Juglone in Oxidative Stress and Cell Signaling
Abstract
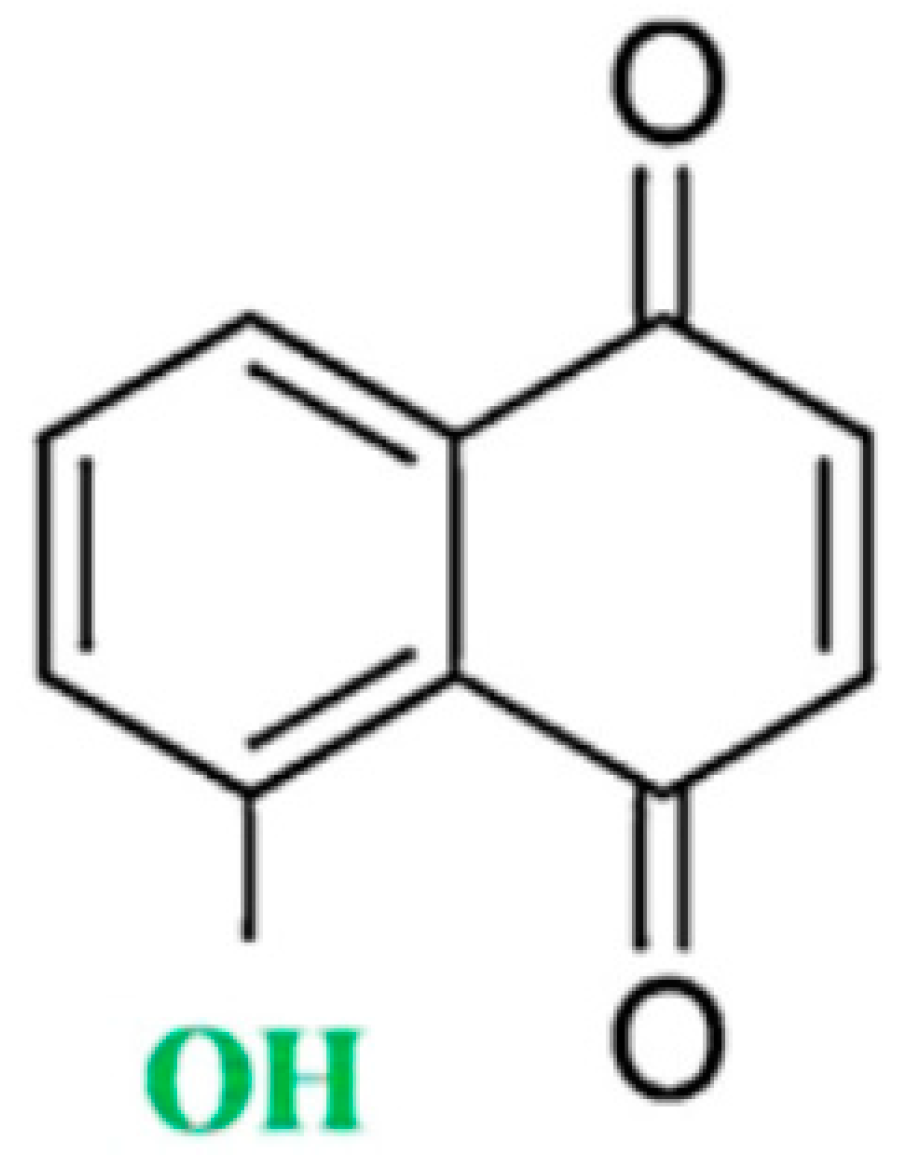
:1. Introduction
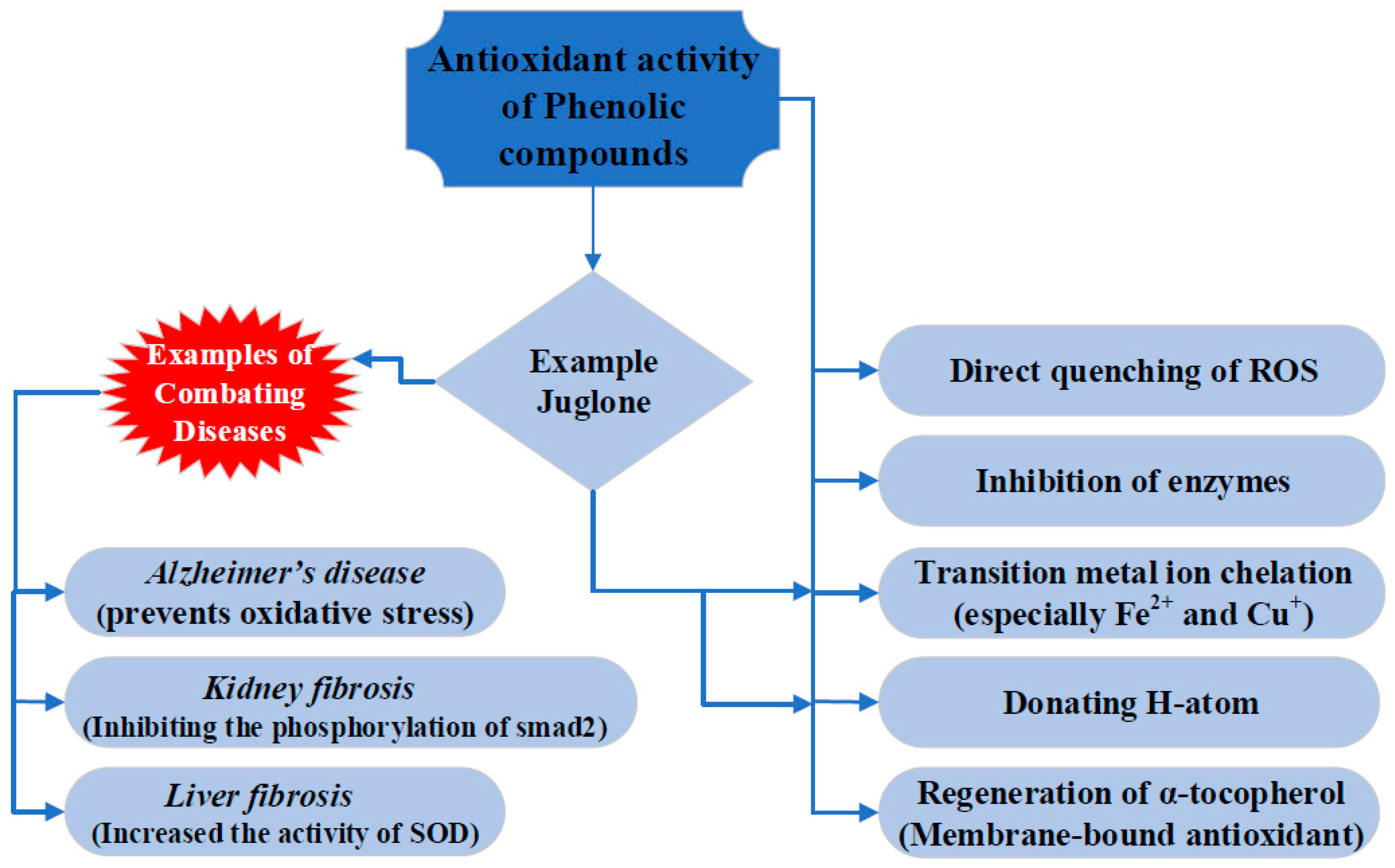
2. Antioxidant Properties of Juglone
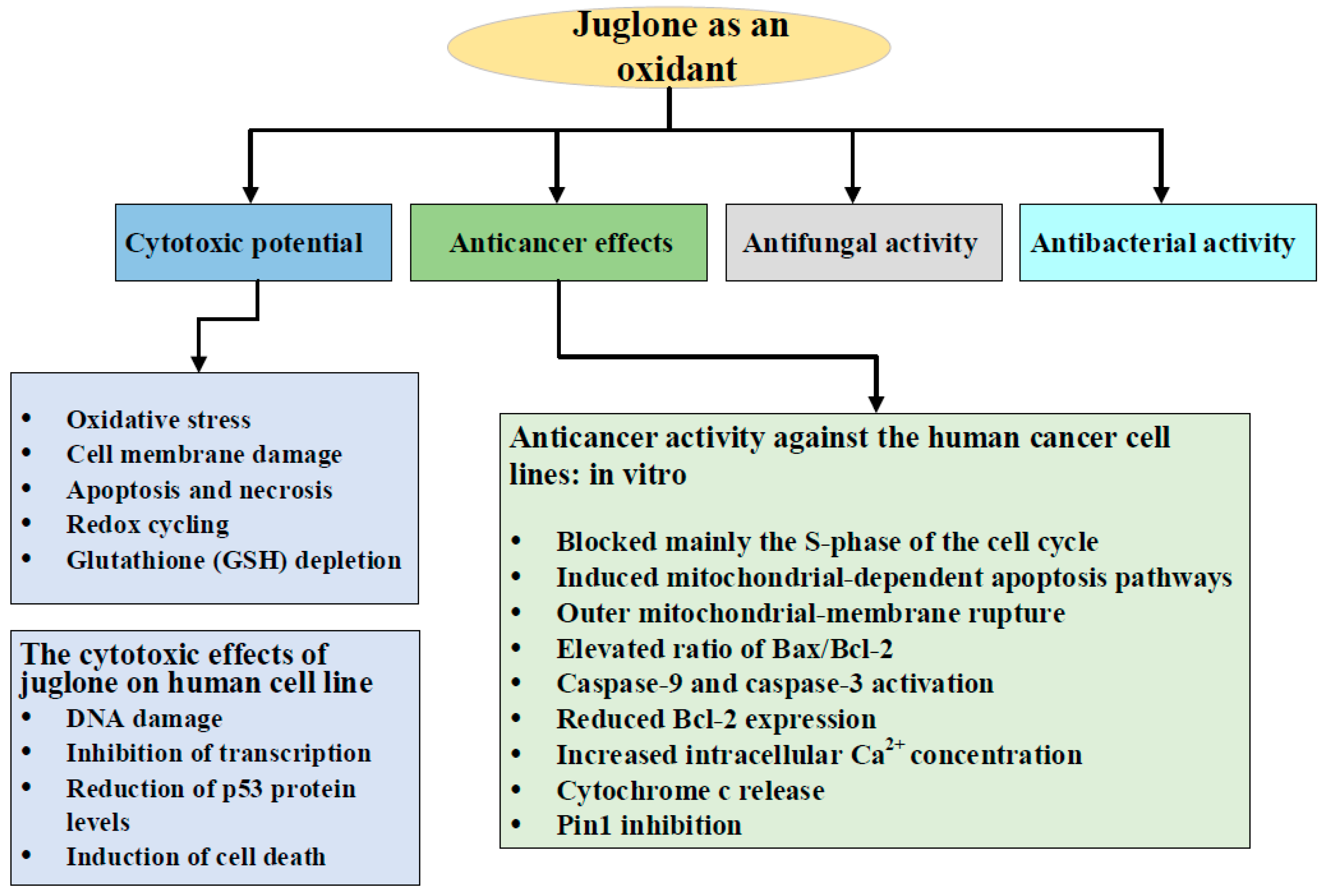
3. Juglone as an Oxidant: Cytotoxic Potential
4. Anticancer Effects of Juglone
4.1. Anticancer Activity against the Human Cancer Cell Lines: in Vitro
4.2. Glioma Cells
4.3. In Vivo Anticancer Effects of Juglone
5. Antimicrobial Activity of Juglone
5.1. Antifungal Activity
5.2. Antibacterial Activity
6. Role of Juglone as an Inhibitor of Peptidyl-Prolyl cis/trans Isomerase, Pin 1
7. Effects of Juglone on Cell Signaling Pathways
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Carrion, J.S.; Fuentes, N.; Gonzalez-Samperiz, P.; Sanchez Quirante, L.; Finlayson, J.C.; Fernandez, S.; Andrade, A. Holocene environmental change in a montane region of southern Europe with a long history of human settlement. Quat. Sci. Rev. 2007, 26, 1455–1475. [Google Scholar] [CrossRef]
- Fathi Azad, F.; Garjani, A.; Motevalian Naeini, A. Study of hypoglycemic activity of the hydroalcholic extract of juglans regia in normal and diabetic rats. Pharma Sci. 2006, 2, 13–17. [Google Scholar]
- Ma, Y.; Njike, V.Y.; Millet, J.; Dutta, S.; Doughty, K.; Treu, J.A.; Katz, D.L. Effects of walnut consumption on endothelial function in type 2 diabetic subjects. A randomized controlled cross over trial. Diabetes Care 2010, 33, 227–232. [Google Scholar] [CrossRef] [PubMed]
- Aronis, K.N.; Vamvini, M.T.; Chamberland, J.P.; Sweeney, L.L.; Brennan, A.M.; Magkos, F.; Mantzoros, C.S. Short-term walnut consumption increases circulating total adiponectin and apolipoprotein A concentrations, but does not affect markers of inflammation or vascular injury in obese humans with the metabolic syndrome: Data from a double-blinded, randomized, placebo-controlled study. Metabolism 2012, 61, 577–582. [Google Scholar] [CrossRef] [PubMed]
- Delaviz, H.; Mohammadi, J.; Ghalamfarsa, G.; Mohammadi, B.; Farhadi, N. A review study on phytochemistry and pharmacology applications of Juglans regia plant. Pharmacogn. Rev. 2017, 11, 145–152. [Google Scholar] [CrossRef]
- Halvorsen, B.L.; Holte, K.; Myhrstad, M.C.; Barikmo, I.; Hvattum, E.; Remberg, S.F.; Wold, A.B.; Haffner, K.; Baugerød, H.; Andersen, L.F.; et al. A systematic screening of total antioxidants in dietary plants. J. Nutr. 2002, 132, 461–471. [Google Scholar] [CrossRef] [PubMed]
- Pellegrini, N.; Serafini, M.; Salvatore, S.; Del Rio, D.; Bianchi, M.; Brighenti, F. Total antioxidant capacity of spices, dried fruits, nuts, pulses, cereals and sweets consumed in Italy assessed by three different in vitro assays. Mol. Nutr. Food Res. 2006, 50, 1030–1038. [Google Scholar] [CrossRef]
- Espin, J.C.; Garcia-Conesa, M.T.; Tomás-Barberán, F.A. Nutraceuticals: Facts and fiction. Phytochemistry 2007, 68, 2986–3008. [Google Scholar] [CrossRef]
- Vinson, J.A.; Cai, Y. Nuts, especially walnuts, have both antioxidant quantity and efficacy and exhibit significant potential health benefits. Food Funct. 2012, 3, 134–140. [Google Scholar] [CrossRef]
- Nahrstedt, A.; Vetter, U.; Hammerschmidt, F.J. Composition of the steam distillation product from the leaves of Juglans regia. Planta Medica 1981, 42, 313–332. [Google Scholar] [CrossRef]
- Hirakawa, K.; Ogiue, E.; Motoyoshiya, J.; Yajima, M. Naphthoquinones from juglandaceae. Phytochemistry 1986, 25, 1494–1495. [Google Scholar] [CrossRef]
- Wichtl, M.; Anton, R. Plantes Thérapeutiques. Tradition, Pratique Officinale, Science et Thérapeutique, 2nd ed.; Librairie Eyrolles: Paris, France, 2003. [Google Scholar]
- Sharma, N.; Ghosh, P.; Sharma, U.K.; Sood, S.; Sinha, A.K.; Gulati, A. Microwave-assisted efficient extraction and stability of juglone in different solvents from Juglans regia: Quantification of six phenolic constituents by validated RP-HPLC and evaluation of antimicrobial activity. Anal. Lett. 2009, 42, 2592–2609. [Google Scholar] [CrossRef]
- Inbaraj, J.J.; Chignell, C.F. Cytotoxic action of juglone and plumbagin: A mechanistic study using HaCaT keratinocytes. Chem. Res. Toxicol. 2004, 17, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Colaric, M.; Veberic, R.; Solar, A.; Hudina, M.; Stampar, F. Phenolic acids, syringaldehyde, and juglone in fruits of different cultivars of Juglans regia L. J. Agric. Food Chem. 2005, 53, 6390–6396. [Google Scholar] [CrossRef] [PubMed]
- Soderquist, C.J. Juglone and allelopathy. J. Chem. Educ. 1973, 50, 782–783. [Google Scholar] [CrossRef]
- Moir, M.; Thomson, R.H. Naphthaquinones in Lomatia species. Phytochemistry 1973, 12, 1351–1353. [Google Scholar] [CrossRef]
- Nageshwar, G.; Radhakrishnainah, M.; Narayana, L.L. Chemotaxonomy of Caesalpinia. Curr. Sci. 1984, 53, 813–814. [Google Scholar]
- Lee, C.H.; Lee, H.S. Color alteration and acaricidal activity of juglone isolated from Caesalpinia sappan heartwoods against Dermatophagoides spp. J. Microbiol. Biotechnol. 2006, 16, 1591–1596. [Google Scholar]
- Marichkova, L.; Kumanova, B. Isolation of flavonoids and some accompanying substances from the above-ground part of some Astragalus centralpinus family Leguminosae (Russian). Probl. Farm. 1981, 9, 63–74. [Google Scholar]
- Walnut Toxicity. Available online: www.omafra.gov.on.ca/english/crops/facts/info_walnut_toxicity.htm (accessed on 25 February 2019).
- Vogel, A.; Reinschauer, C. Ueber einen neuen organischen Körper in deu Fruchtschalen der Juglans regia. Neues Repertorium für Pharmacie 1856, 5, 106–110. [Google Scholar]
- Stickney, J.; Hoy, P. Toxic action of black walnut. Trans Wis State Hort Soc. 1881, 11, 166–167. [Google Scholar]
- Auyong, T.K.; Westfall, B.A.; Russell, R.L. Pharmacological aspects of juglone. Toxicon 1963, 1, 235–239. [Google Scholar] [CrossRef]
- Clark, A.M.; Jurgens, T.M.; Hufford, C.D. Antimicrobial activity of juglone. Phytother. Res. 1990, 4, 11–14. [Google Scholar] [CrossRef]
- Sugie, S.; Okamoto, K.; Rahman, K.M.; Tanaka, T.; Kawai, K.; Yamahara, J.; Mori, H. Inhibitory effects of plumbagin and juglone on azoxymethane-induced intestinal carcinogenesis in rats. Cancer Lett. 1998, 127, 177–183. [Google Scholar] [CrossRef]
- Zhang, W.; Liu, A.; Li, Y.; Zhao, X.; Lv, S.; Zhu, W.; Jin, Y. Anticancer activity and mechanism of juglone on human cervical carcinoma HeLa cells. Can. J. Physiol. Pharmacol. 2012, 90, 1553–1558. [Google Scholar] [CrossRef] [PubMed]
- Fang, F.; Chen, S.; Ma, J.; Cui, J.; Li, Q.; Meng, G.; Wang, L. Juglone suppresses epithelial-mesenchymal transition in prostate cancer cells via the protein kinase B/glycogen synthase kinase-3β/Snail signaling pathway. Oncol. Lett. 2018, 16, 2579–2584. [Google Scholar] [CrossRef] [PubMed]
- Chobot, V.; Hadacek, F. Milieu-dependent pro- and antioxidant activity of juglone may explain linear and nonlinear effects on seedling development. J. Chem. Ecol. 2009, 35, 383–930. [Google Scholar] [CrossRef]
- Liu, X.; Chen, Y.; Zhang, Y.; Du, J.; Lv, Y.; Mo, S.; Liu, Y.; Ding, F.; Wu, J.; Li, J. Juglone potentiates TRAIL-induced apoptosis in human melanoma cells via activating the ROS-p38-p53 pathway. Mol. Med. Rep. 2017, 16, 9645–9651. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Liu, K.; Wang, X.F.; Sun, D.J. Juglone reduces growth and migration of U251 glioblastoma cells and disrupts angiogenesis. Oncol. Rep. 2017, 38, 1959–1966. [Google Scholar] [CrossRef]
- Leopoldini, M.; Russo, N.; Chiodo, S.; Toscano, M. Iron chelation by the powerful antioxidant flavonoid quercetin. J. Agric. Food Chem. 2006, 54, 6343–6351. [Google Scholar] [CrossRef]
- Leopoldini, M.; Marino, T.; Russo, N.; Toscano, M. Antioxidant properties of phenolic compounds: H-atom versus electron transfer mechanism. J. Phys. Chem. A. 2004, 108, 4916–4922. [Google Scholar] [CrossRef]
- Tejero, I.; González-García, N.; González-Lafont, À.; Lluch, J.M. Tunneling in green tea: Understanding the antioxidant activity of catechol-containing compounds. A variational transition-state theory study. J. Am. Chem. Soc. 2007, 129, 5846–5854. [Google Scholar] [CrossRef] [PubMed]
- van Acker, S.A.; de Groot, M.J.; van den Berg, D.J.; Tromp, M.N.; Donné-Op den Kelder, G.; van der Vijgh, W.J.; Bast, A. A quantum chemical explanation of the antioxidant activity of flavonoids. Chem. Res. Toxicol. 1996, 9, 1305–1312. [Google Scholar] [CrossRef] [PubMed]
- Russo, N.; Toscano, M.; Uccella, N. Semiempirical molecular modeling into quercetin reactive site: Structural, conformational, and electronic features. J. Agric. Food Chem. 2000, 48, 3232–3237. [Google Scholar] [CrossRef] [PubMed]
- Nenadis, N.; Sigalas, M.P. A DFT study on the radical scavenging activity of maritimetin and related aurones. J. Phys. Chem. A. 2008, 112, 12196–12202. [Google Scholar] [CrossRef] [PubMed]
- Jin, R. A DFT study on the radical scavenging activity of juglone and its derivatives. J. Mol. Struct. Theochem 2010, 939, 9–13. [Google Scholar] [CrossRef]
- de Castro, E.; Hegi de Castro, S.; Johnson, TE. Isolation of long-lived mutants in Caenorhabditis elegans using selection for resistance to juglone. Free Radic. Biol. Med. 2004, 37, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Bentes, A.L.; Borges, R.S.; Monteiro, W.R.; de Macedo, L.G.; Alves, C.N. Structure of dihydrochalcones and related derivatives and their scavenging and antioxidant activity against oxygen and nitrogen radical species. Molecules 2011, 16, 1749–1760. [Google Scholar] [CrossRef]
- Leopoldini, M.; Russo, N.; Toscano, M. The molecular basis of working mechanism of natural polyphenolic antioxidants. Food Chem. 2011, 125, 288–306. [Google Scholar] [CrossRef]
- Galano, A.; Mazzone, G.; Alvarez-Diduk, R.; Marino, T.; Alvarez-Idaboy, J.R.; Russo, N. Food antioxidants: Chemical insights at the molecular level. Annu. Rev. Food Sci. Technol. 2016, 7, 335–352. [Google Scholar] [CrossRef]
- Gülçin, I.; Huyut, Z.; Elmastaş, M.; Aboul-Enein, H.Y. Radical scavenging and antioxidant activity of tannic acid. Arab. J. Chem. 2010, 3, 43–53. [Google Scholar] [CrossRef]
- Tamafo Fouegue, A.D.; Ghogomu, J.N.; Bikélé Mama, D.; Nkungli, N.K.; Younang, E. Structural and antioxidant properties of compounds obtained from Fe2+ chelation by juglone and two of its derivatives: DFT, QTAIM, and NBO studies. Bioinorg. Chem. Appl. 2016, 2016, 8636409. [Google Scholar] [CrossRef]
- Galas, M.C.; Dourlen, P.; Bégard, S.; Ando, K.; Blum, D.; Hamdane, M.; Buée, L. The peptidylprolyl cis/trans-isomerase Pin1 modulates stress-induced dephosphorylation of Tau in neurons. Implication in a pathological mechanism related to Alzheimer disease. J. Biol. Chem. 2006, 281, 19296–19304. [Google Scholar] [CrossRef]
- Pandareesh, M.D.; Chauhan, V.; Chauhan, A. Walnut supplementation in the diet reduces oxidative damage and improves antioxidant status in transgenic mouse model of Alzheimer’s disease. J. Alzheimers Dis. 2018, 64, 1295–1305. [Google Scholar] [CrossRef]
- Li, J.H.; Zhu, H.J.; Huang, X.R.; Lai, K.N.; Johnson, R.J.; Lan, H.Y. Smad7 inhibits fibrotic effect of TGF-β on renal tubular epithelial cells by blocking Smad2 activation. J. Am. Soc. Nephrol. 2002, 13, 1464–1472. [Google Scholar] [CrossRef] [PubMed]
- Djamali, A. Oxidative stress as a common pathway to chronic tubulointerstitial injury in kidney allografts. Am. J. Physiol. Renal Physiol. 2007, 293, F445–F455. [Google Scholar] [CrossRef]
- Reese, S.; Vidyasagar, A.; Jacobson, L.; Acun, Z.; Esnault, S.; Hullett, D.; Malter, J.S.; Djamali, A. The Pin 1 inhibitor juglone attenuates kidney fibrogenesis via Pin 1-independent mechanisms in the unilateral ureteral occlusion model. Fibrogenesis Tissue Repair 2010, 3, 1. [Google Scholar] [CrossRef]
- Zhou, D.J.; Mu, D.; Jiang, M.D.; Zheng, S.M.; Zhang, Y.; He, S.; Weng, M.; Zeng, W.Z. Hepatoprotective effect of juglone on dimethylnitrosamine-induced liver fibrosis and its effect on hepatic antioxidant defence and the expression levels of α-SMA and collagen III. Mol. Med. Rep. 2015, 12, 4095–4102. [Google Scholar] [CrossRef]
- Klotz, L.-O.; Hou, X.; Jacob, C. 1,4-naphthoquinones: From oxidative damage to cellular and inter-cellular signaling. Molecules 2014, 19, 14902–14918. [Google Scholar] [CrossRef]
- Lown, J.W. The mechanism of action of quinone antibiotics. Mol. Cell. Biochem. 1983, 55, 17–40. [Google Scholar] [CrossRef]
- Powis, G. Free radical formation by antitumor quinones. Free Radic. Biol. Med. 1989, 6, 63–101. [Google Scholar] [CrossRef]
- Monks, T.J.; Walker, S.E.; Flynn, L.M.; Conti, C.J.; DiGiovanni, J. Epidermal ornithine decarboxylase induction and mouse skin tumor promotion by quinones. Carcinogenesis 1990, 11, 1795–1801. [Google Scholar] [CrossRef] [PubMed]
- Ollinger, K.; Brunmark, A. Effect of hydroxy substituent position on 1,4-naphthoquinone toxicity to rat hepatocytes. J. Biol. Chem. 1991, 266, 21496–21503. [Google Scholar] [PubMed]
- Parekh, H.; Chavan, S.; Chitnis, M. Modulation of the thiol pools by vitamin K3 and its effect on survival of sensitive and resistant murine tumor cells. Anticancer Drugs 1991, 2, 159–168. [Google Scholar] [CrossRef]
- Tan, D.T.C.; Osman, H.; Mohamad, S.; Kamaruddin, A.H. Synthesis and antibacterial activity of juglone derivatives. J. Chem. Chem. Eng. 2012, 6, 84–89. [Google Scholar]
- Zakavi, F.; Golpasand Hagh, L.; Daraeighadikolaei, A.; Farajzadeh Sheikh, A.; Daraeighadikolaei, A.; Leilavi Shooshtari, Z. Antibacterial effect of juglans regia bark against oral pathologic bacteria. Int. J. Dent. 2013, 2013, 854765. [Google Scholar] [CrossRef] [PubMed]
- Anaissi-Afonso, L.; Oramas-Royo, S.; Ayra-Plasencia, J.; Martín-Rodríguez, P.; García-Luis, J.; Lorenzo-Castrillejo, I.; Fernández-Pérez, L.; Estévez-Braun, A.; Machín, F. Lawsone, juglone, and β-lapachone derivatives with enhanced mitochondrial-based toxicity. ACS Chem. Biol. 2018, 13, 1950–1957. [Google Scholar] [CrossRef] [PubMed]
- Paulsen, M.T.; Ljungman, M. The natural toxin juglone causes degradation of p53 and induces rapid H2AX phosphorylation and cell death in human fibroblasts. Toxicol. Appl. Pharmacol. 2005, 209, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Segura-Aguilar, J.; Jönsson, K.; Tidefelt, U.; Paul, C. The cytotoxic effects of 5-OH-1,4-naphthoquinone and 5,8-diOH-1,4-naphthoquinone on doxorubicin-resistant human leukemia cells (HL-60). Leuk. Res. 1992, 16, 631–637. [Google Scholar] [CrossRef]
- Babula, P.; Adam, V.; Kizek, R.; Sladky, Z.; Havel, L. Naphthoquinones as allelochemical triggers of programmed cell death. Environ. Exp. Bot. 2009, 65, 330–337. [Google Scholar] [CrossRef]
- Aithal, B.K.; Kumar, M.R.; Rao, B.N.; Udupa, N.; Rao, B.S. Juglone, a naphthoquinone from walnut, exerts cytotoxic and genotoxic effects against cultured melanoma tumor cells. Cell Biol. Int. 2009, 33, 1039–1049. [Google Scholar] [CrossRef]
- d’Arcy Doherty, M.; Rodgers, A.; Cohen, G.M. Mechanisms of toxicity of 2- and 5-hydroxy-1,4-naphthoquinone; absence of a role for redox cycling in the toxicity of 2-hydroxy-1,4-naphthoquinone to isolated hepatocytes. J. Appl. Toxicol. 1987, 7, 123–129. [Google Scholar] [CrossRef]
- Zhang, R.; Hirsch, O.; Mohsen, M.; Samuni, A. Effects of nitroxide stable radicals on juglone cytotoxicity. Arch. Biochem. Biophys. 1994, 312, 385–391. [Google Scholar] [CrossRef]
- Lewis, D.C.; Shibamoto, T. Relative metabolism of quinones to semiquinone radicals in xanthine oxidase system. J. Appl. Toxicol. 1989, 9, 291–295. [Google Scholar] [CrossRef]
- Xu, J.; Cheng, Q.; Arnér, E.S.J. Details in the catalytic mechanism of mammalian thioredoxin reductase 1 revealed using point mutations and juglone-coupled enzyme activities. Free Radic. Biol. Med. 2016, 94, 110–120. [Google Scholar] [CrossRef]
- Kappus, H.; Sies, H. Toxic drug effects associated with oxygen metabolism: Redox cycling and lipid peroxidation. Experientia 1981, 37, 1233–1241. [Google Scholar] [CrossRef]
- Kumbhar, A.S.; Padhye, S.B.; Jitender; Kale, R.K. Naturally occurring hydroxy napthoquinones and their iron complexes as modulators of radiation induced lipid peroxidation in synaptosomes. Met Based Drugs 1997, 4, 279–285. [Google Scholar] [CrossRef]
- Gant, T.W.; Rao, D.N.; Mason, R.P.; Cohen, G.M. Redox cycling and sulphydryl arylation; their relative importance in the mechanism of quinone cytotoxicity to isolated hepatocytes. Chem. Biol. Interact. 1988, 65, 157–173. [Google Scholar] [CrossRef]
- Bruins, J.J.; Albada, B.; van Delft, F. Ortho-Quinones and analogues thereof: Highly reactive intermediates for fast and selective biofunctionalization. Chemistry 2018, 24, 4749–4756. [Google Scholar] [CrossRef]
- Babich, H.; Stern, A. In vitro cytotoxicities of 1,4-naphthoquinone and hydroxylated 1,4-naphthoquinones to replicating cells. J. Appl. Toxicol. 1993, 13, 353–358. [Google Scholar] [CrossRef]
- Meskelevicius, D.; Sidlauskas, K.; Bagdonaviciute, R.; Liobikas, J.; Majiene, D. Juglone exerts cytotoxic, anti-proliferative and anti-invasive effects on glioblastoma multiforme in a cell culture model. Anticancer Agents Med. Chem. 2016, 16, 1190–1197. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.L.; Yu, X.F.; Qu, S.C.; Qu, X.R.; Jiang, Y.F.; Sui, D.Y. Juglone, from Juglans mandshruica Maxim, inhibits growth and induces apoptosis in human leukemia cell HL-60 through a reactive oxygen species-dependent mechanism. Food Chem. Toxicol. 2012, 50, 590–596. [Google Scholar] [CrossRef] [PubMed]
- Kviecinski, M.R.; Pedrosa, R.C.; Felipe, K.B.; Farias, M.S.; Glorieux, C.; Valenzuela, M.; Sid, B.; Benites, J.; Valderrama, J.A.; Verrax, J.; et al. Inhibition of cell proliferation and migration by oxidative stress from ascorbate-driven juglone redox cycling in human bladder-derived T24 cells. Biochem. Biophys. Res. Commun. 2012, 421, 268–273. [Google Scholar] [CrossRef]
- Ourique, F.; Kviecinski, M.R.; Felipe, K.B.; Correia, J.F.; Farias, M.S.; Castro, L.S.; Grinevicius, V.M.; Valderrama, J.; Rios, D.; Benites, J.; et al. DNA damage and inhibition of Akt pathway in MCF-7 cells and Ehrlich tumor in mice treated with 1,4-naphthoquinones in combination with ascorbate. Oxid. Med. Cell. Longev. 2015, 2015, 495305. [Google Scholar] [CrossRef] [PubMed]
- Ourique, F.; Kviecinski, M.R.; Zirbel, G.; Castro, L.S.E.P.W.; Gomes Castro, A.J.; Mena Barreto Silva, F.R.; Valderrama, J.A.; Rios, D.; Benites, J.; Calderon, P.B.; et al. In vivo inhibition of tumor progression by 5 hydroxy-1,4-naphthoquinone (juglone) and 2-(4-hydroxyanilino)-1,4-naphthoquinone (Q7) in combination with ascorbate. Biochem. Biophys. Res Commun. 2016, 477, 640–646. [Google Scholar] [CrossRef]
- Sajadimajd, S.; Yazdanparast, R.; Roshanzamir, F. Augmentation of oxidative stress-induced apoptosis in MCF7 cells by ascorbate-tamoxifen and/or ascorbate-juglone treatments. In Vitro Cell. Dev. Biol. Anim. 2016, 52, 193–203. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.J.; Bao, J.L.; Wu, G.S.; Xu, W.S.; Huang, M.Q.; Chen, X.P.; Wang, Y.T. Quinones derived from plant secondary metabolites as anti-cancer agents. Anticancer Agents Med. Chem. 2013, 13, 456–463. [Google Scholar] [CrossRef] [PubMed]
- Driscoll, J.S.; Hazard, G.F.; Wood, H.B.; Goldin, A. Structure-antitumor activity relationships among quinone derivatives. Cancer Chemother. Rep. 1974, 4, 1–362. [Google Scholar]
- Verma, R.P. Anti-cancer activities of 1,4-naphthoquinones: A QSAR study. Anticancer Agents Med. Chem. 2006, 6, 489–499. [Google Scholar] [CrossRef]
- Aubel-Sadron, G.; Londos-Gagliardi, D. Daunorubicin and doxorubicin, anthracycline antibiotics, a physicochemical and biological review. Biochimie 1984, 66, 333–352. [Google Scholar] [CrossRef]
- Widhalm, J.R.; Rhodes, D. Biosynthesis and molecular actions of specialized 1,4-naphthoquinone natural products produced by horticultural plants. Hortic. Res. 2016, 3, 16046. [Google Scholar] [CrossRef]
- Kayashima, T.; Mori, M.; Yoshida, H.; Mizushina, Y.; Matsubara, K. 1,4-Naphthoquinone is a potent inhibitor of human cancer cell growth and angiogenesis. Cancer Lett. 2009, 278, 34–40. [Google Scholar] [CrossRef]
- Zhang, X.B.; Zou, C.L.; Duan, Y.X.; Wu, F.; Li, G. Activity guided isolation and modification of juglone from Juglans regia as potent cytotoxic agent against lung cancer cell lines. BMC Complement. Altern. Med. 2015, 15, 396. [Google Scholar] [CrossRef]
- Kamei, H.; Koide, T.; Kojima, T.; Hashimoto, Y.; Hasegawa, M. Inhibition of cell growth in culture by quinones. Cancer Biother. Radiopharm. 1998, 13, 185–188. [Google Scholar] [CrossRef]
- Xu, H.L.; Yu, X.F.; Qu, S.C.; Zhang, R.; Qu, X.R.; Chen, Y.P.; Ma, X.Y.; Sui, D.Y. Anti-proliferative effect of Juglone from Juglans mandshurica Maxim on human leukemia cell HL-60 by inducing apoptosis through the mitochondria-dependent pathway. Eur. J. Pharmacol. 2010, 645, 14–22. [Google Scholar] [CrossRef]
- Ji, Y.B.; Qu, Z.Y.; Zou, X. Juglone-induced apoptosis in human gastric cancer SGC-7901 cells via the mitochondrial pathway. Exp. Toxicol. Pathol. 2011, 63, 69–78. [Google Scholar] [CrossRef]
- Fang, F.; Qin, Y.; Qi, L.; Fang, Q.; Zhao, L.; Chen, S.; Li, Q.; Zhang, D.; Wang, L. Juglone exerts antitumor effect in ovarian cancer cells. Iran. J. Basic Med. Sci. 2015, 18, 544–548. [Google Scholar]
- Xu, H.; Yu, X.; Qu, S.; Sui, D. Juglone, isolated from Juglans mandshurica Maxim, induces apoptosis via down-regulation of AR expression in human prostate cancer LNCaP cells. Bioorg. Med. Chem. Lett. 2013, 23, 3631–3634. [Google Scholar] [CrossRef]
- Ji, Y.B.; Xin, G.S.; Qu, Z.Y.; Zou, X.; Yu, M. Mechanism of juglone-induced apoptosis of MCF-7 cells by the mitochondrial pathway. Genet. Mol. Res. 2016, 15. [Google Scholar] [CrossRef]
- Bayram, D.; Armagan, İ.; Özgöcmen, M.; Senol, N.; Calapoglu, M. Determination of apoptotic effect of juglone on human bladder cancer TCC-SUP and RT-4 cells: An in vitro study. J. Environ. Pathol. Toxicol. Oncol. 2018, 37, 173–181. [Google Scholar] [CrossRef]
- Mamelak, A.N.; Jacoby, D.B. Targeted delivery of antitumoral therapy to glioma and other malignancies with synthetic chlorotoxin (TM-601). Expert Opin. Drug Deliv. 2007, 4, 175–186. [Google Scholar] [CrossRef] [PubMed]
- Goodenberger, M.L.; Jenkins, R.B. Genetics of adult glioma. Cancer Genet. 2012, 205, 613–621. [Google Scholar] [CrossRef] [PubMed]
- Sidlauskas, K.; Sidlauskiene, R.; Li, N.; Liobikas, J. 5-Hydroxy-1,4-naphthalenedione exerts anticancer effects on glioma cells through interaction with the mitochondrial electron transport chain. Neurosci. Lett. 2017, 639, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Bonjar, G.H.S.; Nik, A.K.; Aghighi, S. Antibacterial and antifungal survey in plants used in indigenous herbal-medicine of south east regions of Iran. J. Biol. Sci. 2004, 4, 405–412. [Google Scholar]
- Wianowska, D.; Garbaczewska, S.; Cieniecka-Roslonkiewicz, A.; Dawidowicz, A.L.; Jankowska, A. Comparison of antifungal activity of extracts from different Juglans regia cultivars and juglone. Microb. Pathog. 2016, 100, 263–267. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Wang, Z.; Wu, R.; Jiang, D.; Bai, B.; Tan, D.; Yan, T.; Sun, X.; Zhang, Q.; Wu, Z. Proteomic analysis of the antibacterial mechanism of action of juglone against Staphylococcus aureus. Nat. Prod. Commun. 2016, 11, 825–827. [Google Scholar] [CrossRef] [PubMed]
- Zmantar, T.; Miladi, H.; Kouidhi, B.; Chaabouni, Y.; Ben Slama, R.; Bakhrouf, A.; Mahdouani, K.; Chaieb, K. Use of juglone as antibacterial and potential efflux pump inhibitors in Staphylococcus aureus isolated from the oral cavity. Microb. Pathog. 2016, 101, 44–49. [Google Scholar] [CrossRef] [PubMed]
- Arasoglu, T.; Mansuroglu, B.; Derman, S.; Gumus, B.; Kocyigit, B.; Acar, T.; Kocacaliskan, I. Enhancement of antifungal activity of juglone (5-hydroxy-1,4-naphthoquinone) using a poly (d,l-lactic-co-glycolic acid) (PLGA) nanoparticle system. J. Agric. Food Chem. 2016, 64, 7087–7094. [Google Scholar] [CrossRef]
- Arasoglu, T.; Derman, S.; Mansuroglu, B.; Yelkenci, G.; Kocyigit, B.; Gumus, B.; Acar, T.; Kocacaliskan, I. Synthesis, characterization and antibacterial activity of juglone encapsulated PLGA nanoparticles. J. Appl. Microbiol. 2017, 123, 1407–1419. [Google Scholar] [CrossRef]
- Hennig, L.; Christner, C.; Kipping, M.; Schelbert, B.; Rücknagel, K.P.; Grabley, S.; Küllertz, G.; Fischer, G. Selective inactivation of parvulin-like peptidyl-prolyl cis/trans isomerases by juglone. Biochemistry 1998, 37, 5953–5960. [Google Scholar] [CrossRef]
- Jordens, J.; Janssens, V.; Longin, S.; Stevens, I.; Martens, E.; Bultynck, G.; Engelborghs, Y.; Lescrinier, E.; Waelkens, E.; Goris, J.; et al. The protein phosphatase 2A phosphatase activator is a novel peptidyl-prolyl cis/trans-isomerase. J. Biol. Chem. 2006, 281, 6349–6357. [Google Scholar] [CrossRef] [PubMed]
- Hamdane, M.; Smet, C.; Sambo, A.V.; Leroy, A.; Wieruszeski, J.M.; Delobel, P.; Maurage, C.A.; Ghestem, A.; Wintjens, R.; Bégard, S.; et al. Pin1: A therapeutic target in Alzheimer neurodegeneration. J. Mol. Neurosci. 2002, 19, 275–287. [Google Scholar] [CrossRef]
- Esnault, S.; Braun, R.K.; Shen, Z.J.; Xiang, Z.; Heninger, E.; Love, R.B.; Sandor, M.; Malter, J.S. Pin1 modulates the type 1 immune response. PLoS ONE 2007, 2, e226. [Google Scholar] [CrossRef]
- Esnault, S.; Rosenthal, L.A.; Shen, Z.J.; Sedgwick, J.B.; Szakaly, R.J.; Sorkness, R.L.; Malter, J.S. A critical role for Pin1 in allergic pulmonary eosinophilia in rats. J. Allergy Clin. Immunol. 2007, 120, 1082–1088. [Google Scholar] [CrossRef] [PubMed]
- Fan, G.; Fan, Y.; Gupta, N.; Matsuura, I.; Liu, F.; Zhou, X.Z.; Lu, K.P.; Gélinas, C. Peptidyl-prolyl isomerase Pin1 markedly enhances the oncogenic activity of the Rel proteins in the nuclear factor-κB family. Cancer Res. 2009, 69, 4589–4597. [Google Scholar] [CrossRef]
- Kim, G.; Khanal, P.; Kim, J.Y.; Yun, H.J.; Lim, S.C.; Shim, J.H.; Choi, H.S. COT phosphorylates prolyl-isomerase Pin1 to promote tumorigenesis in breast cancer. Mol. Carcinog. 2015, 54, 440–448. [Google Scholar] [CrossRef] [PubMed]
- Nechama, M.; Uchida, T.; Mor Yosef-Levi, I.; Silver, J.; Naveh-Many, T. The peptidyl-prolyl isomerase Pin1 determines parathyroid hormone mRNA levels and stability in rat models of secondary hyperparathyroidism. J. Clin. Investig. 2009, 119, 3102–3114. [Google Scholar] [CrossRef] [PubMed]
- Jeong, H.G.; Pokharel, Y.R.; Lim, S.C.; Hwang, Y.P.; Han, E.H.; Yoon, J.H.; Ahn, S.G.; Lee, K.Y.; Kang, K.W. Novel role of Pin1 induction in type II collagen-mediated rheumatoid arthritis. J. Immunol. 2009, 183, 6689–6697. [Google Scholar] [CrossRef]
- Chiasson, V.L.; Munshi, N.; Chatterjee, P.; Young, K.J.; Mitchell, B.M. Pin1 deficiency causes endothelial dysfunction and hypertension. Hypertension 2011, 58, 431–438. [Google Scholar] [CrossRef] [PubMed]
- Costantino, S.; Paneni, F.; Lüscher, T.F.; Cosentino, F. Pin1 inhibitor Juglone prevents diabetic vascular dysfunction. Int. J. Cardiol. 2016, 203, 702–707. [Google Scholar] [CrossRef]
- Liu, M.; Yu, P.; Jiang, H.; Yang, X.; Zhao, J.; Zou, Y.; Ge, J. The essential role of Pin1 via NF-κB signaling in vascular inflammation and atherosclerosis in ApoE-/- mice. Int. J. Mol. Sci. 2017, 18, 644. [Google Scholar] [CrossRef] [PubMed]
- Lv, L.; Ye, M.; Duan, R.; Yuan, K.; Chen, J.; Liang, W.; Zhou, Z.; Zhang, L. Downregulation of Pin1 in human atherosclerosis and its association with vascular smooth muscle cell senescence. J. Vasc. Surg. 2018, 68, 873–883. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Lin, L.; Xu, C.; Chai, D.; Peng, F.; Lin, J. VDR agonist prevents diabetic endothelial dysfunction through inhibition of prolyl isomerase-1-mediated mitochondrial oxidative stress and inflammation. Oxid. Med. Cell. Longev. 2018, 2018, 1714896. [Google Scholar] [CrossRef] [PubMed]
- Lv, L.; Zhang, J.; Zhang, L.; Xue, G.; Wang, P.; Meng, Q.; Liang, W. Essential role of Pin1 via STAT3 signalling and mitochondria-dependent pathways in restenosis in type 2 diabetes. J. Cell. Mol. Med. 2013, 17, 989–1005. [Google Scholar] [CrossRef]
- Ghosh, A.; Saminathan, H.; Kanthasamy, A.; Anantharam, V.; Jin, H.; Sondarva, G.; Harischandra, D.S.; Qian, Z.; Rana, A.; Kanthasamy, A.G. The peptidyl-prolyl isomerase Pin1 up-regulation and proapoptotic function in dopaminergic neurons: Relevance to the pathogenesis of Parkinson disease. J. Biol. Chem. 2013, 288, 21955–21971. [Google Scholar] [CrossRef]
- Liu, X.; Liang, E.; Song, X.; Du, Z.; Zhang, Y.; Zhao, Y. Inhibition of Pin1 alleviates myocardial fibrosis and dysfunction in STZ-induced diabetic mice. Biochem. Biophys. Res. Commun. 2016, 479, 109–115. [Google Scholar] [CrossRef]
- Wu, X.; Li, M.; Chen, S.Q.; Li, S.; Guo, F. Pin1 facilitates isoproterenol-induced cardiac fibrosis and collagen deposition by promoting oxidative stress and activating the MEK1/2-ERK1/2 signal transduction pathway in rats. Int. J. Mol. Med. 2018, 41, 1573–1583. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Huang, D.; Li, L.L.; Ni, P.; Li, X.X.; Wang, B.; Han, Y.N.; Shao, X.Q.; Zhao, D.; Chu, W.F.; et al. TGF-β1-PML SUMOylation-peptidyl-prolyl cis-trans isomerase NIMA-interacting 1 (Pin1) form a positive feedback loop to regulate cardiac fibrosis. J. Cell. Physiol. 2019, 234, 6263–6273. [Google Scholar] [CrossRef] [PubMed]
- Wahedi, H.M.; Park, Y.U.; Moon, E.Y.; Kim, S.Y. Juglone ameliorates skin wound healing by promoting skin cell migration through Rac1/Cdc42/PAK pathway. Wound Repair Regen. 2016, 24, 786–794. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Zhang, H.; Xu, Y.; Zhang, J.; Zhu, W.; Zhang, Y.; Chen, L.; Hua, W.; Mao, Y. Juglone induces apoptosis of tumor stem-like cells through ROS-p38 pathway in glioblastoma. BMC Neurol. 2017, 17, 70. [Google Scholar] [CrossRef]
- Lu, Z.; Chen, H.; Zheng, X.M.; Chen, M.L. Experimental study on the apoptosis of cervical cancer Hela cells induced by juglone through c-Jun N-terminal kinase/c-Jun pathway. Asian Pac. J. Trop. Med. 2017, 10, 572–575. [Google Scholar] [CrossRef]
- Wang, P.; Gao, C.; Wang, W.; Yao, L.P.; Zhang, J.; Zhang, S.D.; Li, J.; Fang, S.H.; Fu, Y.J. Juglone induces apoptosis and autophagy via modulation of mitogen-activated protein kinase pathways in human hepatocellular carcinoma cells. Food Chem. Toxicol. 2018, 116, 40–50. [Google Scholar] [CrossRef]
- Shults, N.V.; Almansour, F.S.; Rybka, V.; Suzuki, D.I.; Suzuki, Y.J. Ligand-mediated dephosphorylation signaling for MAP kinase. Cell. Signal. 2018, 52, 147–154. [Google Scholar] [CrossRef] [PubMed]
- Pektas, M.B.; Turan, O.; Ozturk Bingol, G.; Sumlu, E.; Sadi, G.; Akar, F. High glucose causes vascular dysfunction through Akt/eNOS pathway: Reciprocal modulation by juglone and resveratrol. Can. J. Physiol. Pharmacol. 2018, 96, 757–764. [Google Scholar] [CrossRef]
- Sajadimajd, S.; Yazdanparast, R. Sensitizing effect of juglone is mediated by down regulation of Notch1 signaling pathway in trastuzumab-resistant SKBR3 cells. Apoptosis 2017, 22, 135–144. [Google Scholar] [CrossRef] [PubMed]
- Hill, K.; Fiorito, S.; Taddeo, V.A.; Schulze, A.; Leonhardt, M.; Epifano, F.; Genovese, S. Plumbagin, juglone, and boropinal as novel TRPA1 agonists. J. Nat. Prod. 2016, 79, 697–703. [Google Scholar] [CrossRef] [PubMed]
- García-Añoveros, J.; Nagata, K. TRPA1. Handb. Exp. Pharmacol. 2007, 179, 347–362. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahmad, T.; Suzuki, Y.J. Juglone in Oxidative Stress and Cell Signaling. Antioxidants 2019, 8, 91. https://doi.org/10.3390/antiox8040091
Ahmad T, Suzuki YJ. Juglone in Oxidative Stress and Cell Signaling. Antioxidants. 2019; 8(4):91. https://doi.org/10.3390/antiox8040091
Chicago/Turabian StyleAhmad, Taseer, and Yuichiro J. Suzuki. 2019. "Juglone in Oxidative Stress and Cell Signaling" Antioxidants 8, no. 4: 91. https://doi.org/10.3390/antiox8040091
APA StyleAhmad, T., & Suzuki, Y. J. (2019). Juglone in Oxidative Stress and Cell Signaling. Antioxidants, 8(4), 91. https://doi.org/10.3390/antiox8040091



