Expression of Antioxidant Enzymes in Patients with Uterine Polyp, Myoma, Hyperplasia, and Adenocarcinoma
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Samples and Methods
2.3. Statistical Analysis
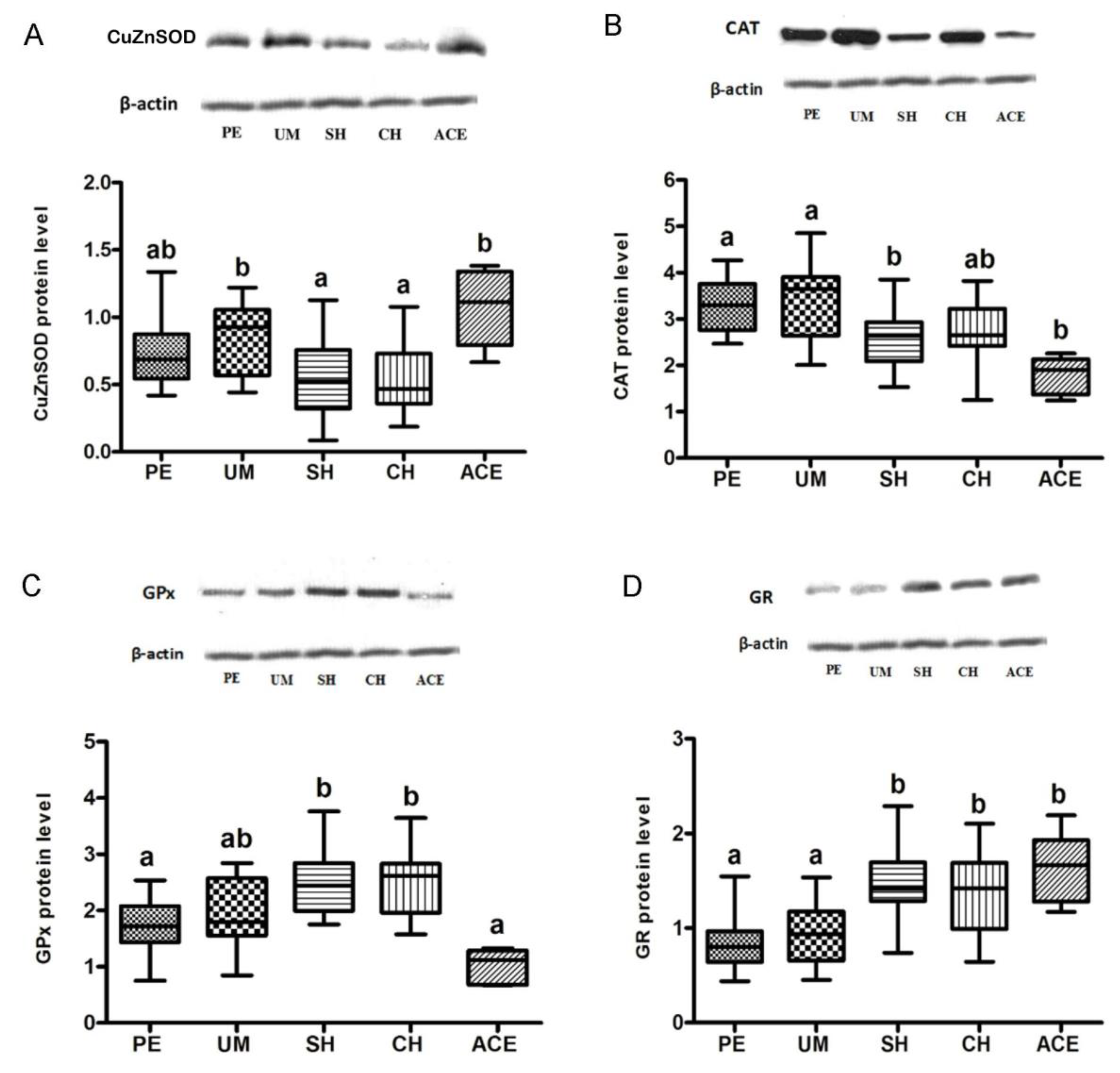
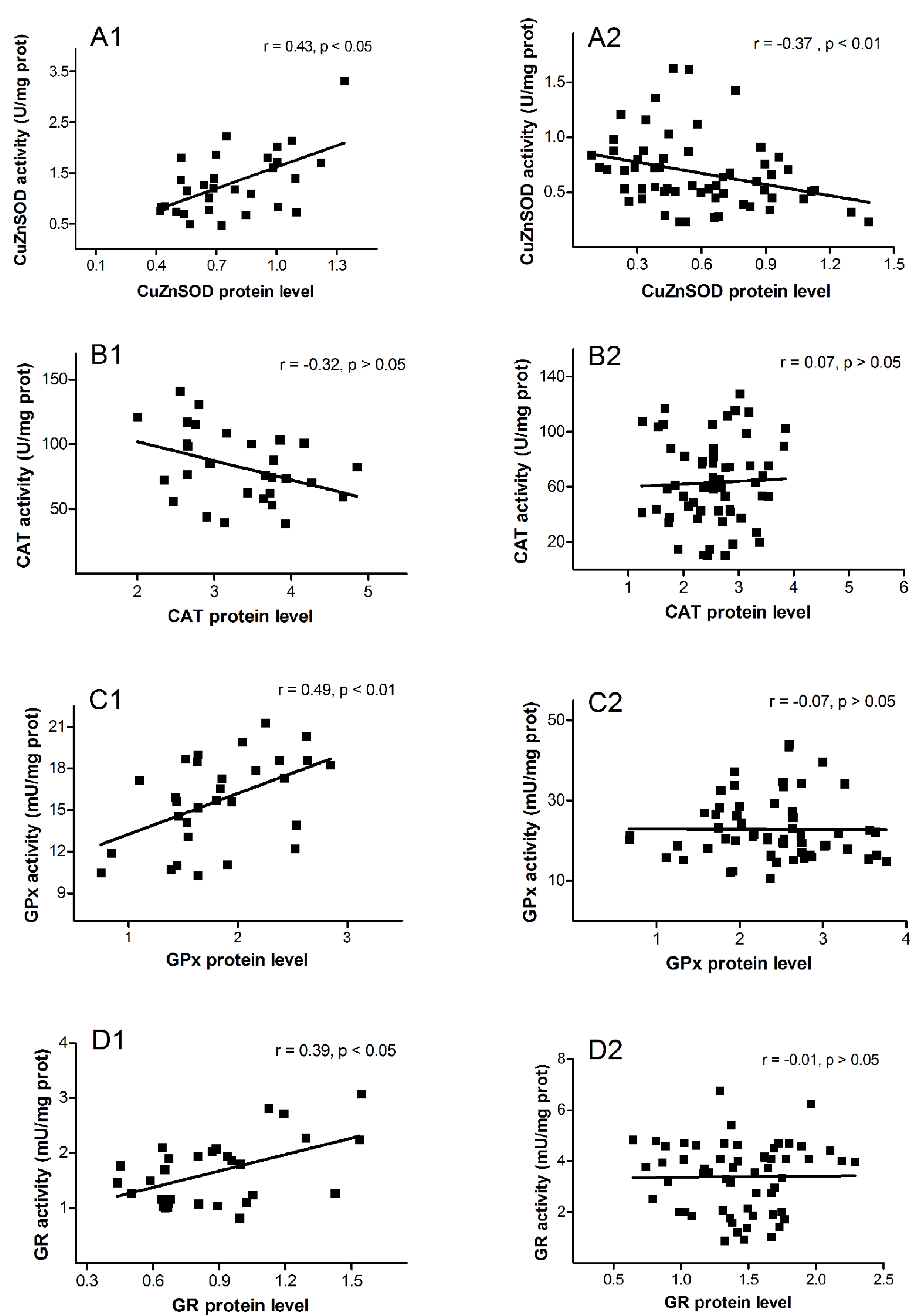
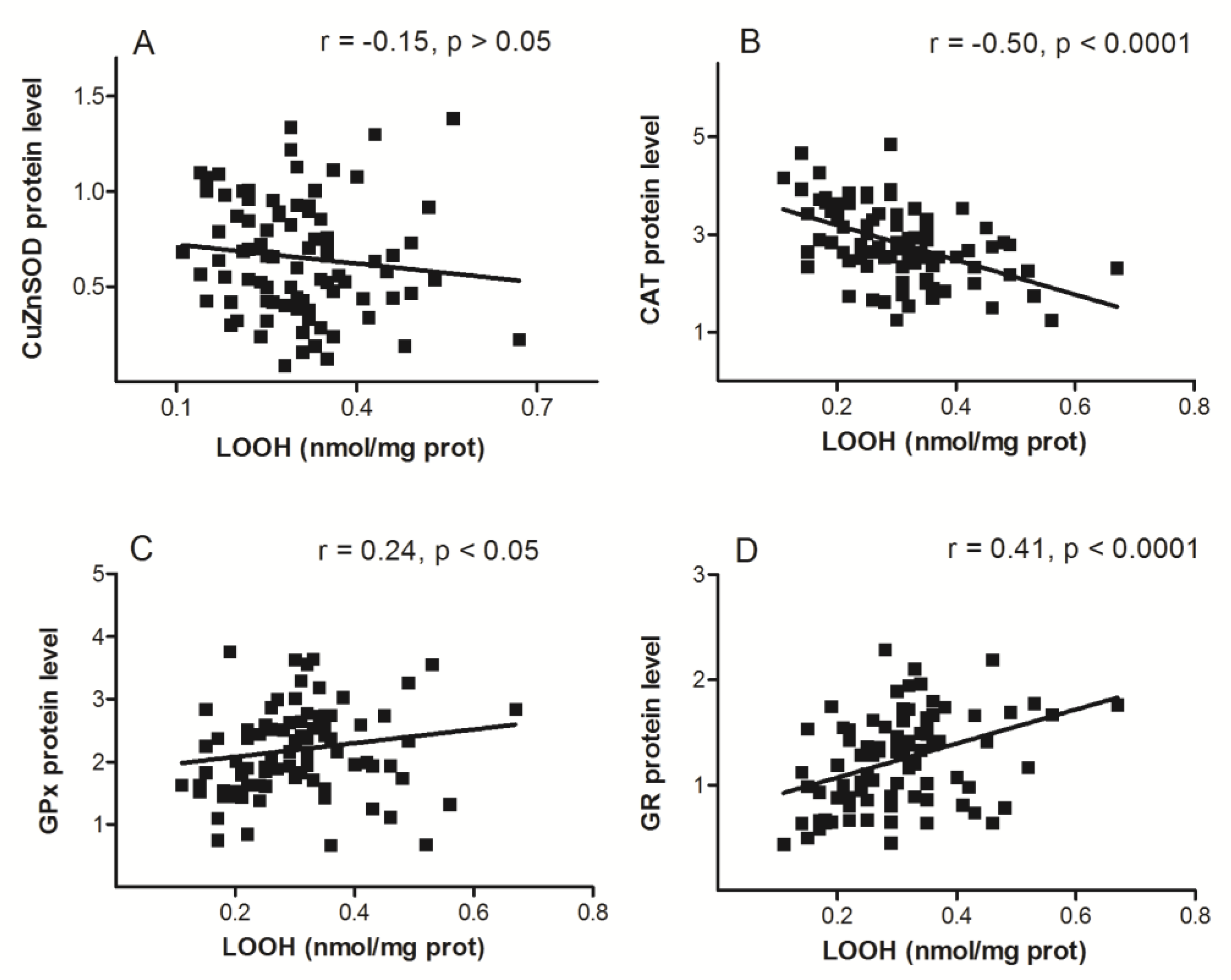
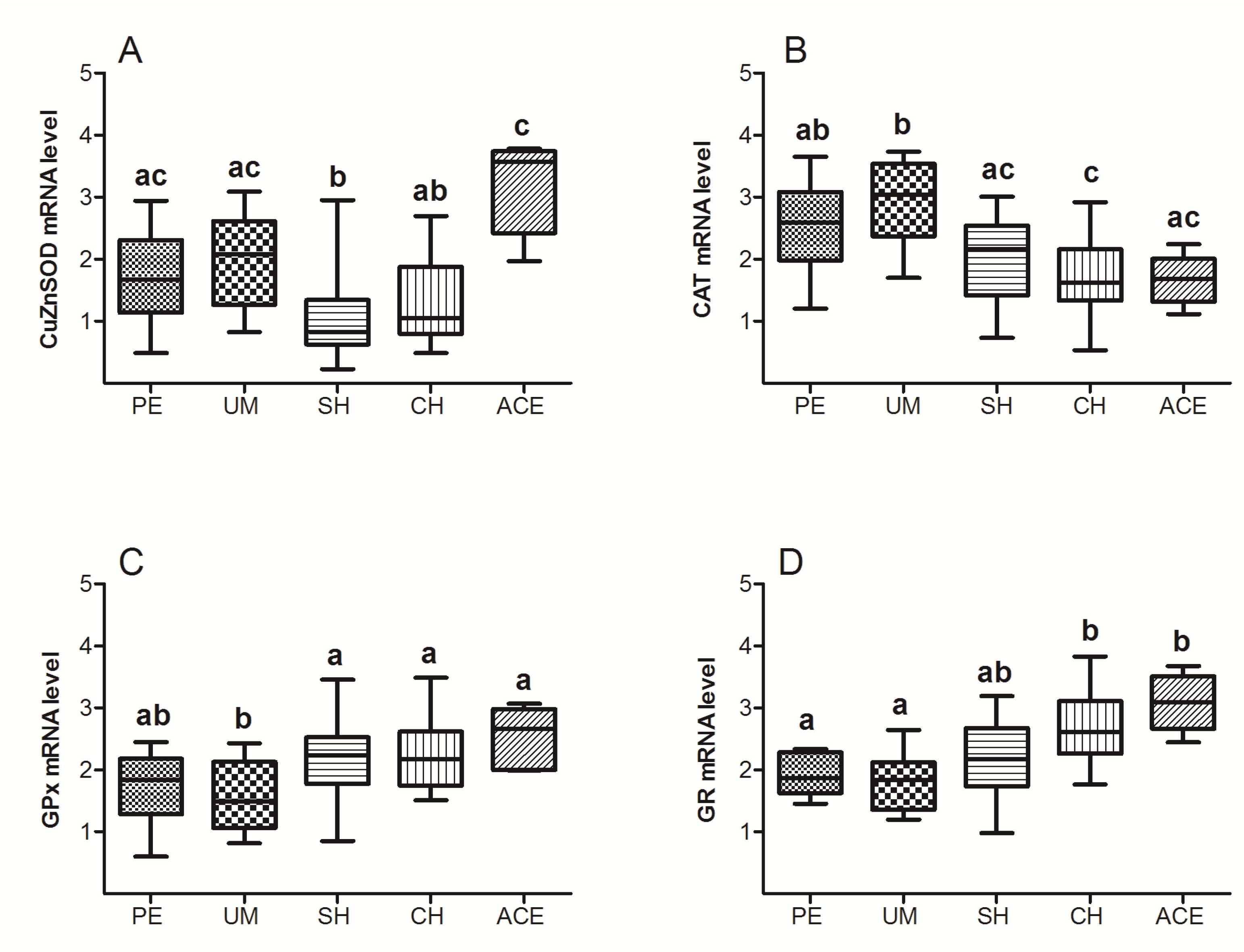
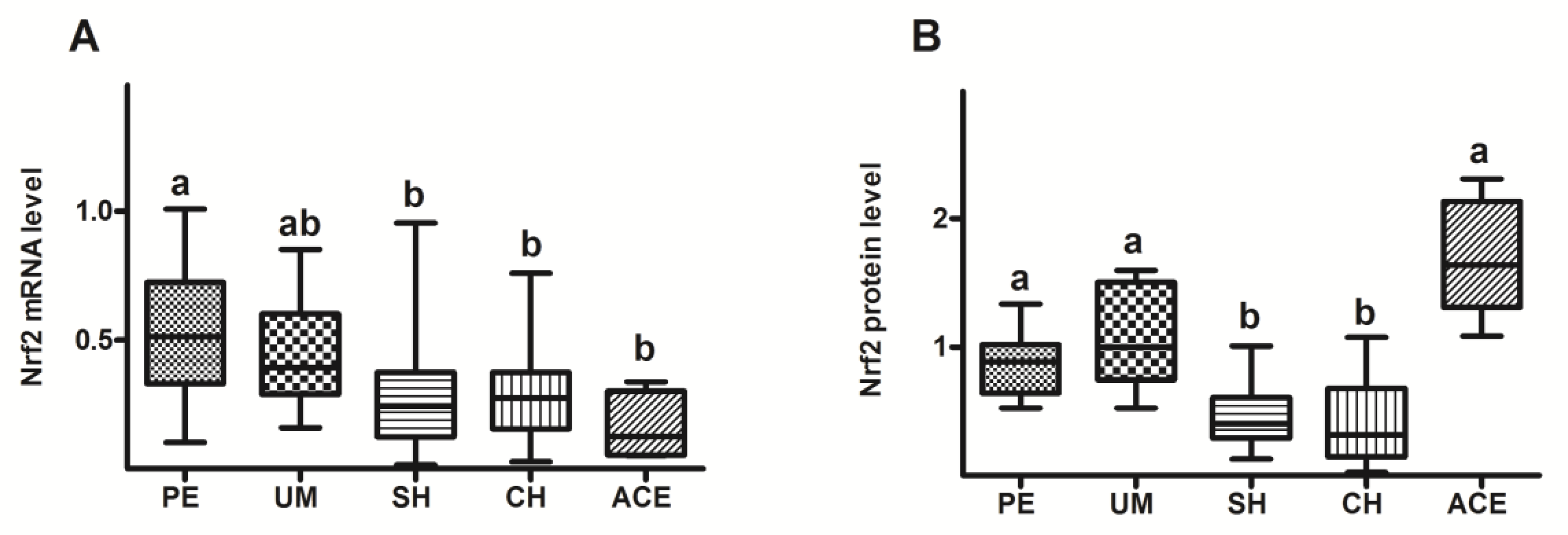
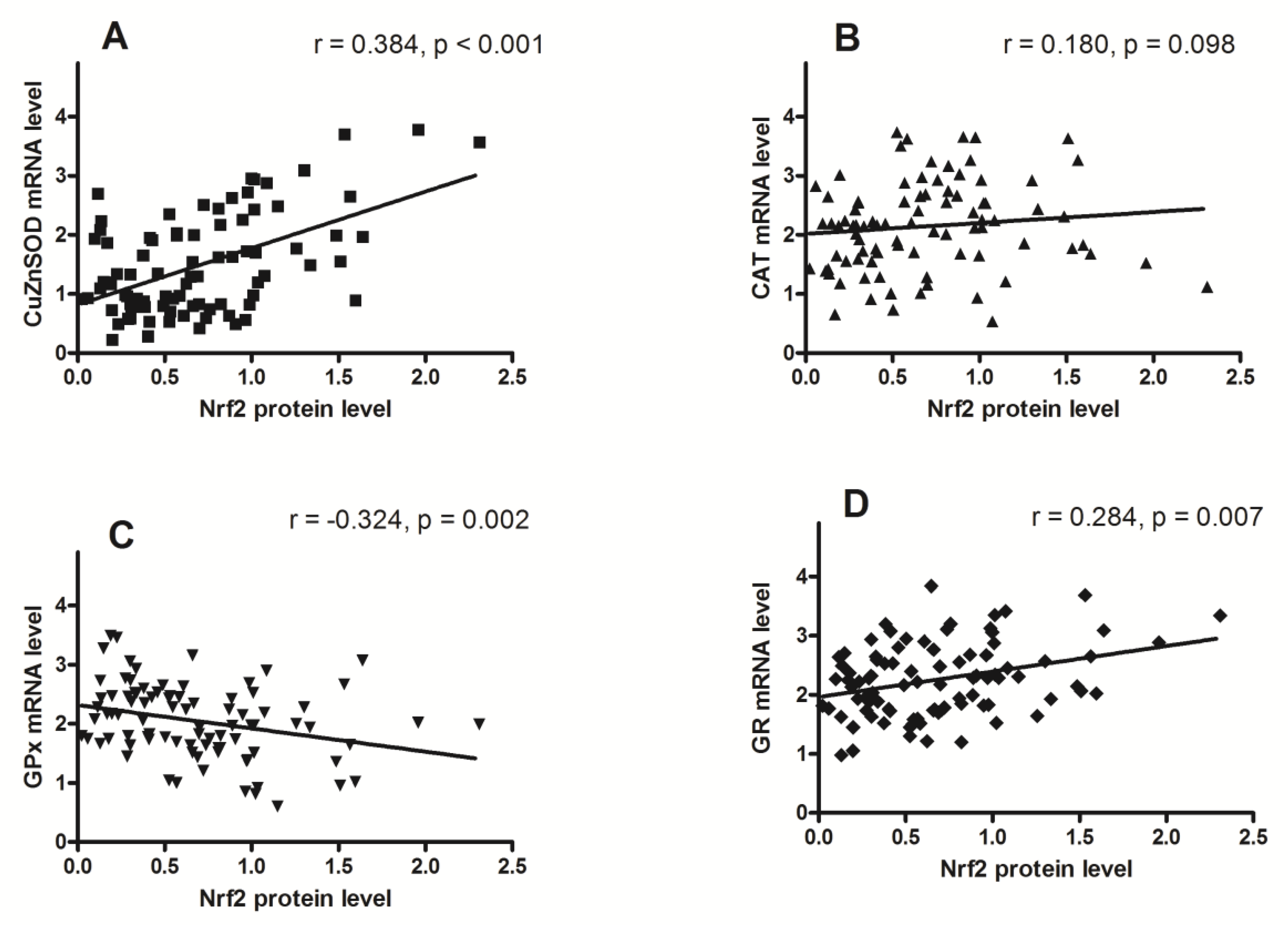
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Halliwell, B.; Gutteridge, J.M.C. Free Radicals in Biology and Medicine, 4th ed.; Oxford University Press: Oxford, UK, 2007. [Google Scholar]
- Zhu, H.; Itoh, K.; Yamamoto, M.; Zweier, J.L.; Li, Y. Role of Nrf2 signaling in regulation of antioxidants and phase 2 enzymes in cardiac fibroblasts: Protection against reactive oxygen and nitrogen species-induced cell injury. FEBS Lett. 2005, 579, 3029–3036. [Google Scholar] [CrossRef]
- Akino, N.; Wada-Hiraike, O.; Terao, H.; Honjoh, H.; Isono, W.; Fu, H.; Hirano, M.; Miyamoto, Y.; Tanikawa, M.; Harada, M.; et al. Activation of Nrf2 might reduce oxidative stress in human granulosa cells. Mol. Cell. Endocrinol. 2018, 470, 96–104. [Google Scholar] [CrossRef]
- Kwak, M.-K.; Wakabayashi, N.; Greenlaw, J.L.; Yamamoto, M.; Kensler, T.W. Antioxidants Enhance Mammalian Proteasome Expression through the Keap1-Nrf2 Signaling Pathway. Mol. Cell. Boil. 2003, 23, 8786–8794. [Google Scholar] [CrossRef]
- Malhotra, D.; Portales-Casamar, E.; Singh, A.; Srivastava, S.; Arenillas, D.; Happel, C.; Shyr, C.; Wakabayashi, N.; Kensler, T.W.; Wasserman, W.W.; et al. Global mapping of binding sites for Nrf2 identifies novel targets in cell survival response through ChIP-Seq profiling and network analysis. Acids Res. 2010, 38, 5718–5734. [Google Scholar] [CrossRef] [PubMed]
- Basak, P.; Sadhukhan, P.; Sarkar, P.; Sil, P.C. Perspectives of the Nrf-2 signaling pathway in cancer progression and therapy. Toxicol. Rep. 2017, 4, 306–318. [Google Scholar] [CrossRef]
- Klaunig, J.E.; Kamendulis, L.M.; Hocevar, B.A. Oxidative stress and oxidative damage in carcinogenesis. Toxicol. Pathol. 2010, 38, 96–109. [Google Scholar] [CrossRef]
- Bray, F.; Loos, A.H.; Oostindier, M.; Weiderpass, E. Geographic and temporal variations in cancer of the corpus uteri: Incidence and mortality in pre- and postmenopausal women in Europe. Int. J. 2005, 117, 123–131. [Google Scholar] [CrossRef]
- Mazur, M.T. Endometrial hyperplasia/adenocarcinoma. A conventional approach. Ann. Diagn. Pathol. 2005, 9, 174–181. [Google Scholar] [PubMed]
- Gültekin, M.; Dogan, N.U.; Aksan, G.; Ozgul, N. Management of endometrial hyperplasia. Minerva Ginecol. 2010, 62, 433–445. [Google Scholar]
- Kwak, M.-K.; Kensler, T.W. Induction of 26S proteasome subunit PSMB5 by the bifunctional inducer 3-methylcholanthrene through the Nrf2-ARE, but not the AhR/Arnt-XRE, pathway. Biochem. Biophys. Res. Commun. 2006, 345, 1350–1357. [Google Scholar] [CrossRef]
- Pejic, S.; Todorovic, A.; Stojiljkovic, V.; Pavlovic, I.; Gavrilovic, L.; Popovic, N.; Pajovic, S.B. Antioxidant status and sex hormones in women with complex endometrial hyperplasia. Cell. Mol. Biol. 2016, 62, 51–56. [Google Scholar]
- Pejić, S.; Todorović, A.; Stojiljković, V.; Pavlovic, I.; Gavrilović, L.; Popovic, N.; Pajovic, S.B. Antioxidant Status and Sex Hormones in Women with Simple Endometrial Hyperplasia. In Basic Principles and Clinical Significance of Oxidative Stress; IntechOpen: London, UK, 2015; Chapter 9; pp. 243–279. [Google Scholar]
- Pejić, S.; Todorović, A.; Stojiljković, V.; Gavrilović, L.; Popović, N.; Pajović, S.B. Antioxidant status in women with uterine leiomyoma: relation with sex hormones. An. Acad. Bras. Ciênc. 2015, 87, 1771–1782. [Google Scholar] [CrossRef]
- Pejić, S.A.; Kasapović, J.D.; Todorović, A.U.; Stojiljković, V.R.; Gavrilović, L.V.; Popović, N.M.; Pajović, S.B. Antioxidant enzymes in women with endometrial polyps: relation with sex hormones. Eur. J. Obstet. Gynecol. 2013, 170, 241–246. [Google Scholar] [CrossRef]
- Pejić, S.; Todorović, A.; Stojiljković, V.; Kasapović, J.; Pajović, S.B. Antioxidant enzymes and lipid peroxidation in endometrium of patients with polyps, myoma, hyperplasia and adenocarcinoma. Reprod. Boil. Endocrinol. 2009, 7, 149. [Google Scholar] [CrossRef]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with the Folin phenol reagent. J. Boil. Chem. 1951, 193, 265–275. [Google Scholar]
- Pejić, S.; Todorović, A.; Stojiljković, V.; Cvetković, D.; Lučić, N.; Radojičić, R.M.; Saičić, Z.S.; Pajovic, S.B. Superoxide dismutase and lipid hydroperoxides in blood and endometrial tissue of patients with benign, hyperplastic and malignant endometrium. An. Acad. Bras. Ciênc. 2008, 80, 515–522. [Google Scholar] [CrossRef]
- Güner, G.A.; Islekel, H.; Oto, Ö.; Hazan, E.; Acikel, U. Evaluation of some antioxidant enzymes in lung carcinoma tissue. Cancer Letters 1996, 103, 233–239. [Google Scholar] [CrossRef]
- Baker, A.M.; Oberley, L.W.; Cohen, M.B. Expression of antioxidant enzymes in human prostatic adenocarcinoma. Prostate 1997, 32, 229–233. [Google Scholar] [CrossRef]
- Oberley, T.D.; Sempf, J.M.; Oberley, L.W. Immunogold analysis of antioxidant enzymes in common renal cancers. Histol. Histopathol. 1996, 11, 153–160. [Google Scholar]
- Oberley, T.D.; Oberley, L.W. Oxygen radicals and cancer. In Free Radicals in Aging; Yu, B.P., Ed.; CRC Press: Boca Raton, FL, USA, 1993; pp. 247–267. [Google Scholar]
- Bostwick, D.G.; Alexander, E.E.; Singh, R.; Shan, A.; Qian, J.; Santella, R.M.; Oberley, L.W.; Yan, T.; Zhong, W.; Jiang, X.; et al. Antioxidant enzyme expression and reactive oxygen species damage in prostatic intraepithelial neoplasia and cancer. Cancer 2000, 89, 123–134. [Google Scholar] [CrossRef]
- Srivastava, A.; Srivastava, S.; Natu, S.; Gupta, A.; Pal, K.; Agarwal, G.; Singh, U.; Goel, M. Lipid peroxidation and antioxidants in different stages of cervical cancer: Prognostic significance. Indian J Cancer 2009, 46, 297. [Google Scholar] [CrossRef]
- Skrzycki, M.; Majewska, M.; Podsiad, M.; Czeczot, H. Expression and activity of superoxide dismutase isoenzymes in colorectal cancer. Biochim. Pol. 2009, 56, 663–670. [Google Scholar] [CrossRef]
- Miao, L.; Clair, D.K.S. Regulation of Superoxide Dismutase Genes: Implications in Diseases. Free. Radic. Boil. Med. 2009, 47, 344–356. [Google Scholar] [CrossRef]
- Milani, P.; Gagliardi, S.; Cova, E.; Cereda, C. SOD1 Transcriptional and Posttranscriptional Regulation and Its Potential Implications in ALS. Neurol. Res. Int. 2011, 2011, 458427. [Google Scholar] [CrossRef]
- Dunning, S.; Hannivoort, R.A.; De Boer, J.F.; Faber, K.N.; Moshage, H.; Buist-Homan, M.; Buist-Homan, M. Superoxide anions and hydrogen peroxide inhibit proliferation of activated rat stellate cells and induce different modes of cell death. Liver Int. 2009, 29, 922–932. [Google Scholar] [CrossRef]
- Clément, M.V.; Stamenkovic, I. Superoxide anion is a natural inhibitor of FAS-mediated cell death. EMBO J. 1996, 15, 216–225. [Google Scholar] [CrossRef]
- Pervaiz, S.; Ramalingam, J.K.; Hirpara, J.L.; Clement, M.-V. Superoxide anion inhibits drug-induced tumor cell death. FEBS Lett. 1999, 459, 343–348. [Google Scholar] [CrossRef]
- Roy, H.B. Superoxide and hydrogen peroxide in relation to mammalian cell proliferation. Free Radic. Biol. Med. 1995, 18, 775–794. [Google Scholar]
- Sato, K.; Ito, K.; Kohara, H.; Yamaguchi, Y.; Adachi, K.; Endo, H. Negative regulation of catalase gene expression in hepatoma cells. Mol. Cell. Boil. 1992, 12, 2525–2533. [Google Scholar] [CrossRef]
- Min, J.Y.; Lim, S.-O.; Jung, G. Downregulation of catalase by reactive oxygen species via hypermethylation of CpG island II on the catalase promoter. FEBS Lett. 2010, 584, 2427–2432. [Google Scholar] [CrossRef]
- Benhusein, G.M.; Mutch, E.; Aburawi, S.; Williams, F.M. Genotoxic effect induced by hydrogen peroxide in human hepatoma cells using comet assay. J. Med. 2010, 5, 1–6. [Google Scholar] [CrossRef]
- Gopalakrishnan, K.; Low, G.K.M.; Ting, A.P.L.; Srikanth, P.; Slijepcevic, P.; Hande, M.P.; Low, G.K.; Ting, A.P. Hydrogen peroxide induced genomic instability in nucleotide excision repair-deficient lymphoblastoid cells. Genome Integr. 2010, 1, 16. [Google Scholar] [CrossRef]
- Li, M.; Zhao, L.; Liu, J.; Liu, A.-L.; Zeng, W.-S.; Luo, S.-Q.; Bai, X.-C.; Zeng, W.; Luo, S.; Bai, X. Hydrogen Peroxide Induces G2Cell Cycle Arrest and Inhibits Cell Proliferation in Osteoblasts. Anat. Rec. 2009, 292, 1107–1113. [Google Scholar] [CrossRef]
- Del Bello, B.; Paolicchi, A.; Comporti, M.; Pompella, A.; Maellaro, E. Hydrogen peroxide produced during γ-glutamyl transpeptidase activity is involved in prevention of apoptosis and maintenance of proliferation in U937 cells. FASEB J. 1999, 13, 69–79. [Google Scholar] [CrossRef]
- Sattler, M.; Winkler, T.; Verma, S.; Byrne, C.H.; Shrikhande, G.; Salgia, R.; Griffin, J.D. Hematopoietic growth factors signal through the formation of reactive oxygen species. Blood 1999, 93, 2928–2935. [Google Scholar]
- Kang, S.W. Epidermal Growth Factor (EGF)-induced Generation of Hydrogen Peroxide. ROLE IN EGF RECEPTOR-MEDIATED TYROSINE PHOSPHORYLATION. J. Boil. Chem. 1997, 272, 217–221. [Google Scholar]
- Krieger-Brauer, H.I.; Kather, H. The stimulus-sensitive H2O2-generating system present in human fat-cell plasma membranes is multireceptor-linked and under antagonistic control by hormones and cytokines. Biochem. J. 1995, 307, 543–548. [Google Scholar] [CrossRef]
- Meng, T.-C.; Fukada, T.; Tonks, N.K. Reversible oxidation and inactivation of protein tyrosine phosphatases in vivo. F1000-Post-publication peer review of the biomedical literature 2002, 9, 387–399. [Google Scholar] [CrossRef]
- Arnao, M.; Acosta, M.; Del Rio, J.; Garcia-Canovas, F. Inactivation of peroxidase by hydrogen peroxide and its protection by a reductant agent. Biochim. et Biophys. (BBA) Struct. Mol. Enzym. 1990, 1038, 85–89. [Google Scholar] [CrossRef]
- Ghadermarzi, M.; Moosavi-Movahedi, A.A. Determination of the Kinetic Parameters for the “Suicide Substrate” Inactivation of Bovine Liver Catalase by Hydrogen Peroxide. J. Enzym. Inhib. 1996, 10, 167–175. [Google Scholar] [CrossRef]
- Finkel, T. Oxygen radicals and signaling. Curr. Opin. Cell. Biol. 1998, 10, 248–253. [Google Scholar] [CrossRef]
- Sundaresan, M.; Yu, Z.X.; Ferrans, V.J.; Irani, K.; Finkel, T. Requirement for generation of H2O2 for platelet-derived growth factor signal transduction. Science 1995, 270, 296–299. [Google Scholar] [CrossRef]
- Pallepati, P.; Averill-Bates, D. Mild thermotolerance induced at 40 degrees c increases antioxidants and protects hela cells against mitochondrial apoptosis induced by hydrogen peroxide: Role of p53. Arch. Biochem. Biophys. 2010, 495, 97–111. [Google Scholar] [CrossRef]
- Yu, J.; Zhang, L. PUMA, a potent killer with or without p53. Oncogene 2008, 27, S71–S83. [Google Scholar] [CrossRef]
- Salmeen, A.; Andersen, J.N.; Myers, M.P.; Meng, T.-C.; Hinks, J.A.; Tonks, N.K.; Barford, D. Redox regulation of protein tyrosine phosphatase 1B involves a sulphenyl-amide intermediate. Nature 2003, 423, 769–773. [Google Scholar] [CrossRef]
- Lee, S.-R.; Kwon, K.-S.; Kim, S.-R.; Rhee, S.G. Reversible Inactivation of Protein-tyrosine Phosphatase 1B in A431 Cells Stimulated with Epidermal Growth Factor. J. Boil. Chem. 1998, 273, 15366–15372. [Google Scholar] [CrossRef]
- Kwon, J.; Lee, S.-R.; Yang, K.-S.; Ahn, Y.; Kim, Y.J.; Stadtman, E.R.; Rhee, S.G. Reversible oxidation and inactivation of the tumor suppressor PTEN in cells stimulated with peptide growth factors. Proc. Natl. Acad. Sci. USA 2004, 101, 16419–16424. [Google Scholar] [CrossRef]
- Hu, Y.J.; Dolan, M.E.; Bae, R.; Yee, H.; Roy, M.; Glickman, R.; Kiremidjian-Schumacher, L.; Diamond, A.M. Allelic Loss at the GPx-1 Locus in Cancer of the Head and Neck. Boil. Element Res. 2004, 101, 097–106. [Google Scholar] [CrossRef]
- Skrzydlewska, E.; Sulkowski, S.; Koda, M.; Zalewski, B.; Kanczuga-Koda, L.; Sulkowska, M. Lipid peroxidation and antioxidant status in colorectal cancer. World J. Gastroenterol. 2005, 11, 403–406. [Google Scholar] [CrossRef]
- Skrzydlewska, E.; Stankiewicz, A.; Sulkowska, M.; Sulkowski, S.; Kasacka, I. Antioxidant Status and Lipid Peroxidation in Colorectal Cancer. J. Toxicol. Environ. Heal. A 2001, 64, 213–222. [Google Scholar] [CrossRef]
- Beevi, S.S.; Rasheed, M.H.; Geetha, A. Evidence of oxidative and nitrosative stress in patients with cervical squamous cell carcinoma. Clin. Chim. 2007, 375, 119–123. [Google Scholar] [CrossRef]
- Skrzycki, M.; Czeczot, H. Superoxide dismutase as a potential therapeutic agent. Adv. Clin. Exp. Med. 2007, 16, 561–568. [Google Scholar]
- Faucher, K.; Rabinovitch-Chable, H.; Cook-Moreau, J.; Barrière, G.; Sturtz, F.; Rigaud, M. Overexpression of human GPX1 modifies Bax to Bcl-2 apoptotic ratio in human endothelial cells. Mol. Cell. Biochem. 2005, 277, 81–87. [Google Scholar] [CrossRef]
- Pergola, P.E.; Raskin, P.; Toto, R.D.; Meyer, C.J.; Huff, J.W.; Grossman, E.B.; Krauth, M.; Ruiz, S.; Audhya, P.; Christ-Schmidt, H.; et al. Bardoxolone Methyl and Kidney Function in CKD with Type 2 Diabetes. New Engl. J. Med. 2011, 365, 327–336. [Google Scholar] [CrossRef]
- Murawaki, Y.; Tsuchiya, H.; Kanbe, T.; Harada, K.; Yashima, K.; Nozaka, K.; Tanida, O.; Kohno, M.; Mukoyama, T.; Nishimuki, E.; et al. Aberrant expression of selenoproteins in the progression of colorectal cancer. Cancer Letters 2008, 259, 218–230. [Google Scholar] [CrossRef]
- Rebsch, C.M.; Penna, F.J.; Copeland, P.R.; Iii, F.J.P. Selenoprotein expression is regulated at multiple levels in prostate cells. Cell Res. 2006, 16, 940–948. [Google Scholar] [CrossRef]
- López-Lázaro, M. Dual role of hydrogen peroxide in cancer: Possible relevance to cancer chemoprevention and therapy. Cancer Letters 2007, 252, 1–8. [Google Scholar] [CrossRef]
- Cao, C.; Leng, Y.; Liu, X.; Yi, Y.; Li, P.; Kufe, D. Catalase Is Regulated by Ubiquitination and Proteosomal Degradation. Role of the c-Abl and Arg Tyrosine Kinases. Biochemistry 2003, 42, 10348–10353. [Google Scholar] [CrossRef]
- Perry, R.R.; Mazetta, J.; Levin, M.; Barranco, S.C. Glutathione levels and variability in breast tumors and normal tissue. Cancer 1993, 72, 783–787. [Google Scholar] [CrossRef]
- Carretero, J.; Obrador, E.; Anasagasti, M.J.; Martin, J.J.; Vidal-Vanaclocha, F.; Estrela, J.M. Growth-associated changes in glutathione content correlate with liver metastatic activity of B16 melanoma cells. Clin. Exp. Metastasis 1999, 17, 567–574. [Google Scholar] [CrossRef]
- Honda, T.; Coppola, S.; Ghibelli, L.; Cho, S.H.; Kagawa, S.; Spurgers, K.B.; Brisbay, S.M.; A Roth, J.; E Meyn, R.; Fang, B.; et al. GSH depletion enhances adenoviral bax-induced apoptosis in lung cancer cells. Gene Ther. 2004, 11, 249–255. [Google Scholar] [CrossRef]
- Sen, C.K.; Packer, L. Antioxidant and redox regulation of gene transcription. FASEB J. 1996, 10, 709–720. [Google Scholar] [CrossRef]
- Cho, C.-S.; Lee, S.; Lee, G.T.; Woo, H.A.; Choi, E.-J.; Rhee, S.G. Irreversible Inactivation of Glutathione Peroxidase 1 and Reversible Inactivation of Peroxiredoxin II by H2O2 in Red Blood Cells. Antioxidants Redox Signal. 2010, 12, 1235–1246. [Google Scholar] [CrossRef]
- Kim, S.Y.; Kwon, O.J.; Park, J.-W. Inactivation of catalase and superoxide dismutase by singlet oxygen derived from photoactivated dye. Biochimie 2001, 83, 437–444. [Google Scholar] [CrossRef]
- Pajovic, S.; Saičić, Z.; Pejić, S.; Kasapović, J.; Stojiljković, V.; Kanazir, D. Antioxidative biomarkers and cancerogenesis. Jugosl. Med. biohemija 2006, 25, 397–402. [Google Scholar] [CrossRef]
- Lecker, S.H.; Goldberg, A.L.; Mitch, W.E. Protein Degradation by the Ubiquitin–Proteasome Pathway in Normal and Disease States. J. Am. Soc. Nephrol. 2006, 17, 1807–1819. [Google Scholar] [CrossRef]
- Chen, N.; Yi, X.; Abushahin, N.; Pang, S.; Zhang, D.; Kong, B.; Zheng, W. Nrf2 expression in endometrial serous carcinomas and its precancers. Int. J. Clin. Exp. Pathol. 2010, 4, 85–96. [Google Scholar]
- Cho, J.-M.; Manandhar, S.; Lee, H.-R.; Park, H.-M.; Kwak, M.-K. Role of the Nrf2-antioxidant system in cytotoxicity mediated by anticancer cisplatin: Implication to cancer cell resistance. Cancer Letters 2008, 260, 96–108. [Google Scholar] [CrossRef]
- Wang, X.-J.; Sun, Z.; Villeneuve, N.F.; Zhang, S.; Zhao, F.; Li, Y.; Chen, W.; Yi, X.; Zheng, W.; Wondrak, G.T.; et al. Nrf2 enhances resistance of cancer cells to chemotherapeutic drugs, the dark side of Nrf2. CARCIN 2008, 29, 1235–1243. [Google Scholar] [CrossRef]
- Nioi, P.; Nguyen, T. A mutation of Keap1 found in breast cancer impairs its ability to repress Nrf2 activity. Biochem. Biophys. Res. Commun. 2007, 362, 816–821. [Google Scholar] [CrossRef]
- Shibata, T.; Kokubu, A.; Gotoh, M.; Ojima, H.; Ohta, T.; Yamamoto, M.; Hirohashi, S. Genetic Alteration of Keap1 Confers Constitutive Nrf2 Activation and Resistance to Chemotherapy in Gallbladder Cancer. Gastroenterology 2008, 135, 1358–1368.e4. [Google Scholar] [CrossRef]
- Chen, W.; Sun, Z.; Wang, X.-J.; Jiang, T.; Huang, Z.; Fang, D.; Zhang, D.D. Direct interaction between Nrf2 and p21Cip1/WAF1 upregulates the Nrf2-mediated antioxidant response. Mol. Cell 2009, 34, 663–673. [Google Scholar] [CrossRef]
- Lau, A.; Wang, X.-J.; Zhao, F.; Villeneuve, N.F.; Wu, T.; Jiang, T.; Sun, Z.; White, E.; Zhang, D.D. A Noncanonical Mechanism of Nrf2 Activation by Autophagy Deficiency: Direct Interaction between Keap1 and p62. Mol. Cell. Boil. 2010, 30, 3275–3285. [Google Scholar] [CrossRef]
- Franco, R.; Schoneveld, O.; Georgakilas, A.G.; Panayiotidis, M.I. Oxidative stress, DNA methylation and carcinogenesis. Cancer Letters 2008, 266, 6–11. [Google Scholar] [CrossRef]
- Faraonio, R.; Vergara, P.; Di Marzo, D.; Pierantoni, M.G.; Napolitano, M.; Russo, T.; Cimino, F. p53 Suppresses the Nrf2-dependent Transcription of Antioxidant Response Genes. J. Boil. Chem. 2006, 281, 39776–39784. [Google Scholar] [CrossRef]
- Liu, G.H.; Qu, J.; Shen, X. Nf-kappab/p65 antagonizes nrf2-are pathway by depriving cbp from nrf2 and facilitating recruitment of hdac3 to mafk. Biochim. Biophys. Acta 2008, 1783, 713–727. [Google Scholar] [CrossRef]
- Levy, S.; Forman, H.J. c-Myc is a Nrf2-interacting protein that negatively regulates phase II genes through their electrophile responsive elements. IUBMB Life 2010, 62, 237–246. [Google Scholar] [CrossRef]
| Age (years) | 25–34 (7) | 35–44 (22) | 45–54 (41) | 55–64 (6) | 65 ± 79 (3) |
|---|---|---|---|---|---|
| Parity (N) | 0 (3) | 1 (6) | 2 (29) | 3 (24) | >3 (17) |
| Abortions (N) | 0(1) | 1 (30) | 2 (16) | 3 (16) | >3 (16) |
| Uterine bleeding 1 (N) | None (26) | MP (19) | MR (15) | MPM (19) | |
| Diagnosis 2 (N) | PE (16) | UM (12) | SH (25) | CH (21) | ACE (5) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Todorović, A.; Pejić, S.; Gavrilović, L.; Pavlović, I.; Stojiljković, V.; Popović, N.; Pajović, S.B. Expression of Antioxidant Enzymes in Patients with Uterine Polyp, Myoma, Hyperplasia, and Adenocarcinoma. Antioxidants 2019, 8, 97. https://doi.org/10.3390/antiox8040097
Todorović A, Pejić S, Gavrilović L, Pavlović I, Stojiljković V, Popović N, Pajović SB. Expression of Antioxidant Enzymes in Patients with Uterine Polyp, Myoma, Hyperplasia, and Adenocarcinoma. Antioxidants. 2019; 8(4):97. https://doi.org/10.3390/antiox8040097
Chicago/Turabian StyleTodorović, Ana, Snežana Pejić, Ljubica Gavrilović, Ivan Pavlović, Vesna Stojiljković, Nataša Popović, and Snežana B. Pajović. 2019. "Expression of Antioxidant Enzymes in Patients with Uterine Polyp, Myoma, Hyperplasia, and Adenocarcinoma" Antioxidants 8, no. 4: 97. https://doi.org/10.3390/antiox8040097
APA StyleTodorović, A., Pejić, S., Gavrilović, L., Pavlović, I., Stojiljković, V., Popović, N., & Pajović, S. B. (2019). Expression of Antioxidant Enzymes in Patients with Uterine Polyp, Myoma, Hyperplasia, and Adenocarcinoma. Antioxidants, 8(4), 97. https://doi.org/10.3390/antiox8040097







