A Review of the Effects of Leucine Metabolite (β-Hydroxy-β-methylbutyrate) Supplementation and Resistance Training on Inflammatory Markers: A New Approach to Oxidative Stress and Cardiovascular Risk Factors
Abstract
1. Introduction
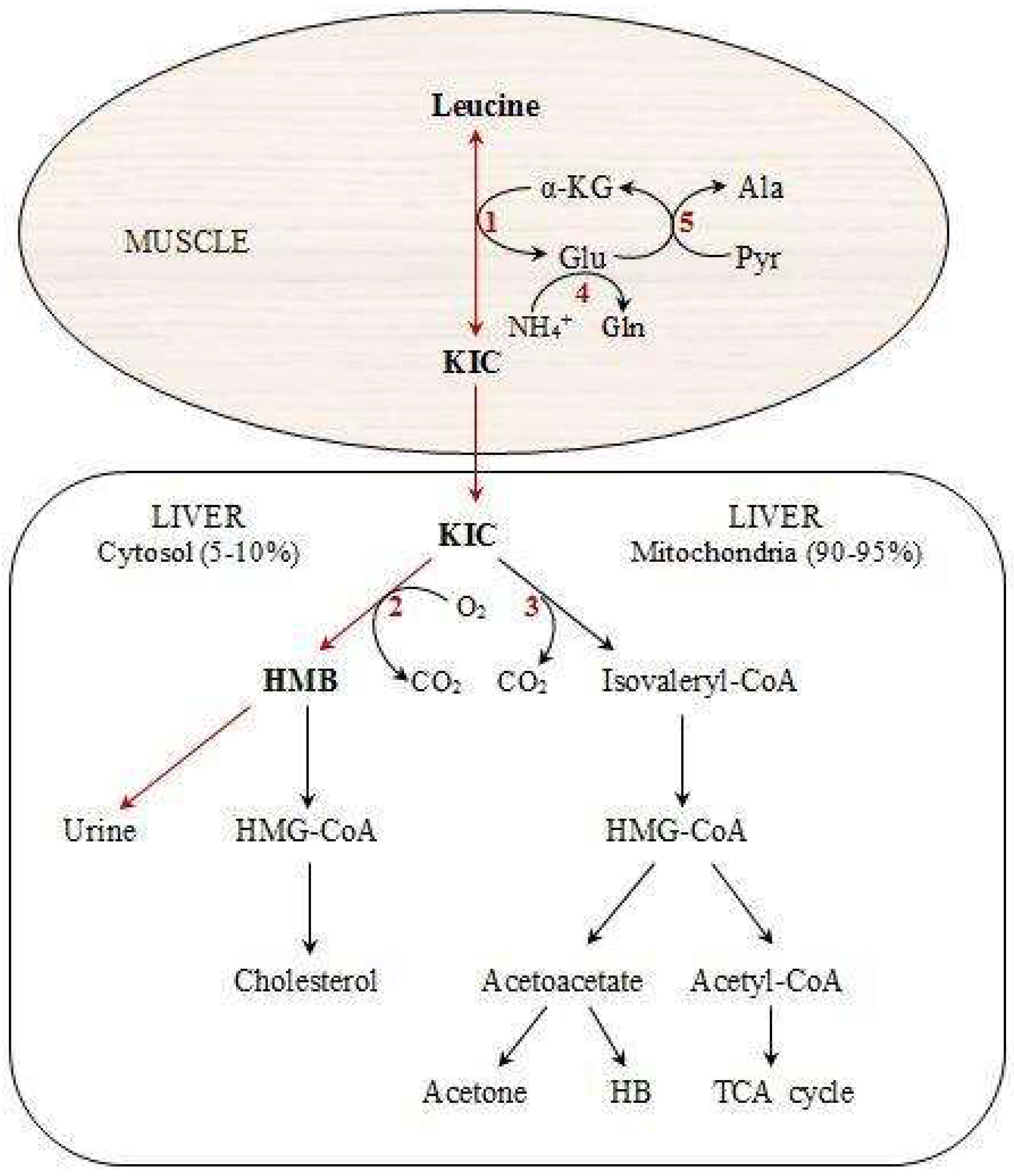
2. A Brief Overview of HMB Metabolism
3. An Overview of Different HMB Effects and Its Potential Mechanisms
4. Effects of HMB on Inflammation
4.1. Effects on Inflammation without Exercise
4.2. Effects on Inflammation Following Exercise
5. An Approach to Oxidative Stress
6. An Approach to Cardiovascular Risk Factors
6.1. Cardiovascular Effects of HMB without Exercise
6.2. Cardiovascular Effects of HMB Following Exercise
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Maughan, R.J.; Depiesse, F.; Geyer, H. The use of dietary supplements by athletes. J. Sports Sci. 2007, 25, S103–S113. [Google Scholar] [CrossRef] [PubMed]
- Thomson, J.S.; Watson, P.E.; Rowlands, D.S. Effects of nine weeks of β-Hydroxy-β-Methylbutyrate supplementation on strength and body composition in resistance trained men. J. Strength Cond. Res. 2009, 23, 827–835. [Google Scholar] [CrossRef] [PubMed]
- Kimura, K.; Cheng, X.W.; Inoue, A.; Hu, L.; Koike, T.; Kuzuya, M. β-hydroxy-β-methylbutyrate facilitates pi3k/akt-dependent mammalian target of rapamycin and foxo1/3a phosphorylations and alleviates tumor necrosis factor α/interferon γ–induced murf-1 expression in c2c12 cells. Nutr. Res. 2014, 34, 368–374. [Google Scholar] [CrossRef] [PubMed]
- Rahimi, M.H.; Mohammadi, H.; Eshaghi, H.; Askari, G.; Miraghajani, M. The effects of beta-hydroxy-beta-methylbutyrate supplementation on recovery following exercise-induced muscle damage: A systematic review and meta-analysis. J. Am. Coll. Nutr. 2018, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Martinez, J.; Santos-Lozano, A.; Garcia-Hermoso, A.; Sadarangani, K.; Cristi-Montero, C. Effects of beta-hydroxy-beta-methylbutyrate supplementation on strength and body composition in trained and competitive athletes: A meta-analysis of randomized controlled trials. J. Sci. Med. Sport 2017, 21, 727–735. [Google Scholar] [CrossRef] [PubMed]
- Palisin, T.; Stacy, J.J. β-Hydroxy-β-methylbutyrate and its use in athletics. Curr. Sports. Med. Rep. 2005, 4, 220–223. [Google Scholar] [CrossRef] [PubMed]
- Lowery, R.P.; Joy, J.M.; Rathmacher, J.A.; Baier, S.M.; Fuller, J.C.; Shelley, M.C.; Jäger, R.; Purpura, M.; Wilson, S.; Wilson, J.M. Interaction of beta-hydroxy-beta-methylbutyrate free acid and adenosine triphosphate on muscle mass, strength, and power in resistance trained individuals. J. Strength Cond. Res. 2016, 30, 1843–1854. [Google Scholar] [CrossRef] [PubMed]
- Stout, J.R.; Fukuda, D.H.; Kendall, K.L.; Smith-Ryan, A.E.; Moon, J.R.; Hoffman, J.R. β-Hydroxy-β-methylbutyrate (HMB) supplementation and resistance exercise significantly reduce abdominal adiposity in healthy elderly men. Exp. Gerontol. 2015, 64, 33–34. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, H.; Gill, P.; Fernandes Filho, J.; Fernandes, L.C. Effects of 12-weeks of supplementation with β-hydroxy-β-methylbutyrate-Ca (HMB-Ca) on athletic performance. J. Exerc. Physiol. Online 2015, 18, 84–94. [Google Scholar]
- Wilson, J.M.; Lowery, R.P.; Joy, J.M.; Andersen, J.; Wilson, S.M.; Stout, J.R.; Duncan, N.; Fuller, J.C.; Baier, S.M.; Naimo, M.A. The effects of 12 weeks of beta-hydroxy-beta-methylbutyrate free acid supplementation on muscle mass, strength, and power in resistance-trained individuals: A randomized, double-blind, placebo-controlled study. Eur. J. Appl. Physiol. 2014, 114, 1217–1227. [Google Scholar] [CrossRef] [PubMed]
- Dunsmore, K.; Lowery, R.P.; Duncan, N.; Davis, G.; Rathmacher, J.; Baier, S.; Sikorski, E.; Morrison, T.; Naimo, M.; Walters, J. Effects of 12 weeks of beta-hydroxy-beta-methylbutyrate free acid Gel supplementation on muscle mass, strength, and power in resistance trained individuals (Abstract). J. Int. Soc. Sports Nutr. 2012, 9. [Google Scholar] [CrossRef]
- Panton, L.B.; Rathmacher, J.A.; Baier, S.; Nissen, S. Nutritional supplementation of the leucine metabolite β-hydroxy-β-methylbutyrate (hmb) during resistance training. Nutrition 2000, 16, 734–739. [Google Scholar] [CrossRef]
- Asadi, A.; Arazi, H.; Suzuki, K. Effects of β-hydroxy-β-methylbutyrate-free acid supplementation on strength, power and hormonal adaptations following resistance training. Nutrients 2017, 9, 1316. [Google Scholar] [CrossRef] [PubMed]
- Durkalec-Michalski, K.; Jeszka, J.; Podgórski, T. The effect of a 12-week beta-hydroxy-beta-methylbutyrate (HMB) supplementation on highly-trained combat sports athletes: A randomised, double-blind, placebo-controlled crossover study. Nutrients 2017, 9, 753. [Google Scholar] [CrossRef] [PubMed]
- Durkalec-Michalski, K.; Jeszka, J. The effect of β-hydroxy-β-methylbutyrate on aerobic capacity and body composition in trained athletes. J. Strength Cond. Res. 2016, 30, 2617–2626. [Google Scholar] [CrossRef] [PubMed]
- Durkalec-Michalski, K.; Jeszka, J. The efficacy of a β-hydroxy-β-methylbutyrate supplementation on physical capacity, body composition and biochemical markers in elite rowers: A randomised, double-blind, placebo-controlled crossover study. J. Int. Soc. Sports Nutr. 2015, 12, 31. [Google Scholar] [CrossRef] [PubMed]
- Portal, S.; Zadik, Z.; Rabinowitz, J.; Pilz-Burstein, R.; Adler-Portal, D.; Meckel, Y.; Cooper, D.M.; Eliakim, A.; Nemet, D. The effect of HMB supplementation on body composition, fitness, hormonal and inflammatory mediators in elite adolescent volleyball players: A prospective randomized, double-blind, placebo-controlled study. Eur. J. Appl. Physiol. 2011, 111, 2261–2269. [Google Scholar] [CrossRef] [PubMed]
- Correia, A.L.M.; de Lima, F.D.; Bottaro, M.; Vieira, A.; da Fonseca, A.C.; Lima, R.M. Pre-exercise β-hydroxy-β-methylbutyrate free-acid supplementation improves work capacity recovery: A randomized, double-blinded, placebo-controlled study. Appl. Physiol. Nutr. Metab. 2018, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, A.M.; Stout, J.R.; Jajtner, A.R.; Townsend, J.R.; Wells, A.J.; Beyer, K.S.; Boone, C.H.; Pruna, G.J.; Mangine, G.T.; Scanlon, T.M. Effects of β-hydroxy-β-methylbutyrate free acid and cold water immersion on post-exercise markers of muscle damage. Amino Acids 2014, 46, 1501–1511. [Google Scholar] [CrossRef] [PubMed]
- Wilson, J.M.; Lowery, R.P.; Joy, J.M.; Walters, J.A.; Baier, S.M.; Fuller, J.C.; Stout, J.R.; Norton, L.E.; Sikorski, E.M.; Wilson, S.M. β-Hydroxy-β-methylbutyrate free acid reduces markers of exercise-induced muscle damage and improves recovery in resistance-trained men. Br. J. Nutr. 2013, 110, 538–544. [Google Scholar] [CrossRef] [PubMed]
- Knitter, A.; Panton, L.; Rathmacher, J.; Petersen, A.; Sharp, R. Effects of β-hydroxy-β-methylbutyrate on muscle damage after a prolonged run. J. Appl. Physiol. 2000, 89, 1340–1344. [Google Scholar] [CrossRef] [PubMed]
- Engelen, M.P.; Deutz, N.E. Is β-hydroxy β-methylbutyrate an effective anabolic agent to improve outcome in older diseased populations? Curr. Opin. Clin. Nutr. Metab. Care 2018, 21, 207–213. [Google Scholar] [CrossRef] [PubMed]
- Silva, V.R.; Belozo, F.L.; Micheletti, T.O.; Conrado, M.; Stout, J.R.; Pimentel, G.D.; Gonzalez, A.M. β-Hydroxy-β-methylbutyrate free acid supplementation may improve recovery and muscle adaptations after resistance training: A systematic review. Nutr. Res. 2017, 45, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Albert, F.J.; Morente-Sánchez, J.; Ortega, F.B.; Castillo, M.J.; Gutiérrez, Á. Usefulness of β-hydroxy-β-methylbutyrate (HMB) supplementation in different sports: An update and practical implications. Nutr. Hosp. 2015, 32, 20–33. [Google Scholar] [PubMed]
- Wilson, J.M.; Fitschen, P.J.; Campbell, B.; Wilson, G.J.; Zanchi, N.; Taylor, L.; Wilborn, C.; Kalman, D.S.; Stout, J.R.; Hoffman, J.R. International Society of Sports Nutrition Position Stand: Beta-hydroxy-beta-methylbutyrate (HMB). J. Int. Soc. Sports Nutr. 2013, 10, 6. [Google Scholar] [CrossRef] [PubMed]
- Zanchi, N.E.; Gerlinger-Romero, F.; Guimaraes-Ferreira, L.; de Siqueira Filho, M.A.; Felitti, V.; Lira, F.S.; Seelaender, M.; Lancha, A.H. HMB supplementation: Clinical and athletic performance-related effects and mechanisms of action. Amino Acids 2011, 40, 1015–1025. [Google Scholar] [CrossRef] [PubMed]
- Garlick, P.J. The role of leucine in the regulation of protein metabolism. J. Nutr. 2005, 135, 1553S–1556S. [Google Scholar] [CrossRef] [PubMed]
- Pinheiro, C.H.J.; Guimaraes-Ferreira, L.; Gerlinger-Romero, F.; Curi, R. An overview on beta-hydroxy-beta-methylbutyrate (HMB) supplementation in skeletal muscle function and sports performance. In Nutrition and Enhanced Sports Performance; Elsevier: Amsterdam, The Netherlands, 2014; pp. 455–463. [Google Scholar]
- Sabourin, P.J.; Bieber, L.L. Formation of beta-hydroxyisovalerate by an alpha-ketoisocaproate oxygenase in human liver. Metabolism 1983, 32, 160–164. [Google Scholar] [CrossRef]
- Van Koevering, M.; Nissen, S. Oxidation of leucine and alpha-ketoisocaproate to beta-hydroxy-beta-methylbutyrate in vivo. Am. J. Physiol. Endocrinol. Metab. 1992, 262, E27–E31. [Google Scholar] [CrossRef] [PubMed]
- Nissen, S.L.; Abumrad, N.N. Nutritional role of the leucine metabolite β-hydroxy β-methylbutyrate (HMB). J. Nutr. Biochem. 1997, 8, 300–311. [Google Scholar] [CrossRef]
- Fuller, J.C.; Sharp, R.L.; Angus, H.F.; Baier, S.M.; Rathmacher, J.A. Free acid gel form of β-hydroxy-β-methylbutyrate (HMB) improves HMB clearance from plasma in human subjects compared with the calcium HMB salt. Br. J. Nutr. 2011, 105, 367–372. [Google Scholar] [CrossRef] [PubMed]
- Vukovich, M.D.; Slater, G.; Macchi, M.B.; Turner, M.J.; Fallon, K.; Boston, T.; Rathmacher, J. β-hydroxy-β-methylbutyrate (HMB) kinetics and the influence of glucose ingestion in humans. J. Nutr. Biochem. 2001, 12, 631–639. [Google Scholar] [CrossRef]
- Fuller, J.C.; Sharp, R.L.; Angus, H.F.; Khoo, P.Y.; Rathmacher, J.A. Comparison of availability and plasma clearance rates of β-hydroxy-β-methylbutyrate delivery in the free acid and calcium salt forms. Br. J. Nutr. 2015, 114, 1403–1409. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, D.; Hossain, T.; Hill, D.; Phillips, B.; Crossland, H.; Williams, J.; Loughna, P.; Churchward-Venne, T.; Breen, L.; Phillips, S. Effects of leucine and its metabolite β-hydroxy-β-methylbutyrate on human skeletal muscle protein metabolism. J. Physiol. 2013, 591, 2911–2923. [Google Scholar] [CrossRef] [PubMed]
- Shreeram, S.; Johns, P.W.; Subramaniam, S.; Ramesh, S.; Vaidyanathan, V.; Puthan, J.K.; Mandal, S.; Mamidi, V.K.; Gelling, R.W. The relative bioavailability of the calcium salt of β-hydroxy-β-methylbutyrate is greater than that of the free fatty acid form in rats–3. J. Nutr. 2014, 144, 1549–1555. [Google Scholar] [CrossRef] [PubMed]
- Kornasio, R.; Riederer, I.; Butler-Browne, G.; Mouly, V.; Uni, Z.; Halevy, O. β-hydroxy-β-methylbutyrate (HMB) stimulates myogenic cell proliferation, differentiation and survival via the MAPK/ERK and PI3K/Akt pathways. Biochim. Biophys. Acta Mol. Cell Res. 2009, 1793, 755–763. [Google Scholar] [CrossRef] [PubMed]
- Gerlinger-Romero, F.; Guimarães-Ferreira, L.; Giannocco, G.; Nunes, M. Chronic supplementation of beta-hydroxy-beta methylbutyrate (HMβ) increases the activity of the GH/IGF-I axis and induces hyperinsulinemia in rats. Growth Horm. IGF Res. 2011, 21, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Smith, H.J.; Mukerji, P.; Tisdale, M.J. Attenuation of proteasome-induced proteolysis in skeletal muscle by β-hydroxy-β-methylbutyrate in cancer-induced muscle loss. Cancer Res. 2005, 65, 277–283. [Google Scholar] [PubMed]
- Kovarik, M.; Muthny, T.; Sispera, L.; Holecek, M. Effects of β-hydroxy-β-methylbutyrate treatment in different types of skeletal muscle of intact and septic rats. J. Physiol. Biochem. 2010, 66, 311–319. [Google Scholar] [CrossRef] [PubMed]
- Eley, H.L.; Russell, S.T.; Tisdale, M.J. Attenuation of depression of muscle protein synthesis induced by lipopolysaccharide, tumor necrosis factor, and angiotensin II by β-hydroxy-β-methylbutyrate. Am. J. Physiol. Endocrinol. Metab. 2008, 295, E1409–E1416. [Google Scholar] [CrossRef] [PubMed]
- Hao, Y.; Jackson, J.R.; Wang, Y.; Edens, N.; Pereira, S.L.; Alway, S.E. β-Hydroxy-β-methylbutyrate reduces myonuclear apoptosis during recovery from hind limb suspension-induced muscle fiber atrophy in aged rats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2011, 301, R701–R715. [Google Scholar] [CrossRef] [PubMed]
- Holeček, M. Beta-hydroxy-beta-methylbutyrate supplementation and skeletal muscle in healthy and muscle-wasting conditions. J. Cachexia Sarcopenia Muscle 2017, 8, 529–541. [Google Scholar] [CrossRef] [PubMed]
- Nissen, S.; Sharp, R.; Ray, M.; Rathmacher, J.; Rice, D.; Fuller, J., Jr.; Connelly, A.; Abumrad, N. Effect of leucine metabolite β-hydroxy-β-methylbutyrate on muscle metabolism during resistance-exercise training. J. Appl. Physiol. 1996, 81, 2095–2104. [Google Scholar] [CrossRef] [PubMed]
- Vallejo, J.; Spence, M.; Cheng, A.-L.; Brotto, L.; Edens, N.K.; Garvey, S.M.; Brotto, M. Cellular and physiological effects of dietary supplementation with β-hydroxy-β-methylbutyrate (HMB) and β-alanine in late middle-aged mice. PLoS ONE 2016, 11, e0150066. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Duan, Y.; Yao, K.; Li, F.; Hou, Y.; Wu, G.; Yin, Y. β-Hydroxy-β-methylbutyrate, mitochondrial biogenesis, and skeletal muscle health. Amino Acids 2016, 48, 653–664. [Google Scholar] [CrossRef] [PubMed]
- Da Justa Pinheiro, C.H.; Gerlinger-Romero, F.; Guimaraes-Ferreira, L.; de Souza, A.L., Jr.; Vitzel, K.F.; Nachbar, R.T.; Nunes, M.T.; Curi, R. Metabolic and functional effects of beta-hydroxy-beta-methylbutyrate (HMB) supplementation in skeletal muscle. Eur. J. Appl. Physiol. 2012, 112, 2531–2537. [Google Scholar] [CrossRef] [PubMed]
- Peterson, A.; Qureshi, M.; Ferket, P.; Fuller, J., Jr. In vitro exposure with β-hydroxy-β-methylbutyrate enhances chicken macrophage growth and function. Vet. Immunol. Immunopathol. 1999, 67, 67–78. [Google Scholar] [CrossRef]
- Clark, R.H.; Feleke, G.; Din, M.; Yasmin, T.; Singh, G.; Khan, F.A.; Rathmacher, J.A. Nutritional treatment for acquired immunodeficiency virus-associated wasting using β-hydroxy β-methylbutyrate, glutamine, and arginine: A randomized, double-blind, placebo-controlled study. J. Parenter. Enter. Nutr. 2000, 24, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, L.; Chien, S.; Huang, M.; Tseng, H.; Chang, C. Anti-inflammatory and anticatabolic effects of short-term beta-hydroxy-beta-methylbutyrate supplementation on chronic obstructive pulmonary disease patients in intensive care unit. Asia Pac. J. Clin. Nutr. 2006, 15, 544–550. [Google Scholar] [PubMed]
- Suzuki, K. Cytokine response to exercise and its modulation. Antioxidants 2018, 7, 17. [Google Scholar] [CrossRef]
- Ma, S.; Suzuki, K. Toll-like receptor 4: Target of lipotoxicity and exercise-Induced anti-inflammatory effect? Ann. Nutr. Food Sci. 2018, 2, 1–3. [Google Scholar]
- Clowes, G.H., Jr.; George, B.C.; Villee, C.A., Jr.; Saravis, C.A. Muscle proteolysis induced by a circulating peptide in patients with sepsis or trauma. N. Engl. J. Med. 1983, 308, 545–552. [Google Scholar] [CrossRef] [PubMed]
- Townsend, J.R.; Fragala, M.S.; Jajtner, A.R.; Gonzalez, A.M.; Wells, A.J.; Mangine, G.T.; Robinson, E.H., IV; McCormack, W.P.; Beyer, K.S.; Pruna, G.J. β-Hydroxy-β-methylbutyrate (HMB)-free acid attenuates circulating TNF-α and TNFR1 expression postresistance exercise. J. Appl. Physiol. 2013, 115, 1173–1182. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, A.M.; Fragala, M.S.; Jajtner, A.R.; Townsend, J.R.; Wells, A.J.; Beyer, K.S.; Boone, C.H.; Pruna, G.J.; Mangine, G.T.; Bohner, J.D. Effects of β-hydroxy-β-methylbutyrate free acid and cold water immersion on expression of CR3 and MIP-1β following resistance exercise. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2014, 306, R483–R489. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, J.R.; Gepner, Y.; Stout, J.R.; Hoffman, M.W.; Ben-Dov, D.; Funk, S.; Daimont, I.; Jajtner, A.R.; Townsend, J.R.; Church, D.D. β-Hydroxy-β-methylbutyrate attenuates cytokine response during sustained military training. Nutr. Res. 2016, 36, 553–563. [Google Scholar] [CrossRef] [PubMed]
- Vulcan, P.R. Role of β-Hydroxy-β-methylbutyrate (HMB) on Inflammation after Eccentric Exercise. Master’s Thesis, Iowa State University, Ames, IA, USA, October 2012. [Google Scholar]
- Arazi, H.; Asadi, A.; Suzuki, K. The effects of beta-hydroxy-beta-methylbutyrate-free acid supplementation and resistance training on oxidative stress markers: A randomized, double-blind, placebo-controlled study. Antioxidants 2018, 7, 76. [Google Scholar] [CrossRef] [PubMed]
- Rudney, H. The biosynthesis of β-hydroxy-β-methylglutaric acid. J. Biol. Chem. 1957, 227, 363–377. [Google Scholar] [PubMed]
- Nissen, S.; Sharp, R.; Panton, L.; Vukovich, M.; Trappe, S.; Fuller, J., Jr. β-hydroxy-β-methylbutyrate (HMB) supplementation in humans is safe and may decrease cardiovascular risk factors. J. Nutr. 2000, 130, 1937–1945. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, L.-C.; Chow, C.-J.; Chang, W.-C.; Liu, T.-H.; Chang, C.-K. Effect of β-hydroxy-β-methylbutyrate on protein metabolism in bed-ridden elderly receiving tube feeding. Asia. Pac. J. Clin. Nutr. 2010, 19, 200–208. [Google Scholar] [PubMed]
- Holecek, M.; Muthny, T.; Kovarik, M.; Sispera, L. Effect of beta-hydroxy-beta-methylbutyrate (HMB) on protein metabolism in whole body and in selected tissues. Food Chem. Toxicol. 2009, 47, 255–259. [Google Scholar] [CrossRef] [PubMed]
- Sharawy, M.H.; El-Awady, M.S.; Megahed, N.; Gameil, N.M. The ergogenic supplement β-hydroxy-β-methylbutyrate (HMB) attenuates insulin resistance through suppressing GLUT-2 in rat liver. Can. J. Physiol. Pharmacol. 2015, 94, 488–497. [Google Scholar] [CrossRef] [PubMed]
- Ravanbakhsh, N.; Torabi, N.; Foadoddini, M. Preventive effects of beta-hydroxy-beta-methyl butyrate on cardiac arrhythmias in rats (Abstract). Ofogh-E-Danesh 2016, 22, 209–214. [Google Scholar]
- Arazi, H.; Rohani, H.; Ghiasi, A.; Davaran, M. The effect of HMB supplementation on cardiovascular risk factors after four weeks of resistance training in amateur athletes. Int. Cardiovasc. Res. J. 2015, 9, 89–93. [Google Scholar]
| Effect | Mechanisms of action |
|---|---|
| Increasing protein synthesis |
|
| Decreasing proteolysis (protein breakdown) |
|
| Enhancing tissue repair | |
| Improving excitation-contraction coupling in muscle cells |
|
| Improving aerobic capacity | |
| Delaying acute muscle fatigue | |
| Increasing ATP and glycogen content in skeletal muscle |
|
| Author (Year) | Subjects | Exercise Protocol | Supplementation | Primary Findings | Conclusion |
|---|---|---|---|---|---|
| Townsend (2013) [54] | 40 resistance-trained men | 4 sets of the squat (80% of 1RM), and dead lift and barbell split-squat (70% of 1RM); as many repetitions as possible (up to 10 repetitions for each set); 90 s rest intervals between sets and exercises | 3 g (1-g serving of HMB-FA, 30 min before, and 2 h and 6 h following the exercise session) | ↑ TNF-α from pre to immediately post-exercise in only PL group; ↑ TNFR1 expression from pre to 30 min post-exercise in only PL group | Acute HMB-FA supplementation may attenuate the initial immune responses to an intense RE, which may reduce subsequent recovery period |
| Vulcan (2012) [57] | Untrained subjects (16 men, 16 women) | 3 sets of 50 eccentric leg extensions from 0° to 90° at a rate of 60°/s on both legs; 2 min rest intervals between sets | Pre-exercise, or pre-exercise and for 4 days (3 servings/day) post-exercise; either HMB-Ca or HMB-FA | ↑ IL-1ra at 48 h, 72 h, and 96 h post-exercise compared to PL; ↑ TNF-α at 48 h, and 72 h post-exercise compared to PL | The role of acute HMB supplementation on reducing the inflammatory response after RE has not been confirmed |
| Gonzalez (2014) [55] | 39 resistance-trained men | 4 sets of the squat, dead lift and barbell split-squat (70–80% of 1RM); as many repetitions as possible (not to exceed 10 repetitions in any set); 90 s rest intervals between sets and exercises | 3 g (1-g serving of HMB-FA, 30 min before, and 2 h and 6 h following the exercise session) | ↔ MIP-1β responses; ↓ peak expression of CR3 at 30 min post-exercise; ↑ percentage of monocytes expressing CR3 for up to 48 h post-exercise | HMB supplementation may alter immune cell mobilization and adhesion mechanisms during tissue recovery after RE |
| Hoffman (2016) [56] | 11 elite combat male soldiers | A combination of different military training including combat skill development, extreme trainings, navigational training with carrying approximately 35 kg of equipment | 3 servings (1 g/serving) of HMB-FA per day at meal time for 23 days | ↓ TNF-α responses; ↓ G-CSF; ↓ IL-10; ↓ INFγ; ↓ IL-8; ↓ CX3CL1 from pre- to post-training | HMB supplementation may attenuate the inflammatory mediators to severe military trainings, and maintain muscle quality |
| Portal (2011) [17] | Adolescent elite volleyball players (14 males, 14 females) | Volleyball training (the early phase of the volleyball season) | 3 g/day HMB supplementation for 7 weeks; the type of HMB has not been mentioned | ↔ IL-10 ↔ IL-1ra from pre- to post-training | HMB supplementation has no significant effect on the inflammatory mediator changes during the initial phases of volleyball training season |
| Arazi (2018) [58] | 16 healthy young males | 3 sets of 8–12 repetitions with 75–85% of 1RM (leg press, knee extension, knee flexion, lat pull-down, bench press, shoulder press, cable biceps curl and triceps push down); 2 sessions/week for 6 weeks; 2 and 3 min rest intervals between sets and exercises, respectively | 3 g/day HMB-FA; one Serving (1 g) with each of 3 separate meals | ↓ MDA and PC from pre- to post-training in both HMB and PL groups; ↔ 8-OHdG in the groups | HMB supplementation has no further improvements related to oxidative stress markers in young males |
| Arazi (2015) [65] | 20 male athletes (without regular resistance training) | 2 sets of 9 exercises (squat, knee extension, knee flexion, leg press, bench press, lat pull- down, shoulder press, cable biceps curl, and triceps push down); 10 repetitions with 80–85% of 1RM; 2 and 3 min rest intervals between sets and exercises, respectively | 3 g/day HMB-Ca; 3 servings (1 g) per day | ↓ TC, LDL, and TG from pre- to post-training in both HMB and PL groups; ↓ systolic BP and ↑ HDL in HMB group; ↔ diastolic BP, RBC, Hb, Hct, MCV, and MCH in the groups | HMB supplementation is safe and does not result in any adverse effects on cardiovascular parameters in male athletes |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arazi, H.; Taati, B.; Suzuki, K. A Review of the Effects of Leucine Metabolite (β-Hydroxy-β-methylbutyrate) Supplementation and Resistance Training on Inflammatory Markers: A New Approach to Oxidative Stress and Cardiovascular Risk Factors. Antioxidants 2018, 7, 148. https://doi.org/10.3390/antiox7100148
Arazi H, Taati B, Suzuki K. A Review of the Effects of Leucine Metabolite (β-Hydroxy-β-methylbutyrate) Supplementation and Resistance Training on Inflammatory Markers: A New Approach to Oxidative Stress and Cardiovascular Risk Factors. Antioxidants. 2018; 7(10):148. https://doi.org/10.3390/antiox7100148
Chicago/Turabian StyleArazi, Hamid, Behzad Taati, and Katsuhiko Suzuki. 2018. "A Review of the Effects of Leucine Metabolite (β-Hydroxy-β-methylbutyrate) Supplementation and Resistance Training on Inflammatory Markers: A New Approach to Oxidative Stress and Cardiovascular Risk Factors" Antioxidants 7, no. 10: 148. https://doi.org/10.3390/antiox7100148
APA StyleArazi, H., Taati, B., & Suzuki, K. (2018). A Review of the Effects of Leucine Metabolite (β-Hydroxy-β-methylbutyrate) Supplementation and Resistance Training on Inflammatory Markers: A New Approach to Oxidative Stress and Cardiovascular Risk Factors. Antioxidants, 7(10), 148. https://doi.org/10.3390/antiox7100148







