Oxidative Stress and Nutraceuticals in the Modulation of the Immune Function: Current Knowledge in Animals of Veterinary Interest
Abstract
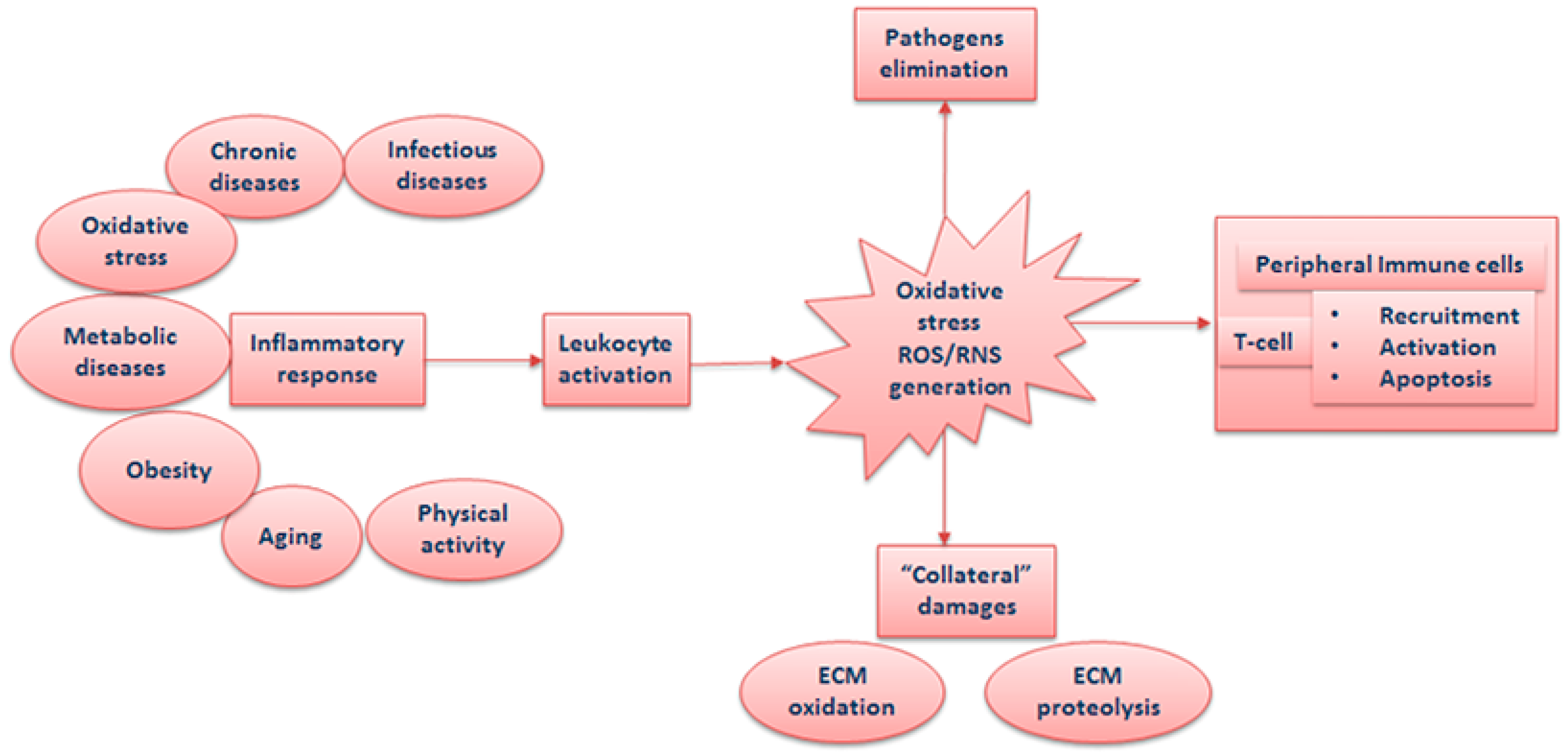
1. Inflammation, Leukocyte Recruitment, and Activation
1.1. Inflammation and Oxidative Stress at the Site of Infection
1.2. Inflammation and Systemic Oxidative Stress
1.3. Adipose Tissue and Oxidative Stress
1.4. Inflammaging
2. Functional Compounds in Animal Nutrition
2.1. Anti-Oxidant Compounds
2.2. Anti-Inflammatory Compounds
2.3. Anti-Inflammatory Compounds in Obesity
3. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| PAMPs | Pathogen-associated molecular patterns |
| LPS | Lipopolysaccharide |
| PRRs | Pattern recognition receptors |
| TLRs | Toll-like receptors |
| DAMPs | Damage-associated molecular patterns |
| AGEs | Advanced glycation end products |
| HDL | High-density lipoproteins |
| LDL | Low-density lipoproteins |
| RAGE | Receptors of AGE |
| ROS | Reactive oxygen species |
| RNS | Reactive nitrogen species |
| NOX | NADPH oxidase |
| MPO | Myeloperoxidase |
| HOCl | Hypochlorous acid |
| OS | Oxidative stress |
| DC | Dendritic cells |
| Treg | Regulatory T cells |
| NFκB | Nuclear factor k B |
| H2O2 | Hydrogen hydroperoxide |
| Se | Selenium |
| EC | Endothelial cells |
| TNFα | Tumor necrosis factor α |
| ICAM-1 | Intercellular adhesion molecule 1 |
| AOPP | Advanced oxidized protein product |
| IBD | Inflammatory bowel disease |
| CVL | Canine visceral leishmaniasis |
| HO-1 | Heme oxygenase-1 |
| MDA | Plasma malondialdehyde |
| GPx | Glutathione peroxidase |
| SOD | Superoxide dismutase |
| CADESI | Canine Atopic Dermatitis Extent and Severity Index |
| FR | Free-radicals |
| SFRP5 | Secreted frizzled-related protein 5 |
| CAT | Catalase |
| FFA | Free-fatty acids |
| ROO• | Peroxyl radicals |
| ROOH | Hydroperoxide radicals |
| PhO• | Phenoxyl radical |
| PTGS2 | Prostaglandin-endoperoxide synthase 2 |
| Nrf2 | NF-E2-related factor 2 |
| KEAP1 | Kelch like ECH associated protein 1 |
| WAT | White adipose tissue |
| BAT | Brown adipose tissue |
| DHA | Docosahexaenoic acid |
| UCP1 | Uncoupling protein 1 |
References
- Tsoupras, A.; Lordan, R.; Zabetakis, I. Inflammation, not cholesterol, is a cause of chronic disease. Nutrients 2018, 10, 604. [Google Scholar] [CrossRef] [PubMed]
- De Lorenzo, G.; Ferrari, S.; Cervone, F.; Okun, E. Extracellular DAMPs in plants and mammals: Immunity, tissue damage and repair. Trends Immunol. 2018, 39, 937–950. [Google Scholar] [CrossRef] [PubMed]
- Medzhitov, R. Origin and physiological roles of inflammation. Nature 2008, 454, 428–435. [Google Scholar] [CrossRef]
- Medzhitov, R.; Janeway, C.A., Jr. Decoding the patterns of self and nonself by the innate immune system. Science 2002, 296, 298–300. [Google Scholar] [CrossRef] [PubMed]
- Fishman, S.L.; Sonmez, H.; Basman, C.; Singh, V.; Poretsky, L. The role of advanced glycation end-products in the development of coronary artery disease in patients with and without diabetes mellitus: A review. Mol. Med. 2018, 24, 59. [Google Scholar] [CrossRef]
- Comazzi, S.; Bertazzolo, W.; Bonfanti, U.; Spagnolo, V.; Sartorelli, P. Advanced glycation end products and sorbitol in blood from differently compensated diabetic dogs. Res. Vet. Sci. 2008, 84, 341–346. [Google Scholar] [CrossRef]
- Gkogkolou, P.; Böhm, M. Advanced glycation end products: Key players in skin aging? Dermato-endocrinology 2012, 4, 259–270. [Google Scholar] [CrossRef]
- Van Dyke, T.E.; Kornman, K.S. Inflammation and factors that may regulate inflammatory response. J. Periodontol. 2008, 79, 1503–1507. [Google Scholar] [CrossRef]
- Peter Libby, M.D. Inflammatory mechanisms: The molecular basis of inflammation and disease. Nutr. Rev. 2007, 65, 140–146. [Google Scholar] [CrossRef]
- Christoffersson, G.; Phillipson, M. The neutrophil: One cell on many missions or many cells with different agendas? Cell Tissue Res. 2018, 371, 415–423. [Google Scholar] [CrossRef]
- Valacchi, G.; Virgili, F.; Cervellati, C.; Pecorelli, A. OxInflammation: From subclinical condition to pathological biomarker. Front. Physiol. 2018, 9, 858. [Google Scholar] [CrossRef] [PubMed]
- Azcutia, V.; Parkos, C.A.; Brazil, J.C. Role of negative regulation of immune signaling pathways in neutrophil function. J. Leukoc. Biol. 2017, 103, 1029–1041. [Google Scholar] [CrossRef] [PubMed]
- Winterbourn, C.C.; Kettle, A.J.; Hampton, M.B. Reactive oxygen species and neutrophil function. Annu. Rev. Biochem. 2016, 85, 765–792. [Google Scholar] [CrossRef] [PubMed]
- Gostner, J.M.; Becker, K.; Fuchs, D.; Sucher, R. Redox regulation of the immune response. Redox Rep. 2013, 18, 88–94. [Google Scholar] [CrossRef] [PubMed]
- Odobasic, D.; Kitching, A.R.; Yang, Y.; O’Sullivan, K.M.; Muljadi, R.C.M.; Edgtton, K.L.; Tan, D.S.Y.; Summers, S.A.; Morand, E.F.; Holdsworth, S.R. Neutrophil myeloperoxidase regulates T-cell-driven tissue inflammation in mice by inhibiting dendritic cell function. Blood 2013, 121, 4195–4204. [Google Scholar] [CrossRef] [PubMed]
- Belikov, A.V.; Schraven, B.; Simeoni, L. T cells and reactive oxygen species. J. Biomed. Sci. 2015, 22, 85. [Google Scholar] [CrossRef] [PubMed]
- Lorne, E.; Zmijewski, J.W.; Zhao, X.; Liu, G.; Tsuruta, Y.; Park, Y.J.; Dupont, H.; Abraham, E. Role of extracellular superoxide in neutrophil activation: Interactions between xanthine oxidase and TLR4 induce proinflammatory cytokine production. Am. J. Physiol. Cell Physiol. 2008, 294, C985–C993. [Google Scholar] [CrossRef]
- Bordignon, M.; Da Dalt, L.; Marinelli, L.; Gabai, G. Advanced oxidation protein products are generated by bovine neutrophils and inhibit free radical production in vitro. Vet. J. 2014, 199, 162–168. [Google Scholar] [CrossRef]
- Yan, Z.; Garg, S.K.; Kipnis, J.; Banerjee, R. Extracellular redox modulation by regulatory T cells. Nat. Chem. Biol. 2009, 5, 721–723. [Google Scholar] [CrossRef]
- Bassel, L.L.; Caswell, J.L. Bovine neutrophils in health and disease. Cell Tissue Res. 2018, 371, 617–637. [Google Scholar] [CrossRef]
- Goff, J.P.; Horst, R.L. Physiological changes at parturition and their relationship to metabolic disorders. J. Dairy Sci. 1997, 80, 1260–1268. [Google Scholar] [CrossRef]
- Aleri, J.W.; Hine, B.C.; Pyman, M.F.; Mansell, P.D.; Wales, W.J.; Mallard, B.; Fisher, A.D. Periparturient immunosuppression and strategies to improve dairy cow health during the periparturient period. Res. Vet. Sci. 2016, 108, 8–17. [Google Scholar] [CrossRef] [PubMed]
- Schwarm, A.; Viergutz, T.; Kuhla, B.; Hammon, H.M.M.; Schweigel-Röntgen, M. Fuel feeds function: Energy balance and bovine peripheral blood mononuclear cell activation. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2013, 164, 101–110. [Google Scholar] [CrossRef] [PubMed]
- Sordillo, L.M.; Aitken, S.L. Impact of oxidative stress on the health and immune function of dairy cattle. Vet. Immunol. Immunopathol. 2009, 128, 104–109. [Google Scholar] [CrossRef] [PubMed]
- Vieira-Neto, A.; Lima, I.R.P.; Lopes, F., Jr.; Lopera, C.; Zimpel, R.; Sinedino, L.D.P.; Jeong, K.C.; Galvão, K.; Thatcher, W.W.; Nelson, C.D.; et al. Use of calcitriol to maintain postpartum blood calcium and improve immune function in dairy cows. J. Dairy Sci. 2017, 100, 5805–5823. [Google Scholar] [CrossRef] [PubMed]
- Maddox, J.F.; Aherne, K.M.; Reddy, C.C.; Sordillo, L.M. Increased neutrophil adherence and adhesion molecule mRNA expression in endothelial cells during selenium deficiency. J. Leukoc. Biol. 1999, 65, 658–664. [Google Scholar] [CrossRef] [PubMed]
- Carneiro, L.C.; Cronin, J.G.; Sheldon, I.M. Mechanisms linking bacterial infections of the bovine endometrium to disease and infertility. Reprod. Biol. 2016, 16, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Celi, P.; Merlo, M.; Da Dalt, L.; Stefani, A.; Barbato, O.; Gabai, G. Relationship between late embryonic mortality and the increase in plasma advanced oxidised protein products (AOPP) in dairy cows. Reprod. Fertil. Dev. 2011, 23, 527–533. [Google Scholar] [CrossRef]
- Celi, P.; Merlo, M.; Barbato, O.; Gabai, G. Relationship between oxidative stress and the success of artificial insemination in dairy cows in a pasture-based system. Vet. J. 2012, 193, 498–502. [Google Scholar] [CrossRef]
- Petzl, W.; Zerbe, H.; Günther, J.; Seyfert, H.M.; Hussen, J.; Schuberth, H.J. Pathogen-specific responses in the bovine udder. Models and immunoprophylactic concepts. Res. Vet. Sci. 2018, 116, 55–61. [Google Scholar] [CrossRef]
- Lombardo, F. Relationship between Leukocyte Subpopulations and Oxidative Stress Markers in Bovine Milk. Veterinary Medicine. Degree Thesis, University of Padua, Padua, Italy, 15 December 2017. [Google Scholar]
- Allenspach, K.; Wieland, B.; Gröne, A.; Gaschen, F. Chronic enteropathies in dogs: Evaluation of risk factors for negative outcome. J. Vet. Intern. Med. 2007, 21, 700–708. [Google Scholar] [CrossRef] [PubMed]
- Simpson, K.W.; Jergens, A.E. Pitfalls and progress in the diagnosis and management of canine inflammatory bowel disease. Vet. Clin. N. Am. Small Anim. Pract. 2011, 41, 381–398. [Google Scholar] [CrossRef] [PubMed]
- Jergens, A.E.; Schreiner, C.A.; Frank, D.E.; Niyo, Y.; Ahrens, F.E.; Eckersall, P.D.; Benson, T.J.; Evans, R. A scoring index for disease activity in canine inflammatory bowel disease. J. Vet. Intern. Med. 2003, 17, 291–297. [Google Scholar] [CrossRef] [PubMed]
- Allenspach, K.; Culverwell, C.; Chan, D. Long-term outcome in dogs with chronic enteropathies: 203 cases. Vet. Rec. 2016, 178, 368. [Google Scholar] [CrossRef] [PubMed]
- Rubio, C.P.; Martínez-Subiela, S.; Hernández-Ruiz, J.; Tvarijonaviciute, A.; Cerón, J.J.; Allenspach, K. Serum biomarkers of oxidative stress in dogs with idiopathic inflammatory bowel disease. Vet. J. 2017, 221, 56–61. [Google Scholar] [CrossRef] [PubMed]
- Lykkesfeldt, J.; Svendsen, O. Oxidants and antioxidants in disease: Oxidative stress in farm animals. Vet. J. 2007, 173, 502–511. [Google Scholar] [CrossRef] [PubMed]
- Celi, P.; Gabai, G. Oxidant/antioxidant balance in animal nutrition and health: The role of protein oxidation. Front. Vet. Sci. 2015, 2, 48. [Google Scholar] [CrossRef] [PubMed]
- Mavangira, V.; Sordillo, L.M. Role of lipid mediators in the regulation of oxidative stress and inflammatory responses in dairy cattle. Res. Vet. Sci. 2018, 116, 4–14. [Google Scholar] [CrossRef]
- Grundy, S.M. What is the contribution of obesity to the metabolic syndrome? Endocrinol. Metab. Clin. N. Am. 2004, 33, 267–282. [Google Scholar] [CrossRef]
- Keaney, J.F.; Larson, M.G.; Vasan, R.S.; Wilson, P.W.F.; Lipinska, I.; Corey, D.; Massaro, J.M.; Sutherland, P.; Vita, J.A.; Benjamin, E.J. Obesity and systemic oxidative stress: Clinical correlates of oxidative stress in the Framingham study. Arterioscler. Thromb. Vasc. Biol. 2003, 23, 434–439. [Google Scholar] [CrossRef]
- Bondia-Pons, I.; Ryan, L.; Martinez, J.A. Oxidative stress and inflammation interactions in human obesity. J. Physiol. Biochem. 2012, 68, 701–711. [Google Scholar] [CrossRef] [PubMed]
- Furukawa, S.; Fujita, T.; Shimabukuro, M.; Iwaki, M.; Yamada, Y.; Nakajima, Y.; Nakayama, O.; Makishima, M.; Matsuda, M.; Shimomura, I. Increased oxidative stress in obesity and its impact on metabolic syndrome. J. Clin. Investig. 2004, 114, 1752–1761. [Google Scholar] [CrossRef] [PubMed]
- Marseglia, L.; Manti, S.; D’Angelo, G.; Nicotera, A.; Parisi, E.; Di Rosa, G.; Gitto, E.; Arrigo, T. Oxidative stress in obesity: A critical component in human diseases. Int. J. Mol. Sci. 2015, 16, 378–400. [Google Scholar] [CrossRef] [PubMed]
- Tenhunen, R.; Marver, H.S.; Schmid, R. The enzymatic conversion of hemoglobin to bilirubin. Trans. Assoc. Am. Phys. 1969, 82, 363–371. [Google Scholar] [PubMed]
- Almeida, B.F.; Silva, K.L.; Chiku, V.M.; Leal, A.A.; Venturin, G.L.; Narciso, L.G.; Fink, M.F.; Eugênio, F.R.; Santos, P.S.; Ciarlini, P.C.; et al. The effects of increased heme oxygenase-1 on the lymphoproliferative response in dogs with visceral leishmaniasis. Immunobiology 2017, 222, 693–703. [Google Scholar] [CrossRef] [PubMed]
- Kapun, A.P.; Salobir, J.; Levart, A.; Kotnik, T.; Svete, A.N. Oxidative stress markers in canine atopic dermatitis. Res. Vet. Sci. 2012, 92, 469–470. [Google Scholar] [CrossRef] [PubMed]
- Alberti, K.G.M.M.; Zimmet, P.Z. Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: Diagnosis and classification of diabetes mellitus. Provisional report of a WHO Consultation. Diabet. Med. 1998, 15, 539–553. [Google Scholar] [CrossRef]
- Krzystek-Korpacka, M.; Patryn, E.; Boehm, D.; Berdowska, I.; Zielinski, B.; Noczynska, A. Advanced oxidation protein products (AOPPs) in juvenile overweight and obesity prior to and following weight reduction. Clin. Biochem. 2008, 41, 943–949. [Google Scholar] [CrossRef]
- Fonseca-Alaniz, M.H.; Takada, J.; Alonso-Vale, M.I.; Lima, F.B. Adipose tissue as an endocrine organ: From theory to practice. J. Pediatr. 2007, 83, S192–S203. [Google Scholar] [CrossRef]
- Wang, B.; Trayhurn, P. Acute and prolonged effects of TNF-α on the expression and secretion of inflammation-related adipokines by human adipocytes differentiated in culture. Pflügers Arch. 2006, 452, 418–427. [Google Scholar] [CrossRef]
- Stienstra, R.; Tack, C.J.; Kanneganti, T.D.; Joosten, L.A.; Netea, M.G. The inflammasome puts obesity in the danger zone. Cell Metab. 2012, 15, 10–18. [Google Scholar] [CrossRef]
- Naugler, W.E.; Karin, M. The wolf in sheep’s clothing: The role of interleukin-6 in immunity, inflammation and cancer. Trends Mol. Med. 2008, 14, 109–119. [Google Scholar] [CrossRef] [PubMed]
- Stenlöf, K.; Wernstedt, I.; Fjällman, T.; Wallenius, V.; Wallenius, K.; Jansson, J.O. Interleukin-6 levels in the central nervous system are negatively correlated with fat mass in overweight/obese subjects. J. Clin. Endocrinol. Metab. 2003, 88, 4379–4383. [Google Scholar] [CrossRef] [PubMed]
- Lavrovsky, Y.; Chatterjee, B.; Clark, R.A.; Roy, A.K. Role of redox-regulated transcription factors in inflammation, aging and age-related diseases. Exp. Gerontol. 2000, 35, 521–532. [Google Scholar] [CrossRef]
- Ruskovska, T.; Bernlohr, D.A. Oxidative stress and protein carbonylation in adipose tissue—Implications for insulin resistance and diabetes mellitus. J. Proteomics 2013, 92, 323–334. [Google Scholar] [CrossRef] [PubMed]
- Ouchi, N.; Higuchi, A.; Ohashi, K.; Oshima, Y.; Gokce, N.; Shibata, R.; Akasaki, Y.; Shimono, A.; Walsh, K. Sfrp5 is an anti-inflammatory adipokine that modulates metabolic dysfunction in obesity. Science 2010, 329, 454–457. [Google Scholar] [CrossRef] [PubMed]
- Amirkhizi, F.; Siassi, F.; Minaie, S.; Djalali, M. Is obesity associated with increased plasma lipid peroxidation and oxidative stress in women? ARYA Atheroscler. J. 2007, 2, 189–192. [Google Scholar]
- Rzheshevsky, A. V Fatal triad: Lipotoxicity, oxidative stress, and phenoptosis. Biochemistry (Mosc.) 2013, 78, 991–1000. [Google Scholar] [CrossRef]
- Saloranta, C.; Franssila-Kallunki, A.; Ekstrand, A.; Taskinen, M.R.; Groop, L. Modulation of hepatic glucose production by non-esterified fatty acids in type 2 (non-insulin-dependent) diabetes mellitus. Diabetologia 1991, 34, 409–415. [Google Scholar] [CrossRef]
- Boden, G.; Chen, X. Effects of fat on glucose uptake and utilization in patients with non-insulin-dependent diabetes. J. Clin. Investig. 1995, 96, 1261–1268. [Google Scholar] [CrossRef]
- Roberts, C.K.; Sindhu, K.K. Oxidative stress and metabolic syndrome. Life Sci. 2009, 84, 705–712. [Google Scholar] [CrossRef] [PubMed]
- Rogge, M.M. The role of impaired mitochondrial lipid oxidation in obesity. Biol. Res. Nurs. 2009, 10, 356–373. [Google Scholar] [CrossRef]
- Duvnjak, M.; Lerotić, I.; Barsić, N.; Tomasić, V.; Virović Jukić, L.; Velagić, V. Pathogenesis and management issues for non-alcoholic fatty liver disease. World J. Gastroenterol. 2007, 13, 4539–4550. [Google Scholar] [CrossRef] [PubMed]
- Goossens, G.H. The role of adipose tissue dysfunction in the pathogenesis of obesity-related insulin resistance. Physiol. Behav. 2008, 94, 206–218. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.I.; Naz, L.; Yasmeen, G. Obesity: An independent risk factor for systemic oxidative stress. Pak. J. Pharm. Sci. 2006, 19, 62–65. [Google Scholar] [PubMed]
- Kuryszko, J.; Sławuta, P.; Sapikowski, G. Secretory function of adipose tissue. Pol. J. Vet. Sci. 2016, 19, 441–446. [Google Scholar] [CrossRef] [PubMed]
- Day, M.J. Ageing, immunosenescence and inflammageing in the dog and cat. J. Comp. Pathol. 2010, 142, S60–S69. [Google Scholar] [CrossRef]
- Mazzatenta, A.; Carluccio, A.; Robbe, D.; Di Giulio, C.; Cellerino, A. The companion dog as a unique translational model for aging. Semin. Cell Dev. Biol. 2017, 70, 141–153. [Google Scholar] [CrossRef]
- Salvioli, S.; Monti, D.; Lanzarini, C.; Conte, M.; Pirazzini, C.; Bacalini, M.G.; Garagnani, P.; Giuliani, C.; Fontanesi, E.; Ostan, R.; et al. Immune system, cell senescence, aging and longevity—Inflamm-aging reappraised. Curr. Pharm. Des. 2013, 19, 1675–1679. [Google Scholar] [CrossRef]
- Franceschi, C.; Bonafè, M.; Valensin, S. Human immunosenescence: The prevailing of innate immunity, the failing of clonotypic immunity, and the filling of immunological space. Vaccine 2000, 18, 1717–1720. [Google Scholar] [CrossRef]
- Larbi, A.; Franceschi, C.; Mazzatti, D.; Solana, R.; Wikby, A.; Pawelec, G. Aging of the immune system as a prognostic factor for human longevity. Physiology 2008, 23, 64–74. [Google Scholar] [CrossRef] [PubMed]
- Leng, S.X.; Xue, Q.-L.; Tian, J.; Huang, Y.; Yeh, S.-H.; Fried, L.P. Associations of neutrophil and monocyte counts with frailty in community-dwelling disabled older women: Results from the women’s health and aging studies I. Exp. Gerontol. 2009, 44, 511–516. [Google Scholar] [CrossRef] [PubMed]
- Mocchegiani, E.; Giacconi, R.; Cipriano, C.; Malavolta, M. NK and NKT cells in aging and longevity: Role of zinc and metallothioneins. J. Clin. Immunol. 2009, 29, 416–425. [Google Scholar] [CrossRef] [PubMed]
- Mathur, S.K.; Schwantes, E.A.; Jarjour, N.N.; Busse, W.W. Age-related changes in eosinophil function in human subjects. Chest 2008, 133, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Wenisch, C.; Patruta, S.; Daxböck, F.; Krause, R.; Hörl, W. Effect of age on human neutrophil function. J. Leukoc. Biol. 2000, 67, 40–45. [Google Scholar] [CrossRef] [PubMed]
- Withers, S.S.; Moore, P.F.; Chang, H.; Choi, J.W.; McSorley, S.J.; Kent, M.S.; Monjazeb, A.M.; Canter, R.J.; Murphy, W.J.; Sparger, E.E.; et al. Multi-color flow cytometry for evaluating age-related changes in memory lymphocyte subsets in dogs. Dev. Comp. Immunol. 2018, 87, 64–74. [Google Scholar] [CrossRef] [PubMed]
- Fujiwara, M.; Yonezawa, T.; Arai, T.; Yamamoto, I.; Ohtsuka, H. Alterations with age in peripheral blood lymphocyte subpopulations and cytokine synthesis in beagles. Vet. Med. 2012, 3, 79–84. [Google Scholar] [CrossRef]
- Mongillo, P.; Bertotto, D.; Pitteri, E.; Stefani, A.; Marinelli, L.; Gabai, G. Peripheral leukocyte populations and oxidative stress biomarkers in aged dogs showing impaired cognitive abilities. Age (Dordr.) 2015, 37, 39. [Google Scholar] [CrossRef]
- Shaw, A.C.; Joshi, S.; Greenwood, H.; Panda, A.; Lord, J.M. Aging of the innate immune system. Curr. Opin. Immunol. 2010, 22, 507–513. [Google Scholar] [CrossRef]
- Boehmer, E.D.; Goral, J.; Faunce, D.E.; Kovacs, E.J. Age-dependent decrease in Toll-like receptor 4-mediated proinflammatory cytokine production and mitogen-activated protein kinase expression. J. Leukoc. Biol. 2004, 75, 342–349. [Google Scholar] [CrossRef]
- Agius, E.; Lacy, K.E.; Vukmanovic-Stejic, M.; Jagger, A.L.; Papageorgiou, A.-P.; Hall, S.; Reed, J.R.; Curnow, S.J.; Fuentes-Duculan, J.; Buckley, C.D.; et al. Decreased TNF-alpha synthesis by macrophages restricts cutaneous immunosurveillance by memory CD4+ T cells during aging. J. Exp. Med. 2009, 206, 1929–1940. [Google Scholar] [CrossRef] [PubMed]
- Fagiolo, U.; Cossarizza, A.; Scala, E.; Fanales-Belasio, E.; Ortolani, C.; Cozzi, E.; Monti, D.; Franceschi, C.; Paganelli, R. Increased cytokine production in mononuclear cells of healthy elderly people. Eur. J. Immunol. 1993, 23, 2375–2378. [Google Scholar] [CrossRef] [PubMed]
- Zanni, F.; Vescovini, R.; Biasini, C.; Fagnoni, F.; Zanlari, L.; Telera, A.; Di Pede, P.; Passeri, G.; Pedrazzoni, M.; Passeri, M.; et al. Marked increase with age of type 1 cytokines within memory and effector/cytotoxic CD8+ T cells in humans: A contribution to understand the relationship between inflammation and immunosenescence. Exp. Gerontol. 2003, 38, 981–987. [Google Scholar] [CrossRef]
- Horiuchi, S.; Wilmoth, J.R. Age patterns of the life table aging rate for major causes of death in Japan, 1951–1990. J. Gerontol. A Biol. Sci. Med. Sci. 1997, 52, B67–B77. [Google Scholar] [CrossRef] [PubMed]
- Haigis, M.C.; Yankner, B.A. The Aging Stress Response. Mol. Cell 2010, 40, 333–344. [Google Scholar] [CrossRef] [PubMed]
- Alexeyev, M.F. Is there more to aging than mitochondrial DNA and reactive oxygen species? FEBS J. 2009, 276, 5768–5787. [Google Scholar] [CrossRef] [PubMed]
- De la Fuente, M.; Hernanz, A.; Guayerbas, N.; Alvarez, P.; Alvarado, C. Changes with age in peritoneal macrophage functions. Implication of leukocytes in the oxidative stress of senescence. Cell. Mol. Biol. (Noisy-le-Grand) 2004, 50, OL683–OL690. [Google Scholar]
- Fujimoto, H.; Kobayashi, H.; Ohno, M. Age-induced reduction in mitochondrial manganese superoxide dismutase activity and tolerance of macrophages against apoptosis induced by oxidized low density lipoprotein. Circ. J. 2010, 74, 353–360. [Google Scholar] [CrossRef]
- Cannizzo, E.S.; Clement, C.C.; Sahu, R.; Follo, C.; Santambrogio, L. Oxidative stress, inflammaging, and immunosenescence. J. Proteomics 2011, 74, 2313–2323. [Google Scholar] [CrossRef]
- Larbi, A.; Dupuis, G.; Douziech, N.; Khalil, A.; Fülöp, T. Low-grade inflammation with aging has consequences for T-lymphocyte signaling. Ann. N. Y. Acad. Sci. 2004, 1030, 125–133. [Google Scholar] [CrossRef]
- Holder, A.; Mella, S.; Palmer, D.B.; Aspinall, R.; Catchpole, B. An age-associated decline in thymic output differs in dog breeds according to their longevity. PLoS ONE 2016, 11, e0165968. [Google Scholar] [CrossRef]
- Head, E.; Liu, J.; Hagen, T.M.; Muggenburg, B.A.; Milgram, N.W.; Ames, B.N.; Cotman, C.W. Oxidative damage increases with age in a canine model of human brain aging. J. Neurochem. 2002, 82, 375–381. [Google Scholar] [CrossRef] [PubMed]
- Moyer, K.L.; Trepanier, L.A. Erythrocyte glutathione and plasma cysteine concentrations in young versus old dogs. J. Am. Vet. Med. Assoc. 2009, 234, 95–99. [Google Scholar] [CrossRef] [PubMed]
- Head, E.; Rofina, J.; Zicker, S. Oxidative stress, aging, and central nervous system disease in the canine model of human brain aging. Vet. Clin. N. Am. Small Anim. Pract. 2008, 38, 167–178. [Google Scholar] [CrossRef] [PubMed]
- Vida, C.; Martinez de Toda, I.; Garrido, A.; Carro, E.; Molina, J.A.; De la Fuente, M. Impairment of several immune functions and redox state in blood cells of Alzheimer’s disease patients. Relevant role of neutrophils in oxidative stress. Front. Immunol. 2018, 8, 1974. [Google Scholar] [CrossRef] [PubMed]
- Acamovic, T.; Brooker, J.D. Biochemistry of plant secondary metabolites and their effects in animals. Proc. Nutr. Soc. 2005, 64, 403–412. [Google Scholar] [CrossRef]
- Colitti, M.; Grasso, S. Nutraceuticals and regulation of adipocyte life: Premises or promises. Biofactors 2014, 40, 398–418. [Google Scholar] [CrossRef] [PubMed]
- Pomari, E.; Stefanon, B.; Colitti, M. Effect of Arctium lappa (burdock) extract on canine dermal fibroblasts. Vet. Immunol. Immunopathol. 2013, 156, 159–166. [Google Scholar] [CrossRef]
- Pomari, E.; Stefanon, B.; Colitti, M. Effect of plant extracts on H2O2-induced inflammatory gene expression in macrophages. J. Inflamm. Res. 2014, 7, 103–112. [Google Scholar] [CrossRef]
- Stefanon, B.; Sgorlon, S.; Gabai, G. Usefulness of nutraceutics in controlling oxidative stress in dairy cows around parturition. Vet. Res. Commun. 2005, 29 (Suppl. 2), 387–390. [Google Scholar] [CrossRef]
- Sgorlon, S.; Stradaioli, G.; Zanin, D.; Stefanon, B. Biochemical and molecular response to antioxidant supplementation in sheep. Small Rumin. Res. 2006, 64, 143–151. [Google Scholar] [CrossRef]
- Sgorlon, S.; Colitti, M.; Asquini, E.; Ferrarini, A.; Pallavicini, A.; Stefanon, B. Administration of botanicals with the diet regulates gene expression in peripheral blood cells of Sarda sheep during ACTH challenge. Domest. Anim. Endocrinol. 2012, 43, 213–226. [Google Scholar] [CrossRef] [PubMed]
- Stefanon, B.; Sgorlon, S.; De Moro, G.; Asquini, E. Action of larch bark in the regulation of cortisol induced stress in sheep. Ital. J. Anim. Sci. 2009, 8, 162–164. [Google Scholar] [CrossRef]
- Sgorlon, S.; Stefanon, B.; Sandri, M.; Colitti, M. Nutrigenomic activity of plant derived compounds in health and disease: Results of a dietary intervention study in dog. Res. Vet. Sci. 2016, 109, 142–148. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Jin, Y.; Du, M.; Liu, W.; Ren, Y.; Zhang, C.; Zhang, J. The effect of dietary grape pomace supplementation on epididymal sperm quality and testicular antioxidant ability in ram lambs. Theriogenology 2017, 97, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Colitti, M.; Stefanon, B. Effect of natural antioxidants on superoxide dismutase and glutathione peroxidase mRNA expression in leukocytes from periparturient dairy cows. Vet. Res. Commun. 2006, 30, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Colitti, M.; Sgorlon, S.; Stradaioli, G.; Farinacci, M.; Gabai, G.; Stefanon, B. Grape polyphenols affect mRNA expression of PGHS-2, TIS11b and FOXO3 in endometrium of heifers under ACTH-induced stress. Theriogenology 2007, 68, 1022–1030. [Google Scholar] [CrossRef] [PubMed]
- Colitti, M.; Gaspardo, B.; Della Pria, A.; Scaini, C.; Stefanon, B. Transcriptome modification of white blood cells after dietary administration of curcumin and non-steroidal anti-inflammatory drug in osteoarthritic affected dogs. Vet. Immunol. Immunopathol. 2012, 147, 136–146. [Google Scholar] [CrossRef] [PubMed]
- Farinacci, M.; Colitti, M.; Sgorlon, S.; Stefanon, B. Immunomodulatory activity of plant residues on ovine neutrophils. Vet. Immunol. Immunopathol. 2008, 126, 54–63. [Google Scholar] [CrossRef]
- Farinacci, M.; Colitti, M.; Stefanon, B. Modulation of ovine neutrophil function and apoptosis by standardized extracts of Echinacea angustifolia, Butea frondosa and Curcuma longa. Vet. Immunol. Immunopathol. 2009, 128, 366–373. [Google Scholar] [CrossRef]
- Farinacci, M.; Gaspardo, B.; Colitti, M.; Stefanon, B. Dietary administration of curcumin modifies trascriptional profile of genes involved in inflammatory cascade in horse leukocytes. Ital. J. Anim. Sci. 2009, 8, 84–86. [Google Scholar] [CrossRef]
- Gladine, C.; Rock, E.; Morand, C.; Bauchart, D.; Durand, D. Bioavailability and antioxidant capacity of plant extracts rich in polyphenols, given as a single acute dose, in sheep made highly susceptible to lipoperoxidation. Br. J. Nutr. 2007, 98, 691–701. [Google Scholar] [CrossRef]
- Montanari, T.; Pošćić, N.; Colitti, M. Factors involved in white-to-brown adipose tissue conversion and in thermogenesis: A review. Obes. Rev. 2017, 18, 495–513. [Google Scholar] [CrossRef] [PubMed]
- Wink, M. Evolution of toxins and antinutritional factors in plants with special emphasis on Leguminosae. In Poisonous Plants and Related Toxins, 1st ed.; Acamovic, T., Stewart, C.S., Pennycott, T.W., Eds.; CABI Publishing: Wallingford, UK, 2004; pp. 1–25. [Google Scholar]
- Bravo, L. Polyphenols: Chemistry, dietary sources, metabolism, and nutritional significance. Nutr. Rev. 1998, 56, 317–333. [Google Scholar] [CrossRef] [PubMed]
- Quideau, S.; Deffieux, D.; Douat-Casassus, C.; Pouységu, L. Plant polyphenols: Chemical properties, biological activities, and synthesis. Angew. Chem. Int. Ed. Engl. 2011, 50, 586–621. [Google Scholar] [CrossRef]
- Gostner, J.M.; Becker, K.; Ueberall, F.; Fuchs, D. The good and bad of antioxidant foods: An immunological perspective. Food Chem. Toxicol. 2015, 80, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Vertuani, S.; Angusti, A.; Manfredini, S. The antioxidants and pro-antioxidants network: An overview. Curr. Pharm. Des. 2004, 10, 1677–1694. [Google Scholar] [CrossRef]
- Fenech, M.; El-Sohemy, A.; Cahill, L.; Ferguson, L.R.; French, T.-A.C.; Tai, E.S.; Milner, J.; Koh, W.-P.; Xie, L.; Zucker, M.; et al. Nutrigenetics and nutrigenomics: Viewpoints on the current status and applications in nutrition research and practice. J. Nutrigenet. Nutrigenomics 2011, 4, 69–89. [Google Scholar] [CrossRef]
- Braicu, C.; Mehterov, N.; Vladimirov, B.; Sarafian, V.; Nabavi, S.M.; Atanasov, A.G.; Berindan-Neagoe, I. Nutrigenomics in cancer: Revisiting the effects of natural compounds. Semin. Cancers Biol. 2017, 46, 84–106. [Google Scholar] [CrossRef] [PubMed]
- Surh, Y.J. Cancer chemoprevention with dietary phytochemicals. Nat. Rev. Cancer 2003, 3, 768–780. [Google Scholar] [CrossRef]
- Barnes, S. Nutritional genomics, polyphenols, diets, and their impact on dietetics. J. Am. Diet. Assoc. 2008, 108, 1888–1895. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, D.K.; Mishra, P.K. Curcumin and its analogues: Potential anticancer agents. Med. Res. Rev. 2010, 30, 818–860. [Google Scholar] [CrossRef] [PubMed]
- Gollucke, A.P.B.; Peres, R.C.; Odair, A.; Ribeiro, D.A. Polyphenols: A nutraceutical approach against diseases. Recent Pat. Food. Nutr. Agric. 2013, 5, 214–219. [Google Scholar] [CrossRef] [PubMed]
- Herman, F.; Westfall, S.; Brathwaite, J.; Pasinetti, G.M. Suppression of presymptomatic oxidative stress and inflammation in neurodegeneration by grape-derived polyphenols. Front. Pharmacol. 2018, 9, 867. [Google Scholar] [CrossRef] [PubMed]
- Sgorlon, S.; Stradioli, G.; Stefanon, B.; Altimer, G.; Della Loggia, R. Dietary grape polyphenols modulate oxidative stress in ageing rabbits. Ital. J. Anim. Sci. 2005, 88, 541–543. [Google Scholar] [CrossRef]
- Pan, M.-H.; Lai, C.-S.; Ho, C.-T. Anti-inflammatory activity of natural dietary flavonoids. Food Funct. 2010, 1, 15–31. [Google Scholar] [CrossRef] [PubMed]
- Salvadó, M.J.; Casanova, E.; Fernández-Iglesias, A.; Arola, L.; Bladé, C. Roles of proanthocyanidin rich extracts in obesity. Food Funct. 2015, 6, 1053–1071. [Google Scholar] [CrossRef]
- Williamson, G. Possible effects of dietary polyphenols on sugar absorption and digestion. Mol. Nutr. Food Res. 2013, 57, 48–57. [Google Scholar] [CrossRef]
- Bauer, R.; Wenzig, E.M.; Stefanon, B.; Farinacci, M.; Sgorlon, S.; Tzika, H.; Kyriakis, S.; Franz, C. Use Larch Wood Material for Treating Inflammation. Patent International Publication Number WO2009/079, 29 September 2010. [Google Scholar]
- Mauro, D.; Procopio, L. Blockade of nitric oxide overproduction and oxidative stress by nigella sativa oil attenuates morphine-induced tolerance. Neurochem. Res. 2011, 36, 924–925. [Google Scholar] [CrossRef]
- Wardyn, J.D.; Ponsford, A.H.; Sanderson, C.M. Dissecting molecular cross-talk between Nrf2 and NF-κB response pathways. Biochem. Soc. Trans. 2015, 43, 621–626. [Google Scholar] [CrossRef]
- Chi, X.; Yao, W.; Xia, H.; Jin, Y.; Li, X.; Cai, J.; Hei, Z. Elevation of HO-1 expression mitigates intestinal ischemia-reperfusion injury and restores tight junction function in a rat liver transplantation model. Oxid. Med. Cell. Longev. 2015, 2015, 986075. [Google Scholar] [CrossRef] [PubMed]
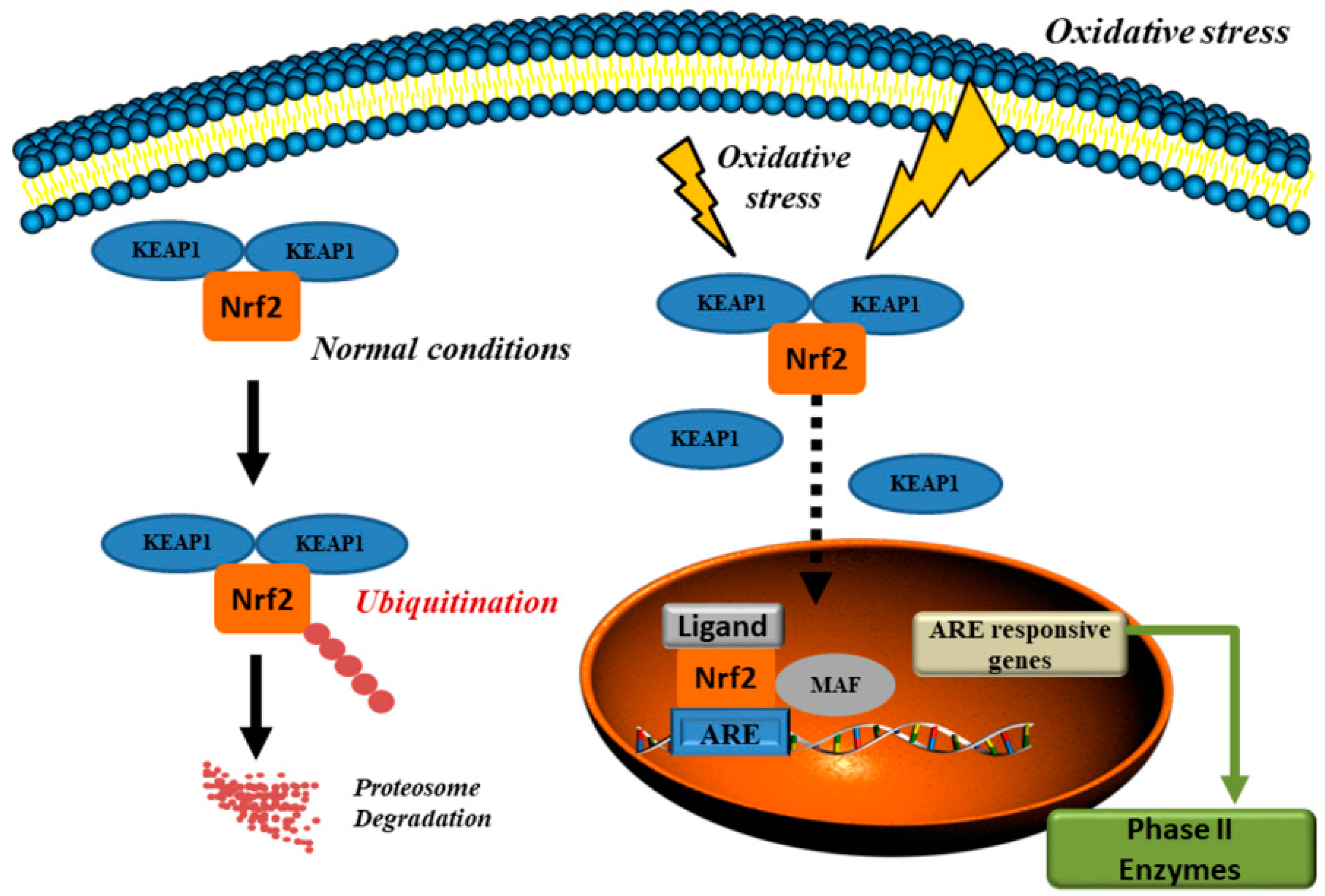
- Ahmed, S.M.; Luo, L.; Namani, A.; Wang, X.J.; Tang, X. Nrf2 signaling pathway: Pivotal roles in inflammation. Biochim. Biophys. Acta Mol. Basis Dis. 2017, 1863, 585–597. [Google Scholar] [CrossRef] [PubMed]
- Itoh, K.; Wakabayashi, N.; Katoh, Y.; Ishii, T.; Igarashi, K.; Engel, J.D.; Yamamoto, M. Keap1 represses nuclear activation of antioxidant responsive elements by Nrf2 through binding to the amino-terminal Neh2 domain. Genes Dev. 1999, 13, 76–86. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, A.; Kang, M.-I.; Okawa, H.; Ohtsuji, M.; Zenke, Y.; Chiba, T.; Igarashi, K.; Yamamoto, M. Oxidative stress sensor Keap1 functions as an adaptor for Cul3-based E3 ligase to regulate proteasomal degradation of Nrf2. Mol. Cell. Biol. 2004, 24, 7130–7139. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.; Wu, R.T.; Wu, T.; Khor, T.-O.; Wang, H.; Kong, A.-N. Sulforaphane suppressed LPS-induced inflammation in mouse peritoneal macrophages through Nrf2 dependent pathway. Biochem. Pharmacol. 2008, 76, 967–973. [Google Scholar] [CrossRef] [PubMed]
- Yamato, O.; Tsuneyoshi, T.; Ushijima, M.; Jikihara, H.; Yabuki, A. Safety and efficacy of aged garlic extract in dogs: Upregulation of the nuclear factor erythroid 2-related factor 2 (Nrf2) signaling pathway and Nrf2-regulated phase II antioxidant enzymes. BMC Vet. Res. 2018, 14, 373. [Google Scholar] [CrossRef] [PubMed]
- Gifford, A.; Towse, T.F.; Walker, R.C.; Avison, M.J.; Welch, E.B. Characterizing active and inactive brown adipose tissue in adult humans using PET-CT and MR imaging. Am. J. Physiol. Endocrinol. 2016, 311, E95–E104. [Google Scholar] [CrossRef]
- Lee, H.; Lee, Y.J.; Choi, H.; Ko, E.H.; Kim, J.W. Reactive oxygen species facilitate adipocyte differentiation by accelerating mitotic clonal expansion. J. Biol. Chem. 2009, 284, 10601–10609. [Google Scholar] [CrossRef]
- Lee, O.H.; Kwon, Y.-I.; Apostolidis, E.; Shetty, K.; Kim, Y.C. Rhodiola-induced inhibition of adipogenesis involves antioxidant enzyme response associated with pentose phosphate pathway. Phutother. Res. 2011, 25, 106–115. [Google Scholar] [CrossRef]
- Pomari, E.; Stefanon, B.; Colitti, M. Effects of two different Rhodiola rosea extracts on primary human visceral adipocytes. Molecules 2015, 20, 8409–8428. [Google Scholar] [CrossRef]
- Witkin, J.M.; Li, X. Curcumin, an active constiuent of the ancient medicinal herb Curcuma longa L.: Some uses and the establishment and biological basis of medical efficacy. CNS Neurol. Disord. Drug Targets 2013, 12, 487–497. [Google Scholar] [CrossRef] [PubMed]
- Colitti, M.; Stefanon, B. Different anti-adipogenic effects of bio-compounds on primary visceral pre-adipocytes and adipocytes. EXCLI J. 2016, 15, 362–377. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Sun, X.B.; Ye, F.; Tian, W.X. Suppression of fatty acid synthase, differentiation and lipid accumulation in adipocytes by curcumin. Mol. Cell. Biochem. 2011, 35, 19–28. [Google Scholar] [CrossRef]
- Kim, C.Y.; Le, T.T.; Chen, C.; Cheng, J.-X.; Kim, K.-H. Curcumin inhibits adipocyte differentiation through modulation of mitotic clonal expansion. J. Nutr. Biochem. 2011, 22, 910–920. [Google Scholar] [CrossRef] [PubMed]
- González-Castejón, M.; García-Carrasco, B.; Fernández-Dacosta, R.; Dávalos, A.; Rodriguez-Casado, A. Reduction of adipogenesis and lipid accumulation by Taraxacum officinale (Dandelion) extracts in 3T3L1 adipocytes: An in vitro study. Phytother. Res. 2014, 28, 745–752. [Google Scholar] [CrossRef] [PubMed]
- Stefanon, B.; Pomari, E.; Colitti, M. Effects of Rosmarinus officinalis extract on human primary omental preadipocytes and adipocytes. Exp. Biol. Med. 2015, 240, 884–895. [Google Scholar] [CrossRef] [PubMed]
- Colitti, M.; Boschi, F.; Montanari, T. Dynamic of lipid droplets and gene expression in response to β-aminoisobutyric acid treatment on 3T3-L1 cells. Eur. J. Histochem. 2018, 62. [Google Scholar] [CrossRef]
- Gao, A.W.; Houtkooper, R.H. Mitochondrial fission: Firing up mitochondria in brown adipose tissue. EMBO J. 2014, 33, 401–402. [Google Scholar] [CrossRef]
| Nitrogen Containing Compounds | Number of Natural Compounds | Non-Nitrogen Containing Compounds | Number of Natural Compounds |
|---|---|---|---|
| Alkaloids | 12,000 | Monoterpenes | 1000 |
| Non protein amino acids | 600 | Sesquiterpenes | 3000 |
| Amines | 100 | Diterpenes | 2000 |
| Cyanogenic glycosides | 100 | Triterpenes, Saponins, Steroids | 4000 |
| Glucosinolates | 100 | Tetraterpens | 350 |
| Flavonoids | 2000 | ||
| Polyacetylenes | 1000 | ||
| Polyketides | 750 | ||
| Phenylpropanes | 1000 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Colitti, M.; Stefanon, B.; Gabai, G.; Gelain, M.E.; Bonsembiante, F. Oxidative Stress and Nutraceuticals in the Modulation of the Immune Function: Current Knowledge in Animals of Veterinary Interest. Antioxidants 2019, 8, 28. https://doi.org/10.3390/antiox8010028
Colitti M, Stefanon B, Gabai G, Gelain ME, Bonsembiante F. Oxidative Stress and Nutraceuticals in the Modulation of the Immune Function: Current Knowledge in Animals of Veterinary Interest. Antioxidants. 2019; 8(1):28. https://doi.org/10.3390/antiox8010028
Chicago/Turabian StyleColitti, Monica, Bruno Stefanon, Gianfranco Gabai, Maria Elena Gelain, and Federico Bonsembiante. 2019. "Oxidative Stress and Nutraceuticals in the Modulation of the Immune Function: Current Knowledge in Animals of Veterinary Interest" Antioxidants 8, no. 1: 28. https://doi.org/10.3390/antiox8010028
APA StyleColitti, M., Stefanon, B., Gabai, G., Gelain, M. E., & Bonsembiante, F. (2019). Oxidative Stress and Nutraceuticals in the Modulation of the Immune Function: Current Knowledge in Animals of Veterinary Interest. Antioxidants, 8(1), 28. https://doi.org/10.3390/antiox8010028








