ||
| 13 | 1.19 (3H, s) | 20.87 | 1.93 (3H, d, J = 1.2) | 19.95 | ||
| 14 | ||||||
| 15 | ||||||
| 3-OH | 4.92 (1H, s,3-OH) | |||||
| 4 | 5 | 6 | ||||
|---|---|---|---|---|---|---|
| Position | δH (J in Hz) | δC | δH (J in Hz) | δC | δH (J in Hz) | δC |
| 1 | 36.25 | 2.67 (1H, tdd, J = 17.3, 6.1, 2.4), 2.52 (1H, ddd, J = 17.3, 5.9, 3.1) | 29.75 | 3.74 (1H, dd, J = 12.5, 4.5) | 80.14 | |
| 2 | 2.54 (1H, d, J = 15.9, H-2α), 2.12 (1H, d, J = 15.9, H-2 β) | 47.14 | 2.29 (1H, m) 1.51 (1H, m | 33.11 | 1.91–1.98 (2H, m, H-2, H-9), 1.83 (1H, dt, J = 14.4, 4) | 28.18 |
| 3 | 198.9.72 | 3.75 (1H, td, J = 12.1, 5.2) | 69.63 | 1.61–1.74 (2H, m) | 41.59 | |
| 4 | 5.93 (1H, s) | 125.98 | 1.42 (1H, m) | 49.81 | 71.64 | |
| 5 | 164.89 | 37.96 | 1.46 (1H, dd, J = 12.2, 6.1) | 48.75 | ||
| 6 | 2.87 (1H, d, J = 8.7) | 54.93 | 2.11 (1H, dd, J = 14.3, 5.8), 1.91 (1H, t, J = 14.8) | 39.52 | 2.05–2.18 (3H, m, H-6β, H-9) | 25.09 |
| 7 | 5.62 (1H, dd, J = 14.3, 8.7) | 126.36 | 3.54 (1H, dd, J = 15.2, 5.7) | 49.78 | 143.67 | |
| 8 | 5.71 (1H, dd, J = 14.3, 5.7) | 139.19 | 197.84 | 5.65 (1H, d, J = 5.1) | 118.32 | |
| 9 | 4.45 (1H, dq, J = 5.7, 6.2) | 65.87 | 5.84 (1H, d, J = 2.3) | 122.75 | 2.05–2.18 (3H, m, H-6β, H-9), 1.91–1.98 (2H, m, H-2, H-9) | 43.81 |
| 10 | 1.32 (3H, d, J = 6.2) | 22.91 | 164.98 | 39.15 | ||
| 11 | 1.05 (3H, s) | 26.78 | 141.57 | 2.44 (1 H, septet, J = 7.4) | 36.32 | |
| 12 | 1.14 (3H, s) | 27.36 | 5.12 (1H, br, t, J = 1.8) 4.91 (1H, br, s) | 110.56 | 1.10 (3H, d, J = 7.5) | 22.98 |
| 13 | 1.98 (3H, s) | 22.42 | 1.83 (3H, br, s) | 19.64 | 1.12 (3H, d, J = 7.5) | 22.75 |
| 14 | 1.26 (3H, s) | 15.38 | 1.02 (3H, s) | 13.53 | ||
| 15 | 1.17 (3H, d, J = 7.4) | 10.32 | 1.23 (3H, s) | 30.43 | ||
| Compound | Affinity (kcal/mol) | Polar Bonds |
|---|---|---|
| 1 | −7.0 | 2 |
| 2 | −6.2 | 1 |
| 3 | −6.7 | 1 |
| 4 | −7.1 | 2 |
| 5 | −7.4 | 2 |
| 6 | −7.0 | 2 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Germoush, M.O.; Elgebaly, H.A.; Hassan, S.; Kamel, E.M.; Bin-Jumah, M.; Mahmoud, A.M. Consumption of Terpenoids-Rich Padina pavonia Extract Attenuates Hyperglycemia, Insulin Resistance and Oxidative Stress, and Upregulates PPARγ in a Rat Model of Type 2 Diabetes. Antioxidants 2020, 9, 22. https://doi.org/10.3390/antiox9010022
Germoush MO, Elgebaly HA, Hassan S, Kamel EM, Bin-Jumah M, Mahmoud AM. Consumption of Terpenoids-Rich Padina pavonia Extract Attenuates Hyperglycemia, Insulin Resistance and Oxidative Stress, and Upregulates PPARγ in a Rat Model of Type 2 Diabetes. Antioxidants. 2020; 9(1):22. https://doi.org/10.3390/antiox9010022
Chicago/Turabian StyleGermoush, Mousa O., Hassan A. Elgebaly, Sherif Hassan, Emadeldin M. Kamel, May Bin-Jumah, and Ayman M. Mahmoud. 2020. "Consumption of Terpenoids-Rich Padina pavonia Extract Attenuates Hyperglycemia, Insulin Resistance and Oxidative Stress, and Upregulates PPARγ in a Rat Model of Type 2 Diabetes" Antioxidants 9, no. 1: 22. https://doi.org/10.3390/antiox9010022
APA StyleGermoush, M. O., Elgebaly, H. A., Hassan, S., Kamel, E. M., Bin-Jumah, M., & Mahmoud, A. M. (2020). Consumption of Terpenoids-Rich Padina pavonia Extract Attenuates Hyperglycemia, Insulin Resistance and Oxidative Stress, and Upregulates PPARγ in a Rat Model of Type 2 Diabetes. Antioxidants, 9(1), 22. https://doi.org/10.3390/antiox9010022







