Effects of Tannase and Ultrasound Treatment on the Bioactive Compounds and Antioxidant Activity of Green Tea Extract
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Preparation of Green Tea Extract
2.3. Treatment of Tannase and Ultrasound
2.4. Design by Response Surface Methodology
2.5. Determination of Antioxidant Activity
2.5.1. Trolox Equivalent Antioxidant Capacity (TEAC) Assay
2.5.2. 2,2-Diphenyl-1-picrylhydrazyl (DPPH) Radical Scavenging Assay
2.5.3. Ferric Reducing Antioxidant Power (FRAP) Assay
2.6. Phytochemical Analysis of the Extracts by HPLC
2.7. Statistical Analysis
3. Results and Discussions
3.1. Results of the Single-Factor Test
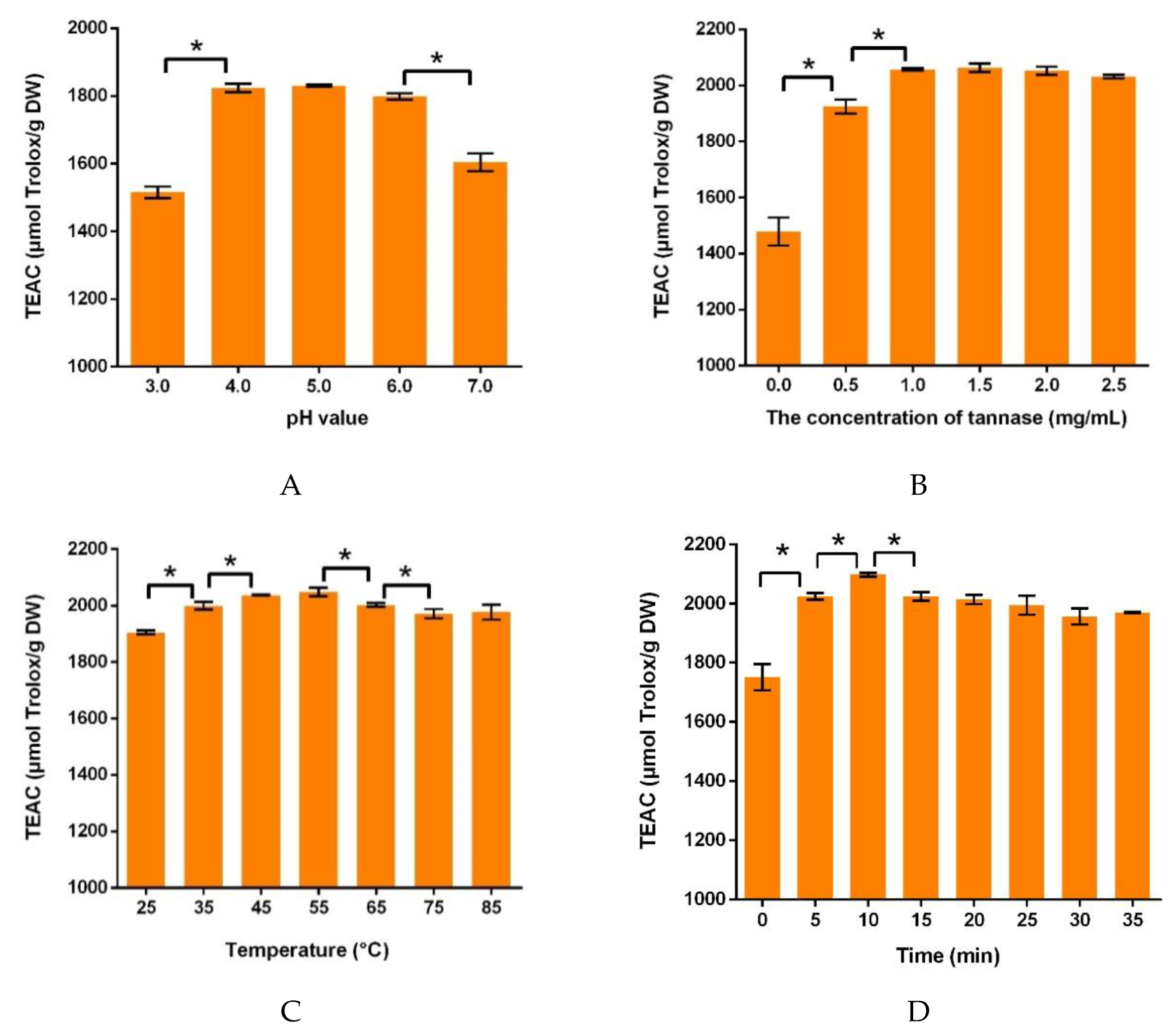
3.1.1. Effects of the pH Value of Buffer Solution on Antioxidant Capacities
3.1.2. Effects of Tannase Concentration on Antioxidant Capacities
3.1.3. Effects of Ultrasonic Temperature on Antioxidant Capacities
3.1.4. Effects of Ultrasonic Time on Antioxidant Capacities
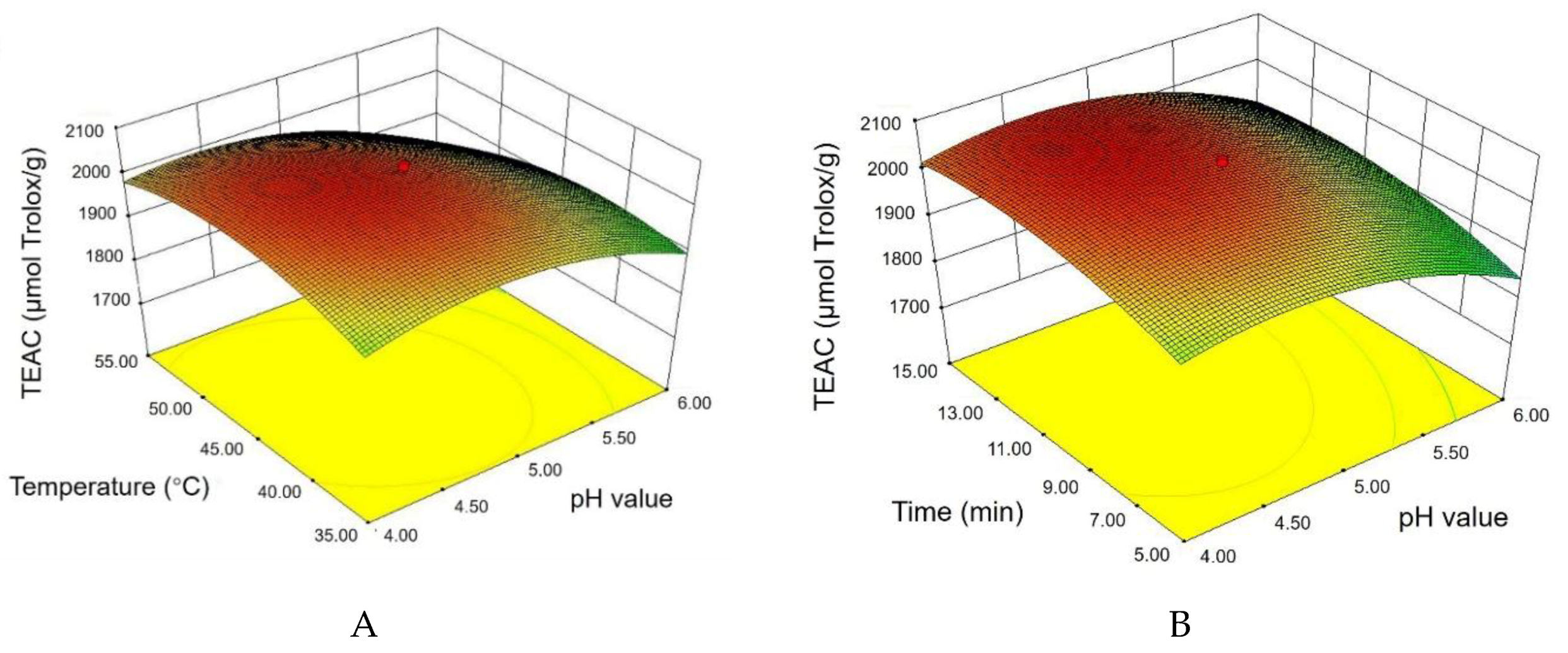
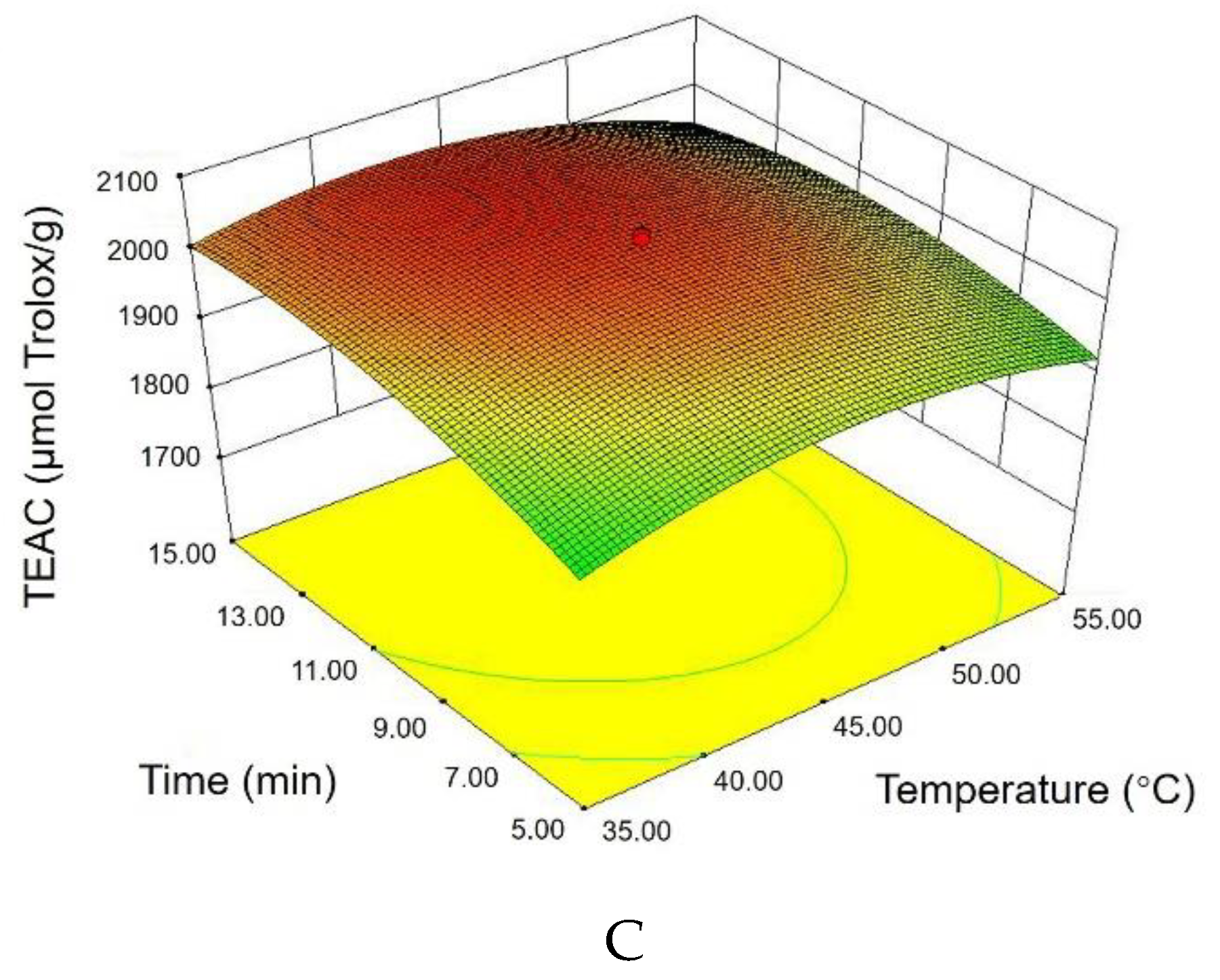
3.2. Results by Response Surface Methodology
3.2.1. Statistical Analysis and Model Fitting
3.2.2. Response Surface Plots and Graphical Analysis
3.2.3. Optimal Treatment Conditions
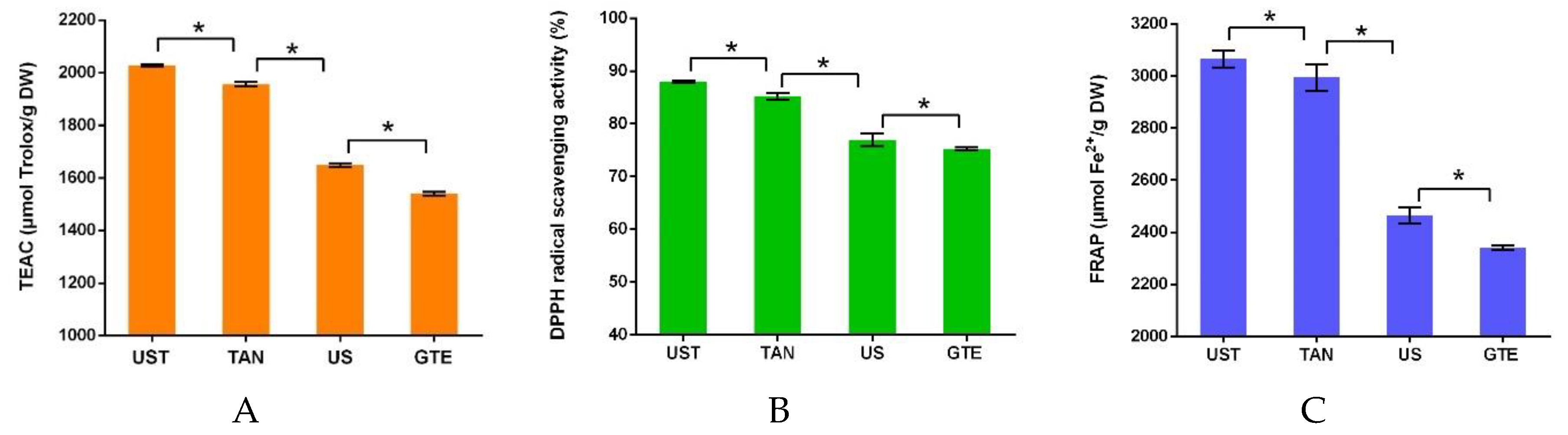
3.3. Comparison of Different Treatments
3.4. Antioxidants of the Extracts
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ende, W.V.D.; Peshev, D.; De Gara, L. Disease prevention by natural antioxidants and prebiotics acting as ROS scavengers in the gastrointestinal tract. Trends Food Sci. Technol. 2011, 22, 689–697. [Google Scholar] [CrossRef]
- Nuutila, A.M.; Puupponen-Pimiä, R.; Aarni, M.; Oksman-Caldentey, K.-M. Comparison of antioxidant activities of onion and garlic extracts by inhibition of lipid peroxidation and radical scavenging activity. Food Chem. 2003, 81, 485–493. [Google Scholar] [CrossRef]
- Paran, E.; Novack, V.; Engelhard, Y.N.; Hazan-Halevy, I. The effects of natural antioxidants from tomato extract in treated but uncontrolled hypertensive patients. Cardiovasc. Drugs Ther. 2009, 23, 145–151. [Google Scholar] [CrossRef] [PubMed]
- Pandey, K.B.; Rizvi, S.I. Plant polyphenols as dietary antioxidants in human health and disease. Oxidative Med. Cell. Longev. 2009, 2, 270–278. [Google Scholar] [CrossRef] [PubMed]
- Li, A.-N.; Li, S.; Zhang, Y.-J.; Xu, X.-R.; Chen, Y.-M.; Li, H.-B. Resources and Biological Activities of Natural Polyphenols. Nutrients 2014, 6, 6020–6047. [Google Scholar] [CrossRef] [PubMed]
- He, F.J.; Nowson, C.A.; Lucas, M.; MacGregor, G.A. Increased consumption of fruit and vegetables is related to a reduced risk of coronary heart disease: Meta-analysis of cohort studies. J. Hum. Hypertens. 2007, 21, 717–728. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.-J.; Gan, R.-Y.; Li, S.; Zhou, Y.; Li, A.-N.; Xu, D.-P.; Li, H.-B. Antioxidant Phytochemicals for the Prevention and Treatment of Chronic Diseases. Molecules 2015, 20, 21138–21156. [Google Scholar] [CrossRef] [PubMed]
- Dauchet, L.; Amouyel, P.; Hercberg, S.; Dallongeville, J. Fruit and vegetable consumption and risk of coronary heart disease: A meta-analysis of cohort studies. J. Nutr. 2006, 136, 2588–2593. [Google Scholar] [CrossRef]
- Shen, Y.; Zhang, H.; Cheng, L.; Wang, L.; Qian, H.; Qi, X. In vitro and in vivo antioxidant activity of polyphenols extracted from black highland barley. Food Chem. 2016, 194, 1003–1012. [Google Scholar] [CrossRef]
- Zhou, Y.; Zheng, J.; Li, Y.; Xu, D.-P.; Li, S.; Chen, Y.-M.; Li, H.-B. Natural Polyphenols for Prevention and Treatment of Cancer. Nutrients 2016, 8, 515. [Google Scholar] [CrossRef]
- Zhou, Y.; Li, Y.; Zhou, T.; Zheng, J.; Li, S.; Li, H.-B. Dietary Natural Products for Prevention and Treatment of Liver Cancer. Nutrients 2016, 8, 156. [Google Scholar] [CrossRef] [PubMed]
- Podmore, I.D.; Griffiths, H.R.; Herbert, K.E.; Mistry, N.; Mistry, P.; Lunec, J. Vitamin C exhibits pro-oxidant properties. Nature 1998, 392, 559. [Google Scholar] [CrossRef] [PubMed]
- Sayin, V.I.; Ibrahim, M.X.; Larsson, E.; Nilsson, J.A.; Lindahl, P.; Bergo, M.O. Antioxidants Accelerate Lung Cancer Progression in Mice. Sci. Transl. Med. 2014, 6, 221ra15. [Google Scholar] [CrossRef] [PubMed]
- Le Gal, K.; Ibrahim, M.X.; Wiel, C.; Sayin, V.I.; Akula, M.K.; Karlsson, C.; Dalin, M.G.; Akyürek, L.M.; Lindahl, P.; Nilsson, J.; et al. Antioxidants can increase melanoma metastasis in mice. Sci. Transl. Med. 2015, 7, 308re8. [Google Scholar] [CrossRef] [PubMed]
- Caleja, C.; Barros, L.; Antonio, A.L.; Oliveira, M.B.P.; Ferreira, I.C. A comparative study between natural and synthetic antioxidants: Evaluation of their performance after incorporation into biscuits. Food Chem. 2017, 216, 342–346. [Google Scholar] [CrossRef] [PubMed]
- Carocho, M.; Ferreira, I.C. A review on antioxidants, prooxidants and related controversy: Natural and synthetic compounds, screening and analysis methodologies and future perspectives. Food Chem. Toxicol. 2013, 51, 15–25. [Google Scholar] [CrossRef]
- Halliwell, B.; Gutteridge, J.M. The definition and measurement of antioxidants in biological systems. Free. Radic. Biol. Med. 1995, 18, 125–126. [Google Scholar] [CrossRef]
- Xu, D.-P.; Li, Y.; Meng, X.; Zhou, T.; Zhou, Y.; Zheng, J.; Zhang, J.-J.; Li, H.-B. Natural Antioxidants in Foods and Medicinal Plants: Extraction, Assessment and Resources. Int. J. Mol. Sci. 2017, 18, 96. [Google Scholar] [CrossRef]
- Apak, R.; Özyürek, M.; Güçlü, K.; Çapanoğlu, E. Antioxidant Activity/Capacity Measurement. 1. Classification, Physicochemical Principles, Mechanisms, and Electron Transfer (ET)-Based Assays. J. Agric. Food Chem. 2016, 64, 997–1027. [Google Scholar] [CrossRef]
- Apak, R.; Özyürek, M.; Güçlü, K.; Çapanoğlu, E. Antioxidant Activity/Capacity Measurement. 2. Hydrogen Atom Transfer (HAT)-Based, Mixed-Mode (Electron Transfer (ET)/HAT), and Lipid Peroxidation Assays. J. Agric. Food Chem. 2016, 64, 1028–1045. [Google Scholar] [CrossRef]
- Apak, R.; Özyürek, M.; Güçlü, K.; Çapanoğlu, E. Antioxidant Activity/Capacity Measurement. 3. Reactive Oxygen and Nitrogen Species (ROS/RNS) Scavenging Assays, Oxidative Stress Biomarkers, and Chromatographic/Chemometric Assays. J. Agric. Food Chem. 2016, 64, 1046–1070. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.; Huo, J.; Jiang, H.; Yang, F. Chemical compositions and bioactivities of crude polysaccharides from tea leaves beyond their useful date. Int. J. Biol. Macromol. 2011, 49, 1143–1151. [Google Scholar] [CrossRef] [PubMed]
- Higdon, J.V.; Frei, B. Tea catechins and polyphenols: Health effects, metabolism, and antioxidant functions. Crit. Rev. Food Sci. Nutr. 2003, 43, 89–143. [Google Scholar] [CrossRef] [PubMed]
- Nagao, T.; Hase, T.; Tokimitsu, I. A Green Tea Extract High in Catechins Reduces Body Fat and Cardiovascular Risks in Humans. Obesity 2007, 15, 1473–1483. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.-Y.; Zhao, C.-N.; Cao, S.-Y.; Tang, G.-Y.; Gan, R.-Y.; Li, H.-B. Effects and mechanisms of tea for the prevention and management of cancers: An updated review. Crit. Rev. Food Sci. Nutr. 2019. [Google Scholar] [CrossRef] [PubMed]
- Cao, S.Y.; Zhao, C.N.; Gan, R.Y.; Xu, X.Y.; Wei, X.L.; Corke, H.; Atanasov, A.G.; Li, H.B. Effects and mechanisms of tea and its bioactive compounds for the prevention and treatment of cardiovascular diseases: An updated review. Antioxidants 2019, 8, 166. [Google Scholar] [CrossRef] [PubMed]
- Meng, J.M.; Cao, S.Y.; Wei, X.L.; Gan, R.Y.; Wang, Y.F.; Cai, S.X.; Xu, X.Y.; Zhang, P.Z.; Li, H.B. Effects and mechanisms of tea for the prevention and management of diabetes mellitus and diabetic complications: An updated review. Antioxidants 2019, 8, 170. [Google Scholar] [CrossRef] [PubMed]
- Al-Awaida, W.J.; Zihlif, M.A.; Al-Ameer, H.J.; Sharab, A.; Akash, M.; Aburubaiha, Z.A.; Fattash, I.A.; Imraish, A.; Ali, K.H. The effect of green tea consumption on the expression of antioxidant- and inflammation-related genes induced by nicotine. J. Food Biochem. 2019, 43, e12874. [Google Scholar] [CrossRef]
- Inami, S.; Takano, M.; Yamamoto, M.; Murakami, D.; Tajika, K.; Yodogawa, K.; Yokoyama, S.; Ohno, N.; Ohba, T.; Sano, J.; et al. Tea catechin consumption reduces circulating oxidized low-density lipoprotein. Int. Heart J. 2007, 48, 725–732. [Google Scholar] [CrossRef]
- Conde, V.R.; Alves, M.G.; Oliveira, P.F.; Silva, B.M. Tea (Camellia sinensis (L.)): A Putative anticancer agent in bladder carcinoma? Anti-Cancer Agents Med. Chem. 2015, 15, 26–36. [Google Scholar] [CrossRef]
- Gan, R.Y.; Li, H.B.; Sui, Z.Q.; Corke, H. Absorption, metabolism, anti-cancer effect and molecular targets of epigallocatechin gallate (EGCG): An updated review. Crit. Rev. Food Sci. Nutr. 2018, 58, 924–941. [Google Scholar] [CrossRef] [PubMed]
- Cavet, M.E.; Harrington, K.L.; Vollmer, T.R.; Ward, K.W.; Zhang, J.-Z. Anti-inflammatory and anti-oxidative effects of the green tea polyphenol epigallocatechin gallate in human corneal epithelial cells. Mol. Vis. 2011, 17, 533–542. [Google Scholar] [PubMed]
- Del Rio, D.; Stewart, A.J.; Mullen, W.; Burns, J.; Lean, M.E.J.; Brighenti, F.; Crozier, A. HPLC-MSnAnalysis of Phenolic Compounds and Purine Alkaloids in Green and Black Tea. J. Agric. Food Chem. 2004, 52, 2807–2815. [Google Scholar] [CrossRef] [PubMed]
- Rusak, G.; Komes, D.; Likić, S.; Horžić, D.; Kovač, M. Phenolic content and antioxidative capacity of green and white tea extracts depending on extraction conditions and the solvent used. Food Chem. 2008, 110, 852–858. [Google Scholar] [CrossRef] [PubMed]
- Peluso, I.; Serafini, M. Antioxidants from black and green tea: From dietary modulation of oxidative stress to pharmacological mechanisms. Br. J. Pharmacol. 2017, 174, 1195–1208. [Google Scholar] [CrossRef] [PubMed]
- Isomura, T.; Suzuki, S.; Origasa, H.; Hosono, A.; Suzuki, M.; Sawada, T.; Terao, S.; Muto, Y.; Koga, T. Liver-related safety assessment of green tea extracts in humans: A systematic review of randomized controlled trials. Eur. J. Clin. Nutr. 2016, 70, 1221–1229. [Google Scholar] [CrossRef] [PubMed]
- Dostal, A.M.; Samavat, H.; Bedell, S.; Torkelson, C.; Wang, R.; Swenson, K.; Le, C.; Wu, A.H.; Ursin, G.; Yuan, J.-M.; et al. The safety of green tea extract supplementation in postmenopausal women at risk for breast cancer: Results of the Minnesota Green Tea Trial. Food Chem. Toxicol. 2015, 83, 26–35. [Google Scholar] [CrossRef]
- Bedrood, Z.; Rameshrad, M.; Hosseinzadeh, H. Toxicological effects of Camellia sinensis (green tea): A review. Phytother. Res. 2018, 32, 1163–1180. [Google Scholar] [CrossRef]
- Mazzanti, G.; Di Sotto, A.; Vitalone, A. Hepatotoxicity of green tea: An update. Arch. Toxicol. 2015, 89, 1175–1191. [Google Scholar] [CrossRef]
- Hu, J.; Webster, D.; Cao, J.; Shao, A. The safety of green tea and green tea extract consumption in adults —Results of a systematic review. Regul. Toxicol. Pharmacol. 2018, 95, 412–433. [Google Scholar] [CrossRef]
- Toushik, S.H.; Lee, K.; Kim, K. Functional Applications of Lignocellulolytic Enzymes in the Fruit and Vegetable Processing Industries. J. Food Sci. 2017, 82, 585–593. [Google Scholar] [CrossRef] [PubMed]
- Roberto, B.S.; Macedo, G.A.; Macedo, J.A.; Martins, I.M.; Nakajima, V.M.; Allwood, J.W.; Stewart, D.; McDougall, G.J. Immobilized tannase treatment alters polyphenolic composition in teas and their potential anti-obesity and hypoglycemic activities in vitro. Food Funct. 2016, 7, 3920–3932. [Google Scholar] [CrossRef] [PubMed]
- Yen, G.-C.; Duh, P.-D.; Tsai, H.-L. Antioxidant and pro-oxidant properties of ascorbic acid and gallic acid. Food Chem. 2002, 79, 307–313. [Google Scholar] [CrossRef]
- Chemat, F.; Huma, Z.E.; Khan, M.K. Applications of ultrasound in food technology: Processing, preservation and extraction. Ultrason. Sonochem. 2011, 18, 813–835. [Google Scholar] [CrossRef] [PubMed]
- Soares, A.D.S.; Augusto, P.E.D.; Júnior, B.R.D.C.L.; Nogueira, C.A.; Vieira, É.N.R.; De Barros, F.A.R.; Stringheta, P.C.; Ramos, A.M. Ultrasound assisted enzymatic hydrolysis of sucrose catalyzed by invertase: Investigation on substrate, enzyme and kinetics parameters. LWT 2019, 107, 164–170. [Google Scholar] [CrossRef]
- Li, A.-N.; Li, S.; Li, Y.; Xu, D.-P.; Li, H.-B. Optimization of Ultrasound-Assisted Extraction of Natural Antioxidants from the Osmanthus fragrans Flower. Molecules 2016, 21, 218. [Google Scholar] [CrossRef] [PubMed]
- Yolmeh, M.; Najafi, M.B.H.; Farhoosh, R. Optimisation of ultrasound-assisted extraction of natural pigment from annatto seeds by response surface methodology (RSM). Food Chem. 2014, 155, 319–324. [Google Scholar] [CrossRef]
- Ye, C.-L.; Jiang, C.-J. Optimization of extraction process of crude polysaccharides from Plantago asiatica L. by response surface methodology. Carbohydr. Polym. 2011, 84, 495–502. [Google Scholar] [CrossRef]
- Zhao, C.N.; Tang, G.Y.; Cao, S.Y.; Xu, X.Y.; Gan, R.Y.; Liu, Q.; Mao, Q.Q.; Shang, A.; Li, H.B. Phenolic profiles and antioxidant activities of 30 tea infusions from green, black, oolong, white, yellow and dark teas. Antioxidants 2019, 8, 215. [Google Scholar] [CrossRef]
- Pastoriza, S.; Pérez-Burillo, S.; Rufián-Henares, J. Ángel How brewing parameters affect the healthy profile of tea. Curr. Opin. Food Sci. 2017, 14, 7–12. [Google Scholar] [CrossRef]
- Xi, J.; Wang, B.S. Optimization of ultrahigh-pressure extraction of polyphenolic antioxidants from green tea by response surface methodology. Food Bioprocess Technol. 2013, 6, 2538–2546. [Google Scholar] [CrossRef]
- Dudonné, S.; Vitrac, X.; Coutière, P.; Woillez, M.; Mérillon, J.-M. Comparative Study of Antioxidant Properties and Total Phenolic Content of 30 Plant Extracts of Industrial Interest Using DPPH, ABTS, FRAP, SOD, and ORAC Assays. J. Agric. Food Chem. 2009, 57, 1768–1774. [Google Scholar] [CrossRef] [PubMed]
- Li, A.-N.; Li, S.; Li, H.-B.; Xu, D.-P.; Xu, X.-R.; Chen, F. Total phenolic contents and antioxidant capacities of 51 edible and wild flowers. J. Funct. Foods 2014, 6, 319–330. [Google Scholar] [CrossRef]
- Tang, G.-Y.; Zhao, C.-N.; Xu, X.-Y.; Gan, R.-Y.; Cao, S.-Y.; Liu, Q.; Shang, A.; Mao, Q.-Q.; Li, H.-B. Phytochemical Composition and Antioxidant Capacity of 30 Chinese Teas. Antioxidants 2019, 8, 180. [Google Scholar] [CrossRef] [PubMed]
- Costa, A.M.; Ribeiro, W.X.; Kato, E.; Monteiro, A.R.G.; Peralta, R.M. Production of tannase by Aspergillus tamarii in submerged cultures. Braz. Arch. Biol. Technol. 2008, 51, 399–404. [Google Scholar] [CrossRef]
- Klibanov, A.M.; Klibanov, A.M.; Klibanov, A.M. Improving enzymes by using them in organic solvents. Nature 2001, 409, 241–246. [Google Scholar] [CrossRef]
- Mahapatra, K.; Nanda, R.K.; Bag, S.S.; Banerjee, R.; Pandey, A.; Szakacs, G. Purification, characterization and some studies on secondary structure of tannase from Aspergillus awamori nakazawa. Process Biochem. 2005, 40, 3251–3254. [Google Scholar] [CrossRef]
- Batra, A.; Saxena, R. Potential tannase producers from the genera Aspergillus and Penicillium. Process Biochem. 2005, 40, 1553–1557. [Google Scholar] [CrossRef]
- Chen, H.; Zhou, X.; Zhang, J. Optimization of enzyme assisted extraction of polysaccharides from Astragalus membranaceus. Carbohydr. Polym. 2014, 111, 567–575. [Google Scholar] [CrossRef]
- Swer, T.L.; Mukhim, C.; Bashir, K.; Chauhan, K. Optimization of enzyme aided extraction of anthocyanins from Prunus nepalensis L. LWT 2018, 91, 382–390. [Google Scholar] [CrossRef]
| Run | pH (X1) | Temperature (X2, °C) | Time (X3, min) | TEAC Value (Y, μmol Trolox/g DW) | |
|---|---|---|---|---|---|
| Actual Value | Predicted Value | ||||
| 1 | 6 (1) | 35 (−1) | 5 (−1) | 1798.00 | 1809.36 |
| 2 | 5 (0) | 45 (0) | 10 (0) | 2001.15 | 2036.55 |
| 3 | 5 (0) | 45 (0) | 18.41 (1.68) | 1993.44 | 1985.88 |
| 4 | 6 (1) | 35 (−1) | 15 (1) | 1930.55 | 1913.58 |
| 5 | 4 (−1) | 35 (−1) | 5 (−1) | 1871.32 | 1874.33 |
| 6 | 5 (0) | 45 (0) | 10 (0) | 2028.64 | 2036.55 |
| 7 | 5 (0) | 45 (0) | 10 (0) | 2056.13 | 2036.55 |
| 8 | 4 (−1) | 55 (1) | 5 (−1) | 1895.76 | 1938.43 |
| 9 | 3.32 (−1.68) | 45 (0) | 10 (0) | 1969.07 | 1937.75 |
| 10 | 4 (−1) | 35 (−1) | 15 (1) | 1953.80 | 1967.27 |
| 11 | 5 (0) | 45 (0) | 10 (0) | 2057.66 | 2036.55 |
| 12 | 5 (0) | 28.18 (−1.68) | 10 (0) | 1898.81 | 1904.75 |
| 13 | 5 (0) | 61.82 (1.68) | 10 (0) | 1912.56 | 1870.27 |
| 14 | 4 (−1) | 55 (1) | 15 (1) | 1943.11 | 1957.46 |
| 15 | 5 (0) | 45 (0) | 10 (0) | 2019.47 | 2036.55 |
| 16 | 5 (0) | 45 (0) | 1.59 (−1.68) | 1911.03 | 1882.23 |
| 17 | 6 (1) | 55 (1) | 5 (−1) | 1765.93 | 1778.17 |
| 18 | 6.68 (1.68) | 45 (0) | 10 (0) | 1762.88 | 1757.84 |
| 19 | 5 (0) | 45 (0) | 10 (0) | 2050.02 | 2036.55 |
| 20 | 6 (1) | 55 (1) | 15 (1) | 1785.79 | 1808.48 |
| Source | Sum of Squares | Df | Mean Square | F Value | P-Value | Significant |
|---|---|---|---|---|---|---|
| Model | 165,385.81 | 9 | 18,376.20 | 19.08 | < 0.0001 | Significant |
| X1-pH | 39,072.88 | 1 | 39,072.88 | 40.56 | < 0.0001 | |
| X2-Temperautre | 1434.63 | 1 | 1434.63 | 1.49 | 0.2503 | |
| X3-Time | 12,967.00 | 1 | 12,967.00 | 13.46 | 0.0043 | |
| X1 X2 | 4540.31 | 1 | 4540.31 | 4.71 | 0.0551 | |
| X1 X3 | 63.69 | 1 | 63.69 | 0.066 | 0.8023 | |
| X2 X3 | 2731.27 | 1 | 2731.27 | 2.84 | 0.1231 | |
| X12 | 64,181.85 | 1 | 64,181.85 | 66.62 | < 0.0001 | |
| X22 | 40,016.60 | 1 | 40,016.60 | 41.54 | < 0.0001 | |
| X32 | 18,924.01 | 1 | 18,924.01 | 19.64 | 0.0013 | |
| Residual | 9633.42 | 10 | 963.34 | |||
| Lack of Fit | 7021.77 | 5 | 1404.35 | 2.69 | 0.1508 | not significant |
| Pure Error | 2611.66 | 5 | 522.33 | |||
| Cor Total | 175,019.24 | 19 |
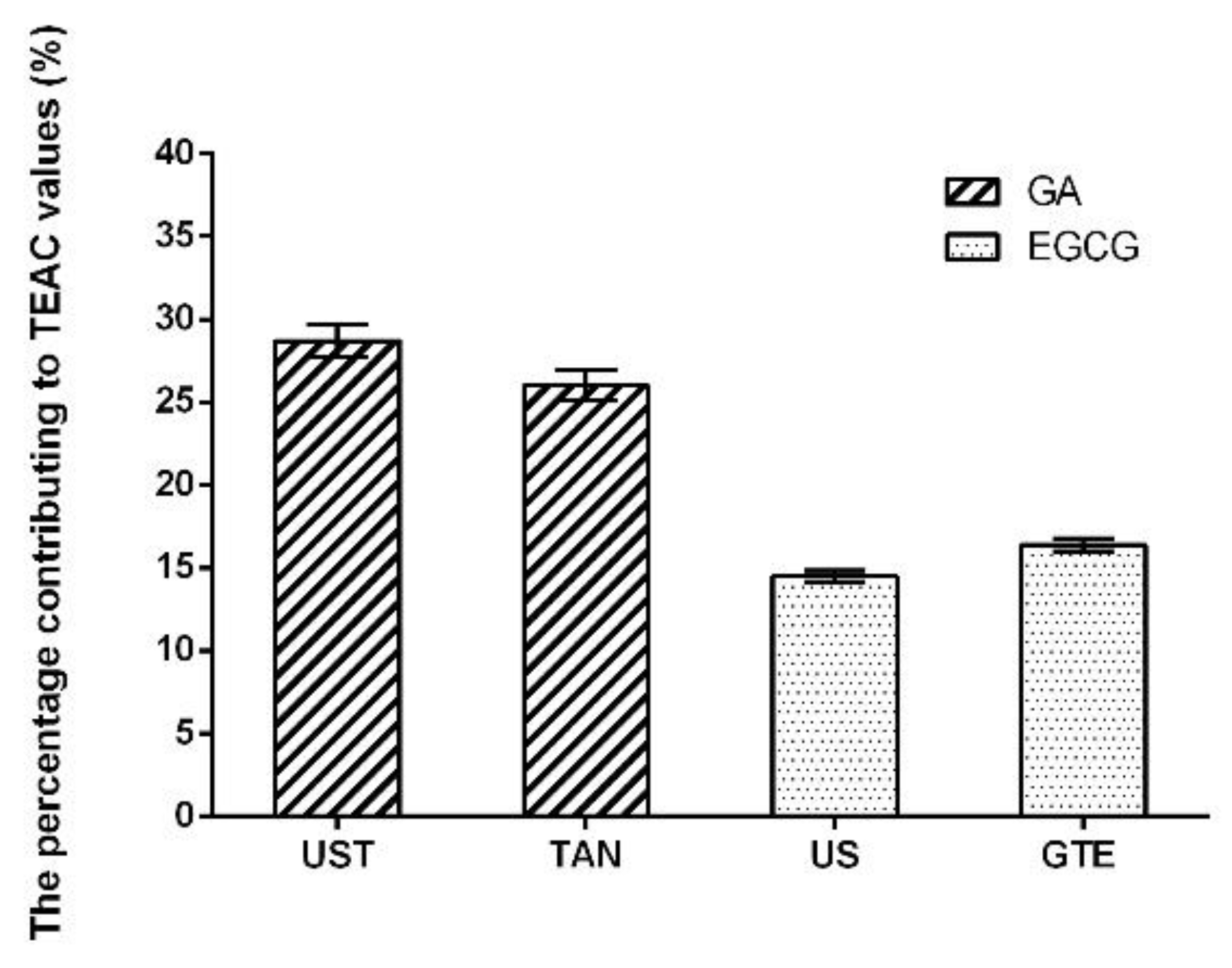
| Antioxidants | UST | TAN | US | GTE |
|---|---|---|---|---|
| Gallic acid | 31.83 ± 0.74 a | 27.82 ± 0.47 b | 2.16 ± 0.05 c | 1.97 ± 0.03 c |
| Gallocatechin | 0.32 ±0.09 | 0.32 ± 0.02 | 0.21 ± 0.07 | 0.21 ± 0.01 |
| Epicatechin | 1.96 ± 0.31 a | 2.12 ± 0.17 a | 0.70 ± 0.06 b | 0.71 ± 0.02 b |
| Epigallocatechin | 2.02 ± 0.73 a | 2.07 ± 0.22 a | 0.29 ± 0.02 b | 0.30 ± 0.01 b |
| Epigallocatechin gallate | 0.16 ± 0.06 c | 0.16 ± 0.10 c | 11.55 ± 0.14 b | 12.18 ± 0.20 a |
| Epicatechin gallate | 0.16 ± 0.02 b | 0.19 ± 0.04 b | 6.33 ± 0.46 a | 6.03 ± 0.60 a |
| Gallocatechin gallate | 0.17 ± 0.11 b | 0.17 ± 0.08 b | 0.71 ± 0.02 a | 0.58 ± 0.09 a |
| Caffeine | 17.00 ± 0.63 a,b | 17.59 ± 0.38 a | 16.60 ± 0.17 b | 16.39 ± 0.32 b |
| Ellagic acid | 1.31 ± 0.04 | 1.32 ± 0.55 | 1.41 ± 0.09 | 0.93 ± 0.19 |
| Astragalin | 0.58 ± 0.02 a | 0.45 ± 0.10 b | 0.53 ± 0.04 a,b | 0.51 ± 0.02 a,b |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, X.-Y.; Meng, J.-M.; Mao, Q.-Q.; Shang, A.; Li, B.-Y.; Zhao, C.-N.; Tang, G.-Y.; Cao, S.-Y.; Wei, X.-L.; Gan, R.-Y.; et al. Effects of Tannase and Ultrasound Treatment on the Bioactive Compounds and Antioxidant Activity of Green Tea Extract. Antioxidants 2019, 8, 362. https://doi.org/10.3390/antiox8090362
Xu X-Y, Meng J-M, Mao Q-Q, Shang A, Li B-Y, Zhao C-N, Tang G-Y, Cao S-Y, Wei X-L, Gan R-Y, et al. Effects of Tannase and Ultrasound Treatment on the Bioactive Compounds and Antioxidant Activity of Green Tea Extract. Antioxidants. 2019; 8(9):362. https://doi.org/10.3390/antiox8090362
Chicago/Turabian StyleXu, Xiao-Yu, Jin-Ming Meng, Qian-Qian Mao, Ao Shang, Bang-Yan Li, Cai-Ning Zhao, Guo-Yi Tang, Shi-Yu Cao, Xin-Lin Wei, Ren-You Gan, and et al. 2019. "Effects of Tannase and Ultrasound Treatment on the Bioactive Compounds and Antioxidant Activity of Green Tea Extract" Antioxidants 8, no. 9: 362. https://doi.org/10.3390/antiox8090362
APA StyleXu, X.-Y., Meng, J.-M., Mao, Q.-Q., Shang, A., Li, B.-Y., Zhao, C.-N., Tang, G.-Y., Cao, S.-Y., Wei, X.-L., Gan, R.-Y., Corke, H., & Li, H.-B. (2019). Effects of Tannase and Ultrasound Treatment on the Bioactive Compounds and Antioxidant Activity of Green Tea Extract. Antioxidants, 8(9), 362. https://doi.org/10.3390/antiox8090362






