Bark of Passiflora edulis Treatment Stimulates Antioxidant Capacity, and Reduces Dyslipidemia and Body Fat in db/db Mice
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals and Experimental Protocol
2.2. Preparation of Chow with Bark of Passiflora Edulis
2.3. Bark of Passiflora Edulis Antioxidants Characterization
2.3.1. Carotenoids
2.3.2. Determination of 2, 2-Diphenyl-1-picrylhydrazyl radical (DPPH) Radical Scavenging Activity
2.3.3. Determination of Total Phenol Content
2.4. Nutritional Parameters
2.5. Metabolic Parameters
2.6. Preparation of Tissues for Oxidative Stress Analysis
2.6.1. Malondialdehyde (MDA)
2.6.2. Antioxidant Capacity
3. Statistical Analysis
4. Results
4.1. Bark of Passiflora Edulis Antioxidants Characterization
4.2. Bark of Passiflora Edulis Reduced Visceral and Subcutaneous Fat Deposit and Adiposity Index
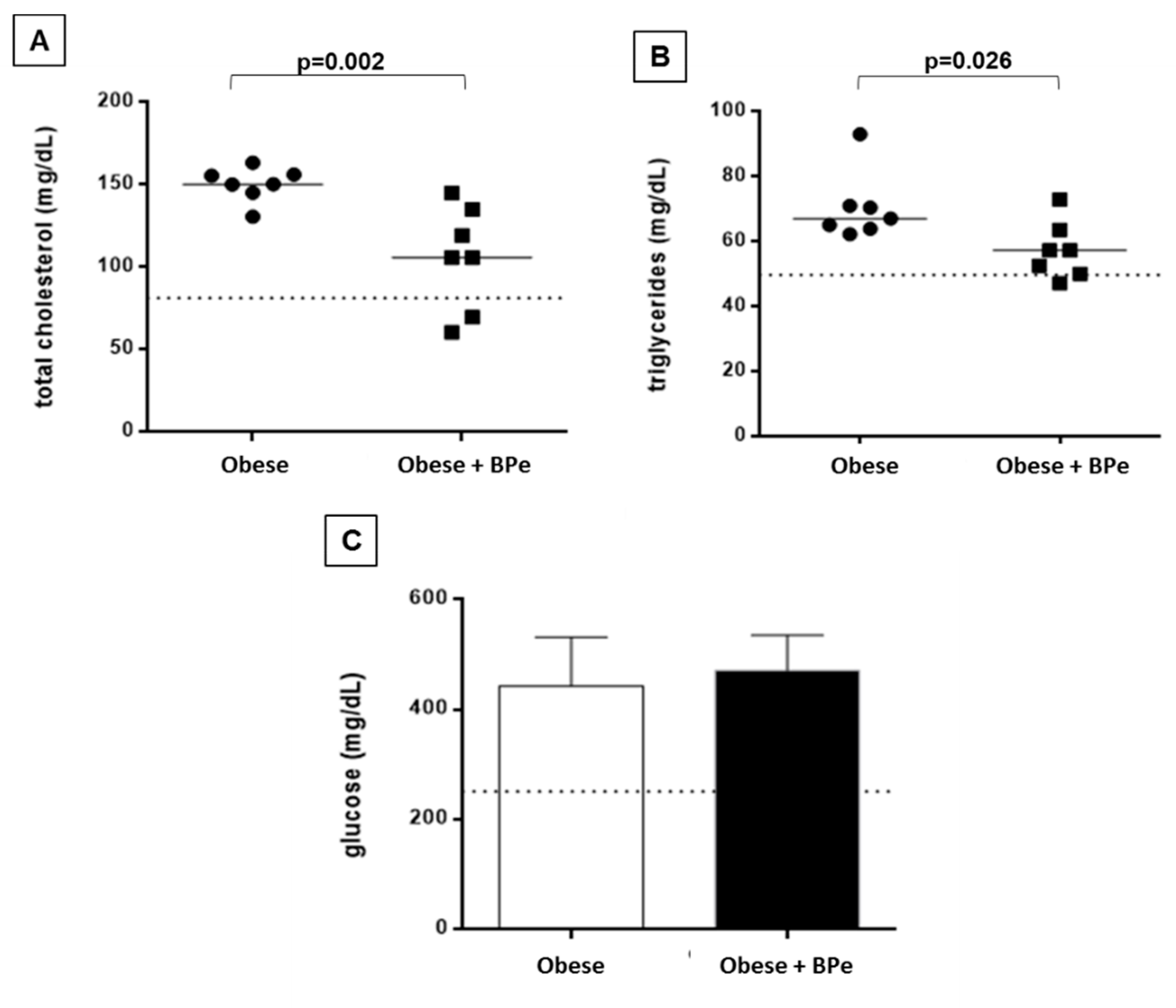
4.3. Bark of Passiflora Edulis Reduced Cholesterol and Triglycerides Levels
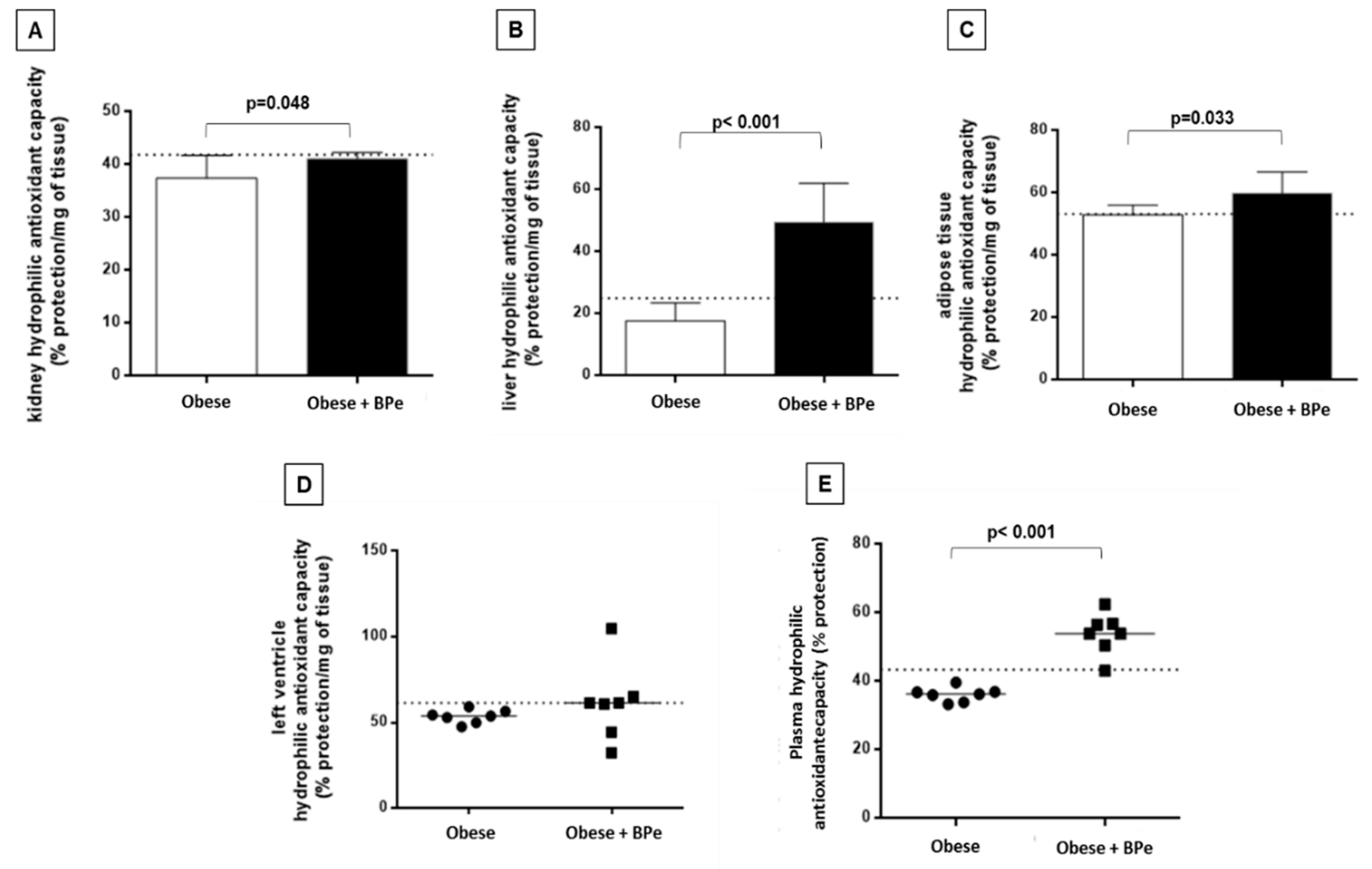
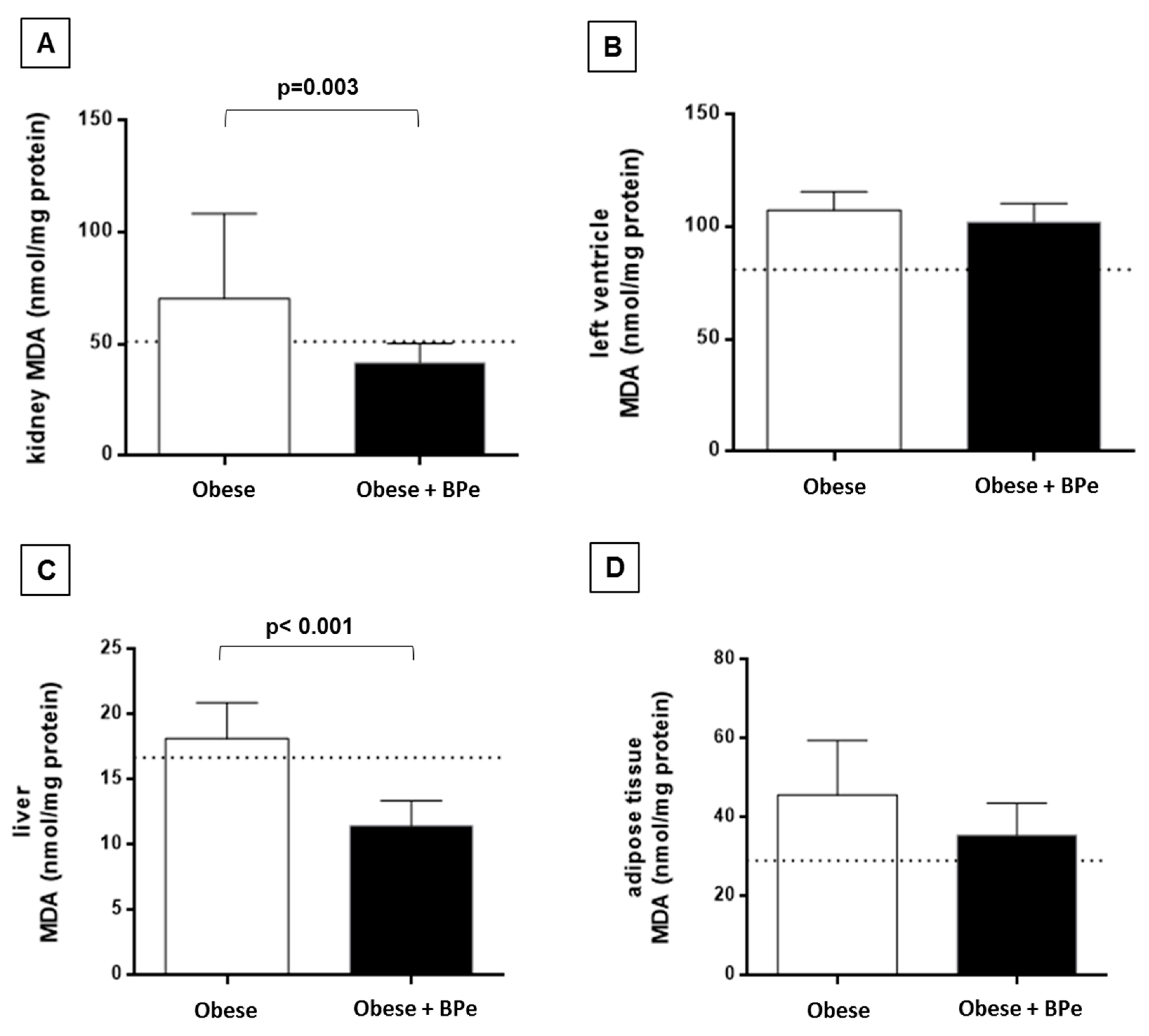
4.4. Bark of Passiflora Edulis Ameliorates the Antioxidant Capacity and Reduces MDA Levels
5. Discussion
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Zhan, J.; Liu, Y.-J.; Cai, L.-B.; Xu, F.-R.; Xie, T.; He, Q.-Q. Fruit and vegetable consumption and risk of cardiovascular disease: A meta-analysis of prospective cohort studies. Crit. Rev. Food Sci. Nutr. 2017, 57, 1650–1663. [Google Scholar] [CrossRef] [PubMed]
- Saita, E.; Kondo, K.; Momiyama, Y. Anti-inflammatory diet for atherosclerosis and coronary artery disease: Antioxidant foods. Clin. Med. Insights Cardiol. 2014, 8 (Suppl. 3), 61–65. [Google Scholar] [CrossRef] [PubMed]
- Mori, T.A. Marine OMEGA-3 fatty acids in the prevention of cardiovascular disease. Fitoterapia 2017, 123, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Drewnowski, A.; Darmon, N. The economics of obesity: Dietary energy density and energy cost. Am. J. Clin. Nutr. 2005, 82, 265–273. [Google Scholar] [CrossRef] [PubMed]
- WHO. World Health Organisation Obesity and Overweight Fact Sheet; WHO: Geneva, Switzerland, 2016. [Google Scholar]
- Glass, C.K.; Olefsky, J.M. Inflammation and lipid signaling in the etiology of insulin resistance. Cell Metab. 2012, 15, 635–645. [Google Scholar] [CrossRef] [PubMed]
- Hirabara, S.M.; Gorjão, R.; Vinolo, M.A.; Rodrigues, A.C.; Nachbar, R.T.; Curi, R. Molecular targets related to inflammation and insulin resistance and potential interventions. J. Biomed. Biotechnol. 2012, 2012, 379024. [Google Scholar] [CrossRef] [PubMed]
- Van de Woestijne, A.P.; Monajemi, H.; Kalkhoven, E.; Visseren, F.L.J. Adipose tissue dysfunction and hypertriglyceridemia: Mechanisms and management. Obes. Rev. 2011, 12, 829–840. [Google Scholar] [CrossRef] [PubMed]
- Klöting, N.; Blüher, M. Adipocyte dysfunction, inflammation and metabolic syndrome. Rev. Endocr. Metab. Disord. 2014, 15, 277–287. [Google Scholar] [CrossRef] [PubMed]
- Forbes, J.M.; Coughlan, M.T.; Cooper, M.E. Oxidative stress as a major culprit in kidney disease in diabetes. Diabetes 2008, 57, 1446–1454. [Google Scholar] [CrossRef] [PubMed]
- Manna, P.; Jain, S.K. Obesity, Oxidative Stress, Adipose Tissue Dysfunction, and the Associated Health Risks: Causes and Therapeutic Strategies. Metab. Syndr. Relat. Disord. 2015, 13, 423–444. [Google Scholar] [CrossRef] [PubMed]
- Santilli, F.; D’Ardes, D.; Davì, G. Oxidative stress in chronic vascular disease: From prediction to prevention. Vasc. Pharmacol. 2015, 74, 23–37. [Google Scholar] [CrossRef] [PubMed]
- Poljsak, B.; Šuput, D.; Milisav, I. Achieving the balance between ROS and antioxidants: When to use the synthetic antioxidants. Oxid. Med. Cell. Longev. 2013, 2013, 956792. [Google Scholar] [CrossRef] [PubMed]
- Montezano, A.C.; Dulak-Lis, M.; Tsiropoulou, S.; Harvey, A.; Briones, A.M.; Touyz, R.M. Oxidative stress and human hypertension: Vascular mechanisms, biomarkers, and novel therapies. Can. J. Cardiol. 2015, 31, 631–641. [Google Scholar] [CrossRef] [PubMed]
- Mangge, H. Antioxidants, inflammation and cardiovascular disease. World J. Cardiol. 2014, 6, 462. [Google Scholar] [CrossRef] [PubMed]
- Pham-Huy, L.A.; He, H.; Pham-Huy, C. Free radicals, antioxidants in disease and health. Int. J. Biomed. Sci. 2008, 4, 89–96. [Google Scholar] [PubMed]
- Ulmer, T.; MacDougal, J.M. Passiflora: Passionflowers of the World; Timber Press: Portland, OR, USA, 2004. [Google Scholar]
- Martin, F.; Nakasone, H. The edible species of Passiflora. Econ. Bot. 1970, 24, 333–343. [Google Scholar] [CrossRef]
- Medina, S.; Collado-González, J.; Ferreres, F.; Londoño-Londoño, J.; Jiménez-Cartagena, C.; Guy, A.; Durand, T.; Galano, J.M.; Gil-Izquierdo, A. Quantification of phytoprostanes–bioactive oxylipins–and phenolic compounds of Passiflora edulis Sims shell using UHPLC-QqQ-MS/MS and LC-IT-DAD-MS/MS. Food Chem. 2017, 229, 1–8. [Google Scholar] [CrossRef] [PubMed]
- De Santana, F.C.; de Oliveira Torres, L.R.; Shinagawa, F.B.; e Silva, A.M.; Yoshime, L.T.; de Melo, I.L.P.; Marcellini, P.S.; Mancini-Filho, J. Optimization of the antioxidant polyphenolic compounds extraction of yellow passion fruit seeds (Passiflora edulis Sims) by response surface methodology. J. Food Sci. Technol. 2017, 54, 3552–3561. [Google Scholar] [CrossRef] [PubMed]
- Correa, E.; Medina, L.; Barros-Monteiro, J.; Valle, N.; Sales, R.; Magalaes, A. The Intake of Fiber Mesocarp Passionfruit (Passiflora Edulis) Lowers Levels of Triglyceride and Cholesterol Decreasing Principally Insulin and Leptin. J. Aging Res. Clin. Pract. 2014, 3, 31–35. [Google Scholar] [PubMed]
- Wong, Y.; Sia, C.; Khoo, H.; Ang, Y.; Chang, S.; Uim, H. Influence of extraction conditions on antioxidant properties of passion fruit (Passiflora edulis) peel. Acta Sci. Pol. Technol. Aliment. 2014, 13, 257–265. [Google Scholar] [CrossRef] [PubMed]
- Galisteo, M.; Duarte, J.; Zarzuelo, A. Effects of dietary fibers on disturbances clustered in the metabolic syndrome. J. Nutr. Biochem. 2008, 19, 71–84. [Google Scholar] [CrossRef] [PubMed]
- Silva, D.; Freitas, A.; Pessoa, C.; Paula, R.; Mesquita, J.; Leal, L.; Brito, G.A.; Gonçalves, D.O.; Viana, G.S. Pectin from Passiflora edulis shows anti-inflammatory action as well as hypoglycemic and hypotriglyceridemic properties in diabetic rats. J. Med. Food 2011, 14, 1118–1126. [Google Scholar] [CrossRef] [PubMed]
- Baú Betim Cazarin, C.; da Silva, J.K.; Colomeu, T.C.; de Lima Zollner, R.; Maróstica Junior, M.R. Capacidade antioxidante e composição química da casca de maracujá (Passiflora edulis). Ciênc. Rural 2014, 44, 1699–1704. [Google Scholar] [CrossRef]
- De Queiroz, M.D.; Janebro, D.I.; da Cunha, M.A.; dos Santos Medeiros, J.; Sabaa-Srur, A.U.; Margareth de Fatima, F.M.; Dos Santos, S.C. Effect of the yellow passion fruit peel flour (Passiflora edulis f. flavicarpa deg.) in insulin sensitivity in type 2 diabetes mellitus patients. Nutr. J. 2012, 11, 89. [Google Scholar] [CrossRef] [PubMed]
- Olfert, E.D.; Cross, B.M.; McWilliam, A.A. Guide to the Care and Use of Experimental Animals; Canadian Council on Animal Care: Ottawa, ON, Canada, 1993; Volume 1. [Google Scholar]
- Lima, E.S.; Schwertz, M.C.; Sobreira, C.R.C.; Borras, M.R.L. Efeito hipoglicemiante da farinha do fruto de maracujá-do-mato (Passiflora nitida Kunth) em ratos normais e diabéticos. Rev. Bras. Plantas Med. 2012, 14, 383–388. [Google Scholar] [CrossRef]
- Yeum, K.; Booth, S.L.; Sadowski, J.A.; Chun, L.; Tang, G.; Krinsky, N.I.; Russell, R.M. Human plasma carotenoid response to the ingestion of controlled diets high in fruits and vegetables. Am. J. Clin. Nutr. 1996, 64, 594–602. [Google Scholar] [CrossRef] [PubMed]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT-Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Horwitz, W. Official Methods of Analysis of the Association of Official analytical Chemists, 14th ed.; Chemists AOOA, Organizador: Washington, DC, USA, 1984. [Google Scholar]
- Pierine, D.T.; Navarro, M.E.L.; Minatel, I.O.; Luvizotto, R.A.M.; Nascimento, A.F.; Ferreira, A.L.A.; Yeum, K.J.; Corrêa, C.R. Lycopene supplementation reduces TNF-α via RAGE in the kidney of obese rats. Nutr. Diabetes 2014, 4, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Beretta, G.; Aldini, G.; Facino, R.M.; Russell, R.M.; Krinsky, N.I.; Yeum, K.J. Total antioxidant performance: A validated fluorescence assay for the measurement of plasma oxidizability. Anal. Biochem. 2006, 354, 290–298. [Google Scholar] [CrossRef] [PubMed]
- Pan, M.-H.; Tung, Y.-C.; Yang, G.; Li, S.; Ho, C.-T. Molecular mechanisms of the anti-obesity effect of bioactive compounds in tea and coffee. Food Funct. 2016, 7, 4481–4491. [Google Scholar] [CrossRef] [PubMed]
- Belke, D.D.; Severson, D.L. Diabetes in Mice with Monogenic Obesity: The db/db Mouse and Its Use in the Study of Cardiac Consequences. In Animal Models in Diabetes Research; Humana Press: Totowa, NJ, USA, 2012; pp. 47–57. [Google Scholar]
- Després, J.P.; Nadeau, A.; Tremblay, A.; Ferland, M.; Moorjani, S.; Lupien, P.J.; Thériault, G.; Pinault, S.; Bouchard, C. Role of deep abdominal fat in the association between regional adipose tissue distribution and glucose tolerance in obese women. Diabetes 1989, 38, 304–309. [Google Scholar] [CrossRef] [PubMed]
- Matsuzawa, Y. Establishment of a concept of visceral fat syndrome and discovery of adiponectin. Proc. Jpn. Acad. Ser. B 2010, 86, 131–141. [Google Scholar] [CrossRef]
- Su, M.L.; He, Y.; Li, Q.S.; Zhu, B.H. Efficacy of acetylshikonin in preventing obesity and hepatic steatosis in db/db mice. Molecules 2016, 21, 976. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Fan, S.; Hu, N.; Gu, M.; Chu, C.; Li, Y.; Lu, X.; Huang, C. Rhein reduces fat weight in db/db mouse and prevents diet-induced obesity in C57Bl/6 mouse through the inhibition of PPAR γ signaling. PPAR Res. 2012, 2012, 374936. [Google Scholar] [CrossRef] [PubMed]
- Leal-Díaz, A.M.; Noriega, L.G.; Torre-Villalvazo, I.; Torres, N.; Alemán-Escondrillas, G.; López-Romero, P.; Sánchez-Tapia, M.; Aguilar-López, M.; Furuzawa-Carballeda, J.; Velázquez-Villegas, L.A.; et al. Aguamiel concentrate from Agave salmiana and its extracted saponins attenuated obesity and hepatic steatosis and increased Akkermansia muciniphila in C57BL6 mice. Sci. Rep. 2016, 6, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.G.; Parks, J.S.; Kang, H.W. Quercetin, a functional compound of onion peel, remodels white adipocytes to brown-like adipocytes. J. Nutr. Biochem. 2017, 42, 62–71. [Google Scholar] [CrossRef] [PubMed]
- Marques, S.D.; Libonati, R.M.; Sabaa-Srur, A.U.; Luo, R.; Shejwalkar, P.; Hara, K.; Dobbs, T.; Smith, R.E. Evaluation of the effects of passion fruit peel flour (Passiflora edulis fo.flavicarpa) on metabolic changes in HIV patients with lipodystrophy syndrome secondary to antiretroviral therapy. Braz. J. Pharmacogn. 2016, 26, 420–426. [Google Scholar] [CrossRef]
- Zeraik, M.L.; Pereira, C.A.M.; Zuin, V.G.; Yariwake, J.H. Maracujá: Um alimento functional? Braz. J. Pharmacogn. 2010, 20, 459–471. [Google Scholar] [CrossRef]
- Samout, N.; Bouzenna, H.; Dhibi, S.; Ncib, S.; Elfeki, A. ScienceDirect Therapeutic effect of apple pectin in obese rats. Biomed. Pharmacother. 2016, 83, 1233–1238. [Google Scholar] [CrossRef] [PubMed]
- Brouns, F.; Theuwissen, E.; Adam, A.; Bell, M.; Berger, A.; Mensink, R.P. Cholesterol-lowering properties of different pectin types in mildly hyper-cholesterolemic men and women. Eur. J. Clin. Nutr. 2012, 66, 591–599. [Google Scholar] [CrossRef] [PubMed]
- Solà, R.; Godàs, G.; Richard, J.L.; Heras, M.; Olivé, S.; Anguera, A.; Salas, J.; Masana, L. Effects of soluble fiber (Ispaghula Husk) on plasma lipids, lipoproteins and apolipoproteins in men with ischemic heart disease. Atheroscler. Suppl. 2001, 2, 88–89. [Google Scholar] [CrossRef]
- Francisqueti, F.V.; Chiaverini, L.C.T.; Santos, K.C.; Minatel, I.O.; Berchieri Ronchi, C.; Ferron, A.J.T.; Ferreira, A.L.; Corrêa, C.R. The role of oxidative stress on the pathophysiology of metabolic syndrome. Rev. Assoc. Med. Bras. 2017, 63, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Young, A.; Lowe, G. Carotenoids—Antioxidant Properties. Antioxidants 2018, 7, 28. [Google Scholar] [CrossRef] [PubMed]
- Fiedor, J.; Burda, K. Potential role of carotenoids as antioxidants in human health and disease. Nutrients 2014, 6, 466–488. [Google Scholar] [CrossRef] [PubMed]
- Ho, C.-T. Phenolic compounds in food. In Phenolic Compounds in Food and Their Effects on Health; ACS Publications: Washington, DC, USA, 1992; pp. 1–45. [Google Scholar]
- Rice-Evans, C.; Miller, N.; Paganga, G. Antioxidant properties of phenolic compounds. Trends Plant Sci. 1997, 2, 152–159. [Google Scholar] [CrossRef]
- Hussain, T.; Tan, B.; Yin, Y.; Blachier, F.; Tossou, M.C.B.; Rahu, N. Oxidative Stress and Inflammation: What Polyphenols Can Do for Us? Oxid. Med. Cell. Longev. 2016, 2016, 7432797. [Google Scholar] [CrossRef] [PubMed]
| Parameters | Content/Activity |
|---|---|
| β-carotene | 0.321 mg/100 mg DW |
| Lutein | 57.53 mg/100 mg DW |
| Cryptoxanthin | 2.23 mg/100 mg DW |
| Total phenolic | 248.9 mg gallic acid equivalents/100 g DW |
| DHPP | 1.1 µM equiv Trolox/100 g DW |
| Variables | Obese | Obese + BPe |
|---|---|---|
| Feed intake (g/day) | 6.50 ± 0.15 | 10.8 ± 0.16 * |
| IBW (g) | 17.6 ± 4.4 | 18.9 ± 4.4 |
| FBW (g) | 53.9 (0.6) | 52.7 (18.5) |
| RAT (g) | 1.87 (0.36) | 1.43 (1.24) |
| EAT (g) | 2.80 ± 0.33 | 2.85 ± 0.80 |
| VAT (g) | 1.80 ± 0.14 | 1.18 ± 0.51 * |
| SAT (g) | 9.11 ± 1.19 | 6.58 ± 2.95 * |
| TF (g) | 15.8 (2.0) | 14.2 (7.9) |
| AI (%) | 29.3 ± 2.5 | 24.66 ± 6.08 * |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Panelli, M.F.; Pierine, D.T.; De Souza, S.L.B.; Ferron, A.J.T.; Garcia, J.L.; Santos, K.C.d.; Belin, M.A.F.; Lima, G.P.P.; Borguini, M.G.; Minatel, I.O.; et al. Bark of Passiflora edulis Treatment Stimulates Antioxidant Capacity, and Reduces Dyslipidemia and Body Fat in db/db Mice. Antioxidants 2018, 7, 120. https://doi.org/10.3390/antiox7090120
Panelli MF, Pierine DT, De Souza SLB, Ferron AJT, Garcia JL, Santos KCd, Belin MAF, Lima GPP, Borguini MG, Minatel IO, et al. Bark of Passiflora edulis Treatment Stimulates Antioxidant Capacity, and Reduces Dyslipidemia and Body Fat in db/db Mice. Antioxidants. 2018; 7(9):120. https://doi.org/10.3390/antiox7090120
Chicago/Turabian StylePanelli, Marielle Fernanda, Damiana Tortolero Pierine, Sérgio Luiz Borges De Souza, Artur Júnio Togneri Ferron, Jéssica Leite Garcia, Klinsmann Carolo dos Santos, Matheus Antônio Filiol Belin, Giuseppina Pace Pereira Lima, Milena Galhardo Borguini, Igor Otávio Minatel, and et al. 2018. "Bark of Passiflora edulis Treatment Stimulates Antioxidant Capacity, and Reduces Dyslipidemia and Body Fat in db/db Mice" Antioxidants 7, no. 9: 120. https://doi.org/10.3390/antiox7090120
APA StylePanelli, M. F., Pierine, D. T., De Souza, S. L. B., Ferron, A. J. T., Garcia, J. L., Santos, K. C. d., Belin, M. A. F., Lima, G. P. P., Borguini, M. G., Minatel, I. O., Cicogna, A. C., Francisqueti, F. V., & Corrêa, C. R. (2018). Bark of Passiflora edulis Treatment Stimulates Antioxidant Capacity, and Reduces Dyslipidemia and Body Fat in db/db Mice. Antioxidants, 7(9), 120. https://doi.org/10.3390/antiox7090120





