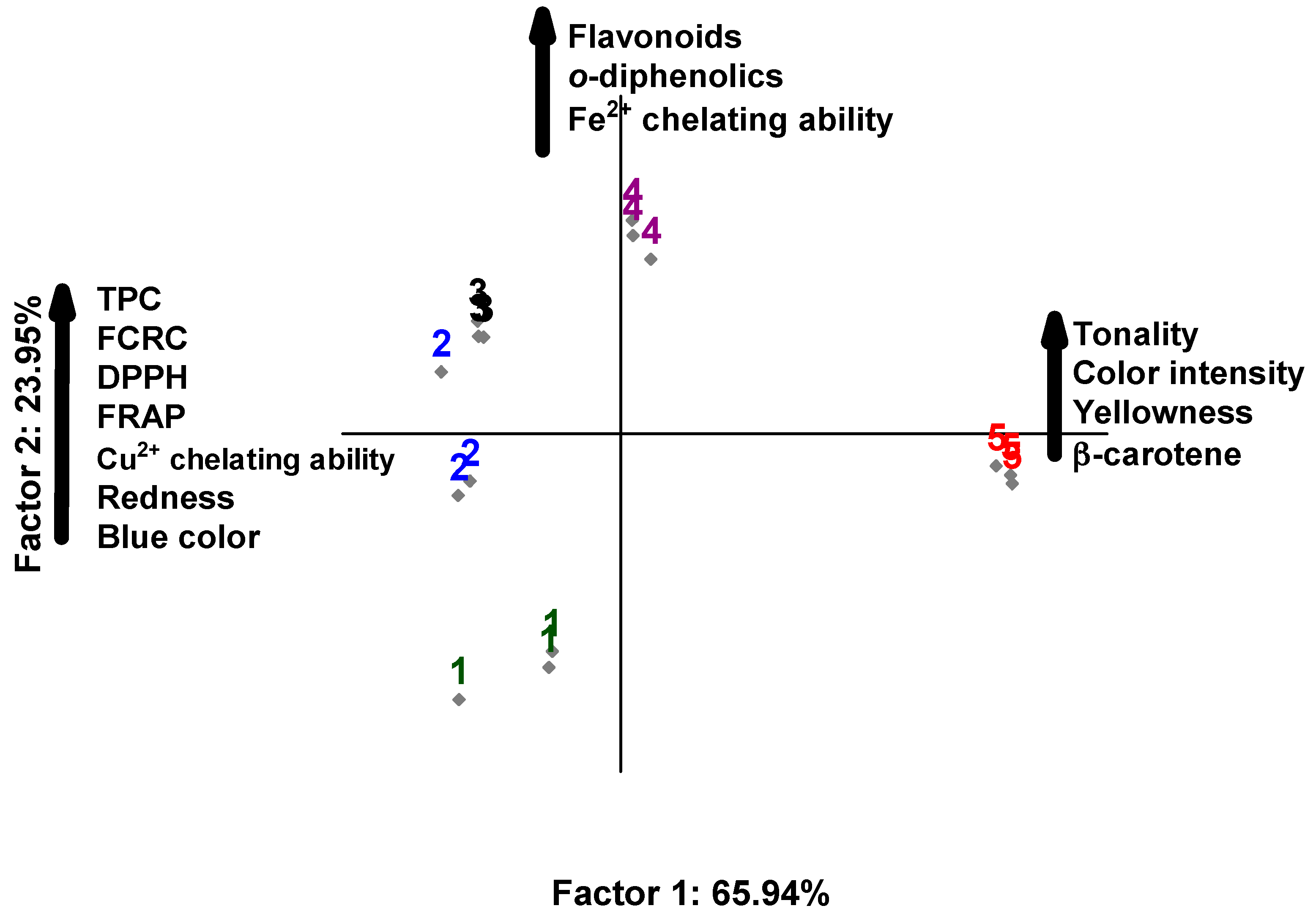
3.1. Chemical Composition, Instrumental Color, and Antioxidant Activity of the Extracts
Table 3 shows the chemical composition and instrumental color of
C. officinalis flower extracts. The β-carotene content ranged from 97 to 637 µg/100 g (
p ≤ 0.05). The highest content of β-carotene was extracted with 100% EtOH, and significant differences were not observed between extracts with 50 and 75% EtOH. Carotenoid contents ranging from 200 to 3510 mg/100 g were quantified in different
C. officinalis flower varieties [
17,
41]. The marigold flowers as well as β-carotene containing α-carotene, γ-carotene, lutein, luteoxanthin, flavoxanthin, rubixanthin, and other carotenoids [
42,
43].
The TPC contents ranged from 123 to 217 mg GAE/100 g (p ≤ 0.05). The highest TPC contents were extracted with 50% H2O:50% EtOH, followed by 75% H2O:25% EtOH and 25% H2O:75% EtOH. The TF contents ranged from 101 to 147 mg QE/100 g (p ≤ 0.05), and the highest content was extracted with 50% H2O:50% EtOH. The ortho-diphenolics content ranged from 331 to 531 mg CAE/100g (p ≤ 0.05). The highest ortho-diphenolics content was extracted with 25% H2O:75% EtOH, and significant differences were not observed between the extracts obtained with 50% H2O:50% EtOH and 100% EtOH.
Ferreira et al. [
15] compared chloroform, ethyl alcohol, methyl alcohol (MeOH), and hydromethanolic (MeOH:H
2O, 70:30 v/v) solvents in the extraction of phenolic compounds from marigold flowers. The authors found that hydromethane was more effective in extracting phenolic compounds because it was highly polar. Pires et al. [
44] quantified in the extracts of marigold petals containing MeOH:H
2O (80:20 v/v) the contents of 1131 mg/100 g of phenolic compounds and 1115 mg/100 g of flavonoids, and in the aqueous extract the contents of 747 mg/100 g and 737 mg/100 g, respectively. The literature shows that methyl alcohol and hydromethanolic solutions extract higher levels of phenolic compounds. However, in this research, it was chosen to use ethyl alcohol because of its low toxicity. In addition, the contents of bioactive compounds (carotenoids and phenolic compounds) may vary due to different
C. officinalis flower varieties and cultivation sites [
41,
45].
The color indices of the extracts were influenced by the combination of the solvents H
2O and EtOH in which the color intensity varied from 1.1 to 2.1 u. a. and the hue ranged from 4.9 to 12.6 u. a. (
p ≤ 0.05). The highest intensity of color and hue was verified in the extract obtained with 100% EtOH which presented the highest content of β-carotene (637 µg/100 g). According to Khalid et al. [
45], carotenoids are mainly responsible for the yellow-orange coloration of
C. officinalis inflorescences. In the correlation analysis, color intensity was significantly correlated (
p ≤ 0.05) with β-carotene (
r = 0.8959),
ortho-diphenolics (
r = 0.6150), and TPC (
r = −0.5393). The hue showed a significant correlation (
p ≤ 0.05) with β-carotene (
r = 0.7874) and TPC (
r = −0.8702).
The yellow, red, and blue pigments of the extracts ranged from 79 to 90%, 7 to 16%, and 3 to 6% (
p ≤ 0.05), respectively. The highest percentage of yellow pigments (90%) was obtained in the extract with 100% EtOH. Higher percentages of red (16%) and blue (6%) pigments were obtained in extracts with 75% H
2O:25% EtOH and 100% H
2O, respectively. The yellow pigments showed significant correlation (
p ≤ 0.05) with β-carotene (
r = 0.7765) and TPC (
r = −0.7516). The red and blue pigments correlated significantly (
p ≤ 0.05) with β-carotene,
r = −0.7619 and
r = −0.6724, respectively. The blue pigments also correlated with
ortho-diphenolic (
r = −0.5387), and the red pigment with TPC (
r = 0.7909). Olennikov et al. [
14] quantified anthocyanins in some
C. officinalis varieties with intense raspberry-red pigmentation in the central part of the flower.
The chemical antioxidant activity of the extracts was influenced by the different proportions of the solvents H
2O and EtOH (
p ≤ 0.05) (
Table 4). The extract obtained with 75% H
2O:25% EtOH showed higher antioxidant activity through electron transfer evaluated by DPPH, FRAP, and RCFC. In the analysis by FRAP and RCFC, no significant differences were observed between the extracts with 75% H
2O:25% EtOH and 50% H
2O:50% EtOH. Additionally, the metal chelating activity of the extracts was evaluated by inhibiting the formation of the Cu
2+-pyrocatechol violet complex and the Fe
2+-ferrozine complex. The extract with 50% H
2O:50% EtOH showed the highest chelating activity with inhibition of 60% and 53% of the formation of the Cu
2+-pyrocatechol violet complex and Fe
2+-ferrozine complex, respectively.
The antioxidant activity of the extract with 50% H2O:50% EtOH is associated with higher levels of TPC (217 mg GAE/100 g) and TF (QE 147 mg/100 g). C. officinalis flower extracts showed significant correlation (p ≤ 0.05) of antioxidant activity with β-carotene (rDPPH = −0.7911; rFRAP = −0.6637; rCCA = −0.6479), total phenolic content (rDPPH = 0.9109; rFRAP = 0.9677; rRCFC = 0.9663; rCCA = 0.9279), flavonoids (rFRAP = 0.5532; rRCFC = 0.7154; rCCA = 0.5341; rFCA = 0.6371), and with ortho-diphenolics (rFCA = 0.5479).
3.3. Selected Extract: Individual Phenolic Composition and in Vitro Functional Properties
The individual phenolic composition of the hydroalcoholic extract (50:50 v/v) of
C. officinalis flowers were determined by HPLC-DAD (
Figure S1). The phenolic acids quantified were
p-coumaric (5.8 mg/100 g), caffeic (9.2 mg/100 g), ferulic (18.3 mg/100 g), and ellagic acid, 3.7 mg/100 g. The quantified flavonoids were procyanidin A2 (42.5 mg/100 g) and quercetin-3-rutinoside (46.1 mg/100 g). Olennikov et al. [
17] quantified caffeic acid (92 mg/100 g) and quercetin-3-rutinoside (226 mg/100 g) in the hydroalcoholic extract, 60% EtOH, of
C. officinalis flowers var. Greenheart Orange. Pires et al. [
44] quantified caffeic acid (1 mg/100 g) and quercetin-3-rutinoside (30 mg/100 g) in the hydromethanolic extract (MeOH:H
2O, 80:20 v/v) of
C. officinalis petals. Miguel et al. [
29] quantified 22 mg/100 g quercetin-3-rutinoside in the hydromethanolic extract (MeOH:H
2O, 80:20 v/v) of
C. officinalis flowers.
Nineteen phenolic compounds were tentatively identified in the hydroalcoholic extract (50:50 v/v) of
C. officinalis flowers by ESI-MS/MS ([M-H]
-): caffeic acid hexoside (
m/z 341), isomeric form of hydroxyferulic acid hexoside (
m/z 371), caffeoylshikimic acid (
m/z 335), 5-
O-feruloylquinic acid (
m/z 367), quercetin derivative (
m/z 505;
m/z 463; and
m/z 609), quercetin-3-
O-malonylhexoside (
m/z 549), apigenin derivative (
m/z 563), quercetin dihexoside (
m/z 625), ligstroside hexoside (
m/z 685), isorhamnetin derivative (
m/z 477;
m/z 519; and
m/z 623), kaempferol-rhammosyrutinoside (
m/z 739), quercetin-3-
o-rhamnosylrutinoside (
m/z 755), calenduloside G (
m/z 793), calendasaponin B (
m/z 971), and calendasaponin A (
m/z 1117). The obtained mass spectra are shown in
Figure S2. The compounds tentatively identified in this study corroborate data obtained by Miguel et al. [
29] and Faustino et al. [
30].
The antimicrobial activity of the lyophilized marigold extract is presented in
Table 5. The extract showed the inhibitory effect of the growth of
L. monocytogenes,
P. aeruginosa,
S. Typhimurium,
S. Enteritidis,
B. cereus,
E. coli, and
S. aureus (0.27 cm). These results corroborate Efstratiou et al. [
46], who found that the ethanolic extract of
C. officinalis petals inhibited the growth of
P. aeruginosa (0.7 cm),
B. cereus (1 cm),
E. coli (0.3 cm), and
S. aureus (2.2 cm). The hydromethanolic extract (MeOH:H
2O, 80:20 v/v) and the infusion of petals
C. officinalis demonstrated inhibitory effect against Gram-positive and Gram-negative bacteria [
44]. According to Faustino et al. [
30] methanolic extract of some
C. officinalis varieties showed low antibacterial activity due to the lower content of antibacterial phenolic compounds such as caffeic acid.
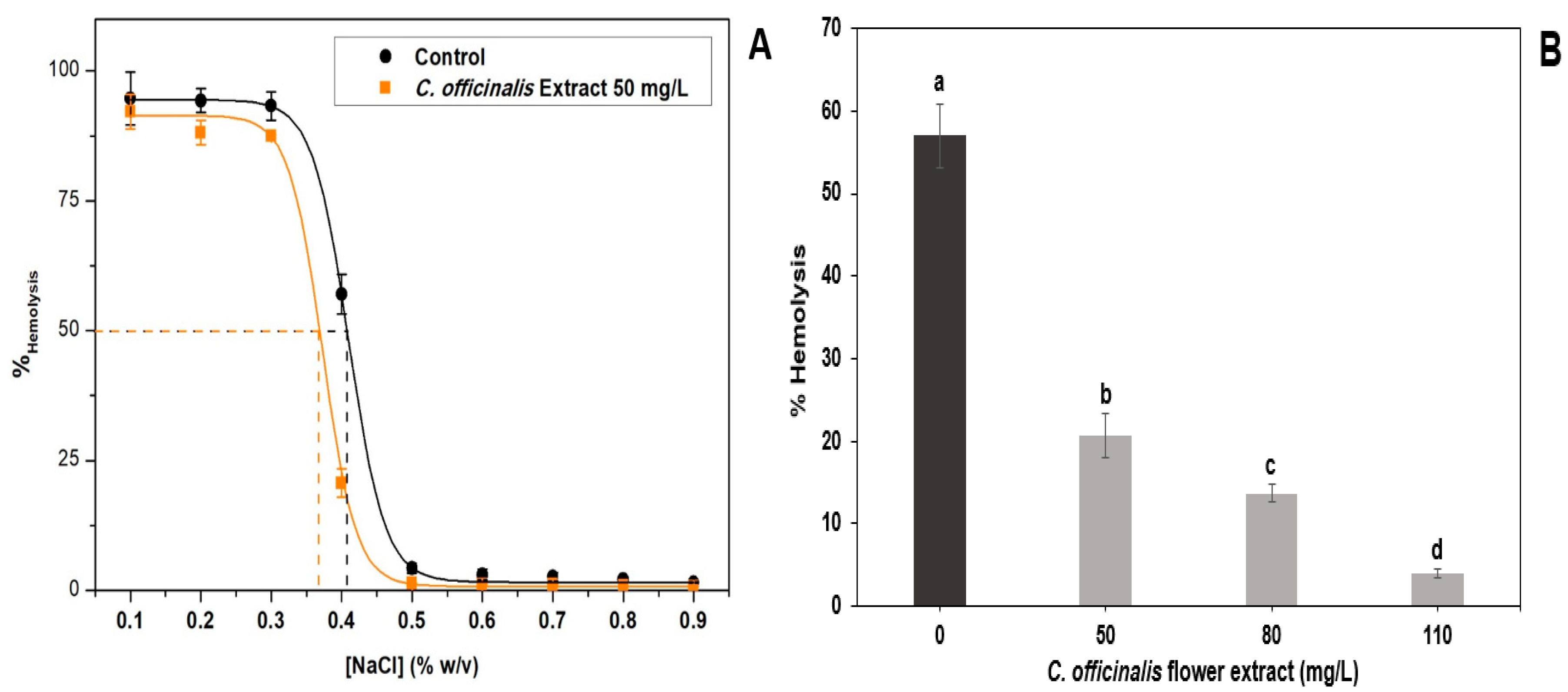
The antihemolytic effect of LME analyzed under hypotonic and isotonic conditions is shown in
Figure 2.
Figure 2A shows that using the 50 mg/L of the LME, it was possible to calculate the H
50, being the NaCl concentration where 50% of the erythrocytes is present in the sample are lysed. The lower this value, the more efficient the extract will be in inhibiting hemolysis caused by low extracellular osmotic pressure. The control, in the absence of the extract, had an H
50 value of 0.413%, while the extract had a lower H
50 value (0.356%). These results suggest that the bioactive compounds present in LME may interact with erythrocyte membrane phospholipids in order to reduce the flow parameter of the lipid bilayer [
47], which increases the resistance of cells to hypotonic hemolysis [
48].
Figure 2B shows that the hemolysis rate is reduced, 0.4% NaCl, with increasing LME concentration (50–110 mg/L). The
C. officinalis flowers are rich in saponins, compounds that have hemolytic activity by solubilizing membranes [
48]. The results showed that the extract showed antihemolytic activity, indicating the absence or low content of saponins in
C. officinalis flowers. The antihemolytic activity of
C. officinalis extracts had already been tested in the literature under oxidative and thermal stress conditions and, in both cases, the hydroalcoholic extract (EtOH:H
2O, 80:20 v/v) was efficient in inhibiting hemolysis [
49].
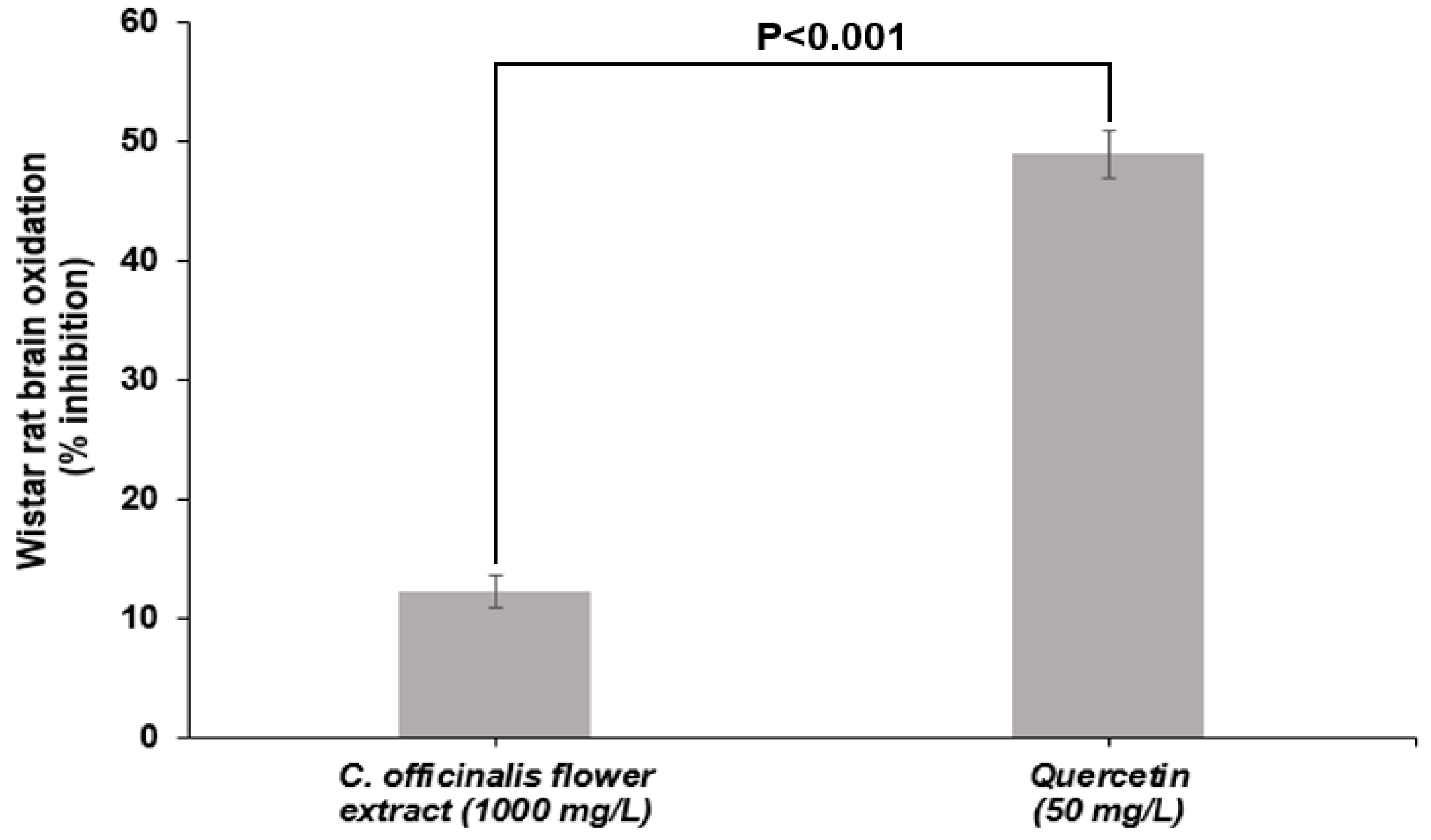
LME showed 12% inhibition of lipid oxidation of Wistar rat brains at a concentration of 1000 mg/L, statistically differing (
p < 0.001) from quercetin inhibition capacity (50 mg/L) (
Figure 3). The superiority of quercetin was already expected since it is a standard of high purity and with a recognized ability to inhibit lipid oxidation. However, the inhibitory capacity presented by the LME is an essential feature because it shows that it is active in biological media inhibiting the oxidation of brain tissue lipids. This test shows the ability of the compounds present in the extract to transfer H
+ atom to free radicals generated by lipid-induced oxidation [
50]. The ability of
C. officinalis extracts to inhibit lipid oxidation was also observed in an in vivo study by Tanideh et al. [
51]. The same authors found that the hydroalcoholic extract (EtOH:H
2O, 80:20 v/v) of
C. officinalis flowers, administered orally to male Sprague–Dawley rats, showed decreased lipid oxidation.
LME, 20 mg/mL, inhibited 27% of α-amylase enzymatic activity, indicating a dose-dependent effect with increasing extract concentration (1–15 mg/mL) (
Figure 4A). The LME, 500 µg/mL, inhibited 43% of α-glucosidase enzymatic activity. The concentrations of 50 to 100 µg/mL and 200 to 300 µg/mL of LME showed no significant differences in the inhibitory activity of α-glucosidase (
Figure 4). The extract from the leaves of
C. officinalis was evaluated in the inhibitory activity of the α-amylase enzyme as described by Olennikov et al. [
52]. The authors found that 38.02 µg/mL of hydroalcoholic extract (EtOH:H
2O, 60:40 v/v) of leaves inhibited 50% of α-amylase activity. In another study, hydroalcoholic extract (50:50 v/v) from
C. officinalis leaves reduced serum glucose levels in diabetic rats [
53].
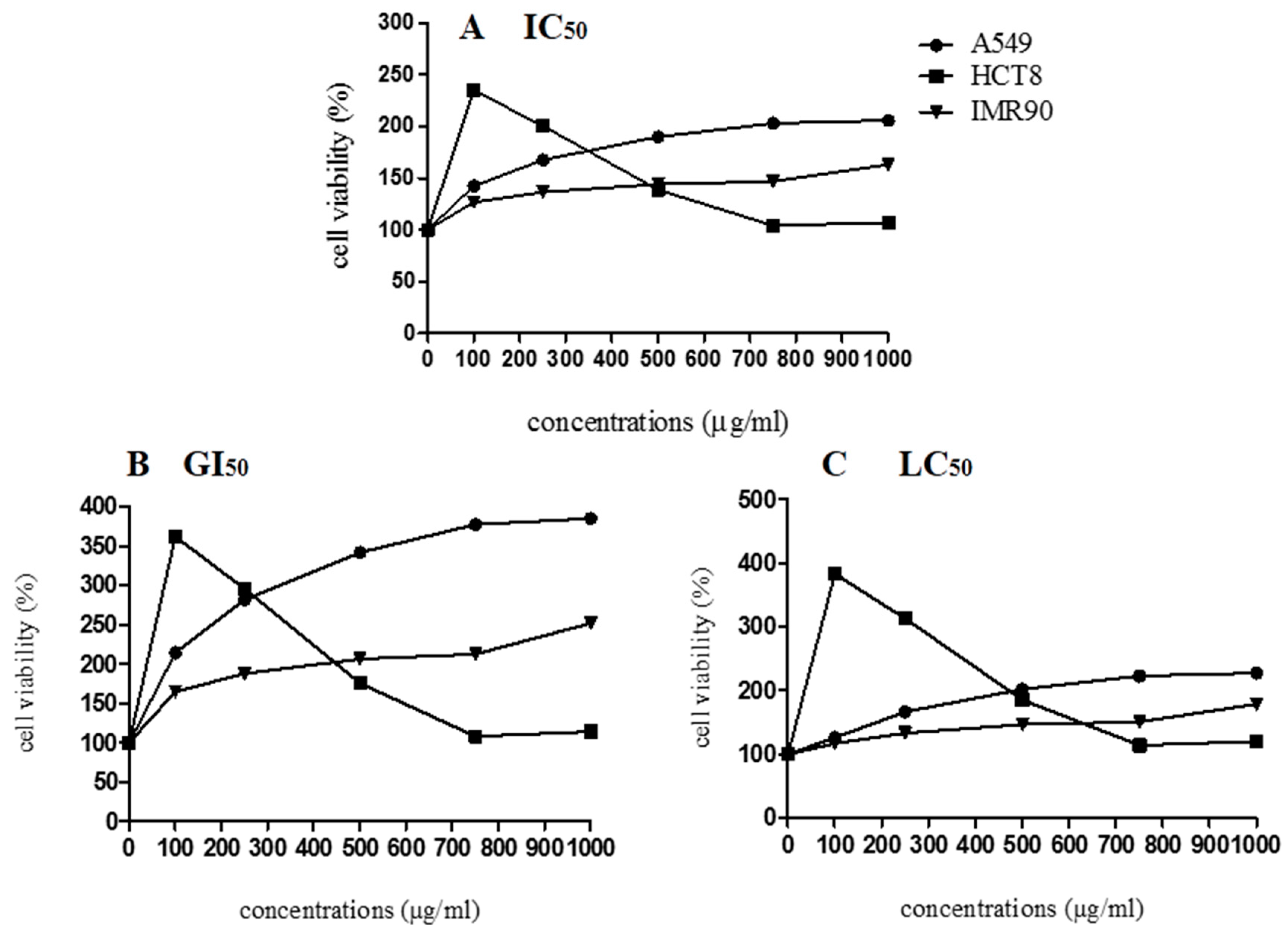
According to the cytotoxicity results (
Figure 5), the lyophilized marigold extract enhanced the cell viability parameter (IC
50), stimulated the growth index (GI
50), and it was not able to cause cell death (LC
50) in both cancerous (HCT8, A549) and noncancerous (IMR90) cell lines, suggesting no apparent cytotoxicity. Similarly, Pires et al. [
44] also observed that not all the
C. officinalis samples inhibited the growth of MCF-7, NCI-H460, HeLa, and HepG2 tumor cells. Herein, considering the cell proliferation behavior, exogenous antioxidants (i.e., flavonoids and carotenoids) may exert cytotoxic effects in several cancerous cell lines, but whether an antioxidant supplement would be helpful, harmful, or neutral depends on the specific antioxidant, its dose, the chemotherapy drugs being used, and the type and stage of cancer [
54]. Regarding the growth of A549, IMR90, and HCT8 cells, we hypothesize that the bioactive compounds in
C. officinalis may favor mitogenic mechanisms, which involves complex pathways that can be related to overexpression of essential cell cycle regulatory proteins, such as cyclin-dependent kinases,
d-type cyclins, polo-like kinases, and aurora kinases, which stimulate the cell cycle progression in the M/G1/S phases [
55]. Bearing these concerns, more research is needed to explain the possible mechanisms involved in
C. officinalis-induced cell growth.
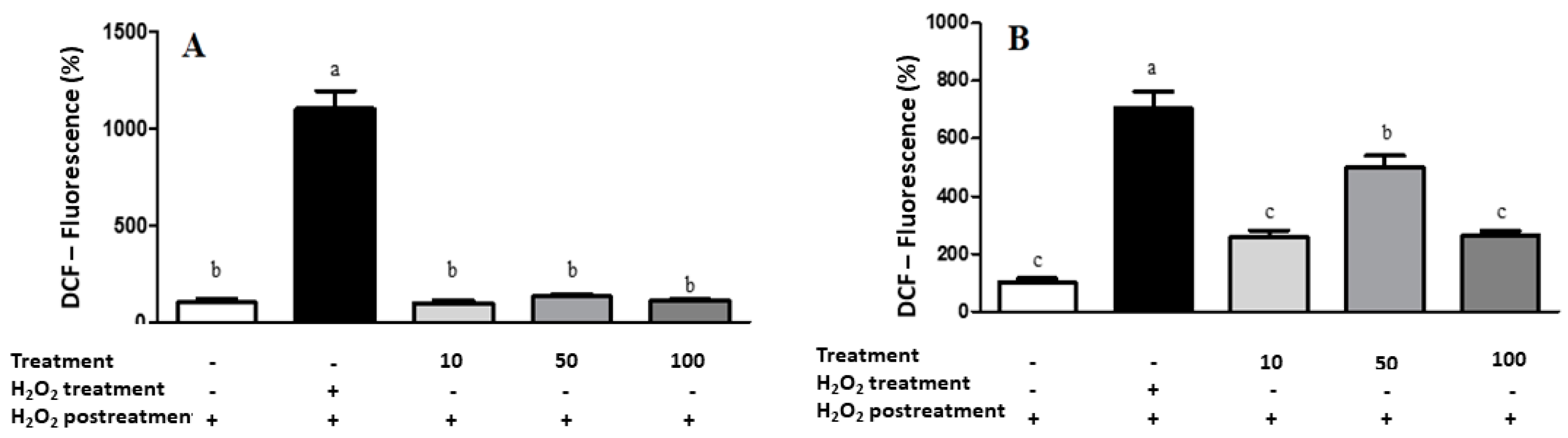
Regarding the cell-based antioxidant assay, LME did not induce ROS generation (
Figure 6) at the higher tested concentrations in both cancerous (A549) and noncancerous (IMR90) cells. Palozza et al. [
56] observed that carotenoids in the
C. officinalis extract have already been associated with antiproliferative activity and cytotoxicity through their prooxidant ability. Thus, although the
C. officinalis extract presented an interesting chemical profile, antioxidant, antimicrobial, and anti-hemolytic capacities, it did not exhibit cytotoxic effects against the cancerous cell lines.
3.4. Characterization of the Organic Yogurt with LME
The TPC and antioxidant activity (DPPH and total reducing capacity (TRC)) of organic yogurts manufactured with different concentrations of LME are shown in
Table 6. Increasing LME concentration, 0.25 to 1.5 g/100 g, increased the TPC and
in vitro antioxidant activity of organic yogurts, and thus indicating a dose-dependence effect. These results corroborate with Demirkol et al. [
57] who determined an increase in TPC (20 to 52 mg/100 g yogurt) and DPPH value (263 to 1100 mg/mL for 50% inhibition of the radical) in yogurt added with grape pomace (1 to 5 g/100 g milk). Abdel-Hamid et al. [
58] measured an increase in antioxidant activity using the DPPH (43 to 78% radical inhibition) and ABTS (54 to 91% radical inhibition) assays in probiotic yogurt supplemented with 0.5 to 2% of
Siraitia grosvenorii fruit extract.
The proximal composition, physicochemical characterization, color, and instrumental texture of organic yogurt with LME (0.25 g LME/100 g) and without extract (0 g LME/100 g, control) is presented in
Table 7. The protein, lipid, ash content, titratable acidity, hardness, and consistency of organic yogurt manufactured with LME did not show statistically significant differences with the control. The organic yogurts with and without LME show statistically significant differences (
p ≤ 0.05) in carbohydrate content, moisture, energy, and pH. These results are similar to those found by Karnopp et al. [
2] in organic yogurt manufactured with Bordeaux grape skin flour. The same authors found the following properties in the yogurt: carbohydrate (9.35 g/100 g), protein (3.80 g/100 g), lipid (6.23 g/100 g), ash (0.81 g/100 g), moisture (76.49 g/100 g) contents, pH (4.19), titratable acidity (0.61 g lactic acid/100 g), hardness (20.88 g), and consistency (493.13 g/s). The incorporation of the LME in the organic yogurt caused a color change (L*, a*, and b*) compared to the control (
p < 0.001) (
Table 7). Demirkol et al. [
57] also found a reduction in the brightness (L*) and increased redness (a*) in yogurt manufactured with grape pomace flour. According to Pires et al. [
6], edible flower petal extracts have great potential for use as natural colorants and are an excellent source of bioactive compounds to develop innovative products with novel sensory characteristics and antioxidant activity.
The organic yogurt with 0.25 g LME/100 g presented a general sensory acceptance rate of 80.4%. The average scores given for odor (7.6 ± 1.5), taste (6.4 ± 2.2), consistency (7.2 ± 2.0), color (7.7 ± 1.5), and overall impression (7.2 ± 1.7) were ranked between “slightly liked” and “moderately liked “. The bitter residual taste reported by some assessors may have influenced the taste attribute (average grade <7). All evaluators (100%, n = 76) consider yogurt a healthy product. Regarding the commercial value of the yogurt, 75% of assessors would pay between R$ 2.00 and R$ 3.00 for yogurt rich in natural antioxidant compounds (100 g) compared to yogurt without natural antioxidants, and 75% would pay between R$ 2.00 and R$ 3.00 for organic yogurt (100 g) compared to conventional yogurt.